Abstract
Although rotavirus infection is usually acute and self-limiting, it can cause chronic infection with severe diseases in immunocompromised patients, including organ transplantation recipients and cancer patients irrespective of pediatric or adult patients. Since no approved medication against rotavirus infection is available, this study screened a library of safe-in-man broad-spectrum antivirals. We identified gemcitabine, a widely used anti-cancer drug, as a potent inhibitor of rotavirus infection. We confirmed this effect in 2D cell cultures and 3D cultured human intestinal organoids with both laboratory-adapted rotavirus strains and five clinical isolates. Supplementation of UTP or uridine largely abolished the anti-rotavirus activity of gemcitabine, suggesting its function through inhibition of pyrimidine biosynthesis pathway. Our results support repositioning of gemcitabine for treating rotavirus infection, especially for infected cancer patients.
Keywords: Broad-spectrum antiviral agents, Gemcitabine, Rotavirus, Human intestinal organoids, Pyrimidine biosynthesis pathway
Highlights
-
•
Gemcitabine, a widely used anti-cancer drug, has potent antiviral activity against rotavirus infection.
-
•
The antiviral effect of gemcitabine has been confirmed with both laboratory-adapted strains and clinical isolates.
-
•
Gemcitabine exerts its anti-rotavirus effect through inhibiting pyrimidine biosynthesis pathway.
Rotavirus infection is the leading cause of severe dehydrating gastroenteritis among children under five-year-old (Greenberg and Estes, 2009). Although rotavirus infection is usually acute and self-limiting, it can cause chronic infection with severe diseases in immunocompromised patients, in particular organ transplantation recipients irrespective of pediatric or adult patients (Yin et al., 2015b). In addition, cancer patients have compromised immune system especially when undergoing chemotherapy or radiotherapy treatment, which make them prone to infections with worse outcomes (Hotchkiss and Moldawer, 2014). Rotavirus infections have been widely reported in pediatric or adult cancer patients causing prolonged diarrhea (Akhtar et al., 2018; Ghosh et al., 2017). Therefore, specific and effective antiviral treatment is urgently needed for these special populations who are infected with rotavirus, but there are no FDA-approved medications available against rotavirus infection.
Developing new drugs usually takes more than ten years with enormous investment and high risk of failure. Given that only the specific population with rotavirus infection require antiviral treatment, the pharmaceutical industry will likely not develop new anti-rotavirus drugs. We propose that repurposing existing drugs represents a cost-effective approach to identify antiviral treatment that can readily benefit patients (Qu et al., 2019). In this study, we screened a library of safe-in-man broad-spectrum antiviral agents (BSAAs, https://drugvirus.info) (Andersen et al., 2020; Ianevski et al., 2018) on rotavirus infection in experimental models. These compounds are known to target viruses belonging to two or more viral families and have passed phase 1 clinical trials. This greatly enhances the probability of identifying novel activities of some of these agents against rotavirus infection and facilitates their clinical translation.
