Refutation of TRPA1-mediated lysosomal calcium release in sensory neurons, emphasizing the requirement and influx of extracellular calcium through TRPA1 for activation.
Abstract
Shang et al. (2016. J. Cell Biol. https://doi.org/10.1083/jcb.201603081) reported that activation of lysosomal TRPA1 channels led to intracellular calcium transients and CGRP release from DRG neurons. We argue that both findings are more likely due to influx of insufficiently buffered extracellular calcium rather than lysosomal release.
Introduction
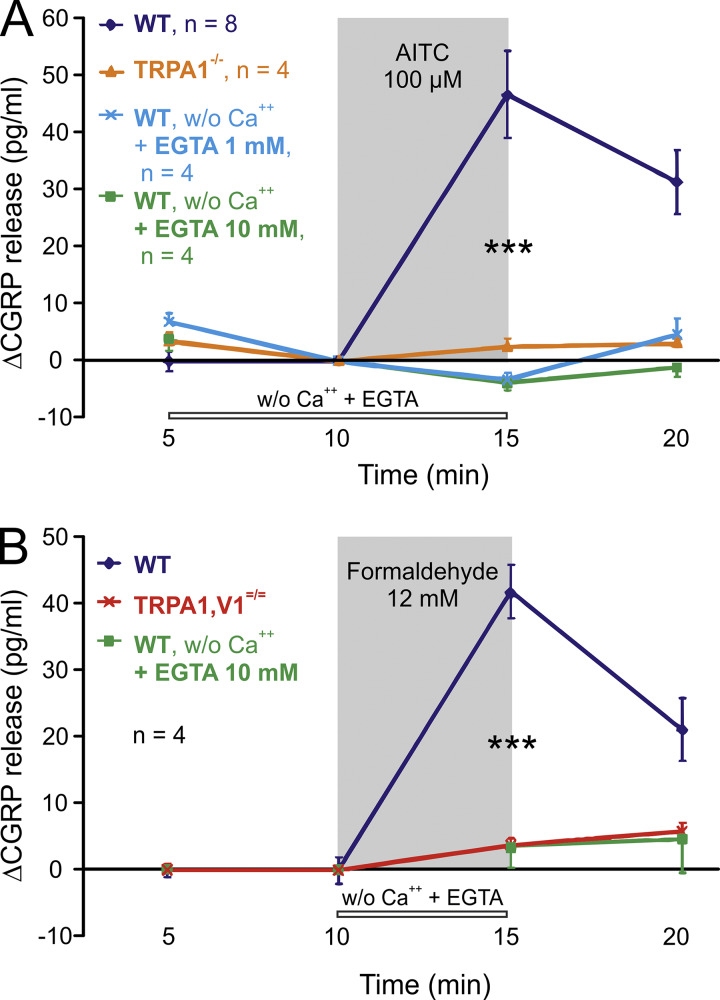
A recent article (Shang et al., 2016) proposed functional expression of the chemoreceptive ion channel TRPA1 (transient receptor potential channel A1) in calcium-storing lysosomes of nociceptive mouse dorsal root ganglion (DRG) neurons: The agonist allyl isothiocyanate (AITC) increased cytosolic calcium in the nominal absence of extracellular calcium ions that would otherwise flow in through the activated TRPA1 channels in the plasma membrane. 40% of the overall stimulated calcium rise was attributed to lysosomal release and reported as sufficient to evoke vesicular exocytosis of calcitonin gene–related peptide (CGRP) from the neuronal somata. Cultured rodent DRG nerve cell bodies have shown merit as surrogates for their inaccessible peripheral terminals, but they remain what they are: a model, with differences in geometry, epigenetics, protein infrastructure, environment, and function compared with their peripheral nerve endings. In those terminals, in the trachea for example, we do not find detectable CGRP release upon TRPA1 activation by AITC and formaldehyde, if extracellular calcium is safely removed (Fig. 1). This physiological result is in accord with CGRP measurements of spinal sensory terminals stimulated with various electrophilic TRPA1 agonists and of DRG neurons stimulated with bradykinin, which also acts through TRPA1 activation (Vasko et al., 1994; Bandell et al., 2004; Quallo, 2015; Benemei et al., 2017; Li Puma et al., 2019).
Figure 1.
Stimulated CGRP release requires extracellular calcium ions and TRPA1 expression in isolated mouse trachea. (A) AITC 100 µM, methods as in Kichko and Reeh (2009); ANOVA and Tukey´s honestly significant difference test. Constitutive basal CGRP secretion ranged between 7 and 15 pg/ml in wild-type C57BL/6 and congenic TRPA1−/− mice. (B) Formaldehyde 12 mM, wild-type mice, and TRPA1/V1 double-knockouts. Constitutive basal CGRP was below the detection limit (5 pg/ml) in both congenic mouse strains. ***, P < 0.001.
