Abstract
Background
The COVID-19 pandemic continues to escalate. There is urgent need to stratify patients. Understanding risk of deterioration will assist in admission and discharge decisions, and help selection for clinical studies to indicate where risk of therapy-related complications is justified.
Methods
An observational cohort of patients acutely admitted to two London hospitals with COVID-19 and positive SARS-CoV-2 swab results was assessed. Demographic details, clinical data, comorbidities, blood parameters and chest radiograph severity scores were collected from electronic health records. Endpoints assessed were critical care admission and death. A risk score was developed to predict outcomes.
Findings
Analyses included 1,157 patients. Older age, male sex, comorbidities, respiratory rate, oxygenation, radiographic severity, higher neutrophils, higher CRP and lower albumin at presentation predicted critical care admission and mortality. Non-white ethnicity predicted critical care admission but not death. Social deprivation was not predictive of outcome. A risk score was developed incorporating twelve characteristics: age>40, male, non-white ethnicity, oxygen saturations<93%, radiological severity score>3, neutrophil count>8.0 x109/L, CRP>40 mg/L, albumin<34 g/L, creatinine>100 µmol/L, diabetes mellitus, hypertension and chronic lung disease. Risk scores of 4 or higher corresponded to a 28-day cumulative incidence of critical care admission or death of 40.7% (95% CI: 37.1 to 44.4), versus 12.4% (95% CI: 8.2 to 16.7) for scores less than 4.
Interpretation
Our study identified predictors of critical care admission and death in people admitted to hospital with COVID-19. These predictors were incorporated into a risk score that will inform clinical care and stratify patients for clinical trials.
Introduction
SARS-CoV-2 is a large enveloped RNA virus that is responsible for the pandemic disease COVID-19. The disease is mild or asymptomatic in many patients, however a minority develop a severe pneumonia and progress to respiratory failure.1 The first large case series from China, where the outbreak originated, reported that 6% of people admitted to hospital required critical care support.2 Data from Europe and the United States indicate proportions needing critical care are higher.3 In the UK, large cities saw early, rapid escalation in numbers. London had a very sharp increase in cases of COVID-19 in early March 2020.4
In patients who progress to respiratory failure, the disease follows a characteristic pattern.5 The illness starts with fever, cough and myalgia, persisting for a week in a stable pattern. Around 7–12 days after symptom onset there is a progressive escalation in breathlessness, with persisting fevers and cough, and the development of multi-system dysfunction. Laboratory evaluation reveals evidence of a hyperinflammatory state with immunological markers suggestive of an exaggerated response to the virus. Understanding which patients are most likely to develop severe disease will enable informed decisions about admission and discharge.
Global efforts to find treatments for COVID-19 are exploring anti-viral and immune modulatory strategies. Evaluating immune modulators in the context of active infection carries the potential for both harm and benefit. Experience from the SARS outbreak (2003–2004) indicated evidence of harm from using corticosteroids, with increased rates of secondary bacterial infection.6 In 2020, there are many targeted immune modulators available, inhibiting cytokine pathways, complement activation and cellular immune components. The use of targeted immune modulation is appealing, but concerns remain that these strategies may cause more harm than benefit if used widely. In autoimmune diseases, there is a wealth of data around the safety of immune modulators. Evidence suggests targeted therapies confer a small but significant increased risk of infection, with some agents appearing more hazardous than others (e.g. in the UK biologics cohort, anti-IL6 users had higher infection rates than other biologics).7 The greatest risk of infection with immune modulators is apparent early after starting therapy, making concerns around use of these agents in COVID-19 relevant.
In design of clinical trials of immune modulators for COVID-19, it is essential that equipoise exists when selecting therapeutic strategies. Patients with COVID-19 must be selected based upon risk of developing severe complications, to justify the potential hazards of immune modulation. Multiple studies have reported on clinical characteristics that predict severe COVID-19, including older age, male sex and specific comorbidities (e.g. hypertension and diabetes mellitus).8 It has been suggested both in the UK and the US that a disproportionate number of Black, Asian and Ethnic Minority (BAME) patients are admitted with severe COVID-19, but the relationship between ethnicity and disease severity remains unclear.9
This study describes the demographic and clinical risk factors for critical care admission or death in a sample of patients admitted with COVID-19. The aim is to take initial steps in developing a tool that identifies patients at risk of deterioration. This tool is intended for two purposes: 1) embedding in the electronic health record (EHR) as a clinical decision support tool; 2) patient selection for immune modulator clinical trials in COVID-19.