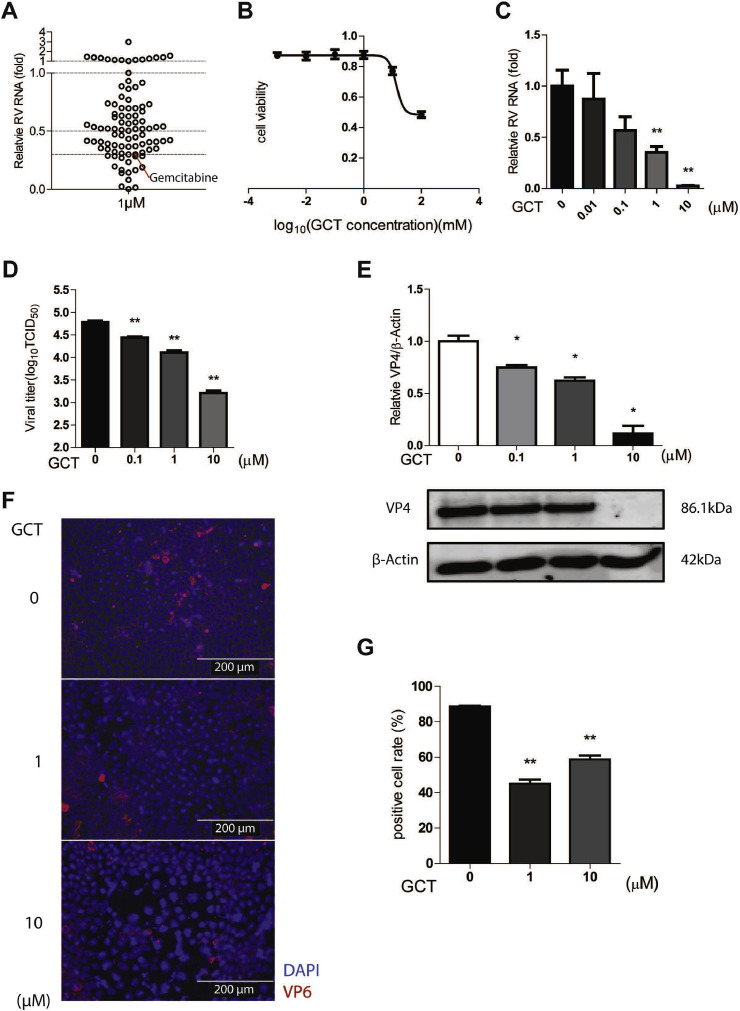
We first screened 94 BSAAs in human intestinal Caco2 cell line infected with simian rotavirus SA11 strain (Table S1). To minimize non-specific effects on host cells, we used low concentration of 1 μM and treated for 48 h. By qRT-PCR (primers listed in Table S2) quantification of rotavirus genomic RNA, we identified 43 candidates exerting over 50% inhibitory effects, and 17 with inhibition over 70% (Fig. 1 A). Among these, gemcitabine was one of the most effective candidates (Fig. 1A). It is a cytidine analog that has been widely used for cancer treatment (Cerqueira et al., 2007; Zhang et al., 2019). It has been shown to inhibit a broad range of RNA viruses including severe acute respiratory syndrome coronavirus (SARS-CoV), Middle East respiratory syndrome coronavirus (MERS-CoV), Zika virus and hepatitis C virus (HCV) in experimental models (Beran et al., 2012; Dyall et al., 2014; Kuivanen et al., 2017). Nucleotide and nucleoside analogues are excellent examples of BSAAs that have been widely used in the clinic for treating infections of RNA and DNA viruses (Ianevski et al., 2018). We have previously demonstrated that nucleotide, including purine and pyrimidine, biosynthesis pathways are essential in regulating rotavirus infection and can be pharmacologically targeted (Chen et al., 2019; Yin et al., 2016, 2018a). As a cytidine analog, gemcitabine has been reported to inhibit pyrimidine biosynthesis, resulting in nucleotide depletion (Lee et al., 2017). Therefore, we focused on the effects and mode-of-action of gemcitabine on rotavirus infection in this study.
Fig. 1.
Screening BSAAs identified gemcitabine as a potent inhibitor against rotavirus infection. (A) Human intestinal Caco2 cells were infected with SA11 rotavirus. After infection, cells were treated with 94 BSAAs at 1 μM for 48 h qRT-PCR analysis of viral RNA revealed that 43 reagents exerted ≥50% inhibitory activity, and 17 of them exerted ≥70% inhibitory activity against rotavirus replication. Gemcitabine (GCT) inhibited rotavirus replication by 70%. (B) 50% cytotoxic concentration (CC50) curves of GCT were determined by MTT assays on Caco2 cell line. (C) Dose-dependent inhibitory activity of GCT on rotavirus infected Caco2 cell line. qRT-PCR data were normalized to housekeeping gene GAPDH and presented relative to the control (CTR) (set as 1) (n = 6). (D) The supernatant of each well under GCT treatment was harvested after freezing and thawing for three times, virus titer from different groups was measured by TCID50 assay (n = 6). (E) Rotavirus infected Caco2 cells were treated by different concentrations of GCT, and protein samples were harvested after 48 h. The expression of viral structural protein VP4 was stained and quantified by Western blot assay (n = 4). (F) Indirect fluorescence microscope analysis of viral structural protein VP6 (red) upon treatment with GCT. Nuclei were visualized by DAPI (blue). (G) The ratio of VP6 positive cells/total cell number was quantified under 40* vision field (Supplementary Fig. S2) (n = 6). Data represent means ± SEM. *P < 0.05; **P < 0.01; ***P < 0.001.
