Abstract
Background
The evaluation of circulating tumour DNA (ctDNA) from clinical blood samples, liquid biopsy, offers several diagnostic advantages compared with traditional tissue biopsy, such as shorter processing time, reduced patient risk and the opportunity to assess tumour heterogeneity. The historically poor sensitivity of ctDNA testing, has restricted its integration into routine clinical practice for non-metastatic disease. The early kinetics of ctDNA during radical radiotherapy for localised NSCLC have not been described with ultra-deep next generation sequencing previously.
Materials and methods
Patients with CT/PET-staged locally advanced, NSCLC prospectively consented to undergo serial venepuncture during the first week of radical radiotherapy alone. All patients received 55Gy in 20 fractions. Plasma samples were processed using the commercially available Roche AVENIO Expanded kit (Roche Sequencing Solutions, Pleasanton, CA, US) which targets 77 genes.
Results
Tumour-specific mutations were found in all patients (1 in 3 patients; 2 in 1 patient, and 3 in 1 patient). The variant allele frequency of these mutations ranged from 0.05–3.35%. In 2 patients there was a transient increase in ctDNA levels at the 72 h timepoint compared to baseline. In all patients there was a non-significant decrease in ctDNA levels at the 7-day timepoint in comparison to baseline (p = 0.4627).
Conclusion
This study demonstrates the feasibility of applying ctDNA-optimised NGS protocols through specified time-points in a small homogenous cohort of patients with localised lung cancer treated with radiotherapy. Studies are required to assess ctDNA kinetics as a predictive biomarker in radiotherapy. Priming tumours for liquid biopsy using radiation warrants further exploration.
Keywords: ctDNA, Ultra-deep NGS, Radical radiotherapy, Lung cancer
Introduction
Circulating tumour DNA (ctDNA) describes tumour-derived DNA fragments released into peripheral blood through necrosis, apoptosis and spontaneous release [1]. The term ‘liquid biopsy’ has been used to describe the evaluation of total cell-free DNA (cfDNA) from clinical blood samples, and compared with traditional tissue biopsy, liquid biopsy can be faster, less invasive and more comprehensive in terms of reflecting tumour heterogeneity. The ctDNA must be identified amongst the cfDNA produced by non-malignant cells from around the body [2]. There are conflicting data for total cell-free circulating DNA trends during treatment, and cfDNA is less useful as a prognostic biomarker [3].
The half-life of ctDNA is estimated to be up to 2 hours, and is dependent on factors including cell turnover, tumour size, excretion in bodily fluids and degradation rate by circulating nucleases [4]. Therefore, in non-metastatic cancer, concentration ranges of ctDNA, considered as fractions of total cell-free DNA, vary between tumour types, ranging from undetectable in prostate cancer [5] to 0.02–3.2% in non-small cell lung cancer (NSCLC) [6]. The historically poor sensitivity of ctDNA testing, has restricted its integration into routine clinical practice in non-metastatic disease [7].
The emerging potential clinical utilities of ctDNA in lung cancer management include screening [8], histological and molecular subtyping [9, 10], disease burden assessment [11], overall prognosis [12, 13], systemic treatment response assessment [14] (oncogenic-driven cases included [15, 16]), identification of resistance mechanisms [17] and response to local consolidative radiotherapy [18].
Although approximately 45% stage I-III NSCLC cases receive radical radiotherapy as multi- or single modality treatment [19], there is only one published series on the impact of radiotherapy on cfDNA in NSCLC [20]. Furthermore, the paucity of data on ctDNA dynamics during radiotherapy across other tumour sites means there are few transferable lessons about any possible interplay in NSCLC [20–26] As the anti-tumour activity of radiotherapy is achieved through DNA damage-mediated cell death, it is expected that an interaction will be observed. Additional evidence for transient ctDNA increases on commencing treatment may support the evaluation of radiotherapy as a preparatory procedure for liquid biopsy. This prospective pilot study aimed to demonstrate the suitability of ultra-deep NGS ctDNA quantitation for examination of the relationship between radiotherapy delivery and ctDNA dynamics in non-metastatic NSCLC.
Methods and materials
Patient selection
Patients receiving radical radiotherapy alone for locally advanced, histologically confirmed NSCLC provided consent for serial venepuncture during the first week of treatment. All patients were deemed unsuitable for concurrent chemoradiation. Routine diagnostic investigations included an 18-FDG-PET-CT for all patients, and TNM8 staging was applied [27]. Patients were approached at the radiotherapy consent clinic regarding study participation. Routine clinical assessments were recorded including ECOG PS and smoking history. Response assessments were carried out by a Consultant Radiologist with expertise in lung cancer.
Radiotherapy
All patients received 55Gy in 20 fractions over 4 weeks planned with the intensity modulated radiotherapy technique and delivered as 6MV arc therapy, with daily online cone beam-CT image guidance, treating Monday to Friday for 4 weeks. All target volumes were subject to peer review [28]. Patients were routinely clinically assessed once per week by an Oncologist or Radiographer.
