Abstract
Background
People with cancer experience a variety of symptoms as a result of their disease and the therapies involved in its management. Inadequate symptom management has implications for patient outcomes including functioning, psychological well‐being, and quality of life (QoL). Attempts to reduce the incidence and severity of cancer symptoms have involved the development and testing of psycho‐educational interventions to enhance patients' symptom self‐management. With the trend for care to be provided nearer patients' homes, telephone‐delivered psycho‐educational interventions have evolved to provide support for the management of a range of cancer symptoms. Early indications suggest that these can reduce symptom severity and distress through enhanced symptom self‐management.
Objectives
To assess the effectiveness of telephone‐delivered interventions for reducing symptoms associated with cancer and its treatment. To determine which symptoms are most responsive to telephone interventions. To determine whether certain configurations (e.g. with/without additional support such as face‐to‐face, printed or electronic resources) and duration/frequency of intervention calls mediate observed cancer symptom outcome effects.
Search methods
We searched the following databases: the Cochrane Central Register of Controlled Trials (CENTRAL; 2019, Issue 1); MEDLINE via OVID (1946 to January 2019); Embase via OVID (1980 to January 2019); (CINAHL) via Athens (1982 to January 2019); British Nursing Index (1984 to January 2019); and PsycINFO (1989 to January 2019). We searched conference proceedings to identify published abstracts, as well as SIGLE and trial registers for unpublished studies. We searched the reference lists of all included articles for additional relevant studies. Finally, we handsearched the following journals: Cancer, Journal of Clinical Oncology, Psycho‐oncology, Cancer Practice, Cancer Nursing, Oncology Nursing Forum, Journal of Pain and Symptom Management, and Palliative Medicine. We restricted our search to publications published in English.
Selection criteria
We included randomised controlled trials (RCTs) and quasi‐RCTs that compared one or more telephone interventions with one other, or with other types of interventions (e.g. a face‐to‐face intervention) and/or usual care, with the stated aim of addressing any physical or psychological symptoms of cancer and its treatment, which recruited adults (over 18 years) with a clinical diagnosis of cancer, regardless of tumour type, stage of cancer, type of treatment, and time of recruitment (e.g. before, during, or after treatment).
Data collection and analysis
We used Cochrane methods for trial selection, data extraction and analysis. When possible, anxiety, depressive symptoms, fatigue, emotional distress, pain, uncertainty, sexually‐related and lung cancer symptoms as well as secondary outcomes are reported as standardised mean differences (SMDs) with 95% confidence intervals (CIs), and we presented a descriptive synthesis of study findings. We reported on findings according to symptoms addressed and intervention types (e.g. telephone only, telephone combined with other elements). As many studies included small samples, and because baseline scores for study outcomes often varied for intervention and control groups, we used change scores and associated standard deviations. The certainty of the evidence for each outcome was interpreted using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach.
Main results
Thirty‐two studies were eligible for inclusion; most had moderate risk of bias,often related to blinding. Collectively, researchers recruited 6250 people and studied interventions in people with a variety of cancer types and across the disease trajectory, although many participants had breast cancer or early‐stage cancer and/or were starting treatment. Studies measured symptoms of anxiety, depression, emotional distress, uncertainty, fatigue, and pain, as well as sexually‐related symptoms and general symptom intensity and/or distress.
Interventions were primarily delivered by nurses (n = 24), most of whom (n = 16) had a background in oncology, research, or psychiatry. Ten interventions were delivered solely by telephone; the rest combined telephone with additional elements (i.e. face‐to‐face consultations and digital/online/printed resources). The number of calls delivered ranged from 1 to 18; most interventions provided three or four calls.
Twenty‐one studies provided evidence on effectiveness of telephone‐delivered interventions and the majority appeared to reduce symptoms of depression compared to control. Nine studies contributed quantitative change scores (CSs) and associated standard deviation results (or these could be calculated). Likewise, many telephone interventions appeared effective when compared to control in reducing anxiety (16 studies; 5 contributed quantitative CS results); fatigue (9 studies; 6 contributed to quantitative CS results); and emotional distress (7 studies; 5 contributed quantitative CS results). Due to significant clinical heterogeneity with regards to interventions introduced, study participants recruited, and outcomes measured, meta‐analysis was not conducted.
For other symptoms (uncertainty, pain, sexually‐related symptoms, dyspnoea, and general symptom experience), evidence was limited; similarly meta‐analysis was not possible, and results from individual studies were largely conflicting, making conclusions about their management through telephone‐delivered interventions difficult to draw. Heterogeneity was considerable across all trials for all outcomes.
Overall, the certainty of evidence was very low for all outcomes in the review. Outcomes were all downgraded due to concerns about overall risk of bias profiles being frequently unclear, uncertainty in effect estimates and due to some inconsistencies in results and general heterogeneity.
Unsubstantiated evidence suggests that telephone interventions in some capacity may have a place in symptom management for adults with cancer. However, in the absence of reliable and homogeneous evidence, caution is needed in interpreting the narrative synthesis. Further, there were no clear patterns across studies regarding which forms of interventions (telephone alone versus augmented with other elements) are most effective. It is impossible to conclude with any certainty which forms of telephone intervention are most effective in managing the range of cancer‐related symptoms that people with cancer experience.
Authors' conclusions
Telephone interventions provide a convenient way of supporting self‐management of cancer‐related symptoms for adults with cancer. These interventions are becoming more important with the shift of care closer to patients' homes, the need for resource/cost containment, and the potential for voluntary sector providers to deliver healthcare interventions. Some evidence supports the use of telephone‐delivered interventions for symptom management for adults with cancer; most evidence relates to four commonly experienced symptoms ‐ depression, anxiety, emotional distress, and fatigue. Some telephone‐delivered interventions were augmented by combining them with face‐to‐face meetings and provision of printed or digital materials. Review authors were unable to determine whether telephone alone or in combination with other elements provides optimal reduction in symptoms; it appears most likely that this will vary by symptom. It is noteworthy that, despite the potential for telephone interventions to deliver cost savings, none of the studies reviewed included any form of health economic evaluation.
Further robust and adequately reported trials are needed across all cancer‐related symptoms, as the certainty of evidence generated in studies within this review was very low, and reporting was of variable quality. Researchers must strive to reduce variability between studies in the future. Studies in this review are characterised by clinical and methodological diversity; the level of this diversity hindered comparison across studies. At the very least, efforts should be made to standardise outcome measures. Finally, studies were compromised by inclusion of small samples, inadequate concealment of group allocation, lack of observer blinding, and short length of follow‐up. Consequently, conclusions related to symptoms most amenable to management by telephone‐delivered interventions are tentative.
Plain language summary
Telephone interventions for managing symptoms in adults with cancer
Background People with cancer experience a variety of symptoms caused by their disease and its treatment. Symptoms can include depression, anxiety, fatigue and pain. These are often managed, day‐to‐day, by patients or their family members. If symptoms are not well managed, this can lead to other problems, such as difficulties in carrying out everyday tasks, poor sleep and poor quality of life.
Cancer professionals have developed psychological and educational treatments to help people to manage cancer symptoms. These treatments (or interventions) can be delivered by telephone (telephone interventions) in the patients’ homes instead of face‐to‐face in hospital.
What questions does this review aim to answer? This Cochrane Review aimed to answer the following questions.
1. Are telephone interventions for adults with cancer effective in relieving symptoms of cancer and cancer treatment?
2. Which symptoms are most reduced when telephone interventions are used?
3. What parts of telephone interventions have the most impact in reducing cancer symptoms?
In this review, telephone interventions were interventions given only, or mainly, by telephone. They were given by health professionals. As well as telephone contact, they could include face‐to‐face contact, or printed, digital or online information, such as, leaflets, computer programs and websites.
How did we answer these questions? We searched medical databases and journals to find all randomised controlled trials that used a telephone intervention to reduce any cancer symptoms. Randomised controlled trials allocate people randomly to one treatment or another; they provide the most reliable evidence. Studies could compare telephone interventions with another telephone intervention, with another type of intervention (e.g. face‐to‐face), or with usual care. Participants in these studies were adults with any kind of cancer at any stage.
Results We included 32 studies with a total of 6250 participants. Most studies (21) were from the USA. Nine studies recruited women with breast cancer, 11 included people with breast, colorectal, lung, or prostate cancer. Fourteen studies included people with early‐stage cancer. Nurses provided interventions in 24 studies. Only 10 studies delivered interventions solely by telephone, and 16 studies combined telephone calls with other materials (printed or digital). Studies measured symptoms of depression, anxiety, emotional distress, uncertainty, fatigue, pain, sexual symptoms, and breathlessness. They also measured the effect of all the symptoms together (the general symptom experience).
Most studies compared a telephone intervention with usual care alone or usual care with additional support. Eight studies compared two telephone interventions against each other; some also compared these with usual care.
Because the studies were so different from each other, we could not combine the results into one analysis for each symptom. However, some studies measured changes in symptoms using standardised or similar scales. They recorded participants’ scale scores at the beginning of the intervention, during the intervention, and at the end, resulting in a ‘change score’. We analysed the results from studies that recorded change scores.
What does evidence from the review tell us? Twenty‐one studies provided evidence on depression compared to usual care or other interventions, but only nine provided change scores. These found that telephone interventions appeared to reduce symptoms of depression. Likewise, telephone interventions appeared effective compared to usual care or other interventions in reducing anxiety (16 studies; 5 contributed change scores); fatigue (9 studies; 6 contributed change scores); and emotional distress (7 studies; 5 contributed change scores).
Evidence for other symptoms was limited, making it difficult to draw conclusions.
Certainty of the evidence Telephone interventions appear to relieve some symptoms of cancer and cancer treatment, however, the studies were small and very different from each other, so our confidence (certainty) in the evidence is very low. It is unclear whether telephone interventions alone, or combined with face‐to‐face meetings, or printed or audio materials, are most effective in reducing the many symptoms that people with cancer experience.
Conclusions Telephone interventions are convenient for patients, their families and healthcare workers but the results of our review were not conclusive. Further, rigorous research on this topic would help to answer our review questions.
Search date This review includes evidence published up to January 2019.
Summary of findings
Summary of findings 1. Summary of findings.
| Telephone interventions compared with control interventions for symptom management in adults with cancer | ||||
|
Patient or population: individuals with any cancer at any stage Settings: randomised controlled trials Intervention: telephone interventions with or without additional support Comparison: control intervention | ||||
| Outcomes | Risks | Effects of interventions | No. of participants (studies) | Certainty of the evidence (GRADE) |
| Anxiety | We could not present illustrative absolute effects because a representative control group risk could not be ascertained from the studies nor from any external source. Furthermore, results were reported in narrative form and varied considerably |
Effect measures (using change score (CS)) ranged from: SMD ‐5.1 (95% CI ‐6.1 to ‐4.1) for breast cancer to SMD ‐0.3 (95% CI ‐0.3 to 0.9) for prostate cancer Other cancer sites including colorectal and lung and trials including participants with mixed cancers The 5 trials reporting data where change scores could be calculated were generally very heterogeneous in terms of demographics, including age and gender (gender specific or mixed cancers), FIGO stage (early or advanced disease), and delivery of interventions and controls. This may have differed in the number of telephone calls and whether additional management components were used in intervention arms, and in controls being sufficiently different to consider any data synthesis methods |
277 participants
(5 studies) Sample sizes were often small, and baseline outcome values for intervention and control groups largely differed widely in 11 further studies. Therefore displaying only studies that used change scores seemed appropriate, and in future updates of the review, meta‐analytical approaches will be attempted |
⊕⊝⊝⊝ Very lowa,b,c |
| Depression |
Effect measures (CS) ranged from: SMD ‐2.2 (95% CI ‐2.7 to ‐1.7) for colorectal cancer to SMD 0.3 (95% CI 0.04 to 0.5) for mixed cancers Other cancer sites including breast, lung, and prostate cancer. There was scope in the breast and mixed cancer subgroups to potentially pool results, but even within these more restrictive analyses, there was considerable heterogeneity, imprecision, and inconsistency across trials. Therefore results were reported by single trials, and results were presented narratively The 9 trials reporting data where change scores could be calculated were generally very heterogeneous in terms of demographics, including age and gender (gender specific or mixed cancers), FIGO stage (early or advanced disease), and delivery of interventions and controls. This may have differed in the number of telephone calls and whether additional management components were used in intervention arms, and in controls being sufficiently different to consider any data synthesis methods |
1059 participants
(9 studies) Sample sizes were often small, and baseline outcome values for intervention and control groups largely differed widely in 12 further studies. Therefore displaying only studies that used change scores seemed appropriate, and in future updates of the review, attempts at meta‐analysis will be made |
⊕⊝⊝⊝ Very lowa,b,c | |
| Fatigue |
Effect measures (CS) ranged from: SMD ‐0.9 (95% CI ‐1.5 to ‐0.3) for breast cancer to SMD 0.0 (95% CI ‐0.2 to 0.2) for mixed cancers Another cancer site, including prostate cancer. There was scope in the mixed cancer subgroup to potentially pool results, but even within these more restrictive analyses, there were sufficient clinical differences between trials to justify not using this approach. Therefore results were reported by single trials and are presented narratively The 6 trials reporting data where change scores could be calculated were generally very heterogeneous in terms of demographics, including age and gender (gender specific or mixed cancers), FIGO stage (early or advanced disease), and delivery of interventions and controls |
895 participants
(6 studies) Sample sizes were often small, and baseline outcome values for intervention and control groups largely differed widely in 3 further studies. Therefore displaying only studies that used change scores seemed appropriate, and in future updates of the review, attempts at meta‐analysis will be made |
⊕⊝⊝⊝ Very lowa,b,c | |
| Emotional distress |
SMDs (CS) in each individual trial all indicated uncertainty as to whether telephone interventions or control interventions were best for minimising emotional distress (all estimates were imprecise) Cancer sites included breast, prostate, and mixed cancers. There was scope in the breast cancer subgroup to potentially pool results, but there were sufficient clinical differences between the 2 trials in terms of including participants at different stages and ages to justify not using this approach. Therefore results were reported by single trials and are presented narratively The 5 trials reporting data where change scores could be calculated were generally very heterogeneous in terms of demographics, including age and gender (gender specific or mixed cancers), FIGO stage (early or advanced disease), and delivery of interventions and controls. This may have differed in the number of telephone calls and whether additional management components were used in intervention arms, and in controls being sufficiently different to consider any data synthesis methods |
968 participants
(5 studies) Sample sizes were often small, and baseline outcome values for intervention and control groups largely differed widely in 2 further studies. Therefore displaying only studies that used change scores seemed appropriate, and in future updates of the review, attempts at meta‐analysis will be made |
⊕⊝⊝⊝ Very lowa,b,c | |
| Other outcomes included uncertainty, pain, sexually related symptoms, dyspnoea, and general symptoms. Data for any of these outcomes were not pooled due to considerable heterogeneity across all aspects. Magnitudes of effect were not reported | Studies for each outcome ranged from 2 to 6 (10 were included in the wider 'general symptoms' outcome) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; CS: change score; FIGO: International Federation of Gynecology and Obstetrics; SMD: standardised mean difference. | ||||
| GRADE Working Group grades of evidence. High certainty: further research is very unlikely to change our confidence in the estimate of effect. Moderate certainty: further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low certainty: further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low certainty: we are very uncertain about the estimate. | ||||
aDowndraded by one level due to concerns about overall risk of bias being unclear or high.
bDowndraded by one level due to concerns about precision.
cDowndraded by one level due to inconsistencies in results and general heterogeneity.
Background
Description of the condition
People with cancer often experience a variety of symptoms as a result of their disease and its treatment (Harrington 2010;Kim 2012;Van Lancker 2014). As much cancer treatment is delivered on an ambulatory basis, patients and family members are largely responsible for their day‐to‐day management (Dodd 2000;McPherson 2014). Inadequate symptom management can result in early discontinuation of, or delays in, treatment (Cleeland 2009), and it has considerable implications for patient outcomes including functioning, psychological well‐being, and quality of life (Dodd 2001;Glover 1995;Laugsand 2011).
Symptoms often manifest concurrently and appear related to one another. Symptom clusters ‐ where three or more related symptoms manifest concurrently (Dodd 2001) ‐ have become the subject of much contemporary research, with some evidence suggesting that they may have prognostic capabilities or may influence cancer outcomes (Cheville 2010). It is estimated that 40% of oncology patients experience more than one symptom at any one time (Kim 2009), and that as disease progresses, symptom burden rises. One study of 1000 people with cancer admitted to a palliative care unit determined that people experienced differing numbers of symptoms on admission, ranging between 1 and 29. However, the median number of symptoms that people presented with was 11 (Walsh 2000).
Attempts to reduce the incidence and severity of cancer symptoms have involved the development and testing of psycho‐educational interventions to enhance patients' symptom self‐management. These interventions may include therapeutic elements such as information exchange, problem‐solving, coping skills training, and facilitating expression of emotions and concerns (Barsevick 2002). Although traditionally delivered face‐to‐face, interventions are increasingly being delivered by telephone (e.g. Freeman 2015), online (e.g. Steel 2016), or by mobile phone (e.g. Kearney 2009). These alternative modes of delivery are convenient for health professionals and patients alike. Integral to these is the delivery of supportive, interactive care provided by health professionals that provides patients with information about symptom management and support and encouragement in adopting effective self‐care.
Description of the intervention
This review evaluates the effectiveness of telephone interventions delivered to people with cancer, with the aim of improving symptoms of the disease and/or its treatment. These interventions are typically educational or psychologically based in nature and may entail cognitive‐behavioural, motivational, or supportive elements to facilitate patient management of symptoms. They can be delivered to patients alone or in conjunction with informal carers (family or friends). Further, they can be supplemented with face‐to‐face contact with health professionals and digital/online/printed educational materials.
Such interventions are gaining in popularity as health systems worldwide are challenged fiscally from having to care for increasingly ageing populations with limited available resources and soaring pharmacological and other healthcare costs. Interventions delivered by telephone are feasible and acceptable to patients and offer health services a cheaper alternative to interventions delivered face‐to‐face.
How the intervention might work
Telephone interventions for symptom management may vary in terms of the symptom(s) they address, the theoretical frameworks underpinning them, the length of time over which they are delivered, and the training/qualifications of persons providing the telephone contact. However, whatever their make‐up, telephone interventions are united in their potential for providing timely information and support to promote behaviour change and/or adherence with prescribed medications and/or recommended self‐care, thereby enhancing patient outcomes and quality of life.
Why it is important to do this review
Although historically, information and support in managing symptoms were delivered face‐to‐face, increasingly this is not the case. The trend is for care to be provided nearer patients' homes, meaning that people with cancer are typically seeing hospital‐based staff less often. Thus, there is a greater requirement for information and support in symptom management to be provided by other means, such as by telephone. Telephone interventions have been developed for a range of cancer symptoms (Scura 2004). Early indications suggest that these interventions have benefit, as they:
reduce symptom severity;
reduce symptom distress;
enhance self‐management of symptoms; and
facilitate adaptation to symptoms.
However, evidence published to date has not been subject to rigorous systematic review. Four previous literature reviews have explored allied topics. Cox 2003 and Dickinson 2014 appraised and synthesised literature related to cancer follow‐up (by telephone and through use of technology, respectively). Gotay 1998 reviewed outcomes of psychosocial support provided by telephone, and Galway 2012 reviewed psychosocial interventions (but did not focus on their delivery by telephone). Thus, none of these reviews explicitly analysed literature specifically evaluating telephone‐delivered interventions for cancer symptoms. Further, the Gotay 1998 and Cox 2003 reviews are very much out‐of‐date. Thus, there is good justification to undertake a Cochrane systematic review to explore the effectiveness of telephone‐delivered interventions for cancer symptoms.
Objectives
To assess the effectiveness of telephone‐delivered interventions for reducing symptoms associated with cancer and its treatment. To determine which symptoms are most responsive to telephone interventions. To determine whether certain configurations (e.g. with/without additional support such as face‐to‐face, printed or electronic resources) and duration/frequency of intervention calls mediate observed cancer symptom outcome effects.
Methods
Criteria for considering studies for this review
Types of studies
Randomised control trials (RCTs) and quasi‐RCTs that:
compared a telephone intervention with other types of interventions (e.g. a face‐to‐face intervention) and/or usual care; or
compared different models of telephone interventions (i.e. with different content) against each other and/or a third arm comprising usual care.
Types of participants
We included studies evaluating telephone interventions for adult men and women (over 18 years of age) with a clinical diagnosis of cancer, regardless of tumour type, stage of disease, type of treatment, and time of recruitment (e.g. before, during, or after anticancer treatment).
We excluded studies that did not focus on cancer patients, or in which only a portion of the sample consisted of cancer patients.
Types of interventions
We included telephone interventions comprising any number of telephone calls delivered by any health or social care professional to cancer patients, with the stated aim of addressing any physical or psychological symptoms of cancer and its treatment. The interventions were referred to by study author(s) as psychological, psychosocial, psycho‐educational, non‐pharmacological, or supportive.
We excluded interventions that:
were not primarily delivered by telephone (e.g. the main form of contact was face‐to‐face and the patient received a single telephone call to monitor progress), although we did include telephone interventions supported with printed/digital/online materials;
aimed to improve patients' general well‐being or adaptation to cancer including managing fear of recurrence (a common concern following cancer) (i.e. interventions that were not aimed primarily at improving cancer symptoms);
evaluated triaging or monitoring care or treatment compliance; or
were not delivered by a health or social care professional, or if details of the background of the person delivering the intervention could not be obtained.
Some interventions within the review incorporated elements other than telephone support. Thus, we categorised them according to whether they comprised solely telephone intervention or included additional supportive elements (e.g. face‐to‐face consultation, printed materials).
Types of outcome measures
We included data related to symptoms associated with cancer and its treatment, measured by standardised instruments that measured symptoms related to cancer with some evidence of validity and reliability.
Primary outcomes
Anxiety (measured by validated instruments such as the Hospital Anxiety and Depression Scale (HADS) or the State Trait Anxiety Inventory (STAI))
Depressive symptoms (measured by validated instruments such as the Hospital Anxiety and Depression Scale (HADS); the Beck Depression Inventory (BDI); or the Centre for Epidemiologic Studies Depression Scale (CES‐D))
Emotional distress (measured by validated instruments such as the Profile of Mood States (POMS))
Uncertainty from being diagnosed with, and treated for, cancer (as measured by validated instruments such as the Mischel Uncertainty in Illness Scale)
Fatigue (measured by validated instruments such as the Brief Fatigue Inventory (BFI); the Multi‐dimensional Fatigue Inventory (MFI); or the Piper Fatigue Scale)
Pain (measured by validated instruments such as the Brief Pain Inventory (BPI))
Nausea/ vomiting (measured by validated instruments such as the Index of Nausea, Vomiting and Retching (INVR))
Sexually‐related symptoms (measured by validated instruments such as the Index of Sexual Satisfaction; the Female Sexual Function Index; or the International Index of Erectile Function)
Lung cancer symptoms (measured by validated instruments such as the Functional Assessment of Cancer Therapy ‐ Lung Cancer (FACT‐L) or the Memorial Symptom Assessment Scale (MSAS) for dyspnoea items)
Secondary outcomes
Symptom experience
Symptom distress (as measured by validated instruments such as the General Symptom Distress Scale)
Search methods for identification of studies
We applied no language restriction for this review, so non‐English publications were to be translated if necessary. This was not needed.
Electronic searches
We searched the following databases.
Cochrane Central Register of Controlled Trials (CENTRAL; 2019, Issue 1), in the Cochrane Library (Appendix 1).
MEDLINE via OVID (1946 to January 2019) (Appendix 2).
Embase via OVID (1980 to January 2019) (Appendix 3).
Cumulative Index to Nursing and Allied Health Literature (CINAHL) via Athens (1982 to January 2019).
British Nursing Index (1984 to January 2019).
PsycINFO (1989 to January 2019).
The search strategies are provided in the appendices.