We next tested a series of gemcitabine concentrations (0.01–10 μM) in the Caco2 cell model to assess both antiviral and cytotoxic effects. We confirmed the potent anti-rotavirus effect and observed a large therapeutic window between cytotoxic and antiviral activities, as shown the CC50 value of 13.58 mM and IC50 value of 0.12 μM (Fig. 1B and C). We performed same experiments in the monkey MA104 cell line that is widely used for propagating rotavirus in laboratory. Similar trends were observed with CC50 value of 0.36 mM and IC50 value of 3.98 μM (Supplementary Fig. S1). By harvesting supernatant of Caco2 cells at 48 h post-treatment, we performed TCID50 assay to determine the titers of secreted viruses. Consistently, the titers of produced rotavirus with infectivity were significantly reduced by gemcitabine treatment (Fig. 1D). At protein level, we found potent inhibition of viral protein 4 (VP4) expression determined by Western blotting (Fig. 1E). Furthermore, immunofluorescent staining of viral capsid protein (VP6) showed significant reduction of the number of infected Caco2 cells by gemcitabine treatment (Fig. 1F and G, Supplementary Fig. S2).
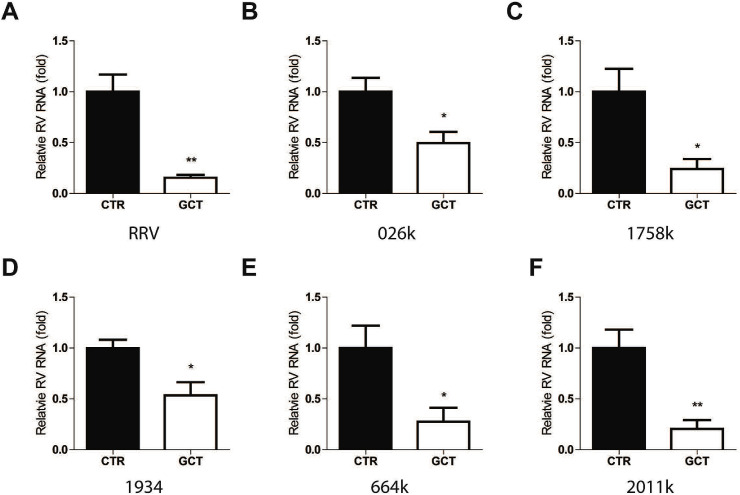
The responsiveness to antiviral therapy can vary dramatically in patients. This mainly attributes to host and viral factors. The nucleoside analog ribavirin has been used for treating HCV infection for decades. Reduced cellular uptake by the host and mutagenesis of the viral genome have been linked to treatment resistance in chronic HCV patients (Ibarra et al., 2011). As recently repositioned for treating chronic hepatitis E virus (HEV) infection (Kamar et al., 2017), viral mutagenesis during ribavirin treatment or pre-deposition of resistance mutations are thought to contribute to treatment failure (Ikram et al., 2018). Our previous studies have evaluated the effects of the nucleoside analog ribavirin and mycophenolic acid (MPA), and the antiviral cytokine interferon alpha (IFN-α) on rotavirus in cell culture models. We found the responsiveness to these agents dramatically vary among different clinical isolates from potent, moderate, minimal to even pro-viral effects (Yin et al., 2015a, 2016). This imposes major challenges for clinical application as how to personalize the selection of potential responders. Thus, we extended our evaluation of gemcitabine to different rotavirus strains/isolates. We found that gemcitabine significantly inhibits the replication of rhesus rotavirus RRV strain (Fig. 2 A) and five clinical isolates (Fig. 2B–F). Based on these results, we postulate that the response to gemcitabine in rotavirus patients could be universally effective.