Methods
Study oversight
The primary dataset was extracted as part of a service evaluation to assist local care planning. A fully anonymised dataset, compliant with Information Commissioner's Office requirements for de-identification, was created.10 The project operated under London South East Research Ethics Committee (reference 18/LO/2048) approval granted to the King's Electronic Records Research Interface (KERRI); specific work on COVID-19 research was reviewed with expert patient input on a virtual committee with Caldicott Guardian oversight.
Data sources
The study represents an observational cohort of two hospitals (King's College Hospital and Princess Royal University Hospital), using data captured through routine care in a single EHR instance (Sunrise Clinical Manager, Allscripts). The admissions came, predominantly, from a South London catchment population of approximately 1.2 million. EHR data were extracted for all patients with a positive reverse transcription polymerase chain reaction (RT-PCR) oronasopharyngeal swab for SARS-CoV-2 between 1st March and 17th April 2020. Patients with clinically suspected COVID-19 but without a positive swab were excluded.
Self-identified ethnicity was coded as White, Black (including Black African and Black Caribbean), Asian, or other (including Mixed). Patient postal code was linked to the 2019 Index of Multiple Deprivation (IMD), an area level composite score of socioeconomic position. IMD is calculated from local data on income, employment, education, crime and living environment.11 The following comorbidities were manually validated through review of medical records: active malignancy, chronic kidney disease, diabetes mellitus, hypertension, ischaemic heart disease, chronic lung disease (predominantly asthma, COPD and interstitial lung disease).
Chest radiographs were assessed using an adapted radiographic assessment of lung oedema (RALE) score for COVID-19, as introduced by Wong et al.12 The severity score attributes a number between 0–4 to each lung depending on extent of consolidation or ground glass opacities (0 = no involvement, 1 = <25%, 2 = 25–49%, 3 = 50–75%, 4 = >75% involvement). Values for each lung were summed to produce a final score ranging from 0–8. Correlation between lungs was high (r = 0.65; κ=0.44). The first 200 radiographs were assessed by two independent clinicians. Inter-rater concordance demonstrated high agreement (90.5%). Single reading was undertaken for remaining radiographs.
Study endpoints
The primary outcome for analyses was either transfer to a critical care unit bed or death, assessed in days from admission. In the time frame of analysis, almost all patients admitted to critical care were mechanically ventilated, as non-invasive ventilation was being infrequently used due to concerns around aerosol-generation and staff risk.
Statistical methods
Time-to-event data were analysed using competing risks regression models, estimated using the approach of Fine and gray.13 Where the event of interest was death during admission, the competing risk was discharge, and where the event of interest was critical care admission, the competing risks were death or discharge. Where no event was recorded, data were censored three days before the date data were extracted from the EHR (17th April 2020). Associations between demographic and clinical variables assessed at admission were adjusted for age and gender. Potential non-linear associations between continuous variables and events were explored using restricted cubic splines and accounted for in models using polynomial terms where indicated.
The samples were randomly split into equally sized training and testing sets, stratified by hospital to ensure equal balance. Risk models were fitted in the training sample using standard binary logistic regression and penalised models using ridge and least absolute shrinkage and selection operator (LASSO) regularisation techniques. Risk scores were estimated using these models in the testing and training sample. Performance was assessed using the Brier score, the area under the receiver operator characteristic curve (AUROC), and calibration plots. Variable selection for a simple risk score was based on variables with non-zero coefficients in the LASSO model, where lambda was selected based on the Bayesian Information Criterion. The predictive accuracy of this risk score was compared to the best fitting LASSO model in the training and testing sample.
All analyses were undertaken in Stata 15.1.
Role of funding source
There are no funding sources to be reported. JG had full access to all the data in the study and had final responsibility for the decision to submit for publication. All authors agree to be accountable for all aspects of the work in ensuring questions related to the accuracy or integrity of the work are appropriately investigated and resolved.
Results
In total, 1173 patients with positive SARS-CoV-2 results were admitted during the time window, with data available for 1157 patients. Demographic and clinical information are described in Table 1 . Median age was 71 years, 57.6% were male. Overall, 51.8% of patients were white. Ethnicity varied across the two hospitals, reflective of local demographics. At admission, 45.1% required supplemental oxygen. The most prevalent comorbidities were hypertension (52.9%) and diabetes mellitus (35.3%). Radiographic severity scores ranged from 0–8, with a median of 2 (interquartile range 1 to 4, with right skew).
Table 1.
