Purpose of review
Tuberculous meningitis (TBM) is associated with significant mortality and morbidity yet is difficult to diagnose and treat. We reviewed original research published in the last 2 years, since 1 January 2018, which we considered to have a major impact in advancing diagnosis, treatment and understanding of the pathophysiology of TBM meningitis in children and adults.
Recent findings
Studies have sought to identify a high sensitivity diagnostic test for TBM, with new data on modified Ziehl--Neelsen staining, urinary and cerebrospinal fluid (CSF) lipoarabinomannan and GeneXpert Ultra. Recent studies on CSF biomarkers provide a better understanding of the detrimental inflammatory cascade and neuromarkers of brain damage and suggest potential for novel host-directed therapy. Tryptophan metabolism appears to affect outcome and requires further study. Increased clinical trials activity in TBM focuses on optimizing antituberculosis drug regimens and adjuvant therapy; however, there are few planned paediatric trials.
Summary
Tuberculous meningitis still kills or disables around half of sufferers. Although some progress has been made, there remains a need for more sensitive diagnostic tests, better drug therapy, improved management of complications and understanding of host-directed therapy if outcomes are to improve.
Keywords: biomarkers, clinical trials, diagnosis, management, neurocritical, tuberculous meningitis
INTRODUCTION
In 2018, tuberculosis (TB) affected 10 million people worldwide and killed 1.5 million [1]. The most fatal form of TB, tuberculous meningitis (TBM), occurs in 1–5% of those with TB. The highest risk populations are children under 5 years of age, the HIV co-infected, and immunocompromised. The disease burden in children may be increasing: a recent retrospective study from a high TB burden setting showing an alarming increase of TBM hospital admissions following global shortages of BCG (Bacille Calmette--Guerin) vaccine [2].
Despite advances in anti-TB chemotherapy, mortality from TBM remains unacceptably high (adults 50%, children 20%) [3,4]. Mortality in children is lower than in adults, but their developing brains render them at risk of unique age-specific neurological sequelae. Early diagnosis and timely initiation of appropriate therapy predict good outcome. But questions remain: ‘how can we diagnose TBM?’ and ‘what is appropriate therapy?’ Judicious neurocritical care of raised intracranial pressure (ICP) and blood pressure protect the brain from further damage, yet evidence guiding management is lacking. There have been recent efforts to improve understanding of pathophysiology with novel studies on biomarker signatures in TBM [5,6].
We searched PubMed for the term ‘tuberculous meningitis’ and found 328 publications between 1 January 2018 and the 7 December 2019. Here we provide an update on the diagnostics, pathophysiology, prognostics and management of TBM in adults and children. We highlight the current gaps in knowledge and the future trajectory of TBM clinical research.
Box 1.

no caption available
DIAGNOSTICS
Low bacterial numbers in cerebrospinal fluid (CSF) lead to challenges in Mycobacterium tuberculosis (Mtb) detection and diagnostic confirmation of TBM, particularly in children in whom large CSF volumes can be difficult to obtain. Current tests remain insufficiently sensitive to rule out TBM when they are negative. Comparing diagnostic test performance is confounded by the lack of a single gold standard reference test for TBM. As a result, use of the uniform case definition for clinical research [7] for test comparison is now commonplace.
New TBM diagnostics frequently seek to improve upon older tests. Ziehl--Neelsen smear microscopy of CSF for acid-fast bacilli is cheap and widely available, yet often insensitive unless performed by experienced microscopists using large volumes of CSF centrifuged at high speeds to concentrate Mtb. In Vietnam, South Africa and Indonesia, 618 individuals were enrolled into a prospective comparison of conventional Ziehl--Neelsen smear, modified Ziehl--Neelsen smear (using cytospin and permeabilization), GeneXpert MTB/RIF (Xpert) and mycobacterial culture [8▪]. Against a reference standard of definite, probable and possible TBM [7], sensitivities of conventional Ziehl--Neelsen, modified Ziehl--Neelsen, Xpert and mycobacterial culture were 33.9, 34.5, 25.1 and 31.8%, respectively [8▪]. Ziehl--Neelsen smear modifications did not improve diagnostic sensitivity.
