Li and Wang present an updated overview of ribosome heterogeneity and its functional implications in selective mRNA translational control in stem cells and development.
Abstract
Translation control is critical to regulate protein expression. By directly adjusting protein levels, cells can quickly respond to dynamic transitions during stem cell differentiation and embryonic development. Ribosomes are multisubunit cellular assemblies that mediate translation. Previously seen as invariant machines with the same composition of components in all conditions, recent studies indicate that ribosomes are heterogeneous and that different ribosome types can preferentially translate specific subsets of mRNAs. Such heterogeneity and specialized translation functions are very important in stem cells and development, as they allow cells to quickly respond to stimuli through direct changes of protein abundance. In this review, we discuss ribosome heterogeneity that arises from multiple features of rRNAs, including rRNA variants and rRNA modifications, and ribosomal proteins, including their stoichiometry, compositions, paralogues, and posttranslational modifications. We also discuss alterations of ribosome-associated proteins (RAPs), with a particular focus on their consequent specialized translational control in stem cells and development.
Introduction
Protein and mRNA amounts are poorly correlated, indicating the importance of posttranscriptional regulation (Khan et al., 2013). Recently, translational control has been found to play a dominant role in the regulation of gene expression. Particularly, during dynamic cell fate transitions, such as embryonic development or responses to internal and external stimuli, translation control enables cells to quickly respond by directly adjusting protein abundance before a new chromatin state and transcriptional network form. The chromatin state and many critical mRNAs of embryonic stem cells (ESCs) are stringently subjected to translational control in maintaining self-renewal and pluripotency as well as controlling developmental signaling pathways, cellular differentiation, and embryonic development across many species (Bulut-Karslioglu et al., 2018; Fujii et al., 2017; Kondrashov et al., 2011; Lee et al., 2013b; Slaidina and Lehmann, 2014).
Ribosomes are complex macromolecular assemblies that are the central sites for translation in cells. Using mRNA as a template, one ribosome reads each codon of the mRNA and pairs the codon with the appropriate amino acid provided by an aminoacyl-tRNA to form a polypeptide chain. The ribosome was viewed as a ubiquitous, invariant entity serving for mRNA translation without alterations or specific regulatory functions. However, recent studies have shown ribosome heterogeneity and specialized functions of ribosomes in controlling selective subsets of mRNAs (Xue et al., 2015; Ferretti et al., 2017; Locati et al., 2017a; Shi et al., 2017; Simsek et al., 2017). The ribosome consists of two major components, the small ribosomal subunit (SSU), containing the decoding center to read mRNA and monitor the complementarity between tRNA and mRNA, and the large subunit (LSU), responsible for peptide bond formation and containing three binding sites for tRNA and polypeptide exit tunnel. Each subunit consists of specific ribosomal RNA (rRNA; SSU: 18S; LSU: 5.8S, 28S, and 5S) and ribosomal proteins (RPs; SSU: ∼34 RPs, known as RPS proteins; LSU: ∼51 RPs, known as RPL proteins; Braschi et al., 2019; Baßler and E. Hurt, 2019; Klinge and Woolford, 2019; Fig. 1). Variations in these components and their assemblies can give rise to heterogeneity in ribosomes, and heterogeneous ribosomes could function as key regulatory players in the translational control of stem cells and development.
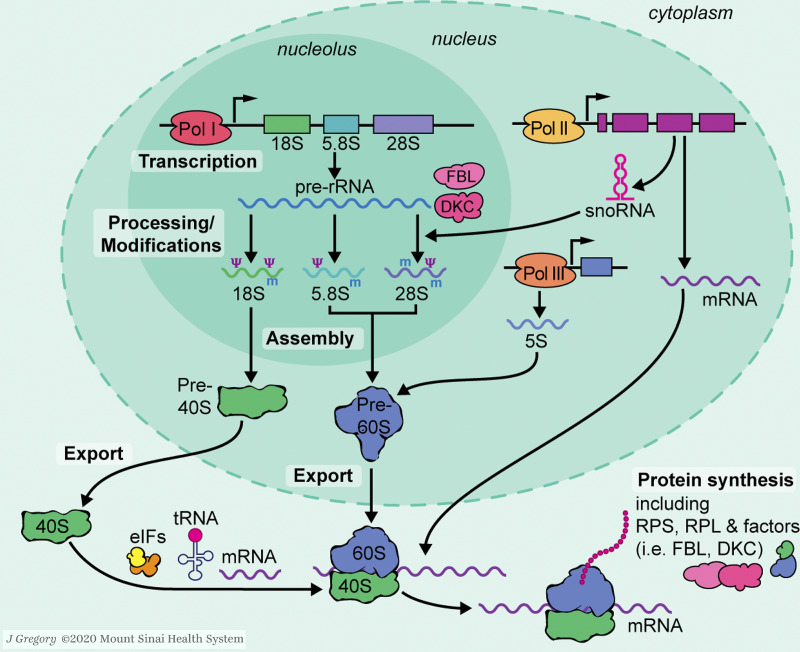
Figure 1.
Schematic diagram of ribosome biogenesis. In the nucleolus, RNA Pol I transcribes a polycistronic pre-rRNA, which is subsequently processed and modified into mature rRNAs (18S, 5.8S, and 28S). Another rRNA, 5S, is transcribed by RNA Pol III in the nucleoplasm. RNA Pol II transcribes snoRNAs (which aid the maturation of pre-rRNA) and mRNAs in the nucleus. mRNAs are transported to the cytoplasm and then translated by ribosomes. RPs and assembly factors (such as FBL and DKC) are also products of mRNA translation. RPs can be reimported to the nucleus to assemble pre40S/60S with rRNAs, which are then exported to the cytoplasm and matured to function in mRNA translation. Illustration by Jill K. Gregory.Fig. 1 is reprinted with permission from Mount Sinai Health System.
Historically, the idea of heterogeneous ribosomes with specific functions originated from the earlier “ribosome filter hypothesis” (Mauro and Edelman, 2002). This hypothesis states that ribosome heterogeneity enables specific mRNA–rRNA and mRNA–RP interactions, resulting in differential rates of mRNA translation. Subsequently, a “ribosome code,” analogous to the “histone code” that regulates transcription, was proposed based on the discovery that different RP paralogs have distinct roles in specific translation regulation (Komili et al., 2007). The tenet of both theories is that heterogeneous ribosome composition confers specific translation regulation, providing another level of complexity in the regulation of gene expression. Based on this tenet, we discuss what constitutes the ribosomal heterogeneity (Fig. 2) and how this heterogeneity regulates mRNA translation in stem cells and development.
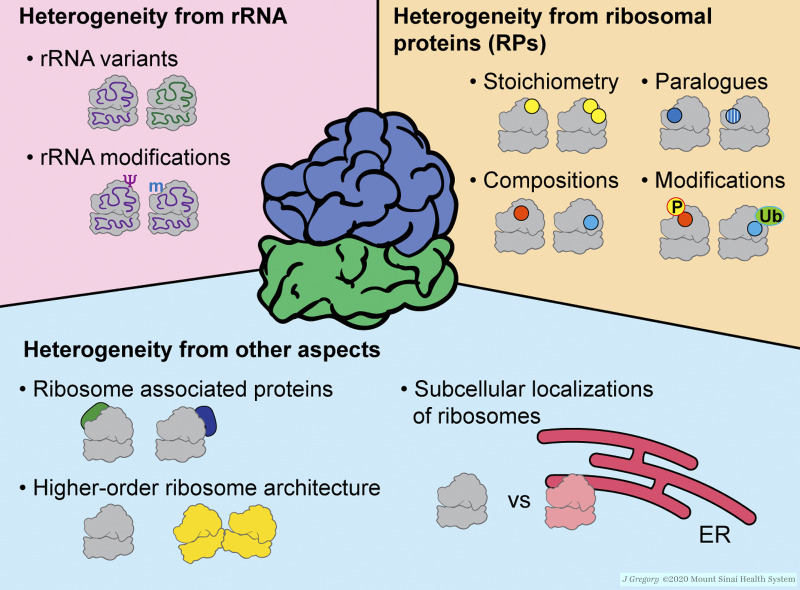
Figure 2.
Components that contribute to ribosome heterogeneity. Ribosome heterogeneity can arise from rRNA, RPs, and other aspects. The details of each component, including examples with their consequent specialized translation and functions, are described in the main text. Illustration by Jill K. Gregory.Fig. 2 is reprinted with permission from Mount Sinai Health System.
In this review, we start with the introduction of global translation control in stem cell differentiation, followed by a discussion on our current knowledge on heterogeneous ribosomes and their specialized translation control in stem cells and development, with a particular focus on vertebrates.
Global translation control in stem cell differentiation
Stem cells require a low global protein synthesis rate to maintain their overall homeostasis. During ESC differentiation into embryoid bodies (EBs), polysome content and translation efficiency are elevated on a genome-wide scale (Sampath et al., 2008; Ingolia et al., 2011). A similar increase upon differentiation is reported in other stem cell systems. Hematopoietic stem cells (HSCs) have a significantly lower protein synthesis rate than most other hematopoietic cells. This low rate of protein synthesis is tightly controlled and is essential for the maintenance of HSCs, as a modest increase or decrease in protein synthesis is sufficient to impair HSC function (Signer et al., 2014). Similar observations have been reported in various tissue-specific stem cell maintenance and differentiation systems, such as hair follicle stem cells (Blanco et al., 2016), muscle stem cells (satellite cells; Zismanov et al., 2016), neural stem cells (Bonaguidi et al., 2011), germline stem cells (GSCs; Sanchez et al., 2016), hepatocytic differentiation (Parent and Beretta, 2008), and spermiogenesis (Yanagiya et al., 2010).
Interestingly, while global translation increases in early differentiation, it decreases in later differentiation. For instance, during GSC differentiation in Drosophila, differentiating germ cells consistently present increased protein synthesis rates from GSCs to 2-cell cysts, reaching the maximum at 4-cell cysts. However, after 4-cell cysts, protein synthesis rates decrease, and 16-cell cysts have an even lower rate than GSCs (Sanchez et al., 2016). Similar observations have been made in human ESC (hESC)-to-cardiomyocyte differentiation (Pereira et al., 2019), mouse neural stem cell differentiation (Baser et al., 2019), the neural progenitor differentiation in mouse forebrain (Chau et al., 2018), and mouse ESC (mESC)-EB differentiation (You et al., 2015). Consistent with these observations, despite globally increased protein synthesis during early differentiation, both RPs (Ingolia et al., 2011) and rRNA synthesis (Savić et al., 2014; Woolnough et al., 2016) are repressed, foreshadowing the reduced protein synthesis in later differentiation. Altogether, these studies show that protein synthesis is dynamically controlled during development and demonstrate that translational control is a key modulator of stem cell maintenance and differentiation.
rRNA heterogeneity and specialized functions
rRNA accounts for >55% of the mass of ribosomes and composes around 80% of total RNA in most cells (Blanco and Blanco, 2017). RNA Polymerase (Pol) I, in the nucleolus, mediates transcription of a single rRNA precursor (prerRNA), which is subsequently cleaved into 18S, 5.8S, and 28S rRNA. On the other hand, 5S rRNA is transcribed by RNA Pol III in the nucleoplasm (Lafontaine, 2015; Fig. 1).