Demographic and clinical characteristics on admission to hospital
Numbers are median (interquartile range) or n (%). Oxygen saturation based upon bedside pulse oximetry. .
| Characteristic | n missing | Hospital A (N = 674) | Hospital B (N = 483) | Total (N = 1157) |
|---|---|---|---|---|
| Age | 0 | 67 (55,80) | 74 (59,85) | 71 (57,82) |
| Male | 0 | 384 (57.0%) | 282 (58.4%) | 666 (57.6%) |
| Ethnicity | 145 | |||
| White | 186 (30.4%) | 338 (84.5%) | 524 (51.8%) | |
| Black or Black British | 303 (49.5%) | 27 (6.8%) | 330 (32.6%) | |
| Asian or Asian British | 37 (6.1%) | 19 (4.8%) | 56 (5.5%) | |
| Other or mixed | 86 (14.1%) | 16 (4.0%) | 247 (10.1%) | |
| Socially deprived area | 50 | 354 (56.0%) | 85 (17.9%) | 439 (39.7%) |
| O2 required | 0 | 300 (44.5%) | 222 (46.0%) | 522 (45.1%) |
| Respiratory rate | 51 | 20 (18,23) | 19 (18,20) | 20 (18,22) |
| Radiological score | 16 | 3 (2,4) | 2 (1,4) | 2 (1,4) |
| Lymphocytes cells x109/L | 0 | 1.0 (0.7,1.4) | 0.9 (0.6,1.3) | 1.0 (0.7,1.4) |
| Neutrophils cells x109/L | 1 | 5.4 (3.9,7.8) | 5.7 (3.8,8.1) | 5.5 (3.9,7.9) |
| CRP mg/L | 19 | 88.7 (42.5,151.4) | 70.5 (31.0,144.0) | 80.0 (37.0,149.0) |
| Albumin g/L | 10 | 37 (34,40) | 37 (34,40) | 37 (34,40) |
| Creatinine µmol/L | 5 | 96 (73,142) | 90 (70,124) | 93 (72,133) |
| Active malignancy | 1 | 53 (7.9%) | 65 (13.5%) | 118 (10.2%) |
| Chronic kidney disease | 1 | 116 (17.2%) | 73 (15.1%) | 189 (16.3%) |
| Diabetes mellitus | 1 | 281 (41.7%) | 127 (26.3%) | 408 (35.3%) |
| hypertension | 1 | 390 (57.9%) | 221 (45.9%) | 611 (52.9%) |
| ischaemic heart disease | 2 | 86 (12.8%) | 66 (13.7%) | 152 (13.2%) |
| Chronic lung disease | 1 | 131 (19.4%) | 103 (21.4%) | 234 (20.2%) |
During 9955 person-days of follow up 244 people died, 157 were admitted to critical care, and 617 were discharged. At the censoring date, 296 people remained in hospital. Table 2 displays cumulative incidence of outcomes at specified days. Cumulative incidence for critical care admission by 7 days was 9.8% (95%CI: 8.15, 11.5%). Cumulative incidence of death at 14 and 28 days was 20.3% (95%CI: 17.9%, 22.6%) and 26.1% (95%CI: 23.1%, 29.1%), respectively. Cumulative incidence of discharge at 14 and 28 days was 51.3% (95%CI: 48.4%, 54.2%) and 63.9% (95%CI: 60.5%, 67.3%), respectively. Rates of death and discharge were similar across the two hospitals but were lower for critical care admission in Hospital B (Supplementary Figures S1 and S2).
Table 2.
Cumulative incidence of death, critical care admission and discharge.