The lipoarabinomannan (LAM) antigen is found in the cell wall of Mtb, and its detection represents an alternative to conventional TBM diagnostics. A prospective study of 550 adults (86% HIV co-infected) with suspected TBM in Zambia compared the diagnostic accuracies of CSF LAM and urinary LAM [Alere Determine TB LAM Ag assay (AlereLAM), Abbott, Chicago, Illinois, USA], against a reference standard of positive CSF mycobacterial culture [9▪]. Diagnostic sensitivities of CSF and urinary LAM were 21.9 and 24.1% respectively, suggesting, at least in this setting that these tests lack the sensitivity required for TBM diagnosis. In a subsequent study of 59 HIV co-infected individuals with suspected TBM in Uganda, lumbar CSF TB AlereLAM was compared against reference standards of positive GeneXpert MTB/RIF Ultra (Ultra), and definite plus probable TBM [10]. Whilst highly specific against these standards (96 and 95%, respectively), sensitivities of TB LAM were poor at 33 and 24%, respectively. However, a novel urine-based LAM assay with high-affinity Mtb specific antibodies and a silver-amplification step [Fujifilm SILVAMP, TB LAM (FujiLAM), Fujifilm, Tokyo, Japan] has a detection limit 30 times lower than AlereLAM [11]. In hospitalized adults with HIV co-infection, the FujiLAM had 35% higher sensitivity for TB diagnosis, and comparable specificity, to the AlereLAM [12▪]. Importantly, FujiLAM has yet to be assessed for the diagnosis of suspected TBM, HIV-uninfected or paediatric populations.
Xpert, recently superseded by Xpert Ultra [13], represents perhaps the most valuable diagnostic test for TBM currently available. Xpert Ultra are rapid PCR-based tests, crucially identifying rifampicin resistance when positive, guiding early therapy. Xpert Ultra is now a well established part of TBM testing. Ultra contains a larger reaction chamber than Xpert Ultra, with two additional different multicopy amplification targets [14]. Ultra has diagnostic superiority over Xpert in pulmonary TB, especially in samples expected to be paucibacillary [15]. Rapid endorsement by WHO [13] of Xpert Ultra came in 2017 following studies demonstrating its improved sensitivity in TB diagnosis [16▪]. An initial study suggested diagnostic superiority of Ultra over Xpert in 23 HIV co-infected adults with TBM [16▪]. Ultra and Xpert sensitivities for Mtb detection in CSF were 70 and 43%, respectively, against a reference standard of definite and probable TBM. Subsequent small studies have further assessed Ultra and Xpert for extrapulmonary TB diagnosis [17–19]. In 43 HIV uninfected adults with suspected TBM in China, Ultra had a higher sensitivity for Mtb detection in CSF than Xpert [19/43 (44.2%) vs. 8/43 (18.6%), respectively], against a reference standard of definite, probable and possible TBM. However, many confirmed cases in the Ultra group were confirmed by Ultra only. Paediatric data on the performance of Xpert Ultra in TBM is lacking. In a recent paediatric study of 28 children with definite, probable or possible TBM, diagnostic sensitivity of Xpert was 46.2% (6/13) against a total TBM diagnostic score of at least 10 points [20]. Large studies comparing Ultra and Xpert for TBM diagnosis in both adults and children are expected to be published soon.
PATHOPHYSIOLOGY
Corticosteroids are a widely used adjunctive therapy in the treatment of TBM but how they improve outcome remains unknown. Recent insights into possible mechanisms driving the inflammatory response in TBM include the regulated balance of inflammatory eicosanoids and host protective mediators (specialized pro-resolving mediators). A study using a lipid-mediator profiling approach demonstrated a specialized pro-resolving mediator profile in CSF associated with disease severity and mortality in adults with TBM. Furthermore, the prothrombotic mediator thromboxane A2 was reduced in CSF in adults with TBM randomly assigned aspirin compared with those who received placebo [21] (Fig. 1).
FIGURE 1.
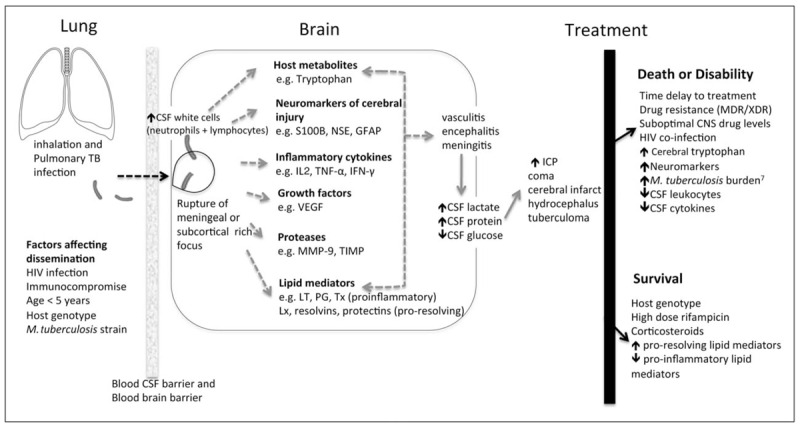
Overview of the pathophysiology of tuberculosis meningitis∗. Mortality of TBM is attributed to Mycobacterium tuberculosis and its interaction with the host immune response. Once M. tuberculosis enters the brain or meninges, a detrimental immunoinflammatory response including inflammatory cytokines, proteases, lipid mediators, neuromarkers and tryptophan metabolites, is triggered [1–6]. This leads to the cerebral disorder and complications known to occur in TBM. Knowledge gaps exist in the mechanism of bacterial invasion into the CNS and the underlying biologic pathways (dotted arrows) leading to the disease process. CSF, cerebrospinal fluid; GFAP, glial fibrillary acidic protein; HIV, human immunodeficiency virus; ICP, intracranial pressure; IFN, interferon; IL, interleukin; LT, leukotriene; Lx, lipoxins; MDR, multidrug-resistant; MMP, matrix metalloproteinases; NSE, neuron-specific enolase; PG, prostaglandin; TB, tuberculosis; TIMP, tissue inhibitor of matrix metalloproteinases; TNF, tumour necrosis factor; Tx, thromboxane; VEGF, vascular endothelial growth factor; XDR, extensively drug-resistant. Adapted from Thwaites and Tran [27]. Figure references [6,21,22▪▪,23–27].