Ribosomal DNA (rDNA) genes encode rRNAs and comprise hundreds of transcription units consisting of tandem repeats. Due to their large copy numbers and the highly repetitive nature of rDNA genes, reliable sequencing and a comparison among different species and tissues are challenging. Recent advances in sequencing technologies and bioinformatics analyses have improved the accuracy and consistency of the sequencing mapping to rDNA loci, revealing the extensive variation of rDNA copy numbers across individuals in many species, including human and mouse (Parks et al., 2018). In the human genome, 45S rDNAs are clustered in nucleolus organizer regions, which are located in five chromosomes (chromosomes 13, 14, 15, 21, and 22). The diploid copy number of the 45S rDNA varies from ∼60 to >800 units. The 5S rDNA array resides on chromosome 1 and the diploid copy number ranges from ∼10 to >400 units (Yu and Lemos, 2016). Analysis of copy-number variation in normal tissues and tumors of human suggests that most cancers are subjected to coupled 5S rDNA amplification and 45S rDNA loss, which are associated with increased cell proliferation rates and nucleolar activity (Wang and Lemos, 2017). Analysis of rRNA, the product of rDNA transcription, shows a reduced transcription in the early ACTIVIN A–induced hESC differentiation (48 h; Woolnough et al., 2016) and in mouse NPC differentiation (Savić et al., 2014), whereas another study shows an approximate 20% increase in 5 day–differentiated EBs compared with mESCs (Sampath et al., 2008). Additionally, HTATSF1 (HIV-1 Tat-specific factor 1), a nucleic acid–binding protein that functions as a cofactor for the stimulation of transcriptional elongation, affects rRNA transcription and processing to modulate overall protein synthesis in controlling ESC pluripotency and differentiation (Corsini et al., 2018). Furthermore, beyond its simple expression level change during differentiation, rRNA modulation can also change the cell fate, growth, and proliferation, which is shown in the study of female Drosophila GSCs and their daughters (Zhang et al., 2014) and of mESCs (Watanabe-Susaki et al., 2014). These results indicate that the copy-number variation of rDNA, together with the expression level difference of its product, rRNA, exerts regulatory effects in different cell types.
After transcription, rRNAs are modified and assembled with RPs to form preribosomal subunits, which are then exported from the nucleus to the cytoplasm (Fig. 1). The steps involved in these processes can specify alternative rRNA molecules into ribosomes, changing the functional specificity of the translation machinery and providing opportunities that ribosome heterogeneity can arise.
Heterogeneity from rRNA variants
Alternative rRNA molecules have been widely reported in archaea, bacteria, and simple eukaryotes (Kim et al., 2008; Yano et al., 2013; López-López et al., 2007; Dimarco et al., 2012; Gunderson et al., 1987). Recent studies have also discovered rRNA heterogeneity in organisms with higher complexity. Humans and mice exhibit vast rDNA variations both across and within individuals (Parks et al., 2018). Specifically in mice, rRNA variants are differentially expressed between tissues, and an analysis indicates that their tissue-specific expression is conserved in humans (Parks et al., 2018; Tseng et al., 2008). These results suggest that rRNA variants can be developmentally regulated. Indeed, in zebrafish, there are at least two rRNA systems comprising distinct subtypes of all four rRNA species. There are maternal-type and somatic-type 5S and 45S rDNA transcription units at different chromosomal locations. Between two developmental subtypes, all four mature rRNA species have sequence differences. During oogenesis, maternal-type rRNAs are expressed and accumulated, and these rRNAs are replaced by the somatic-type rRNAs during embryogenesis (Locati et al., 2017a, 2017b). In silico analysis indicates that 5′ UTRs of maternally expressed mRNAs preferentially bind to certain regions present in the maternal-type 18S rRNA, whereas somatic-type 18S rRNA has a better affinity for maternally nonexpressed mRNAs (Locati et al., 2017b; Fig. 3 A).
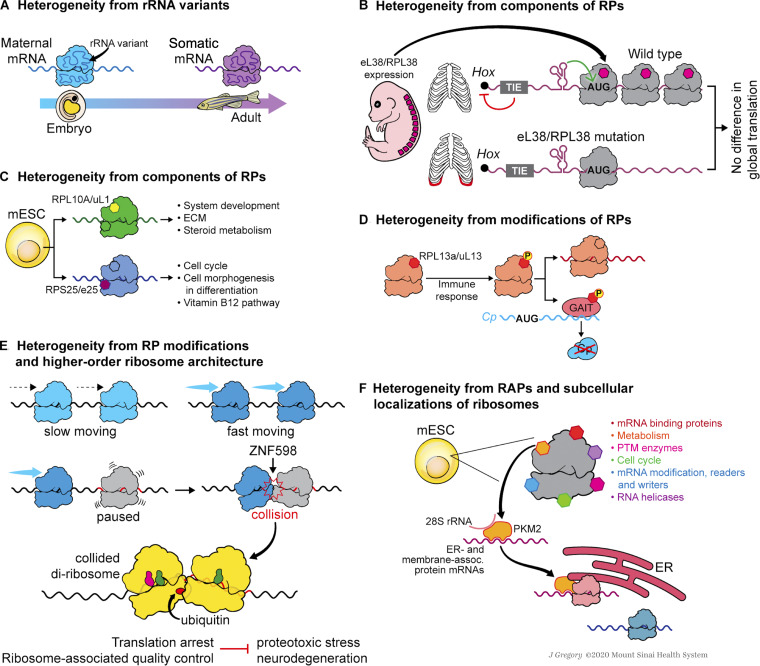
Figure 3.
Ribosome heterogeneity in stem cells and development. (A) During zebrafish embryogenesis, ribosomes switch from a maternal-type rRNA variant to a somatic-type rRNA variant; the former variant has a better affinity for maternal mRNAs, while the latter one prefers maternally nonexpressed mRNAs. (B) RPL38/eL38 is important for the translation of Hox mRNAs, which are necessary for axial skeletal patterning. Within the 5′ UTRs of these Hox mRNAs, a translation inhibitory element (TIE) blocks the canonical cap-dependent translation, and a structured IRES element promotes the recruitment of RPL38/eL38-containing ribosomes. (C) In mESCs, ribosomes containing RPL10A/uL1 or RPS25/eS25 participate in the translation of mRNAs involved in specific cellular pathways. (D) Upon the innate immune response, RPL13A/uL13 is phosphorylated and released from the ribosome. The released phosphorylated RPL13A/uL13 is incorporated into GAIT complex to inhibit the translation of specific inflammation-related ceruloplasmin (Cp) mRNA. (E) The ubiquitin ligase ZNF598 transfers ubiquitin to RPs of the collided diribosomes. Such ubiquitylation is required for translation arrest and the activation of ribosome-associated quality control, the failure of which leads to proteotoxic stress and neurodegeneration. (F) In mESCs, PKM2, as a RAP, is enriched on ER-associated ribosomes and binds mRNAs of ER-associated and membrane-associated proteins to promote their translation. Illustration by Jill K. Gregory.Fig. 3 is reprinted with permission from Mount Sinai Health System.
Heterogeneity from rRNA modifications
rRNA is extensively modified posttranscriptionally, adding another regulatory layer to its heterogeneity. Considering the extensive interactions between rRNA and various proteins, rRNA modifications are potentially able to modulate the ribosome assembly and fine-tune its specificity in translation (Sharma and Lafontaine, 2015; Sloan et al., 2017). The most common rRNA modifications are ribose 2′-O methylation (2′-O-Me) and pseudouridylation (isomerization of uridine to 5-ribosyl isomer, pseudouridine, ψ). Other identified rRNA modifications include acetylation and methylation of the nucleotide base. Most of these modifications are constitutively present in very conserved regions, important for the maintenance of ribosome structure and function (Decatur and Fournier, 2002; Natchiar et al., 2017). The identification of sites amenable to partial (or substoichiometric) rRNA modification highlights rRNA modifications as a major source of ribosome heterogeneity (Birkedal et al., 2015; Taoka et al., 2016). Furthermore, rRNA modifications are mostly guided by small nucleolar RNAs (snoRNAs), which show tissue-specific expression, providing another source for ribosome heterogeneity (Castle et al., 2010).
Mounting evidence has linked changes in rRNA modification, including defects in the regulators of rRNA modification, to developmental regulation in multiple species. For example, loss of function of three snoRNAs in zebrafish reduces the snoRNA-guided methylation of the target nucleotides in rRNA, and the compromised rRNA modification, even at a single site, causes profound morphological defects and embryonic lethality (Higa-Nakamine et al., 2012). The snoRNA-guided methylation on rRNA is regulated by NPM1, which directly binds snoRNAs and the rRNA 2′-O-methyltransferase fibrillarin (FBL). With unchanged snoRNA abundance and FBL levels, the depletion of NPM1 in human erythroleukemia cells decreases 2′-O-Me levels and some internal ribosome entry site (IRES)–translated proteins. Its depletion also reduces colony-formation potential and increases erythroid differentiation. Moreover, Npm1 inactivation in mouse adult HSCs leads to bone marrow failure. These results suggest the important functions of NPM1-mediated 2′-O-Me in HSC maintenance and erythroid development (Nachmani et al., 2019). Pseudouridylation of rRNA is also reported to have functional importance. Impaired rRNA pseudouridylation by mutations in the Dkc1 gene that encodes dyskerin, a pseudouridine synthase, perturbs the translation of mRNAs containing IRES elements in mice and human cells (Yoon et al., 2006). The catalytically impaired DKC1 leads to defects in ribosome activity with decreased translational fidelity and IRES-dependent translational initiation (Jack et al., 2011). Likewise, the catalytic activity of dyskerin is also required for HSC differentiation (Bellodi et al., 2013). In addition, the alterations in these rRNA modifications have been widely characterized in cancers and other conditions (Dimitrova et al., 2019; Kadumuri and Janga, 2018; Huang et al., 2020; Tusup et al., 2018). A systematic mapping of rRNA 2′-O-Me in paired cancer cell lines (HCT116 p53 +/+ and −/−) identified 106 modified sites on mature rRNAs and revealed only some of these 2′-O-Me sites are vulnerable to variation. Interestingly, these vulnerable sites lie peripherally on ribosomal subunits while other sites are present at the core, which are made more robustly. These results indicate that those vulnerable sites of rRNA 2′-O-Me are subject to specific regulation in cancers (Sharma et al., 2017; Natchiar et al., 2018).
Of note, studies using either qRT-PCR (Sampath et al., 2008; Corsini et al., 2018) or northern blots (Zhang et al., 2014) to measure rRNA levels with certain simple rRNA probes only show the change of rRNA abundance without focusing on its heterogeneity (i.e., rRNA variants or modifications), which overlooked the heterogeneous nature of the changed rRNA population. Thus, rRNA expression level changes, such as during stem cell differentiation and development, may potentially reflect the change of rRNA heterogeneity (i.e., rRNA variants or modifications) in these processes. Additionally, in most cases, it remains unclear whether these rRNA variants and rRNAs with changed modifications are incorporated into translationally active ribosomes or not. All of these warrant the additional scrutiny on functional significance of such heterogeneity in these processes.
RP heterogeneity and specialized functions
RPs extensively interact with rRNAs, making up the ribosomal subunits involved in the translation process. Compared with rRNA, the variance in RPs is more complex. The eukaryotic ribosome has 70–80 RPs with various RP composition and stoichiometry of specific RPs in individual ribosomes, and some RPs have paralogous genes. RPs also have distinct posttranslational modifications (PTMs). Variance from all these elements and the small sizes of many RPs (median of ∼150 amino acids) challenge accurate quantification of candidate RPs with mass spectrometry–based proteomics methods. Also, ribosomal subunit fractions contain immature ribosome biogenesis particles, and there are free RPs not assembled into ribosomes, which complicate the RP-related analysis. The development of technologies has enabled more advanced quantitative strategies, and many of these strategies have been employed to study RPs, extending our knowledge of translational control exerted by heterogeneous RPs in stem cells and development (Peterson et al., 2012; Slavov et al., 2015; Shi et al., 2017; Simsek et al., 2017).
