Abstract
Recently, an increasing number of novel drugs were approved in oncology and hematology. Nevertheless, pharmacology progress comes with a variety of side effects, of which cytokine release syndrome (CRS) is a potential complication of some immunotherapies that can lead to multiorgan failure if not diagnosed and treated accordingly. CRS generally occurs with therapies that lead to highly activated T cells, like chimeric antigen receptor T cells or in the case of bispecific T-cell engaging antibodies. This, in turn, leads to a proinflammatory state with subsequent organ damage. To better manage CRS there is a need for specific therapies or to repurpose strategies that are already known to be useful in similar situations. Current management strategies for CRS are represented by anticytokine directed therapies and corticosteroids. Based on its pathophysiology and the resemblance of CRS to sepsis and septic shock, as well as based on the principles of initiation of continuous renal replacement therapy (CRRT) in sepsis, we propose the rationale of using CRRT therapy as an adjunct treatment in CRS where all the other approaches have failed in controlling the clinically significant manifestations.
Keywords: immunotherapy
Rationale for the hypothesis
Current statistics report an increase in the incidence of hematological and oncological malignancies,1 but treatment with immune-based therapies have also risen in both use and efficacy. These are nevertheless accompanied by a variety of side effects, one of which is tightly linked to the mechanism implicated in chimeric antigen receptor T cells (CAR-T) cell therapy2 and treatment with T cell engaging bispecific antibodies3: cytokine release syndrome (CRS). CRS, also known as cytokine-associated toxicity, is a systemic inflammatory response syndrome following massive cytokine release into the bloodstream.4 This can be triggered by the administration of immune-based therapies such as CAR-T cell-based therapy for B-cell malignancies, as well as other monoclonal antibody-based drugs.5 6 Fitzgerald et al have reported that 92% of acute lymphoblastic leukemia (ALL) patients treated with an anti-CD19 CAR T cell therapy revealed that 18 (46%) developed acute kidney injury (AKI)-related with grade 3–4 CRS. Of those 18 patients, 13 were noted to have a clinical improvement following tocilizumab administration. Thus, five of them possibly having an option of subsequent therapies for CRS.7 This represents one of the most serious adverse events of these therapies and it can become a life-threatening complication, leading to multiorgan failure. A condition similar to CRS from a pathophysiological and clinical perspective is represented by sepsis. Because of this, we hypothesize that CRS might benefit from therapeutical strategies that are already in use in sepsis, like continuous renal replacement therapy (CRRT) which, in the current review, might be a therapeutic option for refractory CRS.8
Pathophysiology of CRS
As an overview, CRS emerges when interferon gamma (IFN-γ) secreted by activated T cells polarize macrophages to an M1 phenotype, leading them to secrete several proinflammatory cytokines, of which a significant portion of the literature implicates interleukin-6 (IL-6) as a central mediator of toxicity. IL-6 is a cytokine, produced by a myriad of cells and tissues represented, but not limited by macrophages, T cells and hepatic tissue9 10
Two main inferences can be drawn from these affirmations. First, IFN-γ activation of macrophages is similar to the effect induced by lipopolysaccharide on macrophages in the case of sepsis, thus leading to a logical possible link between these two conditions. Second, the main events implicated in CRS are represented by the activation of T cells and M1 polarization of macrophages and thus cytokines upregulated or downregulated by these cells can represent important pathophysiologic events. For T cells, it is important to mention the central role of IL-2 autocrine signaling, which leads to a positive regulatory loop, leading to more subsequent T-cell activation. Moreover, an important cytokine secreted by activated T cells is IFN-γ, which acts on macrophages leading to an M1 polarized phenotype, characterized by the upregulation of IL-6 and tumor necrosis factor α (TNFα).11
IL-6 signaling occurs through two different mechanisms. The first requires binding to cell-associated gp130 (CD130), which is broadly expressed, and the membrane-bound IL-6 receptor (IL-6R) (CD126).10 IL-6R is cell associated on macrophages, neutrophils, hepatocytes and some T cells and mediates classic IL-6 signaling, which predominates when IL-6 levels are low. However, when IL-6 levels are elevated, soluble IL-6R (sIL-6R; trans-IL-6 signaling) can also initiate trans-signaling, which occurs on a much wider array of cells, resulting in activation of the JAK/STAT pathway. Current models report that the anti-inflammatory properties of IL-6 are mediated via classic signaling, whereas proinflammatory responses occur as a result of trans-IL-6 signaling via the soluble receptor.12 High levels of IL-6, present in the context of CRS, most likely initiate a proinflammatory IL-6-mediated signaling cascade. IL-6 bound to the sIL-6R can associate with membrane-bound gp130, resulting in activation of the JAK/STAT pathway. Since gp130 is broadly expressed across many effector cells, high IL-6 levels result in a more robust immune activation13 (figure 1).
Figure 1.
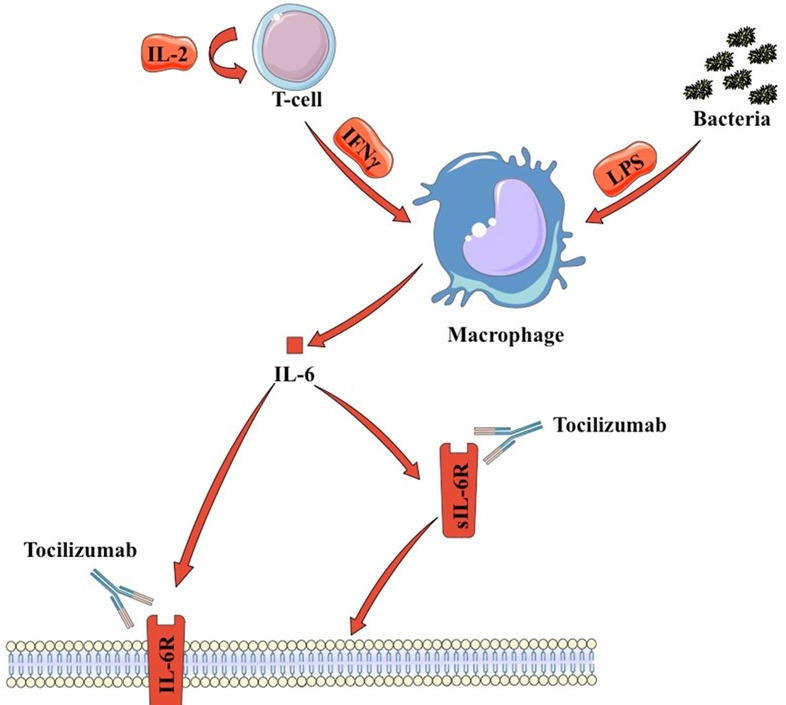
IL-6 secretion in CRS and sepsis. CRS, cytokine release syndrome; IFN-γ, interferon-γ; IL-6, interleukin-6; LPS, lipopolysaccharide; sIL-6R, soluble IL-6 receptor.