Fig. 2.
Antiviral effect of gemcitabine against RRV rotavirus strain and clinical isolates. qRT-PCR analysis of rotavirus RNA upon treatment of gemcitabine (GCT) at 1 μM for 48 h in laboratory rotavirus strain RRV (A), clinically isolated rotavirus strain 026k (B), 1758k (C), 1934 (D), 664k (E), and 2011k (F). Data were normalized to housekeeping gene GAPDH and are presented relative to the control (CTR) (set as 1). Data represent means ± SEM; n = 6; *P < 0.05; **P < 0.01.
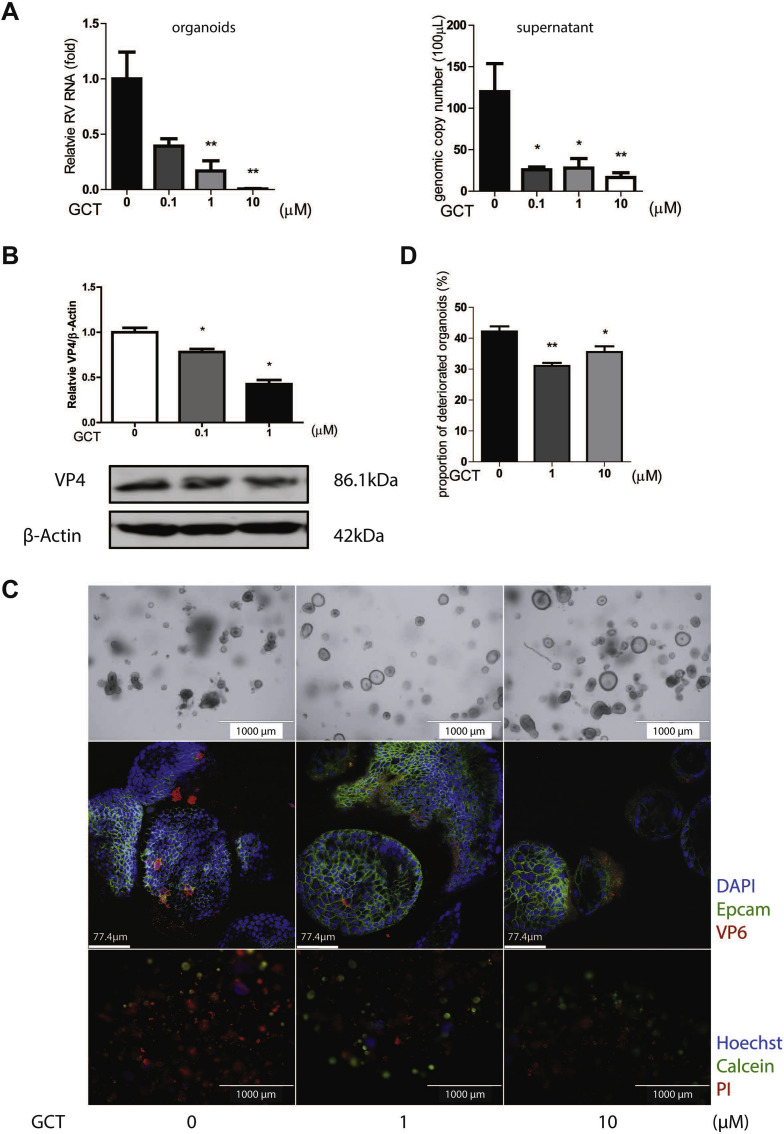
We have previously established modeling of rotavirus infection in intestinal organoids, which allows the study of virus-host interactions and assessment of antiviral drugs (Yin et al., 2015a, 2016, 2018a, 2018b). Intestinal organoids, also called mini-gut, are stem cells-derived epithelial cultured in 3D structure. These organoids are much better in recapitulating the architecture, composition, diversity, organization and functionality of cell types of the intestine. Treatment with gemcitabine in rotavirus inoculated human intestinal organoids potently inhibited viral RNA synthesis and secretion of rotaviruses with approximately 80% inhibitory effects for both at 1 μM concentration treated for 48 h (Fig. 3 A; Supplementary Fig. S3). This effect was further confirmed at VP4 protein level by Western blot assay (Fig. 3B). Importantly, rotavirus infection led to morphological and pathological changes in organoids. Based on optical imaging, we observed that rotavirus infected organoids without gemcitabine treatment showed opaque, wizened and disorganized morphology. In contrast, gemcitabine treated groups, in particular with 1 μM concentration, most of the organoids were hyaline and in a spheroidal shape (Fig. 3C upper panel). In confocal immunostaining, VP6 protein was detected in all groups, but the intensity and frequency of the viral protein were lower in treatment groups (Fig. 3C middle panel). Fluorescence staining of cell viability by Propidium Iodide (PI) showed that cell death (determined as described in Supplementary Fig. S4) in un-treated group is more obvious, and dead cells were diffused in almost all organoids (Fig. 3C lower panel). This is consistent with the percentage of deteriorated organoids (Fig. 3D). Thus, inhibition of rotavirus replication by gemcitabine can protect organoids from rotavirus induced cytopathogenesis.