Searching other resources
We searched conference proceedings to identify published abstracts, along with SIGLE (System for Information on Grey Literature in Europe) and trial registers for unpublished studies. We searched the reference lists of all included articles to identify additional relevant studies. Finally, we handsearched the following journals from 2007 to 2019.
Cancer.
Journal of Clinical Oncology.
Psycho‐oncology.
Cancer Practice.
Cancer Nursing.
Oncology Nursing Forum.
Journal of Pain and Symptom Management.
Palliative Medicine.
We found no additional studies, We found the studies identified for this review from the databases listed above.
Data collection and analysis
Selection of studies
Two review authors (from the pool of AEH, VHP, AC, KC, and ER) independently assessed the potential relevance of all titles and abstracts identified through the literature searches. We retrieved in full text studies identified by either review author as potentially relevant. Two review authors (of AEH, VHP, AC, and ER) independently assessed each of these studies against the review inclusion and exclusion criteria. A third review author resolved disagreements. Studies that appeared eligible for inclusion but were subsequently judged to not meet the selection criteria were detailed in the Characteristics of excluded studies table, including the specific reason(s) for exclusion (e.g. intervention not delivered by health or social care professionals).
Data extraction and management
We used standardised data extraction forms to extract all available data. Two independent review authors extracted data from each included study. We checked the forms against each other, and when we noted discrepancies, we referred to the original papers. We addressed unresolved discrepancies through discussion and consensus, involving the entire review team when necessary. We contacted study authors to obtain missing data. We extracted and reported on the following data.
Geographic location.
Sample demography (age, gender, tumour type, disease stage, treatment).
Number of participants (including those lost to follow‐up).
Details of randomisation and allocation concealment.
Aim of the intervention.
Details of the intervention (number and frequency of telephone calls; duration of calls; health or social care professional(s) delivering intervention; incorporation of additional elements (face‐to‐face contacts, printed/digital/online materials, email contact)).
Details of control/usual care.
Primary and secondary outcome measures.
Time points at which outcomes were collected and reported (frequency, length of follow‐up).
Reported statistics used to assess validity of results.
Quality assurance processes used to ensure uniformity of intervention delivery (e.g. if intervention providers were trained and/or supervised; if a protocol was used; if an integrity check was described).
When possible, all data extracted were those relevant to an intention‐to‐treat analysis in which participants were analysed in the groups to which they were assigned. When study authors reported on the same piece of research in a series of publications, we considered the main study as the one that depicted the study design in detail and reported on primary outcomes of the study.
We managed data using Review Manager 5.3 (RevMan 2014).
Assessment of risk of bias in included studies
All review authors independently assessed and reported potential bias for each trial using the data extraction form. A third review author (ER) resolved any conflicts. We used the following criteria from the Cochrane Handbook for Systematic Reviews of Interventions as a guide for assessment.
Random sequence generation.
Allocation concealment.
Blinding of participant, providers, outcome assessors, data analysts.
Completeness of outcome data; adequate if less than 20% of patients were lost to follow‐up and reasons for loss to follow‐up were similar in both treatment arms.
Selective reporting/intention‐to‐treat analysis.
Other potential sources of bias.
We incorporated results of the assessment into the review through systematic narrative description and commentary about each of these domains. Further, we constructed a risk of bias graph (Figure 1) and a risk of bias summary (Figure 2).
1.

Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
2.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Measures of treatment effect
We processed data in accordance with guidance provided by the Cochrane Handbook for Systematic Reviews of Interventions. We analysed all outcomes in the review (anxiety, depressive symptoms, emotional distress, fatigue, pain, uncertainty, sexually‐related issues) as continuous variables, reflecting how they were presented by study authors. As some studies had small samples, and because baseline scores for study outcomes tended to vary between intervention and control, we determined to input change scores and their associated standard deviations into the analyses. We extracted these statistics (when reported) and analysed them alongside (1) baseline, endpoint, and follow‐up mean scores and associated standard deviations of outcomes of interest; (2) P values; and (3) numbers of patients who provided data at each assessment point to estimate the standardised mean difference (SMD) of change scores between treatment arms and its standard error.
Dealing with missing data
We did not impute missing outcome data for any of the outcomes other than to calculate missing standard deviations of change scores, as few authors reported these. We imputed these values using the approach of Follmann 1992 and Abrams 2005, as detailed in Section 16.1.3.2 of the Cochrane Handbook for Systematic Reviews of Interventions. This entailed calculating the correlation coefficient from one study in the symptom group that reported study outcomes in detail ‐ including the standard deviation of the change score ‐ and then using the summary statistics to determine standard deviation of change from baseline across other studies. Two study authors provided sufficient detail to enable calculation of standard deviations of change scores for symptoms of anxiety, depression, emotional distress, and fatigue (Downe‐Wamboldt 2007; Ream 2015).
Assessment of heterogeneity
We assessed and found considerable heterogeneity in included studies in terms of (1) interventions introduced; (2) types of participants; and (3) outcomes measured. All interventions were delivered primarily by telephone, but some also incorporated face‐to‐face elements and/or printed, digital, or online materials. Interventions varied in length and frequency and were provided at different times in the cancer journey from treatment to survivorship. There was some standardisation with regards to outcomes measured.
We did not assess methodological and statistical heterogeneity due to considerable clinical heterogeneity across trials. Most studies compared a telephone intervention with usual care, five compared two different interventions with usual care (Badger 2007; Dong 2018; Girgis 2009; Livingston 2010; Thomas 2012), and six compared two interventions without a usual care arm (Badger 2013a; Badger 2013b; Chambers 2014; Reese 2018; Sikorskii 2007; Watson 2017).
For future updates, we plan to assess statistical heterogeneity between study outcomes by visually inspecting forest plots and by calculating the I² statistic (estimation of the percentage of heterogeneity between trials that cannot be ascribed to sampling variation (Higgins 2003)), and when possible, by conducting subgroup analyses (see later). If we find evidence of substantial heterogeneity, we will investigate and report the possible reasons for this.
Data synthesis
We did not perform meta‐analyses due to considerable heterogeneity.
For future updates of the review, we will do the following.
We will use random‐effects models with inverse variance weighting for all meta‐analyses (DerSimonian 1986).
We will calculate SMDs in outcomes between telephone and control groups (rather than mean differences, if appropriate) to take account of the different scales used across studies to measure different symptom outcomes.
When we are unable to obtain required data to incorporate studies into meta‐analyses, or when we identify insufficient studies related to management of a particular symptom, we will continue to report study findings in a narrative fashion.
Subgroup analysis and investigation of heterogeneity
For future updates, we will present subgroups on forest plots and will aim to determine whether there is a difference in outcomes according to whether telephone interventions are provided on their own or in conjunction with other elements (e.g. printed materials, face‐to‐face meetings).
Sensitivity analysis
We found an insufficient number of studies (and no meta‐analyses were conducted) to allow review authors to undertake sensitivity analysis to determine the effect of including/excluding studies with high risk of bias (e.g. as a result of inadequate concealment of allocation).
Results
Description of studies
See Characteristics of included studies and Characteristics of excluded studies.
Results of the search
After duplicates and clearly irrelevant articles were eliminated, the electronic and manual search (to January 2019) yielded 78 studies that were potentially eligible for inclusion. After assessing the full text of studies against the inclusion criteria, we excluded 45 studies, leaving 33 studies for inclusion in the review. Gil 2006 was nested in the Mishel 2005 study because it reported on long‐term outcomes of this study. The PRISMA flow chart is presented in Figure 3.
3.
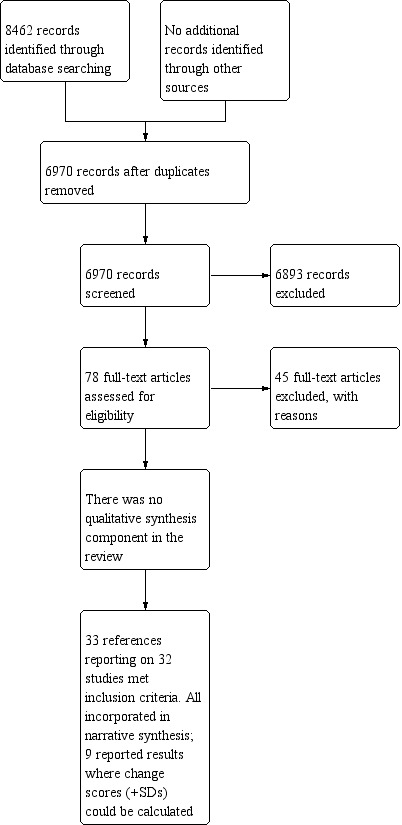
Study flow diagram.
Included studies
Study design
We included 31 randomised controlled trials (RCTs); the final study employed a repeated‐measures experimental design but did not use random principles for assignment to intervention and control groups (Badger 2005). Ten studies appeared adequately powered (i.e. study authors had determined the sample size required to show effect and had managed to recruit to this target) (Allard 2007; Chambers 2014; Girgis 2009; Kroenke 2010; Molassiotis 2009; Mosher 2016; Sherwood 2005; Sikorskii 2007; Traeger 2015; Yates 2005). Three further studies calculated sample sizes required, but due to under‐recruitment (Livingston 2010; Watson 2017), or higher than predicted attrition (Thomas 2012), these studies were underpowered. Most studies did not specify required sample size to attain adequate power. Five were pilot studies with small sample sizes (Badger 2005; Badr 2015; Bailey 2004; Reese 2014; Reese 2018).
Most studies compared a telephone‐delivered intervention with usual care. However, of the included studies, five compared an intervention delivered via telephone with an attentional, rather than usual care, control (Badger 2005; Badger 2007; Barsevick 2004; Barsevick 2010; Mosher 2016). A further study provided a wait‐list control whereby participants in the control group received the intervention on completion of study assessments (Reese 2014). Four studies compared a telephone‐delivered intervention against augmented usual care, which incorporated additional elements including passive referral to a help line; education/support; nutrition information; or extra information about symptoms reported by patients given to the treating oncologist (Barsevick 2004; Kroenke 2010; Mosher 2016; Porter 2011). Six studies compared two/three interventions but incorporated no control group (Badger 2013a; Badger 2013b; Chambers 2014; Reese 2018; Sikorskii 2007; Watson 2017). One was an equivalence trial (Watson 2017). Eight studies included three study arms that incorporated telephone‐delivered intervention(s) with alternative intervention with/without a control group (usual care or attentional control) (Badger 2007; Badger 2013a; Chambers 2015; Dong 2018; Girgis 2009; Livingston 2010; Mishel 2002; Thomas 2012).
Contact with study authors
We contacted five study authors to clarify issues around provision of the intervention; we subsequently excluded two articles from the review, as data obtained from the study author made it clear that these studies were ineligible. We included three studies once the nature of the interventions and who delivered them were clarified (Badger 2013a; Reese 2014; Reese 2018). Further, we contacted one study author with regards to the Mishel 2002 study, to query numbers of participants in each study arm.
Sample size
Sample sizes ranged from 23 in Reese 2014 to 575 in Mishel 2005, with a total of 6250 cancer patients recruited across the 32 studies.
Setting
Twenty‐one studies were conducted in the USA (Allen 2002; Badger 2005; Badger 2007; Badger 2013a; Badger 2013b; Badr 2015; Bailey 2004; Barsevick 2004; Barsevick 2010; Kroenke 2010; Mishel 2002; Mishel 2005; Mosher 2016; Porter 2011; Rawl 2002; Reese 2014; Reese 2018; Sherwood 2005; Sikorskii 2007; Thomas 2012; Traeger 2015). Four were conducted in Australia (Chambers 2014; Chambers 2015; Girgis 2009; Yates 2005), two in Canada (Allard 2007; Downe‐Wamboldt 2007), three in the UK (Molassiotis 2009; Ream 2015; Watson 2017), and one in China (Dong 2018), and one recruited across two countries ‐ Australia and Canada (Livingston 2010).
Participants
Nine studies addressed breast cancer exclusively (Allard 2007; Allen 2002; Badger 2005; Badger 2007; Badger 2013a; Badger 2013b; Mishel 2005; Reese 2018; Yates 2005), three solely recruited men with prostate cancer (Bailey 2004; Chambers 2015; Mishel 2002), three recruited only lung cancer patients (Badr 2015; Mosher 2016; Porter 2011), and two recruited only people diagnosed with colorectal cancer (Dong 2018; Reese 2014).
Eleven studies included heterogeneous samples of cancer patients, most commonly with a combination of breast, colorectal, lung, and prostate cancer (Barsevick 2010; Chambers 2014; Downe‐Wamboldt 2007; Girgis 2009; Kroenke 2010; Livingston 2010; Molassiotis 2009; Rawl 2002; Ream 2015; Traeger 2015; Watson 2017); four studies did not specify the type of cancer diagnosis patients had received (Barsevick 2004; Sherwood 2005; Sikorskii 2007; Thomas 2012).
In addition to including people with cancer, 11 studies addressed partners or carers of cancer patients; five included partners of women with breast cancer (Badger 2005; Badger 2007; Badger 2013a; Badger 2013b; Reese 2018), one included partners of men with prostate cancer (Chambers 2015), one included partners of patients treated for colorectal cancer (Reese 2014), one included partners of people diagnosed with lung cancer (Badr 2015), two included carers of people diagnosed with lung cancer (Mosher 2016; Porter 2011), and one included carers of people with a range of cancers (Chambers 2014). Outcomes for the partners/carers reported in these studies are not included in this review.
Most studies (n = 14) recruited patients with early‐stage cancer. Remaining studies recruited patients with early/locally advanced cancer (n = 2) (Badger 2005; Reese 2018), or people with advanced cancer (n = 4) (Badr 2015; Girgis 2009; Molassiotis 2009; Sherwood 2005), or they did not (n = 11) specify the disease stage for eligible patients (Badger 2005; Barsevick 2010; Chambers 2014; Dong 2018; Kroenke 2010; Mosher 2016; Sikorskii 2007; Ream 2015; Reese 2014; Thomas 2012; Watson 2017). One study specifically targeted cancer survivors who were between five and nine years post treatment (Mishel 2005).
Most studies recruited consecutive patients irrespective of symptom intensity; few (n = 6) recruited people whose symptoms had attained a threshold level (Chambers 2014; Dong 2018; Kroenke 2010; Mosher 2016; Ream 2015; Reese 2018 ).
Symptoms
Interventions introduced across studies aimed to reduce a variety of symptoms caused by cancer and its treatment; psychological and emotional symptoms were frequently assessed. Sixteen studies measured effects of telephone‐delivered interventions on anxiety (Badger 2007; Badger 2013b; Badr 2015; Bailey 2004; Dong 2018; Girgis 2009; Livingston 2010; Molassiotis 2009; Mosher 2016; Porter 2011: Rawl 2002; Ream 2015; Reese 2018; Traeger 2015; Watson 2017; Yates 2005). Depressive symptoms was an outcome measured in 21 studies (Badger 2005; Badger 2007; Badger 2013a; Badger 2013b; Badr 2015; Bailey 2004; Barsevick 2010; Dong 2018; Downe‐Wamboldt 2007; Girgis 2009; Kroenke 2010; Livingston 2010; Molassiotis 2009; Mosher 2016; Porter 2011: Rawl 2002; Ream 2015; Reese 2018; Traeger 2015; Watson 2017; Yates 2005). Seven studies focused more broadly on emotional distress (Allard 2007; Allen 2002;Bailey 2004; Chambers 2014; Downe‐Wamboldt 2007; Livingston 2010; Mishel 2005). Finally, three interventions aimed to alleviate uncertainty resulting from diagnosis of, and treatment for, cancer (Bailey 2004; Mishel 2002; Mishel 2005).
With regards to symptoms with a physical element, nine studies measured the impact of interventions on fatigue (Badger 2005; Badger 2013b; Bailey 2004; Barsevick 2004; Barsevick 2010; Molassiotis 2009; Mosher 2016; Ream 2015; Yates 2005), six on cancer‐related pain (Barsevick 2010; Kroenke 2010; Molassiotis 2009; Mosher 2016; Porter 2011: Thomas 2012), three on sexuallly‐related symptoms (Chambers 2015; Reese 2014; Reese 2018), and two on dyspnoea (Mosher 2016; Porter 2011).
Ten studies measured general symptom experience and reported overall symptom intensity and/or symptom distress (Badger 2013a; Badger 2013b; Barsevick 2010; Kroenke 2010; Mishel 2002; Molassiotis 2009; Porter 2011Sherwood 2005; Sikorskii 2007; Traeger 2015).
Many symptoms were incorporated as secondary outcomes across studies. For example, a study evaluating a telephone‐delivered intervention for fatigue may also have incorporated anxiety and/or depressive symptoms as secondary outcomes.
Intervention format
A summary of intervention characteristics is provided in the table titled Characteristics of included studies. All interventions were delivered primarily by telephone to participants in their homes. However, only ten were delivered solely by telephone (Allard 2007; Badger 2005; Badger 2007; Badger 2013b; Bailey 2004; Dong 2018; Downe‐Wamboldt 2007; Livingston 2010; Reese 2014; Traeger 2015).
Sixteen of the remaining study interventions were delivered by telephone in combination with printed materials and/or online/digital materials (Badger 2013a; Badr 2015; Barsevick 2004; Barsevick 2010; Chambers 2014; Chambers 2015; Girgis 2009; Mishel 2002; Mishel 2005; Mosher 2016; Porter 2011; Ream 2015; Reese 2018; Sikorskii 2007; Thomas 2012; Watson 2017).
Two studies combined telephone calls with face‐to‐face sessions (Molassiotis 2009; Sherwood 2005), and three combined telephone calls with both face‐to‐face sessions and digital/printed materials (Allen 2002; Rawl 2002; Yates 2005). One final study evaluated an intervention that incorporated automated symptom monitoring and prescribing recommendations (regarding depressive symptoms and fatigue) made to participants by oncologists, in addition to telephone calls (Kroenke 2010).
Health professionals delivering interventions
Most interventions (n = 24) were delivered by nurses (Allard 2007; Allen 2002; Badger 2005; Badger 2007; Bailey 2004; Barsevick 2004; Barsevick 2010; Chambers 2014; Chambers 2015; Downe‐Wamboldt 2007; Girgis 2009; Kroenke 2010; Livingston 2010; Mishel 2002; Mishel 2005; Molassiotis 2009; Porter 2011, Rawl 2002; Ream 2015; Sherwood 2005; Sikorskii 2007; Thomas 2012; Traeger 2015; Yates 2005), including:
oncology nurses (Barsevick 2010; Chambers 2014; Chambers 2015; Girgis 2009; Livingston 2010; Rawl 2002; Ream 2015; Sherwood 2005; Sikorskii 2007; Thomas 2012; Traeger 2015; Yates 2005);
research nurses (Allen 2002; Barsevick 2004); and
psychiatric nurses (Badger 2005; Badger 2007).
Eight studies did not specify the specialty or training of nurses who delivered the interventions (Allard 2007; Bailey 2004; Downe‐Wamboldt 2007; Kroenke 2010; Mishel 2002; Mishel 2005; Molassiotis 2009; Porter 2011). In nine studies, the nurses had received training in other skills necessary for intervention delivery, including counselling (Badger 2005; Badger 2007; Barsevick 2004; Downe‐Wamboldt 2007; Girgis 2009; Livingston 2010; Ream 2015), communication (Chambers 2015), and education (Kroenke 2010).
In nine studies, interventions were delivered by professionals other than nurses. These included psychologists (Chambers 2014; Dong 2018; Reese 2014; Reese 2018; Watson 2017), social workers (Badger 2013a; Mosher 2016), a mental health counsellor (Badr 2015), and master's prepared social workers and para‐professionals/psychologists/counsellors (Badger 2013b).
Theoretical basis of interventions
Twenty‐one studies used theoretical models to inform the telephone intervention: Self‐Regulation Theory (Allard 2007; Rawl 2002; Ream 2015), Interpersonal Therapy (Badger 2005; Badger 2007; Badger 2013a), the Stress Process Model (Badger 2013b), the Common Sense Model of Illness (Barsevick 2004; Barsevick 2010), the Transtheoretical Model (Thomas 2012), Self‐Determination Theory (Badr 2015), Cognitive‐Behavioural Theory/Coping Skills Training (Downe‐Wamboldt 2007; Porter 2011; Sherwood 2005), the PRECEDE Model of Health Behaviour (Yates 2005), Social Cognitive Theory (Mosher 2016), the Theory of Uncertainty in Illness (Bailey 2004; Mishel 2002; Mishel 2005), and the Cognitive‐Behavioural and Sex Therapy Theory (Reese 2014; Reese 2018).
The theoretical basis informing the design of the interventions was unclear ‐ or unreported ‐ in 11 studies (Allen 2002; Chambers 2014; Chambers 2015; Dong 2018; Girgis 2009; Kroenke 2010; Livingston 2010; Molassiotis 2009; Sikorskii 2007; Traeger 2015; Watson 2017). Although Allen 2002 did not specify the theoretical basis for the intervention provided, the intervention model for this study was based around problem‐solving and made use of motivational techniques in its delivery. Finally, Sikorskii 2007 employed a multi‐dimensional interactive approach, which drew on various strategies around coping, re‐framing, providing education, and eliciting support for adapting to, or overcoming, cancer‐related symptoms.
Number of calls/duration/timing
Most (n = 29) of the interventions provided a standardised number of telephone calls to participants; these ranged from one call in Chambers 2014 to 18 calls delivered weekly over 18 weeks in Molassiotis 2009. In the remaining three studies, the number of calls provided varied by need (Downe‐Wamboldt 2007; Watson 2017), or the numbers of calls and the intervals between them were unclear (Girgis 2009).
Although most (n = 17) intervention calls were delivered weekly, three interventions were delivered every other week (Allen 2002; Rawl 2002; Thomas 2012). One intervention delivered three calls over three successive cycles of chemotherapy (Ream 2015), one provided four calls over two successive cycles of chemotherapy (Traeger 2015), and seven others provided a series of calls with increasing time intervals between them (Chambers 2015; Girgis 2009; Kroenke 2010; Livingston 2010; Porter 2011; Sherwood 2005; Sikorskii 2007). In one study, calls were made over a three‐month period at participants' convenience (Downe‐Wamboldt 2007), and in another, up to eight calls were again scheduled over an approximate 12‐week period (Watson 2017). Finally, Chambers 2014 made five calls but did not specify the timing of these calls.
Most frequently, the intervention comprised three or four calls (Allen 2002; Badger 2013a; Barsevick 2004; Barsevick 2010; Girgis 2009; Livingston 2010; Mishel 2005; Mosher 2016; Ream 2015; Reese 2014; Reese 2018; Sherwood 2005; Thomas 2012; Traeger 2015; Watson 2017). In three studies, participants received more than 10 calls: Porter 2011 delivered 14; Molassiotis 2009 provided 18; and although Kroenke 2010 planned to deliver four calls, automated symptom reports with high symptom scores triggered extra calls ‐ participants received on average 11 calls.
Twenty‐two studies reported the duration of calls provided (Badger 2005; Badger 2007; Badger 2013a; Badger 2013b; Badr 2015; Bailey 2004; Barsevick 2004; Chambers 2014; Chambers 2015; Dong 2018; Kroenke 2010; Livingston 2010; Molassiotis 2009; Mosher 2016; Porter 2011; Rawl 2002; Ream 2015; Reese 2014; Reese 2018; Thomas 2012; Traeger 2015; Yates 2005); duration ranged on average from 10 minutes in Yates 2005 to 70 minutes in Reese 2018.
Recording and documentation of calls
Only 15 studies recorded telephone calls in some way for quality assurance (Badger 2005; Badger 2007; Badger 2013b; Barsevick 2004; Barsevick 2010; Chambers 2014; Chambers 2015; Livingston 2010; Mosher 2016; Porter 2011, Rawl 2002; Ream 2015; Reese 2018; Sherwood 2005; Traeger 2015). Downe‐Wamboldt 2007 assessed fidelity by reviewing the interventionist's notes to determine goals recorded and degree of problem‐solving achieved. Watson 2017 addressed fidelity by observing some of the intervention sessions.