Furthermore, the cytokines which are elevated in the serum of patients experiencing CRS are listed in table 1.
Table 1.
Cytokines elevated in CRS
| Cytokine | Molecular weight (kDa) | |
| Interleukin-6 (IL-6) | 26 | Proinflammatory |
| IL-10 | 18 | Anti-inflammatory |
| Interferon-gamma | 35 | Proinflammatory |
| Tumor necrosis factor-α monomer | 17 | Proinflammatory |
| IL-1β | 17 | Proinflammatory |
| IL-2 | 17 | Proinflammatory |
| IL-2 receptor | Proinflammatory | |
| IL-8, MCP-1, and MIP-1b | IL-8–8 kDa | Reported in patients treated with CART-19 and blinatumomab |
CART, chimeric antigen receptor T cells; CRS, cytokine release syndrome.
Clinical presentation of CRS
In the clinical setting, CRS presents itself with a variety of symptoms ranging from mild, flu-like symptoms, to severe life-threatening complications with onset within minutes to hours, depending on the inducing agent and the immune system activation.14 Mild symptoms, non-specific of CRS, include fever, fatigue, headache, rash, arthralgia and myalgia. Severe symptoms are characterized by high fever exceeding 40°C with multiorgan system failure and disseminated intravascular coagulopathy.15 The main organ systems involved are respiratory (ranging from symptoms such as tachypnea, dyspnea, cough to acute respiratory distress syndrome with severe impairment of ventilation and oxygenation requiring sedation, intubation of the airway and mechanical ventilation); cardiac (cardiac failure with pulmonary edema); renal (AKI/failure); hepatic failure and neurological (confusion, aphasia, hemiparesis, cranial nerve palsies, seizures and coma).16
Neurotoxicity is the second most common adverse event after CAR-T cell therapy being named ‘CAR T cell-related encephalopathy syndrome’ (CRES). CRES does not seem to be directly related to CRS since is currently considered an independent event, as the two complications have been described as being separate time events in some studies.17 18 Common laboratory abnormalities in patients with CRS include cytopenias, elevated creatinine and liver enzymes, deranged coagulation parameters and a high CRP.6
In rare cases, CRS can overlap with clinical signs and laboratory abnormalities that resemble hemophagocytic lymphohistiocytosis (HLH)/macrophage activation syndrome (MAS). In these cases, the main cytokines observed to play an important role are represented by IL-6, IFN-γ and IL-10, which are in accordance with classic CRS. Patients with CRS-associated HLH display the typical clinical and laboratory findings of HLH/MAS such as high fevers, splenomegaly, highly elevated ferritin levels, coagulopathy and hypertriglyceridemia.19
After treatment with blinatumomab in B-cell ALL, almost 25% of CRS patients had a diagnosis of HLH.20 21
Moreover, because of the overlap in clinical symptoms between CRS and sepsis and because the population of patients receiving CAR-T cell therapy or T-cell engaging bispecific antibodies is at higher risk of both CRS and sepsis, a thorough differential diagnosis has to be made, with active sepsis screening (table 2).
Table 2.
Differential diagnosis of CRS6
| Familial HLH | Secondary HLH/MAS | CRS related to HLH or MAS | Sepsis | |
| Genetics | Homozygous mutation | Heterozygous mutations in case-reports | Not reported | Not reported |
| Biomarkers | ||||
| IL-10 | Very high | Very high | High | High |
| IFN-alpha | Very high | Very high | Very high | Normal |
| IL-6 | High | High | Very high | Very high |
| Ferritin | Very high | Very high | Very high | High |
| CD 163 | Very high | Very high | High | Not reported |
CRS, cytokine release syndrome; HLH, hemophagocytic lymphohistiocytosis; IFN, interferon; IL-6, interleukin-6; MAS, macrophage activation syndrome.
Current management of CRS
It is currently indicated that patients that develop CRS should be monitored in the intensive care unit (ICU) or high-dependency unit, especially those with grade 3 or 4. The therapeutical agents used in this situation are represented by tocilizumab, either as monotherapy or in combination with corticosteroids. These approaches can be extended to using multiple tocilizumab doses or high-dose corticosteroids. Additionally, although more rarely used, siltuximab, an anti-IL-6 antibody, is part of CRS management protocols in some hospitals.16 22
The most commonly used anti-IL-6 antibody is tocilizumab, a humanized antibody that blocks IL-6 signaling by acting on both IL-6R and sIL-6R, thus having an effect especially in the case of high IL-6 levels.23 Clinically, it was shown to be efficacious as its administration was associated with reversal of CRS in patients treated with either anti-CD19 CAR-T cells or with blinatumomab.3 Currently, tocilizumab represents the standard of care in managing CRS both in clinical practice and clinical trials. This can be administered as a single dose of 8 mg/kg with a maximum of 800 mg infused over an hour. If the symptoms do not ameliorate over the next 24 hours, another dose of tocilizumab, the use of corticosteroids or a combination of those is used.16 Additionally, another anti-IL-6 antibody that can be used in CRS management is siltuximab, which acts directly on IL-6.24 Tocilizumab might increase the risk of infection, thus the subsequent need for differential diagnosis between refractory CRS and sepsis being necessary.25
As mentioned before, corticosteroids can be used if tocilizumab use did not result in any amelioration, either as monotherapy or in combination with tocilizumab. These therapeutic approaches have been studied both in CRS as well as in other inflammatory conditions in which T-cells and/or macrophages are known to be activated. The treatment has good efficacy and is included both in standard-of-care clinical practice as well as clinical trial protocols. Because of the T-cell inhibitory effects of this class of drugs, it is generally considered that corticosteroids would diminish the efficacy of CAR-T cell therapy and, although this has now been shown to be the case some physicians would use corticosteroids only if necessary in this situation. Furthermore, various clinical indications for corticosteroids should be reserved for neurological toxicities and CRS refractory to tocilizumab.26
Aside from the above-mentioned approaches, clinical use includes targeting IL-2R, IL-1R and TNFα for managing CRS. Moreover, inhibitors of MCP-1 and MIP1B are being developed even if not yet used in clinical trials for CRS. As presented, the strategies used for managing CRS generally target either T-cells, macrophages or their communication. Of the mentioned molecules, most target macrophage polarization, except for IL-2R inhibitors, which inhibit T-cell autocrine signaling and their subsequent activation with IFNγ secretion and further M1 polarization of macrophages and secretion of proinflammatory molecules.27
The corresponding clinical picture is graded according to CRS consensus grading (table 3),28 with its associated treatments.9 Still, CRRT is not mentioned as a treatment method in the grading classification, even if according to our hypothesis, patients presenting grades 3 and 4 may benefit from CRRT.