Fig. 3.
Antiviral activity of gemcitabine against rotavirus SA11 in human intestinal organoids. (A) Human intestinal organoids (HIOs) were inoculated with SA11 rotavirus strain and treated with gemcitabine (GCT) for 48 h. The relative cellular rotavirus RNA and secreted viruses calculated as genomic copy number were analyzed by qRT-PCR (n = 6). Standard curve for calculation of genomic copy number is included in Supplementary Fig. S3. (B) The expression of viral structural protein VP4 was stained and quantified by Western blot assay (n = 4). (C) Optical microscopy images, confocal images and indirect fluorescence microscope images of HIOs from mock group and GCT treated groups. In confocal images, VP6 protein was stained in red, green signal represents Epcam, and nuclei were visualized by DAPI (blue). In fluorescence microscope images, HIOs were stained by PI (red) indicating dead cells, Hoechst (blue) for nuclear, and Calcein (green) as live cells. (D) Rate of deteriorated HIOs was calculated as dead organoids/total organoids (n = 6). Data represent means ± SEM; *P < 0.05; **, P < 0.01.
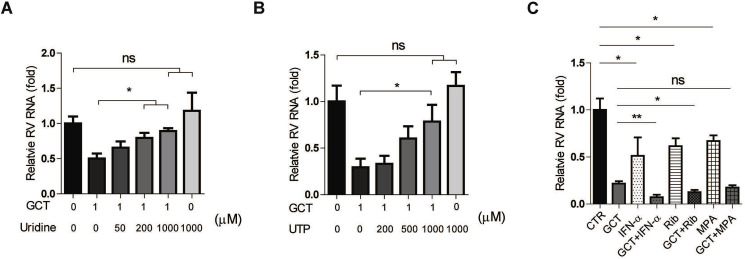
The antiviral activity of gemcitabine has been linked to the inhibition of pyrimidine biosynthesis pathway, especially in salvage pathway (Lee et al., 2017). We thus supplemented UTP or uridine to rotavirus infected Caco2 cell model when treated with gemcitabine. We found that exogenous supplementation of pyrimidine dose-dependently abolished the anti-rotavirus effect of gemcitabine. The anti-rotavirus effect was almost completely abolished by addition of 1000 μM UTP or uridine (Fig. 4 A and B). Hence, the anti-rotavirus activity of gemcitabine was largely dependent on the salvage pyrimidine biosynthesis pathway. Combination approaches are often used in clinic to achieve optimal antiviral efficacy and avoid resistance development. We finally assessed the combinatory effects of gemcitabine with ribavirin, MPA or IFN-α, as we previously have demonstrated the anti-rotavirus effects of these three agents (Yin et al., 2015a, 2016). We found enhanced anti-rotavirus activity when combined with ribavirin or IFN-α, but not MPA (Fig. 4C).
