Abstract
Parkinson's disease (PD) is a neurodegenerative disease characterized by the gradual loss of dopaminergic (DA) neurons in the substantia nigra (SN) and the formation of intracellular Lewy bodies (LB) in the brain, which aggregates α-synuclein (α-Syn) as the main component. The interest of flavonoids as potential neuroprotective agents is increasing due to its high efficiency and low side effects. Baicalin is one of the flavonoid compounds, which is a predominant flavonoid isolated from Scutellaria baicalensis Georgi. However, the key molecular mechanism by which Baicalin can prevent the PD pathogenesis remains unclear. In this study, we used bioinformatic assessment including Gene Ontology (GO) to elucidate the correlation between oxidative stress and PD pathogenesis. RNA-Seq methods were used to examine the global expression profiles of noncoding RNAs and found that C/EBPβ expression was upregulated in PD patients compared with healthy controls. Interestingly, Baicalin could protect DA neurons against reactive oxygen species (ROS) and decreased C/EBPβ and α-synuclein expression in pLVX-Tet3G-α-synuclein SH-SY5Y cells. In a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) induced PD mouse model, the results revealed that treatment with Baicalin improved the PD model's behavioral performance and reduced dopaminergic neuron loss in the substantia nigra, associated with the inactivation of proinflammatory cytokines and oxidative stress. Hence, our study supported that Baicalin repressed C/EBPβ via redox homeostasis, which may be an effective potential treatment for PD.
1. Background
Oxidative stress has been implicated as a key contributor to the progression of Parkinson's disease (PD) [1]. Because of the presence of enzymes such as tyrosine hydroxylase (TH) and monoamine oxidase (MAO), the neurotransmitter dopamine can be a major source of oxidative stress [2]. Although the human brain comprises less than 5% of total body weight, over 20% of the whole body's total oxygen is supplied to it, with part of oxygen subsequently converted into reactive oxygen species (ROS) [3]. Oxidative stress is considered as the common underlying source that leads to cellular dysfunction and demise, the idiopathic and genetic causes of PD [3, 4]. Overexpression of oxidative stress may lead to excitotoxicity, mitochondrial dysfunction, protein misfolding and aggregation, and cellular apoptosis, which are all in vitro indicators of PD [5]. It is also believed that the increased levels of oxidized lipids are the common underlying mechanism that leads to dopaminergic neuronal loss in the substantia nigra (SN) and motor dysfunctions in PD patients [6].
α-Synuclein accumulates in Lewy bodies, which is the hallmark in PD pathology and leads to neurodegeneration and the progression of the clinical symptoms [7]. Although its exact role in neuropathology is unclear, evidence suggests that overexpression of α-synuclein might lead to oxidative stress [8] and neuroinflammation [9]. Then, oxidative stress can modulate the α-synuclein structure, leading to other formations of the protein, including fibrils and oligomers [10], the latter of which can develop into positive regulation of ROS. The phosphorylation of α-synuclein at Ser-129, the major phosphorylation site, has been demonstrated from an animal model study to produce neurotoxic effects [11]. Moreover, oxidative stress leading to ROS production and α-synuclein aggregation is one of the proposed mechanisms for the death of dopaminergic neurons in PD patients [12].
It is also notable that the mitochondrial complex I damage is demonstrated to be one of the primary PD pathological animal models due to the administration of mitochondrial toxins, such as MPTP and rotenone, leading to the formation of α-synuclein aggregates and oxidative stress [13]. Methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), a mitochondrial complex I inhibitor, is metabolized into the toxin 1-methyl-4-phenylpyridine (MPP+) by monoamine oxidase B (MAO-B) and later taken up by dopaminergic neurons, which finally lead to neuronal death and ROS production [14]. In rats, chronic administration of rotenone caused selective nigral dopaminergic neuron loss and a significant reduction in complex I activity while at the same time, the ROS level increased [15]. Altogether, these findings suggest that mitochondrial respiratory chain impairment, in particular, complex I deficiency, and the subsequent increase in ROS production may directly contribute to the pathology of PD.
Usually, it is mitochondria's ability to produce ATP appropriately in response to energy demands [16]. Meanwhile, the transcription factor CCAAT/enhancer-binding protein beta (C/EBPβ), which is expressed in the brain, is also involved in the regulation of ATP synthesis [17]. Remarkably, C/EBPβ−/− mice exhibit resistance to excitotoxicity-induced neuronal cell death, which indicates that C/EBPβ might regulate gene expression implicated in brain damage [18].
There have been many reports discussing effective antioxidant treatment for PD, as well as conventional compounds that possessed antioxidant activity [19, 20]. Therefore, it is reasonable to suggest that targeting oxidative stress may be an effective strategy for PD medicine. Natural compounds have always been attractive targets for discovering new drug candidates, and many flavonoid derivatives are effective in preventing oxidative stress [21]. For instance, Hesperidin, the main flavanone derivative of citrus fruits, can alleviate cognitive impairment and oxidative stress in a mouse model of Alzheimer's disease [22]. Similarly, myricitrin, a flavonoid isolated from Chinese bayberry bark and fruit, demonstrated a protective effect on MPP+ induced mitochondrial dysfunction in a DJ-1-dependent manner in SN4741 cells [23]. Taking into account information about flavonoids, the focus of our paper is to discuss Baicalin, which is also the flavonoid derivatives, the principal component in the roots of Scutellaria radix, known as Huang Qin in Chinese traditional medicine [21, 24]. In recent years, several studies have shown that Baicalin displays a potent neuroprotective effect in various in vitro and in vivo models of neuronal injuries [25]. In particular, Baicalin effectively prevents neurodegenerative diseases through various pharmacological mechanisms, including antiexcitotoxicity, antiapoptosis, and anti-inflammation, promoting the expression of neuronal protective factors [26]. However, the mechanism of which Baicalin can inhibit neurodegeneration and regulate redox homeostasis is unclear. In this study, we used RNA-Seq to examine the global expression profiles of noncoding RNAs in PD patients and healthy controls, and then, we demonstrated that Baicalin could protect cells from neurotoxicity in vitro and in vivo. Subsequently, Gene Ontology analysis displayed that, compared with the healthy controls, many processes overrepresented in PD patients were related to glucocorticoid receptor binding and cellular response to oxidative stress. Our study may help extend understanding of the roles of oxidative stress and provide new research directions for PD.
2. Materials and Methods
2.1. Cell Lines and Cell Culture
Human cell line pLVX-Tet3G-α-synuclein SH-SY5Y was provided by Dr. Jingxing Zhang and supplemented with G418 (100 μg/ml). The cells were cultured in 1640 medium (Life Technologies, USA), supplemented with 10% fetal bovine serum (Hyclone, USA), penicillin (100 U/ml), and streptomycin (100 U/ml) (ABAM Life Technologies, California, USA). Cell cultures were maintained in 5% CO2 and air humidified in a 37°C incubator [27].
2.2. Chemicals and Reagents
Baicalin was purchased from the company (Sigma, USA), and the stock solutions were prepared in dimethyl sulfoxide (DMSO) (Sigma, USA). For in vivo experiments, Baicalin was dissolved in sterile PBS.