Table 3.
2018 CRS consensus grading by Lee et al69
| Grade 1 | Fever* ≥38°C—symptomatic management—intravenous fluids, analgesics, antiemetics, antipyretics possibly antibiotics and a lot of careful watching from the nurses and caregivers and the team. |
| Grade 2 | Fever* ≥38°C, grade 2 creatinine elevation, grade 3 transaminitis, neutropenic fevers and other indications for hospitalization, with hypotension not requiring vasopressors and/or hypoxia requiring low-flow nasal cannula or blow-by oxygen. |
| Grade 3 | Fever* ≥38°C, grade 3 creatinine elevation, grade 4 transaminitis, with hypotension requiring one vasopressor with or without vasopressin and/or hypoxia requiring high-flow nasal cannula, facemask, non-rebreather mask, or Venturi mask not attributable to any other cause. |
| Grade 4 | Fever* ≥38°C, as life-threatening symptoms requiring ventilator support or grade 4 organ toxicity, with hypotension requiring multiple vasopressors (excluding vasopressin) and/or hypoxia requiring positive pressure (eg, CPAP, BiPAP, intubation and mechanical ventilation) not attributable to any other cause. |
| Grade 5 | Death |
*Fever is defined as temperature ≥38°C. In patients who have CRS then receive tocilizumab or steroids, fever is no longer required to grade subsequent CRS severity. In this case, CRS grading is driven by hypotension and/or hypoxia.
CRS, cytokine release syndrome; CPAP, continuous positive airway pressure; BiPAP, bilevel positive airway pressure.
Extracorporeal blood purification therapies
The scope of extracorporeal blood purification therapies has expanded from simple RRT to the treatment of multiple organ dysfunction syndrome management with the intent of utilization over 24 hours.28
Extracorporeal blood purification therapies, may it be filtration, dialysis (diffusion) and adsorption, have been used as a strategy to remove medium molecular weight circulating inflammatory mediators like cytokines (<40 kDa), chemokines and complement during sepsis with AKI and septic shock with multiple organ failure, leading to an improvement of overall morbidity and mortality in this condition.8 By eliminating inflammatory mediators, such as IL-1, IL-6, TNFα from the bloodstream it may limit their cytotoxic activity, improving the migration of leukocytes and possibly modifying the immune cell phenotype, as such playing an immunomodulatory role.29 This hypothesis is supported by the fact that a low level of proinflammatory cytokines has been shown to be associated with low CRS mortality.30
As CRRT leads to the elimination of proinflammatory cytokines from the blood like IL-6, it thus might be used in CRS cases refractory to tocilizumab and corticosteroids.
Working as an artificial kidney, CRRT is a process of slow, isotonic removal of water and solute, providing continuous fluid removal and blood purification by using a semipermeable membrane (filter) with counter-current dialysate fluid to remove fluid and particles via diffusion, convection, as well as adsorption.31 The efficiency of diffusive removal decreases with increasing molecular weight of the solute. Convective clearance is proportional to the ultrafiltration rate and independent of molecular weight up to the cut-off point of the membrane, which is 30–40 kDa for the currently used open hemofiltration membranes.32 In addition to being used as a cytokine removal tool, RRT was used for the correction of electrolyte (hyperkalemia), acid-base status correction (severe metabolic acidosis), promoting recovery of renal function (correction of uremia) and to help correct the overload syndrome from fluid administration. By favoring the clearance of inflammatory mediators and myocardial depression factors, it demonstrates circulatory stability, reduction of the dosage of vasoactive drugs, modulating immune function and overall temperature control.33
The patient’s blood is being continuously recirculated through the extracorporeal circuit at blood flow rates of 150–300 mL/min and, as such, it requires systemic anticoagulation to prevent clotting in the membrane filter. The available options include regional citrate anticoagulation and unfractionated heparin (UFH).34 The regional citrate anticoagulation chelates calcium in the extracorporeal circuit, but it cannot be used in patients with hepatic dysfunction/failure because of impaired metabolic clearance of citrate.35 For UFH, activated partial thromboplastin time 1.5–2 times its normal values or activated clotting time over 220–250 can be used to achieve the target anticoagulation.36 The problem that might be encountered is the thrombocytopenia usually seen in hematological patients, and because of this, the dose of heparin should be reduced.
The use of ultrafiltration greater than that used in supporting renal function, that is, > (35 mL/kg/hour standard-volume hemofiltration (SVHF)) is called high-volume hemofiltration (HVHF). Large and very large ultrafiltration flows (>45 mL/kg/hour) were used to increase the elimination of medium-molecular-mass molecules, for example, cytokines.37 38 In the IVOIRE trial, Joannes-Boyau et al reported no evidence that HVHF at 70 mL/kg/hour when compared with contemporary SVHF at 35 mL/kg/hour, leads to a reduction of 28-day mortality or contributes to early improvements in hemodynamic profile or organ function.39 Thus, this trial does not support the use of HVHF in septic patients complicated by AKI. Furthermore, concerns about the removal of beneficial molecules such as nutrients, proteins, and antimicrobial peptides were associated with HVHF.40 The big question that arises is if the SVHF is enough for the management of CRS?
