Abstract
Hand, foot, and mouth disease (HFMD) is caused by various serotypes of Enterovirus genus. Coxsackievirus A16 (CV-A16) and enterovirus A71 (EV-A71) were known to be the only responsible agents for these epidemics; however, this opinion was challenged after the detection that coxsackievirus A6 (CV-A6) was the responsible species for the outbreak in Finland in 2008. HFMD is frequently seen in Turkey, and no detailed study on its clinical and microbiological epidemiology has previously been reported. The present study addresses this question. Twenty-seven patient samples collected between 2015 and 2017 were included in the study. Typing was conducted by RT-PCR and the sequencing applied directly to patient’s samples and as well as to the viral cultures with pan-enterovirus and serotype-specific primers. The presence of Enterovirus in 12 of 27 HFMD samples was shown with RT-PCR. The causative agent for three of these 12 samples was CV-A16, one of the most frequent two serotypes around the world, and the remaining nine samples was CV-A6. The findings of the study are relevant since it pertains to the molecular epidemiology of HFMD in Turkey, a gateway country where different serotypes might be circulating and transmitted. The findings also support the notion that CV-A6 cases are rising in number, which has caused more severe clinical features and widespread rashes in recent outbreaks.
Key words: hand foot and mouth disease, HFMD, enterovirus, coxsackievirus A6, coxsackievirus A16
Introduction
Hand, foot, and mouth disease (HFMD) is a viral and febrile disease frequently seen in children, generally presenting with a mild course and accompanied by oral rash and rashes on palmar surfaces of hand and feet (Kliegman and Geme 2015). Cases of HFMD are seen all over the world; however, epidemics with serious complications that result in deaths are reported mostly from Western Pacific Region countries (WHO 2011). Enterovirus A species are responsible for more than 90% of HFMD cases. Coxsackievirus A (CV-A) 2, 3, 5, 6, 7, 8, 10, 12, 14, 16, and enterovirus A71 (EV-A71) belong to this group. It is known that epidemics are induced by CV-A16 and EV-A71, while other enteroviruses may cause sporadic cases (Mao et al. 2014; Bian et al. 2015).
CV-A6 was identified, for the first time, as the causative agent for HFMD in an epidemic that emerged in Finland in 2008 (Österback et al. 2009). Following this, the identification of CV-A6 as the major causative agent for HFMD epidemics continued to occur in France, Spain and other European countries in 2009–2011 (Bracho et al. 2011; Mirand et al. 2012). HFMD epidemics caused by CV-A6 were also reported in the Western Pacific Region and the Americas after 2009 (CDC 2012; Chen et al. 2012; Fujimoto et al. 2012; Fonseca et al. 2014; Han et al. 2014; Hu et al. 2015). More severe clinical course and widespread skin rashes were seen in cases caused by CV-A6, different from typical HFMD (Romero 2017).
Cases with no severe clinical course can be diagnosed with clinical findings without the need for microbiological diagnostic tests. However, certain serotypes may lead to severe disease (Romero 2017). Serotypes may vary within and among countries. Therefore, determination of the causative serotype is important for public health as well as for the design and implementation of the preventive measures. Recent increases in movements of people between countries and continents could be influential in the spread of different viral serotypes. Full identification of the causative organism could also provide information for the vaccine development studies in addition to its epidemiological importance (WHO 2011).
The published research on HFMD in Turkey has been generally towards the clinical aspects of the disease (Ekinci et al. 2013; Demirhan et al. 2016; Bucak et al. 2017). Apart from a single case study, no detailed investigations on the viral agent itself have been conducted (Kiratli et al. 2017). In this investigation, we aimed to determine the serotype distribution in HFMD cases in children.
Experimental
Materials and Methods
Patient Selection. Our study cohort consisted of children applied to the outpatient clinics of the Pediatric Infectious Disease Department of Bezmialem Vakif University Hospital between January 2015 and November 2017. These consecutive patients were selected who complained of fever and rash on the hands, mouths or on the body including the legs (Fig. 1, 2 and 3) and who were later clinically diagnosed with HFMD. An approval from Bezmialem Vakıf University Ethics Committee for Experimentation Involving Human Subjects was obtained before the study and informed consent was obtained from all families of the subjects.
Fig. 1.