Telephone calls were additionally documented in some way as part of the intervention (not explicitly for quality purposes): two used patient‐completed worksheets completed after calls (Allen 2002; Badger 2007); four obtained feedback surveys/documentation completed on a computer or via a touchpad telephone (Girgis 2009; Kroenke 2010; Livingston 2010; Sikorskii 2007); one reviewed and assigned homework during intervention calls (Badr 2015); another required the nurse delivering the intervention to keep a diary recording participants' engagement with calls (Ream 2015); and two used patient diaries completed between telephone calls to help tailor the intervention (Barsevick 2004; Barsevick 2010).
Excluded studies
In the Characteristics of excluded studies table, we list the 45 studies excluded after assessment of full text (according to the criteria specified at Types of interventions) and specify the reasons for exclusion.
We excluded studies primarily because:
they did not address management of symptoms (n = 21); or
interventions were not delivered primarily by telephone (n = 12).
Risk of bias in included studies
Assessments of risk of bias and methodological certainty are provided in the Characteristics of included studies table and the Risk of bias graph (Figure 1).
Here we summarise risk of bias.
Adequate sequence generation: fulfilled in 22 studies (Allard 2007; Bailey 2004; Barsevick 2010; Chambers 2014; Chambers 2015; Dong 2018; Downe‐Wamboldt 2007; Girgis 2009; Kroenke 2010; Livingston 2010; Mishel 2005; Molassiotis 2009; Mosher 2016; Porter 2011 Rawl 2002; Ream 2015; Reese 2018; Sikorskii 2007; Thomas 2012; Traeger 2015; Watson 2017; Yates 2005).
Allocation concealment: fulfilled in 10 studies (Barsevick 2010; Chambers 2014; Chambers 2015; Dong 2018; Downe‐Wamboldt 2007; Molassiotis 2009; Porter 2011; Thomas 2012; Traeger 2015; Yates 2005).
Blinding: fulfilled in six studies (Kroenke 2010; Molassiotis 2009; Porter 2011; Rawl 2002; Ream 2015; Sherwood 2005).
Incomplete outcome data assessed: fulfilled in 17 studies (Badger 2007; Badger 2013b; Badr 2015; Bailey 2004; Barsevick 2010; Chambers 2015; Dong 2018; Downe‐Wamboldt 2007; Girgis 2009; Livingston 2010; Mishel 2005; Molassiotis 2009; Ream 2015; Reese 2014; Reese 2018; Traeger 2015; Yates 2005).
Free of selective reporting: fulfilled in 30 studies (Allard 2007; Allen 2002; Badger 2005; Badger 2007; Badger 2013a; Badger 2013b; Badr 2015; Bailey 2004; Barsevick 2004; Barsevick 2010; Chambers 2014; Chambers 2015; Dong 2018; Downe‐Wamboldt 2007; Girgis 2009; Kroenke 2010; Mishel 2002; Mishel 2005; Molassiotis 2009; Mosher 2016; Porter 2011; Rawl 2002; Ream 2015; Reese 2014; Reese 2018; Sherwood 2005; Sikorskii 2007; Thomas 2012; Traeger 2015; Yates 2005).
Other bias: detected in 12 studies (Allard 2007; Allen 2002; Badger 2013b; Badr 2015; Chambers 2014; Dong 2018; Mishel 2005; Porter 2011; Ream 2015; Reese 2014; Traeger 2015; Yates 2005).
Effects of interventions
See: Table 1
1. Findings by symptom
1.1 Anxiety; telephone intervention versus control
Sixteen studies measured effects of their interventions on anxiety (Badger 2007; Badger 2013b; Badr 2015; Bailey 2004; Dong 2018; Girgis 2009; Livingston 2010; Molassiotis 2009; Mosher 2016; Porter 2011; Rawl 2002; Ream 2015; Reese 2018; Traeger 2015; Watson 2017; Yates 2005). These studies used several different validated scales.
Hospital Anxiety and Depression Scale (Girgis 2009; Livingston 2010; Molassiotis 2009; Ream 2015; Watson 2017; Yates 2005).
State version of the State‐Trait Anxiety Inventory (STAI) (Badger 2013b; Rawl 2002).
Trait Anxiety version of the State‐Trait Anxiety Inventory (STAI) ( Porter 2011).
Anxiety subscale of the Profile of Mood States ‐ Short Form (Bailey 2004).
Hamilton Anxiety Scale (HAMA) (Chinese version) (Dong 2018).
Self‐Rating Anxiety Scale (SAS) (Chinese version) (Dong 2018).
Investigator‐designed instrument composed of the Positive and Negative Affect Schedule (PANAS), the Short Form‐12 (SF‐12) Scale, and the Index of Clinical Stress (ICS) (Badger 2007).
6‐item Patient‐Reported Outcomes Measurement Information System short form anxiety measure (Badr 2015).
2‐item Generalised Anxiety Disorder (GAD‐2) Scale (Traeger 2015); 7‐item Generalised Anxiety Disorder (GAD‐7) Scale (Mosher 2016; Reese 2018).
In nine studies, anxiety was a primary outcome (Badger 2007; Badger 2013b; Badr 2015; Girgis 2009; Livingston 2010; Mosher 2016; Porter 2011; Rawl 2002; Watson 2017). Two of these (Badger 2007; Badr 2015), plus three studies in which anxiety was a secondary outcome (Bailey 2004; Dong 2018; Ream 2015), provided data from 277 participants that reported change scores and associated standard deviations ‐ or these could be calculated. Dong 2018 measured anxiety outcomes with two measures (HAMA and SAS); only those related to SAS are displayed on the forest plot. This decision was based on the allied Self‐Rating Depression Scale, which was used to power the study. Three individual studies appeared to suggest that use of a telephone had a significant impact on symptom management of anxiety in participants with various types of cancer (Badger 2007; Badr 2015; Dong 2018), but another two trials found no evidence of a difference (Bailey 2004; Ream 2015). We did not pool the overall effect estimates due to considerable heterogeneity, but we have depicted results of individual trials in the forest plot in Analysis 1.1.
1.1. Analysis.

Comparison 1: Symptoms, Outcome 1: Anxiety
It is interesting to note that Dong 2018, a three‐arm trial incorporating a telephone support arm in addition to telephone‐delivered reminiscence therapy and usual care control, determined that generic telephone support generated similar improvements in anxiety as telephone‐based reminiscence therapy.
Of the trials that did not report a magnitude of effect explicitly nor address baseline imbalance using change scores (or these and their associated standard deviations could not be calculated) ‐ and in which anxiety was a primary outcome ‐ three reported some effect on anxiety. Badger 2013b tested two different forms of telephone‐delivered intervention without usual care control. Although the study determined that interventions were associated with statistically significant decreases in anxiety, without a control group for comparison it is difficult to conclude that these improvements were generated by the intervention rather than by an alternative factor (including passage of time). Watson 2017 also had no control group, but this was an equivalence trial ‐ researchers were seeking to determine equivalence between standard and telephone‐delivered cognitive‐behavioural therapy (CBT) in people with high psychological needs; conventional CBT has established efficacy. Watson 2017 determined that with regards to anxiety, telephone‐delivered and face‐to‐face CBT approaches were equally effective. Finally, Rawl 2002, in the trial report, noted improvements in anxiety nearing statistical significance (P = 0.09). The other four studies did not detect any significant intervention effect (Girgis 2009; Livingston 2010; Mosher 2016; Porter 2011).
1.2 Depressive symptoms; telephone intervention versus control
Twenty‐one studies measured effects of interventions on symptoms of depression (Badger 2005; Badger 2007; Badger 2013a; Badger 2013b; Badr 2015; Bailey 2004; Barsevick 2010; Dong 2018; Downe‐Wamboldt 2007; Girgis 2009; Kroenke 2010; Livingston 2010; Molassiotis 2009; Mosher 2016; Porter 2011; Rawl 2002; Ream 2015; Reese 2018; Traeger 2015; Watson 2017; Yates 2005).
The following validated measurement scales were used to measure depressive symptoms.
Centre for Epidemiologic Studies Depression Scale (CES‐D) (Badger 2005; Badger 2007; Badger 2013a; Badger 2013b; Downe‐Wamboldt 2007; Rawl 2002).
Hospital Anxiety and Depression Scale (Girgis 2009; Livingston 2010; Molassiotis 2009; Ream 2015; Watson 2017; Yates 2005).
Profile of Mood States ‐ Short Form (POMS‐SF) (Bailey 2004; Barsevick 2010).
Hamilton Depression Scale (HAMD) (Chinese version) (Dong 2018).
Self‐Rating Depression Scale (SDS) (Chinese version) (Dong 2018).
Depression severity subscale of the 36‐item Short Form Health Survey (SF‐36) (Kroenke 2010).
Short Form‐12 (SF‐12) (Barsevick 2010).
Hopkins Symptom Checklist (HSCL‐20) (Kroenke 2010).
Patient Health Questionnaire (PHQ‐9) (Kroenke 2010; Mosher 2016; Reese 2018).
2‐item Patient Health Health Questionnaire (PHQ‐2) (Traeger 2015).
Beck Depression Inventory (BDI) (Porter 2011).
6‐item Patient‐Reported Outcomes Measurement Information System short‐form depression measure (Badr 2015).
In all but six studies (Bailey 2004; Barsevick 2010; Molassiotis 2009; Ream 2015; Traeger 2015; Yates 2005), depressive symptoms was the primary outcome. Nine studies, including 1059 participants, reported quantitative results such as change scores ‐ or data enabling these and their associated standard deviations to be calculated (Badger 2005; Badger 2007; Badr 2015; Bailey 2004; Barsevick 2010; Dong 2018; Downe‐Wamboldt 2007; Kroenke 2010; Ream 2015) (Analysis 1.2). Dong 2018 measured depressive symptom outcomes with two measures (HAMD and SDS); only those related to SDS were included in the forest plot. This decision was based on use of the SDS to power the study. Three of these studies had measured depression as a secondary outcome (Bailey 2004; Barsevick 2010; Ream 2015). Four individual studies appeared to suggest that use of a telephone had significant impact on symptom management of depression among participants with various types of cancer (Badger 2007; Badr 2015; Dong 2018; Kroenke 2010); Downe‐Wamboldt 2007 was of borderline significance. Many trials were small and underpowered and did not report magnitude of effect. We did not pool the overall effect estimates due to considerable heterogeneity, but we have depicted the results of individual trials in the forest plot in Analysis 1.2.
1.2. Analysis.

Comparison 1: Symptoms, Outcome 2: Depression
Of trials that did not report a magnitude of effect explicitly nor address baseline imbalance using change scores (or these and their associated standard deviations could not be calculated) ‐ and that had symptoms of depression as a primary outcome ‐ four reported some improvement in the intervention arm. Rawl 2002 measured the impact of a telephone‐delivered intervention on depressive symptoms in patients with newly diagnosed cancer. Researchers found that patients who received the intervention had significantly fewer depressive symptoms (P = 0.05) midway through the intervention when compared to those in the usual care group, although this was not maintained. One month post intervention, the difference was no longer statistically significant (P = 0.07). Porter 2011 detected reduced symptoms of depression in lung cancer patients following provision of both of their two telephone‐delivered interventions (coping skills training (CST) versus education/support) (B = ‐5.55, standard error = 0.28, P = 0.05). As with the intervention's effects on anxiety, levels of depressive symptoms dropped more for patients with stage I cancer given the education/support intervention, and more for patients with stage II‐III cancer given the CST intervention (B = ‐2.38, standard error = 2.86, P = 0.006). Badger 2013a and Badger 2013b reported statistically significant decreases in depressive symptoms over time associated with delivery of two forms of telephone‐delivered intervention. However, neither study incorporated a usual care control for comparison. Finally, Watson 2017, an equivalence trial comparing face‐to‐face and telephone‐delivered CBT, determined that the telephone‐delivered version generated a statistically significant reduction in depressive symptoms equivalent to that seen with CBT delivered in‐person.
Three studies whose primary outcome was reduced depressive symptoms found no significant reductions in these generated by the interventions (Girgis 2009; Livingston 2010; Mosher 2016).
1.3 Fatigue; telephone intervention versus control
Nine studies reported on effects of interventions on fatigue. Six evaluated interventions delivered specifically to reduce fatigue (Badger 2005; Barsevick 2004; Barsevick 2010; Mosher 2016; Ream 2015; Yates 2005). Another three studies measured fatigue as a secondary outcome arising from the interventions (Badger 2013b; Bailey 2004; Molassiotis 2009).
Fatigue was measured using the following measurement tools.
Multi‐Dimensional Fatigue Inventory (MFI) (Badger 2005; Badger 2013b).
Brief Fatigue Inventory (BFI) (Ream 2015).
Fatigue Distress Scale (Ream 2015).
Schwartz Cancer Fatigue Scale (Barsevick 2004).
General Fatigue Scale (GFS) (Barsevick 2004; Barsevick 2010).
Profile of Mood States (POMS), fatigue subscale (Barsevick 2004; Barsevick 2010).
National Cancer Institute Common Toxicity Criteria (NCI‐CTC) ‐ single item (Molassiotis 2009).
Fatigue Symptom Inventory (Mosher 2016).
Six studies reported quantitative results including change scores ‐ or data enabling these and their associated standard deviations to be calculated (Badger 2005; Bailey 2004; Barsevick 2004; Barsevick 2010; Ream 2015; Yates 2005). All but Bailey 2004 incorporated fatigue as a primary outcome. Collectively, trial authors had attained data on 895 participants. Three of the individual studies appeared to suggest that use of the telephone had a significant impact on symptom management of fatigue for participants with various types of cancer (Badger 2005; Barsevick 2004; Yates 2005). Although the intervention evaluated by Ream 2015 in a feasibility trial did not significantly reduce overall (global) fatigue, study authors reported that it did generate significant reductions in distress caused by the symptom (P < 0.05). Other studies found no evidence of any differences between arms (Bailey 2004; Barsevick 2010). We did not pool the overall effect estimates due to considerable heterogeneity, but we have depicted the results of individual trials in the forest plot in Analysis 1.3.
1.3. Analysis.

Comparison 1: Symptoms, Outcome 3: Fatigue
Molassiotis 2009 reported significant reductions in fatigue generated by a telephone‐delivered home care intervention delivered weekly over 18 weeks to people given a course of oral capecitabine. However, differences in comparison with the usual care control declined over time. Greatest improvement in fatigue was found from cycle 0 to 2 (P = 0.005); by the end of treatment, these benefits were no longer statistically significant (P = 0.93). Badger 2013a, which evaluated two forms of telephone‐delivered health education and counselling (without usual care control), reported statistically significant reductions in fatigue (P < 0.05) through telephone‐delivered interventions. However, the pilot study Mosher 2016 did not detect any statistically significant benefit of a telephone‐delivered intervention aimed at reducing a broad range of symptoms associated with lung cancer (one of which was fatigue).
1.4 Emotional distress; telephone intervention versus control
Seven studies investigated effects of interventions on emotional distress (Allard 2007; Allen 2002; Bailey 2004; Chambers 2014; Downe‐Wamboldt 2007; Livingston 2010; Mishel 2005).
Definitions of emotional distress and tools used to measure it varied across studies. The following measurement tools were used.
Profile of Mood States (POMS) (Allard 2007; Bailey 2004; Mishel 2005).
Brief Symptom Inventory‐18 (BSI‐18) (Chambers 2014).
Medical Outcomes Study 36‐item Short Form (SF‐36), Mental Health Index subscale (Allen 2002).
Derogatis Psychosocial Adjustment to Illness Scale ‐ Self‐Report (PAIS‐SR) (Downe‐Wamboldt 2007).
Rosebaum's Self‐Control Schedule (SCS) (Bailey 2004).
Investigator‐developed scale originally developed for breast cancer patients (Livingston 2010).
Five studies including 968 participants reported quantitative results including change scores ‐ or data enabling these and their associated standard deviations to be calculated (Allard 2007; Allen 2002; Bailey 2004; Downe‐Wamboldt 2007; Mishel 2005) (Analysis 1.4). None of these studies reached significance. We did not pool the overall effect estimates due to considerable heterogeneity, but we have depicted the results of individual trials in the forest plot in Analysis 1.4.
1.4. Analysis.

Comparison 1: Symptoms, Outcome 4: Emotional distress
Livingston 2010 evaluated effects of two telephone interventions on emotional distress but provided insufficient data to enable calculation of change scores (and standard deviations). Study authors report that although both interventions appeared to reduce cancer‐specific distress, neither generated statistically significant improvements when compared with the control. Chambers 2014 determined that both study arms (single‐session oncology nurse‐delivered telephone intervention and five‐session psychologist‐delivered telephone intervention) were associated with decreased emotional distress. However, once again, lack of a 'no treatment' control group renders it impossible to conclude whether this resulted from the interventions over other factors such as passage of time.
1.5 Uncertainty; telephone intervention versus control
Three papers reported the impact of interventions on feelings of uncertainty in relation to a patient's illness (Bailey 2004; Mishel 2002; Mishel 2005). All three based their interventions on the same theoretical framework ‐ Mishel's Uncertainty Theory ‐ and focused on reducing uncertainty through strategies such as cognitive re‐framing of threatening events and problem‐solving strategies. These papers did not report change scores, or it was impossible to calculate these alongside the standard deviation of the change score, and meta‐analysis was not possible.
Uncertainty was measured using the following instruments.
Mishel's Uncertainty in Illness Scale (Mishel 2002).
Self‐Control Scale ‐ Problem‐Solving and Cognitive Re‐framing subscales (Bailey 2004; Mishel 2002; Mishel 2005).
Growth Through Uncertainty Scale (GTUS) (Bailey 2004).
Confusion subscale of the Profile of Mood States ‐ Short Form (POMS‐SF) (Bailey 2004).
Bailey 2004 delivered a watchful waiting intervention by telephone to men with prostate cancer. Researchers detected no statistically significant differences between group overall scores for uncertainty management. However, the intervention group displayed a significant improvement on the 'New view of life' subscale compared to the usual care group (P = 0.02). Mishel 2002 also addressed uncertainty in men with prostate cancer; these investigators delivered two telephone‐delivered interventions to men following surgery for their disease. The interventions differed only in that one group had supplementary delivery of it to a close family member. Study authors reported significant improvements in uncertainty management (F[16,438] = 1.96; P = 0.01), cognitive re‐framing (F[4456] = 3.81; P = 0.005), and problem‐solving (F[4456] 2.40; P = 0.049) in both intervention groups when compared with the control group immediately following intervention delivery, but these differences were not maintained over time.
Later, Mishel 2005 evaluated an uncertainty intervention in long‐term breast cancer survivors and found a statistically significant difference for cognitive re‐framing (proxy for uncertainty management) (P = 0.01). Outcomes were more pronounced in African American women (P = 0.03) than in White women. Late outcomes (20 months) of the intervention were reported in the subsequent publication of Mishel 2005 by Gil 2006; these results confirmed that benefits generated by the intervention were maintained over time (F[1479] = 3.94; P < 0.05, d = 0.06, n2 = 0.008). Further, women in the intervention group reported decreased illness uncertainty, whereas there was no change for women in the control group from baseline to 20 months (Wilk's lambda F(1479) = 4.85; P < 0.03, d = 0.09, n2 = 0.010).
1.6 Pain; telephone intervention versus control
Six studies examined interventions to relieve cancer‐related pain (Barsevick 2010; Kroenke 2010; Molassiotis 2009; Mosher 2016Porter 2011; Thomas 2012). These trials did not report change scores, or it was impossible to calculate these alongside the standard deviation of the change score, and meta‐analysis was not possible. Researchers measured outcomes using the following tools.
Brief Pain Inventory (BPI) (Barsevick 2010; Kroenke 2010; Mosher 2016; Porter 2011; Thomas 2012).
Cancer Institute Common Toxicity Criteria (NCI‐CTC) pain item (Molassiotis 2009).
Bodily Pain Scale from the Medical Outcomes Study 36‐item Short Form (SF‐36) (Kroenke 2010).
Barriers Questionnaire (BQ) (Thomas 2012).
Three of these studies reported statistically significant reductions in pain. Kroenke 2010 measured pain at four study points following telephone intervention (1, 3, 6, and 12 months). Across all points, the intervention group reported significantly greater improvements than the usual care group for pain severity (P < 0.0001), interference in functioning caused by pain (P < 0.0001), and bodily pain (P = 0.004). Thomas 2012 tested effectiveness of two interventions compared to usual care in decreasing intensity of cancer pain by lowering patients' attitudinal barriers to pain management. Trial authors found no differences between groups at the end of the study in terms of barriers to pain management, average pain intensity scores (F = 2.58; P = 0.08), pain relief (F = 2.63; P = 0.07), or overall body pain (F = 2.817; P = 0.062). However, post hoc contrasts demonstrated that the coaching group had significantly lower mean pain interference scores compared to the education and usual care groups (F = 4.53; P = 0.03 and P = 0.02, respectively) at the end of the study. Molassiotis compared an 18‐week home care intervention for people with breast or colorectal cancer given oral chemotherapy. Measures were taken weekly, and across all, the intervention group reported significantly less pain when compared with the control group (ranging between P < 0.0005 and P < 0.001).
The remaining three studies did not generate statistically significant results (Barsevick 2010; Mosher 2016; Porter 2011).
1.7 Sexually‐related symptoms; telephone intervention versus control
Three studies focused on the impact of the intervention on sexually‐related symptoms including sexual function and satisfaction (Chambers 2015; Reese 2014; Reese 2018). These trials did not report change scores, or it was impossible to calculate these alongside the standard deviation of the change score, and meta‐analysis was not possible.
These researchers measured outcomes using the following.
International Index of Erectile Function (Chambers 2015; Reese 2014; Reese 2018).
Female Sexual Function Index (Reese 2014; Reese 2018).
Female Sexual Distress Scale (Reese 2018).
PROMIS SexFS v2. Global Sexual Satisfaction Scale (Reese 2018).
Sexual needs subscale of the Supportive Care Need Survey (Chambers 2015).
Psychological Impact of Erectile Dysfunction ‐ Sexual Experience (Chambers 2015).
Index of Sexual Satisfaction (Reese 2014).
Reese 2014 evaluated a telephone intervention intended to improve physical intimacy and sexual concerns for couples in whom one was diagnosed with colorectal cancer. Study authors reported a large effect size related to sexual functioning associated with the intervention for female (0.85) participants and a moderate effect size for males (0.58). However, they observed no effects of the intervention on sexual distress. They followed this study by evaluating an adapted version of the intervention in breast cancer survivors (adapted to address specific needs of breast cancer survivors and their intimate partners). Similar to their findings from research with people with colorectal cancer (Reese 2014), Reese 2018 reported medium to large positive effects on all sexual outcomes measured.
Conversely, Chambers 2015 found no evidence of differences between intervention and control groups for any of the measures above. However, these researchers did report statistically significant differences at 12 months for use of erectile dysfunction medication. Results demonstrated that participants in the intervention group were 3.14 times more likely to use medical treatment for erectile dysfunction than those in the control group (P = 0.0008).
1.8 Dyspnoea; telephone intervention versus control
Only two studies addressed this symptom (Mosher 2016; Porter 2011), even though people with lung cancer were the target population for a number of interventions. Once more, these investigators did not report change scores, or it was impossible to calculate these alongside the standard deviation of the change score, and meta‐analysis was not possible.
These studies measured outcomes using the following.
Four items on the Memorial Symptom Assessment Scale (MSAS) related to breathlessness (Mosher 2016).
Functional Assessment of Cancer Therapy ‐ Lung Cancer subscale (Porter 2011).
Dyspnoea was solely a primary outcome in Mosher 2016. However, this trial did not report any statistically significant benefit regarding breathlessness arising from a generic telephone‐delivered symptom management intervention ‐ involving carers ‐ when compared with telephone education/support. Conversely, comparison between a telephone‐delivered carer‐assisted coping skills programme and telephone‐delivered education/support conducted by Porter 2011 did generate statistically significant improvements (B = 0.76, standard error = 0.21, P = 0.0003) in lung cancer‐specific symptoms. This subscale included shortness of breath, coughing, weight loss, and loss of appetite. Neither of these studies included a usual care control, making it impossible to posit firm conclusions.