A case report by Liu et al presented a 10-year-old boy with relapsed/refractory (R/R) B-ALL who received CD19 CAR-T-cell therapy and who developed severe CRS, in whom the treatment with tocilizumab and glucocorticoids were ineffective, hemofiltration was initiated, with IL-6 levels rapidly decreasing and with successfully treating CRS. It must be mentioned that the case report presented no discernable kidney injury, the authors arguing that hemofiltration was used in the context of severe CRS and bearing in mind that tocilizumab might increase the risk of exacerbating infection if a repeated dose was to be administered. With no AKI present, but persistent high fever (hyperpyrexia >40°C), liver dysfunction, beginning of sepsis and clinical and laboratory abnormalities of the MAS all of which resolved after CRRT initiation. In this case, veno-venous hemofiltration was used over 24 hours with the patient maintaining normal systolic and diastolic blood pressures. After the mentioned process, 1000 mL of plasma were administered to maintain the colloid osmotic pressure.41
Plasma exchange involves the removal of the patient’s plasma, which is replaced by infusion fluids such as plasma or albumin. Cytokine removal through plasma exchange may reduce the level of inflammatory markers in sepsis and organ failure.42 Xiao et al reported a case involving a patient with R/R ALL who developed life-threatening CRS after receiving an infusion of anti-CD19 CAR-T cells. In combination with glucocorticoid therapy, therapeutic plasma exchange was performed daily for three consecutive days. Throughout the treatment, the patient’s inflammatory factors nearly returned to baseline levels, and his CRS-related symptoms were relieved with the patient eventually being discharged in good health.43
Further research focused on using biocompatible adsorption hemofilter, also known as hemadsorption/hemoperfusion, involving materials with high adsorptive properties and which remove cytokines based on their physical properties. Blood circulates in direct contact with the adsorptive surface, which attracts the molecules through hydrophobic, ionic and van der Waals interactions.44
Currently, available adsorption hemofilter are: polymyxin B endotoxin-adsorbing column (PMX-DHP), marketed as Toraymyxin (Toray Industries, Tokyo, Japan), a polycationic antibiotic column containing multiple polymyxin B-immobilized fibers, which has been shown to neutralize bacterial endotoxins; polymethyl-methacrylate membranes, AN69 surface-treated (AN69ST) membranes, and modified AN69ST membranes coated with immobilized heparin in order to reduce the risk of thrombogenic events (oXiris)and Cytosorb.45
Cytosorb (CytoSorbents Corporation, Monmouth Junction, New Jersey, USA) is a biocompatible absorber that is integrated into the extracorporeal blood pump circuit. The therapeutic goal is the reduction of cytokines and other inflammatory mediators from overwhelming levels to a physiological range. It is a porous polymer sorbent technology with extensive elimination capacity due to a total surface of 40 000–45 000 m2. It adsorbs hydrophobic low and middle molecular substances between 5 and 60 kDa in size referred to as the ‘cytokine sweet spot’, in a concentration-dependent manner. It targets many of the key inflammatory mediators such as IL-1β, IL-6, TNF-α and IL-10.46 When in high concentration substances are removed quickly. A single cartridge can be used for up to 24 hours at a time, after which it needs replacement with a new cartridge as long as the therapy continues.47 Reduction of IL-6 levels has been reported, consistent with the findings of Kellum et al examining the effect of CS on IL-6/other cytokines in brain-dead potential donors.48 Two randomized controlled trials (RCTs) have also shown a reduction of IL-6 levels.49 50
In a rat sepsis model, Maeda et al have used continuous hemofiltration with polyester polymer alloy as a cytokine adsorber to remove IL-6 from the bloodstream efficiently.51 Furthermore, a recently published in vitro study compared the adsorptive capacities of Toraymyxin, oXiris and CytoSorb. The comparison focused on the removal of sepsis-associated mediators and endotoxins. Removal rates of 27 proinflammatory and anti-inflammatory mediators (including IL-6, TNF-α, IL-1β, IL-4 and IL-10) were similar between oXiris and CytoSorb and were higher with CytoSorb and oXiris versus Toraymyxin and similar endotoxin removal to Toraymyxin.52
Coupled plasma filtration adsorption (CPFA) is a new method of combining extracorporeal blood purification methods. Separation of plasma from the cellular components of blood with a highly permeable filter is followed by slowly going through the adsorptive material to remove several different cytokines. Then, the reinfusion of the purified plasma returns before the hemofilter to provide CRRT for renal/fluid support. The advantage of CPFA is that it has improved biocompatibility because there is no contact between blood cells and the sorbent material. Still, evidence supporting its effectiveness is scarce. Because it is expensive, it requires well-trained staff, labor intensive and associated with multiple technical issues that often lead to undertreatment.53 54
Two recent studies reported the incidence of AKI after CAR-T cell therapies. The first one identified the incidence of AKI post-CAR-T cell therapy in 46 adult patients with non-Hodgkin’s lymphoma, of whom 14 developed AKI (30%). The cumulative incidence of any grade AKI by day 100 was 30% (95% CI 16.9% to 43.9%) with grade 1 AKI incidence at 21.7% (95% CI 9.7% to 33.8%) and grade 2–3 AKI incidence at 8.7% (95% CI 0.4% to 17%). The overall incidence of CRS was 78.3% (95% CI 66% to 90.5%) out of which 13% (95% CI 3.3% to 22.8%) developed grade 3–4 CRS with a higher incidence of AKI. From the 14 patients with AKI, 3 died within 3 weeks of AKI onset due to progression of the underlying disease, 10 had the return of their kidney function to baseline and 1 had a serum creatinine 1.5 times higher than baseline by the end of follow-up. In this study, CRS was managed with tocilizumab and corticosteroids, with none of the patients who developed severe AKI requiring RRT.55
The second study consisted of 78 patients receiving CAR-T therapy for the treatment of diffuse large B-cell lymphoma, in which AKI occurred in 15 patients (19%) and CRS in 85%, treated with tocilizumab and dexamethasone. Of those with AKI, eight had decreased kidney perfusion, six developed acute tubular necrosis and 1 patient had urinary obstruction related to disease progression. Those with acute tubular necrosis and obstruction had the longest lengths of stay and highest 60-day mortality. Three of six with stage 3 AKI required RRT, but all three ultimately died in the hospital within 30 days (2 within 24 hours of initiating RRT).56
The multitude of CAR-T constructs that are available has different rates of CRS because of their distinct costimulatory domains which lead to differential rates of cell expansion and activation. As such, it is mandatory to know that AKI is likely to differ based on both patient characteristics and differences in CAR-T products. Also, chemotherapy, lymphodepletion regimen used, the need for intravenous contrast, concomitant nephrotoxic drugs, tumor lysis syndrome, urinary obstruction, sepsis, poor case management could lead to AKI and other organ failures requiring RRT as salvage therapy.