2.3. Bioinformatic Analysis
Differentially expressed genes (DEGs) were determined from a treated versus control comparison of log2-transformed expression measurements using the R package (http://www.bioconductor.org/packages/release/bioc/html/edgeR.html), and the resulting p values were adjusted using Benjamini and Hochberg's approach for controlling the false discovery rate (FDR) [28]. Differentially expressed genes (DEGs) with statistical significance were identified through volcano plot filtering. The thresholds for DEG were absolute log2 fold change > 1 and p value < 0.01. Hierarchical clustering was performed using pheatmap package in R. To understand the potential biological functions of DEGs, we used clusterProfiler on R platform (https://bioconductor.org/packages/release/bioc/html/clusterProfiler.html). GO terms with corrected p value less than 0.05 were considered significantly enriched by DEGs.
2.4. Quantitative Real-Time PCR
Total RNA was extracted using a TRIzol reagent (Invitrogen, California, USA) according to the manufacturer's instructions. Reverse transcription was performed with SuperScript III reverse transcriptase (Life Technologies, #18080085), and primers were designed and purchased from TaqMan: CEBPB (Hs00270923_s1), Cebpb (Mm00843434_s1), SNCA (Hs00240906_m1), Snca (Mm01188700_m1), Il1b (Mm00434228_m1), Il6 (Mm00446190_m1), TNF-α (Mm00443258_m1), and TGFβ (Mm01178820_m1). Real-time PCR was performed with a TaqMan Universal Master Mix Kit (Life Technologies, #4304473) by ABI 7500 Fast Real-Time PCR System. The relative quantification of the target genes was calculated by the comparative cycle threshold (CT) (2−ΔΔCT) method. The expression of GAPDH was used as an endogenous control. The relative quantification of gene expression was calculated by the 2-ΔΔCT method. All tests were performed in triplicate [29].
2.5. Protein Extraction and Western Blot Analysis
After the cell treatment under different conditions, the cells were harvested and the total proteins were extracted. Equal amounts of the proteins were loaded on SDS-PAGE gels, and the western blot assays were performed as previously described. α-Synuclein (#610787, BD Biosciences, USA), α-synuclein pS-129 (ab51253, Abcam, USA), p-C/EBPβ (#3084, CST, USA), C/EBPβ (#7962, Santa Cruz, USA), TH (#2792 CST, USA), and cleaved caspase-3 (#9664, CST, USA) were used at a final concentration of 1 mg/ml and were incubated overnight at 4°C in the presence of 5% nonfat milk powder. β-Actin (#3700, CST, USA) was used as the loading control [30].
2.6. Cell Viability Assay
Cell cytotoxicity was assessed in vitro using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. After different treatments, 20 μl of MTT (5 mg/ml in PBS, Sigma, USA) was added to each well and the plates were incubated for 2 h. The resulting formazan product was dissolved with DMSO, and the absorbance at a wavelength of 490 nm was read using a microplate reader (BioTek Instruments Inc., USA) [31]. All tests were performed in triplicate.
2.7. Intracellular ROS Measurement
The level of intracellular reactive oxygen species (ROS) was detected by the DCFH-DA method. After different treatments, cells were collected and then incubated with 10 μM DCFH-DA (ROS dye, #C6827, Invitrogen, USA) for 1 hour at 37°C. The fluorescence intensity was measured by a microplate reader (BioTek Instruments Inc., USA) with settings at excitation and emission equal to 485/535 nm, and all tests were performed in triplicate [32].
2.8. Lactate Dehydrogenase (LDH) Cytotoxicity Assay
The level of LDH was detected by LDH assay kits (Promega Corporation, USA). After different treatments, the 100 μl cell medium of each sample was collected and incubated with 50 μl of the CytoTox 96® Reagent for 30 minutes at room temperature. Then, 50 μl of Stop Solution was added to each well, and the absorbance at 490 nm was recorded. All tests were performed in triplicate.
2.9. Protein Carbonyl Assay Measurements and GSH/GSSG Ratio
After different treatments, the protein carbonyl level and GSH/GSSG ratio were measured from cell homogenates using a Protein Carbonyl Assay Kit (#ab126287, Abcam, Cambridge, MA, USA) and GSH/GSSG-Glo™ (Promega Corporation, USA), respectively, according to the manufacturer's guidelines [33]. All tests were performed in triplicate.
2.10. JC-1 Mitochondrial Membrane Potential Assay
After different treatments, the cells were washed by PBS and stained with 10 μM JC-1 (Cayman, USA) for 1 hour at 37°C. Finally, the cells were photographed with a fluorescence microscope (Nikon, Japan) at Ex488 nm/Em535 nm and Ex 540 mm/Em570 nm [34].
2.11. In Vivo Mouse Model Experiments
Male C57BL/6 mice (weighing 20–30 g) were purchased from Shanghai SLAC Laboratory Animal, housed, and maintained at constant temperature and humidity with a 12 h light/dark cycle in Tongji University. Three-month-old mice (8 per group) were injected a daily i.p. injection of MPTP (30 mg/kg) or saline treatment for 5 days [35] and then i.p. injection with 20 mg/kg and 40 mg/kg Baicalin for 2 weeks. Motor impairments were tested with rotarod tests and grid tests after Baicalin treatment (8 mice per group). In the rotarod tests, mice were trained for 2 min at a speed of 4 r.p.m. and then performed eight trials for a maximum of 5 min with increasing speed starting from 4 r.p.m. to 40 r.p.m. The fall-off time was recorded. For inverted grid tests, mice were placed in the center of a 30 × 30 cm screen with 1 cm wide mesh. The screen was inverted head-over-tail and placed on supports 40 cm above an open cage with deep bedding. Mice were timed until they released their grip or remained for 60 s.
2.12. Tissue Preparation
After 2 weeks of treatment and behavior test, mice (3 per group) intended for immunofluorescence (IF) staining and immunohistochemistry (IHC) analysis were euthanasia and transcardially perfused with PBS followed by 4% paraformaldehyde (PFA) in PBS. Brains were postfixed for 24 h in 4% PFA at 4°C and transferred to a solution of 30% sucrose in PBS for 24 h at 4°C. The coronal section of SN and STR was sectioned as 30 μm free-floating sections on a cryostat (Leica CM3050) and kept in PBS at 4°C [36]. The mouse brains intended for cell lysis (2 per group) and mRNA (3 per group) were transcardially perfused with ice-cold PBS and later performed western blotting and quantitative real-time PCR individually.
2.13. Immunofluorescence Staining
The number of TH- and GFAP-positive cells in the substantia nigra was estimated using a random sampling stereological counting method. Images were sampled from at least four different points within each substantia nigra section [37]. TH (ab6211), GFAP (ab7260), and DAPI (ab104139) were from Abcam, USA. All immunoreactive cells were counted regardless of the intensity of labeling. The slides were photographed with a fluorescence microscope (Nikon, Japan).