A potential role for tryptophan metabolism in TBM outcome has recently been demonstrated. In an observational cohort study of the metabolomes of 33 HIV uninfected Indonesian adults with TBM, CSF tryptophan concentrations were lower in individuals who survived, compared with those who died (by nine times), and compared with controls (by 31 times) [22▪▪]. More than 10 genetic loci were identified as being predictive of CSF tryptophan concentrations in the TBM cohort suggesting the influence of host genotype on outcome.
Biomarker studies of cerebral injury in paediatric TBM have helped gain further insight. In a study of 44 children with TBM-associated hydrocephalus, 11 healthy controls and 9 children with pulmonary TB, neuromarkers of brain damage; S100B, neuron-specific enolase and glial fibrillary acidic protein, were elevated for at least 3 weeks in the TBM group [6]. Neuromarker concentrations increased over time and predicted poor outcome. Furthermore, high concentrations of neuromarkers and inflammatory markers were uniquely compartmentalized to the site of infection (ventricular CSF) but not identified in lumbar CSF or serum. A subsequent transcriptomic study demonstrated this distinct biomarker signature to be unique to TBM when compared with non-TBM controls and was associated with neuronal excitotoxicity [28▪▪]. For the first time, this offers insight into the possible mechanism of cerebral injury in TBM and potential impact on critical neurodevelopment of the immature brain in young children.
PROGNOSIS
Prognostic models have been developed, which may aid clinicians in identifying patients at the greatest risk of death [29]. However, disease complications may change the likelihood of a poor outcome but are not captured by models, which use only baseline data. Using data from 1048 adults with TBM from three randomized controlled trials and one prospective observational study at two Vietnamese hospitals from 2004 to 2016, a dynamic prediction model was developed where follow-up measurements of Glasgow coma score (GCS) and plasma sodium were incorporated [30▪]. In HIV-uninfected and HIV co-infected individuals, higher GCS indicated lower mortality with hazard ratios of 0.76 (95% CI 0.71–0.81) and 0.85 (95% CI 0.81–0.91), respectively. In HIV-uninfected individuals, plasma sodium concentrations of 140 mmol/l; higher than normal for TBM, were associated with worse prognosis from baseline until day 10 of treatment than plasma sodium concentrations of 135 mmol/l. However, by day 30 these higher sodium concentrations were associated with improved survival. In HIV co-infected individuals, higher sodium concentrations associated with better prognosis across all prediction time points. No recent modelling studies predict prognosis in children.
TUBERCULOUS MENINGITIS CRITICAL ILLNESS
The evidence base guiding supportive, medical and neurosurgical management of critically ill individuals with TBM is limited, with numerous research gaps recently highlighted [31]. TBM-associated hyponatraemia is common and may contribute to raised ICP. In a recent randomized trial in India, intravenous and oral salt with or without fludrocortisone was used in the treatment of 37 adults with TBM-associated hyponatraemia secondary to cerebral salt wasting. In the fludrocortisone group, correction of hyponatraemia was faster (4 vs. 15 days); however, severe hypokalaemia and hypertension developed in two patients requiring discontinuation of fludrocortisone in these cases [32▪].
In TBM, hydrocephalus, cerebral infarction, paradoxical reactions including tuberculomas, neurological immune reconstitution inflammatory syndrome in HIV co-infected individuals and seizures may all reduce GCS, a tool routinely used for patient assessment. The cause of seizures in TBM is multifactorial; data regarding the cause and timing of seizures in patients with TBM is scarce, and their incidence appears to vary substantially between populations [31]. A recent study of Indian adults with TBM described seizures in 34% (27/79) of patients, and abnormal electroencephalogram changes were observed in 85% (17/20) of patients who had seizures and an electroencephalogram performed [33].