The pitfalls of CRRT are its adverse events, such as the need for properly prepared personnel, increased cost, catheter complications, risks of bleeding/clotting, hemodynamic instability, a drop in platelet count (thrombocytopenias), antibiotics removal with the need for dosing adjustments, loss of microelements and vitamins, electrolyte imbalances- hypophosphatemia, hypokalemia; hypothermia and the need for well design local protocols.57 58
Further studies should address the implementation of CRRT in patients with CRS. The questions that should be answered are deciding the optimal timing to start the RRT in CRS, how often would the filter need to be changed, what’s the duration of therapy, should there be a need for measuring the cytokine concentrations using the peak elevation as a starting point, are there any genetic predisposition to developing CRS and many more.
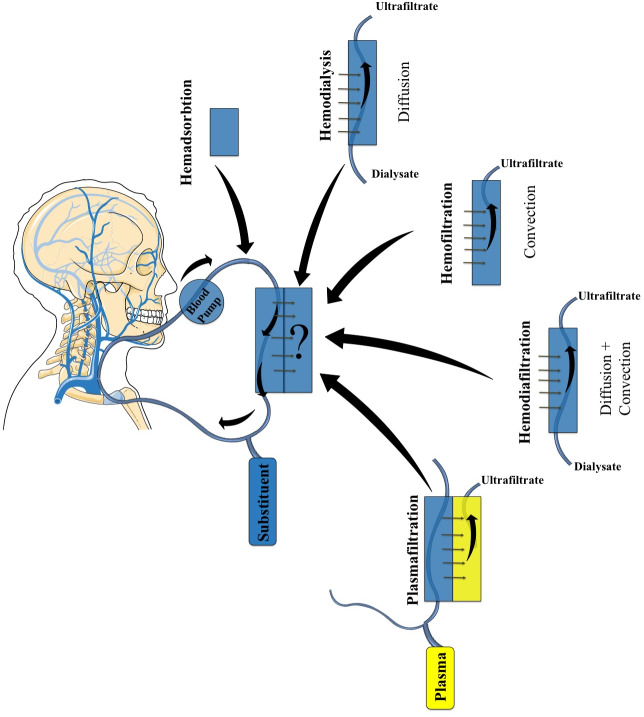
Still, this is far too little information and more research must be carried out, both in the preclinical setting, as well as in large clinical trials. Figure 2 depicts the types of continuous RRTs.
Figure 2.
Different methods for extracorporeal blood purification therapies, each having a similar core and a variation to the technique with its physicochemical principles mentioned. The small arrows represented near the tubular structures show the flow of the blood, while the big arrow depicted from each method to the ‘?’ represents the different variations of these methods.
Proof-of-concept for the hypothesis
To demonstrate the efficacy of the presented approach, we present our clinical experience of an 8-year-old male patient with very severe aplastic anemia unresponsive to antithymocyte globulin which underwent haploidentical hematopoietic stem cell transplant from his father.59–63 The conditioning was performed using thiotepa, melphalan and fludarabine, followed by the infusion of 8.58×106 CD34/kg peripheral blood stem cells. Graft versus host disease prophylaxis was performed with cyclophosphamide in days 3 and 4 posttransplant and with Prograf and mycophenolate mofetil on day 5.64–68 Neutrophil engraftment day was 24, while platelet engraftment was observed on day 25. Chimerism was assessed on day 27 and revealed a 100% donor chimerism. On day 29 both neutrophils and platelets started decreasing and it was revealed to be caused by an infection with BK virus (BKV). As of day 27, BKV was negative in plasma and urine but became positive in both when assessed on day 34, with a subsequent increase in the number of viral copies in day 41. Associated with this, it was observed that day 36 chimerism assessment dropped to 15% donor chimerism. Because of the viral infection, there was a donor lymphocyte infusion performed on day 42. In day 44, we assessed IL-6 levels, which were shown to be over the limit of 1000 pg/mL (normal <3.8 pg/mL). Corticosteroids were used only because tocilizumab was not available. On day 52, the patient presented the following toxicities by grade: grade 2: cardiac, renal, gastrointestinal, skin and coagulation; grade 3: respiratory and hepatic and grade 4: neurotoxicity. On day 53, it has been decided to initiate a CRRT with Cytosorb filter which was followed by the IL-6 level dropping to 8.4 pg/mL at day 60 and with regaining control and amelioration of the clinical symptoms, but with severe pancytopenia due to graft failure. The CRRT mode ordered was continuous veno-venous hemodiafiltration with Cytosorb adsorber with an effluent rate of 25–30 mL/kg/hour and blood flow range of 5–10 mL/kg/min. Because of severe thrombocytopenia, systemic anticoagulation was only used in bolus dose at the beginning of the procedure and not followed by continuous infusion of the anticoagulant. There was no dialysis needed, only hemofiltration, and the procedure lasted 24 hours, without any incidents. Afterwise, on day 69, the patient developed bacterial septic shock and died. Thus, although this case ended with exitus, CRRT with Cytosorb helped reverse some of the toxicities caused by IL-6.
Conclusions
Managing CRS remains a therapeutic challenge and close communication of hematology physicians with the ICU and nephrology teams will be essential in approaching future challenges.
CRS represents a severe adverse event following immunotherapy or cellular therapies, with strategies insufficiently developed to manage this condition. Nonetheless, there are still patients with CRS that are refractory to tocilizumab and corticosteroids which succumb to this complication, leading to the need for the next line of management strategy. In the current manuscript, we propose that this next line be represented by CRRT as it has been demonstrated to be efficacious in sepsis, a condition with a similar pathophysiological mechanism. The questions that remain unanswered are deciding the optimal timing to start the CRRT in CRS, how often would the filter need to be changed, what’s the duration of therapy, should there be a need for measuring the cytokine concentrations using the peak elevation as a starting point, and can we predict the development of severe CRS?