Heterogeneity from stoichiometry of RPs
Various expression levels of different RPs are reported in multiple human tissues and some regions of the mouse embryo (Bortoluzzi et al., 2002; Kondrashov et al., 2011). For example, some ribosomal subunits were identified as stem cell markers due to their highly enriched expression levels in mESCs (e.g., Rps2/uS5 and Rpl13/eL13; Sharov et al., 2003), hESCs (e.g., RPL13A/uL13; Richards et al., 2004), and inner cell mass of human blastocysts (e.g., RPL14/eL14, RPL7A/eL8, RPL19/eL19, and RPL32/eL32; Adjaye et al., 2005). In different tissues and developmental conditions, cells can alter the stoichiometry of RPs assembled into the ribosome to modify its translation specificity. RP stoichiometry in ribosomes was identified in bacteria and yeast, which shows that one or more copies of certain RPs are present in the large ribosomal subunit and that substoichiometry of these RPs sensitizes yeast to cold temperature and affects the translation of certain mRNAs (Ballesta and Remacha, 1996; Remacha et al., 1995). In mESCs, quantification of RPs between monosome and polysome fractions using tandem mass tags mass spectrometry revealed a low polysome-to-monosome ratio, the characteristic feature of ESCs (Slavov et al., 2015). The examination of each RP revealed differential stoichiometry among mouse RPs in monosomes and polysomes. The stoichiometry among RPs also depends on the number of ribosomes bound per mRNA and the growth conditions. These results also indicated that the differential RP stoichiometry and related biological functions are conserved between mice and humans. Subsequently, an absolute protein quantification using selected reaction monitoring–based proteomics strategy was employed to determine the exact RP stoichiometry in mESCs (Shi et al., 2017). This quantification included 15 RPs, among which 4 RPs (RPS7/eS7, RPS25/eS25, RPL10A/uL1, and RPL38/eL38) were identified as significantly substoichiometric in polysomes. A comparison of the RP stoichiometry in polysomes to free ribosomal subunits identified some RPs with a higher (RPS26/eS26, RPL10/uL16, RPL38/eL38, and RPL40/eL40) or lower (RPS7/eS7, RPS25/eS25, and RPL10A/uL1) abundance in polysomes compared with free subunits (Shi et al., 2017). During EB differentiation, RPS18/uS13, RPL7A/eL8, and RPL36A/eL42 protein levels are originally high in mESCs, transiently decrease upon differentiation, and increase again later (Corsini et al., 2018). The differential RP expression levels may also be important for extraribosomal functions, as will be discussed later.
Heterogeneity from compositions of RPs
Various compositions of RPs are one important source of ribosomal heterogeneity. Dysregulation of RP composition affects translation and leads to developmental defects.
For instance, in zebrafish, an individual knockdown of 21 RP genes revealed that depletion of 19 RPs caused obvious developmental deformities, with varying degrees of abnormality in the eyes, ears, brain, and body trunk (Uechi et al., 2006). In mice, deficiency in RPL24/eL24 leads to down-regulation of protein synthesis as well as cellular proliferation and causes physical anomalies, including the kinked tail and preaxial polydactyly (extra digits; Oliver et al., 2004). Mechanistically, variations in RP composition affect the translation specificity and consequently elicit relevant cellular phenotypes. Translational preference of ribosomes with different RP compositions remains an area of active research, and some recent studies have addressed RP-specific translational regulation in stem cells and development.
One example is RPL38/eL38. Deletion in the Rpl38/eL38 gene is identified as the causative mutation for Tail short (Ts) hemizygous mice. The Rpl38/eL38 mutant mice exhibit skeletal patterning defects with the presence of 14 instead of 13 ribs, suggesting that the first lumbar vertebra is transformed anteriorly into a thoracic vertebra. In Rpl38/eL38 mutant embryos, global translation is unchanged, but the translation of a select subset of Homeobox (Hox) mRNAs, the critical regulators controlling axial skeletal morphology, is impaired (Kondrashov et al., 2011). In studying the RNA sequences within the 5′ UTRs of these Hox mRNAs, two RNA regulons were uncovered: a translation inhibitory element (near the 5′ cap) that blocks the canonical cap-dependent translation and a structured IRES element (near the main start codon) that facilitates the recruitment of ribosomes containing RPL38/eL38 to initiate translation. Such two-pronged translational regulation confers specific controls in embryonic development by using specialized ribosomes (Xue et al., 2015; Fig. 3 B).
Similar to the preference of RPL38/eL38-containing ribosomes to certain IRES RNA elements, the ribosomes carrying RPS25/eS25 are also required for translation initiation of multiple classes of viral IRESs, such as the hepatitis C virus IRES and the cricket paralysis virus intergenic region IRES (Landry et al., 2009; Hertz et al., 2013). A chemical cross-linking study revealed the strong interaction between RPS25/eS25 and cricket paralysis virus intergenic region IRES (Nishiyama et al., 2007), and a cryoelectron microscopy study showed the direct contact of RPS25/eS25 to hepatitis C virus IRES (Quade et al., 2015), suggesting that RPS25/eS25 can recognize certain IRES elements and control the relevant translation initiation. To identify mRNAs bound by RPS25/eS25-containing ribosomes in mESCs, “RPS25/eS25-Ribo-seq” was performed by a ribosome profiling protocol using FLAG-immunoprecipitation of cellular extracts from a knock-in mESC line harboring an endogenously tagged Rps25/eS25-3xFLAG allele (Shi et al., 2017). The results showed that the bound transcripts are enriched in specific cellular processes, such as the cell cycle, cell morphogenesis in differentiation, and vitamin B12 pathway. Shi et al. also performed “RPL10A/uL1-Ribo-seq” and found that in mESCs, RPL10A/uL1-containing ribosomes preferentially translate distinct subpools of mRNAs, including those important for system development and extracellular matrix organization. They further showed that RPL10A/uL1-containing ribosomes can regulate specific translation through IRES elements (Shi et al., 2017; Fig. 3 C).
Besides the RPs discussed, there are other RPs that are involved in IRES-mediated translation. In mouse erythroblasts, depletion of Rps19/eS19 or Rpl11/uL5 affects the IRES-dependent translation of Csde1 and Bag1. The lacking of Bag1 is embryonic lethal, and low Csde1 expression inhibits erythroid proliferation and differentiation (Horos et al., 2012). Another example is RACK1, an RP of the 40S ribosomal subunit. In Drosophila, RACK1 is ubiquitously expressed in developing embryos, and its loss is embryonic lethal (Kadrmas et al., 2007). In mice, homozygous deletion of Rack1 leads to embryonic lethality at gastrulation, while heterozygous mice are viable, but with skin pigmentation defects (Volta et al., 2013). RACK1 is essential for IRES-dependent translation in Drosophila and in human hepatoma cells (Majzoub et al., 2014). Future studies should look into the mechanisms underlying RACK1-mediated IRES-dependent translation in embryonic development. Apart from IRES-mediated cap-independent translation, viral transcripts can also be translated in a cap-dependent manner by specialized ribosomes through recognition of other cis-regulatory RNA motifs. For example, RPL40/eL40 recognizes specific cis-regulatory determinant in vesicular stomatitis virus mRNAs, whose translation is activated in a cap-dependent mode (Lee et al., 2013a). The Kozak sequence is another cis-regulatory RNA motif that can be preferentially recognized by specialized ribosomes. This sequence is well known to be important in the translation initiation (Kozak, 1986). The mRNAs with full Kozak consensus are recognized by RPS26/eS26-containing ribosomes for translation initiation, while the mRNAs with less conserved Kozak sequence are translated by RPS26/eS26-depleted ribosomes (Ferretti et al., 2017). Such specialized translation regulation is consistent with the finding that Rps26/eS26 resides upstream of the translation initiation site and directly interacts with the residues before the start codon (Pisarev et al., 2008). In humans, RPS26/eS26 is frequently mutated in Diamond–Blackfan anemia (Boria et al., 2010) and the study in yeast shows that the accumulation of Rps26/eS26-deficient ribosomes is part of the response to multiple stresses (Ferretti et al., 2017). The relevant functions of RPS26/eS26 in distinct tissues and developmental stages need further investigation. Moreover, because some RPs have been shown to have some extra-ribosomal functions (Warner and McIntosh, 2009; Zhou et al., 2015), further work such as the identification of specific RP on the ribosome is necessary to properly ascribe the phenotypes caused by RP dysregulation to specialized ribosome-mediated translational control. Otherwise, extraribosomal roles of RPs cannot be ruled out.
Heterogeneity from paralogs of RPs
The presence of paralogous genes of RPs produces more complexity to RP heterogeneity. Paralogues of RP coding genes widely exist in bacteria, yeast, and plants. In contrast, most mammalian genes coding for RPs are represented in a single copy, with only a few exceptions. One exception is RPL22/eL22. In mice, RPL22/eL22 can repress the expression of its paralog, Rpl22/eL22-like 1 (Rpl22l1/eL22l1), by binding to an internal hairpin structure in Rpl22l1/eL22l1 mRNA and destabilizing it. In the Rpl22/eL22−/− mice, Rpl22l1/eL22l1 has a significantly compensatory increase in its expression and RPL22L1/eL22L1 can be incorporated into ribosomes (O’Leary et al., 2013). Although RPL22/eL22 and RPL22L1/eL22L1 share >70% identical amino acid sequence, there are differences in their developmental functions and organismal phenotypes. For example, Rpl22/eL22−/− mice are phenotypically normal except for specific defects in T and B cell development (Anderson et al., 2007), whereas Rpl22l1/eL22l1 deficiency is embryonic lethal (Zhang et al., 2017), indicating that they cannot fully compensate for each other’s roles. Moreover, RPL22/eL22 and RPL22L1/eL22L1 can perform extraribosomal functions during gastrulation, through antagonistically controlling the splicing of Smad2 pre-mRNA (Zhang et al., 2017). They also play distinct and antagonistic roles in hematopoiesis by binding to Smad1 mRNA with opposing effects on Smad1 expression (Zhang et al., 2013). Further studies are needed to find out whether these posttranscriptional regulations are through ribosomal or extraribosomal effects.
Another example is RPS4/eS4. In humans, there are three genes encoding RPS4/eS4, one (RPS4X/eS4X) on the X chromosome and the other two (RPS4Y1/eS4Y1 and RPS4Y2/eS4Y2) on the Y chromosome (Fisher et al., 1990; Skaletsky et al., 2003; Lopes et al., 2010). RPS4X/eS4X and RPS4Y1/eS4Y1 are ubiquitously expressed, while RPS4Y2/eS4Y2 is specifically expressed in testis and prostate (Lopes et al., 2010). The polypyrimidine tract characterizes the promoters of the constitutively expressed RP genes, including the promoter of RPS4Y1/eS4Y1. This tract is disrupted in RPS4Y2/eS4Y2, which may explain the loss of its constitutive expression. Comparative structure modeling showed very similar structures of the three proteins with a unique hydrogen bond in RPS4Y2/eS4Y2 between its central and C-terminal domains. The C-terminal domain is more exposed at the ribosome surface, and this difference may enable the interaction of RPS4Y2/eS4Y2 with different RPs or extraribosomal factors, exerting testis-specific functions (Lopes et al., 2010). A paralog of RPS4/eS4 exhibiting testis-prominent expression is also identified in mice, known as Rps4/eS4-like, but it is an autosomal gene while the mouse Rps4/eS4 is located on X chromosome as human RPS4X/eS4X (Sugihara et al., 2013). The expression pattern of all RPs and their paralogs across 22 different mouse tissues indicates that expression levels of RP paralogs have a much higher variability across tissues than other RPs (Wong et al., 2014). The results in both mouse and human cell lines also indicate that RP paralogs evolved more recently to additionally provide a greater level of gene expression control (Guimaraes and Zavolan, 2016; Wong et al., 2014).
Heterogeneity from modifications of RPs
PTMs of RPs introduce another layer of heterogeneity to ribosomes. It is a relatively simple way to modify ribosomes compared with energy-consuming de novo protein synthesis and assembly of ribosomes with new RPs. PTMs are covalent modifications that can change the structure, stability, subcellular localization, interacting partners, and functions of modified RPs and consequently contribute to translational control in stem cells and development. Proteomics studies have identified numerous PTMs on RPs, including phosphorylation, ubiquitination, methylation, acetylation, and hydroxylation (Simsek and Barna, 2017).
The phosphorylation of mammalian RPS6/eS6 is the first inducible PTM identified on ribosomes, which was discovered in 1974 from rat liver samples (Gressner and Wool, 1974). RPS6/eS6 phosphorylation is downstream of cellular signal pathways that respond to multiple external stimuli, including the PI3K and mTOR pathways (Meyuhas, 2015). Physiological or pharmacological stimuli in mice that activate neurons result in an increase of RPS6/eS6 phosphorylation, which led to the use of phosphorylated RPS6/eS6 as a marker of activated neurons (Kelleher et al., 2004; Knight et al., 2012). Although RPS6/eS6 phosphorylation is involved in multiple growth signals and stimuli, Rps6/eS6 phosphorylation-deficient mice are viable with only subtle, tissue-specific phenotypes (Ruvinsky et al., 2005, 2009). The second example of phosphorylated RP is RPL13A/uL13, identified from human monocytic cells. RPL13A/uL13 is phosphorylated upon the innate immune response, and this PTM leads to the release of RPL13A/uL13 from the ribosome (Mukhopadhyay et al., 2008). Released phosphorylated RPL13A/uL13 has an extraribosomal function as part of the GAIT complex to bind and inhibit the translation of specific inflammation-related mRNAs (Mazumder et al., 2003; Kapasi et al., 2007; Fig. 3 D). Other examples of RP phosphorylation include RPL12/uL11 phosphorylation regulating translation of mitosis-related transcripts in human proliferating cells (Imami et al., 2018), RPS15/uS19 phosphorylation important for the neurotoxicity phenotype in Parkinson’s disease (Martin et al., 2014), and RACK1 phosphorylation induced upon poxvirus infection (Jha et al., 2017).