Sample Collection & Processing
The time, date and location of blood draws were agreed with each patient in consideration with their radiotherapy appointments and no additional hospital visits were required as part of the study. Patients provided 20 mL of blood at 3 different time periods i) immediately prior to fraction 1, ii) 72 h after fraction 1 and iii) 7 days after fraction 1, as illustrated in the study schema in Fig. 1. Samples were transferred to the local Biobank where all blood samples were processed within a 2-h time period. Each blood sample was initially collected via a vacutainer system into 2 × 10 mL EDTA tubes and transported in ambient temperature to the dedicated Biobank laboratory. The EDTA tubes were centrifuged at 2000 g for 10 min producing 10 mL of plasma and a 1 mL buffy coat sample (which was not available for processing). The plasma was then decanted into a 15 mL conical tube and centrifuged again at 2000 g for 10 min to produce cell free plasma and frozen at − 80 °C.
Fig. 1.
Schema of plasma collection time-points. (blue arrows = venepuncture time-points)
Library Generation & Next-Generation Sequencing
Plasma samples were processed using the commercially available Roche AVENIO Expanded kit (Roche Sequencing Solutions, Pleasanton, CA, US) as per the manufacturer’s protocol. Briefly, DNA was extracted using the AVENIO cfDNA Isolation Kit. Libraries were prepared, hybridised and analysed according to the AVENIO ctDNA Analysis Kits Reagent Workflow User Guide (Version 1.1; Software Version 1.0.0). Following extraction, DNA was washed using the AVENIO cfDNA Isolation Kit and quantified using the Qubit High Sensitivity assay kit and Qubit 2.0 Fluorometer (Thermo Fisher Scientific, Massachusetts, USA). In order to assess average fragment size, the 4200 TapeStation System using the D1000 ScreenTape with D1000 Reagents (Agilent Technologies, California, USA) was used. Library preparation involved an adapter ligation, a bead clean up, PCR amplification and final bead clean up step and incubated overnight with the AVENIO ctDNA Expanded Panel (Roche Sequencing Solutions, Pleasanton, CA, US). Hybridised libraries underwent a streptavidin bead clean up, PCR amplification and a final bead clean up step before DNA quantification. The pooled libraries were diluted to 4 nM, sequenced on the Illumina NextSeq 500 (Illumina Inc., San Diego, CA, US) and data was analysed using Roche AVENIO ctDNA Analysis Software Version 1.0.0 (Roche Sequencing Solutions, Pleasanton, CA, US). This software includes bioinformatics methods from CAPP-Seq and integrated digital error suppression providing analytical sensitivity and specificity of > 99% at allele frequencies down to 0.5–1% [29]. Roche AVENIO Expanded panel targets 77 genes including single nucleotide variants (SNVs), indels, fusion genes and copy number variants (CNVs) [30]. Between 4.3 and 5 mL of plasma were used to extract cfDNA and an average of 30 ng (range 13–42 ng) of cfDNA was extracted. Sequencing yielded an average of 49,528,620 reads per sample during 300 cycles (range 41,813,828-54,599,643) and the mean unique sequencing depth was 5054x ± 1098 SD.
Statistical analysis
Statistical significance of ctDNA levels between timepoints was calculated using the non-parametric Friedman test. Whilst day 3 and day 7 measurements follow normal distribution (parametric data), baseline measurements do not (non-parametric data).
Ethics & Governance
Samples in this study were acquired from the local Biobank who have ethical approval (REC reference 16/NI/0030) for the collection and release of human tissue samples and de-identified data from consented patients [31].
Results
Clinical characteristics
Five patients aged 63–85 with histologically confirmed NSCLC stage II-III disease were enrolled onto this study between February and June 2019. Standard molecular analysis for our centre was complete in 1 of 3 applicable patients, in a case of adenocarcinoma (Table 1). All patients completed radiotherapy without experiencing grade 3 or above toxicity.
Table 1.
Clinical characteristics of patient cohort
| Patient | Age | ECOG PS | Co-morbidities | Tobacco | T-stage | N-stage | M-stage | Histology | EGFR | ALK | PD-L1 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 82 | 1 | sciatica | Ex (60 PYH) | 2a | 2 | 0 | adenocarcinoma | unk | unk | unk |
| 2 | 68 | 1 | TIA, hypothyroidism, dyslipidaemia, MS | Ex (unk) | 2a | 1 | 0 | squamous cell | N/A | N/A | 1–49% |
| 3 | 83 | 1 | macular degeneration, BPH, dyslipidaemia, HTN | Ex (unk) | 4 | 2 | 0 | adenosquamous | unk | unk | unk |
| 4 | 61 | 2 | MI, Raynaud’s | Active | 3 | 2 | 0 | squamous cell | N/A | N/A | < 1% |
| 5 | 69 | 0 | N/A | Ex (unk) | 3 | 1 | 0 | adenocarcinoma | WT | WT | > 50% |
(ECOG PS Eastern Cooperative Oncology Group Performance Status, PYH pack year history, unk unknown, WT wild-type, TIA transient ischaemic attack, MS multiple sclerosis, HTN hypertension, BPH benign prostatic hyperplasia, MI myocardial infarction, N/A not applicable)
ctDNA analysis
The median time from sample collection to processing was 25 mins (10–45), and the median time from processing to freezing was 30 mins (20–50). Potential tumour-specific mutations were found in all patients at baseline (1 in 3 patients; 2 in 1 patient, and 3 in 1 patient) and then tracked 3 days and 7 days post radiation as detailed in Table 2. The variant allele frequency (VAF) of these mutations ranged from 0.05–3.35%, consistent with somatic mutations originating in ctDNA. Mutations detectable in plasma were decreased at 7 days in all patients. In 2 patients there was a transient increase in ctDNA levels at the 72 h timepoint compared to baseline (Fig. 2). Mean ctDNA levels for all patients show a slight increase at 72 h and a decrease at day 7 in comparison to the baseline measurement. This numerical difference is not significant, p = 0.4627, based on a one-way ANOVA test.