Acknowledgments
The authors acknowledge the support of the School for Doctoral Studies – Iuliu Hatieganu University of Medicine and Pharmacy in Cluj Napoca, Romania.
Footnotes
Contributors: All authors contributed the manuscript concept and writing. CC, HE and CT contributed to the presented concept. CC, SP, TT, PT, CV, SI and DD contributed to manuscript writing. DT, ES, AT and AC contributed to the clinical management of the patient. OES, HE and CT supervised the manuscript.
Funding: CC was financed from an internal grant of the School of Doctoral Studies - Iuliu Hatieganu University (PCD 2019-2021). CT was also supported by two national research grants of the Romanian Government: the first one awarded for Frontiers Research Projects 2018-2022 (grant number PN-III-P4-ID-PCCF-2016-112), awarded to the Babes Bolyai University in collaboration with the Ion Chiricuta Oncology Institute Cluj Napoca; as well as a second one, awarded for Young Research Teams 2020-2022 (grant number PN-III-P1-1.1-TE2019-0271) awarded to the Iuliu Hatieganu University of Medicine and Pharmacy Cluj Napoca. CT, DD, AT and OES were also financed by an international collaborative grant of the European Economic Space between Romania and Iceland 2020–2022 (grant number 19-COP-0031).
Competing interests: None declared.
Patient consent for publication: Not required.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Krok-Schoen JL, Fisher JL, Stephens JA, et al. Incidence and survival of hematological cancers among adults ages ≥75 years. Cancer Med 2018;7:3425–33. 10.1002/cam4.1461 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bonifant CL, Jackson HJ, Brentjens RJ, et al. Toxicity and management in car T-cell therapy. Molecular Therapy - Oncolytics 2016;3:16011 10.1038/mto.2016.11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Teachey DT, Rheingold SR, Maude SL, et al. Cytokine release syndrome after blinatumomab treatment related to abnormal macrophage activation and ameliorated with cytokine-directed therapy. Blood 2013;121:5154–7. 10.1182/blood-2013-02-485623 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Murthy H, Iqbal M, Chavez JC, et al. Cytokine release syndrome: current perspectives. Immunotargets Ther 2019;8:43–52. 10.2147/ITT.S202015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Frey N, Porter D. Cytokine release syndrome with chimeric antigen receptor T cell therapy. Biology of Blood and Marrow Transplantation 2019;25:e123–7. 10.1016/j.bbmt.2018.12.756 [DOI] [PubMed] [Google Scholar]
- 6.Shimabukuro-Vornhagen A, Gödel P, Subklewe M, et al. Cytokine release syndrome. J Immunother Cancer 2018;6:56 10.1186/s40425-018-0343-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fitzgerald JC, Weiss SL, Maude SL, et al. Cytokine release syndrome after chimeric antigen receptor T cell therapy for acute lymphoblastic leukemia. Crit Care Med 2017;45:e124–31. 10.1097/CCM.0000000000002053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ankawi G, Neri M, Zhang J, et al. Extracorporeal techniques for the treatment of critically ill patients with sepsis beyond conventional blood purification therapy: the promises and the pitfalls. Critical Care 2018;22:262 10.1186/s13054-018-2181-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Riegler LL, Jones GP, Lee DW. Current approaches in the grading and management of cytokine release syndrome after chimeric antigen receptor T-cell therapy]]>. Ther Clin Risk Manag 2019;15:323–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tanaka T, Narazaki M, Kishimoto T. Immunotherapeutic implications of IL-6 blockade for cytokine storm. Immunotherapy 2016;8:959–70. 10.2217/imt-2016-0020 [DOI] [PubMed] [Google Scholar]
- 11.Matthys P, Dillen C, Proost P, et al. Modification of the anti-CD3-induced cytokine release syndrome by anti-interferon-γ or anti-interleukin-6 antibody treatment: protective effects and biphasic changes in blood cytokine levels. Eur J Immunol 1993;23:2209–16. 10.1002/eji.1830230924 [DOI] [PubMed] [Google Scholar]
- 12.Rose-John S. Il-6 trans-signaling via the soluble IL-6 receptor: importance for the pro-inflammatory activities of IL-6. Int J Biol Sci 2012;8:1237–47. 10.7150/ijbs.4989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Norelli M, Camisa B, Barbiera G, et al. Monocyte-Derived IL-1 and IL-6 are differentially required for cytokine-release syndrome and neurotoxicity due to CAR T cells. Nat Med 2018;24:739–48. 10.1038/s41591-018-0036-4 [DOI] [PubMed] [Google Scholar]
- 14.Grupp SA, Kalos M, Barrett D, et al. Chimeric antigen Receptor–Modified T cells for acute lymphoid leukemia. N Engl J Med 2013;368:1509–18. 10.1056/NEJMoa1215134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hunter CA, Jones SA. Il-6 as a keystone cytokine in health and disease. Nat Immunol 2015;16:448–57. 10.1038/ni.3153 [DOI] [PubMed] [Google Scholar]
- 16.Lee DW, Gardner R, Porter DL, et al. Current concepts in the diagnosis and management of cytokine release syndrome. Blood 2014;124:188–95. 10.1182/blood-2014-05-552729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Maude SL, Frey N, Shaw PA, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med 2014;371:1507–17. 10.1056/NEJMoa1407222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Davila ML, Riviere I, Wang X, et al. Efficacy and toxicity management of 19-28z CAR T cell therapy in B cell acute lymphoblastic leukemia. Sci Transl Med 2014;6:224–5. 10.1126/scitranslmed.3008226 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kantarjian H, Stein A, Gökbuget N, et al. Blinatumomab versus chemotherapy for advanced acute lymphoblastic leukemia. N Engl J Med 2017;376:836–47. 10.1056/NEJMoa1609783 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kantarjian HM, Stein AS, Bargou RC, et al. Blinatumomab treatment of older adults with relapsed/refractory B-precursor acute lymphoblastic leukemia: results from 2 phase 2 studies. Cancer 2016;122:2178–85. 10.1002/cncr.30031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Topp MS, Gökbuget N, Stein AS, et al. Safety and activity of blinatumomab for adult patients with relapsed or refractory B-precursor acute lymphoblastic leukaemia: a multicentre, single-arm, phase 2 study. Lancet Oncol 2015;16:57–66. 10.1016/S1470-2045(14)71170-2 [DOI] [PubMed] [Google Scholar]
