Abstract
Gastrointestinal stromal tumours (GIST) are the most common gastrointestinal mesenchymal tumours of the gastrointestinal tract. They are diagnosed by the expression of markers like CD 117, CD 34, DOG-1 and PDGFRA. The identification of these mutations has resulted in a better understanding of their oncogenic mechanisms. Few studies have shown the high incidence of a second malignancy including papillary thyroid cancer (PTC) in known patients of GIST. Literature review on pathogenesis of GIST and PTC showed that PTC targeting Protein Kinase C theta (PKCθ) plays role in both PTC and GIST. Further studies have also shown that, apart from somatic and familial gastrointestinal stromal tumours, PDGFRA is associated with many other malignancies including PTC. These studies explain the common genetic pathway involved in the development of GIST and PTC in same patient. In spite of common genetic association between GIST and other malignancies, none of the standard protocols recommends screening for second malignancy. In this article, we present the details of four patients who had associated GIST and PTC at the same time or developed during follow-up.
Keywords: Gastrointestinal stromal tumour (GIST), Papillary thyroid carcinoma, KIT mutation, PDGFRA, Protein kinase C theta (PKCθ)
Introduction
Gastrointestinal stromal tumours (GISTs) have been separated from smooth muscle and neural tumours of the gastrointestinal tract (GIT) as a biologically distinctive tumour type. They are the most common gastrointestinal mesenchymal tumours of the GIT and are known to be refractory to conventional chemotherapy or radiation. They are diagnosed by the expression of markers like CD 117, CD 34, DOG-1 and PDGFRA. The identification of these mutations has resulted in a better understanding of their oncogenic mechanisms. Both traditional and minimally invasive surgeries are used to remove these tumours with minimal morbidity and excellent postoperative outcomes. The revolutionary use of specific, molecularly targeted therapies, such as imatinib, reduces the frequency of disease recurrence when used as an adjuvant following complete resection. Neoadjuvant treatment with these agents appears to stabilize disease in the majority of patients and may reduce the extent of surgical resection required for subsequent complete tumour removal [1].
Few studies have shown the high incidence of a second malignancy in patients of GIST. Among them most common are tumours of GI tract, genitourinary tract, breast cancers and haematological cancers. These studies also have shown a very small incidence of papillary thyroid cancer (PTC) in patients who have been treated for GIST, but none of them have quoted the genetic pathways causing GIST and PTC in same patient [2, 3].
In this article, we present the details of four patients presenting with both GIST and PTC (Table 1); along with this, we also reviewed the literature on probable genetic cause(s) for this combination.
Table 1.
Details of the GISTs in the four patients
| Age/sex | Mode of presentation | Site of GIST | Size of GIST | Mitotic index | |
|---|---|---|---|---|---|
| Patient 1 | 56/F | Incidentally detected on imaging | Stomach | 3 × 3 cm | 2/50hpf |
| Patient 2 | 40/M | Melaena | Stomach | 6 × 4 cm | 6/50hpf |
| Patient 3 | 45/F | Melaena | Jejunum | 4 × 4 cm | 8/50hpf |
| Patient 4 | 52/F | Melaena | Stomach | 5 × 5 cm | 0/50hpf |
Case Series
Over a 6 months’ period, 4 patients suffering from both GIST and PTC presented in our department. Two patients were incidentally detected, and the other 2 patients of GIST were found to have PTC on screening, based on the experience in the previous 2 cases.
The first patient was a 56-year-old female who had undergone total thyroidectomy for PTC 6 months back and came with a chronic discharging sinus at the operated site. Patient was planned for exploration of the neck sinus under general anaesthesia. During work-up for anaesthesia fitness, the patient had to undergo a CT scan of the thorax. During this CT scan, the upper abdominal CT cuts revealed a 3-cm × 3-cm incidental mass in the stomach wall which was suspicious of GIST. Endoscopic ultrasound guided FNAC was inconclusive. The patient underwent wedge resection of the stomach with the mass. The final histopathology showed a low grade GIST.
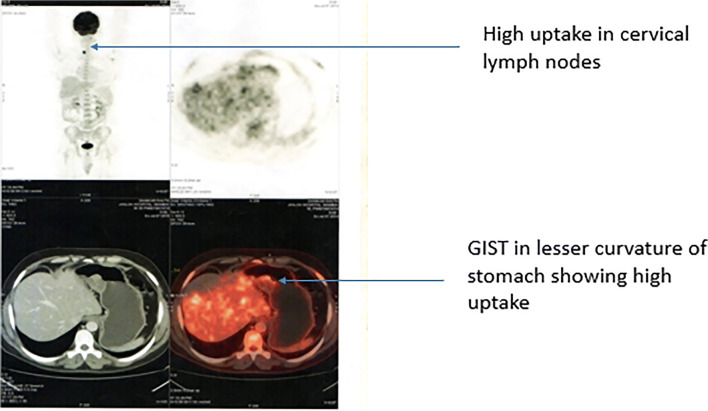
The second patient was a 40-year-old male patient who presented with frequent melaena and severe anaemia. On evaluation, he was found to have a 6-cm × 4-cm stomach GIST. His PET-CT scan showed a PET avid lesion at the site of the stomach GIST and another PET avid lesion in right lobe of thyroid. Ultrasonography (USG) of the neck showed a 3-cm × 2-cm lesion which on FNAC proved to be PTC (Fig. 1).