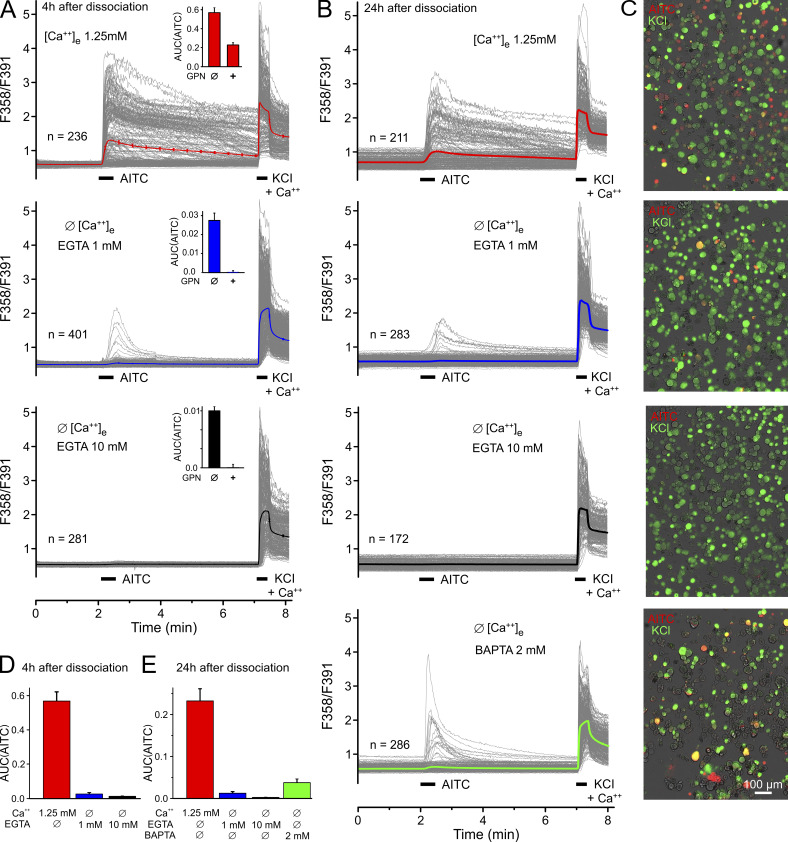
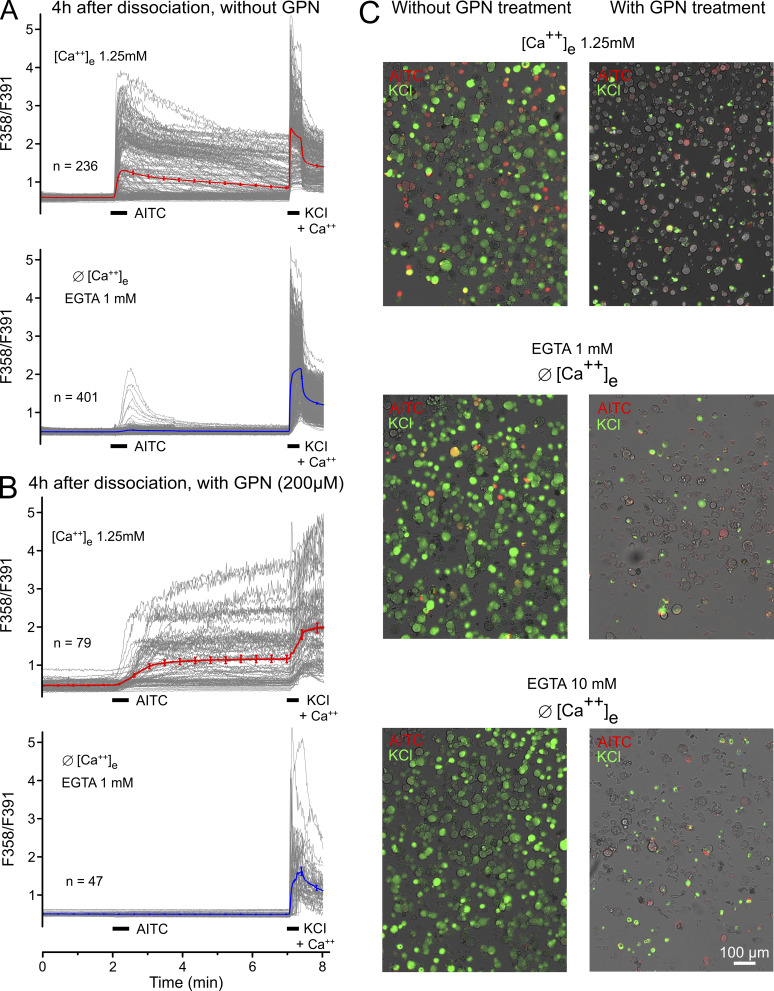
To examine this discrepancy in DRG neurons, we replicated experiments from Shang et al. (2016). However, there were two purpose-designed changes in our approach. We employed complete bath exchange using 10 mM in comparison to 1 mM EGTA as extracellular calcium buffer; Shang et al. (2016) puffed the extracellular solution onto the cells and used 1 mM EGTA. We consider 1 mM insufficient to detach and chelate all loosely attached calcium ions binding to the anionic extracellular moieties of membrane proteins. We repeated the key experiment of Shang et al. (2016) (their Fig. 1 C), stimulating hundreds of mouse DRG neurons (cultured for 4–6 h) with 100 µM AITC in the presence of 1 mM EGTA and the absence of calcium (Fig. 2, A and D). Indeed, 14% of the cells responded with small calcium transients, apparently indicating some intracellular calcium release. However, we did not observe any calcium transients in the presence of 10 mM EGTA, while 35% of DRG neurons responded with a large calcium influx in the presence of extracellular calcium. Using DRG neurons that had been rested overnight in culture, 4% responded to AITC with calcium transients in the presence of 1 mM EGTA, and no calcium transients were observed in the presence of 10 mM EGTA (Fig. 2, B, C, and E). Hsu and Lee (2015) used 5 mM EGTA in calcium-free external solution and found no intracellular calcium increase in response to 100 µM AITC applied to sensory neurons of the jugular ganglion. Given the limited selectivity of many TRPA1 agonists, including AITC (Gees et al., 2013), it is important that other electrophilic TRPA1 agonists also did not increase cytosolic calcium in DRG neurons in the safe absence of external calcium (Eberhardt et al., 2012; Fischer et al., 2015).
