Abstract
To understand the molecular epidemiology and antibiotic resistance of air and clinical isolates of Acinetobacter baumannii, the intensive care unit settings of a hospital in Northern China were surveyed in 2014. Twenty non-duplicate A. baumannii isolates were obtained from patients and five isolates of airborne A. baumannii were obtained from the wards’ corridors. Pulsed field gel electrophoresis (PFGE) and multilocus sequence typing (MLST) were used to analyze the homology relationships of isolates. Resistance and resistance genes were detected by drug susceptibility test and PCR. The results demonstrated that all isolates can be classified into eight PFGE types and four sequence types (ST208, ST195, ST369 and ST530). A pair of isolates from patients (TAaba004) and from the air (TAaba012) that share 100% similarity in PFGE was identified, indicating that air might be a potential and important transmission route for A. baumannii. More than 80% of the isolates were resistant to carbapenems and aminoglycoside antibiotics. Twenty-four isolates, which were resistant to carbapenems, carried the blaOXA-23-like gene. The data indicated that air might be an alternative way for the transmission of A. baumannii. Hospitals should pay more attention to this route, and design new measures accordingly.
Key words: Acinetobacter baumannii, molecular epidemiology, antibiotic resistance, airborne, nosocomial infection
Introduction
Outbreaks caused by multidrug resistant Acinetobacter baumannii strains have been intensively documented worldwide (Zarrilli et al. 2013). With the increasing number of carbapenem-resistant A. baumannii, outbreaks of A. baumannii were expanding. One data showed that resistance to carbapenem increased from 1.0% in 2003 to 58.0% in 2008 in Detroit Medical Center health system (Reddy et al. 2010). In China, A. baumannii has recently become an important pathogen causing the nosocomial infection in hospitals. The infection rate of A. baumannii is always in the top three in total clinical isolates of Gram-negative bacilli, second only to Escherichia coli and Klebsiella pneumoniae. In 2014, A. baumannii isolated from nonfermenting Gram-negative bacilli was always in the first place in clinical tests (Wang et al. 2013; Hu et al. 2014; Hu et al. 2015).
The analysis results of multilocus sequence typing (MLST) demonstrated that multiple STs were prevalent in the outbreak regions. Ying reported that ST208 was the most prevalent, followed by ST191 and ST729 in the 398 isolates collected in seven regions of South China in 2012 (Ying et al. 2015). Through the whole genome sequencing, it was found that blaOXA-23-producing A. baumannii ST208 strains emerged and rapidly spread in Hangzhou First People’s Hospital (Chen et al. 2018). Just recently, Jeon et al. (2018) reported that sequence type (ST) 191 was the predominant clone, followed by the ST208 and ST369 in the hospital in Korea.
At present, A. baumannii strains were mainly isolated from the clinical specimens, as blood, urine, tissues, and sputum (Bogaerts et al. 2006; Zong et al. 2008; Mendes et al. 2009; Yang et al. 2010; Chen et al. 2013). In our laboratory, we have been focused on the epidemiology of A. baumannii isolates from sputum (Jiang et al. 2013; Jiang et al. 2014). However, some reports have shown that A. baumannii can survive and transmit in the air (Allen et al. 1987; Obbard et al. 2003). To date, the homology relationship between the airborne A. baumannii isolates from hospital air and those isolated from patients has not been extensively studied. In this study, twenty-five isolates of non-duplicate A. baumannii were obtained from patients and the air of wards’ corridors during May – Jun. and Oct. – Nov. of 2014. The molecular epidemiological characterization of the isolated A. baumannii, such as drug resistance, and types of resistance genes, was compared to understand the relationship between the airborne A. baumannii and those isolated from patients.
Experimental
Materials and Methods
Ethical approval. Samples of patients were acquired during a routine checkup by medical staffs. The study was approved by the Ethics Committee of Tai’an City Central Hospital and carried out in accordance with the approved guidelines.