Fig. 1.
A PET avid GIST on the lesser curvature of stomach and one more PET avid lesion in the thyroid region [second case]
These two cases in a short period of time made us think whether there is any relation between GIST and PTC. After going through the literature, we could find a definite association of both these conditions. Hence, we decided to routinely screen all patients of GIST for PTC with USG of neck and FNAC if required.
The third patient was a 45-year-old female, with a known case of jejunal GIST who had undergone segmental resection of jejunum 4 years back. She had come for routine follow-up. A PET-CT scan and USG neck apart from routine blood investigations were advised. PET-CT showed no recurrence in any part of the body but there were few non-PET avid nodules in both lobes of thyroid. USG neck showed largest nodule to be 2.5 cm × 2.5 cm in right lobe (Fig. 2). FNAC was done from this largest nodule which confirmed the PTC.
Fig. 2.
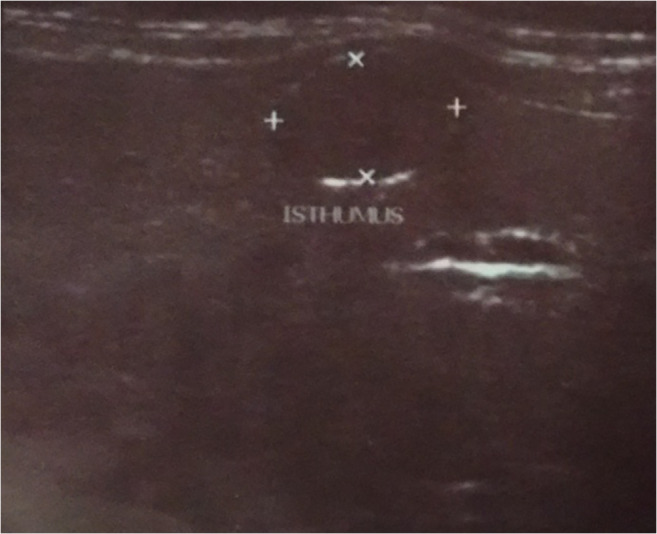
USG neck of an already proven case of GIST showing a hypoechoic lesion in the thyroid. Subsequently, FNAC of this lesion proved it to be PTC
Similarly, the fourth patient in this series was a 52-year-old female patient who had undergone distal gastrectomy for stomach GIST 1 year back and she was on regular adjuvant treatment with Imatinib. Her PET-CT also showed non-PET avid nodules in thyroid with no recurrence in any other part. USG-guided FNAC from the largest nodule revealed PTC.
Discussion
Most of the GIST cases are sporadic and there are very few reported syndromes of autosomal-dominant hereditary GIST that results from inheritance of a germ-line KIT or platelet-derived growth factor receptor (PDGFR) mutation [4]. Apart from these, there are few syndromes in which affected individuals may develop GIST along with other types of tumours. Examples include malignant peripheral nerve sheath tumour in neurofibromatosis [5], paraganglioma and pulmonary chondroma in Carney’s triad [6], and paraganglioma in the Carney–Stratakis syndrome [7]. But none of the syndromes included GIST and PTC.
Few studies have reported that it is not only PTC but also cancers of many other sites that are common among patients of GIST. Murphy et al. analysed 6112 patients of GIST and found that 1047 (17.1%) patients had second malignancy with standardized incidence ratios for papillary thyroid cancer as 5.16. Forty-four percent of the patients were diagnosed before while 66% after the diagnosis of GIST [8]. Pandurengan et al. [2] published their observation in 783 patients of GIST. Out of 783 patients, 153 (20%) were identified with at least one additional primary. Second malignancies observed were cancers of the prostate, breast, oesophagus, colorectal, kidney, melanoma, thyroid and lung. But they could detect only 3 patients who developed PTC. The 5-year survival was 68% for patients with primaries before GIST and 61% for patients with primaries after GIST [2]. A similar study was conducted by Kramer et al. [3] and reported the incidence of second malignancy in GIST patients to be as high as 31.9%. This study also showed origin of second malignancy from variety of organs including GI tract, GU tract, thyroid, head, and neck. They reported only 3 cases of PTC among 836 total patients of GIST.