Figure 2.
Mouse DRGs do not respond to 100 µM AITC in a nominally Ca-free environment with 10 mM EGTA. 100 µM AITC and 60 mM KCl (bars) were applied during calcium imaging; Ca-free condition was established during 10 min after washout of external Fura-2-AM and terminated just before KCl application. The ratio time courses of single neurons are shown in gray, and the average in bold and color. (A) Neurons were imaged 4–6 h after dissociation and incubation. The inserts show AITC responses after 200 µM GPN pretreatment for 40 min (+) in comparison to control (Ø); note the very different ordinate scales. (B) Neurons were incubated overnight before Ca imaging. BAPTA as calcium buffer reduced the number of responsive neurons to 8% (vs. 30% in Shang et al. [2016]). (C) Overlays of the transmission image (gray) and the response to AITC (red) and KCl (green) of representative experiments. Neurons responding to both applications appear in yellow. (D) Area under the curve of Ca2+ transients in response to AITC quantified from A. (E) Area under the curve of Ca2+ transients in response to AITC quantified from B, including the BAPTA effect. Data from A and photomicrographs from C also appear in Fig. S2, A and C, to allow for comparison. Error bars represent SEM values of the counted (n) neurons.
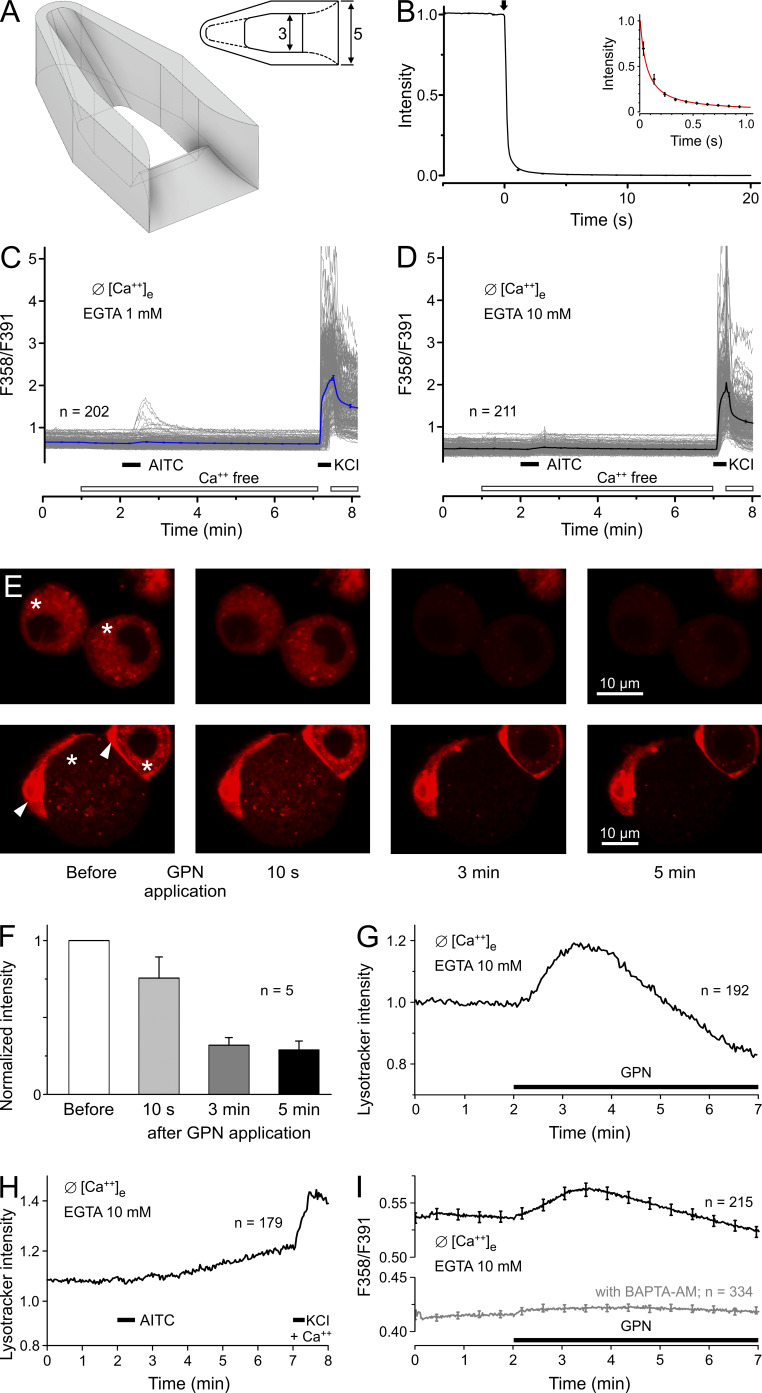
Our procedure to achieve calcium-free conditions involves a 10-min washout of extracellular calcium with EGTA buffer, and this might deplete the intracellular stores. However, using a novel custom-made miniature dish accommodating the freshly dissociated DRG neurons, we achieved nominally calcium-free conditions within <1 s (Fig. 3, A and B). Maintaining these conditions, AITC was applied after 1 min and slightly activated 6 of 202 neurons in the presence of 1 mM EGTA; in the presence of 10 mM EGTA, 0 of 211 neurons exhibited calcium transients in response to AITC (Fig. 3, C and D).