Table 2.
amount of DNA assessed, coding and amino acid changes, sequencing depths and VAFs for each sample
| Sample ID | Isolated DNA Mass (ng) | Gene | Coding Change | Amino Acid Change | Allele Fraction | No. Mutant Molecules per mL | Unique depth |
|---|---|---|---|---|---|---|---|
| Patient 1-day 1 | 22.49 | IDH2 | c.419G > A | p.Arg140Gln | 0.17% | 2.6 | 4506 |
| Patient 1-day 3 | 23.21 | IDH2 | c.419G > A | p.Arg140Gln | 0.05% | 0.783 | 4622 |
| Patient 2-day 1 | 38.61 | TP53 | c.772G > T | p.Glu258* | 0.06% | 1.67 | 6361 |
| Patient 2-day 3 | 35.69 | Variant ND | N/A | N/A | N/A | N/A | 5625 |
| Patient 2-day 7 | 37.83 | Variant ND | N/A | N/A | N/A | N/A | 5334 |
| Patient 3-day 1 | 30.16 | EGFR | c.2573 T > G | p.Leu858Arg | 3.35% | 66.7 | 4579 |
| Patient 3-day 3 | 39.65 | EGFR | c.2573 T > G | p.Leu858Arg | 2.82% | 73.7 | 4911 |
| Patient 3-day 7 | 40.37 | EGFR | c.2573 T > G | p.Leu858Arg | 1.75% | 51.9 | 6116 |
| Patient 4-day 1 | 26.33 | TP53 | c.473G > C | p.Arg158Pro | 0.40% | 6.98 | 5630 |
| Patient 4-day 1 | 26.33 | SMAD4 | c.931C > T | p.Gln311* | 0.68% | 11.8 | 5630 |
| Patient 4-day 1 | 26.33 | BRAF | c.1780G > A | p.Asp594Asn | 1.13% | 19.7 | 5630 |
| Patient 4-day 3 | 43.42 | TP53 | c.473G > C | p.Arg158Pro | 1.00% | 28.7 | 6782 |
| Patient 4-day 3 | 43.42 | TP53 | c.434 T > C | p.Leu145Pro | 0.11% | 3.16 | 6782 |
| Patient 4-day 3 | 43.42 | SMAD4 | c.931C > T | p.Gln311* | 0.60% | 17.2 | 6782 |
| Patient 4-day 3 | 43.42 | BRAF | c.1780G > A | p.Asp594Asn | 1.85% | 53.2 | 6782 |
| Patient 4-day 7 | 32.89 | TP53 | c.473G > C | p.Arg158Pro | 0.68% | 17.1 | 5662 |
| Patient 4-day 7 | 32.89 | SMAD4 | c.931C > T | p.Gln311* | 0.56% | 14.1 | 5662 |
| Patient 4-day 7 | 32.89 | BRAF | c.1780G > A | p.Asp594Asn | 1.61% | 40.7 | 5662 |
| Patient 5-day 1 | 17.94 | TP53 | c.743G > A | p.Arg248Gln | 1.12% | 13.3 | 3806 |
| Patient 5-day 1 | 17.94 | TP53 | c.476C > T | p.Ala159Val | 0.26% | 3.05 | 3806 |
| Patient 5-day 3 | 13.13 | TP53 | c.743G > A | p.Arg248Gln | 1.56% | 13.5 | 2430 |
| Patient 5-day 7 | 33.09 | TP53 | c.743G > A | p.Arg248Gln | 1.36% | 29.7 | 5143 |
| Patient 5-day 7 | 33.09 | TP53 | c.476C > T | p.Ala159Val | 0.22% | 4.81 | 5143 |
(*ND Not detected, N/A Not applicable)
Fig. 2.