Fig. 4.
The effects of nucleotide supplementation and combination with other antivirals on the anti-rotavirus activity of gemcitabine. Supplementation of exogenous uridine (A) and UTP (B) attenuates the anti-rotavirus effect of gemcitabine (GCT) in Caco2 cell model (n = 6). (C) The combinatory effects of 1 μM GCT with 1000 U IFN-α, 1 μg/ml ribavirin (Rib) or 0.1 μg/ml mycophenolic acid (MPA), respectively (n = 6). Data represent means ± SEM; *P < 0.05; **P < 0.01.
In summary, we have identified gemcitabine as a potent inhibitor against rotavirus infection through screening of a BSAA library. The antiviral activity was largely dependent on the pyrimidine biosynthesis pathway. Because gemcitabine has been widely used as chemotherapy for cancer patients, and these patients are at risk of infections (Hotchkiss and Moldawer, 2014). The use of gemcitabine in cancer patients probably mitigates the risk of viral infections in general. Our results support repositioning of gemcitabine for treating rotavirus infection, especially in infected cancer patients. Furthermore, the approach of discovering new antiviral therapy from BSAAs bears essential implications in combating emerging viral pathogens, such as the ongoing coronavirus disease COVID-19 pandemic (Mahase, 2020).
Grant support
This research was supported by a VIDI grant (No. 91719300) from the Netherlands Organization for Scientific Research (NWO) to Q. Pan, and the China Scholarship Council for funding Ph.D. fellowships (201606760056 to S.C., 201808370170 to P.L., 201903250082 to Y.W.), the European Regional Development Fund for the Mobilitas Pluss Project MOBTT39 (to D.K.) and a grant from the Dutch Society for the Replacement of Animal Testing and ZONMW (11 4021 502) to M. Peppelenbosch.
Declaration of competing interest
The authors declare that they have no competing interests.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.antiviral.2020.104823.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- Akhtar T., Cargill J., Gerrard C., Shaw F., Cunliffe N.A., Cooke R.P.D., Pizer B. Detection of rotavirus in paediatric oncology patients with diarrhoea: the impact of rotavirus vaccine. J. Hosp. Infect. 2018;99:185–187. doi: 10.1016/j.jhin.2017.12.022. [DOI] [PubMed] [Google Scholar]
- Andersen P.I., Ianevski A., Lysvand H., Vitkauskiene A., Oksenych V., Bjoras M., Telling K., Lutsar I., Dampis U., Irie Y., Tenson T., Kantele A., Kainov D.E. Discovery and development of safe-in-man broad-spectrum antiviral agents. Int. J. Infect. Dis. 2020;93:268–276. doi: 10.1016/j.ijid.2020.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beran R.K., Sharma R., Corsa A.C., Tian Y., Golde J., Lundgaard G., Delaney W.E.t., Zhong W., Greenstein A.E. Cellular growth kinetics distinguish a cyclophilin inhibitor from an HSP90 inhibitor as a selective inhibitor of hepatitis C virus. PloS One. 2012;7 doi: 10.1371/journal.pone.0030286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cerqueira N.M., Fernandes P.A., Ramos M.J. Understanding ribonucleotide reductase inactivation by gemcitabine. Chemistry. 2007;13:8507–8515. doi: 10.1002/chem.200700260. [DOI] [PubMed] [Google Scholar]