Figure 3.
After 1 min without external calcium, DRGs respond minimally in 1 mM EGTA but not in 10 mM EGTA. (A) Schematic drawing of the purpose-designed perfusion chamber generated by stereolithography using the 3D printer Form 2 (Formlabs Boston/Berlin) with 25-µm resolution and Fusion 360 software. The 2D view from the top provides dimensions in millimeters; the perfusion capillary with 1.5-mm outer diameter was placed at the left end. (B) The intensity time course of transmitted light after a switch (arrow) from a clear solution to one containing methylene blue was recorded seven times at 10 Hz. The solution was gravity driven at a flowrate of 1 ml/min and removed by a suction tube placed next to the outlet. The inset shows a power-law fit to the curve during the first second. (C) DRGs were measured 4–6 h after plating using the described perfusion chamber. The solution in the chamber was changed from calcium-containing to calcium-free 1 min before exposure to 100 µM AITC. The nominally calcium-free solution contained EGTA 1 mM. (D) 10 mM EGTA eliminated the responses to AITC. (E) DRG cells, neurons of different size (labeled *). and attached satellite cells (labeled with arrowheads), were loaded with LysoTracker and imaged by confocal microscopy under nominally calcium-free conditions (+ 10 mM EGTA). Images were acquired before and during 200 µM GPN application as indicated. The images show the rapid fading of pH-dependent fluorescence in four representative neurons, indicating lysosome disruption. (F) The normalized LysoTracker fluorescence intensity of five neurons dropped upon GPN application to ~25% within 5 min. (G) The same protocol was applied using wide-field microfluorimetry, allowing observation of more neurons. After a transient increase under GPN, LysoTracker fluorescence faded, indicating mass disruption of lysosomes. (H) Neurons stained with LysoTracker show no visible effect of 30-s AITC application (100 µM) in the absence of extracellular calcium. (I) Fura-2 calcium imaging reveals the small intracellular release of calcium ions from disintegrating lysosomes evoked by 200 µM GPN. Neurons pretreated with the intracellular calcium chelator BAPTA-AM for 30 min showed no response to GPN application. Error bars represent SEM values of the counted (n) neurons.
Residual calcium ions in nominally calcium-free physiological buffers result from inevitable contaminations of the commercial American Chemical Society–grade constituents, which can amount to 12 µM calcium in a final solution (Patton et al., 2004). Exactly this extracellular concentration is sufficient to cause a significant potentiation of agonist-evoked rTRPA1 currents. This potentiation depends on the influx of calcium ions through the membrane channel; intracellular calcium is an agonist of TRPA1, and half-maximal activation of the channel is achieved by 225 nM calcium (Wang et al., 2008). The problem of contamination can be minimized by raising the EGTA concentration, for example from 1 to 10 mM, which reduces the relative error from 12% to 1% in case of an assumed 10 µM calcium contamination (Patton et al., 2004).