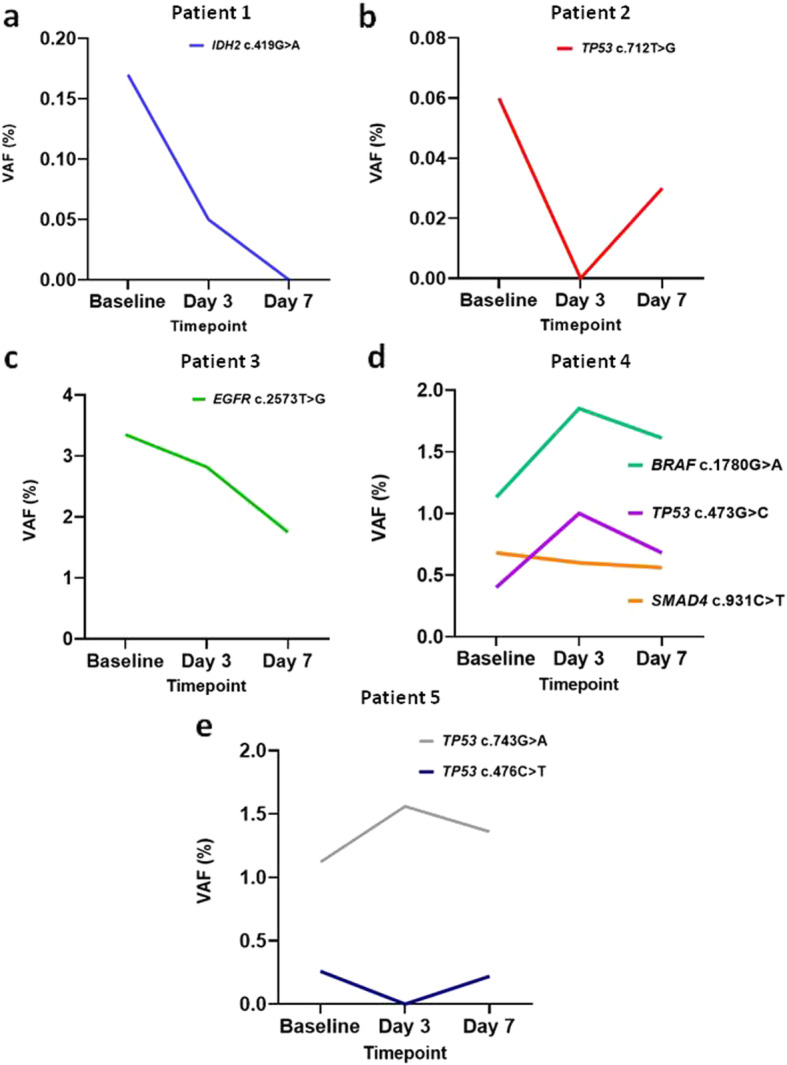
Line graphs depicting the VAF of mutations (labelled on each graph) detected in patients 1 to 5 (a-e, respectively) at baseline, 3 days and 7 days mid-treatment
Clinical outcomes
At 3 months post-treatment 4 of 5 patients were alive (Table 3). Patient 3’s tumour exhibited the least response to radiotherapy as assessed by changes in volume and the patient expired from disseminated relapse at 3 months following treatment, without accessing EGFR-targeted therapy due to poor fitness. According to volumetric assessment of the primary and nodes at 3 months confirmed a good response in all other patients. All patients had a decrease in primary tumour volume and the mean decrease was − 60% (− 8% to − 95%). Patient 4 had an increase in some nodes after radiotherapy but appearances were in keeping with cystic change (21 Hounsfield Units), and therefore a good treatment response. In Table 3, if more than one node was FDG-avid, the largest node’s size was recorded.
Table 3.
Clinical outcomes of study participants
| Patient | 6-Month ECOG PS | Baseline Volumes (cm3) | Volumes at 3 Months (cm3) | Net Change in Primary (%) | ||
|---|---|---|---|---|---|---|
| Primary | Node | Primary | Node | |||
| 1 | unk | 36.1 | 0.10 | 17.2 | 0.01 | −52% |
| 2 | unk | 8.2 | 0.06 | 3.1 | 0.00 | −62% |
| 3 | 5 | 187.4 | 0.27 | 172.0 | 0.27 | −8% |
| 4 | unk | 33.5 | 2.14 | 1.8 | 2.57 | −95% |
| 5 | 0 | 22.4 | 1.43 | 3.6 | 0.07 | − 84% |
Discussion
In recent years, liquid biopsy has obtained a place in standard clinical care for its role in selection of patients for third generation tyrosine kinase inhibitors in EGFR-mutant NSCLC after progression on first/second generation treatment, by detection of the p.T790M mutation [32]. The potential for ctDNA analysis as a predictive or prognostic biomarker continues to expand in lung cancer, but most of the clinical utility of ctDNA remains unvalidated [33].
Compared to needle tissue biopsy, liquid biopsy results in reduced risk to the patient and reduced processing time [34]. Poor sensitivity of ctDNA testing methodologies such as allele-specific PCR restricted uptake of this technique previously, however modern next-generation sequencing (NGS) can overcome this issue with sensitivities in excess of 90% reported [6].
In academic studies of metastatic lung cancer, ctDNA levels have been shown to decrease during treatment with cytotoxic [14] and targeted agents [15]. Trough values during systemic therapy have been correlated with disease-free interval and raised levels off-treatment appear to pre-empt clinical relapse by 5.2 months [35].
Similarly, following surgery ctDNA levels are known to plummet within 1 day [36]. Levels immediately post-surgery can indicate minimal residual disease detection, which may guide the clinical management [37]. Radiological relapse post-surgery has been predicted by ctDNA increases up to 5.2 months prior [38]. However, multiple studies have also shown that patients with undetectable ctDNA can develop relapse [35].
There is only one known published report of cfDNA monitoring during radiotherapy for lung cancer [20]. In this study of 17 patients, cfDNA was assessed after each quarter of the radiotherapy treatment course with digital PCR, and NGS where possible (2 cases of thoracic radiotherapy; 2 cases of cranial stereotactic radiosurgery). A net decrease in cfDNA was observed in most cases after radiotherapy, with a modest increase observed during the course of radiotherapy. The 2 patients receiving thoracic radiotherapy who had ctDNA analysis completed by NGS had no detectable ctDNA prior to treatment. After commencing treatment ctDNA became detectable and rose during the first week, followed by a subsequent decrease. This study had a heterogenous population of radiotherapy treatments for early stage primary tumours and brain metastases, and time-points for plasma sampling varied considerably. Thoracic radiotherapy was planned with a 3D-conformal technique, brain radiotherapy was completed with a stereotactic approach.