- 22.Maude SL, Barrett D, Teachey DT, et al. Managing cytokine release syndrome associated with novel T cell-engaging therapies. The Cancer Journal 2014;20:119–22. 10.1097/PPO.0000000000000035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kotch C, Barrett D, Teachey DT. Tocilizumab for the treatment of chimeric antigen receptor T cell-induced cytokine release syndrome. Expert Rev Clin Immunol 2019;15:813–22. 10.1080/1744666X.2019.1629904 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chen F, Teachey DT, Pequignot E, et al. Measuring IL-6 and sIL-6R in serum from patients treated with tocilizumab and/or siltuximab following CAR T cell therapy. J Immunol Methods 2016;434:1–8. 10.1016/j.jim.2016.03.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pawar A, Desai RJ, Solomon DH, et al. Risk of serious infections in tocilizumab versus other biologic drugs in patients with rheumatoid arthritis: a multidatabase cohort study. Ann Rheum Dis 2019;78:456–64. 10.1136/annrheumdis-2018-214367 [DOI] [PubMed] [Google Scholar]
- 26.Brudno JN, Kochenderfer JN. Toxicities of chimeric antigen receptor T cells: recognition and management. Blood 2016;127:3321–30. 10.1182/blood-2016-04-703751 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lamb YN, Deeks ED. Sarilumab: a review in moderate to severe rheumatoid arthritis. Drugs 2018;78:929–40. 10.1007/s40265-018-0929-z [DOI] [PubMed] [Google Scholar]
- 28.Pannu N, Gibney RTN. Renal replacement therapy in the intensive care unit. Ther Clin Risk Manag 2005;1:141–50. 10.2147/tcrm.1.2.141.62908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rimmelé T, Kaynar A, McLaughlin JN, et al. Leukocyte capture and modulation of cell-mediated immunity during human sepsis: an ex vivo study. Critical Care 2013;17:R59 10.1186/cc12587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang H-J, Wang P, Li N, et al. Effects of continuous renal replacement therapy on serum cytokines, neutrophil gelatinase-associated lipocalin, and prognosis in patients with severe acute kidney injury after cardiac surgery. Oncotarget 2017;8:10628–36. 10.18632/oncotarget.13254 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Joannes-Boyau O, Velly L, Ichai C. Optimizing continuous renal replacement therapy in the ICU. Curr Opin Crit Care 2018;24:476–82. 10.1097/MCC.0000000000000564 [DOI] [PubMed] [Google Scholar]
- 32.Honore PM, Spapen HD. What a clinician should know about a renal replacement membrane? J Transl Intern Med 2018;6:62–5. 10.2478/jtim-2018-0016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tandukar S, Palevsky PM. Continuous renal replacement therapy: who, when, why, and how. Chest 2019;155:626–38. 10.1016/j.chest.2018.09.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tolwani AJ, Wille KM. Anticoagulation for continuous renal replacement therapy. Semin Dial 2009;22:141–5. 10.1111/j.1525-139X.2008.00545.x [DOI] [PubMed] [Google Scholar]
- 35.von Brecht JH, Flanigan MJ, Freeman RM, et al. Regional anticoagulation: hemodialysis with hypertonic trisodium citrate. Am J Kidney Dis 1986;8:196–201. 10.1016/S0272-6386(86)80025-7 [DOI] [PubMed] [Google Scholar]
- 36.Bellomo R, Teede H, Boyce N. Anticoagulant regimens in acute continuous hemodiafiltration: a comparative study. Intensive Care Med 1993;19:329–32. 10.1007/BF01694706 [DOI] [PubMed] [Google Scholar]
- 37.Bellomo R, Cass A, et al. , RENAL Replacement Therapy Study Investigators . Intensity of continuous renal-replacement therapy in critically ill patients. N Engl J Med 2009;361:1627–38. [DOI] [PubMed] [Google Scholar]
- 38.Rimmelé T, Kellum JA. High-Volume hemofiltration in the intensive care unit: a blood purification therapy. Anesthesiology 2012;116:1377–87. [DOI] [PubMed] [Google Scholar]
- 39.Joannes-Boyau O, Honoré PM, Perez P, et al. High-Volume versus standard-volume haemofiltration for septic shock patients with acute kidney injury (IVOIRE study): a multicentre randomized controlled trial. Intensive Care Med 2013;39:1535–46. 10.1007/s00134-013-2967-z [DOI] [PubMed] [Google Scholar]
- 40.Hattori N, Oda S. Cytokine-adsorbing hemofilter: old but new modality for septic acute kidney injury. Ren Replace Ther 2016;2:41 10.1186/s41100-016-0051-1 [DOI] [Google Scholar]
- 41.Liu Y, Chen X, Wang D, et al. Hemofiltration successfully eliminates severe cytokine release syndrome following CD19 CAR-T-Cell therapy. J Immunother 2018;41:406–10. 10.1097/CJI.0000000000000243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Reeves JH, Butt WW, Shann F, et al. Continuous plasmafiltration in sepsis syndrome. Plasmafiltration in sepsis Study Group. Crit Care Med 1999;27:2096–104. 10.1097/00003246-199910000-00003 [DOI] [PubMed] [Google Scholar]
- 43.Xiao X, He X, Li Q, et al. Plasma exchange can be an alternative therapeutic modality for severe cytokine release syndrome after chimeric antigen Receptor-T cell infusion: a case report. Clinical Cancer Research 2019;25:29–34. 10.1158/1078-0432.CCR-18-1379 [DOI] [PubMed] [Google Scholar]
- 44.Ronco C. Sorbents: from bench to bedside can we combine membrane separation processes and adsorbent based solute removal? Int J Artif Organs 2006;29:819–22. 10.1177/039139880602900901 [DOI] [PubMed] [Google Scholar]
- 45.Bonavia A, Groff A, Karamchandani K, et al. Clinical utility of extracorporeal cytokine Hemoadsorption therapy: a literature review. Blood Purif 2018;46:337–49. 10.1159/000492379 [DOI] [PubMed] [Google Scholar]
- 46.The Adsorber [Internet] CytoSorbents Europe GmbH. Available: https://cytosorb-therapy.com/en/the-adsorber/ [Accessed 14 Feb 2020].