2.14. Immunohistochemistry Staining
Sections were prepared, and the expression of 4-HNE in the striatum and substantia nigra was assessed using a technique that has been reported previously [38]. In brief, the endogenous peroxidase activity was inactivated with 10% methanol and 3% hydrogen peroxide (H2O2) solution in PBS, pH 7.4, for 10 min, and nonspecific binding was blocked with 10% normal serum in TBS containing 0.5% Triton X-100. The sections were incubated overnight at 4°C with anti-4-HNE antibody (1 : 200 dilution in 1% normal serum in PBS containing 0.5% Triton X-100; ab46545, Abcam). The sections were then incubated with HRP-conjugated secondary antibody for 1 h. The slides were identified following DAB incubation for 10 min at room temperature. Finally, photographs were taken using a microscope (Nikon, Japan).
2.15. Statistical Analysis
Data visualization and analysis were performed with GraphPad Prism 6 (GraphPad Software Inc., La Jolla, CA, USA). Statistical analysis was performed using either Student's t-test or one-way ANOVA. Significant difference among groups was assessed as ∗p < 0.05, ∗∗p < 0.01, and ∗∗∗p < 0.001.
3. Results
3.1. The Functional Annotation of Bioinformatic Analysis in PD Patients
To profile differentially expressed mRNAs of PD patients and healthy controls, we obtained blood samples from both groups. Then, the whole-genome sequencing and alteration were analyzed. The mRNA was upregulated or downregulated, respectively, by more than twofold in PD patients vs. healthy controls (p < 0.05) (Figure 1(a)). Among them, the results showed that C/EBPβ was upregulated in PD patient blood samples compared with healthy controls (Figure 1(b)). Gene Ontology (GO) is a commonly used bioinformatic tool that provides comprehensive information on gene function of individual genomic products based on defined features [39]. This analysis was performed to establish the role of enrichment in molecular functions (MF), biological processes (BP), and cellular components (CC) in the interaction networks of C/EBPβ. The enrichment analysis showed that for biological processes in the C/EBPβ system, most of the genes were enriched at “cellular response to oxidative stress.” The cellular component analysis showed that most of the genes were “nuclear chromatin.” The typical molecular functions were “glucocorticoid receptor binding” and “protein heterodimerization activity” (Figure 1(c)). Hence, these results indicated that oxidative stress can be an effective target for PD research and presented more possible research directions for in-depth investigation.
Figure 1.

Expression profiles and Gene Ontology (GO) terms for differentially expressed mRNAs between PD patients and healthy control blood samples. (a) Volcano analysis exhibited differentially expressed mRNAs. Blue dots illustrated downregulated genes, and red dots illustrated upregulated genes. (b) C/EBPβ was upregulated in PD patient samples compared to healthy control samples. (c) GO enrichment analysis for biological processes, cellular component, and molecular function in the interaction networks of C/EBPβ.
3.2. Baicalin Inhibits the Toxicity and Oxidative Stress in pLVX-Tet3G-α-Synuclein SH-SY5Y Cell Lines
To examine the neurotoxicity of α-synuclein in vitro, the Doxycycline inducible pLVX-Tet3G-α-synuclein stable cell line was constructed, and the transfected efficiency was validated by mRNA and western blotting, as presented in Figures 2(a) and 2(b). To identify natural compounds capable of inhibiting the toxicity, we initiated a cell-based assay to screen chemicals extracted from Chinese herbal medicine [25]. The toxicity was evaluated by the release of lactate dehydrogenase (LDH). We found that 25 μM or 50 μM groups of Baicalin (Figure 2(c)) showed the significant ability (p = 0.036 and p = 0.02 individually) to protect cells from Dox-induced cell death (Figure 2(d)). Further study indicated that Baicalin exerted the protection effect on Dox-induced cells in a dose-dependent manner (Figure 2(e)). The quantitative oxidative stress level analysis showed that the 50 μM Baicalin group decreased the ROS level (Figure 2(f)) (p = 0.007) and carbonyl expression (Figure 2(g)) (p = 0.039) while increasing the GSH/GSSH level (p = 0.048) (Figure 2(h)) compared to the Dox-induced group. Hence, Baicalin inhibited toxicity and balanced the redox homeostasis in pLVX-Tet3G-α-synuclein SH-SY5Y cell lines.
Figure 2.

Baicalin inhibited toxicity and oxidative stress in pLVX-Tet3G-α-synuclein SH-SY5Y cell lines. (a) The mRNA level of α-synuclein in pLVX-Tet3G-α-synuclein SH-SY5Y stable cell line. (b) The protein level of α-synuclein in pLVX-Tet3G-α-synuclein SH-SY5Y stable cell line. (c) The chemical structure of Baicalin. (d) The LDH level of Baicalin. (e) Cell viability by MTT assay. (f) The ROS level, (g) carbonyl expression, and (h) GSH/GSSG level of Baicalin. The pLVX-Tet3G-α-synuclein SH-SY5Y cells were incubated with various concentrations (0–50 μM) of Baicalin for 24 h. All data represented the mean and standard error of three independent experiments. ∗p < 0.05; ∗∗p < 0.01; ∗∗∗p < 0.001.
3.3. Baicalin Protects against Dox-Induced Mitochondrial Dysfunctions
To determine whether Baicalin possesses the preventive effects against mitochondrial dysfunctions, parameters of mitochondrial function were studied in pLVX-Tet3G-α-synuclein SH-SY5Y cell lines. Mitochondrial membrane potential (ΔψM) is an important parameter of the mitochondrial function used as an indicator of cell health [40]. JC-1 is a dye that can selectively enter into mitochondria and reversibly change color from green to red as the membrane potential increases [41]. In Figure 3(a), Dox-induced JC-1 monomers were detected, which means that damaged or unhealthy cells were upregulated. On the other hand, increasing the concentration of Baicalin could induce JC-1 aggregates (healthy cells), especially at 50 μM (p = 0.024). These results indicated that Baicalin could protect membrane potential from Dox-induced toxicity. To assess whether Baicalin regulates CEBPB and SNCA mRNA expression, we conducted quantitative RT-PCR (qRT-PCR) assays with pLVX-Tet3G-α-synuclein SH-SY5Y cells and found that CEBPB and SNCA were upregulated after DOX treatment and it can be reversed by Baicalin in a dose-dependent way (Figure 3(b)). Immunoblotting showed that TH proteins revealed a dose-dependent elevation while p-C/EBPβ, C/EBPβ, α-synuclein, and α-synuclein pS129 decreased in pLVX-Tet3G-α-synuclein SH-SY5Y cells upon Baicalin treatment. The results demonstrated that Baicalin could reverse Dox-induced α-synuclein aggregation. Interestingly, Dox-induced caspase-3 activation indicates that active oxidative stress triggers apoptosis. On the other hand, Baicalin reduced caspase-3 activation in a dose-dependent manner (Figure 3(c)).
Figure 3.