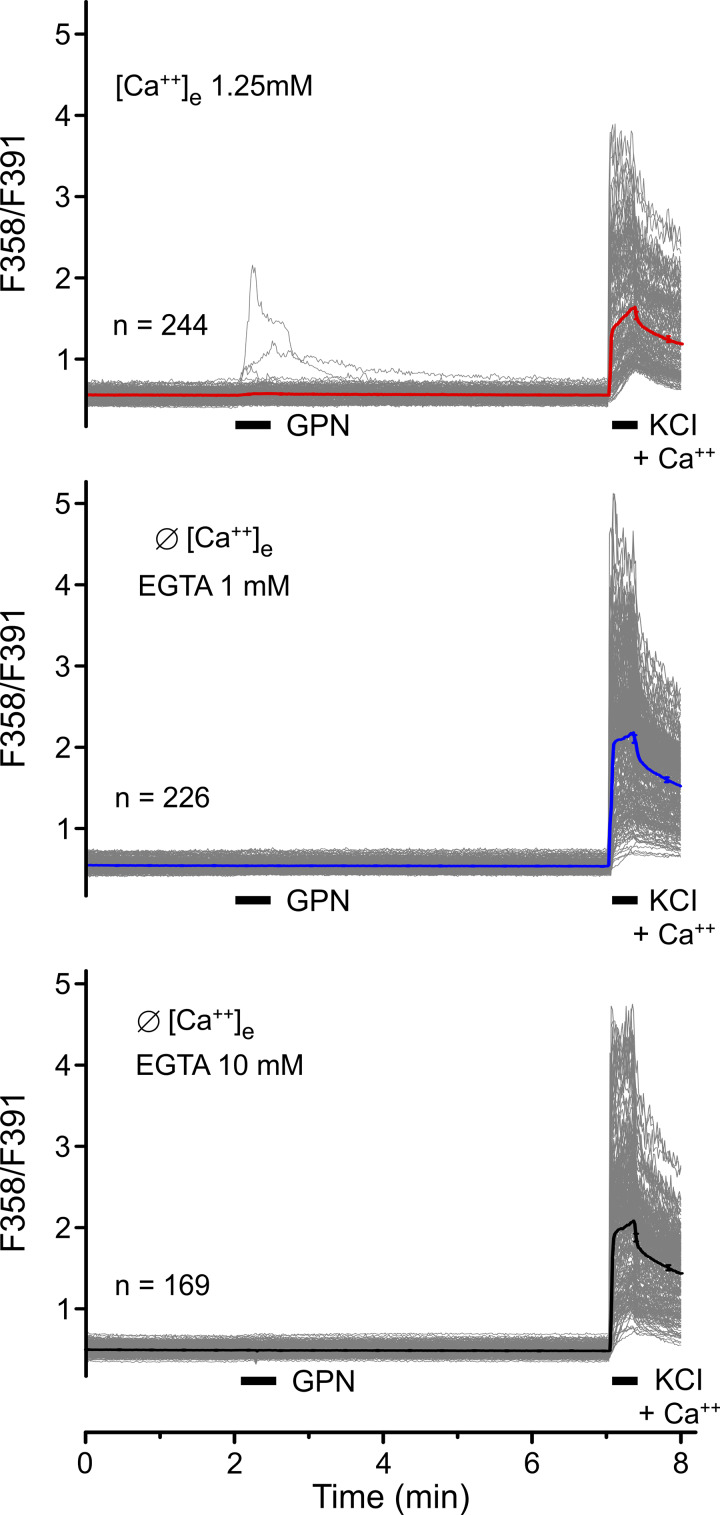
Shang et al. (2016) used glycylphenylalanine 2-naphtylamide (GPN) to osmotically disrupt the lysosomes, which caused a small cytosolic calcium transient (Fig. 3 C in Shang et al. [2016]; and our Fig. S1). We used Fura-2 wide-field microfluorimetry and confirmed GPN-induced intracellular release of calcium ions that is quenched by pretreatment with the membrane permeable BAPTA-AM (Fig. 3 I). We also confirmed that GPN disrupts lysosomes in DRG neurons using LysoTracker, a fluorescent dye for tracking acidic organelles (Fig. 3, E–G). In addition, we found that the membrane-permeable AITC had no effect on LysoTracker fluorescence (Fig. 3 H), although TRPA1 should pass protons when activated (Ye et al., 2018). As shown in Shang et al. (2016), we observed that prolonged GPN pretreatment of the DRG neurons reduced the large AITC response in the presence of external calcium as much as the removal of extracellular calcium (Fig. 3, G and H, in Shang et al. [2016]; and inserts in our Fig. 2 A). However, in our hands, the yield of responsive neurons was also greatly reduced (mean n = 318 vs. 63 per field of view), as was the KCl control response of the cells that underwent the GPN pretreatment (Fig. S2). These findings may suggest off-target effects of GPN, compromising the ability of the DRG neurons to respond to AITC and to nonspecific depolarization.
Figure S1.
Wide-field Fura-2 microfluorimetry data from freshly dissociated mouse DRG neurons. In the absence of external calcium, we do not observe calcium transients in neurons stimulated by brief (30-s) applications of the lysosome disruptor GPN (200 µM) using 1 mM (middle panel) or 10 mM (lower panel) EGTA as external calcium buffer. In the presence of external calcium (upper panel), 3 of 244 neurons showed small and variable responses. These findings are in contrast to small cytosolic calcium transients evoked by 5-min application of GPN (Fig. 3 I), which paralleled the disruption of lysosomes (Fig. 3 G) with a mean time to peak calcium increase of 100 s.
Figure S2.