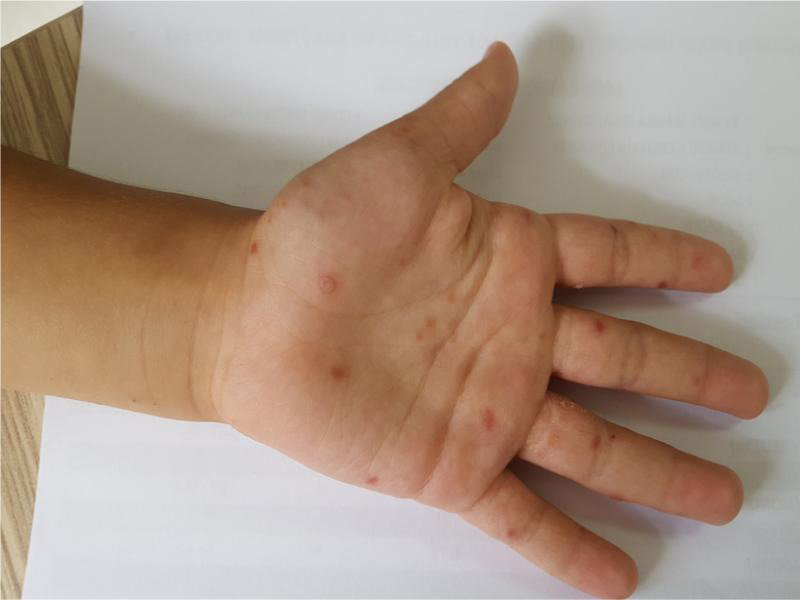
Patients with typical lesions on the hands.
Fig. 2.
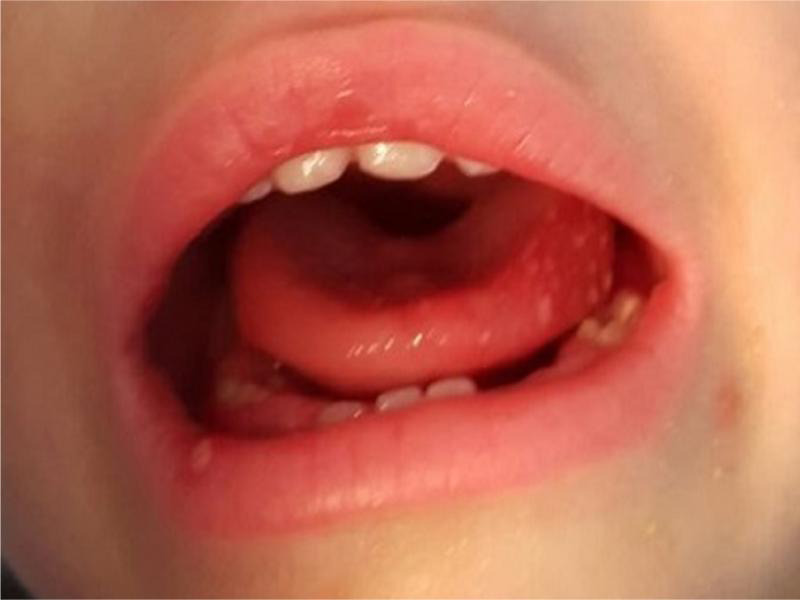
Patients with mouth ulcers.
Fig. 3.
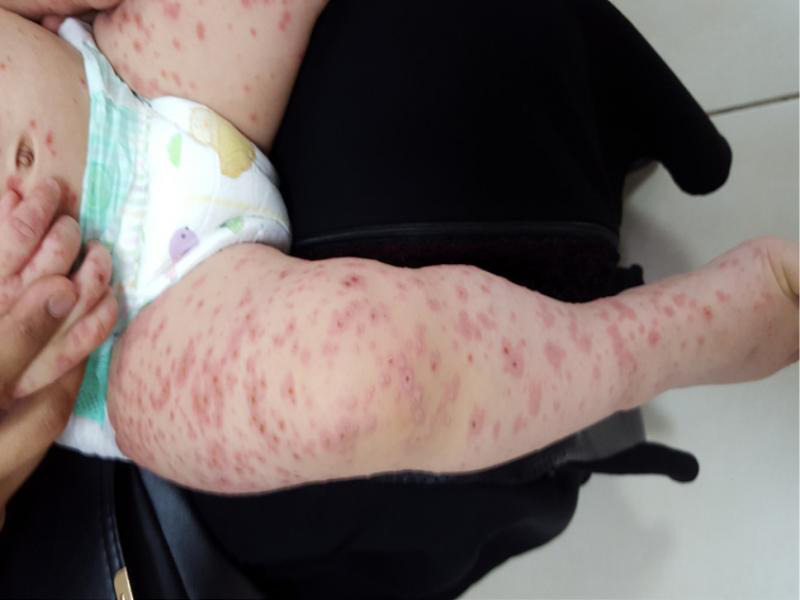
Patient with generalized lesions on the legs.