Baicalin protected Dox-induced mitochondrial dysfunction. (a) The representative image of pLVX-Tet3G-α-synuclein SH-SY5Y cell with an accumulation of JC-1 staining after incubation with various concentrations (0–50 μM) of Baicalin for 24 h. Scale bars: 50 μm. (b) The mRNA level of pLVX-Tet3G-α-synuclein SH-SY5Y cells treated with various concentrations (0–50 μM) of Baicalin for 24 h. (c) Western blot analysis of pLVX-Tet3G-α-synuclein SH-SY5Y cell treated with various concentrations (0–50 μM) of Baicalin for 24 h. All data represented the mean and standard error of three independent experiments. ∗p < 0.05; ∗∗p < 0.01; ∗∗∗p < 0.001.
3.4. Baicalin Protects Dopaminergic Neurons and Rescues Motor Dysfunction against MPTP-Induced Neurotoxicity In Vivo
To investigate the in vivo roles of Baicalin, three-month-old C57BL mice (8 per group) were treated with MPTP (i.p., 30 mg/kg) or saline for 5 days. Motor behavioral tests showed that MPTP incurred significant motor disorder, which was ameliorated upon two weeks of 20 mg/kg and 40 mg/kg Baicalin treatment. Remarkably, in the 40 mg/kg Baicalin group, MPTP elicited in the rotarod test (p = 0.042) and grid test (p = 0.047) were significantly less severe than the control group (Figures 4(a) and 4(b)), supporting the fact that Baicalin was highly neuroprotective and prevented MPTP-elicited motor dysfunctions in mice. We also monitored dopaminergic neuron loss after Baicalin treatment. Immunofluorescence staining showed that dopaminergic neurons in SN were substantially diminished by MPTP as compared to the vehicle group. Again, Baicalin attenuated the loss of dopaminergic neurons (Figure 4(c)). The maximal neuroprotective effects occurred with the 40 mg/kg Baicalin group (p = 0.012).
Figure 4.
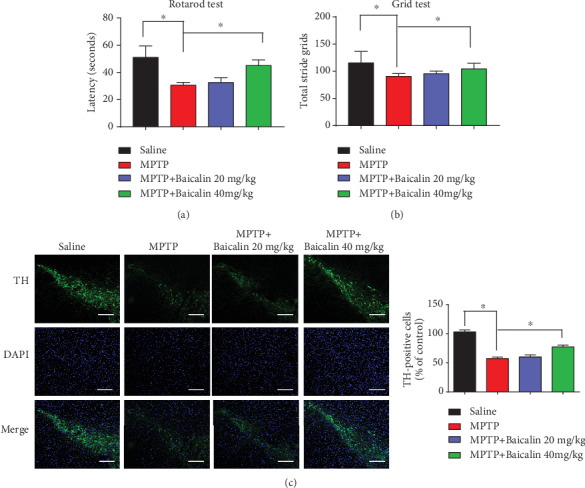
Baicalin protected dopaminergic neurons and rescues motor dysfunction against MPTP-induced neurotoxicity in vivo. (a) Rotarod tests and (b) grid tests were conducted by a blinded observer after two weeks of 20 mg/kg and 40 mg/kg Baicalin treatment. Data were the mean ± SEM (n = 8 per group). (c) TH staining of the substantia nigra (SN) of the above mice. Scale bars: 200 μm. All data represented the mean and standard error of three independent experiments. ∗p < 0.05; ∗∗p < 0.01; ∗∗∗p < 0.001.
3.5. Baicalin Regulates the Redox Balance in the MPTP Treatment Group
For manifesting the oxidative stress in Parkinson's disease, we employed 4-HNE staining, one of the most bioactive and studied lipid peroxidation biomarkers [42]. IHC analysis demonstrated that Baicalin group mice possessed the least oxidative stress among the experimental groups, provoked by MPTP treatment, in alignment with its prominent neuroprotective activity in the striatum and substantia nigra (Figure 5(a)). We observed that Cebpb (p = 0.021) and Snca (p = 0.0042) mRNA expression decreased after 40 mg/kg Baicalin treatment in SN compared to the MPTP group (Figure 5(b)). Immunoblotting analysis revealed the comparable protein levels expressed in the mouse brains. MPTP treatment elicited prominent dopaminergic neuronal TH loss, which was partially alleviated by Baicalin. As expected, α-synuclein pS129, α-synuclein, p-C/EBPβ, and C/EBPβ were strongly upregulated in the MPTP group after Baicalin treatment (Figure 5(c)). Hence, Baicalin possesses strong antioxidative activity, rescuing dopaminergic neurons from MPTP-induced cell death.
Figure 5.
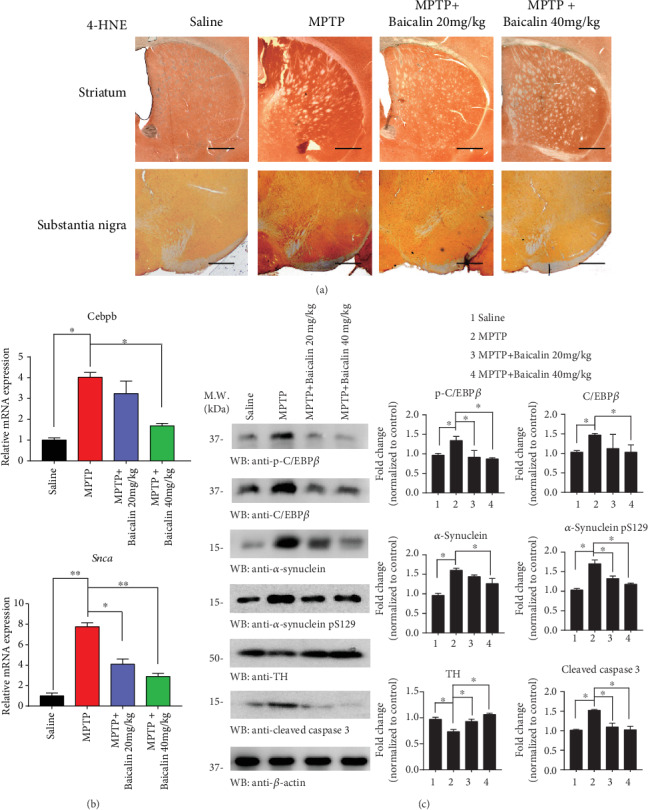
Baicalin protected dopaminergic neuron oxidative stress against MPTP treatment. (a) Immunohistochemical analysis for assessment of 4-HNE in the substantia nigra (SN) and striatum of saline, MPTP, MPTP+20 mg/kg Baicalin, and MPTP+40 mg/kg Baicalin groups. Scale bars: 500 μm. (b) The Cebpb and Snca mRNA levels of represented groups. (c) SN lysates were probed with various indicated antibodies. The band densitometric data of WB. All data represented the mean and standard error of three independent experiments. ∗p < 0.05; ∗∗p < 0.01; ∗∗∗p < 0.001.