| Days since admission | Death |
Critical care |
Discharge |
|||
|---|---|---|---|---|---|---|
| Estimate | 95% CI | Estimate | 95% CI | Estimate | 95% CI | |
| 2 | 3.3% | (2.5%,4.1%) | 5.9% | (4.8%,11.0%) | 7.7% | (6.5%,8.8%) |
| 7 | 13.6% | (11.7%,15.5%) | 9.8% | (8.1%,11.5%) | 33.1% | (30.5%,35.6%) |
| 14 | 20.3% | (17.9%,22.6%) | 11.5% | (9.7%,13.3%) | 51.3% | (48.4%,54.2%) |
| 21 | 23.9% | (21.3%,26.6%) | 12.4% | (10.4%,14.3%) | 59.5% | (56.4%,62.7%) |
| 28 | 26.1% | (23.1%,29.1%) | 12.8% | (10.7%,14.8%) | 63.9% | (60.5%,67.3%) |
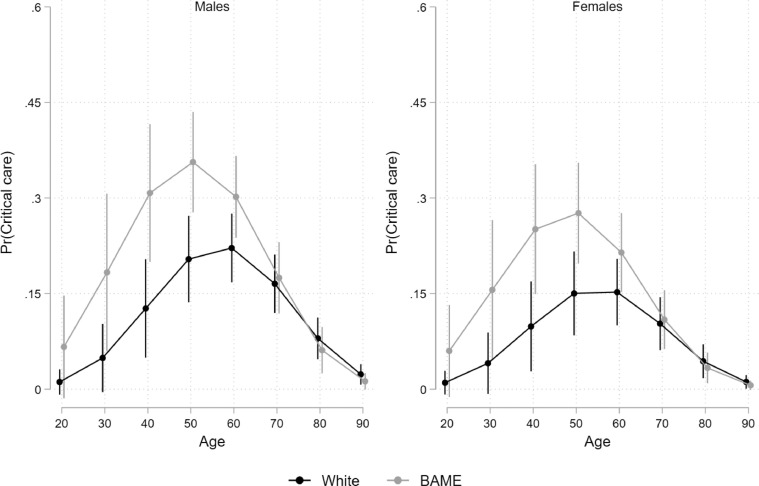
Demographic and clinical predictors for critical care admission and/or death are detailed in Table 3 . Demographic predictors included older age and male sex, in addition to non-white ethnicity for critical care admission only. Young non-white patients were more likely to be admitted to critical care. Risk of death increased with age and was higher in males compared to females. There was a clear age effect with incidence of critical care admission peaking between 50 and 60 years of age and being highest for ethnic minority males (Fig. 1 and Supplementary Figures S3–S5). Mortality differences were not significant between ethnic groups. Deprivation was not an independent predictor of either outcome.
Table 3.
Predictors of death and critical care admission. Estimates are adjusted for age and sex.
| Characteristic | Death |
Critical care |
Death or critical care |
||||||
|---|---|---|---|---|---|---|---|---|---|
| HR | p | 95%CI | HR | p | 95%CI | HR | p | 95%CI | |
| Age* | 2.51 | 0.000 | (1.62,3.91) | 1.51 | 0.006 | (1.12,2.01) | 1.53 | 0.281 | (0.70,3.30) |
| Age2 | 0.94 | 0.041 | (0.88,1.00) | 0.83 | 0.000 | (0.78,0.88) | 0.99 | 0.369 | (0.95,1.02) |
| Male | 1.59 | 0.000 | (1.23,2.06) | 1.41 | 0.043 | (1.01,1.96) | 1.51 | 0.000 | (1.22,1.86) |
| BAME | 1.19 | 0.241 | (0.89,1.58) | 1.53 | 0.007 | (1.12,2.09) | 1.20 | 0.093 | (0.97,1.50) |
| IMD | 1.06 | 0.658 | (0.81,1.39) | 1.07 | 0.672 | (0.78,1.46) | 1.05 | 0.666 | (0.85,1.30) |
| O2 required | 2.03 | 0.000 | (1.57,2.61) | 1.20 | 0.246 | (0.88,1.65) | 1.78 | 0.000 | (1.45,2.19) |
| O2 saturation | 0.88 | 0.000 | (0.84,0.92) | 0.93 | 0.014 | (0.88,0.99) | 0.89 | 0.000 | (0.87,0.92) |
| Respiratory rate | 1.02 | 0.039 | (1.00,1.05) | 1.06 | 0.000 | (1.04,1.07) | 1.03 | 0.004 | (1.01,1.06) |
| Radiological score | 1.23 | 0.000 | (1.16,1.31) | 1.49 | 0.000 | (1.39,1.59) | 1.35 | 0.000 | (1.29,1.42) |
| Lymphocytes* x109/L | 0.46 | 0.010 | (0.26,0.84) | 0.59 | 0.113 | (0.30,1.13) | 0.52 | 0.004 | (0.33,0.82) |
| Lymphocytes2 | 1.28 | 0.001 | (1.11,1.48) | 1.17 | 0.072 | (0.99,1.39) | 1.23 | 0.000 | (1.11,1.37) |
| Neutrophils x109/L | 1.06 | 0.000 | (1.02,1.09) | 1.09 | 0.000 | (1.05,1.13) | 1.08 | 0.000 | (1.06,1.11) |
| CRP* mg/L | 1.06 | 0.000 | (1.02,1.09) | 1.05 | 0.000 | (1.03,1.06) | 1.05 | 0.000 | (1.04,1.06) |
| Albumin g/L | 0.93 | 0.000 | (0.91,0.96) | 0.92 | 0.000 | (0.89,0.96) | 0.93 | 0.000 | (0.91,0.95) |
| Creatinine* µmol/L | 1.01 | 0.000 | (1.00,1.02) | 1.01 | 0.072 | (1.00,1.01) | 1.01 | 0.000 | (1.01,1.01) |
| Active Malignancy | 1.15 | 0.444 | (0.81,1.63) | 0.81 | 0.532 | (0.42,1.56) | 1.05 | 0.772 | (0.77,1.42) |