1.9 General symptom experience; telephone intervention versus control
General symptom intensity and/or symptom distress scores were study outcomes in 10 of the studies incorporated in the review (Badger 2013a; Badger 2013b; Barsevick 2010; Kroenke 2010; Mishel 2002; Molassiotis 2009; Porter 2011; Sherwood 2005; Sikorskii 2007; Traeger 2015). The following tools were used to measure this.
General Symptom Distress Scale (Badger 2013a; Badger 2013b).
Side Effect Checklist (SCL) (Barsevick 2010).
Symptom Distress Scale (Mishel 2002).
National Cancer Institute Common Toxicity Criteria (NCI‐CTC) (Molassiotis 2009).
Functional Assessment of Cancer Therapy ‐ Lung Cancer (FACT‐L) lung cancer‐specific subscale (Porter 2011).
Memorial Symptom Assessment Scale ‐ Short Form (MSAS‐SF) (Traeger 2015).
Various investigator‐developed symptom severity scales (Kroenke 2010; Sherwood 2005; Sikorskii 2007).
Again, these studies did not report change scores, or it was impossible to calculate these alongside the standard deviation of the change score, and meta‐analysis was not possible. Of the 10 studies, only two reported positive effects of telephone interventions on general symptom experience (Badger 2013b; Kroenke 2010). Kroenke 2010 measured study participants' physical symptom burden and calculated an overall symptom score. Study authors determined that the telephone intervention for pain and depression that was introduced generated statistically significant reductions in general symptom severity by the end of the study when compared with the control (M ‐1.0, 95% confidence interval (CI) ‐2.7 to ‐0.7; P = 0.014). The other study that reported statistically significant improvements in general symptom experience evaluated two different telephone interventions for Latina women with breast cancer (Badger 2013b). Badger 2013b determined that both interventions (telephone interpersonal counselling (TIP‐C) and telephone health education (THE)) generated significant improvements in general symptom distress, with participants in the THE group reporting slightly greater improvements (F[1.60, 55.83] = 17.13; P < 0.001) than TIP‐C participants (F[2.66] = 7.63; P < 0.01).
None of the eight remaining studies generated statistically significant results. However, although Molassiotis 2009 detected no significant improvement ‐ when introducing telephone‐delivered symptom‐focused interventions for people with colorectal or breast cancer receiving oral chemotherapy ‐ in a composite symptom score derived from nine symptoms (oral mucositis, hand‐foot syndrome, diarrhoea, constipation, nausea, vomiting, pain, fatigue, and insomnia), they did find improvements across most individual symptoms over time. During treatment cycles 1 and 2, the intervention group reported significant improvements across all symptoms (P < 0.0005 to 0.005), except for hand‐foot syndrome (P = 0.08) and vomiting (P = 0.062). These improvements were maintained until the sixth cycle for all symptoms except fatigue.
Discussion
Summary of main results
Telephone‐delivered interventions are growing in popularity within cancer care, arguably reflecting the trend for health care to be delivered in day care settings with limited capacity for delivering interventions face‐to‐face. This review incorporated data from 6250 people with cancer across 32 studies. Most of the interventions incorporated had been developed for ‐ or tested ability to reduce ‐ psychological symptoms, notably depressive symptoms (n = 21), anxiety (n = 16), and emotional distress (n = 7). The predominance of telephone‐delivered interventions for psychological symptoms is unsurprising; telephone counselling has been shown to be effective in reducing psychological symptoms including depression and anxiety in patient populations other than those with diagnosed cancer (Reese 2002), in people specifically with breast cancer (Chen 2018), and more recently among carers (Lins 2014). Not all studies had theoretical frameworks underpinning the intervention that was delivered and evaluated; those that did have such a framework used one from a diverse array of 11 differing frameworks.
Twenty‐one of the 32 studies reported a significant effect (P < 0.05) in favour of the intervention condition generated by the telephone‐delivered intervention (Allard 2007; Allen 2002; Badger 2007; Badger 2013a; Badger 2013b; Badr 2015; Bailey 2004; Barsevick 2004; Barsevick 2010; Chambers 2015; Dong 2018; Girgis 2009; Kroenke 2010; Mishel 2002; Mishel 2005; Molassiotis 2009; Rawl 2002; Porter 2011; Thomas 2012; Watson 2017; Yates 2005). However, meta‐analyses were not possible for any of the outcomes due to considerable heterogeneity. Findings provided in the review suggest that telephone‐delivered interventions may be effective in addressing some common symptoms associated with cancer, but this would need to be established in future updates of the review, when more evidence is available for each cancer type. At present, no firm conclusions can be drawn. It is encouraging, however, that findings from the review appear to be largely consistent with those from the Lins 2014 and Chen 2018 systematic reviews. The only difference is related to anxiety; neither Chen 2018 nor Lins 2014 reported positive outcomes for anxiety. Reasons for this are unclear, although populations eligible for inclusion in these reviews differed.
Further, this review has demonstrated that telephone‐delivered interventions are being developed for managing a range of physical as well as psychological cancer‐related symptoms. Symptoms including pain, dyspnoea, and sexually‐related symptoms, in addition to fatigue, have been subject to investigation. Sexually‐related symptoms are a relatively recent addition to the range of symptoms being addressed in this area of research. This mirrors research more generally into sexually‐related symptoms in cancer care. They have been orphan symptoms ‐ comparatively neglected ‐ when compared with other cancer‐related symptoms (White 2011). Further, this review identified that, with regards to telephone‐delivered interventions, dyspnoea could also be viewed as an orphan symptom. This may reflect perceived challenges of dealing with a complex and often acute symptom like dyspnoea by telephone. It may be that brief psycho‐educational interventions that are delivered primarily by telephone may be unsuitable for this group of cancer patients. However, further research is needed before such conclusions can be drawn.
For a number of symptoms ‐ uncertainty, pain, dyspnoea, and sexually‐related symptoms ‐ data from the studies incorporated are inconsistent, making conclusions regarding their management through telephone‐delivered interventions difficult to draw; around half failed to generate statistically significant findings.
Most interventions were delivered over a relatively short period, typically ranging from two to eight weeks; most frequently, interventions were provided weekly. However, lack of data prevented comparison of effectiveness by length or frequency of the intervention (dose). Likewise, evidence was insufficient to enable comparison by stage of disease. Some preliminary evidence from Porter 2011 suggests that different forms of interventions may be more/less effective according to stage of disease. This theory needs further investigation. Differential responses to interventions are important to determine. Although likely to be more cost‐effective than interventions delivered face‐to‐face (cost‐effectiveness of these interventions has not been reported on), those delivered by telephone remain relatively costly in terms of health professional time. It is clearly optimal to target interventions towards people who would benefit most, to enable effective resource use.
Telephone‐delivered interventions may provide greater benefit for particular people. Arguably, people with high symptom intensity could benefit the most, as they would also have high possibility for symptom reduction. However, as few studies specifically targeted people with moderate or greater symptoms, it is not possible to draw conclusions about the relative efficacy of interventions according to symptom burden. Further, the telephone can provide a degree of anonymity that may be helpful with certain symptoms (e.g. sexually‐related ones), and no requirement for face‐to‐face contact is likely to be of particular importance to people whose immunity is compromised through disease and/or treatment. However, data are insufficient to enable these hypotheses to be confirmed or refuted. It would appear that telephone‐delivered interventions may also generate practical benefit, for example, being particularly pertinent for people living at a distance from treatment centres, or for those with fatigue for whom the effort required to attend delivery of an intervention in person may be prohibitive.
Many people with cancer report feelings of fatigue and the negative effect this can have on their motivation for symptom self‐management (Ahlberg 2003; Ream 2015). Further, health professionals may find it more difficult to motivate patients in their patient group to adopt behaviour change when delivering interventions remotely (e.g. by telephone) rather than in person. This would suggest that techniques such as motivational interviewing (MI) could offer important benefit in telephone‐delivered interventions for symptom self‐management ‐ in particular, when patients may be experiencing fatigue. MI was used to generate behaviour change in two studies within the review (Thomas 2012; Ream 2015). Two further trial authors referred to techniques within their interventions aimed at enhancing motivation to change behaviour (Allen 2002; Yates 2005). Incorporation of goal‐setting and monitoring used by MI, as well as interventions based around it, provides impetus for behaviour change.
All interventions were delivered by health and social care professionals, most by nurses. Most papers (n = 22) referred to training needed by those delivering interventions ‐ notably in counselling skills, communication, and education. This is not attained without cost. Unfortunately, the papers in this review did not feature any form of cost evaluation; thus it is impossible to determine the cost implications of interventions such as these. This is an important topic that needs consideration in future research.
Overall completeness and applicability of evidence
This is the first review to appraise and synthesise findings from studies evaluating telephone‐delivered interventions for management of cancer‐related symptoms. This review indicates that such interventions may be effective in ameliorating cancer‐related symptoms ‐ most notably, depressive symptoms, fatigue, and emotional distress. Some symptoms (uncertainty, pain, dyspnoea, sexually‐related symptoms) have been subject to relatively less research; robust conclusions cannot be drawn from available evidence.
This review does have some limitations. First, it is limited by the narrative nature of the synthesis and the relatively small number of studies addressing certain symptoms, such as sexually‐related symptoms and dyspnoea, which rendered study conclusions difficult to determine. Further, it was impossible to determine whether better outcomes were achieved if the telephone was used alongside other elements (e.g. face‐to‐face contact, printed/digital/online materials). Although some studies that were incorporated into the review reported findings that failed to attain statistical significance, the review is likely to have been subject to a degree of publication bias. Finally, studies incorporated into the review were largely conducted in the United States, Australia, and the United Kingdom ‐ thus their findings may not translate well to other continents and nations. The findings presented need to be viewed in light of these limitations.
Further, symptoms are multi‐dimensional. This review focused on severity of symptoms; it did not address the distress generated by symptoms. Several studies in this review measured and reported on symptom distress, in addition to intensity. A symptom may be of low intensity but still may give rise to considerable distress. This may be important clinically ‐ arguably symptoms should be prioritised according to how distressing they are. The authors of this review are unable to draw conclusions about the effectiveness of telephone interventions with regards to symptom distress.
This review did incorporate psychological symptoms including uncertainty. However, it did not include fear of recurrence, as this is a concern associated with cancer that can give rise to symptoms like anxiety and uncertainty (rather than being a symptom per se). A recent study has evaluated an intervention for fear of recurrence delivered by telephone (Dieng 2016). We did not include this paper in the review.
Quality of the evidence
We used the GRADE approach to interpret findings based on the certainty of evidence generated across the included studies. Most of the 32 trials that met our inclusion criteria were at moderate risk of bias. All but one were randomised controlled trials, so the evidence generated by them should be interpreted as highest certainty available, despite blinding not being achieved across almost all of these trials; lack of blinding can give rise to performance or detection bias. However, it is acknowledged that in research of this nature, blinding of participants would be possible only if a form of sham intervention was delivered to those in the control group. This in itself introduces difficulties, as some of the effects of a telephone‐delivered intervention are likely to reflect characteristics of the telephone conversation (that would be experienced by those in the sham intervention control). This could render effects of the telephone intervention difficult to detect. Alternatively, an intervention could be tested against an active control for which the control group receives an intervention of known positive effect.
Review authors noted heterogeneity in results generated by the interventions studied (i.e. inconsistency within the body of evidence). This is likely to reflect differences between the interventions themselves. Although all were predominantly delivered by telephone, they varied in length, in content, and in some cases in incorporation of additional elements such as face‐to‐face contact or additional supportive media. Further, the cancer populations studied varied, and this appeared to introduce additional levels of heterogeneity. Therefore, meta‐analyses were not performed.
Overall, the certainty of evidence was very low for all outcomes in the review, as we are very uncertain about the estimates. We downgraded all outcomes due to concerns about overall risk of bias profiles being unclear or high, imprecision, inconsistency in results, and general heterogeneity.
Potential biases in the review process
We performed a comprehensive search, including a thorough search of the grey literature, and two review authors independently sifted all studies and extracted study data. We restricted the review to randomised controlled trials (RCT) and one quasi‐RCT. By searching a wide range of databases and grey literature, we attempted to ensure that we did not overlook any relevant evidence.
A threat to the validity of the review is likely to be publication bias; studies that did not find the treatment to be effective may not have been published. We did not report any meta‐analyses due to considerable clinical and methodological heterogeneity. Meta‐analysis may have led to misleading results that lacked any kind of generalisability.
Agreements and disagreements with other studies or reviews
The results of this review are largely consistent with those published previously by Chen 2018. That review evaluated tele‐health interventions for quality of life and psychological outcomes in people with breast cancer. Those review authors similarly reported that compared with usual care, telephone‐delivered interventions led to statistically significantly less depressive symptoms, distress, and perceived stress. Consistency across reviews such as these does provide some confidence in the veracity of findings. However, unlike the present review, Chen 2018 reported that anxiety did not differ between tele‐health and usual care control. As with this review, the findings of Chen 2018 have to be considered with a degree of caution owing to between‐study heterogeneity.
Authors' conclusions
Implications for practice.
This review found some evidence supporting use of telephone‐delivered interventions for managing cancer‐related symptoms ‐ most evidence is related to managing anxiety, depressive symptoms, emotional distress, or fatigue. This would suggest that telephone interventions should be considered as one component for managing these cancer‐related symptoms. Arguably, these interventions would not need to be provided within statutory services; they could be provided by voluntary sector providers, who frequently provide patients support and information via health professional‐delivered telephone help lines. We found limited evidence regarding potential management of some symptoms by telephone‐delivered intervention, notably regarding sexually‐related symptoms and pain.
Interventions evaluated in this review varied considerably in terms of (1) the number of calls provided; (2) the length and timing of calls; (3) the content of calls; and (4) provision or not of additional supportive material. It appears that for some symptoms (i.e. depressive symptoms), telephone‐only interventions may be indicated. However, given the small, mostly biased studies ‐ and lack of meta‐analysis ‐ this conclusion is tentative. For other symptoms, arguably those for which greater behaviour change may be required (e.g. fatigue), it may be the case that additional supportive materials are required to optimise outcomes. It has not been possible however through this review to conclude how many calls, delivered over what period, of particular duration and content, are required to generate effect. It appears that it may be beneficial for those delivering interventions to be trained in motivational interviewing skills.
It is noteworthy that most studies included within the review did not target samples with high baseline symptom burden. Arguably, it would be wise to do so, as there could be greater possibility for symptom reduction in such patient groups and greater associated cost savings.
Healthcare services are facing an unprecedented level of austerity that is likely to persist given ageing communities and finite health expenditure. There is a requirement for care to shift closer to home, and for cost containment to be applied. Telephone‐delivered interventions could make an important contribution to both requirements. Further research is needed to determine clearly which symptoms are most amenable to be managed through telephone‐delivered interventions, which patients should be targeted to achieve clinically important and sustained benefit, and which interventions are most cost‐effective.
There is risk of bias in the studies reported on in this review (most studies were at risk of some bias). When considered alongside other factors including between‐study heterogeneity and imprecise estimates of effect, the overall certainty of evidence is best considered very low, as we are very uncertain about the estimates. A lot of the heterogeneity was due to different cancer sites. Thus, the review findings need to be interpreted with a degree of caution. Consistency between this review and that of Chen 2018 is however encouraging and can lend support to the conclusions drawn.
Implications for research.
Further work is necessary to determine:
which cancer‐related symptoms are amenable to management by telephone‐delivered intervention; particular attention needs to be directed towards symptoms that have been subject to little research (e.g. sexually‐related symptoms, dyspnoea);
what augmentation of telephone‐delivered interventions (e.g. with face‐to‐face meetings or additional resources) works, and in what particular circumstances;
cost implications of delivering interventions by telephone to manage cancer‐related symptoms;
consensus on the most appropriate measures to be used to measure outcomes and minimal clinically important differences for each one;
consensus on standardising reporting of the intervention theory underpinning the intervention and its components;
longevity of effects generated by telephone‐delivered interventions for cancer‐related symptoms;
effectiveness of telephone‐delivered interventions in reducing fear of recurrence (a phenomenon that is increasingly being written about in the literature but is not deemed a symptom and thus was excluded from this review); and
effectiveness of telephone‐delivered interventions for enhancing quality of life for people with cancer.
Future research is needed to address methodological limitations identified in the included studies. Notably, future randomised controlled trials need to have better methodological conduct and design to minimise risk of bias and to provide more extensive reporting of pertinent outcomes. Further, they need sufficient statistical power and length of follow‐up to generate much needed definitive evidence concerning efficacy of telephone‐delivered interventions for management of cancer symptoms across diverse patient groups.
History
Protocol first published: Issue 1, 2009 Review first published: Issue 6, 2020
Acknowledgements
The review authors wish to thank the Cochrane Gynaecological, Neuro‐oncology, and Orphan Cancers editorial team: Jo Morrison and Robin Grant (Co‐ordinating Editors) for clinical and editorial advice, Gail Quinn (Managing Editor) and Tracey Harrison (Assistant Managing Editor) for editorial support, and Jo Platt (Information Specialist) for assistance with the search.
This project was supported by the National Institute for Health Research, via Cochrane infrastructure funding to the Cochrane Gynaecological, Neuro‐oncology, and Orphan Cancers Group. The views and opinions expressed therein are those of the review authors and do not necessarily reflect those of the Systematic Reviews Programme, NIHR, NHS, or the Department of Health.
We would like to acknowledge Shirley Jefferson and Gian Gargaro for scoping the literature before development of the protocol.
The review authors and the Cochrane Gynaecological, Neuro‐oncology, and Orphan Cancers Team are grateful to the following peer reviewers for their time and comments: Helen Bulbeck, Karen Galway, and Alasdair Roodey.
Appendices
Appendix 1. CENTRAL search strategy
#1 MeSH descriptor Neoplasms explode all trees #2 neoplasm* or cancer* or carcinoma* or tumour* or adenocarcinoma* or leukemi* or leukaemi* or lymphoma* or tumor* or malignan* or myeloma* #3 MeSH descriptor Radiotherapy explode all trees #4 radiotherap* or radiation or radiochemotherap* or chemoradi* or chemotherap* #5 (#1 OR #2 OR #3 OR #4) #6 MeSH descriptor Telemedicine explode all trees #7 telemedicine or (tele next medicine) #8 teleconsultation or (tele next consultation) #9 telephone* #10 phone* #11 cellphone* #12 remote* near/5 consultation* #13 (#6 OR #7 OR #8 OR #9 OR #10 OR #11 OR #12) #14 (#5 AND #13)
Appendix 2. MEDLINE search strategy
1 exp Neoplasms/ 2 (neoplasm* or cancer* or carcinoma* or tumour* or adenocarcinoma* or leukemi* or leukaemi* or lymphoma* or tumor* or malignan* or myeloma*).mp. 3 exp Radiotherapy/ 4 (radiotherap* or radiation or radiochemotherap* or chemoradi* or chemotherap*).mp. 5 1 or 2 or 3 or 4 6 exp Telemedicine/ 7 (telemedicine or (tele adj medicine)).mp. 8 (teleconsultation or (tele adj consultation)).mp. 9 (remote* adj5 consultation*).mp. 10 telephone*.mp. 11 phone*.mp. 12 cellphone*.mp. 13 8 or 6 or 11 or 7 or 10 or 9 or 12 14 "randomized controlled trial".pt. 15 "controlled clinical trial".pt. 16 randomized.ab. 17 randomly.ab. 18 trial.ab. 19 groups.ab. 20 18 or 19 or 16 or 17 or 15 or 14 21 13 and 20 and 5
Appendix 3. Embase search strategy
1 exp Neoplasm/ 2 (neoplasm* or cancer* or carcinoma* or tumour* or adenocarcinoma* or leukemi* or leukaemi* or lymphoma* or tumor* or malignan* or myeloma*).mp. 3 exp Radiotherapy/ 4 (radiotherap* or radiation or radiochemotherap* or chemoradi* or chemotherap*).mp 5 1 or 2 or 3 or 4 6 exp Telemedicine/ 7 (telemedicine or (tele adj medicine)).mp. 8 (teleconsultation or (tele adj consultation)).mp. 9 (remote* adj5 consultation*).mp. 10 telephone*.mp. 11 phone*.mp. 12 cellphone*.mp. 13 8 or 6 or 11 or 7 or 10 or 9 or 12 14 exp Controlled Clinical Trial/ 15 randomized.ab. 16 randomly.ab. 17 trial.ab. 18 groups.ab. 19 18 or 16 or 17 or 15 or 14 20 19 and 13 and 5
Data and analyses
Comparison 1. Symptoms.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1.1 Anxiety | 5 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.1.1 Breast cancer (Telephone only) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.1.2 Colorectal cancer (Telephone only) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.1.3 Lung cancer (Telephone plus other) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.1.4 Prostate cancer (Telephone only) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.1.5 Mixed cancers (Telephone plus other) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.2 Depression | 9 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.2.1 Breast cancer (Telephone only) | 2 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.2.2 Colorectal cancer (Telephone only) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.2.3 Lung cancer (Telephone plus other) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.2.4 Prostate cancer (Telephone only) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.2.5 Mixed cancers (Telephone only) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.2.6 Mixed cancers (Telephone plus other) | 3 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.3 Fatigue | 6 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.3.1 Breast cancer (Telephone only) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.3.2 Breast cancer (Telephone plus other) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.3.3 Prostate cancer (Telephone only) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.3.4 Mixed cancers (Telephone plus other) | 3 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.4 Emotional distress | 5 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.4.1 Breast cancer (Telephone only) | 2 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.4.2 Breast cancer (Telephone plus other) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.4.3 Prostate cancer (Telephone only) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 1.4.4 Mixed cancers (Telephone only) | 1 | Std. Mean Difference (IV, Random, 95% CI) | Totals not selected |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Allard 2007.
| Study characteristics | ||
| Methods |
Setting: 5 regional centres in different geographic areas in Quebec, Canada Recruitment: eligible women were over the age of 18, were French speaking, and were diagnosed with primary breast cancer or a suspected lesion. They were scheduled to undergo day surgery and had no hearing impairment and no access to a phone at home. Staff nurses identified eligible patients who were given a pamphlet describing the study and subsequently, if willing to participate, were telephoned by a member of the research team Randomisation: randomised clinical block trial |
|
| Participants | 117 women with breast cancer aged 18 years or older | |
| Interventions |
Intervention: based on self‐regulation theory, which emphasises concrete objective information and regulation of emotional and functional processes to achieve optimal coping. Comprising 2 telephone sessions delivered weekly for 2 weeks during the immediate postoperative period. In each session, participants were asked to identify and describe symptoms in concrete terms. Actions taken by women to manage each symptom were rated according to their effectiveness from 1 to 5 (1 = not effective, 5 = very effective). Effective actions were encouraged; ineffective actions were explored and women encouraged to find other, potentially helpful, actions. The interventionist suggested new or additional self‐care strategies and acknowledged feelings expressed by women during the call Control group: usual care consisting of perioperative teaching and a follow‐up phone call within 24 hours of discharge from the hospital Interventionist: the intervention was delivered by the principal investigator, who is a trained nurse |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | Some participants may have already started adjuvant hormonal therapies when recruited for this study | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | The research assistant randomly allocated the women with or without axillary node dissection to the experimental or control group at each site using a table of random numbers |
| Allocation concealment (selection bias) | Unclear risk | The method of concealment is not described; however, the research assistant allocated participants using a table of random numbers, and the research assistant did not perform the intervention. Detection bias may exist, as it is unclear if the intervener or the research assistant collected the data |
| Blinding (performance bias and detection bias) All outcomes | High risk | Owing to the nature of the intervention, study participants were not blinded. It is unclear who collected the data and by what method |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Attrition rate is reported. Reasons for attrition are not reported |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting of outcomes |
| Other bias | Unclear risk | The process of screening was reliant on staff nurses identifying participants at the doctor's office. It is unclear if all eligible participants were approached; some bias may exist, as these staff nurses may have excluded or not approached participants for many reasons |
Allen 2002.
