Abstract
A boy exhibiting conjugated hyperbilirubinemia from birth, with elevated serum gamma-glutamyl transpeptidase activity (GGT), developed liver failure unusually early (7mo); GGT concomitantly normalized. ABCB4 disease was suspected, but no ABCB4 lesion was found. The boy was instead homozygous for ABCB11 variant c.1213 T>C (p.(Cys405Arg)), which is predicted to affect protein function. Both ABCB4 and ABCB11 were normally expressed in the explanted liver, with intralobular cholestasis; however, large-duct sclerosing cholangiopathy and ductal-plate malformation also were present. The primary-cilium constituent doublecortin domain containing 2 (DCDC2) was not expressed. Co-existence of ABCB11 disease and DCDC2 disease was proposed. Further testing identified homozygosity for the canonical-receptor splice-site variant c.294-2A>G (p.?) in DCDC2. Our report emphasizes the need to integrate clinical, histological, and genetic data in patients with neonatal cholestasis.
Subject terms: Genetics research, Primary sclerosing cholangitis
Introduction
Progressive familial intrahepatic cholestasis (PFIC) comprises disorders of bile formation leading to fibrosis and cirrhosis; most patients present as infants. So far, six PFIC-associated genes, including ATP-binding cassette subfamily B member 11 (ABCB11), are known [1, 2]. ABCB11 disease is histopathologically characterized by lobular disarray with anisocytosis, giant-cell change, and rosetting of hepatocytes; bile pigment lies in hepatocyte cytoplasm and canalicular lumina, and bile salt export pump (BSEP, encoded by ABCB11) often cannot be detected, while gamma-glutamyl transpeptidase (GGT) is expressed at bile-canaliculus walls [3]. Serum activity of GGT is disproportionately low for the degree of conjugated hyperbilirubinemia [1, 4]. Liver transplantation (LTx) is the only curative therapy. However, unless hepatocellular carcinoma develops, LTx is rarely required in infants or toddlers with ABCB11-related PFIC [5–8].
Neonatal sclerosing cholangitis (NSC) is a rare disorder manifest principally as cholestasis. GGT activity is high; clinical features in infancy suggest extrahepatic biliary atresia. However, cholangiography identifies a patent although distorted biliary tract. NSC progresses to fibrosis and cirrhosis. Variants in doublecortin domain containing 2 (DCDC2) are found in some cases of NSC [9, 10]. DCDC2 encodes DCDC2, a constituent of primary cilia [11]. LTx, when required, occurs during adolescence [9, 10]. DCDC2-disease patients may have renal as well as hepatobiliary disease [11, 12] with deafness another prominent feature [11, 12]. Histopathologic features of DCDC2 disease in smaller portal tracts include hypoplasia of portal-venule radicles, with cholangiocyte injury (anisocytosis, disordered polarity), periductal fibrosis, and interlobular bile duct loss. In larger portal tracts (seen only in hepatectomy material), chronic inflammation and biliary-tract ectasia with excessive numbers of anastomosing biliary profiles accompany bridging fibrosis and cirrhosis. DCDC2 expression is lacking in cases studied to date. Ultrastructural study identifies deficiency of primary cilia at cholangiocyte apical membranes [10].
We report an infant with cholestasis and exceptionally early liver failure that we consider due to homozygous variants in both ABCB11 and DCDC2.
Methods
The parents gave written informed consent for molecular research investigations, as approved by the local ethics committee. Clinical exome sequencing was performed with patient leukocyte-derived DNA using the Trusight One Panel enrichment kit (Illumina, Cambridge, UK) on an Illumina instrument to generate 100-bp paired-end reads. These were aligned to the human reference genome and evaluated with SeqNext software (JSI Systems, Ettenheim, Germany) and the CNV Detective [13].