3.6. Baicalin Protects against Neuroinflammation and Oxidative Stress Triggered by MPTP Treatment
Activated microglia and increased levels of inflammatory mediators are detected in the striatum of deceased PD patients [43, 44]; meanwhile, a large body of animal studies supports the contributory role of inflammation in dopaminergic cell loss [45]. Quantification of neuroinflammation revealed that IL-1β, IL-6, and TNF-α mRNA levels were decreased after Baicalin treatment, while the TGF-β mRNA level was increased (Figure 6(a)). Increased expression of GFAP is considered as markers of ROS production and the inflammatory process. In Figure 6(b), immunofluorescence staining of GFAP in the striatum showed a remarkably higher expression of GFAP-positive astrocytes in MPTP treatment when compared to the saline group (p = 0.0025). However, Baicalin decreased the GFAP-positive cells as well (p = 0.032). Again, our results indicated that Baicalin reduced oxidative stress in the MPTP treatment group.
Figure 6.

Baicalin protected against neuroinflammation and oxidative stress triggered by MPTP treatment. (a) IL-1β, IL-6, TNF-α, and TGF-β mRNA levels of SN measured by q-RTPCR in saline, MPTP, and MPTP+40 mg/kg Baicalin groups. (b) Immunofluorescence staining to detect the expression of the glial fibrillary acidic protein- (GFAP-) positive astrocyte (green) in saline, MPTP, and MPTP+40 mg/kg Baicalin groups. Scale bars: 100 μm. All data represented the mean and standard error of three independent experiments. ∗p < 0.05; ∗∗p < 0.01; ∗∗∗p < 0.001.
4. Discussion
Over thousands of years, Chinese herbs have been used by the Chinese pharmacologists as treatment of diseases, including cancer [46], climacteric syndrome [47], schizophrenia [48], Alzheimer's disease [49], and Parkinson's disease [50]. Baicalin is one of the most important flavonoid compounds, which is mainly isolated from the root of Scutellaria baicalensis Georgi, which is an indispensable Chinese medicinal herb [51]. Besides, Baicalin does not display any significant toxicity to the mice at a dose even of 15 g/kg [52], which indicates its low toxicity, making it highly acceptable and safe for application in humans. However, one major limitation in the clinical application of Baicalin is their poor oral bioavailability and low aqueous solubility. There is increasing evidence that Baicalin plays an important role in various diseases, such as cardiovascular disease, depression, Alzheimer's disease, and Parkinson's disease. Baicalin alleviates cardiac dysfunction and myocardial remodeling in a chronic pressure overload mouse model [53]. In AD, Baicalin can reduce Alzheimer-like pathological changes and memory impairment caused by amyloid β1-42. Jin et al. reported that Baicalin can reduce cognitive impairment and protect neurons from microglia-mediated neuroinflammation TLR4/NF-κB signaling [54]. In a recent study, Baicalin inhibited TLR4 expression through the PI3K/AKT/FoxO1 pathway and improved depression-like behavior [55]. Noticeably, Baicalein, which is the aglycone of Baicalin, inhibits α-synuclein fibrillation and disaggregates the preformed fibrils [56]. The medical mechanism of Baicalin inhibiting α-synuclein aggregates is still unclear. In our experiment, an in vitro experimental model, Baicalin (50 μM) protected pLVX-Tet3G-α-synuclein SH-SY5Y cells against Dox-induced toxicity and also prevented the loss of cell viability (Figure 2(e)). Other researchers also found that lack of α-synuclein in mice is associated with reduced vulnerability to MPTP and reduced dopaminergic neuronal cell death [57]. The previous study also showed that abnormal posture, gait, and stiffness of the limbs are directly related to the loss of dopaminergic neurons, which is the root cause of PD [58]. Baicalin and deferoxamine can reduce iron accumulation in SN of Parkinson's disease rats [59]. In our results, we showed that Baicalin blocked α-synuclein expression and aggregation and protected dopaminergic neurons and rescues motor dysfunction against MPTP-induced neurotoxicity in vivo (Figure 4); presumably, Baicalin's therapeutic efficacy might partially result from its inhibition against α-synuclein.
Additionally, Baicalin may sustain redox homeostasis by protecting mitochondrial systems after treating with MPP+ [60]. Maintaining homeostasis is essential for preventing and curing disease [61]. Unbalanced homeostasis with the more oxidized environment, for example, higher oxidative stress, facilitated more cell death. In our research, Baicalin inactivated ROS (25 μM and 50 μM) production, carbonyl expression, and lactate dehydrogenase (LDH) level, meanwhile, alternated in GSH/GSSG ratio (Figures 2(d) and 2(f)–2(h)), facilitating a dramatically less oxidized environment for cell survival. Interestingly, Baicalein was effective in blocking the Dox-induced toxicity on the mitochondrial membrane potential (Figure 3(a)), resulting in increased ATP synthesis.
Currently, with the advent of next-generation sequencing technologies, RNA-Seq is gradually replacing microarrays for the detection of transcript expression profiling [62]. Although a lot of papers reported the RNA networks in PD, microarray methods were primarily employed [63, 64]. In this study, we used RNA-Seq to examine the global expression profiles of noncoding RNAs. Using RNA-Seq technology, we found that C/EBPβ was upregulated in PD patient's blood samples (Figure 1(b)). Moreover, the target mRNAs of differentially expressed mRNAs were mostly involved in “glucocorticoid receptor binding,” “cellular response to oxidative stress,” and “protein localization to the mitochondria” (Figure 1(c)). These results were partly consistent with those of our data. Also, RNA-Seq data showed new findings in that regulation of “protein heterodimerization” and “nuclear chromatin” were involved in the pathogenesis of PD, which are interesting targets for further investigation.
Our previous work demonstrated that CEBP/β, which is the transcription factor of α-synuclein and MAO-B, mediates the pathogenesis of Parkinson's disease [65]. C/EBPβ is one of the family members of transcription factors in the basic-leucine zipper (bZIP) class. In glial cells, C/EBPβ regulates the proinflammatory program. Because of its role in neuroinflammation, C/EBPβ is a potential target for the treatment of neurodegenerative disorders [66]. In our study, Baicalin repressed DOX-induced C/EBPβ level and α-synuclein as indicated in mRNA and protein level (Figures 3(b) and 3(c)). In addition, it was demonstrated that Baicalin inhibited MPTP-triggered inflammatory mediators, including the classical proinflammatory triad of IL-1β, IL-6, and TNF-α (Figure 6(a)). Notably, Baicalin was involved in the regulation of proinflammatory gene expression in glial activation, especially GFAP expression, and played a key role in the reduction of neurotoxic effects by inactivating microglia (Figure 6(b)).
There may be some other limitations to our study that should be shortly mentioned: (1) Further studies are necessary to reveal how Baicalin regulates the inactivation of glial cells in PD models. (2) Although flavonoids are nonspecific inhibitors [67], our future research will focus on this basis and look for structural elements with high specificity and beneficial effects in PD treatment.
5. Conclusions
Our data strongly supported that the analysis of the gene expression profile may enable the identification of targets for PD diagnosis and treatment. The results confirmed that C/EBPβ and oxidative stress played important roles in the progression of PD. Meanwhile, Baicalin functioned as an antioxidant for dictating the expression of C/EBPβ and regulated oxidative stress in the pLVX-Tet3G-α-synuclein SH-SY5Y cell model and MPTP treatment mouse model, further indicating Baicalin's neuroprotective effect in neurotoxin-triggered Parkinson's disease.