| Study characteristics | ||
| Methods |
Setting: a network of sites (hospitals and private oncology clinics) in Rhode Island, Massachusetts, Pennsylvania, New Jersey, USA Recruitment: eligible women were aged 50 or younger, had no prior history of breast carcinoma, had stage I‐IIIA tumours, and were beginning their first course of chemotherapy treatment. Medical staff provided information about the study to eligible women. Women interested in participating were sent a letter describing the study, followed by a telephone call several days later to answer questions and invite participation. Women who agreed to participate were asked to complete a baseline telephone interview and were mailed questionnaires. They were also asked to nominate a primary support person (PSP) to participate as a partner in the intervention. Nominated partners were then contacted by research staff to invite their participation and to complete similar surveys Randomisation: RCT. Women were stratified by recruitment site location and PSP status (spouse/significant other, other family member or friend, no participating PSP) |
|
| Participants | 164 women with breast cancer younger than 50 years | |
| Interventions |
Intervention: based on motivational technique, using an investigator‐developed “home care training model”. Nurse delivered to patient and other supportive person if nominated. The intervention consisted of 5 interactive components: problem orientation, problem definition, generation of alternatives, decision‐making, and solutions implementation. Patients were given a printed manual with chapters that paralleled the intervention components. The intervention was conducted as 1 × 2‐hour individual face‐to‐face training session held in the participant's home, followed by 4 telephone contacts 2 weeks apart, and a final 2‐hour individual face‐to‐face training session held in the participant's home, where the nurse took a less active role while letting the participant and the PSP take over. In each session, worksheets were completed and interventionist feedback was provided. Participants could instigate telephone contact to discuss additional questions Control group: usual care (not described) Interventionist: oncology research nurse |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Generation of random sequence is not described |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | High risk | Owing to the nature of the intervention, study participants were not blinded. It is unclear who collected the data and by what method |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Attrition rate is reported. Reasons for attrition are not reported |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting of outcomes |
| Other bias | High risk | Due to unavoidable delays in completing all 6 sessions for some participants receiving the intervention, women in the experimental group completed both of their follow‐up assessments approximately 1 month later than those in the control arm |
Badger 2005.
| Study characteristics | ||
| Methods |
Setting: academic cancer centres and urban private oncology offices in Arizona, USA Recruitment: eligible women had a diagnosis of stage I‐III breast cancer, were receiving adjuvant treatment, were able to speak English and talk on the telephone, were married, and had a partner, who were also willing to take part in the intervention and were employed at the time of the study. Participants were recruited from a local cancer centre, oncologists' offices, and support groups, and through self‐referral after reading brochures displayed in the various settings Randomisation: pilot quasi‐randomised controlled trial (experimental with repeated measures) |
|
| Participants | 48 women in their mid‐50s with breast cancer | |
| Interventions |
Intervention: telephone interpersonal counselling (TIP‐C). Based on theories of interpersonal therapy and cancer education. Participants received 6 weekly telephone calls × 30 minutes (approximately) while they were undergoing treatment for breast cancer. Sessions focused on cancer education, interpersonal role disputes, social support, awareness, management of depressive symptoms, and role transitions. Partners of participants received 3 telephone‐delivered TIP‐C sessions (weeks 1, 3, and 5) during the same 6‐week period as the women. These sessions also focused on issues such as cancer education, role disputes, role transitions, and social support
Control group: participants in the usual care arm received a resource list about cancer and brief, focused telephone calls (6 for women and 3 for their partners × 5 to 10 minutes) to inquire about general well‐being and to answer general questions (no counselling) Interventionist: nurse counsellors (master's prepared clinical nurse specialists in psychiatric/mental health nursing who had additional oncology training) |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | Pilot study, quasi‐randomised sample |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | High risk | It is unclear if study participants were blinded. Interveners were not blinded. It is unclear who collected the data and by what method |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Attrition is not reported |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting of outcomes |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Badger 2007.
| Study characteristics | ||
| Methods |
Setting: Arizona, USA (not specified) Recruitment: eligible women had a diagnosis of stage I‐III breast cancer, were receiving adjuvant treatment, were able to speak English and talk on the telephone, had no physical or psychological disabilities, and had a partner who was available and willing to take part in the intervention. Participants were recruited from a local cancer centre, oncologists' offices, and support groups, and through self‐referral after reading brochures displayed in the various settings Randomisation: 3‐arm RCT |
|
| Participants | 96 women with breast cancer and their supportive partners | |
| Interventions |
Intervention: participants and their partners were randomised to receive 1 of 3 programmes: (1) telephone interpersonal counselling (TIP‐C); (2) self‐managed exercise (SE); or (3) attention control (AC). In the TIP‐C arm, participants received 6 × weekly calls, which consisted of cancer education, social support, and awareness and management of depressive and anxiety symptoms. Partners received 3 × calls (every other week of the intervention) to discuss their well‐being and relationship. Participants in the SE arm also received 6 × weekly calls. The calls encouraged participants to exercise and recorded information about progress and intensity of exercise. The SE partners also received calls every other week to encourage exercise and to track progress
Control group: the AC group received printed information about breast cancer and 6 brief weekly calls. Their partners received 3 biweekly calls during the same period. The calls averaged 7 minutes in duration. If these participants reported any problems, they were directed to the primary physician. No counselling or encouragement to exercise was offered Interventionist: psychiatric nurse counsellors |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | The generation of random sequence is not described |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | High risk | It is not clear whether participants were blinded to group. Interveners were not blinded. It is unclear who collected the data and by what method |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Loss to follow‐up < 20%; similar reasons between groups |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting of outcomes |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Badger 2013a.
| Study characteristics | ||
| Methods |
Setting: Arizona, USA (not specified) Recruitment: eligible women had a diagnosis of breast cancer, were currently receiving treatment, were at least 21 years of age, had access to a telephone, and had a supportive partner (SP) who was available and willing to take part in the intervention. SPs were not restricted to spouses. Participants were recruited from a local cancer centre, oncologists' offices, and support groups, and through self‐referral |
|
| Participants | 52 women with breast cancer and their supportive partners | |
| Interventions |
Interventions: women and their supportive partners were randomly assigned to 1 of 3 interventions: (1) telephone health education (THE), (2) telephone interpersonal counselling (TIC), or (3) videophone interpersonal counselling (VIC). All participants received 8 weekly telephone calls of 30 minutes in their preferred language (English or Spanish), and their SP received 4 biweekly sessions. Participants in THE received leaflets on breast cancer terminology, treatments, side effect management, nutrition, physical activity, and resources. These materials were reviewed with participants and SPs over the telephone. Participants in the TIC and VIC arms received counselling via video or telephone, which addressed mood and affect management, emotional expression, interpersonal communication, relationships with family and providers, social support, and referral to support service as required Interventionist: an information specialist provided THE; TIC and VIC were provided by a social worker |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | The generation of random sequence is not described |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | High risk | It is unclear if participants were blinded to group. Interveners were not blinded |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Over 20% loss to follow‐up; significantly higher attrition in the THE group compared to the TIC or VIC group |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting of outcomes |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Badger 2013b.
| Study characteristics | ||
| Methods |
Setting: Arizona, USA (not specified) Recruitment: eligible women had a diagnosis of stage I‐III breast cancer, were receiving adjuvant treatment, spoke Spanish or English, had no physical or psychological disabilities, had access to a telephone, and had a supportive partner (SP) who was available and willing to take part in the intervention. Eligible SPs were 21 years of age or older, spoke Spanish or English, had no physical or psychological disabilities, and had access to a telephone. Participants were recruited from a local cancer centre, oncologists' offices, and support groups, and through self‐referral Randomisation: RCT (3‐wave repeated measure with a between‐subjects factor) |
|
| Participants | 90 Latina women with breast cancer and their supportive partners | |
| Interventions |
Intervention: women and their supportive partners were randomly assigned to 1 of 2 interventions: (1) telephone interpersonal counselling (TIP‐C), or (2) telephone health education (THE). The TIP‐C intervention addressed mood and affect management, emotional expression, interpersonal communication and relationships, social support, and cancer information. Participants received 8 weekly phone calls × 30 minutes (approximately) in their preferred language (Spanish or English), and their SPs received 4 sessions every other week. Interventions were tailored to the cultural values and beliefs of participants Control group: participants in the THE group also received 8 weekly phone calls in their preferred language (Spanish or English), and their SPs received 4 biweekly sessions. The THE focused on normal breast health and breast cancer, routine tests for diagnosis and prevention and associated terminology, treatment, side effects of treatment and strategies to combat these side effects, lifestyle interventions such as nutrition and physical activity, and referrals and resources Interventionist: bilingual, bicultural master’s‐prepared social workers |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | The generation of random sequence is not described |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | High risk | It is not clear whether participants were blinded to group. Interveners were not blinded. It is unclear who collected the data and by what method |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Loss to follow‐up < 20%; similar reasons between groups |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting of outcomes |
| Other bias | Unclear risk | Participants received $20 for completing assessments |
Badr 2015.
| Study characteristics | ||
| Methods |
Setting: Mount Sinai Hospital, New York, USA Recruitment: eligible participants had advanced lung cancer (LC), were within 1 month of treatment initiation, were spending more than 50% of their time out of bed each day, and had a partner (or other close family member) whom they identified as their primary carer. Participants and carers had to be at least 18 years old and able to read and understand English, and had to provide informed consent Randomisation: 2‐arm RCT |
|
| Participants | 39 lung cancer participants and their partners | |
| Interventions | Participants and their partners were randomised to receive usual medical care or a psychosocial intervention Psychosocial intervention: participants and carers in the intervention group received a manual, covering the topics of self‐care, stress and coping, symptom management, effective communication, problem‐solving, and maintaining and enhancing relationships. In addition, they participated in 6 × weekly 60‐minute telephone counselling sessions Usual medical care: primary palliative care was provided by the participant’s medical oncologist, including basic management of pain and other symptoms, including depression and anxiety, with referral to outpatient supportive oncology practice if required Interventionist: trained interventionist with a master’s degree in mental health counselling |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | The generation of random sequence is not described |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | High risk | Owing to the nature of the intervention, study participants and interveners were not blinded. It is unclear who collected the data and by what method |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Loss to follow‐up < 20%; similar reasons between groups |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting of outcomes |
| Other bias | Unclear risk | Participants received $20 gift cards on completing surveys |
Bailey 2004.
| Study characteristics | ||
| Methods |
Setting: North Carolina, USA (not specified). Recruitment: eligible men were diagnosed with stage B1, B2, or C1 prostate cancer and had been in watchful waiting from 1 to 124 months. Participants were recruited from the urology practices of 3 physicians at 1 hospital. The sample was a convenience sample, which was then randomised to intervention and control groups Randomisation: pilot RCT |
|
| Participants | 41 men with prostate cancer | |
| Interventions |
Intervention: based on Mishel's Reconceptualised Uncertainty in Illness Theory. Participants in the intervention group received telephone calls from the interventionist weekly × 5 weeks. During each call, participants' problems were identified and the nature of their uncertainty was assessed, then the intervention was delivered. The intervention consisted of 4 elements: (1) re‐framing uncertainty through encouraging probabilistic thinking, discouraging negative perspectives and providing information, (2) helping incorporate uncertainty into life structure through emphasising empowering activities, promoting self‐care, and encouraging assertive communication with healthcare providers about symptom concerns, (3) supporting participants' belief in future treatment, and (4) encouraging self‐monitoring and vigilance Control group: usual care (not described in detail). Had access to naturally occurring sources of support Interventionist: male nurse |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | A table of random numbers was used to assign men to either an experimental group that received the intervention or a control group that received usual care |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | High risk | Nurse data collectors did not deliver the intervention, but owing to the nature of the intervention, study participants were not blinded |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Loss to follow‐up < 20%; similar reasons between groups |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting of outcomes |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Barsevick 2004.
| Study characteristics | ||
| Methods |
Setting: a university health science centre in Utah and a comprehensive cancer centre in Philadelphia, USA Recruitment: eligible participants spoke English; were beginning curative or local control treatment for breast, lung, colorectal, advanced prostate, gynaecological, or testicular cancer; and planned to receive at least 3 cycles of chemotherapy, 6 weeks of radiotherapy, or concurrent chemotherapy and radiotherapy. Prior treatment other than surgery had to be completed at least 1 month previously. Participants were excluded if they were having stem cell transplantation, interleukins, interferons, or tumour necrosis factor; had chronic fatigue syndrome; were enrolled in another study involving a psycho‐educational intervention; had overt evidence of psychiatric disorder; or had initiated treatment for anaemia or depression in the past 3 weeks Randomisation: RCT |
|
| Participants | 396 men and women with various cancer diagnoses | |
| Interventions |
Intervention: the Telephone Energy Conservation and Activity Management (ECAM) intervention was based on the Common Sense Model, which guided participants through 3 stages of information‐processing to help them manage cancer‐related fatigue. The intervention consisted of 3 weekly telephone sessions commencing at the beginning of chemotherapy treatment or at week 3 to 5 during radiation therapy. Session 1 consisted of coping skills training. Participants kept a journal and prioritised their usual activities. During session 2, participants were assisted to develop a care plan to minimise the interference of fatigue. In session 3, participants appraised and revised the plan. Sessions 1 and 2 each lasted 30 minutes, and session 3 lasted 15 minutes Control group: attention control consisting of information about nutrition and a healthy diet. Participants kept a dietary record for 24 hours Interventionist: research nurse |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | The generation of random sequence is not described |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | High risk | It is unclear whether participants were blinded to group. Interveners were not blinded |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Loss to follow‐up and between‐group differences regarding reasons for it are unclear |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting of outcomes |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Barsevick 2010.
| Study characteristics | ||
| Methods |
Setting: 2 university health centres, a community cancer centre, and a comprehensive cancer centre in Philadelphia, USA Recruitment: eligible patients were 18 years of age or older and were beginning a new chemotherapy regimen with at least 2 drugs administered intravenously in a cyclical manner for breast, lung, colorectal, prostate, gynaecological, bladder, or testicular cancer or lymphoma. Prior treatment other than surgery had to be completed at least 1 month previously, and the individual could receive concurrent radiotherapy. Participants had to be able to read and write English. Individuals were excluded if they were having marrow or stem cell transplantation, interleukins, interferons, or tumour necrosis factor; had chronic fatigue syndrome; were being treated for diagnosed sleep disorder; were enrolled in another study involving a psycho‐educational intervention; had a communication impairment; had overt evidence of psychiatric disorder; or had initiated treatment for anaemia or depression in the past 3 weeks. Potential participants were approached by telephone or in the clinic, and the study was explained Randomisation: RCT. Participants were stratified by diagnosis (breast cancer vs non‐breast cancer) at each site and then were randomly assigned to intervention or control group |
|
| Participants | 292 men and women aged 18 years or older with various cancers | |
| Interventions |
Intervention: The EASE intervention is based on the Common Sense Model and includes providing information about fatigue and sleep disturbance, coping skills training, and appraisal of used coping strategies. Participants received 3 telephone sessions during the second, third, and fourth weeks after the first chemotherapy treatment. Participants also received a handbook with information about symptoms and examples of energy conservation and sleep management strategies. Between sessions 1 and 2, participants completed a daily diary of their symptoms and sleep patterns, as well as a priority list of usual activities Control group: attention control consisting of information about nutrition and a healthy diet. Participants kept a dietary record for 24 hours in preparation for session 2. The 3 control sessions were equivalent to the intervention session in terms of the amount of time spent with the individual Interventionist: research nurse |
|
| Outcomes |
Method of assessing outcome measures:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Random assignments were generated by the statistician |
| Allocation concealment (selection bias) | Low risk | Random assignments were generated by the statistician and placed in sealed envelopes that were numbered and selected sequentially for each stratification group |
| Blinding (performance bias and detection bias) All outcomes | High risk | It is unclear whether participants were blinded to group. Interveners were not blinded. It is unclear who collected the data and by what method |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Loss to follow‐up < 20%; similar reasons between groups |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting of outcomes |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Chambers 2014.
| Study characteristics | ||
| Methods |
Setting: community‐based; 2 large and well‐established state‐based Cancer Helplines in Australia Recruitment: eligible participants were adult patients and carers who called cancer information and support Cancer Helplines in 2 Australian states (Queensland and New South Wales). Patients and carers were independent callers and, therefore, were not a dyad. Study inclusion criteria included (a) having a score of 4 or greater on the Distress Thermometer, (b) being able to read and speak English, and (c) having no previous history of head injury and/or dementia. Individuals under current psychiatric care and those who presented with grief or bereavement were excluded. The Cancer Helpline operator offered callers who met selection criteria entry into the study at the time of the call Randomisation: randomised trial. Participants were randomised following completion of baseline measures. Randomisation was stratified by participant and carer and state |
|
| Participants | 354 participants with various cancer diagnoses and 336 carers | |
| Interventions |
Intervention: participants and carers randomised between (a) single nurse‐delivered telephone self‐management session and (b) 5 telephone CBT sessions delivered by a psychologist Both groups were provided a self‐management resource kit comprising:
|
|
| Outcomes |
Method of assessing outcome measures:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomisation was stratified by participant and carer and state (Queensland vs New South Wales) and occurred in blocks of 10, with each condition randomly generated 5 times within each block to ensure an unpredictable allocation sequence with equal numbers of participants in each group at the completion of each block |
| Allocation concealment (selection bias) | Low risk | Unpredictable allocation sequence was undertaken by the project manager and was concealed from investigators |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Project staff tracking assessments were blinded to treatment allocation |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Loss to follow‐up over 20%; loss to follow‐up considerably higher in the 5‐session psychologist‐delivered CBT sessions intervention group |
| Selective reporting (reporting bias) | Low risk | Balanced reporting for all measures |
| Other bias | Unclear risk | 93% completed the single‐intervention session with the nurse; only 53% completed all 5 sessions in the psychologist arm |
Chambers 2015.
| Study characteristics | ||
| Methods |
Setting: private clinics, public/private hospitals, and public service announcements in Queensland, Australia Recruitment: eligible participants were men who had been scheduled for/had undergone radical prostatectomy within the last 12 months and their female partners. Participants were required to be able to read and write in English, without any other concurrent cancer, and with no history of head injury, dementia, or psychiatric illness Randomisation: randomised in blocks of 12, with each condition randomly generated 4 times within each block |
|
| Participants | 189 men with prostate cancer and their female partners | |
| Interventions |
Intervention: men and their partners were randomly assigned to 1 of 3 groups: (a) couples‐based peer‐delivered telephone support, (b) couples‐based nurse‐delivered telephone counselling, or (c) usual care. All participants in the intervention arms received at least 6 sessions of telephone support/counselling by nurse counsellors or peer support volunteers. Couples recruited pre‐surgery received an additional 2 calls before undergoing surgery. Both intervention arms included skills training in couple communication and conjoint coping, in addition to a DVD with tip sheets. The nurse counselling intervention included educating about prostate cancer, menopause, and sexuality; providing behavioural homework on increasing expression of affection and non‐demanding sexual touch; challenging negative beliefs about prostate cancer, ageing, and sexuality; and choosing and integrating a medical treatment for erectile treatment into the relationship. The peer support intervention included psycho‐education about diagnosis; common experiences with surgery and recovery; managing side effects; improving communication; maintaining intimacy; sexual problems; and managing erectile dysfunction. Men and their partners were required to attend all sessions. Participants randomised to usual care received standard medical management and published participant education materials Interventionist: 2 prostate cancer nurse counsellors |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomisation occurred in blocks of 12, with each condition randomly generated four times with each block to ensure an unpredictable allocation sequence |
| Allocation concealment (selection bias) | Low risk | Sequence was undertaken by the project manager and concealed from investigators |
| Blinding (performance bias and detection bias) All outcomes | High risk | Owing to the nature of the intervention, participants and interveners were not blinded |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Loss to follow‐up < 20%; similar reasons between groups |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting of outcomes |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Dong 2018.
| Study characteristics | ||
| Methods |
Setting: China Recruitment: All participants had colorectal cancer diagnosis confirmed by pathology and/or cytology before enrolment. Inclusion criteria were (1) ≥ 18 years of age; (2) Self‐Rating Depression Scale score of 50 to 70; (3) willing to comply with study protocols; (4) expected survival time over 6 months; (5) awareness of personal disease status; (6) understanding of the study and being voluntarily recruited; (5) no history of other malignant tumours or personal psychiatric conditions, family history of psychosis, or special medication history of psychoactive drugs; and (6) not treated with antidepressant drugs Randomisation: randomised by computer‐generated random numbers to 1 of 3 arms ‐ 2 treatment arms, 1 control arm |
|
| Participants | 135 colorectal cancer participants | |
| Interventions | Before recruitment, all participated in a 90‐minute physiological and psychological education lecture based on the SIGN guidelines on colorectal cancer Control: participants in the control group received usual care Interventions: participants in intervention arms received 6 weekly telephone sessions of 20 to 40 minutes. The content of sessions for the 2 intervention arms is given below: Telephone Support (TS): each week, participants were asked questions related to changes in condition, new symptoms, fear of recurrence, treatment and side effects, genetic risk, self‐care (diet, support groups, finances), and family concerns Telephone‐Based Reminiscence Therapy (TBR): Week 1: the psychologist guided the participant to reminisce about people who had a positive influence on his/her life. Week 2: the participant reminisced about happy times in the past. Week 3: the participant talked about his or her past achievements and the significance of these achievements. Week 4: the participant recalled the important turning points in his/her life and the influence of each. Week 5: the participant talked about his/her struggles with cancer and their positive significance. Week 6: the participant talked about his/her hopes for the future Interventionist: psychologist |
|
| Outcomes |
Method of assessing outcome measures:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated random numbers (EPIDAT 3.1) |
| Allocation concealment (selection bias) | Low risk | Random numbers were provided in numbered opaque envelopes by an external staff member, ensuring that assessors were blinded to treatment group assignment |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | No details |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No loss to follow‐up |
| Selective reporting (reporting bias) | Low risk | Balanced reporting for all measures |
| Other bias | Unclear risk | Did not collect pathological grading and staging data for colorectal cancer, and previous studies showed that the severity of depression in colorectal cancer participants is related to pathological grade |
Downe‐Wamboldt 2007.
| Study characteristics | ||
| Methods |
Setting: academic cancer centre, Canada Recruitment: eligible participants were 50 years or older, had surgery within the past 3 months, were receiving care from the Academic Clinic's Oncology Program, were living within 200 km of the academic clinic, had access to a telephone, were aware of their diagnosis, understood English, and were able to provide informed consent. Women with breast cancer stage I or II who had a lumpectomy or a mastectomy were eligible, as were men with prostate cancer stage T1 or T2M0 who had had a radical prostatectomy, and individuals with small cell lung cancer stage I, II, or IIIA. Patients receiving chemotherapy were excluded. Participants were recruited from the offices of private surgical oncologists and urologists and site‐specific clinics (breast, lung, and prostate) at an academic cancer centre clinic Randomisation: RCT |
|
| Participants | 175 men or women aged 50 years or older with breast, lung, or prostate cancer | |
| Interventions |
Intervention: individualised telephone problem‐solving counselling based on cognitive‐behavioural principles. A series of counselling sessions were offered at participants' convenience over 3 months. Collaborative in nature; the participant and the interventionist worked together to define participant concerns, identify solutions, and evaluate consequences of these solutions Control group: usual care, including diagnostic and follow‐up care at 3‐month intervals Interventionist: nurse counsellor |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Participants were randomised to groups using concealed random computer‐generated numbers that randomly blocked after every second and fourth subject to ensure equivalent numbers of subjects in both arms of the trial |
| Allocation concealment (selection bias) | Low risk | Participants were randomised to groups using concealed random computer‐generated numbers that randomly blocked after every second and fourth subject to ensure equivalent numbers of subjects in both arms of the trial |
| Blinding (performance bias and detection bias) All outcomes | High risk | Owing to the nature of the intervention, participants and personnel were not blinded to the intervention |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Loss to follow‐up < 20%; similar reasons between groups |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting of outcomes |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Girgis 2009.