The presence and segregation of variants were tested by PCR amplification and Sanger sequencing of single exons. Designations are based on NCBI reference sequences for transcripts NM_003742.2 (ABCB11) and NM_016356.3 (DCDC2), and genomic sequence NG_012829.2 (DCDC2). Nucleotide numbering uses +1 as the A of the reference-sequence ATG translation initiation codon (codon 1). Variant information was submitted to the LOVD database (https://databases.lovd.nl/shared/individuals/00275554). Allele frequencies for variants were taken from gnomAD (https://gnomad.broadinstitute.org) and ExAC databases (http://exac.broadinstitute.org/). Significance of missense-change sequence variation was predicted using four web-based programs, Mutation Taster (http://www.mutationtaster.org), PolyPhen-2 (http://genetics.bwh.harvard.edu/pph2/), PROVEAN (http://provean.jcvi.org/index.php/), and CADD (complete annotation dependent depletion; https://cadd.gs.washington.edu/). BSEP modeling used the Mutagenesis Wizard module in MacPyMol (Schrödinger, Munich, Germany) and P-glycoprotein structure (PDB-Code 4M1M [14]).
Clinical description
A male infant, third-born of distantly consanguineous parents, was jaundiced from term birth. Details of the pregnancy are unavailable; the siblings are well. On hospitalization for scabies aged 7mo, he was icteric with mild hepatosplenomegaly (total bilirubin [TB] 9.43 mg/dl; direct bilirubin [DB] 7.78 mg/dl; GGT 251 U/L, expected 2–21; total serum bile acids 366 µmol/l, ≤10). AST and ALT were elevated (~460 and 187 U/L), as was the international normalized ratio (INR) at 2.25. Jaundice and elevated GGT activity reportedly had been present since birth. No signs of renal disease were apparent. Serum creatinine was slightly reduced at 0.1 mg/dl (0.16–0.39). Biliary atresia, viral infection, autoimmunity, alpha-1-antitrypsin deficiency, and urea cycle disorder were excluded. On sonography, the spleen was enlarged at 9.4 cm (<7 cm). The kidneys appeared normal. Magnetic resonance cholangiopancreatography identified a patent biliary duct without distortion. PFIC3 was suspected and “PFIC panel screening” followed. Liver biopsy was not undertaken. We received the patient for LTx evaluation.
At age 8 months, the prothrombin time was 19% (lower level of normal [LLN] 81); the INR was 3.9, an activated partial thromboplastin time was 102 s (upper level of normal [ULN] 42), with serum fibrinogen 130 mg/dL (LLN 202), antithrombin-III <20% (LLN 94), and serum albumin 3122 mg/dL (LLN 3800). TB and DB were respectively 26.81 mg/dl (ULN 1) and 21.57 mg/dl (ULN 0.3); AST was 1378 U/L (ULN 77), ALT 262 U/L (ULN 49), ALP 1028 U/L (ULN 469), and LDH 614 U/L (ULN 450). GGT was within normal range.
Split LTx (segments II–III) from the father was uneventful, with Roux-en-Y placement. At age 2 years, the patient’s growth and psychomotor development were age-appropriate and graft function was normal. Hearing loss was not detected on sensorineural testing.
Results and discussion