Source of the isolates. From May 12 to Jun. 5 and Oct. 11 to Nov. 15, 2014, A. baumannii isolates were obtained from patients hospitalized in four different wards, including the intensive care unit (ICU), emergency intensive care unit (EICU), respiratory intensive care unit (RICU) and geriatrics ward (GW). Meanwhile, airborne A. baumannii isolates were obtained from the air of the wards and adjacent corridors using the Andersen 6-stage air sampler. Each stage of the sampler has a plate with 400 holes of uniform diameter, according to the method described in previous studies (Andersen et al. 1958; Obbard et al. 2003). In brief, the sampler was placed in the center of wards and corridors (closed) at a height of 1.5 m above the ground. Airborne A. baumannii was collected at a calibrated flow rate of 28.3 l/min for 20 min, and MacConkey agar (Oxoid, Basingstoke, England) was used as the sampling medium. At each sampling site, four duplicate air samples were collected once per week. The number of colonies was about 30–300 in each stage. Temperature and relative humidity were recorded throughout the sampling periods (temperature 20–25°C; relative humidity 73–83%).
Bacterial identification and drug susceptibility test. Bacterial identification was performed by using an VITEK-2 bacterial instrument (BioMerieux, Lyons, France) following the operation instruction. Drug susceptibility test was performed by two different methods: the sensitivity of tigecycline and polymyxin B was detected by the Etest method (AB Biodisk, Solna, Sweden); the sensitivity of the remaining fourteen antibiotics was detected using the minimal inhibitory concentration method. The susceptibility testing result of tigecycline was explained according to Food and Drug Administration (FDA) guideline (http://www.fda.org.uk/sitemap.aspx). The criteria of the susceptibility of other agents were adapted from the Clinical and Laboratory Standards Institute (CLSI) (http://clsi.org/standards/). The criteria of multidrug resistant (MDR) A. baumannii, extensively drug-resistant (XDR) A. baumannii and pandrug-resistant (PDR) A. baumannii were referred to the previously described (Magiorakos et al. 2012).
Pulsed field gel electrophoresis (PFGE). Clonal relatedness of 25 A. baumannii strains isolated in this study was determined by PFGE (Ribot et al. 2006). Briefly, the bacterial chromosomal DNA was digested with 60 U of ApaI (Takara, Dalian, China) in a 37°C. Salmonella serotype Braenderup strain (H9812) was digested with XbaI (Takara, Dalian, China) and used as the molecular weight standard, the DNA fragments were separated on agarose gel using a clamped homogeneous electric field electrophoresis-Mapper XA system (Bio-Rad, California, USA). The PFGE images were analyzed using the Gel Doc software (Bio-Rad, California, USA) and the genetic relatedness of the A. baumannii isolates were evaluated using the BioNumerics software (Applied Maths, Sint-Martens-Latem, Belgium). The cutoff level of 85% was set to define clonal clustering of the PFGE types.
Multilocus sequence typing (MLST). The sequence types (STs) of A. baumannii strains were determined by MLST using the previously described method (Bartual et al. 2005). In brief, the internal fragments of seven housekeeping genes, including citrate synthase (gltA), DNA gyrase subunit B (gyrB), glucose dehydrogenase B (gdhB), homologous recombination factor (recA), 60-kDa chaperonin (cpn60), glucose-6-phosphate isomerase (gpi) and RNA polymerase σ70 factor (rpoD) were amplified by PCR. The positive sequences were further analyzed using the A. baumannii PubMLST database (http://pubmlst.org/abaumannii).
Table I.