| Chronic kidney disease | 1.34 | 0.052 | (1.00,1.79) | 0.90 | 0.697 | (0.54,1.51) | 1.27 | 0.063 | (0.99,1.63) |
| Diabetes mellitus | 1.24 | 0.109 | (0.95,1.60) | 1.42 | 0.029 | (1.04,1.95) | 1.20 | 0.092 | (0.97,1.48) |
| Hypertension | 1.28 | 0.062 | (0.99,1.67) | 1.26 | 0.186 | (0.90,1.76) | 1.53 | 0.000 | (1.24,1.90) |
| Ischaemic heart disease | 1.26 | 0.137 | (0.93,1.72) | 0.88 | 0.649 | (0.51,1.52) | 1.13 | 0.359 | (0.87,1.49) |
| Chronic lung disease | 1.37 | 0.032 | (1.03,1.84) | 1.26 | 0.232 | (0.86,1.85) | 1.32 | 0.021 | (1.04,1.67) |
*Age, CRP and creatinine hazard ratios are for 10-point increases. **Age and lymphocyte count have a non-linear association with outcome and are also entered with polynomial terms (Age2, Lymphocytes2. Oxygen saturation based upon bedside pulse oximetry. BAME = Black, Asian and minority ethnic; IMD = index of multiple deprivation.
Fig. 1.
Association between age, gender, ethnicity and risk of critical care admission
Pr(Critical care) = probability of admission to critical care. BAME = Black, Asian and minority ethnic.
Clinical predictors for critical care admission and/or death included respiratory rate, pulse oximetry saturations and oxygen requirement. Predictive laboratory markers included higher neutrophil counts, higher CRP, lower albumin and renal impairment. Lymphocyte counts showed a non-linear association, with both low and high lymphocytes associating with critical care admission and death (Supplementary Figure S6). Radiographic severity was predictive of outcomes. For each unit increase on the 8-point ordinal scale, the hazard increased by 1.35 (95% CI 1.29, 1.42). Background comorbidities (diabetes mellitus, hypertension and chronic lung disease) were all related to death or critical care admission, with effect sizes of similar magnitude across all comorbidities.
Risk models were estimated using a training sample including half the patients from each hospital in a standard logistic regression model, a model with LASSO regularisation, and a model with ridge regularisation. Model performance in the test sample were approximately the same across each of the methods (Supplementary Figure S7). A risk score was formed as a count of variables with non-zero coefficients from the LASSO model; this performed almost as well and demonstrated acceptable discrimination of risk for death and critical care admission (Supplementary Table S1).
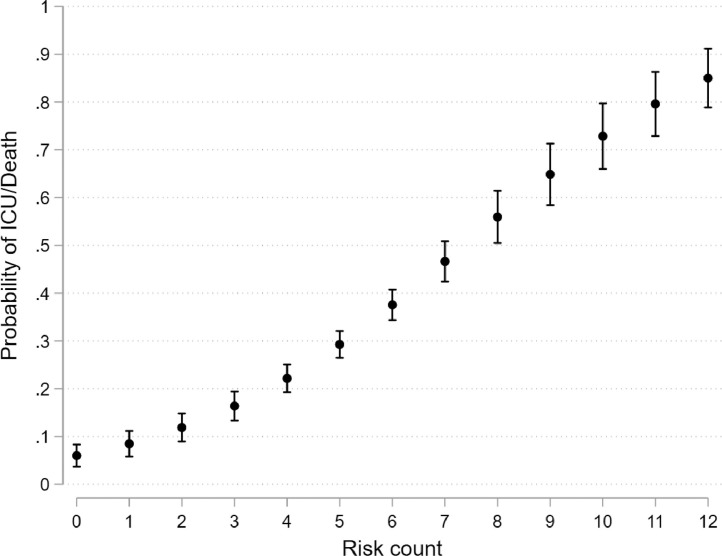
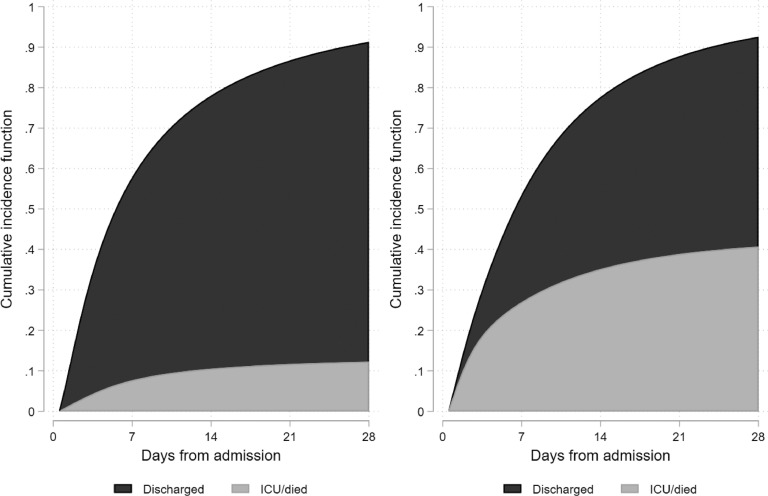
The risk score included the following twelve parameters: age greater than 40, male gender, non-white ethnicity, oxygen saturation less than 93%, radiological severity score greater than 3, neutrophil count greater than 8.0 x109/L, CRP greater than 40 mg/L, albumin less than 34 g/L, creatinine greater than 100 µmol/L, diabetes mellitus, hypertension and chronic lung disease. The probability of death or critical care admission by increasing risk count is shown in Fig. 2 . Risk scores of 4 or higher corresponded to a 28-day cumulative incidence of critical care admission or death of 40.7% (95% CI: 37.1 to 44.4), versus 12.4% (95% CI: 8.2 to 16.7) for scores less than 4 (Fig. 3 ). A weighted count using the LASSO coefficients performed marginally better at discriminating between those at very high risk (Supplementary Figure S8 and Supplementary Table S2).