Detection and monitoring of raised ICP, a common endpoint of TBM neurocomplications, is limited by global availability of gold standard invasive monitoring methods. Further evidence to support use of noninvasive ICP monitoring techniques, such as optic nerve sheath ultrasound, is needed. Optimal management of raised ICP is uncertain; previous neurosurgical intervention studies for TBM-associated hydrocephalus have largely focused on children. The permanent relief of raised ICP in TBM-associated obstructive hydrocephalus can be achieved through ventriculoperitoneal shunting or endoscopic third ventriculostomy (ETV), yet no study to date has shown one technique to be consistently superior to the other in TBM. In a recent study of TBM-associated hydrocephalus in India, children were randomized to ETV or ventriculoperitoneal shunting [34]. Success rates were 65% (17/26) and 61% (16/26), respectively. Poor outcomes for both procedures were linked to increased disease severity. A recent study assessed outcomes in individuals (predominantly adults) with TBM-associated hydrocephalus and HIV co-infection, either receiving (n = 15) or not receiving (n = 15), antiretroviral therapy (ART) [35▪]. In the ART and non-ART groups there were 4/15 (27%) and 10/15 (67%) deaths, respectively. Whilst this suggests outcomes after ventriculoperitoneal shunting for TBM-associated hydrocephalus are better once ART is commenced, CD4 counts in the ART group had generally recovered well. Outcomes in patients newly starting ART after HIV diagnosis at the time of TBM presentation are uncertain.
Given the complex and challenging nature of critical illness in TBM, patient assessment proformas and a priorities checklist have recently been developed [36] with the aim of standardizing clinical review and prioritizing likely causes of acute deterioration. However, these tools require validating.
CLINICAL TRIALS
Whilst many questions regarding optimal TBM management remain unanswered, the current TBM research field is dynamic. A search of clinicaltrials.gov and ISRCTN trial registries identified 11 relevant prospective randomized studies or clinical trials in TBM (Table 1). Current trials in TBM can generally be separated into those aiming to optimize Mtb killing, and those using adjunctive therapies to prevent and manage disease complications. The uncertainty surrounding optimal anti-TB chemotherapy regimens and doses in TBM is underlined by the current trial panorama, where optimized anti-TB chemotherapy trials dominate. Given the poor central nervous system penetration of rifampicin, and its importance as a first line therapy, multiple studies of high-dose rifampicin, with or without linezolid, are underway or due to start. High-dose isoniazid therapy in rapid acetylators is also under investigation.
Table 1.
Registered current and future trials in tuberculosis meningitis, listed by start date
| Trial short title | Design | Study population | Intervention arms(s) | Countries recruiting | Sample size | Recruitment period | Primary outcome | Clinical trial registry number |
| TBM-KIDS | Randomised, open-label trial phase II | Age ≥6 months Age < 12 years Weight >6 kg Clinical TBM | Control: standard anti-TB therapy Interventions: High-dose rifampicin (30 mg/kg) containing regimen, OR high dose rifampicin (30 mg/kg) containing regimen plus substitution of ethambutol for levofloxacin (20 mg/kg) | India, Malawi | 120 | February 2017 -- ongoing | PK, functional, neurocognitive and safety endpoints | NCT02958709 |
| ACT HIV | Randomised, double blind, placebo controlled phase III trial | Age ≥18 years Clinical TBM HIV co-infected | Control: standard of care without dexamethasone Intervention: adjunctive dexamethasone tapering regimen | Vietnam, Indonesia | 520 | June 2017 -- ongoing | Overall survival until 12 months | NCT03092817 |
| RiFT | Randomised, open-label, phase II trial | Age ≥18 years Clinical TBM | Control: standard anti-TB therapy Interventions: regimens containing high dose (20 mg/kg) intravenous then oral 35 mg/kg rifampicin OR high dose (35 mg/kg) oral rifampicin | Uganda | 60 | September 2017 -- ongoing | PK parameters, composite safety endpoint | ISRCTN 42218549 |
| LAST ACT | LTA4H genotype stratified, randomised (CC or CT genotypes), double blind, placebo-controlled phase III noninferiority trial | Age ≥18 years Clinical TBM HIV-uninfected | Control: standard of care without dexamethasone Intervention: adjunctive dexamethasone tapering regimen | Vietnam | 720 | February 2018 -- ongoing | All-cause mortality or new neurological event until 12 months | NCT03100786 |
| Optimizing anti-TB therapy in adults with TBM | NAT2 stratified, randomised, parallel group trial | Age 18–65 years Clinical TBM | Control: standard anti-TB therapy in normal acetylators Interventions: rapid acetylators randomised to standard isoniazid containing regimen OR high dose isoniazid containing regimen | China | 676 | March 2019 -- ongoing | Number of participants with death or severe disability at 12 months | NCT03787940 |