Here we report a small, prospective, observational pilot study of patients with lung cancer, where the feasibility of applying NGS ctDNA analysis in non-metastatic NSCLC patients receiving radiotherapy was demonstrated. The detected mutations were identified at baseline and subsequent time-points during radiotherapy for all cases, with values ranging from 0.06 to 3.35%. All patients in this feasibility study were found to have reduced ctDNA (ranging from 0 to 1.75%) at 7 days from commencing radiotherapy (5 fractions delivered), although the difference was not statistically significant. This is in keeping with an absolute reduction in viable tumour cells available to release DNA, or impaired DNA release processes in remaining tumour cells. Other possibilities include cellular senescence in response to sub-lethal DNA damage, and differential lethality in tumour clones prone to DNA secretion.
In keeping with this feasibility analysis, unpublished data (n = 55) presented at the ASTRO Conference 2017 examining ctDNA in stage I-III NSCLC managed with surgery, radiotherapy and chemoradiation demonstrated a decrease with treatment generally [26]. Mid-treatment and post-treatment ctDNA levels during (chemo-)radiation correlated with progression-free and overall survival. In another unpublished NSCLC cohort, approx. 40% (5/12) patients demonstrated elevated ctDNA early in the course of stereotactic radiotherapy [25].
The pilot findings are also in keeping with evaluations of head and neck cancer patients, where viral-associated DNA, considered ctDNA, appeared to decrease during the course of radical radiotherapy [23, 24]. Furthermore, subsets of patients (approx. 20%) in both studies exhibited a transient rise prior to the eventual decrease (3/14 [23] and 2/10 [24]). In addition, transient rises in ctDNA have been observed following systemic therapy, such as immunotherapy in melanoma [39], neoadjuvant combined cytotoxic/biologic combination therapy in breast carcinoma [40] and tyrosine kinase inhibition in EGFR-mutant NSCLC [41].
This pilot study demonstrates the feasibility of applying ctDNA-optimised NGS protocols through specified time-points in a small, homogenous cohort of patients treated with modern radiotherapy planning for locally advanced NSCLC. However, as the kinetics of ctDNA during radical radiotherapy have not been hitherto described in relation to lung cancer with high-quality NGS, it was not possible to optimise the plasma collection time-points in this study. It is reasonable to hypothesise that transient rises in ctDNA may have occurred within the 72 h before venepuncture, and therefore were not detected. Acknowledging ctDNA’s half-life of up to 2 h, it is also possible that the processing times achieved (median 25 min to processing from collection, median 30 min further to freezing) affected the VAF values produced. However, EDTA tubes processed within 6 h have been found to have similar performance than cell-stabilising tubes for ctDNA analysis [42].
A key weakness of our study was the inability to differentiate between clonal haematopoiesis of indeterminate potential (CHIP) [43] and tumour-related mutations, due to the lack of tumour tissue or leukocytes available for NGS. IDH2 and TP53 genes, both identified in this study, are associated with CHIP. However, given the variation in levels observed over the short study period we believe that these mutations are unlikely to reflect to CHIP in these patients. Furthermore, the frequency of TP53 mutations in this study (60%) is more in line with NSCLC (expected frequency of 60–70%) than with CHIP (expected frequency 2–5%).
Further investigation is required to address the many questions surrounding the interaction of radiotherapy with ctDNA kinetics if the full clinical utility of this technology-enabled assessment is to be realised. If ctDNA kinetics prove to have prognostic capability, surveillance strategies could be individualised for following each patient’s treatment. Early trends may be predictive for response to radiation therapy, and such a predictive biomarker could inform discussions with patients about dose-escalation/acceleration and addition of concurrent drugs. Whether the low dose bath effect of VMAT radiotherapy affects total cfDNA is not known and investigation of this is warranted. It is worth noting that cfDNA increases reduce VAF of ctDNA, given that this is a proportion, although this was not measured in our small sample set. Future investigations of ctDNA kinetics during radical radiotherapy should involve large patient cohorts powered to ensure the interpretation of their results can be justified.
In the oligometastatic setting, transient elevations in ctDNA following local consolidative radiotherapy could provide opportunity for understanding the uncontrolled tumour cell clones. In this way, a radiation-primed liquid biopsy would enable a non-invasive method of understanding mechanisms of resistance. Similarly, ongoing exploration of tumour evolution in lung cancer by the TRACERx group may create further opportunities for application of ctDNA in the future [44].
In summary this was a prospective observational pilot study of ultra-deep NGS ctDNA analysis in a cohort of stage II-III NSCLC undergoing the first week of state-of-the-art curative-intent radiotherapy. The feasibility of ctDNA analysis was shown in this small patient cohort with high-quality NGS and a larger study with a range of dose-fractionations, time-points and disease stages, including matched tissue analysis, will help addressing the clinical relevance of ctDNA monitoring during radiotherapy.
Conclusion
This pilot study of ultra-deep NGS ctDNA analysis in non-metastatic NSCLC in the first week of radical radiotherapy demonstrated the feasibility of this approach. All included patients had detectable ctDNA at baseline, and had reduced levels at 7 days. A non-significant transient ctDNA increase at 72 h preceded the decrease observed at 7 days in 2 patients, in keeping with trends in the other limited data. Such temporary increases may represent a surge of cellular lethality very early (< 1 week) in courses of radiotherapy.
Acknowledgements
Not applicable.
Availability of supporting data
All pertinent results are contained within this manuscript.