| Study characteristics | ||
| Methods |
Setting: New South Wales, Australia Recruitment: eligible participants were 18 years of age or older, had non‐localised breast or colorectal cancer, were diagnosed within the last 6 months, resided in New South Wales province at diagnosis, were able to complete study measures in English, and were physically and mentally capable of study participation. Potential participants were identified by the New South Wales Central Cancer Registry, and eligibility was confirmed by participants' treating physicians. Eligible individuals were sent a letter from the CCR seeking consent to receive study information from the research team. If a signed consent form was returned, the participant was contacted by telephone to complete baseline measures Randomisation: parallel factorial RCT |
|
| Participants | 356 men and women aged 18 years or older with non‐localised breast or colorectal cancer | |
| Interventions |
Intervention: based on the notion of providing feedback for patient‐reported outcomes (PROs) to clinicians to improve supportive care for cancer patients. Participants completed 3 computer‐assisted telephone interviews at baseline and at 3 and 6 months. Patient‐reported outcomes collected during these interviews were summarised onto a feedback sheet by a specially developed computer programme, which identified issues of concern (e.g. severe symptoms). In the TCW arm, feedback sheets were forwarded to telephone care workers (TCWs), who then telephoned participants to discuss reported issues of concern and used a modified version of the Cancer Helpline database to refer participants to appropriate resources/services consistent with recommended feedback sheet strategies. TCWs also followed up with participants at 6‐week intervals to assess coping. In the oncologist/GP arm, 2 hard copies of feedback sheets were mailed to participants’ nominated oncologists and GPs for discussion at their next appointments. Clinicians were asked to keep 1 feedback sheet for their records and to return the second, which indicated which issues of concern were discussed and whether any actions were taken Control group: usual care (not described) Interventionist: oncology nurses with telephone counselling training |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Random assignment to one of the three study groups, by using a computer‐generated algorithm, occurred at completion of the baseline CATI and included stratification by sex and tumour type |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | High risk | Owing to the nature of the intervention, participants and personnel were not blinded |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Loss to follow‐up < 20%; similar reasons between groups |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting on outcomes |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Kroenke 2010.
| Study characteristics | ||
| Methods |
Setting: Indiana, USA Recruitment: eligible participants had a diagnosis of cancer and were screened positive for either pain or depression with scores of 6 or higher for cancer‐related pain, or 10 or higher for depression, as measured using 2 validated measurement instruments. Participants were recruited from 16 urban and rural oncology practices. Participants presenting for oncology clinic visits underwent an eligibility interview, and eligible participants willing to participate provided audiotaped oral informed consent (followed up by posted written consent forms) and completed a baseline interview Randomisation: RCT; block‐randomised in block sizes of 4, 8, and 12 and stratified by symptom (pain vs depression) |
|
| Participants | 405 men and women suffering from cancer‐related depression or pain | |
| Interventions |
Intervention: consisted of centralised tele‐care management coupled with automated home‐based symptom monitoring by interactive voice recording or Internet. Participants received a baseline call and 3 follow‐up calls (1, 4, and 12 weeks) during the first 3 months of treatment, as well as triggered telephone calls when automated monitoring indicated inadequate symptom improvement, non‐adherence to medication, adverse effects, suicidal ideation, or a participant request to be contacted. The automated symptom monitoring survey was administered twice a week for the first 3 weeks, then weekly during weeks 4 through 11, twice a month during months 3 through 6, and once a month during months 7 through 12 Control group: usual care. Participants in this arm were informed of their depressive and pain symptoms, and their screening results were provided to the oncologist. No further attempts were made by study personnel to influence depression or pain management unless a psychiatric emergency arose (e.g. suicidal ideation was detected on baseline or follow‐up outcome assessment) Interventionist: nurse care manager trained in assessing symptom response and medication adherence, in providing pain and depression‐specific education, and in making treatment adjustments according to evidence‐based guidelines |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomisation was computer‐generated in randomly varying block sizes of 4, 8, and 12 and was stratified by symptom type (pain only, depression only, or both pain and depression) |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | Low risk | Outcome assessments were conducted by a research assistant blinded to study group |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Loss to follow‐up 34% |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting on outcomes |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Livingston 2010.
| Study characteristics | ||
| Methods |
Setting: Victoria, Australia Recruitment: eligible participants were men recently diagnosed with colorectal or prostate cancer stage I‐III, who spoke sufficient English, had no psychiatric illness, and had a prognosis longer than 52 weeks. Participants were recruited at the end of the consultation, at which time they received their diagnosis. Specialists followed a standardised script and introduced participants to the concept of the Cancer Helpline and stated that they would arrange for the Helpline to contact them, or (if in the usual care group) recommended that participants contact the Helpline themselves. The specialist provided a referral slip detailing the number of outcalls the participants would receive, or the Helpline toll‐free number to call themselves. The specialist then advised participants that a study was being conducted about the needs of men with cancer, gave them an information package about the study, and asked them to consider taking part Randomisation: RCT. Block randomisation: medical specialists were randomised to 1 of 3 trial arms and then enrolled 15 consecutive participants to that arm, 15 participants to the next arm, and so forth. Specialists were blinded to which arm they were referring to |
|
| Participants | 571 men with colorectal or prostate cancer | |
| Interventions |
Intervention: consisted of 2 different interventions: (1) active referral ‐ 4 outcalls, in which men received 4 telephone calls from the Cancer Helpline within 1 week of diagnosis, and at 6 weeks, 3 months, and 6 months post diagnosis, and (2) active referral ‐ 1 outcall, in which men received 1 telephone call within 1 week of diagnosis. In both arms, participants were called and offered to discuss 10 different topics: the cancer diagnosis; treatment/management issues; what to expect from surgery; management of side effects; communication with the specialist; partner/family issues; psychological/emotional and communication concerns; understanding cancer language; diet and nutrition; other support services; and availability of printed resources. If participants did not mention a topic, the cancer nurse raised the topic. All topics discussed and information requested were documented Control group: passive referral arm; designed to resemble usual care. Participants were referred to the Cancer Helpline, with contact at their initiative Interventionist: Cancer Helpline cancer nurses with postgraduate counselling qualifications and minimum 5 years clinical oncology experience |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Participants were block randomised (15 at a time) to each of the 3 study arms by specialist clinicians via computer‐generated random numbers produced by the project co‐ordinator under supervision of the investigators |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | High risk | Owing to the nature of the intervention, participants and personnel were not blinded |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Loss to follow‐up < 20%; similar reasons between groups |
| Selective reporting (reporting bias) | Unclear risk | Significant differences between active referral ‐ 4 outcalls and passive referral arms were presented. Non‐significant differences between the active referral ‐ 1 outcall and active referral ‐ 4 outcalls arms were mentioned but were not presented. However, lack of effect of the active referral intervention is discussed |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Mishel 2002.
| Study characteristics | ||
| Methods |
Setting: central and eastern North Carolina, USA Recruitment: eligible patients were African American and Caucasian men who were diagnosed with localised prostate cancer and were within 2 weeks of catheter removal post surgery and/or < 3 weeks of starting radiation therapy, had no major cognitive impairment, had no concurrent other cancer, had access to a telephone, had a family member willing to participate in the study, and planned to reside in their current community for 12 months. Participants were recruited from 9 treatment facilities. Members of the research team introduced the study to participants during a clinic visit, and interested participants were subsequently mailed an information package and were called to determine willingness to participate Randomisation: RCT using 3 × 2 block randomisation |
|
| Participants | 239 men with prostate cancer | |
| Interventions |
Intervention: based on the Theory of Uncertainty in Illness, which assumes that uncertainty occurs when participants lack the information or knowledge needed to fully understand their illness and treatments. Through cognitive re‐framing, participants can learn to cope better with uncertainty. The intervention consisted of 8 × weekly phone calls, which were delivered in different ways in the 2 intervention arms: (1) direct, in which only the participant received the intervention, and (2) supplemented, with supplemented delivery to a close family member. An assessment was made by telephone to identify the participant's cancer‐related concerns. In the supplemented arm, the spouse or designated family support person also received a weekly phone call for 8 weeks from a nurse who conducted a similar assessment of family members’ concerns about the participant using a list similar to that used for the participants. Based on identified concerns, an appropriate intervention was then selected from a standardised list, which included validating and reinforcing views and behaviours, providing information, activating resources, teaching symptom management strategies, structuring expectations, applying problem‐solving, and teaching assertion techniques for communicating with health care providers. These interventions were applied to concerns about diagnosis, treatment, response to treatment, living with cancer, caring for oneself, and social/lifestyle issues. Printed materials, audiotapes, and videotapes for managing specific problems were mailed to participants after each weekly call. Any materials that were sent to the participant were reviewed at the next phone call Control group: usual care, which included printed general health information not related to prostate cancer or the side effects of treatment Interventionist: trained nurses matched with the participant and the family member by ethnicity and gender |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | The generation of random sequence is not described |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | "Data collectors who were not involved in the delivery of the intervention collected all data". It is unclear if participants were blinded to group |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Low loss to follow‐up, but reasons for this are not described |
| Selective reporting (reporting bias) | Low risk | Results of all measures are reported, including non‐significant findings. However, the paper focuses slightly more on significant differences found at T2 than on non‐significant differences at T3 |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Mishel 2005.
| Study characteristics | ||
| Methods |
Setting: cancer centres and hospitals in North Carolina, USA Recruitment: eligible participants were African American and Caucasian women who were 5 to 9 years post treatment for breast cancer, had no cognitive impairment, had no concurrent treatment for another cancer, were recurrence free, could be on tamoxifen or similar agents, had access to a telephone, and planned to reside in their current community for 2 years following entry into the study. Participants were identified using the tumour registries from 13 cancer centres and hospitals. Introductory letters were sent to these women to ask permission to release their name and contact details to the research team, who then mailed letters about the study to interested women and later called them to determine willingness to participate. Additional African American women were recruited from the community via announcements in local media and community volunteers Randomisation: RCT, block‐randomised by ethnicity |
|
| Participants | 509 African American and Caucasian breast cancer survivors | |
| Interventions |
Intervention: based on the Theory of Uncertainty in Illness, which assumes that uncertainty occurs when participants lack the information or knowledge needed to fully understand their illness and treatments. Through cognitive re‐framing, participants can learn to cope better with uncertainty. The intervention had 2 main components: (1) cognitive strategies delivered via audiotapes to teach active emotion‐focused coping responses to threats of recurrence; and (2) behavioural strategies packaged in a self‐help manual designed to provide management skills, information and resources on long‐term treatment side effects, and cancer resources. Using a standardised protocol, interventionists guided women through the intervention over the course of 4 weekly telephone calls. The women practised 1 of the 4 cognitive coping skills each week during the calls: (1) relaxation, (2) pleasant imagery, (3) calming self‐talk, and (4) distraction. The women were instructed to use these skills when confronting a trigger of fear of recurrence, or when anticipating such a trigger. During the third and fourth telephone calls, women were guided in use of the manual, which contained behavioural strategies in the form of information about long‐term treatment side effects. During each telephone session, women were encouraged to practise the skills by listening to audiotapes and using the manual until the next call Control group: usual care. No attempt was made to limit exposure to naturally occurring learning contexts such as media coverage of cancer issues, public health programmes, or cancer support groups Interventionist: nurses (specific training not described) |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | The SAS program Proc Plan was used to construct a randomisation plan |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | High risk | Owing to the nature of the intervention, participants and personnel were not blinded |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Low loss to follow‐up, but reasons for this are not described |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting on outcomes |
| Other bias | Unclear risk | At each data collection point, each woman received a "gasoline card worth $20" |
Molassiotis 2009.
| Study characteristics | ||
| Methods |
Setting: Manchester, UK Recruitment: eligible participants were 18 years of age or older, were diagnosed with breast or colorectal cancer with a life expectancy longer than 6 months and starting oral capecitabine, were able to self‐care and communicate in English, lived within 35 miles of the hospital, and had no past experience in home care nursing programmes. Participants were recruited Randomisation: RCT |
|
| Participants | 164 men and women aged 18 years or older with colorectal or breast cancer | |
| Interventions |
Intervention: no theoretical basis; developed on best evidence for symptom management from the nursing literature. Participants received a home care nursing (HCN) programme for 18 weeks. The HCN programme consisted of symptom assessment, participant education, and/or treatment of symptoms. The nurse conducted 1 standard home visit (1 to 1.5 hours) during the first week of capecitabine treatment, in which chemotherapy and its adverse effects were discussed, questions participants had were sought and answered, and support was given. Subsequent home visits were offered when participants experienced multiple grade 3 toxicities or had difficulty coping with chemotherapy. All home care participants also received 1 monitoring phone call per week during all cycles (i.e. a minimum of 18 phone contacts during the intervention). These lasted between 10 and 25 minutes and included toxicity assessment followed by discussion of possible strategies to deal with reported symptoms. Participants also had access to a 24‐hour, on‐call specialist nursing service Control group: usual care consisting of information about oral capecitabine and its adverse effects provided by clinicians and accompanied by printed information. Relevant medications to relieve symptoms were prescribed as needed. Participants were given the hospital's 24‐hour emergency hotline phone number Interventionist: nurses with training and experience in home care and in cancer care who received additional training relevant to the intervention |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Random assignment was carried out by an independent statistician through a computer‐generated program |
| Allocation concealment (selection bias) | Low risk | No clinician or researcher could anticipate or direct allocation |
| Blinding (performance bias and detection bias) All outcomes | Low risk | Interventionist and participants not blinded, but outcome assessor blinded |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Loss to follow‐up < 20%; similar reasons between groups |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting on outcomes |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Mosher 2016.
| Study characteristics | ||
| Methods |
Setting: Indiana, USA Recruitment: eligible participants were diagnosed with small cell or non‐small cell lung cancer; had at least 1 symptom of moderate severity, defined by validated cutoff points for depressive symptoms, anxiety, pain, vitality, or breathlessness; were fluent in English; and had a consenting carer Randomisation: RCT; randomisation by a person not involved with study delivery using an SAS procedure |
|
| Participants | 106 dyads comprising a man/woman with symptomatic lung cancer and his/her carer | |
| Interventions |
Intervention: based on Social‐Cognitive Theory. Participants took part in four 45‐minute telephone symptom management (TSM) sessions. Both dyad members participated simultaneously through speaker phone. Received instruction in symptom management strategies. Mailed handouts detailing major points discussed during sessions, along with home practice assignments and digital media with instructions for relaxation exercises. Primary goal of intervention was to teach participants and carers evidence‐based cognitive‐behavioural and emotion‐focused strategies for managing symptoms. All sessions had dual focus on participant and carer concerns Control group: four 45‐minute telephone sessions delivered to dyads by speaker phone aimed at directing participants to resources for practical health information and psychosocial services Interventionist: clinical social workers trained by a PhD psychologist |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Generated by an individual independent from the study. Stratified by participant gender and performance status |
| Allocation concealment (selection bias) | Unclear risk | No details |
| Blinding (performance bias and detection bias) All outcomes | Low risk | Research assistants blind to study condition conducted all assignments by telephone |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Loss to follow‐up over 20%; reasons similar between groups |
| Selective reporting (reporting bias) | Low risk | Balanced reporting for all measures |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Porter 2011.
| Study characteristics | ||
| Methods |
Setting: North Carolina, USA Recruitment: eligible participants had a diagnosis of non‐small cell lung cancer stage I‐III or limited‐stage small cell lung cancer, had no other cancers in the past 5 years, were able to read and speak English, and had a carer who was also willing to participate. Participants were recruited from a university hospital oncology programme and from community oncology clinics Randomisation: RCT |
|
| Participants | 233 lung cancer patients and their carers | |
| Interventions |
Intervention: coping skills training based on cognitive‐behavioural therapy principles. Participants and their carers received either (1) carer‐assisted coping skills training, or (2) education/support involving the carer. Both arms consisted of 14 × 45‐minute telephone‐based sessions delivered to the participant and the carer simultaneously. Sessions were conducted over an 8‐month period tapered from weekly (sessions 1 to 3) to biweekly (sessions 4 to 10) to monthly (sessions 11 to 14). Telephone sessions were supplemented with printed and digital/audiovisual materials with instructions for progressive muscle relaxation. Sessions 1 to 7 focused on specific coping skills; sessions 8 to 12 focused on smoking cessation, relaxation and imagery exercises, and application of coping skills to particular challenges faced by the participant and/or carer; session 11 focused specifically on the carer, encouraging the carer to explore his/her own resources and sources of stress and discussing how to use coping skills to ease the burden of care giving; sessions 13 and 14 focused on maintenance strategies including a review of coping skills learned and how to maintain regular practice of skills to prevent and cope with possible setbacks Control group: participants and carers in the education/support arm received information about lung cancer and its treatment via a presentation and discussion format. Sessions were supplemented with handouts summarising major points and listing additional resources (e.g. websites, books) that participants could access if desired. Participants in this condition did not receive any training in coping skills Interventionist: both treatment conditions were delivered by registered nurses trained by PhD‐level psychologists and medical oncologists |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomisation assignments were generated by an individual not involved in the study via a random‐numbers table |
| Allocation concealment (selection bias) | Low risk | Assignments were concealed in envelopes that were not opened until participants had completed their pre‐treatment evaluation |
| Blinding (performance bias and detection bias) All outcomes | Low risk | Research assistants conducting assessments were blind to treatment condition |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | High loss to follow‐up; reasons for this not described in detail |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting |
| Other bias | Unclear risk | Lack of a no treatment control group |
Rawl 2002.
| Study characteristics | ||
| Methods |
Setting: urban, tertiary cancer centre and community‐based cancer centre in a medium‐sized midwestern city, USA Recruitment: eligible participants were aged 18 years or older; were newly diagnosed with breast, colorectal, or lung cancer; were undergoing chemotherapy; had identified carers; and spoke English. Participants were recruited by a trained recruiter, who explained the study Randomisation: RCT |
|
| Participants | 109 men and women with breast, colon, or lung cancer and their partners | |
| Interventions |
Intervention: based on self‐regulation theory, which suggests that concrete, objective information in combination with appropriate emotional support can help minimise disruption of a participant's usual activities resulting in decreased psychological distress. A computer programme guided the clinical intervention, which consisted of 5 face‐to‐face visits and 4 telephone calls delivered alternately every second week over 18 weeks. During the initial visit, the nurse took a brief history, assessed the participant's symptoms, and developed a plan of care tailored to the participant. In each following visit/call, nurses assessed participants' physical, mental, and resource needs and their symptoms, and tailored interventions to participants using the computer programme. Effectiveness of interventions used to cope with these needs was assessed, and nurses offered emotional and counselling support to participants Control group: usual care, which consisted of verbal information about what to expect from chemotherapy and symptoms that should be reported to the doctor. Participants received any education normally delivered during chemotherapy but no attention outside of medical visits Interventionist: master's prepared oncology nurse specialist trained specifically in the intervention protocol and in use of the computer‐based nursing system |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Group assignment was generated by computer |
| Allocation concealment (selection bias) | High risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | Low risk | Interviewers who collected outcome data were blind to respondents' group assignments |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Attrition over 20%, and the intervention had twice the level of attrition as the control. Nine withdrew from the study due to the time‐consuming nature of the intervention |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Ream 2015.
| Study characteristics | ||
| Methods |
Setting: UK Recruitment: patients were eligible if they were receiving their first course of intravenous (IV) chemotherapy for treatment of breast or colorectal cancer or a lymphoma, were aged 18 years or over, had experienced at least moderate fatigue during previous treatment cycles, and had sufficient proficiency in English to complete the study. Participants were recruited when they attended their third cycle of chemotherapy Randomisation: RCT via a table of random numbers |
|
| Participants | 44 participants with breast or colorectal cancer were recruited to the trial; 37 participants completed the trial | |
| Interventions |
Intervention: the Beating Fatigue by Telephone intervention comprised education on fatigue; assessment and monitoring of fatigue; coaching in self‐care; and provision of emotional support. Participants in this arm were provided with a Coping with Fatigue booklet (Macmillan Cancer Support) and an investigator‐designed handbook and fatigue diary. Telephone calls were scripted and specifically addressed fatigue through motivational interviewing techniques. Participants received 3 telephone calls Control: people randomised to the control group were provided usual care: basic screening for fatigue and limited self‐care advice without onward referral Interventionist: a cancer nurse with experience of working on a Cancer Helpline |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Participants were randomised by simple randomisation via a table of random numbers between intervention and usual care |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | Low risk | Researchers collecting outcome measures were blind to the intervention. Health professionals providing care were blind to the intervention |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Loss to follow‐up < 20%; similar reasons between groups |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting |
| Other bias | Unclear risk | The intervention group had higher scores at baseline compared to the control group in all variables under investigation ‐ given small sample size, this may be important |
Reese 2014.
| Study characteristics | ||
| Methods |
Setting: Baltimore, USA (not specified) Recruitment: eligible patients had undergone surgery/other treatment for colorectal cancer and had answered yes to having sexual concerns. In addition, they were required to be over 21 years old with a partner of at least 1 year, with sufficient reading/writing skills in English. Partners were required to be over 21 with sufficient English to participate Randomisation: RCT stratified by participant gender and current ostomy use |
|
| Participants | 18 participants treated for colorectal cancer and their partners | |
| Interventions |
Intervention: participants and their partners were randomly assigned to the intimacy enhancement (IE) intervention or a wait‐list control group (WL). The IE intervention consisted of 4 50‐minute (weekly) telephone sessions, which included techniques from sex therapy and couple/marital therapy such as sensual touching exercises, improving sexual communication, identifying/challenging overly negative or rigid sexually related cognitions, and intimacy‐building activities Control: after both assessments had been completed in the waiting‐list condition, the couple was able to participate in the IE intervention Interventionist: previous paper outlining this intervention says, "all sessions of the intervention were delivered by the first author, J.B.R., a clinical psychologist" |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Randomisation was stratified by participant gender and ostomy use |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | High risk | Owing to the nature of the intervention, participants and interveners were not blinded |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Loss to follow‐up < 20%; similar reasons between groups |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting |
| Other bias | Unclear risk | Couples were given $25 if they completed both assessments |
Reese 2018.
| Study characteristics | ||
| Methods |
Setting: USA Recruitment: breast cancer survivors were identified through providers’ schedules and the institutional tumour registry. Women were eligible if they (a) were age ≥ 21years, (b) were in a stable relationship (i.e. living with a romantic partner for ≥ 6 months) that could involve sexual activity, (c) had completed active treatment 6 months to 5 years ago for non‐recurrent stage I–III breast cancer (current use of endocrine therapy was acceptable), and (d) scored ≥ 3 on the Patient Care Monitor sexual concerns screening item. Women were excluded if they had a past history of any cancer other than non‐melanoma skin cancer, an ECOG performance score > 2 (or were judged as being too ill to participate), or overt cognitive dysfunction or psychiatric disturbance; were pregnant; or were currently engaged in couple or marital therapy; or if either they or their partner had hearing impairment or were not able to speak English. No exclusions were made based on sexual orientation Randomisation: 2‐arm RCT |
|
| Participants | 29 women with breast cancer and their partners (20 IE, 9 LHT) | |
| Interventions | Both arms consisted of four 60 to 75 minute weekly sessions delivered by telephone to both members of the couple by the interventionist and included session handouts for both survivors and partners sent in advance of the scheduled session Intimacy Enhancement (IE) arm: 4 sessions of education and skills training to address survivors’ sexual concerns and to improve intimacy. Techniques include sensual touching exercises adapted to incorporate specific instructions for how to deal with thoughts and feelings about breast touching during these exercises, communication skills with regard to sex and intimacy, identifying and challenging overly negative or inflexible sexually related cognitions, and broadening the repertoire of both sexual and non‐sexual intimacy building activities. Each of the 4 sessions includes a detailed agenda and topic, 1 or more skills practice sessions, and assignment of home practice exercises Living Healthy Together (LHT) arm: education and support across a range of topics: education on finding support and the breast cancer experience, what it means to “live healthy together” (session 1), stress and stress management (session 2), fatigue and sleep (session 3), and diet and nutrition (session 4). Each session consisted of interactive discussion and activities including self‐assessment and discussions both with the interventionist and between members of the couple in relation to the session topic Interventionist: trained health care professionals (social work or counselling/psychology) |
|
| Outcomes |
Method of assessing outcome measures:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Study biostatistician generated the randomisation sequence (2:1 ratio) |
| Allocation concealment (selection bias) | High risk | Project manager assigned participants to interventions |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | No details |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Study retention 97% |
| Selective reporting (reporting bias) | Low risk | Balanced reporting for all measures |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Sherwood 2005.