| LASER-TBM | Randomised, parallel group, multiarm phase IIa trial | Age ≥18 years Clinical TBM HIV co-infected | Control: standard anti-TB therapy Interventions: high-dose regimen containing 35 mg/kg oral rifampicin, and oral linezolid 1.2 g, OR high-dose regimen plus aspirin 1 g | South Africa | 100 | June 2019 -- ongoing | Treatment related adverse events | NCT03927313 |
| SIMPLE | Randomised, open label phase II trial | Age ≥18 years Clinical TBM | control: anti-TB regimen containing 35 mg/kg rifampicin (high dose) Interventions: High-dose rifampicin regimen with oral linezolid 600 mg, OR high-dose rifampicin regimen with oral linezolid 1.2 g | Indonesia | 36 | June 2019 -- ongoing | Linezolid blood and CSF PK | NCT03537495 |
| HARVEST | Randomised, double-blinded, placebo-controlled, phase III trial | Age ≥18 years Clinical TBM | Control: standard fixed dose anti-TB therapy Intervention: Regimen containing additional four oral rifampicin 300 mg capsules | Indonesia, South Africa, Uganda | 500 | Estimated recruitment start - January 2020 | 6-month survival | ISRCTN 15668391 |
| SURE | Multifactorial randomiization (×2), open label phase III trial | Age >28 days Age <15 years Clinical TBM | Control: standard 12-month anti-TB therapy Interventions: 6-month intensified therapy Oral aspirin 20 mg/kg or placebo | India, Uganda, Vietnam, Zambia, Zimbabwe | 400 | Estimated recruitment start - February 2020 | All-cause mortality at 48 weeks, neuro-development at 48 weeks | ISRCTN40829906 |
| ALTER | Randomised, open-label, phase II trial | Age >18 years Clinical TBM | Four arms: standard dose rifampicin containing regimen with/without oral linezolid 1.2 g, or high-dose (35 mg/kg) rifampicin with/without oral linezolid 1.2 g | Uganda | 60 | Estimated recruitment start -- April 2020 | CSF PK | NCT04021121 |
| INTENSE- TBM | Randomised, placebo controlled (for aspirin), phase III, 2 × 2 factorial superiority trial | Age ≥15 years Clinical TBM | Four arms: standard anti-TB therapy OR high dose (35 mg/kg) oral rifampicin containing therapy and plus oral linezolid 1.2 g added to standard anti-TB therapy, with/without aspirin 200 mg | Ivory Coast Madagascar South Africa Uganda | 768 | Estimated recruitment start -- April 2020 | Rate of all-cause death until 40 weeks | NCT04145258 |
CSF, cerebrospinal fluid; ISRCTN, prefix to unique clinical trials number in the International Standard Randomised Controlled Trial Number registry; LTA4H, leukotriene A4 hydrolase; NAT2, N-acetyltransferase type 2; NCT, prefix to unique clinical trials number on clinicaltrials.gov; PK, pharmacokinetic; TBM, tuberculous meningitis.
An evidence base is forming for adjunctive aspirin therapy in TBM. Aspirin may reduce the incidence and promote the resolution of TBM-associated brain infarcts, as shown in a recent phase 2 trial of adjunctive aspirin therapy in 120 HIV-uninfected adults with TBM in Vietnam [37]. Large phase 3 trials are required. In the adult LASER-TBM trial (NCT03927313), high-dose rifampicin and linezolid will be trialed with or without 1000 mg adjunctive aspirin for 8 weeks. In the SURE trial (ISRCTN40829906), children with TBM will be randomized to 6 or 12-month anti-TB chemotherapy, and will also undergo a second randomization to aspirin 20 mg/kg or placebo. Adjunctive dexamethasone is now commonly used in HIV-uninfected individuals with TBM. Whether all HIV-uninfected individuals with TBM should receive corticosteroids, and whether there is benefit in HIV co-infected individuals, is currently under investigation [38,39]. LAST ACT (NCT03100786) is the first study to personalize anti-inflammatory therapy in TBM by host leukotriene A4 hydrolase genotype.
Most therapeutic advances in TBM have occurred in adult studies, however, drug pharmacokinetics and TBM treatment outcomes are different in children. Children generally metabolize drugs differently; drug clearance being inversely proportional to age [40]. Although children suffer lower mortality than adults, other treatment outcomes, such as neurodevelopmental disabilities are paediatric-specific [40]. In addition to age-appropriate pharmacokinetic data, clinical trials, which characterize neurodevelopmental and cognitive outcomes in paediatric TBM, are needed to evaluate drug efficacy appropriately. Conducting such clinical trials in a real world setting has been challenging with recent data from the TBM-KIDS (NCT02958709) trial showing that only 21/3371 (<1%) children screened with clinical meningoencephalitis met enrolment [41]. Notably, this trial included children with confirmed and probable TBM; potentially excluding those with early-stage disease who met the definition of possible TBM. To date TBM-KIDS, an open label randomized trial in Indian and Malawian children evaluating efficacy of high-dose (30 mg/kg) oral rifampicin, and SURE, a factorial and noninferiority trial evaluating the efficacy of shortened intensified anti-TB chemotherapy and adjuvant aspirin, represent the only clinical trials in paediatric TBM.