Fig. 2.
Estimated probabilities of death or critical care admission by increasing risk count
ICU = critical care admission.
Fig. 3.
Cumulative incidence of critical care admission or death versus discharge from hospital in patients with unweighted risk scores of less than 4 (Left) and 4 or higher (Right)
ICU = critical care admission.
Discussion
This study utilises a large observational cohort of 1157 COVID-19 patients, admitted to two London hospitals, to develop a risk score that identifies patients most likely to die or require critical care admission. This tool will assist clinicians in identifying patients at highest risk of severe disease and those most appropriate to enrol into therapeutic studies.
A systematic review identified ten models reporting predictors for COVID-19 disease severity, all from Chinese populations and deemed at high risk of bias.8 Reported predictors of severe disease included older age, male sex, elevated CRP, abnormal lymphocyte count and radiographic (computed tomography) features, in line with our study. Our study has several advantages: (1) large sample size; (2) heterogenous population, including a significant proportion of BAME patients, facilitating identification of novel demographic predictors of severe disease; (3) utilisation of an ordinal scoring system for chest radiographs; (4) development of an internally validated risk score to stratify patients into higher and lower risk prognostic groups.
A useful cut-point on the risk score to inform clinical care and enable stratification of patients for clinical trials seems to be around 4. Risk scores of 4 or higher corresponded to a 28-day cumulative incidence of critical care admission or death of 40.7% (95% CI: 37.1 to 44.4), versus 12.4% (95% CI: 8.2 to 16.7) in those scoring less than 4. It is important to note that this score is unlikely to generalise to the community population and should only be used in the assessment of patients admitted to hospital.
Our study characterised important predictors of critical admission and death. Several characteristics demonstrated non-linear relationships with the outcomes, notably age and lymphocyte count. Age was included in the risk score as binary predictor (above/below 40 years), while lymphocyte count was excluded from the risk score. The observation that both high and low lymphocyte counts correlate with poorer outcomes may be in part explained by changes in lymphocyte count during disease progression, which is not accounted for in the analyses.
Chest radiograph severity was one of the most striking additional predictors and a key finding. The extent of radiographic infiltrate was predictive of critical care admission and death. Chest radiography was used over other imaging modalities because of accessibility and simplicity to interpret. This simple modality provides broad utility in a variety of healthcare settings.