Sample Collection and Storage. Samples were taken from at least two lesions in oral mucosa or trunk of patients with clinical symptoms of HFMD. Sterile cotton swabs wetted with saline (0.9% NaCl) were used for sampling. The samples were stored at –80°C in 1.5 ml sterile tubes containing phosphate buffered saline (PBS) until further processed.
RNA isolation. Direct-zol RNA MiniPrep Kit (Zymo Research, Irvine, CA, USA) was used for nucleic acid extraction directly from patient samples and from supernatants obtained after viral culture. Manufacturer’s instructions were followed for the entire procedure.
Reverse transcription. SensiFAST cDNA Synthesis Kit (Bioline, London, UK) was used to synthesize cDNA from isolated viral RNAs using random primers and the manufacturer’s instructions were followed at all steps. cDNA was purified from reverse transcription products with Zymo Research DNA Clean & Concentrator™-5 kit in accordance with the manufacturer’s instructions.
Designing primers and PCR. Pan-enterovirus primers specific to the Enterovirus genus targeting 5’UTR were designed for nested-PCR on basis of previously published studies (Ge et al. 2013). In addition, specific primers targeting VP1 gene were used for EV-A71 and CV-A16 (Table I). A commercial kit (Illustra™ Hot Start Master Mix, GE Healthcare, Buckinghamshire, UK) was used for PCR. The reaction mixture contained 0.5 μmol forward primer, 0.5 μmol reverse primer, 1 μl (1–4 μg/μl) post clean-up purified cDNA, and 22 μl DNase-RNase free water added to the master mix kept ready in PCR tubes. Veriti® thermal cycler (Applied Biosystems, Foster City, CA, USA), was programmed as 94°C for 3 min initial incubation, 30 cycles of 94°C for 30 sec, 59°C for 30 sec, 72°C for 30 sec and 72°C for 5 min in order to amplify the Pan-EV, CV-A16 and EV-A71 specific products.
Table I.
Primers used for PCR.
| Target Serotype | Orientation | Sequence |
|---|---|---|
| Pan-EV-5’UTR Outer | Forward | 5’-CYTTGTGCGCCTGTTTT-3’ |
| Reverse | 5’-ATTGTCACCATAAGCAGCC-3’ | |
| Pan-EV-5’UTR-Inner | Forward | 5’-CAAGYACTTCTGTMWCCCC-3’ |
| Reverse | 5’-CCCAAAGTAGTCGGTTCC-3’ | |
| EV-A71-VP1 | Forward | 5’-AGAGCATGATTGAGACACG-3’ |
| Reverse | 5’-RTCTTTCTCYTGTTTGTGTTC-3’ | |
| CV-A16-VP1 | Forward | 5’-TGCAGACATGATTGACCAG-3’ |
| Reverse | 5’-TGCCTACAGTTCTGATGCTA-3’ |
The amplification products and the 100 bp marker were loaded into 1.5% agarose gel, electrophoresed for 40 minutes under 80 V, and analyzed in Molecular Imager Gel Doc XR System (BioRad, Hercules, CA, U.S.A.) imaging device.
Cell culture. Vero E6 cells were cultured in vitro with DMEM (Gibco, Thermo Fisher Scientific, Waltham, MA, USA) containing 10% fetal bovine serum (Gibco) in 5% CO2 medium at 37°C. Samples were filtered through 0.45 μm filters and then added to monolayers of Vero E6 cell cultures. The infected cells were examined after 3 days of incubation for evaluation of Enterovirus specific cytopathic effects. Reverse-transcription PCR was performed with cell culture supernatants to confirm the PCR results of the patient samples.
Sanger sequencing and phylogenetic analysis. The sequencing of the amplified product was carried out by the Sentegen Biyotek (Ankara, Turkey) to determine the serotype of Enterovirus detected in the samples.
The sequences of PCR products were assembled and edited using BioEdit version 7.0.5 (http://www.mbio.ncsu.edu/BioEdit/bioedit.html). This sequence was compared with the sequences deposited in GenBank of the National Center for Biotechnology Information (NCBI) by means of the BLASTN 2.2.19 option. The consensus sequence was compiled in a FASTA file for the phylogenetic analysis and aligned with the Clustal W 1.8.1. profile mode included in the Mega 4.0.2 (https://www.megasoftware.net/mega4/) software.
Results
Eleven of 27 children included in the study were female and 16 were male. The ages ranged from 4 months to 9 years, and 21 of the 27 children were aged between 1 and 3 years (78%). Eighteen of the patients (67%) applied to the hospital during the summer months. In the cohorts, neither systemic blisters nor high fever was noted. None of our patients needed hospitalization. There were no severe complications such as meningitis and encephalitis. Only three of 27 patients applied to the hospital with nail shedding after the recovery period of approximately six weeks.