| Study characteristics | ||
| Methods |
Setting: Michigan, Indiana, and Ohio, USA Recruitment: eligible participants were aged 21 years or older; were newly diagnosed with any solid tumour or non‐Hodgkin's lymphoma stage III, IV, or recurrent; were undergoing chemotherapy; had no cognitive disabilities; had access to a telephone; and were able to read and speak English. Participants were recruited from 6 urban cancer centres by trained recruiters, who explained the study and obtained written consent Randomisation: RCT |
|
| Participants | 124 men and women aged 21 years or older with various cancers | |
| Interventions |
Intervention: based on cognitive‐behavioural theory, which states that an individual's perception of a situation affects his/her behaviour and beliefs regarding ability to control it. Participants' ability to manage symptoms can therefore be improved by changing their perception of them. The intervention consisted of 5 contacts spread over 8 weeks. The first and last contacts were face‐to‐face and were used mainly to establish rapport and to facilitate closure. The second, third, and fourth contacts were delivered by telephone. During each contact, nurses assessed symptoms and participants rated the severity of symptoms. Participants selected symptoms they would like to focus on, and nurses tailored a list of interventions. Nurses helped participants re‐frame attitudes and beliefs and proposed cognitive‐behavioural strategies. Participants then agreed to implement the interventions. Participants were responsible for choosing and implementing the strategies Control group: usual care (not described) Interventionist: nurse with experience in oncology trained in the study protocol |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Following baseline data collection, a stratified randomisation schema was used to randomly assign participants from each recruitment site |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | Low risk | Data collection interviewers were not nurses and were not aware of which arm of the study participants were in |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Attrition over 20%; reasons for this not reported |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Sikorskii 2007.
| Study characteristics | ||
| Methods |
Setting: Michigan and Indiana, USA Recruitment: eligible patients were 21 years or older, had a diagnosis of a solid tumour cancer or non‐Hodgkin's lymphoma, were undergoing chemotherapy, were able to speak and read English, and had access to a touch‐tone telephone. Participants were recruited from 2 comprehensive cancer centres, 2 community cancer oncology programme, and 6 hospital‐affiliated community oncology centres by nurses from the clinical trial offices at these sites Randomisation: RCT; participants were randomised according to recruitment location and site of cancer |
|
| Participants | 435 men and women with various cancers | |
| Interventions |
Intervention: no specified theoretical basis but incorporates elements of coping, re‐framing, education, and eliciting support for adapting to or overcoming problems such as managing cancer symptoms. Consisted of (1) an automated telephone symptom management (ATSM) intervention delivered via an automated system, or (2) nurse‐assisted symptom management (NASM). Participants in both arms received a total of 6 telephone calls over 8 weeks; weekly calls during the first 4 weeks; then 2 biweekly calls. For participants assigned to the NASM group, nurses delivered up to 4 strategies for each symptom, supplemented with references to a symptom management guide. At each subsequent contact, assigned strategies were evaluated. If a strategy was not tried, or was tried but was found not helpful, participants were counselled as to how they might fit strategy into their daily activities, or they were offered different strategies. Successful strategies were reinforced and continued. In the ATSM arm, a pre‐recorded female voice queried participants regarding severity of the 17 symptoms. To rate severity, participants pressed the appropriate numbers on their telephone keypads. For symptoms rated at 4 or higher, participants were directed to the section of the symptom management guide that informed them about strategies to manage each symptom Control group: no usual care group Interventionist: experienced cancer nurses; specific training not described |
|
| Outcomes |
Methods for assessing outcomes
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomisation was conducted via a computer minimisation programme |
| Allocation concealment (selection bias) | Unclear risk | Not stated |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not stated |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Loss to follow‐up 20%; reasons not detailed |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Thomas 2012.
| Study characteristics | ||
| Methods |
Setting: California and New Jersey, USA Recruitment: eligible participants were 18 years of age or older, were able to read and understand English, had access to a telephone, had a life expectancy longer than 6 months, had cancer‐related pain with an average pain intensity score of 2 or higher (on a 0 to 10 scale), had no cognitive or psychiatric condition, had no substance abuse problem, had no severe pain unrelated to their cancer, or resided in a setting where they could not self‐administer pain medication (e.g. nursing home). Potential participants were identified by clinical staff and were recruited from 6 outpatient oncology clinics (4 Veterans Affairs facilities, 1 county hospital, and 1 community‐based practice) by a member of the research team Randomisation: RCT; participants were stratified by pain intensity and treatment via permuted blocks with variable sizes |
|
| Participants | 318 men and women aged 18 years or older with various types of cancer‐related pain | |
| Interventions |
Intervention: based on change theory, specifically the Transtheoretical Model, in which behavioural change is a function of a person's state of readiness or motivation to modify a particular behaviour. Uses principles of coaching and motivational interviewing to modify participants' attitudes towards pain management. Participants received either (1) an educational intervention or (2) a coaching intervention. Both groups viewed a video on overcoming attitudinal barriers and received a pamphlet on managing cancer pain. In addition, all participants received 4 × 30 minute biweekly telephone calls conducted over 6 weeks. In the coaching group, the calls explored participants' beliefs about pain, use of pain medication, non‐pharmacological pain management strategies, and communication about pain management. In the educational and usual care groups, the calls were for attention control (content not described) Control group: usual care. Participants viewed a video on cancer produced by the American Cancer Society Interventionist: advanced practice oncology nurse with expertise in cancer pain management trained in the Transtheoretical Model and in motivational interviewing by a cognitive‐behavioural psychologist, and in the specific coaching protocol used in the study. Research associates were trained in providing attention control telephone calls for the control group |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Participants were randomised via permuted blocks with variable sizes |
| Allocation concealment (selection bias) | Low risk | Participants and clinicians were blinded to participants' group assignments |
| Blinding (performance bias and detection bias) All outcomes | High risk | Owing to the nature of the intervention, participants and personnel were not blinded |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Loss to follow‐up over 20%; no detail provided on reasons for loss to follow‐up between study groups |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
Traeger 2015.
| Study characteristics | ||
| Methods |
Setting: Massachusetts, USA Recruitment: eligible participants were 18 years or older; had a diagnosis of stage I‐III breast, lung, or colorectal cancer and were scheduled to start chemotherapy; and were able to respond to questionnaires in English. Participants were recruited by a study co‐ordinator during their first chemotherapy visit. They were recruited consecutively until target enrolment was reached Randomisation: by a clinical trials office (1:1 allocation sequence) with stratification by tumour type |
|
| Participants | 120 participants initiating chemotherapy for non‐metastatic disease (60 breast cancer; 30 colorectal cancer; 30 lung cancer) | |
| Interventions | Intervention: provocative telephone‐based nursing guidance and support during first 2 chemotherapy cycles. Interventionists had a brief outline to structure calls but were encouraged to complete them according to clinical judgement to reflect participant‐centred practice. Two calls were provided during the first week after first chemotherapy administration, and 2 in the first week after the second cycle. Calls lasted around 15 minutes | |
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Dana Faber/Harvard Cancer Center Quality Assurance Officer for clinical trials randomly assigned participants |
| Allocation concealment (selection bias) | Unclear risk | For control participants, the nurse practitioner was not notified of study enrolment or group assignment |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | No details |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Study retention rate 97.5% |
| Selective reporting (reporting bias) | Low risk | Balanced reporting of all measures |
| Other bias | High risk | If the electronic health record showed a documented in‐person oncology visit during the intervention delivery window, the in‐person visit was counted as a completed call |
Watson 2017.
| Study characteristics | ||
| Methods |
Setting: UK Recruitment: a consecutive series of participants referred to the Royal Marsden Hospital's Psychological Care Service over an 18‐month period by clinical staff Randomisation: prospective randomised equivalence trial |
|
| Participants | 118 cancer participants greater than 8 weeks post diagnosis with a minimum prognosis greater than 3 months who were considered to have high psychological needs were randomised. Sixty participants were randomised to telephone‐CBT (T‐CBT), and 58 to treatment as usual‐CBT (TAU‐CBT) (43 and 35 provided complete analysable data) | |
| Interventions | Prospective randomised equivalence trial comparing TAU‐CBT with T‐CBT. A no‐treatment control group was not used, given prior data indicating the efficacy of standard care CBT for cancer participants Telephone CBT Participants telephoned at pre‐arranged times and sessions scheduled. Up to 8 sessions offered therapy over an approximate 12‐week period. Core therapy components include:
Also provided a patient workbook and a digital relaxation resource CBT Face‐to‐Face Treatment As Usual (TAU‐CBT) Not described Interventionist: level 3/4 mental health professionals/psychologists |
|
| Outcomes |
Method of assessing outcome measures:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomisation by independent statistician, stratified by therapist |
| Allocation concealment (selection bias) | Unclear risk | No details |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | No details |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Attrition over 20%; loss to follow‐up higher in TAU‐CBT |
| Selective reporting (reporting bias) | Low risk | Balanced reporting for all measures |
| Other bias | Unclear risk | Referred based on clinician judgement of high psychological need – not screened |
Yates 2005.
| Study characteristics | ||
| Methods |
Setting: Melbourne and Brisbane, Australia Recruitment: eligible patients were aged 18 years or older, had stage I or II breast cancer, were commencing adjuvant chemotherapy, and had an Eastern Cooperative Oncology Group performance rating of 1or 2; their haemoglobin level was at least 11.6 g/mL. Participants were recruited from 5 outpatient clinics at 3 major metropolitan hospitals Randomisation: RCT |
|
| Participants | 109 women aged 18 years or older with breast cancer | |
| Interventions |
Intervention: psycho‐educational intervention aiming to improve participants' knowledge and skills to enable them to perform self‐care behaviours designed to minimise fatigue. Consisted of 3 sessions tailored to participants' individual needs and circumstances. The first session consisted of a 20‐minute face‐to‐face meeting in the clinic at the participant's second chemotherapy course, and focused on techniques of information‐giving, problem‐solving, rehearsal, and reinforcement. The second and third sessions were delivered by telephone, were conducted a week apart, and lasted 10 minutes each. In these sessions, participants' fatigue management strategies were reviewed. Printed information supplemented the face‐to‐face/telephone interactions Control group: participants received general cancer education in the form of verbal and printed information about cancer in sessions equivalent in number and timing to the sessions provided to the intervention group Interventionist: oncology nurses trained in the research programme and in the fatigue management or control intervention |
|
| Outcomes |
Methods for assessing outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Participants were randomly assigned to intervention or control conditions through a central telephone system via computer‐generated random numbers |
| Allocation concealment (selection bias) | Low risk | Group allocation was concealed from research assistants involved in recruitment and in baseline and follow‐up assessments |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not stated |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Loss to follow‐up < 20%; similar reasons between groups |
| Selective reporting (reporting bias) | Low risk | There appears to be no selective reporting |
| Other bias | Low risk | The trial appears to be free of other problems that could put it at high risk of bias |
CBT: cognitive‐behavioural therapy.
ECOG: Eastern Cooperative Oncology Group.
GP: general practitioner.
RCT: randomised controlled trial.
SIGN: Scottish Intercollegiate Guidelines Network.
TAU: treatment as usual.
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Aranda 2006 | Not primarily telephone intervention |
| Ashing‐Giwa 2008 | Not focused on symptom management |
| Bakitas 2009 | Not focused on symptom management |
| Beaver 2006 | Not an RCT |
| Beaver 2009 | Not focused on symptom management |
| Beney 2002 | Not focused on symptom management |
| Bohnenkamp 2004 | Not primarily telephone intervention |
| Budin 2008 | Not focused on symptom management |
| Campbell 2007 | Not focused on symptom management |
| Cheville 2013 | Not focused on symptom management |
| Coleman 2005 | Not focused on symptom management |
| Collie 2006 | Not primarily telephone intervention |
| Craddock 1999 | Not focused on symptom management |
| Dalton 2004 | Not primarily telephone intervention |
| De Wit 1997 | Not primarily telephone intervention |
| DuHamel 2015 | Not primarily telephone intervention |
| Given 2008 | Subanalysis of included article (Sikorskii 2007) |
| Haddad 2003 | Not focused on symptom management |
| Hagiopan 1990 | Not focused on symptom management |
| Halbert 2004 | Not exclusively cancer patients |
| Hanks 2002 | Not exclusively cancer patients |
| Harrison 2011 | Not focused on symptom management |
| Hawkes 2009 | Not an RCT |
| Hawkins 2010 | Not focused on symptom management |
| Hayes 2012 | Not focused on symptom management |
| Kearney 2009 | Not primarily telephone intervention |
| Kimman 2010 | Not focused on symptom management |
| Koller 2013 | Not primarily telephone intervention |
| Kornblinth 2006 | Not delivered by an HCP |
| Marcus 2010 | Not delivered by an HCP |
| McCorkle 2009 | Not primarily telephone intervention |
| Meneses 2009 | Not primarily telephone intervention |
| Nguyen 2018 | Not primarily telephone intervention |
| Park 2012 | Not focused on symptom management |
| Rustoen 2014 | Not focused on symptom management |
| Salonen 2009 | Not focused on symptom management |
| Sandgren 2000 | Not focused on symptom management |
| Sandgren 2003 | Not focused on symptom management |
| Sandgren 2007 | Not focused on symptom management |
| Santacroce 2010 | Not adults |
| Sikorskii 2009 | Duplicate of included article (Sikorskii 2007) |
| Sikorskii 2009b | Duplicate of included article (Sikorskii 2007) |
| Spoelstra 2013 | Not delivered by an HCP |
| Williams 2004 | Not primarily telephone intervention |
| Winger 2018 | Secondary analysis of included article (Mosher 2016) |
HCP: health care practitioner.
RCT: randomised controlled trial.
Differences between protocol and review
The protocol stated that the review would incorporate quality of life as a secondary outcome. However, quality of life resulting from a telephone‐delivered intervention for symptom management proved an oft measured outcome that warrants a review in its own right. Consequently, these data have not been reported here. Rather, general symptom experience is presented as the secondary outcome.
Contributions of authors
ER: background, objectives, and review development; data extraction and analysis; manuscript writing. AR: background, objectives, and review development; data extraction; manuscript review. AH, VHP, AC, KC: literature search; organising/co‐ordinating of data extraction; data extraction and analysis; manuscript writing. TW, AF: data extraction; manuscript review.
Sources of support
Internal sources
King's College, London, UK
University of Surrey, Guildford, UK
External sources
NCRI Supportive & Palliative Care (SuPaC) Research Collaborative Capacity Building Grant, UK
Cochrane Gynaecological, Neuro‐oncology, and Orphan Cancers, UK
Declarations of interest
Emma Ream: principal investigator of a trial that assessed the effectiveness of a telephone intervention for cancer patients experiencing fatigue. Amanda Amanda Euesden Hughes: none known. Anna Cox: none known. Katy Skarparis: none known. Alison Richardson: none known. Vibe H Pedersen: none known. Theresa Wiseman: none known. Angus Forbes: none known. Andrew Bryant: none known.
New
References
References to studies included in this review
Allard 2007 {published data only}
- Allard N. Day surgery for breast cancer: effects of a psychoeducational telephone intervention on functional status and emotional distress. Oncology Nursing Forum 2007;34(1):133-41. [DOI] [PubMed] [Google Scholar]
Allen 2002 {published data only}
- Allen S, Shah A, Nezu A, Nezu C, Ciambrone D, Hogan J, et al. A problem-solving approach to stress reduction among younger women with breast carcinoma: a randomized controlled trial. Cancer 2002;94(12):3089-100. [DOI] [PubMed] [Google Scholar]
Badger 2005 {published data only}
- Badger T, Segrin C, Meek P, Lopez A, Bonham E, Sieger A. Telephone interpersonal counseling with women with breast cancer: symptom management and quality of life. Oncology Nursing Forum 2005;32:273-9. [DOI] [PubMed] [Google Scholar]
Badger 2007 {published data only}
- Badger T, Segrin C, Dorros S, Meek P, Lopez A. Depression and anxiety in women with breast cancer and their partners. Nursing Research 2007;56:44-53. [DOI] [PubMed] [Google Scholar]
Badger 2013a {published data only}
- Badger T, Segrin C, Pasvogel A, Lopez AM. The effect of psychosocial interventions delivered by telephone and videophone on quality of life in early-stage breast cancer survivors and their supportive partners. Journal of Telemedicine and Telecare 2013;19(5):260-5. [DOI] [PubMed] [Google Scholar]
Badger 2013b {published data only}
- Badger T, Segrin C, Hepworth J, Pasvogel A, Weihs K, Lopez A. Telephone-delivered health education and interpersonal counseling improve quality of life for Latinas with breast cancer and their supportive partners. Psycho-Oncology 2012;22(5):1035–42. [DOI] [PubMed] [Google Scholar]
Badr 2015 {published data only}
- Badr H, Smith CB, Goldstein NE, Gomez JE, Redd WH. Dyadic psychosocial intervention for advanced lung cancer patients and their family care-givers: results of a randomized pilot trial. Cancer 2015;121(1):150-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Bailey 2004 {published data only}
- Bailey D, Mishel M, Belyea M, Stewart J, Mohler J. Uncertainty intervention for watchful waiting in prostate cancer. Cancer Nursing 2004;27:339-46. [DOI] [PubMed] [Google Scholar]
Barsevick 2004 {published data only}
- Barsevick A, Dudley W, Beck S, Sweeney C, Whitmer K, Nail L. A randomized clinical trial of energy conservation for patients with cancer-related fatigue. Cancer 2004;100(6):1302–10. [DOI] [PubMed] [Google Scholar]
Barsevick 2010 {published data only}
- Barsevick A, Stewart K, Beck S, Dudley W, Wong B, Berger A, et al. Efficacy of an intervention for fatigue and sleep disturbance during cancer chemotherapy. Journal of Pain Symptom Management 2010;40(2):200–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
Chambers 2014 {published data only}
- Chambers S, Girgis A, Occhipinti S, Hutchison S, Turner J, Mcdowell M, et al. A randomised trial comparing two low-intensity psychological interventions for distressed patients with cancer and their caregivers. Oncology Nursing Forum 2014;41(4):E256-66. [DOI] [PubMed] [Google Scholar]
Chambers 2015 {published data only}
- Chambers SK, Occhipinti S, Schover L, Nielson L, Zajglewicz L, Clutton S. A randomised controlled trial of a couples-based sexuality intervention for men with localised prostate cancer and their female partners. Psycho-oncology 2015;24(7):748-56. [DOI] [PubMed] [Google Scholar]
Dong 2018 {published data only}
- Dong X, Sun G, ZhanJ, Liu F, Ma S, Li P, et al. Telephone-based reminiscence therapy for colorectal cancer patients undergoing postoperative chemotherapy complicated with depression: a three-arm randomised controlled trial. Supportive Care in Cancer 2018;DOI: 10.1007/s00520-018-4566-6:1-9. [DOI] [PubMed] [Google Scholar]
Downe‐Wamboldt 2007 {published data only}
- Downe-Wamboldt B, Butler L, Melanson P, Coulter L, Singleton J, Keefe J, et al. The effects and expense of augmenting usual cancer clinic care with telephone problem-solving counseling. Cancer Nursing 2007;30(6):441-53. [DOI] [PubMed] [Google Scholar]
Girgis 2009 {published data only}
- Girgis A, Breen S, Stacey F, Lecathelinais C. Impact of two supportive care interventions on anxiety, depression, quality of life, and unmet needs in patients with nonlocalized breast and colorectal cancers. Journal of Clinical Oncology 2009;27(36):6180-90. [DOI] [PubMed] [Google Scholar]
Kroenke 2010 {published data only}
- Kroenke K, Theobald D, Wu J, Norton K, Morrison G, Carpenter J, et al. Effect of telecare management on pain and depression in patients with cancer. A randomized trial. Journal of American Medical Association 2010;304(2):163-71. [DOI] [PMC free article] [PubMed] [Google Scholar]
Livingston 2010 {published data only}
- Livingston P, White V, Hayman J, Maunsell E, Dunn S, Hill D. The psychological impact of a specialist referral and telephone intervention on male cancer patients: a randomised controlled trial. Psycho-oncology 2010;19(6):617–25. [DOI] [PubMed] [Google Scholar]
Mishel 2002 {published data only}
- Mishel M, Belyea M, Germino B, Stewart J, Balley JR D, Robertson C, et al. Helping patients with localized prostate carcinoma manage uncertainty and treatment side effects: nurse-delivered psychoeducational intervention over the telephone. Cancer 2002;94(6):1854-66. [DOI] [PubMed] [Google Scholar]
Mishel 2005 {published data only}
- Mishel M, Germino B, Gil K, Belyea M, Laney I, Stewart J, et al. Benefits from an uncertainty management intervention for African-American and Caucasian older long-term breast cancer survivors. Psycho-oncology 2005;14(11):962-78. [DOI] [PubMed] [Google Scholar]
Molassiotis 2009 {published data only}
- Molassiotis A, Brearley S, Saunders M, Craven O, Wardley A, Farrell C, et al. Effectiveness of a home care nursing program in the symptom management of patients with colorectal and breast cancer receiving oral chemotherapy: a randomized, controlled trial. Journal of Clinical Oncology 2009;27(36):6191-8. [DOI] [PubMed] [Google Scholar]
Mosher 2016 {published data only}
- Mosher C, Winger J, Hanna N, Jalal S, Einhorn L, Birdas T, et al. Randomized pilot trial of a telephone symptom management intervention for symptomatic lung cancer patients and their family caregivers. Journal of Pain and Symptom Management 2016;52(4):469-82. [DOI] [PMC free article] [PubMed] [Google Scholar]