- Chen S., Ding S., Yin Y., Xu L., Li P., Peppelenbosch M.P., Pan Q., Wang W. Suppression of pyrimidine biosynthesis by targeting DHODH enzyme robustly inhibits rotavirus replication. Antivir. Res. 2019;167:35–44. doi: 10.1016/j.antiviral.2019.04.005. [DOI] [PubMed] [Google Scholar]
- Dyall J., Coleman C.M., Hart B.J., Venkataraman T., Holbrook M.R., Kindrachuk J., Johnson R.F., Olinger G.G., Jr., Jahrling P.B., Laidlaw M., Johansen L.M., Lear-Rooney C.M., Glass P.J., Hensley L.E., Frieman M.B. Repurposing of clinically developed drugs for treatment of Middle East respiratory syndrome coronavirus infection. Antimicrob. Agents Chemother. 2014;58:4885–4893. doi: 10.1128/AAC.03036-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh N., Malik F.A., Daver R.G., Vanichanan J., Okhuysen P.C. Viral associated diarrhea in immunocompromised and cancer patients at a large comprehensive cancer center: a 10-year retrospective study. Infect. Dis. (Lond.) 2017;49:113–119. doi: 10.1080/23744235.2016.1224384. [DOI] [PubMed] [Google Scholar]
- Greenberg H.B., Estes M.K. Rotaviruses: from pathogenesis to vaccination. Gastroenterology. 2009;136:1939–1951. doi: 10.1053/j.gastro.2009.02.076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hotchkiss R.S., Moldawer L.L. Parallels between cancer and infectious disease. N. Engl. J. Med. 2014;371:380–383. doi: 10.1056/NEJMcibr1404664. [DOI] [PubMed] [Google Scholar]
- Ianevski A., Zusinaite E., Kuivanen S., Strand M., Lysvand H., Teppor M., Kakkola L., Paavilainen H., Laajala M., Kallio-Kokko H., Valkonen M., Kantele A., Telling K., Lutsar I., Letjuka P., Metelitsa N., Oksenych V., Bjoras M., Nordbo S.A., Dumpis U., Vitkauskiene A., Ohrmalm C., Bondeson K., Bergqvist A., Aittokallio T., Cox R.J., Evander M., Hukkanen V., Marjomaki V., Julkunen I., Vapalahti O., Tenson T., Merits A., Kainov D. Novel activities of safe-in-human broad-spectrum antiviral agents. Antivir. Res. 2018;154:174–182. doi: 10.1016/j.antiviral.2018.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ibarra K.D., Jain M.K., Pfeiffer J.K. Host-based ribavirin resistance influences hepatitis C virus replication and treatment response. J. Virol. 2011;85:7273–7283. doi: 10.1128/JVI.00629-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikram A., Hakim M.S., Zhou J.H., Wang W., Peppelenbosch M.P., Pan Q. Genotype-specific acquisition, evolution and adaptation of characteristic mutations in hepatitis E virus. Virulence. 2018;9:121–132. doi: 10.1080/21505594.2017.1358349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamar N., Wang W., Dalton H.R., Pan Q. Direct-acting antiviral therapy for hepatitis E virus? Lancet Gastroenterol. Hepatol. 2017;2:154–155. doi: 10.1016/S2468-1253(16)30242-4. [DOI] [PubMed] [Google Scholar]
- Kuivanen S., Bespalov M.M., Nandania J., Ianevski A., Velagapudi V., De Brabander J.K., Kainov D.E., Vapalahti O. Obatoclax, saliphenylhalamide and gemcitabine inhibit Zika virus infection in vitro and differentially affect cellular signaling, transcription and metabolism. Antivir. Res. 2017;139:117–128. doi: 10.1016/j.antiviral.2016.12.022. [DOI] [PubMed] [Google Scholar]