A total of 27 patient samples, 15 from the 2015–2016 season and 12 from 2017, were included in the study. The nested PCR results of 12 patients, sampled in 2015–2016, revealed products with a length of 530 and 389 bp (Fig. 4 and 5) that corresponded to the pan-enterovirus outer and inner primers, respectively. This finding was interpreted as proof of the Enterovirus content in the samples. No positive results were observed after PCR performed with the species-specific primers targeting CV-A16 and EV-A71 viruses. To confirm the presence of enteroviral genetic material, all 12 PCR positive products were sequenced. Sequencing revealed that nine of these samples were consistent with the CV-A6 sequences but the remaining samples were not (Table II). Afterward, a confirmatory species-specific PCR affirmed that the samples were indeed CV-A6 (Fig. 6).
Fig. 4.
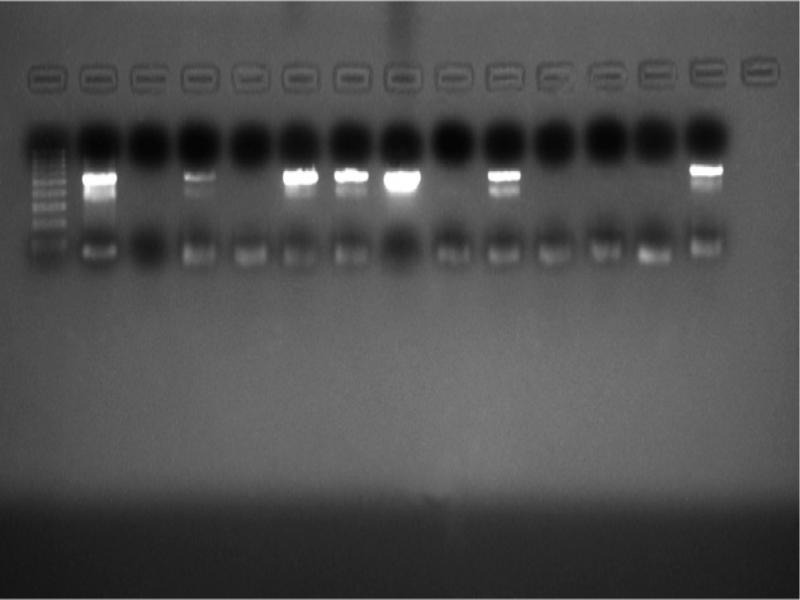
The samples were subjected to the pan-enterovirus specific nested PCR with outer primers where 530 bp products were amplified. Left to right, line 1: 100 bp marker, lines 2 to 5: HFM-2 to HFM-5, line 6: HFM-7, line 7: HFM-9, lines 8 to 14: HFM-10 to HFM-16.
Fig. 5.
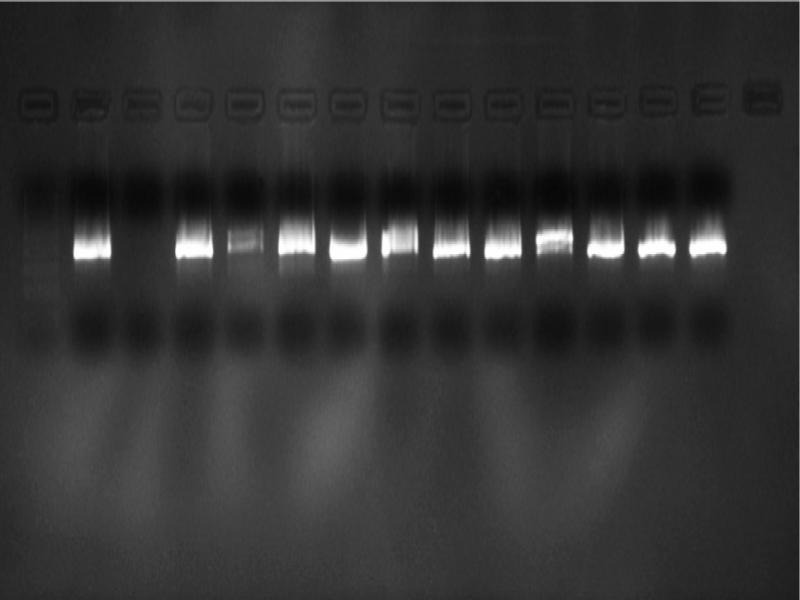
The samples were subjected to the pan-enterovirus specific nested PCR with inner primers where 389 bp products were amplified. Left to right, line 1: 100 bp marker, lines 2 to 5: HFM-2 to HFM-5, line 6: HFM-7, line 7: HFM-9, lines 8 to 14: HFM-10 to HFM-16.