Non-white ethnicity was predictive of critical care admission, especially amongst younger patients, but did not predict death. The association between ethnicity and disease severity is likely to be complex and could be affected by multiple factors including socioeconomic and demographic factors, comorbidity profiles or genetic traits. Our study was not designed to investigate this association in detail. We found no obvious association between area level social deprivation and disease severity. A key driver underlying the link between deprivation and health outcomes is thought to be patient candidacy, which refers to patient and clinician-perceived eligibility for healthcare services. Those with higher deprivation typically have lower candidacy, acting as a barrier to healthcare engagement.14 However, we did not have individual patient-level data to explore this aspect. An increased prevalence of comorbidities such as diabetes mellitus and hypertension in BAME individuals could also be a contributing factor.15
Our study has important limitations. We limited our cohort to confirmed SARS-CoV-2 patients who were not admitted, potentially excluding up to 20% of COVID-19 cases.16 We did not directly confirm if critical care patients were mechanically ventilated or on continuous positive airway pressure support, nor were we able to provide information on other organ support (renal replacement therapy or extracorporeal membrane oxygenation). UK data suggest two thirds of COVID-19 patients in critical care are mechanically ventilated, with the rest on non-invasive ventilation.17 Many patients were still in hospital at the time of analysis and their final outcomes are not yet known.
Our non-white patient population was predominantly black ethnicity, limiting robust generalisations across other non-white ethnic groups. In addition, we have not linked to community records. UK national policy is for all patients to be managed at home in self-isolation for the first seven days after symptom onset;18 this likely reduced the number of patients presenting with early non-severe disease, and will have imposed a sampling bias that may limit generalisability to other countries. Finally, the risk score needs to be externally validated in other datasets. As more data become available, it will be possible to refine and strengthen the model. The mechanistic underpinnings of the associations that we observed will require additional studies.
Conclusion
Our study identifies demographic and clinical predictors of critical care admission or death in people with COVID-19 who present to emergency departments and incorporates them into a clinically usable risk score. Plain chest radiograph severity was also highly predictive of poor outcomes. Non-white ethnicity predicted an increased risk of critical admission, especially in males under 60 years. The risk score will help inform clinical care and enable stratification of patients for clinical trials.
Research in context
Evidence before the study
Prior to our analyses, we searched PubMed and Google Scholar for manuscripts describing risk prediction models for COVID-19. On April 7th, 2020, the BMJ published a systematic review of predictors of COVID-19 and prognostic models. Our own search and the BMJ publication confirmed significant limitations of published literature in terms of bias, population heterogeneity, and sample size. Reported demographic predictors for developing severe COVID-19 included older age and male sex. Clinical predictors of severe COVID-19 included abnormal neutrophil and lymphocyte counts, elevated CRP, and computed tomography findings. Ethnicity differences in outcomes were widely reported in the media.
Added value of this study
We analysed patient characteristics for 1157 people admitted to hospital with COVID-19 to identify predictors of outcome. Plain chest radiography severity scores were highly predictive of death and critical care admission. Non-white ethnicity predicted critical care admission. The large sample size permitted development and validation of a risk prediction score using survival modelling. Risk scores of 4/12 or higher corresponded to a 28-day cumulative incidence of critical care admission or death of 40.7% (95% CI: 37.1 to 44.4), versus 12.4% (95% CI: 8.2 to 16.7) for scores less than 4.
Implications of all the available evidence
The findings will help inform admission and discharge decisions, providing frontline clinicians with a tool to identify patients most at risk of deterioration. Our findings have potential to identify patients for clinical trials of immune modulators, where the risk-benefit in all COVID-19 patients is less clear.
Funding
None
Contributions
JG, SN, RB, AB, IC, BC, RJ, CR, MR, RS, LS, SW, MY, JT, AS, FC contributed to the design of the work and acquisition of the data. JG, SN, RB, AB, IC, BC, RJ, CR, MR, RS, LS, SW, MY, JT, AS, FC drafted the work and were involved in revising the manuscript in preparation for submission, in addition to final approval of the version to be published.
Data sharing statement
The authors commit to making the relevant anonymised patient-level data available on reasonable request following publication via the King's College London Research Data Management system.
Declaration of Competing Interest
All authors have completed an ICMJE conflict of interest form and report no conflicts of interest.
Acknowledgements
We are grateful for the work of the King's College Hospital Information and Communication Technology Department. AMS is supported by the British Heart Foundation (CH/1999001/11735) and the National Institute for Health Research (NIHR) Biomedical Research Centre at Guy's & St Thomas’ NHS Foundation Trust and King's College London with King's College Hospital NHS Foundation Trust (IS-BRC-1215-20006). We would like to thank all the clinicians managing the patients.
Footnotes
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.jinf.2020.05.064.