Another extensively studied PTM on ribosomes is ubiquitylation, whereby a ubiquitin is covalently attached to a substrate protein singularly (monoubiquitylation) or in long chains (polyubiquitylation). Ubiquitin is a highly conserved small peptide and is highly enriched in cells, representing up to 5% of the total cellular protein content (Park and Ryu, 2014). Interestingly, RPs are important sources for such a big ubiquitin pool. In both humans and mice, there are four genes encoding ubiquitin: UBB and UBC encoding polyubiquitin precursor with ubiquitin repeats and RPS27A/eS31 and UBA52 encoding ubiquitin as in-frame fusions to RPS27A/eS31 and RPL40/eL40, respectively (Park and Ryu, 2014). These RP-ubiquitin fusions (ubiquitin-S27A/L40) are identified across the full breadth of eukaryotic diversity (Sibbald et al., 2019). Ubiquitin can be posttranslationally cleaved from RP–ubiquitin fusions. An early study shows that the transient association between these ubiquitin molecules and their originally fused proteins can promote their incorporation into the nascent ribosome to adjust the ribosome structure and is required for efficient ribosome biogenesis, suggesting specific roles of the ubiquitin generated from RP-ubiquitin fusions (Finley et al., 1989). Consistent with this indication, a recent study shows that the deficiency of the RPL40/eL40–ubiquitin fusion gene UBA52 does not change the global ubiquitin level but decreases the ubiquitylation of ribosomes, which is required for efficient translation. Consequently, the deficiency of Uba52 in mice is embryonic lethal (Kobayashi et al., 2016).
Besides being sources to ubiquitin, RPs can also be ubiquitylated as substrates. Ubiquitylation of RPs can mark them for degradation by the proteasome (degradative ubiquitylation) or alter their functions (regulatory ubiquitylation). Excess RPs that are not assembled into ribosomes can be polyubiquitylated and degraded. This regulation is very important for proper ribosome functions, as RPs are usually expressed at high levels beyond what is actually needed (Sung et al., 2016; Lam et al., 2007). During erythroid differentiation, the expression of UBE2O, a hybrid E2 (ubiquitin-conjugating)–E3 (ubiquitin ligase) enzyme, is induced in reticulocytes. UBE2O ubiquitinates some RPs for degradation, driving the transition from a complex to a simple proteome, which is needed for the transformation of reticulocytes into erythrocytes (Nguyen et al., 2017; Yanagitani et al., 2017). Thus, UBE2O is very important in the differentiation of red blood cells, and its deficiency in mice causes anemia (Nguyen et al., 2017). Moreover, several RPs have been shown to undergo regulatory ubiquitylation in response to cellular stresses. For instance, the site-specific regulatory ubiquitylations of RPS2/uS5, RPS3/uS3, and RPS20/uS10, which occur on assembled cytoplasmic ribosomes, are stimulated by activation of the unfolded protein response (UPR), a cellular response to proteotoxic stress in the ER. These regulatory ribosomal ubiquitylations occur on a timescale coincident with eIF2α phosphorylation, which reduces protein synthesis. Defective ubiquitylation of these RPs sensitizes cells to UPR-induced cell death (Higgins et al., 2015). Interestingly, two of these ubiquitylated RPs, RPS3/uS3 and RPS20/uS10, together with an LSU, RPL10/uL16, are found to be ufmylated by UFM1 (ubiquitin-fold modifier 1) in mESCs (Simsek et al., 2017). UFM1 is a recently identified ubiquitin-like modifier. Mice lacking UFL1, a UFM1 E3 ligase, exhibit severe anemia, cytopenia, and death (Zhang et al., 2015), and similarly, mice without UBA5, a UFM1 E1 activating enzyme, die in utero with severe fetal anemia (Tatsumi et al., 2011). The molecular function of this modification remains to be defined, but the overlap of these two modifications, ufmylation and ubiquitylation, on RPS3/uS3 and RPS20/uS10 are very intriguing, indicating their combinatorial presence and potential cooperative function on the same RP.
The transfer of ubiquitin to protein substrates is directly catalyzed by a ubiquitin ligase and regulated by other factors. ZNF598, a ubiquitin ligase, is required for the regulatory ubiquitylation of RPS10/eS10 and RPS20/uS10. RACK1, a 40S RP, regulates the ubiquitylation of RPS2/uS5, RPS3/uS3, and RPS20/uS10 (Sundaramoorthy et al., 2017). The ubiquitylation of RPs regulated by ZNF598 and RACK1 happens on the 40S–40S interface of the collided diribosomes (Juszkiewicz et al., 2018). Diribosomes are usually formed in stalled ribosomes by which a trailing ribosome encounters a slower leading ribosome. A long stretch of adenosine nucleotides, like the poly(A) tail, stall ribosomes and inhibit downstream translation, which requires the regulatory ubiquitylation of certain RPs. These events activate the ribosome-associated quality control (RQC) pathway, which can resolve the stalled ribosomes and recycle ribosomal subunits. This is an evolved failsafe mechanism to protect ribosomes from accidentally translating into the poly(A) tail (Juszkiewicz and Hegde, 2017; Sundaramoorthy et al., 2017; Garzia et al., 2017; Brandman and Hegde, 2016). Hel2, the mammalian homologue of ZNF598 in yeast, can recognize the stalled ribosomes at poly(A) tail as well as the dicodon CGA-CCG, which can efficiently cause ribosomal stalling. It then ubiquitylates RPS20/uS10, directing the ribosome to RQC pathway (Matsuo et al., 2017; Ikeuchi et al., 2019). The failure of this process results in protein aggregation and proteotoxic stress in yeast and neurodegeneration in mice (Shen et al., 2015; Chu et al., 2009; Choe et al., 2016; Yonashiro et al., 2016; Defenouillère et al., 2016; Fig. 3 E). These findings strongly suggest that the structural unit for RQC is a diribosome and indicate that mRNA translation can be regulated, particularly monitored, by a higher-order ribosome architecture, which may serve as another layer of ribosome heterogeneity.
RAP heterogeneity and specialized functions
In addition to rRNAs and canonical core RPs, ribosomes have hundreds of ribosome-associated proteins (RAPs), which tremendously increase the size and complexity of ribosomes and further expand ribosomal heterogeneity and functional diversity on mRNA translation. The structure on ribosomal surface is important for such RAP assembly. The eukaryotic ribosomes have variable expansion segments (ESs) in their rRNAs, some of which resemble tentacle-like extensions exposed on the ribosome surface. ES27L, one of the largest ESs, can recruit nascent peptide processing enzymes that are required for translation fidelity (Fujii et al., 2018). The termini of human ESs 7 and 27 are exposed on the ribosomal surface and contain tandem G-tracts that can form highly stable G-quadruplexes. These G-quadruplexes can bind to RAPs, including helicases (such as DDX3, CNBP, DDX21, and DDX17, all of which are reported to be able to unfold G-quadruplexes) and some heterogeneous nuclear ribonucleoproteins (Mestre-Fos et al., 2019).
Several identified RAPs have been shown to specifically regulate translation of specific subsets of mRNAs. A well-known example is FMRP, the loss of which causes fragile X syndrome. FMRP directly binds to the coding regions of transcripts associated with synaptic signaling and autism spectrum disorders (Darnell et al., 2011), possibly through the specific binding of its RGG motif (arginine-glycine-rich box) to guanine-quadruplex–containing RNAs (Vasilyev et al., 2015). FMRP can also directly interact with RPL5 protein, and this interaction happens near the binding sites for tRNA and translation elongation factors, precluding their incorporation to the ribosome and inhibiting the translation of FMRP targets (Chen et al., 2014). To determine the magnitude and the components of the “ribo-interactome,” an affinity enrichment for endogenously tagged ribosome subunits in mESCs was performed (Simsek et al., 2017). This interactome is composed of >400 proteins, including RPs and translation initiation and elongation factors, which have been expected, as well as many other proteins with known functions, such as RNA helicases, RNA modifiers, cis-regulatory element readers, protein modifiers, and proteins involved in cell cycle, cellular metabolism, and mRNA stability. These proteins may specialize the structure and function of their associated ribosomes and change their translation efficiency on certain subset of mRNAs. For example, the ribo-interactome includes well-known readers (YTHDF1, YTHDF3, and eIF3) of N6-methyladenosine modifications on mRNAs. 5′ UTR N6-methyladenosine is up-regulated upon diverse cellular stresses, and it can promote cap-independent translation in response to cellular stresses (Meyer et al., 2015). Future work is required to provide the direct validation of such RAP-induced translation specialization. PKM2 is another interesting partner identified and further studied in this ribo-interactome (Simsek et al., 2017; Fig. 3 F). In mESCs, PKM2 is enriched on ER-associated ribosomes and directly binds ER-associated mRNAs to facilitate their translation. A primary role for the ER is in the biogenesis of secretory and membrane proteins, which is well established by many studies showing the subcellular distributions of transcripts between cytosol and ER compartments (Reid and Nicchitta, 2011, 2015; Voigt et al., 2017). A very recent study shows that several ER membrane proteins, like SEC61β (a subunit of the SEC61 translocon) and LRRC59, are associated with ER-bound ribosomes. The mRNAs translated by SEC61β- or LRRC59-labeled ribosomes reveal commonly shared and some specifically enriched transcripts, indicating the contribution of these RAPs to translational specialization (Hoffman et al., 2019). These intriguing findings suggest RAPs can associate with subpools of ribosomes in different cellular compartments to display preferential translation, indicating another layer of ribosomal heterogeneity arising from spatial regulation of ribosomes.
Ribosome concentration model
Even though extensive studies have shown the significance of ribosomal heterogeneity to gene-specific regulations of translation, how ribosomes contribute to specific mRNA translation remains a matter of debate. Apart from ribosomal heterogeneity, many reports show that much of the specificity observed in mRNA translation resulting from ribosomes could be explained by a ribosome concentration model (Mills and Green, 2017). In this model, ribosome concentration can affect global and mRNA-specific translational control. Variable dependence of mRNAs on cellular ribosome concentration is mostly due to their difference in the translation initiation rates that results from mRNA-specific features (such as IRES elements, 5′ UTR structures; Lodish, 1974; Noderer et al., 2014). mRNAs with poor translation initiation rates are typically more sensitive to ribosome concentration fluctuations, and certain cell types and tissues that reply on the protein levels of such mRNAs are consequently more vulnerable to perturbations in ribosome concentration (Lodish, 1974; Mills and Green, 2017), such as the important erythroid transcription factor GATA1 in the context of Diamond–Blackfan anemia (Khajuria et al., 2018; Ludwig et al., 2014). The ribosome concentration model was further supported by a recent study demonstrating a preferential translation of synaptic mRNAs by monosomes, but not polysomes, in neuronal processes (Biever et al., 2020). For more information on the ribosome concentration model, readers are referred to the review by Mills and Green (2017). The models of ribosome heterogeneity and ribosome concentration are not mutually exclusive. While our current review highlights impacts of ribosomes on mRNA-specific translational control in stem cells and development, we should not ignore the possibility of other reasonable models, which may complement each other and/or be tightly coordinated to control mRNA translation.