Abbreviations
- ctDNA
Circulating tumour DNA
- NSCLC
Non-small cell lung cancer
- CT/PET
Computed tomography/positron emission tomography
- NGS
Next generation sequencing
- cfDNA
Cell-free DNA
- ECOG PS
Eastern Cooperative Oncology Group Performance Status
- TNM8
Tumour-Node-Metastasis 8th Edition
- MV
Megavoltage
- EDTA
Ethylenediaminetetraacetic acid
- SNV
Single nucleotide variant
- CNV
Copy number variant
- REC
Research Ethics Committee
- VAF
Variant allele frequency
- ASTRO
American Society for Radiation Oncology
Authors’ contributions
Conception = DG and GH; study design GW, GH, DG, KS; procedures and analysis GW, PM, TL, LM; interpretation = all authors; preparation of manuscript = GW; re-drafting = all authors. The author(s) read and approved the final manuscript.
Authors’ information
GW is a Wellcome-HRB Irish Clinical Academic Training Fellow and PhD candidate. LM is a PhD candidate. JM is a Consultant Clinical Oncologist. PM is a senior Biobank Nurse. TL is a Consultant Radiologist. KS is a Senior Lecturer. GH is a clinical academic and Associate Professor. DG is a Professor of Genomic Medicine.
Funding
This study was funded by the Experimental Cancer Medicine Centre Pump-Priming fund, and by the Wellcome-HRB Irish Clinical Academic Training Programme.
Ethics approval and consent to participate
Patients were prospectively consented to undergo serial venepuncture during the first week of radical radiotherapy alone, and for their clinical data to be used in conjunction with plasma analysis.
Consent for publication
No de-anonymised patient information contained within.
Competing interests
GH wishes to declare $2000.00 in speaker bureau fee from AstraZeneca (received August 2019) – donated directly to his institution. All other authors have no conflicts of interest to declare.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
G. M. Walls, Email: g.walls@qub.ac.uk
L. McConnell, Email: lmcconnell13@qub.ac.uk
J. McAleese, Email: jonathan.mcaleese@belfasttrust.hscni.net
P. Murray, Email: paul.murray@belfasttrust.hscni.net
T. B. Lynch, Email: tom.lynch@belfasttrust.hscni.net
K. Savage, Email: k.savage@qub.ac.uk
G. G. Hanna, Email: gerry.hanna@petermac.org
D. Gonzalez de Castro, Email: d.gonzalezdecastro@qub.ac.uk.
References
- 1.Papadopoulos N. Pathophysiology of ctDNA release into the circulation and its characteristics: what is important for clinical applications BT - tumor liquid biopsies. In: Schaffner F, editor. Merlin J-L, von Bubnoff N, editors. Cham: Springer International Publishing; 2020. pp. 163–180. [DOI] [PubMed] [Google Scholar]
- 2.Ignatiadis M, Lee M, Jeffrey SS. Circulating tumor cells and circulating tumor DNA: challenges and opportunities on the path to clinical utility. Clin Cancer Res. 2015;21(21):4786–4800. doi: 10.1158/1078-0432.CCR-14-1190. [DOI] [PubMed] [Google Scholar]
- 3.Lee YJ, Yoon KA, Han JY, Kim HT, Yun T, Lee GK, et al. Circulating cell-free DNA in plasma of never smokers with advanced lung adenocarcinoma receiving gefitinib or standard chemotherapy as first-line therapy. Clin Cancer Res. 2011;17(15):5179–5187. doi: 10.1158/1078-0432.CCR-11-0400. [DOI] [PubMed] [Google Scholar]
- 4.Diehl F, Schmidt K, Choti MA, Romans K, Goodman S, Li M, et al. Circulating mutant DNA to assess tumor dynamics. Nat Med. 2008;14(9):985–990. doi: 10.1038/nm.1789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hennigan ST, Trostel SY, Terrigino NT, Voznesensky OS, Schaefer RJ, Whitlock NC, et al. Low abundance of circulating tumor DNA in localized prostate Cancer. JCO Precis Oncol. 2019;3:1–13. doi: 10.1200/PO.19.00176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Newman AM, Bratman SV, To J. Wynne JF, Eclov NCW, Modlin LA, et al. An ultrasensitive method for quantitating circulating tumor DNA with broad patient coverage. Nat Med. 2014;20(5):548–554. doi: 10.1038/nm.3519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Herbreteau G, Vallée A, Charpentier S, Normanno N, Hofman P, Denis MG. Circulating free tumor DNA in non-small cell lung cancer (NSCLC): clinical application and future perspectives. J Thorac Dis. 2019;11(Suppl 1):S113–S126. doi: 10.21037/jtd.2018.12.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cohen JD, Li L, Wang Y, Thoburn C, Afsari B, Danilova L, et al. Detection and localization of surgically resectable cancers with a multi-analyte blood test. Science (80- ). 2018;359(6378):926–930. [DOI] [PMC free article] [PubMed]
- 9.Zhang B, Niu X, Zhang Q, Wang C, Liu B, Yue D, et al. Circulating tumor DNA detection is correlated to histologic types in patients with early-stage non-small-cell lung cancer. Lung Cancer. 2019;134(December 2018):108–116. doi: 10.1016/j.lungcan.2019.05.034. [DOI] [PubMed] [Google Scholar]
- 10.Sabari JK, Offin M, Stephens D, Ni A, Lee A, Pavlakis N, et al. A prospective study of circulating tumor DNA to guide matched targeted therapy in lung cancers. J Natl Cancer Inst. 2019;111(6):575–583. doi: 10.1093/jnci/djy156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schweizer MT, Gulati R, Beightol M, Konnick EQ, Cheng HH, Klemfuss N, et al. Clinical determinants for successful circulating tumor DNA analysis in prostate cancer. Prostate. 2019;79(7):701–708. doi: 10.1002/pros.23778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tissot C, Toffart AC, Villar S, Souquet PJ, Merle P, Moro-Sibilot D, et al. Circulating free DNA concentration is an independent prognostic biomarker in lung cancer. Eur Respir J. 2015;46(6):1773–1780. doi: 10.1183/13993003.00676-2015. [DOI] [PubMed] [Google Scholar]