Porter 2011 {published data only}
- Porter L, Keefe F, Garst J, Baucom D, Mcbride C, Mckee D, et al. Caregiver-assisted coping skills training for lung cancer: results of a randomized clinical trial. Journal of Pain Symptom Management 2011;41(1):1-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
Rawl 2002 {published data only}
- Rawl S, Given B, Given C, Champion V, Kozachik S, Barton D, et al. Intervention to improve psychological functioning for newly diagnosed patients with cancer. Oncology Nursing Forum Online 2002;29(6):967-75. [DOI] [PubMed] [Google Scholar]
Ream 2015 {published data only}
- Ream E, Gargaro G, Barsevick A, Richardson A. Management of cancer-related fatigue during chemotherapy through telephone motivational interviewing: modeling and randomized exploratory trial. Patient Education and Counseling 2015;98:199-206. [DOI] [PubMed] [Google Scholar]
Reese 2014 {published data only}
- Reese JB, Porter LS, Regan KR, Keefe FJ, Azad NS, Diaz LA, et al. A randomised pilot trial of a telephone-based couples intervention for physical intimacy and sexual concerns in colorectal cancer. Psycho-oncology 2014;23:1005-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
Reese 2018 {published data only}
- Reese J, Clegg Smith K, Handorf E, Sorice K, Bober S, Bantug E, et al. A randomized pilot trial of a couple-based intervention addressing sexual concerns for breast cancer survivors. Journal of Psychosocial Oncology 2018;9:1005-13. [DOI: DOI: 10.1080/07347332.2018.1510869] [DOI] [PMC free article] [PubMed] [Google Scholar]
Sherwood 2005 {published data only}
- Sherwood P, Given BA, Given CW, Champion VL, Doorenbos AZ, Azzouz F, et al. A cognitive behavioral intervention for symptom management in patients with advanced cancer. Oncology Nursing Forum 2005;32(6):1190-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Sikorskii 2007 {published data only}
- Sikorskii A, Given C, Given B, Jeon S, Decker V, Decker D, et al. Symptom management for cancer patients: a trial comparing two multimodal interventions. Journal of Pain and Symptom Management 2007;34(3):253-64. [DOI] [PMC free article] [PubMed] [Google Scholar]
Thomas 2012 {published data only}
- Thomas M, Elliott J, Rao S, Fahey K, Paul S, Miaskowski C. A randomized, clinical trial of education or motivational-interviewing–based coaching compared to usual care to improve cancer pain management. Oncology Nursing Forum 2012;39(1):39-49. [DOI] [PubMed] [Google Scholar]
Traeger 2015 {published data only}
- Traeger L, Mcdonnell T, McCarty C, Greer J, El-Jawahri A, Temel J. Nursing intervention to enhance outpatient chemotherapy symptom management: patient reported outcomes of a randomized controlled trial. Cancer 2015;1:3905-13. [DOI] [PubMed] [Google Scholar]
Watson 2017 {published data only}
- Watson M, White C, Lynch A, Mohammed K. Telephone‐delivered individual cognitive behavioural therapy for cancer patients: an equivalence randomised trial. Psycho-oncology 2017;26:301-8. [DOI] [PubMed] [Google Scholar]
Yates 2005 {published data only}
- Yates P, Aranda S, Hargraves M, Mirolo B, Clavarino A, McLachlan S, et al. Randomized controlled trial of an educational intervention for managing fatigue in women receiving adjuvant chemotherapy for early-stage breast cancer. Journal of Clinical Oncology 2005;23(25):6027-36. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Aranda 2006 {published data only}
- Aranda S, Schofield P, Weih L, Milne D, Yates P, Faulkner R. Meeting the support and information needs of women with advanced breast cancer: a randomised controlled trial. British Journal of Cancer 2006;95:667-73. [DOI] [PMC free article] [PubMed] [Google Scholar]
Ashing‐Giwa 2008 {published data only}
- Ashing-GiwaK T. Enhancing physical well-being and overall quality of life among underserved Latina-American cervical cancer survivors: feasibility study. Journal of Cancer Survivorship 2008;2:215–23. [DOI] [PubMed] [Google Scholar]
Bakitas 2009 {published data only}
- Bakitas M, Hegel MT, Balan S, Barnett KN, Brokaw FC, Byock IR, et al. The project ENABLE II randomized controlled trial to improve palliative care for rural patients with advanced cancer: baseline findings, methodological challenges and solutions. Palliative & Supportive Care 2009;7:75-86. [DOI] [PMC free article] [PubMed] [Google Scholar]
Beaver 2006 {published data only}
- Beaver K, Twomey M, Whitham G, Foy S, Luke KA. Meeting the information needs of women with breast cancer: piloting a nurse-led intervention. European Journal of Oncology Nursing 2006;10:378–90. [DOI] [PubMed] [Google Scholar]
Beaver 2009 {published data only}
- Beaver K, Tysver-Robinson D, Campbell M, Twomey M, Williamson S, Hindley A, et al. Comparing hospital and telephone follow-up after treatment for breast cancer: randomised equivalence trial. British Medical Journal 2009;338(a3147):1-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Beney 2002 {published data only}
- Beney J, Devine EB, Chow V, Ignoffo RJ, Mitsunaga L, Shahkarami M, et al. Effect of telephone follow-up on the physical well-being dimension of quality of life in patients with cancer. Pharmacotherapy 2002;22(10):1301–11. [DOI] [PubMed] [Google Scholar]
Bohnenkamp 2004 {published data only}
- Bohnekamp SK, McDonald P, Lopez AM, Krupinski E, Blackett A. Traditional versus telenursing outpatient management of patients with cancer with new ostomies. Oncology Nursing Forum 2004;31(5):1005-10. [DOI] [PubMed] [Google Scholar]
Budin 2008 {published data only}
- Budin WC, Hoskins CN, Haber J, Sherman DW, Maislin G, Cater JR, et al. Education, counseling, and adjustment among patients and partners: a randomized clinical trial. Nursing Research 2008;57(3):199-213. [DOI] [PubMed] [Google Scholar]
Campbell 2007 {published data only}
- Campbell LC, Keefe FJ, Scipio C, McKee DC, Edwards CL, Herman SH, et al. Facilitating research participation and improving quality of life for African American prostate cancer survivors and their intimate partners. American Cancer Society 2007;109(2):414-24. [DOI] [PubMed] [Google Scholar]
Cheville 2013 {published data only}
- Cheville A, Kollasch J, Vandenberg J, Shen T, Grothey A, Gamble G, et al. A home-based exercise program to improve function, fatigue, and sleep quality in patients with stage IV lung and colorectal cancer: a randomized controlled trial. Journal of Pain and Symptom Management 2013;45(5):811-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
Coleman 2005 {published data only}
- Coleman EA, Tulman L, Samarel N, Wilmoth MC, Rickel L, Rickel M, et al. The effect of telephone social support and education on adaptation to breast cancer during the year following diagnosis. Oncology Nursing Forum 2005;32(4):822-9. [DOI] [PubMed] [Google Scholar]
Collie 2006 {published data only}
- Collie K, Kreshka MA, Ferrier S, Parsons R, Graddy K, Avram S, et al. Videoconferencing for delivery of breast cancer support groups to women living in rural communities: a pilot study. Psycho-oncology 2007;16:778–82. [DOI] [PubMed] [Google Scholar]
Craddock 1999 {published data only}
- Craddock R, Adams P, Usui W, Mitchell L. An intervention to increase use and effectiveness of self-care measures for breast cancer chemotherapy patients. Cancer Nursing 1999;22(4):312-9. [DOI] [PubMed] [Google Scholar]
Dalton 2004 {published data only}
- Dalton JA, Keefe FJ, Carlson J, Youngblood R . Tailoring cognitive-behavioral treatment for cancer pain. Pain Management Nursing 2004;5(1):3-18. [DOI] [PubMed] [Google Scholar]
De Wit 1997 {published data only}
- De Wit R, Van Dam F, Zandbelt L, Van Buuren A, Van Der Heijden K, Leenhouts G, et al. A pain education program for chronic cancer pain patients: follow-up results from a randomized controlled trial. Pain 1997;73:55-69. [DOI] [PubMed] [Google Scholar]
DuHamel 2015 {published data only}
- Duhamel K, Schuler T, Nelson C, Philip E, Temple L, Schover L, et al. The sexual health of female rectal and anal cancer survivors: results of a pilot randomised psycho-educational intervention trial. Journal of Cancer Survivorship 2016;10(3):553-63. [DOI] [PMC free article] [PubMed] [Google Scholar]
Given 2008 {published data only}
- Given CW, Sikorski A, Tamkus D, Given B, You M, McCorkle R, et al. Managing symptoms among patients with breast cancer during chemotherapy: results of a two-arm behavioral trial. Journal of Clinical Oncology 2008;26(36):5855-62. [DOI] [PMC free article] [PubMed] [Google Scholar]
Haddad 2003 {published data only}
- Haddad P, Wilson P, Wong R, Williams D, Sharma N, Soban F, et al. The success of data collection in the palliative setting - telephone or clinic follow-up? Supportive Care in Cancer 2003;11:555-9. [DOI] [PubMed] [Google Scholar]
Hagiopan 1990 {published data only}
- Hagopian GA, Rubenstein JH. Effects of telephone call interventions on patients' well-being in a radiation therapy department. Cancer Nursing 1990;13(6):339-44. [PubMed] [Google Scholar]
Halbert 2004 {published data only}
- Halbert CH, Wenzel L, Lerman C, Peshkin BN, Narod S, Marcus A, et al. Predictors of participation in psychosocial telephone counseling following genetic testing for BRCA1and BRCA2 mutations. Cancer Epidemiology, Biomarkers & Prevention 2004;13(5):875-81. [PubMed] [Google Scholar]
Hanks 2002 {published data only}
- Hanks GW, Robbins M, Sharp D, Forbes K, Done K, Peters TJ, et al. The imPaCT study: a randomised controlled trial to evaluate a hospital palliative care team. British Journal of Cancer 2002;87:733–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Harrison 2011 {published data only}
- Harrison JD, Young JM, Soloman MJ, Butow PN, Secomb R, Masya L. Randomized pilot evaluation of the supportive care intervention “CONNECT” for people following surgery for colorectal cancer. Diseases of the Colon & Rectum 2011;54(5):622-31. [DOI] [PubMed] [Google Scholar]
Hawkes 2009 {published data only}
- Hawkes AL, Gollschewski S, Lynch BM, Chambers S. A telephone-delivered lifestyle intervention for colorectal cancer survivors ‘CanChange’: a pilot study. Psycho-oncology 2009;18:449–55. [DOI] [PubMed] [Google Scholar]
Hawkins 2010 {published data only}
- Hawkins RP, Pingree S, Shawa B, Serlin RC, Swoboda C, Han JY, et al. Mediating processes of two communication interventions for breast cancer patients. Patient Education and Counseling 2010;81s:S48-53. [DOI] [PMC free article] [PubMed] [Google Scholar]
Hayes 2012 {published data only}
- Hayes SC, Rye S, Disipio T, Yates P, Bashford J, Pyke C, et al. Exercise for health: a randomized, controlled trial evaluating the impact of a pragmatic, translational exercise intervention on the quality of life, function and treatment related side-effects following breast cancer. Breast Cancer Research and Treatment 2013;137:175-86. [DOI] [PubMed] [Google Scholar]
Kearney 2009 {published data only}
- Kearney N, McCann L, Norrie J, Taylor L, Gray P, McGee-Lennon M, et al. Evaluation of a mobile phone-based, advanced symptom management system (ASyMS©) in the management of chemotherapy-related toxicity. Supportive Care in Cancer 2009;17:437-44. [DOI] [PubMed] [Google Scholar]
Kimman 2010 {published data only}
- Kimman ML, Dirksen CD, Voogd AC, Falger P, Gijsen BCM, Thuring MT, et al. Nurse-led telephone follow-up and an educational group programme after breast cancer treatment: results of a 2x2 randomised controlled trial. European Journal of Cancer 2011;47:1027-36. [DOI] [PubMed] [Google Scholar]
Koller 2013 {published data only}
- Koller A, Miaskowski C, De Geest S, Opitz O, Spichiger E. Results of a randomized controlled pilot study of a self-management intervention for cancer pain. European Journal of Oncology Nursing 2013;17:284-91. [DOI] [PubMed] [Google Scholar]
Kornblinth 2006 {published data only}
- Kornblith AB, Dowell JM, Herndon JE, Engelman BJ, Bauer-Wu S, Small EJ, et al. Telephone monitoring of distress in patients aged 65 years or older with advanced stage cancer. A cancer and leukemia group B study. Cancer 2006;107:2706–14. [DOI] [PubMed] [Google Scholar]
Marcus 2010 {published data only}
- Marcus AC, Garrett KM, Cella D, Wenzel L, Brady MJ, Fairclough D, et al. Can telephone counseling post-treatment improve psychosocial outcomes among early stage breast cancer survivors? Psycho-oncology 2010;19:923–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
McCorkle 2009 {published data only}
- McCorkle R, Dowd M, Ercolano E, Schulman-Green D, Williams AI, Siefert ML, et al. Effects of a nursing intervention on quality of life outcomes in post-surgical women with gynecological cancers. Psycho-oncology 2009;18:62–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
Meneses 2009 {published data only}
- Meneses K, Meneses P, Azuero A, Loerzei VW, Su X, Hassey LA. Preliminary evaluation of psycho-educational support interventions on quality of life in rural breast cancer survivors post-primary treatment. Cancer Nursing 2009;32(5):385–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
Nguyen 2018 {published data only}
- Nguyen L, Alexander K, Yates P. Psychoeducational Intervention for Symptom Management of Fatigue, Pain, and Sleep Disturbance Cluster Among Cancer Patients: A Pilot Quasi-Experimental Study. Journal of Pain and Symptom Management 2018;55(6):1459-72. [DOI] [PubMed] [Google Scholar]
Park 2012 {published data only}
- Park JH, Bae SH, Jung YS, KIM KS. Quality of life and symptom experience in breast cancer survivors after participating in a psychoeducational support program. A pilot study. Cancer Nursing 2012;35(1):E34-41. [DOI] [PubMed] [Google Scholar]
Rustoen 2014 {published data only}
- Rustoen T, Valeberg B, Kolstad E, WIist E, Paul S, Miaskowski C. A randomized clinical trial of the efficacy of a self-care intervention to improve cancer pain management. Cancer Nursing 2014;37(1):34-43. [DOI] [PubMed] [Google Scholar]
Salonen 2009 {published data only}
- Salonen P, Tarkka MT, Kellokumpu-Lehtinen PL, Aastedt-Kurki P, Luukkaala T, Kaunonen M. Telephone intervention and quality of life in patients with breast cancer. Cancer Nursing 2009;32(3):177-90. [DOI] [PubMed] [Google Scholar]
Sandgren 2000 {published data only}
- Sandgren AK, McCaul KD, King B, O'Donnell S, Foreman G. Telephone therapy for patients with breast cancer. Oncology Nursing Forum 2000;27(4):683-8. [PubMed] [Google Scholar]
Sandgren 2003 {published data only}
- Sandgren AK, McCaul KD. Short-term effects of telephone therapy for breast cancer patient. Health Psychology 2003;22(3):310–15. [DOI] [PubMed] [Google Scholar]
Sandgren 2007 {published data only}
- Sandgren AK, McCaul KD. Long-term telephone therapy outcomes for breast cancer patients. Psycho-oncology 2007;16:38-47. [DOI] [PubMed] [Google Scholar]
Santacroce 2010 {published data only}
- Santacroce SJ, Asmus K, Kadan-Lottick N, Grey M. Feasibility and preliminary outcomes from a pilot study of coping skills training for adolescent-young adult survivors of childhood cancer and their parents. Journal of Pediatric Oncology Nursing 2010;27(1):10-20. [DOI] [PubMed] [Google Scholar]
Sikorskii 2009 {published data only}
- Sikorskii A, Given CW, Given B, Jeon S, You M. Differential symptom reporting by mode of administration of the assessment: automated voice response system versus a live telephone interview. Medical Care 2009;47(8):866–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
Sikorskii 2009b {published data only}
- Sikorskii A, Given CW, You M, Jeon S, Given B. Response analysis for multiple symptoms revealed differences between arms of a symptom management trial. Journal of Clinical Epidemiology 2009;62(7):716–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
Spoelstra 2013 {published data only}
- Spoelstra S, Given B, Given C, Grant M, Sikorskii A, You M, et al. An intervention to improve adherence and management of symptoms for patients prescribed oral chemotherapy agents. Cancer Nursing 2013;36(1):18-28. [DOI] [PubMed] [Google Scholar]
Williams 2004 {published data only}
- Williams SA, Schreier AM. The effect of education in managing side effects in women receiving chemotherapy for treatment of breast cancer. Oncology Nursing Forum 2004;31(1):E16-23. [DOI] [PubMed] [Google Scholar]
Winger 2018 {published data only}
- Winger J, Rand K, Hanna N, Jalal S, Einhorn L, Birdas T, et al. Coping skills practice and symptom change: a secondary analysis of a pilot telephone symptom management intervention for lung cancer patients and their family caregivers. Journal of Pain and Symptom Management 2018;55:1341-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Additional references
Abrams 2005
- Abrams K, Gillies C, Lambert P. Meta-analysis of heterogeneously reported trials assessing change from baseline. Statistics in Medicine 2005;24:3823-44. [DOI] [PubMed] [Google Scholar]
Ahlberg 2003
- Ahlberg K, Ekman T, Gaston-Johansson F, Mock V. Assessment and management of cancer-related fatigue in adults. Lancet 2003;362:640-50. [DOI] [PubMed] [Google Scholar]
Barsevick 2002
- Barsevick A, Sweeney C, Haney E, Chung E. A systematic qualitative analysis of psychoeducational interventions for depression in patients with cancer. Oncology Nursing Forum 2002;1:73-84. [DOI] [PubMed] [Google Scholar]
Chen 2018
- Chen Y, Bing-Sheng G, Li Z, Li X. Effect of telehealth intervention on breast cancer patients’ quality of life and psychological outcomes: a meta-analysis. Journal of Telemedicine and Telecare 2018;24(3):157-67. [DOI] [PubMed] [Google Scholar]
Cheville 2010
- Cheville A, Novotny P, Sloan J, Basford J, Wampfler J, Garces Y, et al. The value of a symptom cluster of fatigue, dyspnea, and cough in predicting clinical outcomes in lung cancer survivors. Journal of Pain and Symptom Management 2011;42(2):213-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
Cleeland 2009
- Cleeland C. Symptom burden: multiple symptoms and their impact as patient-reported outcomes. Journal of the National Cancer Institute Monographs 2009;37:16-21. [DOI] [PubMed] [Google Scholar]
Cox 2003
- Cox K, Wilson E. Follow-up for people with cancer: nurse-led services and telephone interventions. Journal of Advanced Nursing 2003;43:51-61. [DOI] [PubMed] [Google Scholar]
DerSimonian 1986
- DerSimonian R, Laird N. Meta-analysis in clinical trials. Controlled Clinical Trials 1986;7:177-88. [DOI] [PubMed] [Google Scholar]
Dickinson 2014
- Dickinson R, Hall S, Sinclair JE, Bond C, Murchie P. Using technology to deliver cancer follow-up: a systematic review. BMC Cancer 2014;14(3):311. [DOI] [PMC free article] [PubMed] [Google Scholar]
Dieng 2016
- Dieng M, Butow P, Costa D, Morton R, Menzies S, Mireskandari S. Psychoeducational intervention to reduce fear of cancer recurrence in people at high risk of developing another primary melanoma: results of a randomized controlled trial. Journal of Clinical Oncology 2016;34:4405-14. [DOI] [PubMed] [Google Scholar]
Dodd 2000
- Dodd M, Miaskowski C. The PRO-SELF program: a self-care intervention program for patients receiving cancer treatment. Seminars in Oncology Nursing 2000;16:300-8. [DOI] [PubMed] [Google Scholar]
Dodd 2001
- Dodd M, Miaskowski C, Paul S. Symptom clusters and their effect on the functional status of patients with cancer. Oncology Nursing Forum 2001;28:465-70. [PubMed] [Google Scholar]
Follmann 1992
- Follmann D, Elliott P, Suh I, Cutler J. Variance imputation for overviews of clinical trials with continuous response. Journal of Clinical Epidemiology 1992;45:769-73. [DOI] [PubMed] [Google Scholar]
Freeman 2015
- Freeman L, White R, Ratcliff C, Sutton S, Stewart M, Palmer J, et al. A randomized trial comparing live and telemedicine deliveries of an imagery-based behavioral intervention for breast cancer survivors: reducing symptoms and barriers to care. Psycho-oncology 2015;24(8):910-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Galway 2012
- Galway K, Black A, Cantwell M, Cardwell CR, Mills M, Donnelly M. Psychosocial interventions to improve quality of life and emotional wellbeing for recently diagnosed cancer patients. Cochrane Database of Systematic Reviews 2012;11(11):CD007064. [DOI] [PMC free article] [PubMed] [Google Scholar]
Gil 2006
- Gil K, Mishel M, Belyea M, Germino B, Porter L, Clayton M. Benefits of the uncertainty management intervention for African American and White older breast cancer survivors: 20-month outcomes. International Journal of Behavioural Medicine 2006;13(4):286-94. [DOI] [PubMed] [Google Scholar]
Glover 1995
- Glover J, Miaskowski C, Dibble S, Dodd M. Mood states of oncology outpatients: does pain make a difference? Journal of Pain and Symptom Management 1995;10:120-8. [DOI] [PubMed] [Google Scholar]
Gotay 1998
- Gotay C, Bottomley A. Providing psycho-social support by telephone: what is the potential for cancer patients? European Journal of Cancer Care 1998;7:225-31. [DOI] [PubMed] [Google Scholar]
Harrington 2010
- Harrington C, Hansen J, Moskowitz M, Todd B, Feuerstein M. It's not over when it's over: long-term symptoms in cancer survivors - a systematic review. International Journal of Psychiatry in Medicine 2010;40(2):163-81. [DOI] [PubMed] [Google Scholar]
Higgins 2003
- Higgins J, Thompson S, Deeks J, Altan D. Measuring inconsistency in meta-analyses. British Medical Journal 2003;327:557-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
Kim 2004
- Kim J, Dodd M, West C, Paul S, Facione N, Schumacher K, et al. The PRO-SELF pain control program improves patients' knowledge of cancer pain management. Oncology Nursing Forum 2004;31:1137-43. [DOI] [PubMed] [Google Scholar]
Kim 2009
- Kim J, Dodd M, Aouizerat B, Jahan T, Miaskowski C. A review of the prevalence and impact of multiple symptoms in oncology patients. Journal of Pain and Symptom Management 2009;37(4):715-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
Kim 2012
- Kim H, Barsevick A, Fang C, Miaskowski C. Common biological pathways underlying the psychoneurological symptom cluster in cancer patients. Cancer Nursing 2012;35(6):E1-20. [DOI] [PubMed] [Google Scholar]
Laugsand 2011
- Laugsand EA, Jakobsen G, Kaasa S, Klepstad P. Inadequate symptom control in advanced cancer patients across Europe. Support Care in Cancer 2011;19(12):2005-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
Lins 2014
- Lins S, Hayder-Beichel D, Rucker G, Motschall E, Antes G, Meyer G, et al. Efficacy and experiences of telephone counselling for informal carers of people with dementia. Cochrane Database of Systematic Reviews 2014, Issue 9. [DOI: 10.1002/14651858.CD009126.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
McPherson 2014
- McPherson C, Hadjistavropoulos T, Devereaux A, Lobchuk M. A qualitative investigation of the roles and perspectives of older patients with advanced cancer and their family caregivers in managing pain in the home. BMC Palliative Care 2014;13(1):1-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
Reese 2002
- Reese R, Conoley C, Brossart D. Effects of telephone counselling: a field-based investigation. Journal of Counselling Psychology 2002;49:233-42. [Google Scholar]
RevMan 2014 [Computer program]
- The Nordic Cochrane Centre, The Cochrane Collaboration, Copenhagen Review Manager (RevMan). Version 5.3. The Nordic Cochrane Centre, The Cochrane Collaboration, Copenhagen, 2014.
Scura 2004
- Scura K, Budin W, Garging E. Telephone social support and education for adaptation to prostate cancer: a pilot study. Oncology Nursing Forum 2004;31:335-8. [DOI] [PubMed] [Google Scholar]
Steel 2016
- Steel J, Geller D, Kim K, Butterfield L, Spring M, Grady J, et al. Web-based collaborative care intervention to manage cancer-related symptoms in the palliative care setting. Cancer 2016;8:1270-82. [DOI] [PMC free article] [PubMed] [Google Scholar]
Van Lancker 2014
- Van Lancker A, Velghe A, Van Hecke A, Verbrugghe M, Van Den Noortgate N, Grypdonck M, et al. Prevalence of symptoms in older cancer patients receiving palliative care: a systematic review and meta-analysis. Journal of Pain and Symptom Management 2014;47(1):90-104. [DOI] [PubMed] [Google Scholar]
Walsh 2000
- Walsh D, Donnelly S, Rybicki L. The symptoms of advanced cancer: relationship to age, gender, and performance status in 1,000 patients. Supportive Care in Cancer 2000;8(3):175-9. [DOI] [PubMed] [Google Scholar]
White 2011
- White I, Allan H, Faithfull S. Assessment of treatment-induced female sexual morbidity in oncology: is this a part of routine medical follow-up after radical pelvic radiotherapy? British Journal of Cancer 2011;105(7):903-10. [DOI] [PMC free article] [PubMed] [Google Scholar]