Susceptibility profiles of 20 clinical A. baumannii isolates.
| Antibiotics | Susceptible | Resistant | ||
|---|---|---|---|---|
| Number | Rate (%) | Number | Rate (%) | |
| Amikacin | 3 | 15.0% | 17 | 85.0% |
| Ceftazidime | 0 | 0.0% | 20 | 100% |
| Ciprofloxacin | 0 | 0.0% | 20 | 100% |
| Levofloxacin | 0 | 0.0% | 20 | 100% |
| Gentamicin | 0 | 0.0% | 20 | 100% |
| Tobramycin | 3 | 15.0% | 17 | 85.0% |
| Ceftriaxone | 0 | 0.0% | 20 | 100% |
| Cefotaxime | 0 | 0.0% | 20 | 100% |
| Cefepime | 0 | 0.0% | 20 | 100% |
| Imipenem | 0 | 0.0% | 20 | 100% |
| Meropenem | 0 | 0.0% | 20 | 100% |
| Trimethoprim/sulfamethoxazole | 6 | 30.0% | 14 | 100% |
| Piperacillin/tazobactam | 0 | 0.0% | 20 | 100% |
| Tigecycline | 20 | 100% | 0 | 0.0% |
| Polymyxin B | 20 | 100% | 0 | 0.0% |
Table II.
Susceptibility profiles of the airborne and clinical strains with the highest similarity.
| Strain / Antibiotics | TAaba004 | TAaba012 |
|---|---|---|
| Amikacin | R | R |
| Ceftazidime | R | R |
| Ciprofloxacin | R | R |
| Levofloxacin | R | R |
| Gentamicin | R | R |
| Tobramycin | R | R |
| Ceftriaxone | R | R |
| cefotaxime | R | R |
| Cefepime | R | R |
| Imipenem | R | R |
| Meropenem | R | R |
| Trimethoprim/sulfamethoxazole | R | R |
| Piperacillin/tazobactam | R | R |
| Tigecycline | S | S |
| Polymyxin B | S | S |
Note: “R” indicates resistant, “S” indicates susceptible.
Drug resistance genes. Bacterial DNA were extracted and the carbapenemase genes, including blaOXA-23-like, blaOXA-24-like, blaOXA-48, blaOXA-50, blaOXA-51-like, blaOXA-55, blaOXA-58-like, blaOXA-60, and blaOXA-64, the 16S rRNA methylase genes armA and rmtB were amplified by PCR as previously described (Shen et al. 2008; Yang et al. 2011). The positive bands were sequenced and then compared to the Genbank database (Jiang et al. 2014).
Results
Isolation of A. baumannii strains. Twenty A. baumannii clinical strains were isolated from 356 patients and five strains were isolated from 4 out of 64 air samples in duplicate during this experimental period (8 weeks). Wherein two A. baumannii strains (TAaba015 and TAaba014) were isolated from the same air sample from ICU corridor in 08.11.2014.
Drug susceptibility patterns. A total of twenty-five non-duplicate A. baumannii isolates were obtained, including twenty from the clinical samples and five from the adjacent corridors of the wards. Fifteen commonly used antibiotics (amikacin, ceftazidime, ciprofloxacin, levofloxacin, gentamicin, tobramycin, ceftriaxone, cefotaxime, cefepime, imipenem, meropenem, trimethoprim/sulfamethoxazole, piperacillin/tazobactam, tigecycline, and polymyxin B) were selected for drug susceptibility test. The result showed that only one out of the five airborne isolates (TAaba013) was non-MDR A. baumannii, which was only resistant to ceftazidime, ceftriaxone, cefotaxime, and cefepime. All twenty clinical isolates were MDR A. baumannii, and susceptible to tigecycline and polymyxin B.
Drug resistance genes. All isolates had the blaOXA-51-like resistant gene, and twenty-four had the blaOXA-23-like gene except one airborne isolate (TAaba014). However, other seven resistance genes of carbapenemases were not detected in this study. These results indicated that blaOXA-23-like gene was responsible for the carbapenemase resistance. In addition, twenty isolates carried the 16S rRNA methylase gene armA. No rmtB gene was detected in all isolates.