Table II.
Phylogenetic identities of the coxsackievirus A isolates, based on VP1 sequences.
| Isolate code | Primer | Nearest phylogenetic neighbours | Identity percentage |
|---|---|---|---|
| HFM-2 | 153 F/541 R | Coxsackievirus A6 | 99% |
| HFM-5 | 153 F/541 R | Coxsackievirus A6 | 99% |
| HFM-9 | 153 F/541 R | Coxsackievirus A6 | 99% |
| HFM-10 | 153 F/541 R | Coxsackievirus A6 | 99% |
| HFM-11 | 153 F/541 R | Coxsackievirus A6 | 99% |
| HFM-12 | 153 F/541 R | Coxsackievirus A6 | 99% |
| HFM-14 | 153 F/541 R | Coxsackievirus A6 | 99% |
| HFM-15 | 153 F/541 R | Coxsackievirus A6 | 99% |
| HFM-16 | 153 F/541 R | Coxsackievirus A6 | 99% |
| HFM24 | 153 F/541 R | Coxsackievirus A16 | 99% |
| HFM25 | 153 F/541 R | Coxsackievirus A16 | 99% |
| HFM26 | 153 F/541 R | Coxsackievirus A16 | 97% |
Fig. 6.
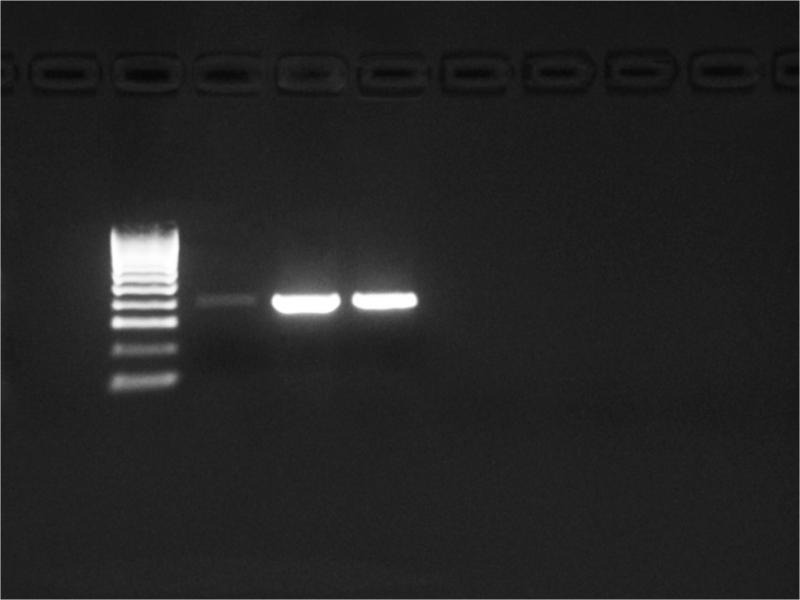
CV-A6 specific primers amplified products of approximately 645 bp.
Twelve patient samples from 2017, were amplified by PCR and seven samples were positive for Enterovirus. These samples were directly subjected to sequencing without serotype-specific PCR for EV-A71 and CV-A16, since the previous samples were negative. Three of the seven samples were CV-A16 according to the results of sequencing (Fig. 7).
Fig. 7.

Phylogenetic analysis of clinical isolates. Phylogenetic analysis of coxsackievirus A6 nucleotide sequences that were obtained using 153F ve 541R primers, showed the relationships between the clinical CV-A6 and the CV-A16 isolate selected from the GenBank. A scale bar indicates branch distances. The phylogenetic tree was constructed using neighbour-joining method and validated with 1000 pseudo-replicates. Poliovirus (NC 002058.3) was selected as out-group.
All the samples were inoculated into the Vero E6 cell line and characteristic cytopathic effects were noticed after 3-days incubation. The supernatant samples were collected and the PCR test was carried out with pan-enterovirus primers (data not shown). It was noticed that the results were compatible with the PCR performed directly from the patient samples.