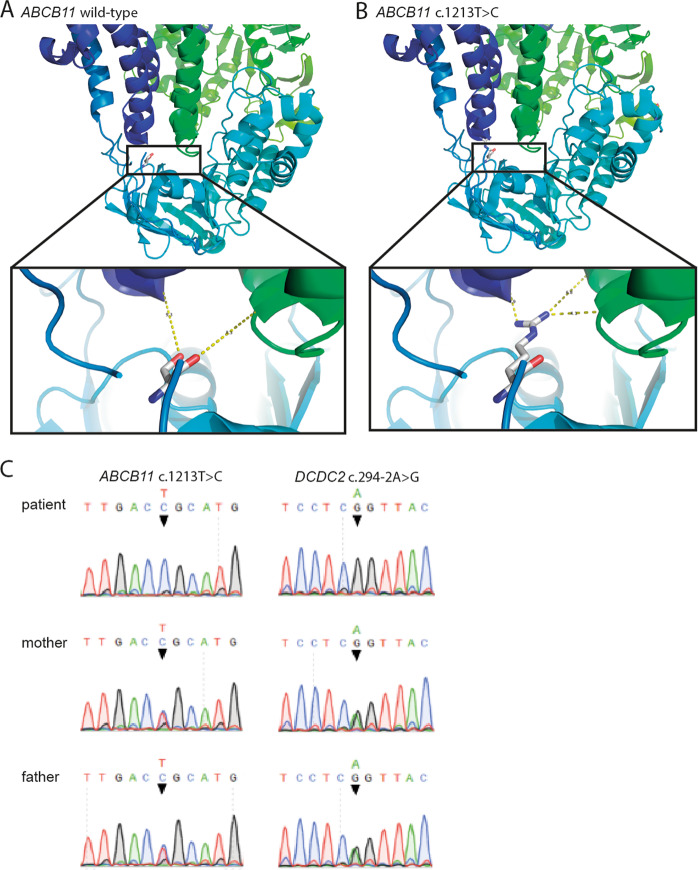
Our patient at presentation had high-GGT intrahepatic cholestasis. The coding regions and flanking intronic regions of ABCB4, ABCB11, and ATP8B1 were initially examined for sequence variants. He proved to harbor a homozygous c.1213 T>C (p.(Cys405Arg)) variant in ABCB11, which has a very low allele frequency of 1:9682 in the African population in the ExAC database. This variant was not observed in other populations despite high sequence coverage around this codon, and all four in silico algorithms employed assess it as affecting protein function. 3D protein modeling suggests that this variant impairs substrate entry at the junction between a transmembrane domain and the nucleotide-binding domain (Fig. 1a, b). However, ACMG guidelines classify the variant as of unknown significance [15], and the clinical course was highly unusual for ABCB11 disease alone. ABCB11 disease is associated with low- or normal-GGT intrahepatic cholestasis [1], and few persons with ABCB11 disease require transplantation in the first postnatal years [5]. These observations suggested an etiology beyond ABCB11 disease.
Fig. 1. BSEP protein modeling and genetic analysis.
a, b The homozygous missense variant c.1213T>C changes cysteine-405 to arginine in human. Cysteine-405 corresponds to serine-373 in murine P-glycoprotein. The structure of murine P-glycoprotein serves to model the identified substitution with the larger, charged and hydrophilic arginine. The mutated residue lies in interhelical domain 6, close to nucleotide-binding domain 1. Distances to glutamine-155 in interhelical domain 1 (dark blue) and serine-905 in interhelical domain 4 (green) are altered, suggesting that this variant may impair substrate entry into BSEP at the transmembrane domain: nucleotide-binding domain junction. c Sequence chromatograms, DCDC2, and ABCB11 of patient and his parents.
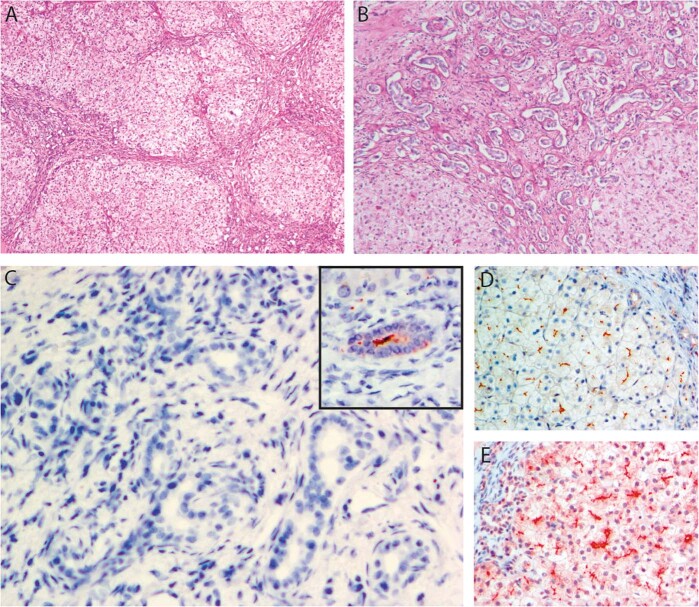
Histopathologic study of the explanted liver revealed micronodular cirrhosis. Chronic hepatocellular and canalicular cholestasis, with giant-cell change of hepatocytes, accompanied ductal-plate malformation peripherally and sclerosing cholangitis near the hilum. BSEP expression was unremarkable (not implying normal function), as was that of MDR3, GGT, and several other canalicular antigens (Fig. 2a–e). Coarsely granular “Byler bile” was not found on ultrastructural study. The findings suggested DCDC2 disease [9, 10]. Immunostaining failed to demonstrate DCDC2 expression. DCDC2 analysis detected homozygosity for the variant NG_012829.2(DCDC2_v001):c.294-2 A>G (p.?), altering a canonical-receptor splice-site and predicted to abolish protein expression (ClinVar database, https://www.ncbi.nlm.nih.gov/clinvar/variation/502281/). The variant is not listed in ExAC and gnomAD databases, although sequencing coverage around this nucleotide position is good. The parents were each heterozygous for the DCDC2 and ABCB11 variants (Fig. 1c).