Appendix. Supplementary materials
References
- 1.Li H., Liu S.-.M., Yu X.-.H., Tang S.-.L., Tang C.-.K. Coronavirus disease 2019 (COVID-19): current status and future perspectives. Int J Antimicrob Agents. 2020 Mar 29 doi: 10.1016/j.ijantimicag.2020.105951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.W-j Guan, Z-y Ni, Hu Y. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020 Feb 28 doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kinross P., Suetens C., Gomes Dias J. Rapidly increasing cumulative incidence of coronavirus disease (COVID-19) in the European Union/European Economic Area and the United Kingdom, 1 January to 15 March 2020. Euro Surveill. Mar 2020;25(11) doi: 10.2807/1560-7917.ES.2020.25.11.2000285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Coronavirus.data.gov.uk [Internet]. Coronavirus (COVID-19) in the UK. 2020. [Cited 2020 Apr 20]. Available from:https://coronavirus.data.gov.uk/#regions.
- 5.Holshue M.L., DeBolt C., Lindquist S. First case of 2019 novel coronavirus in the United States. N Engl J Med. 2020;382(10):929–936. doi: 10.1056/NEJMoa2001191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Auyeung T.W., Lee J.S., Lai W.K. The use of corticosteroid as treatment in SARS was associated with adverse outcomes: a retrospective cohort study. J Infect. 2005;51(2):98–102. doi: 10.1016/j.jinf.2004.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rutherford A.I., Subesinghe S., Hyrich K.L., Galloway J.B. Serious infection across biologic-treated patients with rheumatoid arthritis: results from the British society for rheumatology biologics register for rheumatoid arthritis. Ann Rheum Dis. 2018;77(6):905–910. doi: 10.1136/annrheumdis-2017-212825. [DOI] [PubMed] [Google Scholar]
- 8.Wynants L., Van Calster B., Bonten M.M.J. Prediction models for diagnosis and prognosis of covid-19 infection: systematic review and critical appraisal. BMJ. 2020 Apr 7;369:m1328. doi: 10.1136/bmj.m1328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pareek M., Bangash M.N., Pareek N. Ethnicity and COVID-19: an urgent public health research priority. Lancet. 2020 Apr 21 doi: 10.1016/S0140-6736(20)30922-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ico.org.uk [Internet]. Anonymisation: managing data protection risk code of practice. 2012. [Cited 2020 Apr 20]. Available from:https://ico.org.uk/media/for-organisations/documents/1061/anonymisation-code.pdf.
- 11.Geoportal.statistics.gov.uk [Internet]. Index of Multiple Deprivation (December 2019) Lookup in England. 2019. [Cited 2020 Apr 20]. Available from:https://geoportal.statistics.gov.uk/datasets/index-of-multiple-deprivation-december-2019-lookup-in-england.
- 12.Wong H.Y.F., Lam H.Y.S., Fong A.H.-T. Frequency and distribution of chest radiographic findings in COVID-19 positive patients. Radiology. 2019 Mar 27 doi: 10.1148/radiol.2020201160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fine J.P., Gray R.J. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–509. [Google Scholar]
- 14.Dixon-Woods M., Cavers D., Agarwal S. Conducting a critical interpretive synthesis of the literature on access to healthcare by vulnerable groups. BMC Med Res Methodol. 2006 Jul 26;6:35. doi: 10.1186/1471-2288-6-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Oldroyd J., Banerjee M., Heald A., Cruickshank K. Diabetes and ethnic minorities. Postgrad Med J. 2005;81:486–490. doi: 10.1136/pgmj.2004.029124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Carver C., Jones N.Comparative accuracy of oropharyngeal and nasopharyngeal swabs for diagnosis of COVID-19. Oxford COVID-19 evidence service team centre for evidence based medicine. 2020. [Cited 2020 Apr 20]. Available from:https://www.cebm.net/covid-19/comparative-accuracy-of-oropharyngeal-and-nasopharyngeal-swabs-for-diagnosis-of-covid-19/
- 17.Icnarc.org [Internet]. ICNARC report on COVID-19 in critical care 17 April 2020. 2020. [Cited 2020 Apr 20]. Available from:https://www.icnarc.org/DataServices/Attachments/Download/76a7364b-4b76-ea11-9124-00505601089b.
- 18.Gov.uk [Internet]Stay at home: guidance for households with possible coronavirus (COVID-19) infection. 2020. [Cited 2020 Apr 20]. Available from:https://www.gov.uk/government/publications/covid-19-stay-at-home-guidance/stay-at-home-guidance-for-households-with-possible-coronavirus-covid-19-infection.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.