Acknowledgments
The authors would like to thank Dr. Keqiang Ye from Emory University for revising the manuscript. This article is supported by the “National Key R&D Program of China” (2018YFC1314700 and 2018YFA0108000), National Natural Science Foundation of China (81974196 and 81873779), Shanghai municipal medical and health excellent academic leaders training program (2017BR029), and China Postdoctoral Science Foundation (2018M632168).
Data Availability
The data used to support the findings of this study have not been made available because the data also form part of an ongoing study.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Consent
All subjects agreed to be in the study, and biological specimens were obtained after informed consent with approval from Shanghai Tongji Hospital.
Conflicts of Interest
The authors declare that they have no competing interests.
Authors' Contributions
Kecheng Lei and Yijue Shen contributed equally to this work. They carried out the experiment and wrote the manuscript. Yijing He and Weifang Tong helped with animal work. Jingxing Zhang offered the cell lines. Liwen Zhang and Yichun Xu performed the analytic calculations. Lingjing Jin supervised the project. Kecheng Lei and Yijue Shen are co-first authors.
References
- 1.Al Shahrani M., Heales S., Hargreaves I., Orford M. Oxidative stress: mechanistic insights into inherited mitochondrial disorders and Parkinson’s disease. Journal of Clinical Medicine. 2017;6(11):p. 100. doi: 10.3390/jcm6110100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mahy N., Andrés N., Andrade C., Saura J. Age-related changes of MAO-A and -B distribution in human and mouse brain. Neurobiology. 2000;8(1):47–54. [PubMed] [Google Scholar]
- 3.Crotty G. F., Ascherio A., Schwarzschild M. A. Targeting urate to reduce oxidative stress in Parkinson disease. Experimental Neurology. 2017;298(Part B):210–224. doi: 10.1016/j.expneurol.2017.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hauser D. N., Hastings T. G. Mitochondrial dysfunction and oxidative stress in Parkinson's disease and monogenic parkinsonism. Neurobiology of Disease. 2013;51:35–42. doi: 10.1016/j.nbd.2012.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Olanow C. W. The pathogenesis of cell death in Parkinson's disease–2007. Movement Disorders. 2007;22(S17):S335–S342. doi: 10.1002/mds.21675. [DOI] [PubMed] [Google Scholar]
- 6.Barzilai A., Melamed E. Molecular mechanisms of selective dopaminergic neuronal death in Parkinson's disease. Trends in Molecular Medicine. 2003;9(3):126–132. doi: 10.1016/S1471-4914(03)00020-0. [DOI] [PubMed] [Google Scholar]
- 7.Schulz-Schaeffer W. J. The synaptic pathology of α-synuclein aggregation in dementia with Lewy bodies, Parkinson’s disease and Parkinson’s disease dementia. Acta Neuropathologica. 2010;120(2):131–143. doi: 10.1007/s00401-010-0711-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hashimoto M., Hsu L. J., Rockenstein E., Takenouchi T., Mallory M., Masliah E. α-Synuclein protects against oxidative stress via inactivation of the c-Jun N-terminal kinase stress-signaling pathway in neuronal cells. Journal of Biological Chemistry. 2002;277(13):11465–11472. doi: 10.1074/jbc.M111428200. [DOI] [PubMed] [Google Scholar]
- 9.Hirsch E. C., Vyas S., Hunot S. Neuroinflammation in Parkinson's disease. Parkinsonism & Related Disorders. 2012;18:S210–S212. doi: 10.1016/S1353-8020(11)70065-7. [DOI] [PubMed] [Google Scholar]
- 10.Uversky V. N. A protein-chameleon: conformational plasticity of α-synuclein, a disordered protein involved in neurodegenerative disorders. Journal of Biomolecular Structure and Dynamics. 2003;21(2):211–234. doi: 10.1080/07391102.2003.10506918. [DOI] [PubMed] [Google Scholar]
- 11.Gorbatyuk O. S., Li S., Sullivan L. F., et al. The phosphorylation state of Ser-129 in human α-synuclein determines neurodegeneration in a rat model of Parkinson disease. Proceedings of the National Academy of Sciences of the United States of America. 2008;105(2):763–768. doi: 10.1073/pnas.0711053105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Barnham K. J., Masters C. L., Bush A. I. Neurodegenerative diseases and oxidative stress. Nature Reviews Drug Discovery. 2004;3(3):205–214. doi: 10.1038/nrd1330. [DOI] [PubMed] [Google Scholar]
- 13.Betarbet R., Sherer T. B., Greenamyre J. T. Animal models of Parkinson's disease. BioEssays. 2002;24(4):308–318. doi: 10.1002/bies.10067. [DOI] [PubMed] [Google Scholar]
- 14.Schmidt N., Ferger B. Neurochemical findings in the MPTP model of Parkinson's disease. Journal of Neural Transmission. 2001;108(11):1263–1282. doi: 10.1007/s007020100004. [DOI] [PubMed] [Google Scholar]
- 15.Testa C. M., Sherer T. B., Greenamyre J. T. Rotenone induces oxidative stress and dopaminergic neuron damage in organotypic substantia nigra cultures. Molecular Brain Research. 2005;134(1):109–118. doi: 10.1016/j.molbrainres.2004.11.007. [DOI] [PubMed] [Google Scholar]
- 16.Brand M. D., Nicholls D. G. Assessing mitochondrial dysfunction in cells. Biochemical Journal. 2011;435(2):297–312. doi: 10.1042/BJ20110162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Du Q., Tan Z., Shi F., et al. PGC1α/CEBPB/CPT1A axis promotes radiation resistance of nasopharyngeal carcinoma through activating fatty acid oxidation. Cancer Science. 2019;110(6):2050–2062. doi: 10.1111/cas.14011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang Z.-H., Gong K., Liu X., et al. C/EBPβ regulates delta-secretase expression and mediates pathogenesis in mouse models of Alzheimer's disease. Nature Communications. 2018;9(1, article 1784) doi: 10.1038/s41467-018-04120-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Aquilano K., Baldelli S., Rotilio G., Ciriolo M. R. Role of nitric oxide synthases in Parkinson’s disease: a review on the antioxidant and anti-inflammatory activity of polyphenols. Neurochemical Research. 2008;33(12):2416–2426. doi: 10.1007/s11064-008-9697-6. [DOI] [PubMed] [Google Scholar]
- 20.The Parkinson Study Group. Effects of tocopherol and deprenyl on the progression of disability in early Parkinson's disease. New England Journal of Medicine. 1993;328(3):176–183. doi: 10.1056/NEJM199301213280305. [DOI] [PubMed] [Google Scholar]