Wide-field Fura-2 microfluorimetry data from freshly dissociated mouse DRG neurons. (A and B) Each panel represents pooled results from three to four measurements on two to three different days; n represents the mean number of viable (i.e., KCl-responsive) neurons per field of view. Captions indicate the extracellular calcium concentrations used and presence or absence of 1 mM EGTA; bars indicate the applications of 100 µM AITC and 60 mM KCl to activate TRPA1 and, by unspecific depolarization, voltage-gated calcium channels in the plasma membrane, respectively. (A) Naive DRG neurons without pretreatment, 35% responding with calcium transients to AITC in presence of extracellular calcium (upper panel), 14% in calcium-free condition (+ 1 mM EGTA); the bold and red- or blue-colored traces represent the mean (±SEM). (B) DRG neurons were pretreated with lysosome-disrupting GPN for 30 min during the Fura-2 exposure period in the presence of extracellular calcium, followed by 10-min GPN superfusion with calcium (upper panel) or calcium-free solution (+ 1 mM EGTA; lower panel). The yield of KCl-responsive neurons was strongly reduced compared with naive DRGs; the kinetics of AITC and KCl responses were reduced as well as their amplitudes; no neuron was activated by AITC in calcium-free solution; and the KCl responses were slowed and reduced. (C) Photomicrographs of false-color-stained AITC-responsive (red) and KCl-responsive (green) neurons; double-stained cells appear in orange/yellow, gray cells are unresponsive, which is the dominant color following GPN pretreatment (right column) irrespective of presence (upper row) or absence (lower rows) of extracellular calcium or of 1 or 10 mM EGTA, compared with naive DRG neurons (left column). Data from A and photomicrographs from C are also presented in Fig. 2, A and C.
In conclusion, we propose that in DRG neurons cytosolic calcium increase evoked by AITC in the absence of external calcium, as described by Shang et al. (2016), is actually due to insufficient buffering of extracellular calcium ions and influx through activated TRPA1 channels in the plasma membrane. Whether calcium-induced calcium release plays an amplifying role remains to be elucidated (Ouyang et al., 2005). In addition, we found that TRPA1-evoked CGRP release from isolated peripheral nerves depends entirely on the availability of extracellular calcium.
Materials and methods
Animals and trachea preparation
The experiments were performed using C57BL/6 mice (in-house breeding colony; originally from Jackson Laboratory) and mice carrying targeted deletions of Trpv1 or Trpa1; the initial heterozygous breeding pairs were generous gifts of GlaxoSmithKline (Dr. John Davis, formerly Harlow, UK) or Drs. Kelvin Kwan and David Corey (Harvard University, Cambridge, MA), respectively, and the animals had been continuously back–crossed on C57BL/6 since 2001 or 2006. Mice were housed in group cages in a temperature-controlled environment on a 12-h light–dark cycle and were supplied with water and food ad libitum. Mice of either sex, body weight 15–25 g, were euthanized by exposure to a rising CO2 atmosphere. The trachea was excised from the thyroid cartilage to the carina and hemisected along the midline. One half of the preparation was then used as control, and the other half for experimental variations. The preparations were washed for 30 min at 37°C in synthetic interstitial fluid (SIF; in mM): NaCl 107.8, KCl 3.5, CaCl2 1.53, MgSO4 0.69, NaHCO3 26.2, NaH2PO4 1.67, sodium gluconate 9.64, glucose 5.55, and sucrose 7.6. The fluid had a temperature of 37°C and was gassed with carbogen (95% O2, 5% CO2) to obtain pH 7.4. The husbandry and usage of the animals were approved by the District Government of Mittelfranken (Ansbach, Germany), and experiments were performed in accordance with the guidelines of the International Association for the Study of Pain.
Stimulation and chemicals
After the initial washing period, the isolated trachea was consecutively incubated for 5 min in each of four tubes mounted in a temperature-controlled shaking bath at 37°C. The first two incubations were performed to determine basal CGRP release. The third tube contained the stimulating chemicals, 100 µM AITC (Sigma-Aldrich) or formaldehyde (Sigma-Aldrich), as TRPA1 agonists. Formaldehyde was bought as a saturated aqueous solution (formalin, 37–37.5 mass %, stabilized by methanol 8–12%; Carl Roth). For calcium-free conditions, CaCl2 was omitted from the SIF, and either 1 or 10 mM EGTA (Sigma-Aldrich) was added; this solution was then used to apply the stimulants and as pretreatment in case of subsequent AITC application.
CGRP enzyme immunoassay (EIA)
The CGRP content of the incubation fluid was measured using commercial EIA kits with a detection threshold of 5 pg/ml (Bertin Pharma). For this purpose 100 µl sample fluid was stored on ice and, immediately after the tissue exposure period, mixed with 25 µl of fivefold-concentrated commercial CGRP-EIA buffer that contained a proprietary cocktail of peptidase inhibitors. The antibody reactions took place overnight. The EIA plates were read photometrically using a microplate reader (Dynatech). All results were processed as measured by the EIA in picograms of CGRP per milliliter of SIF. To reduce interindividual variability and day-to-day baseline variability, the data refer to the second individual baseline value (before stimulation). This value was subtracted from all four data points of a typical experiment so that only the absolute change in CGRP release (Δ picograms per milliliter) is displayed in the figures.