CONCLUSION
TBM is difficult to diagnose, optimal drug regimens are unknown, and management of common complications poorly evidence-based. The true global burden of TBM is hard to ascertain, and many cases likely remain undiagnosed [42]. Standardization of research methods and of study reporting, facilitated by global networking through the TBM International Research Consortium, first convened in 2009, represents progress. The consortium has also recently published a collection of reviews and opinion pieces on TBM (https://wellcomeopenresearch.org/collections/tbmeningitis). Recent and new clinical trials are encouraging, however, there is still a long way to go to convert an increasing evidence base into improved clinical outcomes. Few studies exist in paediatric TBM, despite the high burden of disease in this population. Studies of complications causing critical illness of TBM are particularly lacking. Research priorities include developing a high-sensitivity diagnostic test with the ability to exclude TBM. In addition, evidence to support shortened intensified anti-TB chemotherapy regimens would be welcome. Unfortunately, the current prognosis of TBM, particularly of severe cases, remains poor, and mortality and morbidity from this disease unacceptably high.
Acknowledgements
None.
Financial support and sponsorship
The authors are supported by the Wellcome Trust, United Kingdom.
Conflicts of interest
There are no conflicts of interest.
REFERENCES AND RECOMMENDED READING
Papers of particular interest, published within the annual period of review, have been highlighted as:
▪ of special interest
▪▪ of outstanding interest
REFERENCES
- 1.World Health Organization. Global tuberculosis report 2019. 2019. Available at: https://www.who.int/tb/global-report-2019 [Accessed 8 December 2019]. [Google Scholar]
- 2.du Preez K, Seddon JA, Schaaf HS, et al. Global shortages of BCG vaccine and tuberculous meningitis in children. Lancet Glob Heal 2019; 7:e28–e29. [DOI] [PubMed] [Google Scholar]
- 3.Chiang SS, Khan FA, Milstein MB, et al. Treatment outcomes of childhood tuberculous meningitis: a systematic review and meta-analysis. Lancet Infect Dis 2014; 14:947–957. [DOI] [PubMed] [Google Scholar]
- 4.Wilkinson RJ, Rohlwink U, Misra UK, et al. Tuberculous meningitis. Nat Rev Neurol 2017; 13:581–598. [DOI] [PubMed] [Google Scholar]
- 5.Manyelo CM, Solomons RS, Snyders CI, et al. Application of cerebrospinal fluid host protein biosignatures in the diagnosis of tuberculous meningitis in children from a high burden setting. Mediators Inflamm 2019; 2019:7582948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rohlwink UK, Mauff K, Wilkinson KA, et al. Biomarkers of cerebral injury and inflammation in pediatric tuberculous meningitis. Clin Infect Dis 2017; 65:1298–1307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Marais S, Thwaites G, Schoeman JF, et al. Tuberculous meningitis: a uniform case definition for use in clinical research. Lancet Infect Dis 2010; 10:803–812. [DOI] [PubMed] [Google Scholar]
- 8▪.Heemskerk AD, Donovan J, Thu DDA, et al. Improving the microbiological diagnosis of tuberculous meningitis: a prospective, international, multicentre comparison of conventional and modified Ziehl-Neelsen stain, GeneXpert, and culture of cerebrospinal fluid. J Infect 2018; 77:509–515. [DOI] [PMC free article] [PubMed] [Google Scholar]; Initial studies suggested modified Ziehl--Neelsen smear microscopy of CSF would greatly improve conventional Ziehl--Neelsen smear microscopy. However, this study did not support that.
- 9▪.Siddiqi OK, Birbeck GL, Ghebremichael M, et al. Prospective cohort study on performance of cerebrospinal fluid (CSF) Xpert MTB/RIF, CSF lipoarabinomannan (LAM) lateral flow assay (LFA), and urine LAM LFA for diagnosis of tuberculous meningitis in Zambia. J Clin Microbiol 2019; 57: DOI:10.1128/JCM.00652-19. [DOI] [PMC free article] [PubMed] [Google Scholar]; New high-sensitivity diagnostics urgently required for Mtb detection in CSF. Unfortunately, CSF and urinary LAM are not sensitive enough in this study.