- 13.Lv J, Chen Y, Zhou G, Qi Z, Tan KRL, Wang H, et al. Liquid biopsy tracking during sequential chemo-radiotherapy identifies distinct prognostic phenotypes in nasopharyngeal carcinoma. Nat Commun. 2019;10(1):1–10. doi: 10.1038/s41467-019-11853-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kumar S, Guleria R, Singh V, Bharti AC, Mohan A, Das BC. Plasma DNA level in predicting therapeutic efficacy in advanced nonsmall cell lung cancer. Eur Respir J. 2010;36(4):885–892. doi: 10.1183/09031936.00187909. [DOI] [PubMed] [Google Scholar]
- 15.Ding PN, Becker TM, Bray VJ, Chua W, Ma YF, Lynch D, et al. The predictive and prognostic significance of liquid biopsy in advanced epidermal growth factor receptor-mutated non-small cell lung cancer: a prospective study. Lung Cancer. 2019;134(June):187–193. doi: 10.1016/j.lungcan.2019.06.021. [DOI] [PubMed] [Google Scholar]
- 16.Ortiz-Cuaran S, Mezquita L, Swalduz A, Aldea M, Mazieres J, Jovelet C, et al. MA21.07 Circulating Tumor DNA Analysis Depicts Potential Mechanisms of Resistance to BRAF-Targeted Therapies in BRAF+ Non-Small Cell Lung Cancer. J Thorac Oncol. 2019;14(10):S337. [Google Scholar]
- 17.Del Re M, Crucitta S, Gianfilippo G, Passaro A, Petrini I, Restante G, et al. Understanding the mechanisms of resistance in EGFR-positive NSCLC: from tissue to liquid biopsy to guide treatment strategy. Int J Mol Sci. 2019;20(16):3951. doi: 10.3390/ijms20163951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tang C, Lee W-C, Reuben A, Chang L, Tran H, Little L, et al. Immune and circulating tumor DNA profiling following radiation treatment for oligometastatic NSCLC; translational correlatives from a mature randomized phase II trial. Int J Radiat Oncol Biol Phys. 2019. [DOI] [PubMed]
- 19.National Lung Cancer Audit. National Lung Cancer Audit Report 2018. (for the audit period 2017). 2019.
- 20.Kageyama S, Nihei K, Karasawa K, Sawada T, Koizumi F, Yamaguchi S, et al. Radiotherapy increases plasma levels of tumoral cell-free DNA in non-small cell lung cancer patients. Oncotarget. 2018;9(27):19368–19378. doi: 10.18632/oncotarget.25053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Leung SF, Chan KCA, Ma BB, Hui EP, Mo F, Chow KCK, et al. Plasma Epstein-Barr viral DNA load at midpoint of radiotherapy course predicts outcome in advanced-stage nasopharyngeal carcinoma. Ann Oncol. 2014;25(6):1204–1208. doi: 10.1093/annonc/mdu117. [DOI] [PubMed] [Google Scholar]
- 22.Lin JC, Wang WY, Chen KY, Wei YH, Liang WM, Jan JS, et al. Quantification of plasma Epstein-Barr virus DNA in patients with advanced nasopharyngeal carcinoma. N Engl J Med. 2004;350(24):2461–2470. doi: 10.1056/NEJMoa032260. [DOI] [PubMed] [Google Scholar]
- 23.Cao H, Banh A, Kwok S, Shi X, Wu S, Krakow TC, et al. Quantitation of the human papillomavirus DNA in the plasma of patients with Oropharyngeal carcinoma. Int J Radiat Oncol Biol Phys Int J Radiat Oncol Biol Phys March. 2012;1(823):351–358. doi: 10.1016/j.ijrobp.2011.05.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lo YMD, Leung S-F, Chan LYS, Chan ATC, Lo K-W, Johnson PJ, et al. Kinetics of plasma Epstein-Barr virus DNA during radiation therapy for. Cancer Res. 2000;60(9):2351–2355. [PubMed] [Google Scholar]
- 25.Chen EL, Chaudhuri AA, Nabet BY, Chabon JJ, Merriott DJ, Loo BW, et al. Analysis of circulating tumor DNA kinetics during stereotactic ablative radiation therapy for non-small cell lung Cancer. Int J Radiat Oncol. 2018;102(3):e676. [Google Scholar]
- 26.Chaudhuri AA, Lovejoy AF, Chabon JJ, Newman A, Stehr H, Merriott DJ, et al. Circulating tumor DNA analysis during radiation therapy for localized lung Cancer predicts treatment outcome. Int J Radiat Oncol. 2017;99(2):S1–S2. [Google Scholar]
- 27.Detterbeck FC. The eighth edition TNM stage classification for lung cancer: what does it mean on main street? J Thorac Cardiovasc Surg. 2018;155(1):356–359. doi: 10.1016/j.jtcvs.2017.08.138. [DOI] [PubMed] [Google Scholar]
- 28.Rooney KP, McAleese J, Crockett C, Harney J, Eakin RL, Young VAL, et al. The impact of colleague peer review on the radiotherapy treatment planning process in the radical treatment of lung cancer. Clin Oncol (R Coll Radiol) 2015;27(9):514–518. doi: 10.1016/j.clon.2015.05.010. [DOI] [PubMed] [Google Scholar]
- 29.Jiang J, Adams H, Yao L, Froehler S, Fang LT, Oster O, et al. Concordance of Genomic Alterations by Next Generation Sequencing ( NGS ) in Tumor Tissue vs . Cell-Free DNA in Stage I-IV NSCLC. J Mol Diagnostics. 2019;2. [DOI] [PubMed]
- 30.Roche Sequencing Solutions. AVENIO ctDNA Expanded Kit Product Specification [Internet]. Available from: https://sequencing.roche.com/en/products-solutions/by-category/assays/ctdna-analysis-kits/ctdna-expanded-kits/specifications.html. Accessed 27 May 2020.