Fig. 2. Histopathologic findings.
Portal fibrosis with abnormal bile ducts and deficiency of portal-venule radicles (a, b). c DCDC2 signal is not detectable at cholangiocytes. Inset, normal DCDC2 expression, control sample. Note intraluminal (ciliary–axonemal) pattern. d, e BSEP and MDR3 expression is unremarkable. a ×40 (all magnifications original), hematoxylin and eosin; b ×100, diastase/periodic acid-Schiff technique; c ×400, mouse monoclonal anti-DCDC2 antibody (Santa Cruz Biotechnology, Santa Cruz, CA; sc-166051); 3-amino-9-ethylcarbazole chromogen, hematoxylin counterstain; d ×200, rabbit polyclonal anti-BSEP antibody (Sigma Life Science, St. Louis, MO; HPA019035), chromogen and counterstain as c; e ×200, anti-MDR3 antibody (Kamiya Biomedical, Seattle, WA; MC-224); chromogen and counterstain as c and d.
Our patient’s genetic status was evaluated sequentially and incrementally, rather than through initial wide-ranging panel screening or exomic analysis. Perhaps to assess patients with intrahepatic cholestasis, whether spontaneous or associated with shifts in hormonal milieu, infection, malignancy, or drug ingestion, for variants in a broad spectrum of “cholestasis genes” will add to understanding of how, and why, such variants do or do not achieve clinical penetrance as pruritus, jaundice, or inflammation and fibrosis—or, with DCDC2 in our patient, as renal disease or hearing loss. Such panel screening is routine in genetic diagnosis in many specialized centers, including ours, but, as with our patient, exceptions occur.
When genetic studies provide an answer to the question of what underlies a patient’s disorder, depending on how the question is asked the answer may not be complete. We were content with the initial answer of “ABCB11 disease”. However, the discrepancy between ABCB11 disease and high-GGT values, like the unusual speed with which our patient’s disorder advanced, might have prompted us to conduct liver biopsy (possibly identifying the ductal-plate malformation), or to request broader sequencing (possibly identifying DCDC2 variant). We accordingly advocate skepticism toward genetic findings when they and clinical or histopathologic observations are not in full accord. Our patient had both NSC and PFIC, an example of the potential complexity of “cholestasis-gene” disease and how histopathologic evaluation can both pose questions—here, suggesting DCDC2 variants as likely responsible for aspects of the patient’s disease—and fail to answer them, as when normal BSEP expression leaves the question of functional impairment of ABCB11 variants open.
Of interest is the fall in GGT activity as the patient entered end-stage liver disease with synthetic failure, a setting associated with normal-range serum GGT activity despite conjugated hyperbilirubinemia. We ascribe this biomarker-value shift to reduced synthesis of bile acids, necessary to elute GGT from canalicular membranes into bile (GGT and bile, leaking between damaged hepatocytes, thus together enter blood plasma), although a contribution from nonspecifically reduced synthesis of BSEP cannot be excluded. Admittedly, normalization of GGT values also could have been due to ursodeoxycholic acid administration. Biliary tract diseases in children are frequently characterized initially by elevated GGT activity that normalizes in late phases of disease, indicating terminal liver failure and exhaustion of the functionally intact hepatobiliary cell pool. Our patient’s course is a reminder that interpretation of GGT values requires assessment of clinical context as well as genetic findings, and that the diagnostic work-up of infantile cholestasis should follow current algorithms [16, 17].