Discussion
Hand, foot, and mouth disease epidemics are mostly caused by EV-A71 and CV-A16. Recently, CV-A6 has been identified as the agent of epidemics causing severe forms of the disease with general skin involvement. Similar epidemics that started in Finland in 2008 were also reported from Singapore in 2009, France and Taiwan in 2010, and the USA, Cuba, Spain, Japan, and China in 2011 (Bian et al. 2015). In this study, CV-A6 was found to be the causative agent in 75% of the cases what was confirmed by sequencing (Table III). This result is consistent with the current literature that reports an increase in CV-A6 incidence (Bian et al. 2015). HFMD is not a mandatory disease to report to public health authorities in Turkey. Therefore, complete data on the causative agents of the cases throughout the country is not yet known. Only a single case study identifying the causative agent was reported from Turkey (Kiratli et al. 2017). In that study, the identification of CV-16 in a 39 years old male patient was performed. The present study identified CV-A16 in 25% of the pediatric samples (Table III). A study conducted in China between 2011 and 2015 on 2130 samples positive for Enterovirus demonstrated that CV-A16 was identified in 32%, and EV-A71 – in 30% of the samples (Sun et al. 2017). It has been reported that EV-A71 infections may lead to neurological complications (e.g. aseptic meningitis, acute flaccid paralysis, and brainstem encephalitis), and mortality. All of the cases in our study were from outpatient services and no EV-A71 was identified in any patients. The literature reported severe cases with CV-A6 but none of our patients had a complicated course of the disease and needed hospitalization (Yang et al. 2014). Another clinical feature of CV-A6 is onychomadesis, (Wei et al. 2011) and in our cohorts, only one of the confirmed CV-A6 cases was re-admitted to the hospital at the 6th week after the discharge with complaints of onychomadesis.
Table III.
The patients’ samples and their positivity for coxsackievirus A6 and A16.
| Year | No. of samples | Enterovirus positivity | Coxsackievirus A6 positivity | Coxsackievirus A16 positivity |
|---|---|---|---|---|
| 2015–2016 | 15 | 12 | 9 | 0 |
| 2017 | 12 | 7 | 0 | 3 |
| Total | 27 | 19 | 9 | 3 |
The frequency of HFMD emergences peak twice in a year in countries with a tropical climate; however, it can be observed throughout the year. It exhibits a single annual rise in summer or fall in countries located at higher latitudes (Blomqvist et al. 2010; Huang et al. 2015; Wang et al. 2017). In Turkey, individual studies showed that the frequency of HFMD rises during summer (Topkarcı 2013; Uğraş et al. 2014; Yorulmaz and Onat 2017). In parallel with the previous research, in the present study, it has been observed that the majority of HFMD cases (67%) applied to our hospital during summer months.
Hand, foot, and mouth disease frequently affects children under the age of 5. Previous studies show that more than half of the individuals affected by epidemics were aged between 1 and 3 years (Montes et al. 2013; Huang et al. 2015). Seventy-eight percent of 27 children included in the current study was aged between 1 and 3 years. Epidemiological studies investigating HFMD have shown that the incidence is 1.5–2 times higher among boys compared to girls (Huang et al. 2015; Sun et al. 2017). In this study, similarly, the ratio of boys/girls was 16/11.
One of the limitations of the current study is that our cohorts were from a single city and the numbers of the patient were rather small. Therefore, our result might not reflect the overall HFMD epidemiology of all country. Secondly, only 12 sequencing results could be used to make the evolutionary trees to establish the relationship of the CV-A6, or CV-A16 in Turkey in relation to others in the world. Therefore further studies including samples from various regions of Turkey with a larger sampling might shed a better light on the dissemination as well as the phylogenetic relationships of these viral agents. Nevertheless, the data presented in the current study reports for the first time a detailed investigation about the presence of previously unnoticed serotypes on an important public health agent.
Geographically, Turkey acts as a gateway between Asia and Europe, and receives an influx of tourists from various countries across the world during summer months, the time that the incidence of HFMD peaks. Therefore, the establishment of a national surveillance system for HFMD and identification of causative serotype agents are important from an epidemiological point of view as well as for the planning and execution of preventive measures including vaccination. Our investigation and reporting might be useful in guiding these efforts.
Acknowledgements
We thank Hamid Ceylan Ph.D. for Blast Search assistance.
Footnotes
Conflict of interest
The authors do not report any financial or personal connections with other persons or organizations, which might negatively affect the contents of this publication and/or claim authorship rights to this publication.