Concluding remarks and future perspectives
Ribosomes are multisubunit molecular machines and directly responsible for the translation of the genetic information into the ultimate functional product proteins. Ribosomes were initially considered as indiscriminate machines that comprise the same components in any cells or conditions. However, studies over the past decades have proven that ribosomes are heterogeneous macromolecular complexes with both general translation functions and also specialized translation roles of specific transcripts. Several related hypothetical theories have been proposed, including the ribosome filter hypothesis (Mauro and Edelman, 2002), ribosome code (Komili et al., 2007), and ribosome concentration model (Mills and Green, 2017), to introduce another regulatory level in the control of gene expression. Ribosomes comprise rRNAs and RPs. Any differences in these components can lead to ribosome heterogeneity; rRNA variants and modifications can give rise to the formation of heterogeneous ribosomes, and ribosomal heterogeneity can also be introduced by RPs through their differential stoichiometry, varying components, existence of paralogs, and numerous PTMs. Apart from these core components, RAPs, which bind to the ribosome surface, can also contribute to ribosome heterogeneity and broaden its regulatory capacity. In addition to these factors, cellular localization of ribosomes and higher-order ribosome architecture may contribute to ribosome heterogeneity as well. On the other hand, different mRNAs have variable sensitivities to subtle alterations in ribosome levels. All of these regulatory controls from ribosomes prove to be an integral regulatory mechanism to modulate and fine tune protein abundance in cells. This mechanism is particularly important in response to internal and external stimuli, like during embryogenesis, as ribosomes can directly change the protein levels in a speedy way, preceding the formation of a new chromatin landscape and transcription program that constitute a secondary response important for long-term adjustment.
For heterogeneous ribosomes, there are many gaps and questions left for future investigations (see text box) to gain comprehensive understanding of this translational machinery in stem cells and development. We should also bear in mind that variable expression levels of certain ribosome-related components (such as rRNA variants, RPs, and RAPs) do not necessarily mean that these components are actually incorporated into ribosomes and result in various functionally distinct populations of ribosomes in cells. Most cases that we discuss here lack such direct verifications. To decipher the heterogeneity in ribosomes and characterize their functions in specialized translation, researchers need techniques to identify heterogeneous ribosomes and specific translated transcripts, as well as the related comprehensive molecular analyses from transcriptome to translatome and proteome. And as this mechanism is extremely critical during cell fate transition, the studies need to be conducted under various cellular conditions to determine the roles of heterogeneous ribosomes during the process. Recently, many advanced techniques have been developed to suit such needs. These techniques are very useful to advance our understanding of heterogeneous ribosomes and their related translation functions, but we still need more advanced technologies. Most mentioned technologies are performed in cultured cells, and related technologies that can be done at the single-cell level and in vivo (such as embryos and tissues) are needed to enable us to study heterogeneity of ribosomes within single cells and during different developmental stages. For example, it would be very interesting to know the changes of ribosome components or other modulations during embryogenesis that may indicate the existence of totipotency-, pluripotency-, multipotency-, unipotency-, and certain differentiation-associated ribosomes, which in turn can be used to achieve “cellular alchemy” for regenerative medicine and disease therapeutics.
Open questions
1. For the genetic information flow based on “the central dogma,” is there a specialized class of rDNAs/rRNAs/RPs that is specifically active (or silent) during stem cell maintenance or differentiation? If so, what are the chromatin, sequence, and epigenetic features of this class of rDNAs/rRNAs/RPs?
2. How is ribosome heterogeneity selectively determined, including alterations from rRNAs and RPs? Multiple regulatory mechanisms should be involved, like transcription and translation. Signals from both outside and inside of cells are also supposed to confer and control the heterogeneity, as ribosome heterogeneity exists not only intercellularly but also intracellularly.
3. Following the previous question, how are heterogeneous ribosomes formed? For example, are ribosomes without certain RP (e.g., RPL38/eL38 and RPS26/eS26) formed from mature ribosomes containing the RP, or are the newly produced ribosomal subunits formed without the RP? Ribosome assembly is an efficient but complicated process (Warner, 1999; Davis and Williamson, 2017). During this process, transient rRNA–RP interactions chaperone rRNA folding, which is essential for pre-rRNA processing and the following rRNA–RP assembly, and many other assembly factors have been identified (Bohnsack and Bohnsack, 2019; Duss et al., 2019; Rodgers et al., 2019; Prattes et al., 2019), indicating that assembly of ribosomes, especially heterogeneous ribosomes, is significantly more complex than previously thought.
4. How does the specific translation happen? Is it through the interaction between ribosomal compositions and cis-regulatory elements in mRNAs, including a specific sequence, modification, or structure (like IRES)? A major knowledge gap exists in our current understanding of these cis-regulatory elements in mRNAs, especially with regard to their interface in trans with different factors and how this interface results in the recruitment of or resistance to certain mRNA subset. These in trans factors include different RNA-binding proteins, such as RPs, RAPs, and translation initiation factors, as well as rRNAs, tRNAs, and noncoding RNAs.
5. How heterogeneous are ribosomes in a certain cell type? To answer this question, we may need to know how diverse the ribosome population needs to be in any given cell. Is it possible that in a certain cell type, one type of the ribosome is enough to maintain cell viability and basic functions? In responses to stress and stimuli, does the extent of ribosomal heterogeneity increase or decrease?
6. rRNAs are transcribed and modified by multiple proteins, and RPs and RAPs are also translation products. It is also interesting to know whether there is a positive/negative feedback loop to enhance/inhibit certain ribosome heterogeneity in stem cells, certain lineage differentiation, or tissue development.
7. Considering the ribosome concentration model, which may complement ribosome heterogeneity, how do these alterations in cellular ribosome concentrations and ribosome compositions affect stem cell fates and development? To explore this question, we need to know how to determine protein synthesis rates of one certain mRNA (1) under gradient concentrations of ribosomes with the same composition or (2) under the same concentration of ribosomes with different compositions. These measurements are very technically challenging, but they are critical to the understanding of cohesive molecular mechanisms connecting quantitative/qualitative regulation of ribosomes to phenotypic consequences of specific mRNA translations in stem cells and development. Additionally, we should not exclude the possibility that translation of one certain mRNA may be robust to all ribosomes with mentioned heterogeneity if ribosome concentrations are not changed, in which the mRNA feature and ribosome concentration have a larger impact on translational control.
Last, but not least, the model of large multisubunit complexes with both general and specific functions is not unique to ribosomes. Another similar well-studied example is the Mediator, a critical regulator that has both general and specialized functions in transcription with altering compositions and activities (Poss et al., 2013; Cramer, 2019). More similar examples are histone octamers in DNA packaging (Soshnev et al., 2016; Talbert and Henikoff, 2017) and proteasomes in protein degradation (Morozov and Karpov, 2018; Bousquet-Dubouch et al., 2011). These examples indicate that heterogeneity may widely exist in such complicated multisubunit complexes, especially the ones with critical cellular functions. Such heterogeneity may enable their specialized functions in orchestrating gene expression at different regulatory levels in stem cells and development, as well as in human diseases. Studies of such heterogeneity and related specialized functions not only reveal novel gene regulatory mechanisms but also bring insights into diagnostics and therapeutics of human diseases.
Acknowledgments
We are grateful to Jill Gregory for the illustrations (Figs. 1, 2, and 3) and members of Wang laboratory for helpful discussions.
This study was supported by the National Institutes of Health (grants GM129157, HD095938, HD097268, and HL146664) and New York State Stem Cell Science (NYSTEM grants C32583GG and C32569GG).
The authors declare no competing financial interests.
Author contributions: D. Li and J. Wang both conceived and wrote the review.
References
- Adjaye J., Huntriss J., Herwig R., BenKahla A., Brink T.C., Wierling C., Hultschig C., Groth D., Yaspo M.-L., Picton H.M., et al. . 2005. Primary differentiation in the human blastocyst: comparative molecular portraits of inner cell mass and trophectoderm cells. Stem Cells. 23:1514–1525. 10.1634/stemcells.2005-0113 [DOI] [PubMed] [Google Scholar]
- Anderson S.J., Lauritsen J.P.H., Hartman M.G., Foushee A.M.D.G., Lefebvre J.M., Shinton S.A., Gerhardt B., Hardy R.R., Oravecz T., and Wiest D.L.. 2007. Ablation of ribosomal protein L22 selectively impairs alphabeta T cell development by activation of a p53-dependent checkpoint. Immunity. 26:759–772. 10.1016/j.immuni.2007.04.012 [DOI] [PubMed] [Google Scholar]
- Ballesta J.P.G., and Remacha M.. 1996. The Large Ribosomal Subunit Stalk as a Regulatory Element of the Eukaryotic Translational Machinery. Prog. Nucleic Acid Res. Mol. Biol. 55:157–193. 10.1016/S0079-6603(08)60193-2 [DOI] [PubMed] [Google Scholar]
- Baser A., Skabkin M., Kleber S., Dang Y., Gülcüler Balta G.S., Kalamakis G., Göpferich M., Ibañez D.C., Schefzik R., Lopez A.S., et al. . 2019. Onset of differentiation is post-transcriptionally controlled in adult neural stem cells. Nature 566:100–104. 10.1038/s41586-019-0888-x [DOI] [PubMed]
- Baßler J., and Hurt E.. 2019. Annual Review of Biochemistry Eukaryotic Ribosome Assembly. Annu. Rev. Biochem. 88:8.1–8.26. [DOI] [PubMed]
- Bellodi C., McMahon M., Contreras A., Juliano D., Kopmar N., Nakamura T., Maltby D., Burlingame A., Savage S.A., Shimamura A., and Ruggero D.. 2013. H/ACA small RNA dysfunctions in disease reveal key roles for noncoding RNA modifications in hematopoietic stem cell differentiation. Cell Rep. 3:1493–1502. 10.1016/j.celrep.2013.04.030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biever A., Glock C., Tushev G., Ciirdaeva E., Dalmay T., Langer J.D., and Schuman E.M.. 2020. Monosomes actively translate synaptic mRNAs in neuronal processes. Science. 367(6477). 10.1126/science.aay4991 [DOI] [PubMed] [Google Scholar]
- Birkedal U., Christensen-Dalsgaard M., Krogh N., Sabarinathan R., Gorodkin J., and Nielsen H.. 2015. Profiling of ribose methylations in RNA by high-throughput sequencing. Angew. Chem. Int. Ed. Engl. 54:451–455. 10.1002/anie.201408362 [DOI] [PubMed] [Google Scholar]