- 21.de Oliveira M. R., Nabavi S. F., Habtemariam S., Orhan I. E., Daglia M., Nabavi S. M. The effects of baicalein and baicalin on mitochondrial function and dynamics: a review. Pharmacological Research. 2015;100:296–308. doi: 10.1016/j.phrs.2015.08.021. [DOI] [PubMed] [Google Scholar]
- 22.Wang D., Liu L., Zhu X., Wu W., Wang Y. Hesperidin alleviates cognitive impairment, mitochondrial dysfunction and oxidative stress in a mouse model of Alzheimer’s disease. Cellular and Molecular Neurobiology. 2014;34(8):1209–1221. doi: 10.1007/s10571-014-0098-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cai Z., Zeng W., Tao K., Lu F., Gao G., Yang Q. Myricitrin alleviates MPP+-induced mitochondrial dysfunction in a DJ-1-dependent manner in SN4741 cells. Biochemical and Biophysical Research Communications. 2015;458(2):227–233. doi: 10.1016/j.bbrc.2015.01.060. [DOI] [PubMed] [Google Scholar]
- 24.Sowndhararajan K., Deepa P., Kim M., Park S. J., Kim S. Neuroprotective and cognitive enhancement potentials of baicalin: a review. Brain Sciences. 2018;8(6):p. 104. doi: 10.3390/brainsci8060104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dong J., Zhang Y., Chen Y., et al. Baicalin inhibits the lethality of ricin in mice by inducing protein oligomerization. Journal of Biological Chemistry. 2015;290(20):12899–12907. doi: 10.1074/jbc.M114.632828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Liang W., Huang X., Chen W. The effects of baicalin and baicalein on cerebral ischemia: a review. Aging and Disease. 2017;8(6):850–867. doi: 10.14336/AD.2017.0829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ahn E. H., Kim D. W., Shin M. J., et al. PEP-1–ribosomal protein S3 protects dopaminergic neurons in an MPTP-induced Parkinson's disease mouse model. Free Radical Biology and Medicine. 2013;55:36–45. doi: 10.1016/j.freeradbiomed.2012.11.008. [DOI] [PubMed] [Google Scholar]
- 28.Xing Y., Zhao Z., Zhu Y., Zhao L., Zhu A., Piao D. Comprehensive analysis of differential expression profiles of mRNAs and lncRNAs and identification of a 14-lncRNA prognostic signature for patients with colon adenocarcinoma. Oncology Reports. 2018;39(5):2365–2375. doi: 10.3892/or.2018.6324. [DOI] [PubMed] [Google Scholar]
- 29.Lei K., Liang X., Gao Y., et al. Lnc-ATB contributes to gastric cancer growth through a miR-141-3p/TGFβ2 feedback loop. Biochemical and Biophysical Research Communications. 2017;484(3):514–521. doi: 10.1016/j.bbrc.2017.01.094. [DOI] [PubMed] [Google Scholar]
- 30.Lei K., du W., Lin S., et al. 3B, a novel photosensitizer, inhibits glycolysis and inflammation via miR-155-5p and breaks the JAK/STAT3/SOCS1 feedback loop in human breast cancer cells. Biomedicine & Pharmacotherapy. 2016;82:141–150. doi: 10.1016/j.biopha.2016.04.049. [DOI] [PubMed] [Google Scholar]
- 31.Lei K., Tan S., Du W., et al. 3B, a novel of photosensitizer, exhibited anti-tumor effects via mitochondrial apoptosis pathway in MCF-7 human breast carcinoma cells. Tumor Biology. 2015;36(7):5597–5606. doi: 10.1007/s13277-015-3231-7. [DOI] [PubMed] [Google Scholar]
- 32.Ahn E. H., Kim D. W., Shin M. J., et al. PEP-1-PEA-15 protects against toxin-induced neuronal damage in a mouse model of Parkinson's disease. Biochimica et Biophysica Acta (BBA)-General Subjects. 2014;1840(6):1686–1700. doi: 10.1016/j.bbagen.2014.01.004. [DOI] [PubMed] [Google Scholar]
- 33.Luo S., Lei K., Xiang D., Ye K. NQO1 is regulated by PTEN in glioblastoma, mediating cell proliferation and oxidative stress. Oxidative Medicine and Cellular Longevity. 2018;2018:16. doi: 10.1155/2018/9146528.9146528 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ding H., Xiong Y., Sun J., Chen C., Gao J., Xu H. Asiatic acid prevents oxidative stress and apoptosis by inhibiting the translocation of α-synuclein into mitochondria. Frontiers in Neuroscience. 2018;12:p. 431. doi: 10.3389/fnins.2018.00431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Li Z., Bao X., Bai X., et al. Design, synthesis, and biological evaluation of phenol bioisosteric analogues of 3-hydroxymorphinan. Scientific Reports. 2019;9(1, article 2247) doi: 10.1038/s41598-019-38911-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Svarcbahs R., Julku U. H., Myöhänen T. T. Inhibition of prolyl oligopeptidase restores spontaneous motor behavior in the α-synuclein virus vector–based Parkinson's disease mouse model by decreasing α-synuclein oligomeric species in mouse brain. Journal of Neuroscience. 2016;36(49):12485–12497. doi: 10.1523/JNEUROSCI.2309-16.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jiang H., Kang S. U., Zhang S., et al. Adult conditional knockout of PGC-1α leads to loss of dopamine neurons. eNeuro. 2016;3(4):ENEURO.0183–ENEU16.2016. doi: 10.1523/ENEURO.0183-16.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Jia R., Kurita-Ochiai T., Oguchi S., Yamamoto M. Periodontal pathogen accelerates lipid peroxidation and atherosclerosis. Journal of Dental Research. 2013;92(3):247–252. doi: 10.1177/0022034513475625. [DOI] [PubMed] [Google Scholar]
- 39.Gene Ontology Consortium. The Gene Ontology (GO) database and informatics resource. Nucleic Acids Research. 2004;32(90001):258D–2261. doi: 10.1093/nar/gkh036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Acton B., Jurisicova A., Jurisica I., Casper R. Alterations in mitochondrial membrane potential during preimplantation stages of mouse and human embryo development. Molecular Human Reproduction. 2004;10(1):23–32. doi: 10.1093/molehr/gah004. [DOI] [PubMed] [Google Scholar]
- 41.Martin M. V., Fiol D., Zabaleta E., Pagnussat G. Arabidopsis thaliana embryo sac mitochondrial membrane potential stain. Bio-Protocol. 2014;4(10) doi: 10.21769/BioProtoc.1128. [DOI] [Google Scholar]
- 42.Guichardant M., Lagarde M. Analysis of biomarkers from lipid peroxidation: a comparative study. European Journal of Lipid Science and Technology. 2009;111(1):75–82. doi: 10.1002/ejlt.200800069. [DOI] [Google Scholar]
- 43.Hald A., Lotharius J. Oxidative stress and inflammation in Parkinson's disease: is there a causal link? Experimental Neurology. 2005;193(2):279–290. doi: 10.1016/j.expneurol.2005.01.013. [DOI] [PubMed] [Google Scholar]