Compliance with ethical standards
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Maggiore G, Bernard O, Riely CA, Hadchouel M, Lemonnier A, Alagille D. Normal serum gamma-glutamyl-transpeptidase activity identifies groups of infants with idiopathic cholestasis with poor prognosis. J Pediatr. 1987;111:251–2. doi: 10.1016/S0022-3476(87)80079-3. [DOI] [PubMed] [Google Scholar]
- 2.Bull LN, Thompson RJ. Progressive familial intrahepatic cholestasis. Clin liver Dis. 2018;22:657–69.. doi: 10.1016/j.cld.2018.06.003. [DOI] [PubMed] [Google Scholar]
- 3.Knisely AS, Gissen P. Trafficking and transporter disorders in pediatric cholestasis. Clin liver Dis. 2010;14:619–33. doi: 10.1016/j.cld.2010.08.001. [DOI] [PubMed] [Google Scholar]
- 4.Sambrotta M, Strautnieks S, Papouli E, Rushton P, Clark BE, Parry DA, et al. Mutations in TJP2 cause progressive cholestatic liver disease. Nat Genet. 2014;46:326–8. doi: 10.1038/ng.2918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pawlikowska L, Strautnieks S, Jankowska I, Czubkowski P, Emerick K, Antoniou A, et al. Differences in presentation and progression between severe FIC1 and BSEP deficiencies. J Hepatol. 2010;53:170–8. doi: 10.1016/j.jhep.2010.01.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Liu Y, Sun LY, Zhu ZJ, Wei L, Qu W, Zeng ZG. Liver transplantation for progressive familial intrahepatic cholestasis. Ann Transplant. 2018;23:666–73. doi: 10.12659/AOT.909941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mehl A, Bohorquez H, Serrano MS, Galliano G, Reichman TW. Liver transplantation and the management of progressive familial intrahepatic cholestasis in children. World J Transplant. 2016;6:278–90. doi: 10.5500/wjt.v6.i2.278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Davit-Spraul A, Fabre M, Branchereau S, Baussan C, Gonzales E, Stieger B, et al. ATP8B1 and ABCB11 analysis in 62 children with normal gamma-glutamyl transferase progressive familial intrahepatic cholestasis (PFIC): phenotypic differences between PFIC1 and PFIC2 and natural history. Hepatology. 2010;51:1645–55.. doi: 10.1002/hep.23539. [DOI] [PubMed] [Google Scholar]
- 9.Girard M, Bizet AA, Lachaux A, Gonzales E, Filhol E, Collardeau-Frachon S, et al. DCDC2 mutations cause neonatal sclerosing cholangitis. Hum Mutat. 2016;37:1025–9. doi: 10.1002/humu.23031. [DOI] [PubMed] [Google Scholar]
- 10.Grammatikopoulos T, Sambrotta M, Strautnieks S, Foskett P, Knisely AS, Wagner B, et al. Mutations in DCDC2 (doublecortin domain containing protein 2) in neonatal sclerosing cholangitis. J Hepatol. 2016;65:1179–87. doi: 10.1016/j.jhep.2016.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schueler M, Braun DA, Chandrasekar G, Gee HY, Klasson TD, Halbritter J, et al. DCDC2 mutations cause a renal-hepatic ciliopathy by disrupting Wnt signaling. Am J Hum Genet. 2015;96:81–92. doi: 10.1016/j.ajhg.2014.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Grati M, Chakchouk I, Ma Q, Bensaid M, Desmidt A, Turki N, et al. A missense mutation in DCDC2 causes human recessive deafness DFNB66, likely by interfering with sensory hair cell and supporting cell cilia length regulation. Hum Mol Genet. 2015;24:2482–91. doi: 10.1093/hmg/ddv009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Povysil G, Tzika A, Vogt J, Haunschmid V, Messiaen L, Zschocke J, et al. panelcn.MOPS: copy-number detection in targeted NGS panel data for clinical diagnostics. Hum Mutat. 2017;38:889–97. doi: 10.1002/humu.23237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Li J, Jaimes KF, Aller SG. Refined structures of mouse P-glycoprotein. Protein Sci. 2014;23:34–46. doi: 10.1002/pro.2387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–24. doi: 10.1038/gim.2015.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nicastro E, Di Giorgio A, Marchetti D, Barboni C, Cereda A, Iascone M, et al. Diagnostic yield of an algorithm for neonatal and infantile cholestasis integrating next-generation sequencing. J Pediatr. 2019;211:54–62 e4.. doi: 10.1016/j.jpeds.2019.04.016. [DOI] [PubMed] [Google Scholar]
- 17.Nicastro E, D’Antiga L. Next generation sequencing in pediatric hepatology and liver transplantation. Liver Transpl. 2018;24:282–93. doi: 10.1002/lt.24964. [DOI] [PubMed] [Google Scholar]