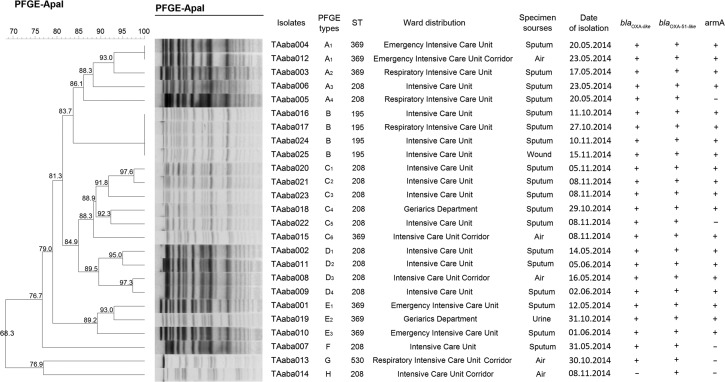
PFGE and MLST. As shown in Fig. 1, the isolates can be clustered into eight PFGE types (A-H) and four STs (ST208, ST369, ST195 and ST530). One airborne isolate (TAaba012) had the same PFGE and ST types as one clinical isolates (TAaba004). They were isolated from the same ward (EICU) during the same period (May – Jun 2014). Similarly, in another ward (intensive care unit, ICU), one airborne isolate (TAaba008) was found to be closely related to three clinical isolates (TAaba002, TAaba009, and TAaba011) in terms of PFGE and ST types (type D and ST208, respectively).
Fig. 1.
Dendrogram showing the PFGE and MLST profiles of the A. baumannii isolates. The dendogram was generated by the BioNumerics software. The sampling date, isolate number, ward and source, PFGE and ST types and resistance genes are shown for each isolate.
In the second sampling period (Oct. 11 to Nov. 15, 2014), we collected three non-reduplicate airborne isolates (TAaba013, TAaba014 and TAaba015). We did not find any clinical isolates with the same PFGE and ST types. In particular, the PFGE and ST types of TAaba013 were different from any isolates obtained in this study.
Discussion
A. baumannii is a predominant cause of nosocomial infections, and the organism has been detected in water, soil, and on the surface of human body. A recent study has shown that some A. baumannii environmental isolates are able to survive in tap, normal saline, and distilled water with a wide range of pH and temperature for a long period of time (Obeidat et al. 2014). It makes it harder for the prevention of A. baumannii spread in a nosocomial environment. A. baumannii is often transmitted through physical objects, like stethoscopes, computers, milk pumps, and headboards of beds. In this study, we investigated the genetic relationship of the A. baumannii isolates from the air and patients, and the possible transmission pathway through the air.
Our results show that one airborne A. baumannii (TAaba012) isolated in the EICU and one clinical isolate (TAaba004) isolated in the same ward shares 100% similarity in PFGE and MLST, indicating A. baumannii might be transmitted through air. Another airborne isolate (TAaba008) obtained in ICU was also found to be the same strain as three clinical isolates (TAaba002, TAaba009, and TAaba011). Based on the genetic relationship of the airborne and clinical isolates, we speculate that there could exist an air-mediated spread of this A. baumannii strain. Moreover, many studies demonstrated that airborne A. baumannii could be detected in ICU and airborne transmission of A. baumannii was possible (Munoz-Price et al. 2013; Spellberg et al. 2013; Yakupogullari et al. 2016).
A total of twenty clinical isolates were obtained during the sampling period of this study, but only one isolate was found in the air, indicating airborne transmission might be affected by many factors, such as length of hospital stay, humidity, temperature, etc. Since our dataset are relatively small, we cannot draw any exclusive conclusion. Moreover, because horizontal gene transfer that can jeopardize clonal strains, PFGE is no longer the most effective technique for A. baumannii typing (Salipante et al. 2015), and our data can only confirm that the same strain was isolated from air and the specimens from patients, which could suggest an alternative route of transmission through air. More systematic studies with a longer period of sampling should be constructed to better understand the airborne pathway.