- 44.Kaur K., Gill J. S., Bansal P. K., Deshmukh R. Neuroinflammation - A major cause for striatal dopaminergic degeneration in Parkinson's disease. Journal of the Neurological Sciences. 2017;381:308–314. doi: 10.1016/j.jns.2017.08.3251. [DOI] [PubMed] [Google Scholar]
- 45.Whitton P. Inflammation as a causative factor in the aetiology of Parkinson's disease. British Journal of Pharmacology. 2007;150(8):963–976. doi: 10.1038/sj.bjp.0707167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.McCulloch M., See C., Shu X. J., et al. Astragalus-based Chinese herbs and platinum-based chemotherapy for advanced non-small-cell lung cancer: meta-analysis of randomized trials. Journal of Clinical Oncology. 2006;24(3):419–430. doi: 10.1200/JCO.2005.03.6392. [DOI] [PubMed] [Google Scholar]
- 47.Eisenhardt S., Fleckenstein J. Traditional Chinese medicine valuably augments therapeutic options in the treatment of climacteric syndrome. Archives of Gynecology and Obstetrics. 2016;294(1):193–200. doi: 10.1007/s00404-016-4078-x. [DOI] [PubMed] [Google Scholar]
- 48.Rathbone J., Zhang L., Zhang M., et al. Chinese herbal medicine for schizophrenia: cochrane systematic review of randomised trials. The British Journal of Psychiatry. 2007;190(5):379–384. doi: 10.1192/bjp.bp.106.026880. [DOI] [PubMed] [Google Scholar]
- 49.Wu T.-Y., Chen C.-P., Jinn T.-R. Traditional Chinese medicines and Alzheimer’s disease. Taiwanese Journal of Obstetrics and Gynecology. 2011;50(2):131–135. doi: 10.1016/j.tjog.2011.04.004. [DOI] [PubMed] [Google Scholar]
- 50.Chen L.-W., Wang Y.-Q., Wei L.-C., Shi M., Chan Y.-S. Chinese herbs and herbal extracts for neuroprotection of dopaminergic neurons and potential therapeutic treatment of Parkinson’s disease. CNS & Neurological Disorders - Drug Targets. 2007;6(4):273–281. doi: 10.2174/187152707781387288. [DOI] [PubMed] [Google Scholar]
- 51.Zhao Q., Chen X.-Y., Martin C. Scutellaria baicalensis, the golden herb from the garden of Chinese medicinal plants. Science Bulletin. 2016;61(18):1391–1398. doi: 10.1007/s11434-016-1136-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zhang X., Cheng Q., Zhang Y. Acute toxicity test of baicalin capsule in mice. Journal of Medical Research. 2006 [Google Scholar]
- 53.Zhang Y., Liao P., Zhu M.’., et al. Baicalin attenuates cardiac dysfunction and myocardial remodeling in a chronic pressure-overload mice model. Cellular Physiology and Biochemistry. 2017;41(3):849–864. doi: 10.1159/000459708. [DOI] [PubMed] [Google Scholar]
- 54.Jin X., Liu M. Y., Zhang D. F., et al. Baicalin mitigates cognitive impairment and protects neurons from microglia-mediated neuroinflammation via suppressingNLRP3 inflammasomes andTLR4/NF‐κB signaling pathway. CNS Neuroscience & Therapeutics. 2019;25(5):575–590. doi: 10.1111/cns.13086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Guo L.-T., Wang S.-Q., Su J., et al. Baicalin ameliorates neuroinflammation-induced depressive-like behavior through inhibition of toll-like receptor 4 expression via the PI3K/AKT/FoxO1 pathway. Journal of Neuroinflammation. 2019;16(1):p. 95. doi: 10.1186/s12974-019-1474-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Caruana M., Högen T., Levin J., Hillmer A., Giese A., Vassallo N. Inhibition and disaggregation of α-synuclein oligomers by natural polyphenolic compounds. FEBS Letters. 2011;585(8):1113–1120. doi: 10.1016/j.febslet.2011.03.046. [DOI] [PubMed] [Google Scholar]
- 57.Klivenyi P., Siwek D., Gardian G., et al. Mice lacking alpha-synuclein are resistant to mitochondrial toxins. Neurobiology of Disease. 2006;21(3):541–548. doi: 10.1016/j.nbd.2005.08.018. [DOI] [PubMed] [Google Scholar]
- 58.Eichhammer P., Johann M., Kharraz A., et al. High-frequency repetitive transcranial magnetic stimulation decreases cigarette smoking. Journal of Clinical Psychiatry. 2003;64(8):951–953. doi: 10.4088/JCP.v64n0815. [DOI] [PubMed] [Google Scholar]
- 59.Xiong P., Chen X., Guo C., Zhang N., Ma B. Baicalin and deferoxamine alleviate iron accumulation in different brain regions of Parkinson's disease rats. Neural Regeneration Research. 2012;7, article 2092 doi: 10.3969/j.issn.1673-5374.2012.27.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Guo A. J. Y., Choi R. C. Y., Cheung A. W. H., et al. Baicalin, a flavone, induces the differentiation of cultured osteoblasts. Journal of Biological Chemistry. 2011;286(32):27882–27893. doi: 10.1074/jbc.M111.236281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ursini F., Maiorino M., Forman H. J. Redox homeostasis: the Golden Mean of healthy living. Redox Biology. 2016;8:205–215. doi: 10.1016/j.redox.2016.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Tarazona S., García-Alcalde F., Dopazo J., Ferrer A., Conesa A. Differential expression in RNA-seq: a matter of depth. Genome Research. 2011;21(12):2213–2223. doi: 10.1101/gr.124321.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Mandel S., Weinreb O., Youdim M. B. H. Using cDNA microarray to assess Parkinson's disease models and the effects of neuroprotective drugs. Trends in Pharmacological Sciences. 2003;24(4):184–191. doi: 10.1016/S0165-6147(03)00067-1. [DOI] [PubMed] [Google Scholar]
- 64.Häbig K., Walter M., Poths S., Riess O., Bonin M. RNA interference of LRRK2–microarray expression analysis of a Parkinson’s disease key player. Neurogenetics. 2008;9(2):83–94. doi: 10.1007/s10048-007-0114-0. [DOI] [PubMed] [Google Scholar]
- 65.Wu Z., Xia Y., Wang Z., et al. C/EBPβ/δ-secretase signaling mediates Parkinson's disease pathogenesis via regulating transcription and proteolytic cleavage of α-synuclein and MAOB. Molecular Psychiatry. 2020 doi: 10.1038/s41380-020-0687-7. [DOI] [PubMed] [Google Scholar]
- 66.Pulido-Salgado M., Vidal-Taboada J. M., Saura J. C/EBPβ and C/EBPδ transcription factors: Basic biology and roles in the CNS. Progress in Neurobiology. 2015;132:1–33. doi: 10.1016/j.pneurobio.2015.06.003. [DOI] [PubMed] [Google Scholar]
- 67.Wright B., Spencer J. P. E., Lovegrove J. A., Gibbins J. M. Insights into dietary flavonoids as molecular templates for the design of anti-platelet drugs. Cardiovascular Research. 2013;97(1):13–22. doi: 10.1093/cvr/cvs304. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used to support the findings of this study have not been made available because the data also form part of an ongoing study.