Literature
- Bian L, Wang Y, Yao X, Mao Q, Xu M, Liang Z. Coxsackievirus A6: a new emerging pathogen causing hand, foot and mouth disease outbreaks worldwide. Expert Rev Anti Infect Ther. 2015. Sep 02;13(9):1061–1071. doi: 10.1586/14787210.2015.1058156Medline [DOI] [PubMed] [Google Scholar]
- Blomqvist S, Klemola P, Kaijalainen S, Paananen A, Simonen ML, Vuorinen T, Roivainen M. Co-circulation of coxsackieviruses A6 and A10 in hand, foot and mouth disease outbreak in Finland. J Clin Virol. 2010. May;48(1):49–54. doi: 10.1016/j.jcv.2010.02.002Medline [DOI] [PubMed] [Google Scholar]
- Bracho MA, González-Candelas F, Valero A, Córdoba J, Salazar A. Enterovirus co-infections and onychomadesis after hand, foot, and mouth disease, Spain, 2008. Emerg Infect Dis. 2011. Dec;17(12): 2223–2231. doi: 10.3201/eid1712.110395Medline [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bucak İH, Tepe B, Almiş H, Köse A, Turgut M. Joint diagnosis of pediatric and dermatology clinics: Prospective observation of thirty-nine patients with hand, foot and mouth disease (in Turkish). Turkderm-Turk Arch Dermatol Venereology. 2017;51:41–45. doi: 10.4274/turkderm.22804 [DOI] [Google Scholar]
- Centers for Disease Control and Prevention (CDC) Notes from the field: severe hand, foot, and mouth disease associated with coxsackievirus A6 – Alabama, Connecticut, California and Nevada, November 2011 – February 2012. MMWR Morb Mortal Wkly Rep. 2012. Mar 30;61(12):213–214.Medline [PubMed] [Google Scholar]
- Chen YJ, Chang SC, Tsao KC, Shih SR, Yang SL, Lin TY, Huang YC. Comparative genomic analysis of coxsackievirus A6 strains of different clinical disease entities. PLoS One. 2012. Dec 26;7(12):e52432. doi: 10.1371/journal.pone.0052432Medline [DOI] [PMC free article] [PubMed] [Google Scholar]
- Demirhan B, Demirbaş Z, Sari T, Toprak A, Navruz M. Handfoot-mouth disease in 14 years-old: A Case Report. Turk J Fam Med & Prim Care. 2016;10(1):49–51. doi: 10.5455/tjfmpc.209819 [DOI] [Google Scholar]
- Ekinci AP, Erbudak E, Baykal C. A significant increase in the frequency of hand-foot-mouth disease in Istanbul in June 2012 (in Turkish). Turkderm-Turk Arch Dermatol Venereology. 2013; 47(4):192. doi: 10.4274/turkderm.19942 [DOI] [Google Scholar]
- Fonseca MC, Sarmiento L, Resik S, Martínez Y, Hung LH, Morier L, Piñón A, Valdéz O, Kourí V, González G. Coxsackievirus A6 and enterovirus 71 causing hand, foot and mouth disease in Cuba, 2011–2013. Arch Virol. 2014. Sep;159(9):2451–2455. doi: 10.1007/s00705-014-2071-xMedline [DOI] [PubMed] [Google Scholar]
- Fujimoto T, Iizuka S, Enomoto M, Abe K, Yamashita K, Hanaoka N, Okabe N, Yoshida H, Yasui Y, Kobayashi M, et al. . Hand, foot, and mouth disease caused by coxsackievirus A6, Japan, 2011. Emerg Infect Dis. 2012. Feb;18(2):337–339. doi: 10.3201/eid1802.111147Medline [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ge S, Yan Q, He S, Zhuang S, Niu J, Xia N. Specific primer amplification of the VP1 region directed by 5’ UTR sequence analysis: enterovirus testing and identification in clinical samples from hand-foot-and-mouth disease patients. J Virol Methods. 2013. Nov;193(2):463–469. doi: 10.1016/j.jviromet.2013.06.009Medline [DOI] [PubMed] [Google Scholar]
- Han JF, Xu S, Zhang Y, Zhu SY, Wu DL, Yang XD, Liu H, Sun BX, Wu XY, Qin CF. Hand, foot, and mouth disease outbreak caused by coxsackievirus A6, China, 2013. J Infect. 2014. Sep;69(3):303–305. doi: 10.1016/j.jinf.2014.03.015Medline [DOI] [PubMed] [Google Scholar]
- Hu YQ, Xie GC, Li DD, Pang LL, Xie J, Wang P, Chen Y, Yang J, Cheng WX, Zhang Q, et al. . Prevalence of coxsackievirus A6 and enterovirus 71 in hand, foot and mouth disease in Nanjing, China in 2013. Pediatr Infect Dis J. 2015. Sep;34(9):951–957. doi: 10.1097/INF.0000000000000794Medline [DOI] [PubMed] [Google Scholar]