- 10.Kwizera R, Cresswell FV, Mugumya G, et al. Performance of Lipoarabinomannan Assay using cerebrospinal fluid for the diagnosis of tuberculous meningitis among HIV patients. Wellcome open Res 2019; 4:123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sigal GB, Pinter A, Lowary TL, et al. A novel sensitive immunoassay targeting the 5-methylthio-d-xylofuranose-lipoarabinomannan epitope meets the WHO's performance target for tuberculosis diagnosis. J Clin Microbiol 2018; 56: DOI:10.1128/JCM.01338-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12▪.Broger T, Sossen B, du Toit E, et al. Novel lipoarabinomannan point-of-care tuberculosis test for people with HIV: a diagnostic accuracy study. Lancet Infect Dis 2019; 19:852–861. [DOI] [PMC free article] [PubMed] [Google Scholar]; Whilst not yet studied for TBM diagnosis, this study offers promise for FujiLAM as a higher sensitivity diagnostic than first generation LAM testing.
- 13.World Health Organisation. WHO | Next-generation Xpert® MTB/RIF Ultra assay recommended by WHO. 2017. Available at: https://www.who.int/tb/features_archive/Xpert-Ultra/en/ (Accessed 29 January 2019) [Google Scholar]
- 14.Foundation for Innovative New Diagnostics (FIND). Report for WHO: a multicentre noninferiority diagnostic accuracy study of the Ultra assay compared to Xpert MTB/RIF assay. Geneva: Foundation for Innovative New Diagnostics (FIND); 2017. 2017. Available at: https://www.finddx.org/wp-content/uploads/2017/03/Ultra-WHO-report_24MAR2017_FINAL.pdf [Accessed 9 December 2019]. [Google Scholar]
- 15.Dorman SE, Schumacher SG, Alland D, et al. Xpert MTB/RIF ultra for detection of Mycobacterium tuberculosis and rifampicin resistance: a prospective multicentre diagnostic accuracy study. Lancet Infect Dis 2018; 18:76–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16▪.Bahr NC, Nuwagira E, Evans EE, et al. ASTRO-CM Trial Team. Diagnostic accuracy of Xpert MTB/RIF Ultra for tuberculous meningitis in HIV-infected adults: a prospective cohort study. Lancet Infect Dis 2018; 18:68–75. [DOI] [PMC free article] [PubMed] [Google Scholar]; First study of Ultra testing of CSF showed improved diagnostic sensitivity over Xpert, for TBM diagnosis.
- 17.Wang G, Wang S, Jiang G, et al. Xpert MTB/RIF Ultra improved the diagnosis of paucibacillary tuberculosis: a prospective cohort study. J Infect 2019; 78:311–316. published online Feb 21. DOI: 10.1016/j.jinf.2019.02.010. [DOI] [PubMed] [Google Scholar]
- 18.Wu X, Tan G, Gao R, et al. Assessment of the Xpert MTB/RIF Ultra assay on rapid diagnosis of extrapulmonary tuberculosis. Int J Infect Dis 2019; 81:91–96. [DOI] [PubMed] [Google Scholar]
- 19.Perez-Risco D, Rodriguez-Temporal D, Valledor-Sanchez I, Alcaide F. Evaluation of the Xpert MTB/RIF ultra assay for direct detection of Mycobacterium tuberculosis complex in smear-negative extrapulmonary samples. J Clin Microbiol 2018; 56: DOI:10.1128/JCM.00659-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jyothy A, Ratageri VH, Illalu S, et al. The utility of CSF Xpert MTB/RIF in diagnosis of tubercular meningitis in children. Indian J Pediatr 2019; 86:1089–1093. [DOI] [PubMed] [Google Scholar]
- 21.Colas RA, Nhat LTH, Thuong NTT, et al. Proresolving mediator profiles in cerebrospinal fluid are linked with disease severity and outcome in adults with tuberculous meningitis. FASEB J 2019; 33:13028–13039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22▪▪.van Laarhoven A, Dian S, Aguirre-Gamboa R, et al. Cerebral tryptophan metabolism and outcome of tuberculous meningitis: an observational cohort study. Lancet Infect Dis 2018; 18:526–535. [DOI] [PubMed] [Google Scholar]; Important study of tryptophan in TBM, previously unknown to be associated with patient outcome, which may lead to development of new therapeutic targets.
- 23.Thuong NTT, Heemskerk D, Tram TTB, et al. Leukotriene A4 hydrolase genotype and HIV infection influence intracerebral inflammation and survival from tuberculous meningitis. J Infect Dis 2017; 215:1020–1028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Misra UK, Kalita J, Singh AP, Prasad S. Vascular endothelial growth factor in tuberculous meningitis. Int J Neurosci 2012; 123:128–132. [DOI] [PubMed] [Google Scholar]
- 25.Rohlwink UK, Walker NF, Ordonez AA, et al. Matrix metalloproteinases in pulmonary and central nervous system tuberculosis-a review. Int J Mol Sci 2019; 20:1350.DOI:10.3390/ijms20061350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Thuong NTT, Vinh DN, Hai HT, et al. Pretreatment cerebrospinal fluid bacterial load correlates with inflammatory response and predicts neurological events during tuberculous meningitis treatment. J Infect Dis 2019; 219:986–995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Thwaites GE, Tran TH. Tuberculous meningitis: many questions, too few answers. Lancet Neurol 2005; 4:160–170. [DOI] [PubMed] [Google Scholar]
- 28▪▪.Rohlwink UK, Figaji A, Wilkinson KA, et al. Tuberculous meningitis in children is characterized by compartmentalized immune responses and neural excitotoxicity. Nat Commun 2019; 10:3767. [DOI] [PMC free article] [PubMed] [Google Scholar]; Paediatric study showing distinct immune response patterns related to cerebral injury in TBM, and compartmentaliization of these responses within ventricular CSF.