Treatment of A. baumannii infections is particularly difficult because of the resistance to a broad range of antibiotics, especially with the increasing of resistant to carbapenems (Lee et al. 2012). Although mechanisms of resistance to carbapenems were very complex, resistance genes play an important role in this process. It has been reported that blaOXA-23-like-producing A. baumannii were found worldwide (Le et al. 2008; Runnegar et al. 2010; Mosqueda et al. 2013). In this study, the analysis showed that all strains had blaOXA-51-like gene, one intrinsic gene in A. baumannii. Moreover, 96% strains (24/25) carried the blaOXA-23-like gene, except TAaba014. These results indicated that both genes were mainly responsible for the carbapenems resistance in this area, especially the blaOXA-23like gene and it was generally believed that blaOXA-23-like gene was responsible for carbapenem resistance in China (Ruan et al. 2010; Liu et al. 2015). Additionally, it has been reported that 16S rRNA methylase gene armA was closely related to the aminoglycosides resistance (Yokoyama et al. 2003), and 80% (20/25) strains had the 16S rRNA methylase gene armA. All of the strains showed the resistance to amikacin, tobramycin and gentamicin. Antibiotic resistance is a serious global public problem.
Taken together, in this study, we isolated twenty clinical A. baumannii from patients and five airborne A. baumannii from the ward environment. Out of the five airborne A. baumannii, one was found to have the same PFGE and ST type with one strain isolated from patients, indicating that the air may be an important and potential transmission way for A. baumannii. Considering the seriousness of nosocomial infections of A. baumannii, prophylactic strategies in controlling the air transmission of A. baumannii should be considered when new measures are designed for controlling nosocomial infections.
Acknowledgements
This research was supported by Natural Science Foundation of Shandong Province (ZR2013HM009).
Footnotes
Author contributions
M. Jiang and Z. Zhang performed the main experiment and wrote the manuscript. Y. Mu and N. Li analyzed data, Z. Zhang and S. Han reviewed the manuscript and approved it.
Conflict of interest statement
The authors declared there was no any conflict of interests.
Literature
- Zarrilli R, Pournaras S, Giannouli M, Tsakris A.. 2013. Global evolution of multidrug-resistant Acinetobacter baumannii clonal lineages. Int J Antimicrob Agents. 41:11–19. [DOI] [PubMed] [Google Scholar]
- Reddy T, Chopra T, Marchaim D, Pogue JM, Alangaden G, Salimnia H, Boikov D, Venezia SN, Akins R, Selman P, et al. 2010. Trends in antimicrobial resistance of Acinetobacter baumannii isolates from a metropolitan Detroit health system. Antimicrob Agents Ch. 54(5):2235–2238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu F, Zhu D, Wang F, Jiang X, Xu Y, Zhang X.. 2015. CHINET 2014 surveillance of bacterial resistance in China. Chin J Infect Chemother. 15:401–410. [Google Scholar]
- Wang F, Zhu D, Hu F, Du Y.. 2013. CHINET 2012 surveillance of bacterial resistance in China. Chin J Infect Chemother. 13:321–330. [Google Scholar]
- Ying J, Lu JW, Zong L, Li AL, Pan RW, Cheng C, Li KP, Chen LQ, Ying JC, Tou HF, et al. 2016. Molecular epidemiology and characterization of genotypes of Acinetobacter baumannii isolates from regions of south China. Jpn J Infect Dis. 69(3):180–185. [DOI] [PubMed] [Google Scholar]