- Blanco A., and Blanco G.. 2017. Nucleic acids. In Medical Biochemistry. Elsevier; 121–140. 10.1016/B978-0-12-803550-4.00006-9 [DOI] [Google Scholar]
- Blanco S., Bandiera R., Popis M., Hussain S., Lombard P., Aleksic J., Sajini A., Tanna H., Cortés-Garrido R., Gkatza N., et al. . 2016. Stem cell function and stress response are controlled by protein synthesis. Nature. 534:335–340. 10.1038/nature18282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohnsack K.E., and Bohnsack M.T.. 2019. RNA-Binding Proteins Chaperone Ribonucleoprotein Complex Assembly to Solve the RNA-Folding Problem. Cell. 179:1248–1250. 10.1016/j.cell.2019.11.011 [DOI] [PubMed] [Google Scholar]
- Bonaguidi M.A., Wheeler M.A., Shapiro J.S., Stadel R.P., Sun G.J., Ming G.L., and Song H.. 2011. In vivo clonal analysis reveals self-renewing and multipotent adult neural stem cell characteristics. Cell. 145:1142–1155. 10.1016/j.cell.2011.05.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boria I., Garelli E., Gazda H.T., Aspesi A., Quarello P., Pavesi E., Ferrante D., Meerpohl J.J., Kartal M., Da Costa L., et al. . 2010. The ribosomal basis of Diamond-Blackfan Anemia: mutation and database update. Hum. Mutat. 31:1269–1279. 10.1002/humu.21383 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bortoluzzi S., d’Alessi F., Romualdi C., and Danieli G.A.. 2002. Differential expression of genes coding for ribosomal proteins in different human tissues. Bioinformatics. 10.1093/bioinformatics/17.12.1152 [DOI] [PubMed] [Google Scholar]
- Bousquet-Dubouch M.P., Fabre B., Monsarrat B., and Burlet-Schiltz O.. 2011. Proteomics to study the diversity and dynamics of proteasome complexes: from fundamentals to the clinic. Expert Rev. Proteomics. 8:459–481. 10.1586/epr.11.41 [DOI] [PubMed] [Google Scholar]
- Brandman O., and Hegde R.S.. 2016. Ribosome-associated protein quality control. Nat. Struct. Mol. Biol. 23:7–15. 10.1038/nsmb.3147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braschi B., Denny P., Gray K., Jones T., Seal R., Tweedie S., Yates B., and Bruford E.. 2019. Genenames.org: the HGNC and VGNC resources in 2019. Nucleic Acids Res. 47(D1):D786–D792. 10.1093/nar/gky930 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bulut-Karslioglu A., Macrae T.A., Oses-Prieto J.A., Covarrubias S., Percharde M., Ku G., Diaz A., McManus M.T., Burlingame A.L., and Ramalho-Santos M.. 2018. The Transcriptionally Permissive Chromatin State of Embryonic Stem Cells Is Acutely Tuned to Translational Output. Cell Stem Cell. 22:369–383.e8. 10.1016/j.stem.2018.02.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castle J.C., Armour C.D., Löwer M., Haynor D., Biery M., Bouzek H., Chen R., Jackson S., Johnson J.M., Rohl C.A., and Raymond C.K.. 2010. Digital genome-wide ncRNA expression, including SnoRNAs, across 11 human tissues using polyA-neutral amplification. PLoS One. 5:e11779 10.1371/journal.pone.0011779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chau K.F., Shannon M.L., Fame R.M., Fonseca E., Mullan H., Johnson M.B., Sendamarai A.K., Springel M.W., Laurent B., and Lehtinen M.K.. 2018. Downregulation of ribosome biogenesis during early forebrain development. eLife. 7:e36998 10.7554/eLife.36998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen E., Sharma M.R., Shi X., Agrawal R.K., and Joseph S.. 2014. Fragile X mental retardation protein regulates translation by binding directly to the ribosome. Mol. Cell. 54:407–417. 10.1016/j.molcel.2014.03.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choe Y.J., Park S.H., Hassemer T., Körner R., Vincenz-Donnelly L., Hayer-Hartl M., and Hartl F.U.. 2016. Failure of RQC machinery causes protein aggregation and proteotoxic stress. Nature. 531:191–195. 10.1038/nature16973 [DOI] [PubMed] [Google Scholar]
- Chu J., Hong N.A., Masuda C.A., Jenkins B.V., Nelms K.A., Goodnow C.C., Glynne R.J., Wu H., Masliah E., Joazeiro C.A.P., and Kay S.A.. 2009. A mouse forward genetics screen identifies LISTERIN as an E3 ubiquitin ligase involved in neurodegeneration. Proc. Natl. Acad. Sci. USA. 106:2097–2103. 10.1073/pnas.0812819106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corsini N.S., Peer A.M., Moeseneder P., Roiuk M., Burkard T.R., Theussl H.-C., Moll I., and Knoblich J.A.. 2018. Coordinated Control of mRNA and rRNA Processing Controls Embryonic Stem Cell Pluripotency and Differentiation. Cell Stem Cell. 22:543–558.e12. 10.1016/j.stem.2018.03.002 [DOI] [PubMed] [Google Scholar]
- Cramer P. 2019. Organization and regulation of gene transcription. Nature. 573:45–54. 10.1038/s41586-019-1517-4 [DOI] [PubMed] [Google Scholar]
- Darnell J.C., Van Driesche S.J., Zhang C., Hung K.Y.S., Mele A., Fraser C.E., Stone E.F., Chen C., Fak J.J., Chi S.W., et al. . 2011. FMRP stalls ribosomal translocation on mRNAs linked to synaptic function and autism. Cell. 146:247–261. 10.1016/j.cell.2011.06.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis J.H., and Williamson J.R.. 2017. Structure and dynamics of bacterial ribosome biogenesis. Philos. Trans. R. Soc. Lond. B Biol. Sci. 372:20160181 10.1098/rstb.2016.0181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Decatur W.A., and Fournier M.J.. 2002. rRNA modifications and ribosome function. Trends Biochem. Sci. 27:344–351. 10.1016/S0968-0004(02)02109-6 [DOI] [PubMed] [Google Scholar]
- Defenouillère Q., Zhang E., Namane A., Mouaikel J., Jacquier A., and Fromont-Racine M.. 2016. Rqc1 and ltn1 prevent c-terminal alanine-threonine tail (cat-tail)-induced protein aggregation by efficient recruitment of cdc48 on stalled 60s subunits. J. Biol. Chem. 291:12245–12253. 10.1074/jbc.M116.722264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dimarco E., Cascone E., Bellavia D., and Caradonna F.. 2012. Functional variants of 5S rRNA in the ribosomes of common sea urchin Paracentrotus lividus. Gene. 508:21–25. 10.1016/j.gene.2012.07.067 [DOI] [PubMed] [Google Scholar]
- Dimitrova D.G., Teysset L., and Carré C.. 2019. RNA 2′-O-Methylation (Nm) modification in human diseases. Genes (Basel). 10:117 10.3390/genes10020117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duss O., Stepanyuk G.A., Puglisi J.D., and Williamson J.R.. 2019. Transient Protein-RNA Interactions Guide Nascent Ribosomal RNA Folding. Cell. 179:1357–1369.e16. 10.1016/j.cell.2019.10.035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferretti M.B., Ghalei H., Ward E.A., Potts E.L., and Karbstein K.. 2017. Rps26 directs mRNA-specific translation by recognition of Kozak sequence elements. Nat. Struct. Mol. Biol. 24:700–707. 10.1038/nsmb.3442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finley D., Bartel B., and Varshavsky A.. 1989. The tails of ubiquitin precursors are ribosomal proteins whose fusion to ubiquitin facilitates ribosome biogenesis. Nature. 338:394–401. 10.1038/338394a0 [DOI] [PubMed] [Google Scholar]
- Fisher E.M.C., Beer-Romero P., Brown L.G., Ridley A., McNeil J.A., Lawrence J.B., Willard H.F., Bieber F.R., and Page D.C.. 1990. Homologous ribosomal protein genes on the human X and Y chromosomes: escape from X inactivation and possible implications for Turner syndrome. Cell. 63:1205–1218. 10.1016/0092-8674(90)90416-C [DOI] [PubMed] [Google Scholar]
- Fujii K., Shi Z., Zhulyn O., Denans N., and Barna M.. 2017. Pervasive translational regulation of the cell signalling circuitry underlies mammalian development. Nat. Commun. 8:14443 10.1038/ncomms14443 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujii K., Susanto T.T., Saurabh S., and Barna M.. 2018. Decoding the Function of Expansion Segments in Ribosomes. Mol. Cell. 72:1013–1020.e6. 10.1016/j.molcel.2018.11.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garzia A., Jafarnejad S.M., Meyer C., Chapat C., Gogakos T., Morozov P., Amiri M., Shapiro M., Molina H., Tuschl T., and Sonenberg N.. 2017. The E3 ubiquitin ligase and RNA-binding protein ZNF598 orchestrates ribosome quality control of premature polyadenylated mRNAs. Nat. Commun. 8:16056 10.1038/ncomms16056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gressner A.M., and Wool I.G.. 1974. The Phosphorylation of Liver Ribosomal Proteins in Vito EVIDENCE THAT ONLY A SINGLE SMALL SUBUNIT PROTEIN (S6) IS PHOSPHORYLATED. J. Biol. Chem. 249:6917–6925. [PubMed] [Google Scholar]
- Guimaraes J.C., and Zavolan M.. 2016. Patterns of ribosomal protein expression specify normal and malignant human cells. Genome Biol. 17:236 10.1186/s13059-016-1104-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gunderson J.H., Sogin M.L., Wollett G., Hollingdale M., De La Cruz V.F., Waters A.P., and McCutchan T.F.. 1987. Structurally distinct, stage-specific ribosomes occur in plasmodium. Science. 13:933–937. 10.1126/science.3672135 [DOI] [PubMed] [Google Scholar]
- Hertz M.I., Landry D.M., Willis A.E., Luo G., and Thompson S.R.. 2013. Ribosomal Protein S25 Dependency Reveals a Common Mechanism for Diverse Internal Ribosome Entry Sites and Ribosome Shunting. Mol. Cell. Biol. 33:1016–1026. 10.1128/MCB.00879-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higa-Nakamine S., Suzuki T., Uechi T., Chakraborty A., Nakajima Y., Nakamura M., Hirano N., Suzuki T., and Kenmochi N.. 2012. Loss of ribosomal RNA modification causes developmental defects in zebrafish. Nucleic Acids Res. 40:391–398. 10.1093/nar/gkr700 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higgins R., Gendron J.M., Rising L., Mak R., Webb K., Kaiser S.E., Zuzow N., Riviere P., Yang B., Fenech E., et al. . 2015. The Unfolded Protein Response Triggers Site-Specific Regulatory Ubiquitylation of 40S Ribosomal Proteins. Mol. Cell. 59:35–49. 10.1016/j.molcel.2015.04.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffman A.M., Chen Q., Zheng T., and Nicchitta C.V.. 2019. Heterogeneous translational landscape of the endoplasmic reticulum revealed by ribosome proximity labeling and transcriptome analysis. J. Biol. Chem. 294:8942–8958. 10.1074/jbc.RA119.007996 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horos R., Ijspeert H., Pospisilova D., Sendtner R., Andrieu-Soler C., Taskesen E., Nieradka A., Cmejla R., Sendtner M., Touw I.P., and von Lindern M.. 2012. Ribosomal deficiencies in Diamond-Blackfan anemia impair translation of transcripts essential for differentiation of murine and human erythroblasts. Blood. 119:262–272. 10.1182/blood-2011-06-358200 [DOI] [PubMed] [Google Scholar]
- Huang H., Weng H., Deng X., and Chen J.. 2020. RNA Modifications in Cancer: Functions, Mechanisms, and Therapeutic Implications. Annu. Rev. Cancer Biol. 4:221–240. 10.1146/annurev-cancerbio-030419-033357 [DOI] [Google Scholar]
- Ikeuchi K., Tesina P., Matsuo Y., Sugiyama T., Cheng J., Saeki Y., Tanaka K., Becker T., Beckmann R., and Inada T.. 2019. Collided ribosomes form a unique structural interface to induce Hel2-driven quality control pathways. EMBO J. 38:e100276 10.15252/embj.2018100276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imami K., Milek M., Bogdanow B., Yasuda T., Kastelic N., Zauber H., Ishihama Y., Landthaler M., and Selbach M.. 2018. Phosphorylation of the Ribosomal Protein RPL12/uL11 Affects Translation during Mitosis. Mol. Cell. 72:84–98.e9. 10.1016/j.molcel.2018.08.019 [DOI] [PubMed] [Google Scholar]