- Lee K., Kim D.E., Jang K.S., Kim S.J., Cho S., Kim C. Gemcitabine, a broad-spectrum antiviral drug, suppresses enterovirus infections through innate immunity induced by the inhibition of pyrimidine biosynthesis and nucleotide depletion. Oncotarget. 2017;8:115315–115325. doi: 10.18632/oncotarget.23258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahase E. Covid-19: WHO declares pandemic because of "alarming levels" of spread, severity, and inaction. BMJ. 2020;368:m1036. doi: 10.1136/bmj.m1036. [DOI] [PubMed] [Google Scholar]
- Qu C., Li Y., Li Y., Yu P., Li P., Donkers J.M., van de Graaf S.F.J., de Man R.A., Peppelenbosch M.P., Pan Q. FDA-drug screening identifies deptropine inhibiting hepatitis E virus involving the NF-kappaB-RIPK1-caspase axis. Antivir. Res. 2019;170:104588. doi: 10.1016/j.antiviral.2019.104588. [DOI] [PubMed] [Google Scholar]
- Yin Y., Bijvelds M., Dang W., Xu L., van der Eijk A.A., Knipping K., Tuysuz N., Dekkers J.F., Wang Y., de Jonge J., Sprengers D., van der Laan L.J., Beekman J.M., Ten Berge D., Metselaar H.J., de Jonge H., Koopmans M.P., Peppelenbosch M.P., Pan Q. Modeling rotavirus infection and antiviral therapy using primary intestinal organoids. Antivir. Res. 2015;123:120–131. doi: 10.1016/j.antiviral.2015.09.010. [DOI] [PubMed] [Google Scholar]
- Yin Y., Chen S., Hakim M.S., Wang W., Xu L., Dang W., Qu C., Verhaar A.P., Su J., Fuhler G.M., Peppelenbosch M.P., Pan Q. 6-Thioguanine inhibits rotavirus replication through suppression of Rac 1 GDP/GTP cycling. Antivir. Res. 2018;156:92–101. doi: 10.1016/j.antiviral.2018.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yin Y., Dang W., Zhou X., Xu L., Wang W., Cao W., Chen S., Su J., Cai X., Xiao S., Peppelenbosch M.P., Pan Q. PI3K-Akt-mTOR axis sustains rotavirus infection via the 4E-BP1 mediated autophagy pathway and represents an antiviral target. Virulence. 2018;9:83–98. doi: 10.1080/21505594.2017.1326443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yin Y., Metselaar H.J., Sprengers D., Peppelenbosch M.P., Pan Q. Rotavirus in organ transplantation: drug-virus-host interactions. Am. J. Transplant. 2015;15:585–593. doi: 10.1111/ajt.13135. [DOI] [PubMed] [Google Scholar]
- Yin Y., Wang Y., Dang W., Xu L., Su J., Zhou X., Wang W., Felczak K., van der Laan L.J., Pankiewicz K.W., van der Eijk A.A., Bijvelds M., Sprengers D., de Jonge H., Koopmans M.P., Metselaar H.J., Peppelenbosch M.P., Pan Q. Mycophenolic acid potently inhibits rotavirus infection with a high barrier to resistance development. Antivir. Res. 2016;133:41–49. doi: 10.1016/j.antiviral.2016.07.017. [DOI] [PubMed] [Google Scholar]
- Zhang Y., Chen L., Hu G.Q., Zhang N., Zhu X.D., Yang K.Y., Jin F., Shi M., Chen Y.P., Hu W.H., Cheng Z.B., Wang S.Y., Tian Y., Wang X.C., Sun Y., Li J.G., Li W.F., Li Y.H., Tang L.L., Mao Y.P., Zhou G.Q., Sun R., Liu X., Guo R., Long G.X., Liang S.Q., Li L., Huang J., Long J.H., Zang J., Liu Q.D., Zou L., Su Q.F., Zheng B.M., Xiao Y., Guo Y., Han F., Mo H.Y., Lv J.W., Du X.J., Xu C., Liu N., Li Y.Q., Chua M.L.K., Xie F.Y., Sun Y., Ma J. Gemcitabine and cisplatin induction chemotherapy in nasopharyngeal carcinoma. N. Engl. J. Med. 2019;381:1124–1135. doi: 10.1056/NEJMoa1905287. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.