Statistical comparisons
Statistical comparisons were performed using Statistica 7 (Statsoft). All time series of experimental values were first analyzed for the effect of chemical stimulation compared with baseline CGRP secretion using nonparametric Wilcoxon matched pairs test. The baseline-corrected (i.e., Δ pg/ml) CGRP values were entered into a one-way ANOVA followed by Tukey’s honestly significant difference test, focusing on the peak values of stimulated CGRP release to compare the chemically stimulated responses in WT, TRPA1−/− knockout mice and double-knockout mice (TRPA1−/− and transient receptor potential channel V1 [TRPV1−/−]). P < 0.05 was considered statistically significant. Data points represent means ± SEM of the given number (n) of experiments on different animals. Triple asterisk symbols in the figures (***) indicate significant differences at P < 0.001.
Cell culture
DRGs were taken from the spinal columns of C57BL/6 mice. After removing DRGs from all spinal levels, the nerves and roots were cut off. The cleaned DRGs were incubated in 0.5% streptomyces protease and 1% clostridium collagenase at 37°C for 30 min (Sigma-Aldrich). The cells were first washed and then mechanically dissociated in serum-free TNB 100 medium supplemented with TNB 100 protein–lipid complex (Biochrom), penicillin, and streptomycin (100 U/ml each; Life Technologies) and murine nerve growth factor (mouse NGF 2.5S, 100 ng/ml; Alomone Labs). They were incubated on glass coverslips coated with poly-d-lysine (200 µg/ml; Sigma-Aldrich) in the above medium at 37°C in a 5% CO2 atmosphere for ∼24 h before the Ca-imaging measurements. Alternatively, the neurons were imaged for 4–6 h after incubation.
Calcium imaging
For Ca imaging, the fluorescent calcium indicator dye Fura-2-AM was used. It was mixed with 0.02% pluronic F-127 (Thermo Fisher Scientific or Biotrend) and diluted to 3 µM in extracellular solution containing (in mM): 1.25 calcium, 145 NaCl, 5 KCl, 10 glucose, 10 Hepes, and 1 MgCl2. DRG cells were loaded for ∼30 min at 37°C with 5% CO2 and then washed for 10 min at room temperature in either the Ca-containing extracellular solution for measurements with calcium or nominally Ca-free solutions containing 2 mM BAPTA, 1 mM EGTA, or 10 mM EGTA. In additional experiments, 3D-printed miniature superfusion chambers were used to accommodate freshly dissociated DRGs neurons; these allowed for rapid and complete bath exchange in <60 s before the stimulation with 100 µM AITC (Fig. 3). In some experiments, BAPTA-AM (10 µM; Merck) was applied to the cells during Fura-2 staining for 30 min and washed out before imaging.
The Ca imaging measurements were performed using an inverted microscope and excitation at 358- and 391-nm wavelengths with a Polychrome V monochromator (Till Photonics) at 1 Hz. A Peltier-cooled slow-scan charge-coupled device camera recorded the fluorescence emission >440 nm, controlled by TillVision software. The software was also used to calculate the fluorescence ratio for all selected neurons with background correction.
During the experiments, a gravity-driven and software-controlled common outlet perfusion system superfused the cells continuously with a flow rate of ∼1 ml/min. The protocols included a final application of 60 mM KCl in calcium-containing solution (even for the Ca-free measurements) to detect all responsive cells.
Lysotracker staining and imaging
DRG cells were loaded after 4–6 h in culture with Lysotracker 50 nM (Lysotracker Red DND-99; Thermo Fisher Scientific) for 1 h at 37°C with 5% CO2. After washing for 10 min at room temperature, Lysotracker fluorescence was monitored at 518 nm every 2 s. Control experiments allowed to correct for a slow but steady signal loss due to bleaching without intervention.
Confocal imaging
The measurements on the confocal microscope (Zeiss LSM 780) were performed to gain a higher resolution for the detection of lysosomes. The DRG cells were loaded with Lysotracker as described above and washed for 10 min at room temperature. The fluorescence was acquired before and after GPN 200 µM application, as indicated (in Fig. 3).
Online supplemental material
Fig. S1: In the absence of external calcium, we do not observe calcium transients in DRG neurons stimulated by the lysosome disruptor GPN. Fig. S2: Following GPN pretreatment, calcium imaging shows a massive reduction of DRG neurons responding to AITC and unspecific depolarisation by KCl.
Acknowledgments
We thank S. Haux-Oertel, A. Kuhn, I. Izidorczyk, and J. Schramm for excellent technical assistance and Birgit Vogler for assistance with confocal microscopy. Dr. Cosmin Ciotu is gratefully acknowledged for constructing the miniature dishes described in Fig. 3.
Funded by Deutsche Forschungsgemeinschaft (Interdisziplinäre Zentrum für Klinische Forschung Erlangen) Project E 27 to M.J.M. Fischer and DFG 704/3-1 to P.W. Reeh.
The authors declare no competing financial interests.
Author contributions: T.I. Kichko, M.J.M. Fischer, and P.W. Reeh: conception and design of research; T.I. Kichko and L. Gebhardt performed experiments; T.I. Kichko, M.J.M. Fischer, L. Gebhardt, and P.W. Reeh analyzed data and interpreted results of experiments; T.I. Kichko and L. Gebhardt prepared the figures; TIK and P.W. Reeh drafted the manuscript; T.I. Kichko, M.J.M. Fischer, L. Gebhardt, and P.W. Reeh approved the final version of the manuscript.