- Chen Q, Zhou JW, Fan JZ, Wu SH, Xu LH, Jiang Y, Ruan Z, Yu YS, Yu DJ, Wang XJ.. 2018. Simultaneous emergence and rapid spread of three OXA-23 producing Acinetobacter baumannii ST208 strains in intensive care units confirmed by whole genome sequencing. Infect Genet Evol. 58:243–250. [DOI] [PubMed] [Google Scholar]
- Jeon H, Kim S, Kim MH, Kim SY, Nam D, Park SC, Park SH, Bae H, Lee HJ, Cho JH, et al. 2018. Molecular epidemiology of carbapenem-resistant Acinetobacter baumannii isolates from a Korean hospital that carry blaOXA-23. Infect Genet Evol. 58:232–236. [DOI] [PubMed] [Google Scholar]
- Bogaerts P, Naas T, Wybo I, Bauraing C, Soetens O, Pierard D, Nordmann P, Glupczynski Y.. 2006. Outbreak of infection by carbapenem-resistant Acinetobacter baumannii producing the carbapenemase OXA-58 in Belgium. J Clin Microbiol. 44:4189–4192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen ZH, Liu WE, Zhang YL, Li YB, Jian ZJ, Deng HL, Zou MX, Liu YY.. 2013. Molecular epidemiology of carbapenem-resistant Acinetobacter spp. from XiangYa Hospital, in Hunan Province, China. J Basic Microbiol. 53:121–127. [DOI] [PubMed] [Google Scholar]
- Mendes RE, Spanu T, Deshpande L, Castanheira M, Jones RN, Fadda G.. 2009. Clonal dissemination of two clusters of Acinetobacter baumannii producing OXA-23 or OXA-58 in Rome, Italy. Clin Microbiol Infect. 15(6):588–592. [DOI] [PubMed] [Google Scholar]
- Yang SC, Chang WJ, Chang YH, Tsai YS, Yang TP, Juan CW, Shiau MY.. 2010. Prevalence of antibiotics resistance and OXA carbapenemases genes in multidrug-resistant Acinetobacter baumannii isolates in central Taiwan. Eur J Clin Microbiol Infect Dis. 29:601–604. [DOI] [PubMed] [Google Scholar]
- Zong Z, Lü X, Valenzuela JK, Partridge SR, Iredell J.. 2008. An outbreak of carbapenem-resistant Acinetobacter baumannii producing OXA-23 carbapenemase in western China. Int J Antimicrob Agents. 31:50–54. [DOI] [PubMed] [Google Scholar]
- Jiang MJ, Zhao SP, Li JM, Zhang FS.. 2013. Molecular epidemiological study and detection of multidrug resistant Acinetobacter baumannii related resistance genes. Afr J Microbiol Res. 7:5496–5502. [Google Scholar]
- Jiang MJ, Zhang ZJ, Zhao SP.. 2014. Epidemiological characteristics and drug resistance analysis of multidrug-resistant Acinetobacter baumannii in a China hospital at a certain time. Pol J Microbiol. 63:275–281. [PubMed] [Google Scholar]
- Allen KD, Green HT.. 1987. Hospital outbreak of multi-resistant Acinetobacter anitratus: an airborne mode of spread? J Hosp Infect. 9:110–119. [DOI] [PubMed] [Google Scholar]
- Obbard JP, Fang LS.. 2003. Airborne concentrations of bacteria in a hospital environment in Singapore. Water Air Soil Pollut. 144:333–341. [Google Scholar]
- Andersen AA. 1958. New sampler for the collection, sizing, and enumeration of viable airborne particles. J Bacteriol. 76:471–484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magiorakos AP, Srinivasan A, Carey RB, Carmeli Y, Falagas ME, Giske CG, Harbarth S, Hindler JF, Kahlmeter G, Olsson-Lijequist B, et al. 2012. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 18(3):268–281. [DOI] [PubMed] [Google Scholar]
- Ribot EM, Fair MA, Gautom R, Cameron DN, Hunter SB, Swaminathan B, Barrett TJ.. 2006. Standardization of pulsed-field gel electrophoresis protocols for the subtyping of Escherichia coli O157:H7, Salmonella, and Shigella for PulseNet. Foodborne Pathog Dis. 3(1):59–67. [DOI] [PubMed] [Google Scholar]