- Ingolia N.T., Lareau L.F., and Weissman J.S.. 2011. Ribosome profiling of mouse embryonic stem cells reveals the complexity and dynamics of mammalian proteomes. Cell. 147:789–802. 10.1016/j.cell.2011.10.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jack K., Bellodi C., Landry D.M., Niederer R.O., Meskauskas A., Musalgaonkar S., Kopmar N., Krasnykh O., Dean A.M., Thompson S.R., et al. . 2011. rRNA pseudouridylation defects affect ribosomal ligand binding and translational fidelity from yeast to human cells. Mol. Cell. 44:660–666. 10.1016/j.molcel.2011.09.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jha S., Rollins M.G., Fuchs G., Procter D.J., Hall E.A., Cozzolino K., Sarnow P., Savas J.N., and Walsh D.. 2017. Trans-kingdom mimicry underlies ribosome customization by a poxvirus kinase. Nature. 546:651–655. 10.1038/nature22814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Juszkiewicz S., and Hegde R.S.. 2017. Initiation of Quality Control during Poly(A) Translation Requires Site-Specific Ribosome Ubiquitination. Mol. Cell. 65:743–750.e4. 10.1016/j.molcel.2016.11.039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Juszkiewicz S., Chandrasekaran V., Lin Z., Kraatz S., Ramakrishnan V., and Hegde R.S.. 2018. ZNF598 Is a Quality Control Sensor of Collided Ribosomes. Mol. Cell. 72:469–481.e7. 10.1016/j.molcel.2018.08.037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kadrmas J.L., Smith M.A., Pronovost S.M., and Beckerle M.C.. 2007. Characterization of RACK1 function in Drosophila development. Dev. Dyn. 236:2207–2215. 10.1002/dvdy.21217 [DOI] [PubMed] [Google Scholar]
- Kadumuri R.V., and Janga S.C.. 2018. Epitranscriptomic Code and Its Alterations in Human Disease. Trends Mol. Med. 24:886–903. 10.1016/j.molmed.2018.07.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kapasi P., Chaudhuri S., Vyas K., Baus D., Komar A.A., Fox P.L., Merrick W.C., and Mazumder B.. 2007. L13a blocks 48S assembly: role of a general initiation factor in mRNA-specific translational control. Mol. Cell. 25:113–126. 10.1016/j.molcel.2006.11.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelleher R.J. III, Govindarajan A., Jung H.Y., Kang H., and Tonegawa S.. 2004. Translational control by MAPK signaling in long-term synaptic plasticity and memory. Cell. 116:467–479. 10.1016/S0092-8674(04)00115-1 [DOI] [PubMed] [Google Scholar]
- Khajuria R.K., Munschauer M., Ulirsch J.C., Fiorini C., Ludwig L.S., McFarland S.K., Abdulhay N.J., Specht H., Keshishian H., Mani D.R., et al. . 2018. Ribosome Levels Selectively Regulate Translation and Lineage Commitment in Human Hematopoiesis. Cell. 173:90–103.e19. 10.1016/j.cell.2018.02.036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan Z., Ford M.J., Cusanovich D.A., Mitrano A., Pritchard J.K., and Gilad Y.. 2013. Primate transcript and protein expression levels evolve under compensatory selection pressures. Science. 342:1100–1104. 10.1126/science.1242379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim H.L., Song W.S., Kim K., and Lee K.. 2008. Characterization of heterogeneous LSU rRNA profiles in Streptomyces coelicolor under different growth stages and conditions. Curr. Microbiol. 57:537–541. 10.1007/s00284-008-9238-1 [DOI] [PubMed] [Google Scholar]
- Klinge S., and Woolford J.L. Jr. 2019. Ribosome assembly coming into focus. Nat. Rev. Mol. Cell Biol. 20:116–131. 10.1038/s41580-018-0078-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knight Z.A., Tan K., Birsoy K., Schmidt S., Garrison J.L., Wysocki R.W., Emiliano A., Ekstrand M.I., and Friedman J.M.. 2012. Molecular profiling of activated neurons by phosphorylated ribosome capture. Cell. 151:1126–1137. 10.1016/j.cell.2012.10.039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi M., Oshima S., Maeyashiki C., Nibe Y., Otsubo K., Matsuzawa Y., Nemoto Y., Nagaishi T., Okamoto R., Tsuchiya K., et al. . 2016. The ubiquitin hybrid gene UBA52 regulates ubiquitination of ribosome and sustains embryonic development. Sci. Rep. 6:36780 10.1038/srep36780 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komili S., Farny N.G., Roth F.P., and Silver P.A.. 2007. Functional specificity among ribosomal proteins regulates gene expression. Cell. 131:557–571. 10.1016/j.cell.2007.08.037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kondrashov N., Pusic A., Stumpf C.R., Shimizu K., Hsieh A.C., Ishijima J., Shiroishi T., and Barna M.. 2011. Ribosome-mediated specificity in Hox mRNA translation and vertebrate tissue patterning. Cell. 145:383–397. 10.1016/j.cell.2011.03.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozak M. 1986. Point mutations define a sequence flanking the AUG initiator codon that modulates translation by eukaryotic ribosomes. Cell. 44:283–292. 10.1016/0092-8674(86)90762-2 [DOI] [PubMed] [Google Scholar]
- Lafontaine D.L.J. 2015. Noncoding RNAs in eukaryotic ribosome biogenesis and function. Nat. Struct. Mol. Biol. 22:11–19. 10.1038/nsmb.2939 [DOI] [PubMed] [Google Scholar]
- Lam Y.W., Lamond A.I., Mann M., and Andersen J.S.. 2007. Analysis of nucleolar protein dynamics reveals the nuclear degradation of ribosomal proteins. Curr. Biol. 17:749–760. 10.1016/j.cub.2007.03.064 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landry D.M., Hertz M.I., and Thompson S.R.. 2009. RPS25 is essential for translation initiation by the Dicistroviridae and hepatitis C viral IRESs. Genes Dev. 23:2753–2764. 10.1101/gad.1832209 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee A.S.-Y., Burdeinick-Kerr R., and Whelan S.P.J.. 2013a A ribosome-specialized translation initiation pathway is required for cap-dependent translation of vesicular stomatitis virus mRNAs. Proc. Natl. Acad. Sci. USA. 110:324–329. 10.1073/pnas.1216454109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee M.T., Bonneau A.R., Takacs C.M., Bazzini A.A., DiVito K.R., Fleming E.S., and Giraldez A.J.. 2013b Nanog, Pou5f1 and SoxB1 activate zygotic gene expression during the maternal-to-zygotic transition. Nature. 503:360–364. 10.1038/nature12632 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Locati M.D., Pagano J.F.B., Ensink W.A., van Olst M., van Leeuwen S., Nehrdich U., Zhu K., Spaink H.P., Girard G., Rauwerda H., et al. . 2017a Linking maternal and somatic 5S rRNA types with different sequence-specific non-LTR retrotransposons. RNA. 23:446–456. 10.1261/rna.059642.116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Locati M.D., Pagano J.F.B., Girard G., Ensink W.A., van Olst M., van Leeuwen S., Nehrdich U., Spaink H.P., Rauwerda H., Jonker M.J., et al. . 2017b Expression of distinct maternal and somatic 5.8S, 18S, and 28S rRNA types during zebrafish development. RNA. 23:1188–1199. 10.1261/rna.061515.117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lodish H.F. 1974. Model for the regulation of mRNA translation applied to haemoglobin synthesis. Nature. 251:385–388. 10.1038/251385a0 [DOI] [PubMed] [Google Scholar]
- Lopes A.M., Miguel R.N., Sargent C.A., Ellis P.J., Amorim A., and Affara N.A.. 2010. The human RPS4 paralogue on Yq11.223 encodes a structurally conserved ribosomal protein and is preferentially expressed during spermatogenesis. BMC Mol. Biol. 11:33 10.1186/1471-2199-11-33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- López-López A., Benlloch S., Bonfá M., Rodríguez-Valera F., and Mira A.. 2007. Intragenomic 16S rDNA divergence in Haloarcula marismortui is an adaptation to different temperatures. J. Mol. Evol. 65:687–696. 10.1007/s00239-007-9047-3 [DOI] [PubMed] [Google Scholar]
- Ludwig L.S., Gazda H.T., Eng J.C., Eichhorn S.W., Thiru P., Ghazvinian R., George T.I., Gotlib J.R., Beggs A.H., Sieff C.A., et al. . 2014. Altered translation of GATA1 in Diamond-Blackfan anemia. Nat. Med. 20:748–753. 10.1038/nm.3557 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Majzoub K., Hafirassou M.L., Meignin C., Goto A., Marzi S., Fedorova A., Verdier Y., Vinh J., Hoffmann J.A., Martin F., et al. . 2014. RACK1 controls IRES-mediated translation of viruses. Cell. 159:1086–1095. 10.1016/j.cell.2014.10.041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin I., Kim J.W., Lee B.D., Kang H.C., Xu J.C., Jia H., Stankowski J., Kim M.S., Zhong J., Kumar M., et al. . 2014. Ribosomal protein s15 phosphorylation mediates LRRK2 neurodegeneration in Parkinson’s disease. Cell. 157:472–485. 10.1016/j.cell.2014.01.064 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuo Y., Ikeuchi K., Saeki Y., Iwasaki S., Schmidt C., Udagawa T., Sato F., Tsuchiya H., Becker T., Tanaka K., et al. . 2017. Ubiquitination of stalled ribosome triggers ribosome-associated quality control. Nat. Commun. 8:159 10.1038/s41467-017-00188-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mauro V.P., and Edelman G.M.. 2002. The ribosome filter hypothesis. Proc. Natl. Acad. Sci. USA. 99:12031–12036. 10.1073/pnas.192442499 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazumder B., Sampath P., Seshadri V., Maitra R.K., DiCorleto P.E., and Fox P.L.. 2003. Regulated release of L13a from the 60S ribosomal subunit as a mechanism of transcript-specific translational control. Cell. 115:187–198. 10.1016/S0092-8674(03)00773-6 [DOI] [PubMed] [Google Scholar]
- Mestre-Fos S., Penev P.I., Suttapitugsakul S., Hu M., Ito C., Petrov A.S., Wartell R.M., Wu R., and Williams L.D.. 2019. G-Quadruplexes in Human Ribosomal RNA. J. Mol. Biol. 431:1940–1955. 10.1016/j.jmb.2019.03.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer K.D., Patil D.P., Zhou J., Zinoviev A., Skabkin M.A., Elemento O., Pestova T.V., Qian S.B., and Jaffrey S.R.. 2015. 5′ UTR m(6)A Promotes Cap-Independent Translation. Cell. 163:999–1010. 10.1016/j.cell.2015.10.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyuhas O. 2015. Ribosomal Protein S6 Phosphorylation: Four Decades of Research. Int. Rev. Cell Mol. Biol. 320:41–73. 10.1016/bs.ircmb.2015.07.006 [DOI] [PubMed] [Google Scholar]
- Mills E.W., and Green R.. 2017. Ribosomopathies: There’s strength in numbers. Science. 358:eaan2755. [DOI] [PubMed]
- Morozov A.V., and Karpov V.L.. 2018. Biological consequences of structural and functional proteasome diversity. Heliyon. 4:e00894 10.1016/j.heliyon.2018.e00894 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mukhopadhyay R., Ray P.S., Arif A., Brady A.K., Kinter M., and Fox P.L.. 2008. DAPK-ZIPK-L13a axis constitutes a negative-feedback module regulating inflammatory gene expression. Mol. Cell. 32:371–382. 10.1016/j.molcel.2008.09.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nachmani D., Bothmer A.H., Grisendi S., Mele A., Bothmer D., Lee J.D., Monteleone E., Cheng K., Zhang Y., Bester A.C., et al. . 2019. Germline NPM1 mutations lead to altered rRNA 2′-O-methylation and cause dyskeratosis congenita. Nat. Genet. 51:1518–1529. 10.1038/s41588-019-0502-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Natchiar S.K., Myasnikov A.G., Kratzat H., Hazemann I., and Klaholz B.P.. 2017. Visualization of chemical modifications in the human 80S ribosome structure. Nature. 551:472–477. 10.1038/nature24482 [DOI] [PubMed] [Google Scholar]
- Natchiar S.K., Myasnikov A.G., Hazemann I., and Klaholz B.P.. 2018. Visualizing the Role of 2′-OH rRNA Methylations in the Human Ribosome Structure. Biomolecules. 8:E125 10.3390/biom8040125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen A.T., Prado M.A., Schmidt P.J., Sendamarai A.K., Wilson-Grady J.T., Min M., Campagna D.R., Tian G., Shi Y., Dederer V., et al. . 2017. UBE2O remodels the proteome during terminal erythroid differentiation. Science. 357:eaan0218. [DOI] [PMC free article] [PubMed]