- Huang X, Wei H, Wu S, Du Y, Liu L, Su J, Xu Y, Wang H, Li X, Wang Y, et al. . Epidemiological and etiological characteristics of hand, foot, and mouth disease in Henan, China, 2008–2013. Sci Rep. 2015. Aug;5(1):8904. doi: 10.1038/srep08904Medline [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiratli K, Topal Y, Saat B, Kemer S, Cevik E. Hand-foot-mouth Disease in an Immunocompetent Adult. CausaPedia. 2017;6(1):1–5. [Google Scholar]
- Kliegman S, Geme SS. Nelson Textbook of Pediatrics, 2-Volume Set, 20th ed. Amsterdam (the Netherlands): Elsevier Inc.; 2015. [Google Scholar]
- Mao Q, Wang Y, Yao X, Bian L, Wu X, Xu M, Liang Z. Coxsackievirus A16: epidemiology, diagnosis, and vaccine. Hum Vaccin Immunother. 2014. Feb;10(2):360–367. doi: 10.4161/hv.27087Medline [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirand A, Henquell C, Archimbaud C, Ughetto S, Antona D, Bailly JL, Peigue-Lafeuille H. Outbreak of hand, foot and mouth disease/herpangina associated with coxsackievirus A6 and A10 infections in 2010, France: a large citywide, prospective observational study. Clin Microbiol Infect. 2012. May;18(5):E110–E118. doi: 10.1111/j.1469-0691.2012.03789.xMedline [DOI] [PubMed] [Google Scholar]
- Montes M, Artieda J, Piñeiro LD, Gastesi M, Diez-Nieves I, Cilla G. Hand, foot, and mouth disease outbreak and coxsackievirus A6, northern Spain, 2011. Emerg Infect Dis. 2013. Apr;19(4):676. doi: 10.3201/eid1904.121589Medline [DOI] [PMC free article] [PubMed] [Google Scholar]
- Österback R, Vuorinen T, Linna M, Susi P, Hyypiä T, Waris M. Coxsackievirus A6 and hand, foot, and mouth disease, Finland. Emerg Infect Dis. 2009. Sep;15(9):1485–1488. doi: 10.3201/eid1509.090438Medline [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romero JR. Hand, foot, and mouth disease and herpangina: an overview [Internet]. Waltham (USA): UpToDate; 2017. [cited 2018 Sep 12]. Available from: https://www.uptodate.com/contents/hand-foot-and-mouth-disease-and-herpangina [Google Scholar]
- Sun Z, Zhang G, Guo P, Liu J, Gao Q, Xu X, Gong L. Epidemiological characterizations, pathogen spectrum and molecular characteristics of Coxsackievirus A16 from patients with HFMD in Yantai, Shandong, China between 2011 and 2015. Hum Vaccin Immunother. 2017. Aug 03;13(8):1831–1838. doi: 10.1080/21645515.2017.1318233Medline [DOI] [PMC free article] [PubMed] [Google Scholar]
- Topkarcı Z. A clinical and demographic properties of hand-foot-mouth disease. Medical Journal of Bakirköy. 2013;9(1):12–15. doi: 10.5350/BTDMJB201309103 [DOI] [Google Scholar]
- Uğraş M, Küçük Ö, Biçer S, Çöl D, Giray T, Erdağ GÇ, Yalvaç Z, Yüce B, Vitrinel A. İki yıllık periyot içinde el-ayak-ağız hastalığı olan çocukların değerlendirilmesi. J Kartal TR. 2014;25:34–38. doi: 10.5505/jkartaltr.2014.48568 [DOI] [Google Scholar]
- Wang P, Zhao H, You F, Zhou H, Goggins WB. Seasonal modeling of hand, foot, and mouth disease as a function of meteorological variations in Chongqing, China. Int J Biometeorol. 2017. Aug; 61(8):411–1419. doi: 10.1007/s00484-017-1318-0Medline [DOI] [PubMed] [Google Scholar]
- Wei SH, Huang YP, Liu MC, Tsou TP, Lin HC, Lin TL, Tsai CY, Chao YN, Chang LY, Hsu CM. An outbreak of coxsackievirus A6 hand, foot, and mouth disease associated with onychomadesis in Taiwan, 2010. BMC Infect Dis. 2011. Dec;11(1):346. doi: 10.1186/1471-2334-11-346Medline [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Health Organization (WHO) A guide to clinical management and public health response for hand, foot and mouth disease (HFMD). Manila (Philippines):WHO Regional Office for the Western Pacific; 2011. [Google Scholar]
- Yang F, Yuan J, Wang X, Li J, Du J, Su H, Zhou B, Jin Q. Severe hand, foot, and mouth disease and coxsackievirus A6-Shenzhen, China. Clin Infect Dis. 2014. Nov 15;59(10):1504–1505. doi: 10.1093/cid/ciu624Medline [DOI] [PubMed] [Google Scholar]
- Yorulmaz A, Onat NK. Clinical and Demographic Properties of Hand-Foot and Mouth Disease. J Clin Anal Med. 2017. May 01; 8(3):219–222. doi: 10.4328/JCAM.4821 [DOI] [Google Scholar]