References
- Bandell M., Story G.M., Hwang S.W., Viswanath V., Eid S.R., Petrus M.J., Earley T.J., and Patapoutian A.. 2004. Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin. Neuron. 41:849–857. 10.1016/S0896-6273(04)00150-3 [DOI] [PubMed] [Google Scholar]
- Benemei S., De Logu F., Li Puma S., Marone I.M., Coppi E., Ugolini F., Liedtke W., Pollastro F., Appendino G., Geppetti P., et al. 2017. The anti-migraine component of butterbur extracts, isopetasin, desensitizes peptidergic nociceptors by acting on TRPA1 cation channel. Br. J. Pharmacol. 174:2897–2911. 10.1111/bph.13917 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eberhardt M.J., Filipovic M.R., Leffler A., de la Roche J., Kistner K., Fischer M.J., Fleming T., Zimmermann K., Ivanovic-Burmazovic I., Nawroth P.P., et al. 2012. Methylglyoxal activates nociceptors through transient receptor potential channel A1 (TRPA1): a possible mechanism of metabolic neuropathies. J. Biol. Chem. 287:28291–28306. 10.1074/jbc.M111.328674 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischer M.J., Soller K.J., Sauer S.K., Kalucka J., Veglia G., and Reeh P.W.. 2015. Formalin evokes calcium transients from the endoplasmatic reticulum. PLoS One. 10 e0123762 10.1371/journal.pone.0123762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gees M., Alpizar Y.A., Boonen B., Sanchez A., Everaerts W., Segal A., Xue F., Janssens A., Owsianik G., Nilius B., et al. 2013. Mechanisms of transient receptor potential vanilloid 1 activation and sensitization by allyl isothiocyanate. Mol. Pharmacol. 84:325–334. 10.1124/mol.113.085548 [DOI] [PubMed] [Google Scholar]
- Hsu C.C., and Lee L.Y.. 2015. Role of calcium ions in the positive interaction between TRPA1 and TRPV1 channels in bronchopulmonary sensory neurons. J. Appl. Physiol. 118:1533–1543. 10.1152/japplphysiol.00043.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kichko T.I., and Reeh P.W.. 2009. TRPV1 controls acid- and heat-induced calcitonin gene-related peptide release and sensitization by bradykinin in the isolated mouse trachea. Eur. J. Neurosci. 29:1896–1904. 10.1111/j.1460-9568.2009.06747.x [DOI] [PubMed] [Google Scholar]
- Li Puma S., Landini L., Macedo S.J. Jr., Seravalli V., Marone I.M., Coppi E., Patacchini R., Geppetti P., Materazzi S., Nassini R., et al. 2019. TRPA1 mediates the antinociceptive properties of the constituent of Crocus sativus L., safranal. J. Cell. Mol. Med. 23:1976–1986. 10.1111/jcmm.14099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ouyang K., Zheng H., Qin X., Zhang C., Yang D., Wang X., Wu C., Zhou Z., and Cheng H.. 2005. Ca2+ sparks and secretion in dorsal root ganglion neurons. Proc. Natl. Acad. Sci. USA. 102:12259–12264. 10.1073/pnas.0408494102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patton C., Thompson S., and Epel D.. 2004. Some precautions in using chelators to buffer metals in biological solutions. Cell Calcium. 35:427–431. 10.1016/j.ceca.2003.10.006 [DOI] [PubMed] [Google Scholar]
- Quallo T.; Gentry C, Bevan S, Broad LM, Mogg AJ . 2015. Activation of transient receptor potential ankyrin 1 induces CGRP release from spinal cord synaptosomes. Pharmacol Res Perspect. 3(6):e00191. 10.1002/prp2.191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shang S., Zhu F., Liu B., Chai Z., Wu Q., Hu M., Wang Y., Huang R., Zhang X., Wu X., et al. 2016. Intracellular TRPA1 mediates Ca2+ release from lysosomes in dorsal root ganglion neurons. J. Cell Biol. 215:369–381. 10.1083/jcb.201603081 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vasko M.R., Campbell W.B., and Waite K.J.. 1994. Prostaglandin E2 enhances bradykinin-stimulated release of neuropeptides from rat sensory neurons in culture. J. Neurosci. 14:4987–4997. 10.1523/JNEUROSCI.14-08-04987.1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y.Y., Chang R.B., Waters H.N., McKemy D.D., and Liman E.R.. 2008. The nociceptor ion channel TRPA1 is potentiated and inactivated by permeating calcium ions. J. Biol. Chem. 283:32691–32703. 10.1074/jbc.M803568200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ye W., Tu Y.H., Cooper A.J., Zhang Z., Katritch V., and Liman E.R.. 2018. Activation Stoichiometry and Pore Architecture of TRPA1 Probed with Channel Concatemers. Sci. Rep. 8:17104 10.1038/s41598-018-35435-y [DOI] [PMC free article] [PubMed] [Google Scholar]