Huang et al. [9] analysed the expression data sets of PTC from The Cancer Genome Atlas (TCGA) Data Portal and demonstrated for the first time that immune responses are significantly enriched and under specific regulation in the direct miRNA2 target network among distinctive PTC variants to different extents. From the conventional miRNA2 target network perspective, they identified miR-34a-5p upregulation among PTC targeting Protein Kinase C theta (PKCθ). Furthermore, considering the ability of PKCθ to regulate KIT expression in gastrointestinal stromal tumours (GIST) [10], miR-34a- 5p targeting to PRKCQ and KIT could augment the effects of miR- 221-3p: and miR-222-3p: KIT interactions, which may accordingly lead to PTC pathogenesis [9]. Thus, they gave sufficient evidence that PKCθ plays role in both PTC and GIST and probably this is the cause in our patients for the association of GISTs and PTCs.
Wei et al. [11] evaluated the role of PDGFRA in thyroid cancer. PDGFRA encodes a cell surface tyrosine kinase receptor for members of the platelet-derived growth factor family. They suggested that this gene plays a role in organ development, wound healing, and tumour progression. Mutations, copy number variations, and polymorphisms in this gene have been found to be associated with somatic and familial gastrointestinal stromal tumours (GIST), as well as other cancers including PTC. Hence, this is a possible cause of PTC developing in PDGFRA-induced GIST patients.
Whether the occurrence of a second tumour in GIST patients is incidental or it has a genetic cause is unknown. More genetic studies are required to find the pathogenesis involved in development of second malignancy. In spite of the known fact that there is high incidence of second malignancy in patients of GIST, none of the standard protocols recommends screening for second malignancy.
PET-CT serves as a good tool to detect recurrence of GIST as well as to detect second tumour in an early stage but 3 out of 4 cases of PTC were non-PET avid in our series. Thus, we believe that all patients of GIST should be screened for second tumour with appropriate modalities. We have experienced difficulty in characterizing the thyroid nodule based on only PET scan. Hence, we recommend USG neck and FNAC of thyroid nodules as part of screening method in cases of GIST to rule out PTC.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Rammohan A. A gist of gastrointestinal stromal tumours: a review. World J Gastrointest Oncol. 2012;5(6):102–112. doi: 10.4251/wjgo.v5.i6.102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pandurengan R, Dumont A, Araujo D, Ludwig J, Ravi V, Patel S, et al. Survival of patients with multiple primary malignancies: a study of 783 patients with gastrointestinal stromal tumor. Ann Oncol. 2010;21(10):2107–2111. doi: 10.1093/annonc/mdq078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kramer K, Wolf S, Mayer B, Schmidt S, Agaimy A, Henne-Bruns D, et al. Frequency, spectrum and prognostic impact of additional malignancies in patients with gastrointestinal stromal tumours. Neoplasia. 2015;17(1):134–140. doi: 10.1016/j.neo.2014.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gupta P, Tewari M, Shukla HS. Gastrointestinal stromal tumor. SurgOncol. 2008;17:129–138. doi: 10.1016/j.suronc.2007.12.002. [DOI] [PubMed] [Google Scholar]
- 5.Fuller CE, Williams GT. Gastrointestinal manifestations of type 1 neurofibromatosis (von Recklinghausen’s disease) Histopathology. 1991;19:1–11. doi: 10.1111/j.1365-2559.1991.tb00888.x. [DOI] [PubMed] [Google Scholar]
- 6.Carney JA. The triad of gastric epithelioidleiomyosarcoma, pulmonary chondroma, and functioning extra-adrenal paraganglioma: a five-year review. Medicine (Baltimore) 1983;62:159–169. doi: 10.1097/00005792-198305000-00003. [DOI] [PubMed] [Google Scholar]
- 7.Carney JA, Sheps SG, Go VL, Gordon H. The triad of gastric leiomyosarcoma, functioning extra-adrenal paraganglioma and pulmonary chondroma. N Engl J Med. 1977;296:1517–1518. doi: 10.1056/NEJM197706302962609. [DOI] [PubMed] [Google Scholar]
- 8.Murphy JD, Ma GL, Baumgartner JM, Madlensky L, Burgoyne AM, Tang, et al. Increased risk of additional cancers among patients with gastrointestinal stromal tumours: a population-based study. Cancer. 2015;121(17):2960–2967. doi: 10.1002/cncr.29434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Huang C-T, et al. MicroRNA-mediated networks underlie immune response regulation in papillary thyroid carcinoma. Sci Rep. 2014;4:6495. doi: 10.1038/srep06495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ou W-b, et al. Protein Kinase C Theta (PKCθ) regulates KIT expression and proliferation in gastrointestinal stromal tumourS. Oncogene. 2008;27.42:5624–5634. doi: 10.1038/onc.2008.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wei W, Lu Z, Li D, Wang Y, Zhu Y, Wang Z, et al. Association of the miR-149 Rs2292832 polymorphism with papillary thyroid cancer risk and clinicopathologic characteristics in a Chinese population. IJMS. 2014;15(11):20968–20981. doi: 10.3390/ijms151120968. [DOI] [PMC free article] [PubMed] [Google Scholar]