- 29.Thao LTP, Heemskerk AD, Geskus RB, et al. Prognostic models for 9-month mortality in tuberculous meningitis. Clin Infect Dis 2018; 66:523–532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30▪.Thao LTP, Wolbers M, Heemskerk AD, et al. Dynamic prediction of death in patients with tuberculous meningitis using time-updated Glasgow coma score and plasma sodium measurements. Clin Infect Dis 2019. [DOI] [PubMed] [Google Scholar]; Dynamic prognostic score, expanding on previous nondynamic prognostic score, allows prognostic calculations after baseline.
- 31.Donovan J, Figaji A, Imran D, et al. The neurocritical care of tuberculous meningitis. Lancet Neurol 2019; 18:771–783. [DOI] [PubMed] [Google Scholar]
- 32▪.Misra UK, Kalita J, Kumar M. Safety and efficacy of fludrocortisone in the treatment of cerebral salt wasting in patients with tuberculous meningitis: a randomized clinical trial. JAMA Neurol 2018; 75:1383–1391. [DOI] [PMC free article] [PubMed] [Google Scholar]; Hyponatraemia in TBM is complicated and best management uncertain. Here fludrocortisone shown to not only aid correction of hyponatraemia but also to associate with adverse events.
- 33.Misra UK, Kumar M, Kalita J. Seizures in tuberculous meningitis. Epilepsy Res 2018; 148:90–95. [DOI] [PubMed] [Google Scholar]
- 34.Aranha A, Choudhary A, Bhaskar S, Gupta LN. A randomized study comparing endoscopic third ventriculostomy versus ventriculoperitoneal shunt in the management of hydrocephalus due to tuberculous meningitis. Asian J Neurosurg 2018; 13:1140–1147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35▪.Harrichandparsad R, Nadvi SS, Suleman Moosa M-Y, Rikus van Dellen J. Outcome of ventriculoperitoneal shunt surgery in human immunodeficiency virus-positive patients on combination antiretroviral therapy with tuberculosis meningitis and hydrocephalus. World Neurosurg 2018; 123:e574–e580. [DOI] [PubMed] [Google Scholar]; Provides evidence to guide VP shunting in HIV co-infected individuals with TBM-associated hydrocephalus.
- 36.Donovan J, Rohlwink UK, Tucker EW, et al. Tuberculous Meningitis International Research Consortium. Checklists to guide the supportive and critical care of tuberculous meningitis. Wellcome Open Res 2019; 4:163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mai NT, Dobbs N, Phu NH, et al. A randomised double blind placebo controlled phase 2 trial of adjunctive aspirin for tuberculous meningitis in HIV-uninfected adults. Elife 2018; 7: pii: e33478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Donovan J, Phu NH, Thao LTP, et al. Adjunctive dexamethasone for the treatment of HIV-uninfected adults with tuberculous meningitis stratified by Leukotriene A4 hydrolase genotype (LAST ACT): study protocol for a randomised double blind placebo controlled noninferiority trial. Wellcome Open Res 2018; 3:32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Donovan J, Phu NH, Mai NTH, et al. Adjunctive dexamethasone for the treatment of HIV-infected adults with tuberculous meningitis (ACT HIV): study protocol for a randomised controlled trial. Wellcome Open Res 2018; 3:31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Garcia-Prats AJ, Svensson EM, Weld ED, et al. Current status of pharmacokinetic and safety studies of multidrug-resistant tuberculosis treatment in children. Int J Tuberc Lung Dis 2018; 22:15–23. [DOI] [PubMed] [Google Scholar]
- 41.Paradkar M, Devaleenal DB, Mvalo T, et al. Challenges in conducting trials for pediatric tuberculous meningitis: lessons from the field. Int J Tuberc Lung Dis 2019; 23:1082–1089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Seddon JA, Thwaites GE. Tuberculous Meningitis International Research Consortium. Tuberculous meningitis: new tools and new approaches required. Wellcome Open Res 2019; 4:181. [DOI] [PMC free article] [PubMed] [Google Scholar]