- 47.The Therapy [Internet] CytoSorbents Europe GmbH. Available: https://cytosorb-therapy.com/en/the-therapy/ [Accessed 14 Feb 2020].
- 48.Kellum JA, Venkataraman R, Powner D, et al. Feasibility study of cytokine removal by hemoadsorption in brain-dead humans*. Crit Care Med 2008;36:268–72. 10.1097/01.CCM.0000291646.34815.BB [DOI] [PubMed] [Google Scholar]
- 49.Schädler D, Pausch C, Heise D, et al. The effect of a novel extracorporeal cytokine hemoadsorption device on IL-6 elimination in septic patients: a randomized controlled trial. PLoS One 2017;12:e0187015 10.1371/journal.pone.0187015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Schädler D, Porzelius C, Jörres A, et al. A multicenter randomized controlled study of an extracorporeal cytokine hemoadsorption device in septic patients. Crit Care 2013;17:P62 10.1186/cc12000 [DOI] [Google Scholar]
- 51.Maeda H, Tomisawa N, Jimbo Y, et al. Efficacy of hemofiltration with PEPA membrane for IL-6 removal in a rat sepsis model. J Artif Organs 2017;20:335–40. 10.1007/s10047-017-0991-4 [DOI] [PubMed] [Google Scholar]
- 52.Malard B, Lambert C, Kellum JA. In vitro comparison of the adsorption of inflammatory mediators by blood purification devices. ICMx 2018;6:12 10.1186/s40635-018-0177-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hassan J, Cader RA, Kong NC, et al. Coupled plasma filtration adsorption (CPFA) plus continuous Veno-Venous haemofiltration (CVVH) versus CVVH alone as an adjunctive therapy in the treatment of sepsis. Excli J 2013;12:681–92. [PMC free article] [PubMed] [Google Scholar]
- 54.Abdul Cader R, Abdul Gafor H, Mohd R, et al. Coupled plasma filtration and adsorption (CPFA): a single center experience. Nephrourol Mon 2013;5:891–6. 10.5812/numonthly.11904 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Gutgarts V, Jain T, Zheng J, et al. Acute kidney injury after CAR-T cell therapy: low incidence and rapid recovery. Biol blood marrow transplant. J Am Soc Blood Marrow Transplant 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Gupta S, Seethapathy H, Strohbehn IA, et al. Acute kidney injury and electrolyte abnormalities after chimeric antigen receptor T-cell (CAR-T) therapy for diffuse large B-cell lymphoma. Am J Kidney Dis 2020. 10.1053/j.ajkd.2019.10.011. [Epub ahead of print: 20 Jan 2020]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Druml W. Metabolic aspects of continuous renal replacement therapies. Kidney Int 1999;56:S56–61. 10.1046/j.1523-1755.56.s72.1.x [DOI] [PubMed] [Google Scholar]
- 58.Kim Z, Goldfarb DS. Continuous renal replacement therapy does not have a clear role in the treatment of poisoning. Nephron Clin Pract 2010;115:c1–6. 10.1159/000286343 [DOI] [PubMed] [Google Scholar]
- 59.Gaballa S, Ge I, El Fakih R, et al. Results of a 2-arm, phase 2 clinical trial using post-transplantation cyclophosphamide for the prevention of graft-versus-host disease in haploidentical donor and mismatched unrelated donor hematopoietic stem cell transplantation. Cancer 2016;122:3316–26. 10.1002/cncr.30180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Tănase A, Tomuleasa C, Mărculescu A, et al. First successful haploidentical stem cell transplantation in Romania. Rom J Intern Med 2016;54:194–200. 10.1515/rjim-2016-0021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Tanase A, Tomuleasa C, Marculescu A, et al. Haploidentical donors: can faster transplantation be Life-Saving for patients with advanced disease? Acta Haematol 2016;135:211–6. 10.1159/000443469 [DOI] [PubMed] [Google Scholar]
- 62.Brammer JE, Khouri I, Gaballa S, et al. Outcomes of Haploidentical Stem Cell Transplantation for Lymphoma with Melphalan-Based Conditioning. Biol Blood Marrow Transplant 2016;22:493–8. 10.1016/j.bbmt.2015.10.015 [DOI] [PubMed] [Google Scholar]
- 63.Colita A, Colita A, Bumbea H, et al. LEAM vs. beam vs. CLV conditioning regimen for autologous stem cell transplantation in malignant lymphomas. retrospective comparison of toxicity and efficacy on 222 patients in the first 100 days after transplant, on behalf of the Romanian Society for bone marrow transplantation. Front Oncol 2019;9:892 10.3389/fonc.2019.00892 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sas V, Moisoiu V, Teodorescu P, et al. Approach to the adult acute lymphoblastic leukemia patient. JCM 2019;8:1175 10.3390/jcm8081175 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Constantinescu C, Bodolea C, Pasca S, et al. Clinical approach to the patient in critical state following immunotherapy and/or stem cell transplantation: guideline for the on-call physician. J Clin Med 2019;8:884. 10.3390/jcm8060884 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Qian L, Dima D, Berce C, et al. Protein dysregulation in graft versus host disease. Oncotarget 2018;9:1483–91. 10.18632/oncotarget.23276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Tomuleasa C, Fuji S, Berce C, et al. Chimeric antigen receptor T-cells for the treatment of B-cell acute lymphoblastic leukemia. Front Immunol 2018;9:239 10.3389/fimmu.2018.00239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Frinc I, Dima D, Chitic M, et al. Transthoracic ultrasonography for the follow-up of a chronic lymphocytic leukemia patient with chemotherapy-induced immunosuppression prior to allogeneic stem cell transplantation. A case report. Med Ultrason 2017;19:330–2. 10.11152/mu-976 [DOI] [PubMed] [Google Scholar]
- 69.Lee DW, Santomasso BD, Locke FL, et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biology of Blood and Marrow Transplantation 2019;25:625–38. 10.1016/j.bbmt.2018.12.758 [DOI] [PMC free article] [PubMed] [Google Scholar]