- Bartual SG, Seifert H, Hippler C, Luzon MAD, Wisplinghoff H, Rodríguez-Valera F.. 2005. Development of a multilocus sequence typing scheme for characterization of clinical isolates of Acinetobacter baumannii. J Clin Microbiol. 43:4382–4390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen J, Zhu D, Wang M.. 2008. The relationship between acquired carbapenemases and resistance of gram-negative bacilli. Chin J Lab Med. 31:408–414. [Google Scholar]
- Yang Y, Ye H, Zhou X.. 2011. Detection of New Delhi metallo-β-lactamase I gene in Klebsiella ozaenae and Acinetobacter baumannii. Chin J Lab Med. 32:1407–1409. [Google Scholar]
- Obeidat N, Jawdat F, Al-Bakri AG, Shehabi AA.. 2014. Major biologic characteristics of Acinetobacter baumannii isolates from hospital environmental and patients’ respiratory tract sources. Am J Infect Control. 42:401–404. [DOI] [PubMed] [Google Scholar]
- Spellberg B, Bonomo RA.. 2013. “Airborne Assault”: A new dimension in Acinetobacter baumannii transmission. Crit Care Med. 41(8):2042–2044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munoz-Price LS, Fajardo-Aquino Y, Arheart KL, Cleary T, DePascale D, Pizano L, Namias N, Rivera JI, O’Hara JA, Doi Y.. 2013. Aerosolization of Acinetobacter baumannii in a trauma ICU. Crit Care Med. 41(8):1915–1918. [DOI] [PubMed] [Google Scholar]
- Yakupogullari Y, Otlu B, Ersoy Y, Kuzucu C, Bayindir Y, Kayabas U, Togal T, Kizilkaya C.. 2016. Is airborne transmission of Acinetobacter baumannii possible: A prospective molecular epidemiologic study in a tertiary care hospital. Am J Infect Control. 44: 1595–1599. [DOI] [PubMed] [Google Scholar]
- Salipante SJ, SenGupta DJ, Cummings LA, Land TA, Hoogestraat DR, Cookson BT.. 2015. Application of whole-genome sequencing for bacterial strain typing in molecular epidemiology. J Clin Microbiol. 53:1072–1079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee JY, Ko KS.. 2012. Antimicrobial resistance and clones of Acinetobacter Species and Pseudomonas aeruginosa. J Bacteriol Virol. 42(1):1. [Google Scholar]
- Le Hello S, Falcot V, Lacassin F, Baumann F, Nordmann P, Naas T.. 2008. Molecular epidemiology of carbapenem-resistant Acinetobacter baumannii in New Caledonia. Clin Microbiol Infect. 14:977–981. [DOI] [PubMed] [Google Scholar]
- Mosqueda N, Espinal P, Cosgaya C, Viota S, Plasensia V, Alvarez-Lerma F, Montero M, Gomez J, Horcajada JP, Vila J, et al. 2013. Globally expanding carbapenemase finally appears in Spain: nosocomial outbreak of Acinetobacter baumannii producing plasmid-encoded OXA-23 in Barcelona, Spain. Antimicrob Agents Chemother. 57:5155–5157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Runnegar N, Sidjabat H, Goh HMS, Nimmo GR, Schembri MA, Paterson DL.. 2010. Molecular epidemiology of multidrug-resistant Acinetobacter baumannii in a single institution over a 10-year period. J Clin Microbiol. 48:4051–4056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu LL, Ji SJ, Ruan Z, Fu Y, Fu YQ, Wang YF, Yu YS.. 2015. Dissemination of blaOXA-23 in Acinetobacter spp. In China: main roles of conjugative plasmid pAZJ221 and Transposon Tn2009. Antimicrob Agents Chemother. 59:1998–2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruan Z, Chen Y, Jiang Y, Zhou H, Zhou Z, Fu Y, Wang H, Wang Y, Yu Y.. 2013. Wide distribution of CC92 carbapenem-resistant and OXA-23-producing Acinetobacter baumannii in multiple provinces of China. Int J Antimicrob Agents. 42:322–328. [DOI] [PubMed] [Google Scholar]
- Yokoyama K, Doi Y, Yamane K, Kurokawa H, Shibata N, Shibayama K, Yagi T, Kato T, Arakawa Y.. 2003. Acquisition of 16S rRNA methylase gene in Pseudomonas aeruginosa. Lancet. 362: 1888–1893. [DOI] [PubMed] [Google Scholar]