- 31.Lewis C, McQuaid S, Clark P, Murray P, McGuigan T, Greene C, et al. The Northern Ireland biobank: a Cancer focused repository of science. Open J Bioresour. 2018;5:1–6. [Google Scholar]
- 32.Jenkins S, Yang JCH, Ramalingam SS, Yu K, Patel S, Weston S, et al. Plasma ctDNA analysis for detection of the EGFR T790M mutation in patients with advanced non–small cell lung Cancer. J Thorac Oncol. 2017;12(7):1061–1070. doi: 10.1016/j.jtho.2017.04.003. [DOI] [PubMed] [Google Scholar]
- 33.Siravegna G, Mussolin B, Venesio T, Marsoni S, Seoane J, Dive C, et al. How liquid biopsies can change clinical practice in oncology. Ann Oncol. 2019;(August):1–11. [DOI] [PubMed]
- 34.Loong HH, Raymond VM, Shiotsu Y, Chua DTT, Teo PML, Yung T, et al. Clinical application of genomic profiling with circulating tumor DNA for Management of Advanced non–Small-cell Lung Cancer in Asia. Clin Lung Cancer. 2018;19(5):e601–e608. doi: 10.1016/j.cllc.2018.04.022. [DOI] [PubMed] [Google Scholar]
- 35.Chaudhuri AA, Chabon JJ, Lovejoy AF, Newman AM, Stehr H, Azad TD, et al. Early detection of molecular residual disease in localized lung cancer by circulating tumor DNA profiling. Cancer Discov. 2017;7(12):1394–1403. doi: 10.1158/2159-8290.CD-17-0716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Guo N, Lou F, Ma Y, Li J, Yang B, Chen W, et al. Circulating tumor DNA detection in lung cancer patients before and after surgery. Sci Rep. 2016;6(January):1–8. doi: 10.1038/srep33519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chae YK, Oh MS. Detection of minimal residual disease using ctDNA in lung Cancer: current evidence and future directions. J Thorac Oncol. 2019;14(1):16–24. doi: 10.1016/j.jtho.2018.09.022. [DOI] [PubMed] [Google Scholar]
- 38.Hu W, Yang Y, Zhang L, Yin J, Huang J, Huang L, et al. Post surgery circulating free tumor DNA is a predictive biomarker for relapse of lung cancer. Cancer Med. 2017;6(5):962–974. doi: 10.1002/cam4.980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Xi L, Pham THT, Payabyab EC, Sherry RM, Rosenberg SA, Raffeld M. Circulating tumor DNA as an early indicator of response to T-cell transfer immunotherapy in metastatic melanoma. Clin Cancer Res. 2016;22(22):5480–5486. doi: 10.1158/1078-0432.CCR-16-0613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Butler TM, Boniface CT, Johnson-Camacho K, Tabatabaei S, Melendez D, Kelley T, et al. Circulating tumor DNA dynamics using patient-customized assays are associated with outcome in neoadjuvantly treated breast cancer. Cold Spring Harb Mol case Stud. 2019;5(2):1–18. doi: 10.1101/mcs.a003772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kato K, Uchida J, Kukita Y, Kumagai T, Nishino K, Inoue T, et al. Transient appearance of circulating tumor DNA associated with de novo treatment. Sci Rep. 2016;6:1–8. doi: 10.1038/srep38639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kang Q, Henry NL, Paoletti C, Jiang H, Vats P, Chinnaiyan AM, et al. Comparative analysis of circulating tumor DNA stability In K3EDTA, Streck, and CellSave blood collection tubes. Clin Biochem. 2016;49(18):1354–1360. doi: 10.1016/j.clinbiochem.2016.03.012. [DOI] [PubMed] [Google Scholar]
- 43.Razavi P, Li BT, Brown DN, Jung B, Hubbell E, Shen R, et al. High-intensity sequencing reveals the sources of plasma circulating cell-free DNA variants. Nat Med. 2019;25(12):1928–1937. doi: 10.1038/s41591-019-0652-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Abbosh C, Birkbak NJ, Wilson GA, Jamal-Hanjani M, Constantin T, Salari R, et al. Phylogenetic ctDNA analysis depicts early-stage lung cancer evolution. Nature. 2017;545(7655):446–451. doi: 10.1038/nature22364. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All pertinent results are contained within this manuscript.