- Nishiyama T., Yamamoto H., Uchiumi T., and Nakashima N.. 2007. Eukaryotic ribosomal protein RPS25 interacts with the conserved loop region in a dicistroviral intergenic internal ribosome entry site. Nucleic Acids Res. 35:1514–1521. 10.1093/nar/gkl1121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noderer W.L., Flockhart R.J., Bhaduri A., Diaz de Arce A.J., Zhang J., Khavari P.A., and Wang C.L.. 2014. Quantitative analysis of mammalian translation initiation sites by FACS-seq. Mol. Syst. Biol. 10:748 10.15252/msb.20145136 [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Leary M.N., Schreiber K.H., Zhang Y., Duc A.C.E., Rao S., Hale J.S., Academia E.C., Shah S.R., Morton J.F., Holstein C.A., et al. . 2013. The ribosomal protein Rpl22 controls ribosome composition by directly repressing expression of its own paralog, Rpl22l1. PLoS Genet. 9:e1003708 10.1371/journal.pgen.1003708 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliver E.R., Saunders T.L., Tarlé S.A., and Glaser T.. 2004. Ribosomal protein L24 defect in belly spot and tail (Bst), a mouse Minute. Development. 131:3907–3920. 10.1242/dev.01268 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parent R., and Beretta L.. 2008. Translational control plays a prominent role in the hepatocytic differentiation of HepaRG liver progenitor cells. Genome Biol. 9:R19 10.1186/gb-2008-9-1-r19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park C.W., and Ryu K.Y.. 2014. Cellular ubiquitin pool dynamics and homeostasis. BMB Rep. 47:475–482. 10.5483/BMBRep.2014.47.9.128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parks M.M., Kurylo C.M., Dass R.A., Bojmar L., Lyden D., Vincent C.T., and Blanchard S.C.. 2018. Variant ribosomal RNA alleles are conserved and exhibit tissue-specific expression. Sci. Adv. 4:eaao0665 10.1126/sciadv.aao0665 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pereira I.T., Spangenberg L., Robert A.W., Amorín R., Stimamiglio M.A., Naya H., and Dallagiovanna B.. 2019. Cardiomyogenic differentiation is fine-tuned by differential mRNA association with polysomes. BMC Genomics. 20:219 10.1186/s12864-019-5550-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson A.C., Russell J.D., Bailey D.J., Westphall M.S., and Coon J.J.. 2012. Parallel reaction monitoring for high resolution and high mass accuracy quantitative, targeted proteomics. Mol. Cell. Proteomics. 11:1475–1488. 10.1074/mcp.O112.020131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pisarev A.V., Kolupaeva V.G., Yusupov M.M., Hellen C.U.T., and Pestova T.V.. 2008. Ribosomal position and contacts of mRNA in eukaryotic translation initiation complexes. EMBO J. 27:1609–1621. 10.1038/emboj.2008.90 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poss Z.C., Ebmeier C.C., and Taatjes D.J.. 2013. The Mediator complex and transcription regulation. Crit. Rev. Biochem. Mol. Biol. 48:575–608. 10.3109/10409238.2013.840259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prattes M., Lo Y.-H., Bergler H., and Stanley R.E.. 2019. Shaping the Nascent Ribosome: AAA-ATPases in Eukaryotic Ribosome Biogenesis. Biomolecules. 9:715 10.3390/biom9110715 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quade N., Boehringer D., Leibundgut M., van den Heuvel J., and Ban N.. 2015. Cryo-EM structure of Hepatitis C virus IRES bound to the human ribosome at 3.9-Å resolution. Nat. Commun. 6:7646 10.1038/ncomms8646 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reid D.W., and Nicchitta C.V.. 2011. Primary Role for Endoplasmic Reticulum-bound Ribosomes in Cellular Translation Identified by Ribosome Profiling. J. Biol. Chem. 287:5518–5527. 10.1074/jbc.M111.312280 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reid D.W., and Nicchitta C.V.. 2015. Diversity and selectivity in mRNA translation on the endoplasmic reticulum. Nat. Rev. Mol. Cell Biol. 16:221–231. 10.1038/nrm3958 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Remacha M., Jimenez-Diaz A., Bermejo B., Rodriguez-Gabriel M.A., Guarinos E., and Ballesta J.P.G.. 1995. Ribosomal Acidic Phosphoproteins P1 and P2 Are Not Required for Cell Viability but Regulate the Pattern of Protein Expression in Saccharomyces cerevisiae. Mol. Cell Biol. 15:4754–4762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richards M., Tan S.P., Tan J.H., Chan W.K., and Bongso A.. 2004. The transcriptome profile of human embryonic stem cells as defined by SAGE. Stem Cells. 22:51–64. 10.1634/stemcells.22-1-51 [DOI] [PubMed] [Google Scholar]
- Rodgers M.L., Woodson S.A., and Jenkins T.C.. 2019. Transcription Increases the Cooperativity of Ribonucleoprotein Assembly In Brief Concomitant single-molecule detection of transcription elongation and protein recruitment reveals dynamic sampling of nascent pre-ribosomal RNA by ribosomal proteins until multiple proteins can bind and commit the elongating pre-rRNA to productive assembly. Cell. 179:1370–1381.e12. 10.1016/j.cell.2019.11.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruvinsky I., Sharon N., Lerer T., Cohen H., Stolovich-Rain M., Nir T., Dor Y., Zisman P., and Meyuhas O.. 2005. Ribosomal protein S6 phosphorylation is a determinant of cell size and glucose homeostasis. Genes Dev. 19:2199–2211. 10.1101/gad.351605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruvinsky I., Katz M., Dreazen A., Gielchinsky Y., Saada A., Freedman N., Mishani E., Zimmerman G., Kasir J., and Meyuhas O.. 2009. Mice deficient in ribosomal protein S6 phosphorylation suffer from muscle weakness that reflects a growth defect and energy deficit. PLoS One. 4:e5618 10.1371/journal.pone.0005618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sampath P., Pritchard D.K., Pabon L., Reinecke H., Schwartz S.M., Morris D.R., and Murry C.E.. 2008. A hierarchical network controls protein translation during murine embryonic stem cell self-renewal and differentiation. Cell Stem Cell. 2:448–460. 10.1016/j.stem.2008.03.013 [DOI] [PubMed] [Google Scholar]
- Sanchez C.G., Teixeira F.K., Czech B., Preall J.B., Zamparini A.L., Seifert J.R.K., Malone C.D., Hannon G.J., and Lehmann R.. 2016. Regulation of Ribosome Biogenesis and Protein Synthesis Controls Germline Stem Cell Differentiation. Cell Stem Cell. 18:276–290. 10.1016/j.stem.2015.11.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savić N., Bär D., Leone S., Frommel S.C., Weber F.A., Vollenweider E., Ferrari E., Ziegler U., Kaech A., Shakhova O., et al. . 2014. lncRNA maturation to initiate heterochromatin formation in the nucleolus is required for exit from pluripotency in ESCs. Cell Stem Cell. 15:720–734. 10.1016/j.stem.2014.10.005 [DOI] [PubMed] [Google Scholar]
- Sharma S., and Lafontaine D.L.J.. 2015. “View From A Bridge”: A New Perspective on Eukaryotic rRNA Base Modification. Trends Biochem. Sci. 40:560–575 (doi.) 10.1016/j.tibs.2015.07.008 [DOI] [PubMed] [Google Scholar]
- Sharma S., Marchand V., Motorin Y., and Lafontaine D.L.J.. 2017. Identification of sites of 2′-O-methylation vulnerability in human ribosomal RNAs by systematic mapping. Sci. Rep. 7:11490 10.1038/s41598-017-09734-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharov A.A., Piao Y., Matoba R., Dudekula D.B., Qian Y., VanBuren V., Falco G., Martin P.R., Stagg C.A., Bassey U.C., et al. . 2003. Transcriptome analysis of mouse stem cells and early embryos. PLoS Biol. 1:E74 10.1371/journal.pbio.0000074 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen P.S., Park J., Qin Y., Li X., Parsawar K., Larson M.H., Cox J., Cheng Y., Lambowitz A.M., Weissman J.S., Brandman O., and Frost A.. 2015. Rqc2p and 60S ribosomal subunits mediate mRNA-independent elongation of nascent chains. Science (80-.). 347:75–78. doi:. 10.1126/science.1259724 [DOI] [PMC free article] [PubMed]
- Shi Z., Fujii K., Kovary K.M., Genuth N.R., Röst H.L., Teruel M.N., and Barna M.. 2017. Heterogeneous Ribosomes Preferentially Translate Distinct Subpools of mRNAs Genome-wide. Mol. Cell. 67:71–83.e7. 10.1016/j.molcel.2017.05.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sibbald S.J., Hopkins J.F., Filloramo G.V., and Archibald J.M.. 2019. Ubiquitin fusion proteins in algae: implications for cell biology and the spread of photosynthesis. BMC Genomics. 20:38 10.1186/s12864-018-5412-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Signer R.A.J., Magee J.A., Salic A., and Morrison S.J.. 2014. Haematopoietic stem cells require a highly regulated protein synthesis rate. Nature. 509:49–54. 10.1038/nature13035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simsek D., and Barna M.. 2017. An emerging role for the ribosome as a nexus for post-translational modifications. Curr. Opin. Cell Biol. 45:92–101. 10.1016/j.ceb.2017.02.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simsek D., Tiu G.C., Flynn R.A., Byeon G.W., Leppek K., Xu A.F., Chang H.Y., and Barna M.. 2017. The Mammalian Ribo-interactome Reveals Ribosome Functional Diversity and Heterogeneity. Cell. 169(6):1051–1065.e18. 10.1016/j.cell.2017.05.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skaletsky H., Kuroda-Kawaguchi T., Minx P.J., Cordum H.S., Hillier L., Brown L.G., Repping S., Pyntikova T., Ali J., Bieri T., et al. . 2003. The male-specific region of the human Y chromosome is a mosaic of discrete sequence classes. Nature. 423:825–837. 10.1038/nature01722 [DOI] [PubMed] [Google Scholar]
- Slaidina M., and Lehmann R.. 2014. Translational control in germline stem cell development. J. Cell Biol. 207:13–21. 10.1083/jcb.201407102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slavov N., Semrau S., Airoldi E., Budnik B., and van Oudenaarden A.. 2015. Differential Stoichiometry among Core Ribosomal Proteins. Cell Rep. 13:865–873. 10.1016/j.celrep.2015.09.056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sloan K.E., Warda A.S., Sharma S., Entian K.-D., Lafontaine D.L.J., and Bohnsack M.T.. 2017. Tuning the ribosome: The influence of rRNA modification on eukaryotic ribosome biogenesis and function. RNA Biol. 14:1138–1152. 10.1080/15476286.2016.1259781 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soshnev A.A., Josefowicz S.Z., and Allis C.D.. 2016. Greater Than the Sum of Parts: Complexity of the Dynamic Epigenome. Mol. Cell. 62:681–694. 10.1016/j.molcel.2016.05.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugihara Y., Sadohara E., Yonezawa K., Kugo M., Oshima K., Matsuda T., and Nadano D.. 2013. Identification and expression of an autosomal paralogue of ribosomal protein S4, X-linked, in mice: potential involvement of testis-specific ribosomal proteins in translation and spermatogenesis. Gene. 521:91–99. 10.1016/j.gene.2013.02.040 [DOI] [PubMed] [Google Scholar]
- Sundaramoorthy E., Leonard M., Mak R., Liao J., Fulzele A., and Bennett E.J.. 2017. ZNF598 and RACK1 Regulate Mammalian Ribosome-Associated Quality Control Function by Mediating Regulatory 40S Ribosomal Ubiquitylation. Mol. Cell. 65:751–760.e4. 10.1016/j.molcel.2016.12.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sung M.K., Porras-Yakushi T.R., Reitsma J.M., Huber F.M., Sweredoski M.J., Hoelz A., Hess S., and Deshaies R.J.. 2016. A conserved quality-control pathway that mediates degradation of unassembled ribosomal proteins. eLife. 5:e19105 10.7554/eLife.19105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Talbert P.B., and Henikoff S.. 2017. Histone variants on the move: substrates for chromatin dynamics. Nat. Rev. Mol. Cell Biol. 18:115–126. 10.1038/nrm.2016.148 [DOI] [PubMed] [Google Scholar]
- Taoka M., Nobe Y., Yamaki Y., Yamauchi Y., Ishikawa H., Takahashi N., Nakayama H., and Isobe T.. 2016. The complete chemical structure of Saccharomyces cerevisiae rRNA: partial pseudouridylation of U2345 in 25S rRNA by snoRNA snR9. Nucleic Acids Res. 44:8951–8961. 10.1093/nar/gkw564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tatsumi K., Yamamoto-Mukai H., Shimizu R., Waguri S., Sou Y.S., Sakamoto A., Taya C., Shitara H., Hara T., Chung C.H., et al. . 2011. The Ufm1-activating enzyme Uba5 is indispensable for erythroid differentiation in mice. Nat. Commun. 2:181 10.1038/ncomms1182 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tseng H., Chou W., Wang J., Zhang X., Zhang S., and Schultz R.M.. 2008. Mouse ribosomal RNA genes contain multiple differentially regulated variants. PLoS One. 3:e1843 10.1371/journal.pone.0001843 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tusup M., Kundig T., and Pascolo S.. 2018. Epitranscriptomics of cancer. World J. Clin. Oncol. 9:42–55. 10.5306/wjco.v9.i3.42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uechi T., Nakajima Y., Nakao A., Torihara H., Chakraborty A., Inoue K., and Kenmochi N.. 2006. Ribosomal protein gene knockdown causes developmental defects in zebrafish. PLoS One. 1:e37 10.1371/journal.pone.0000037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vasilyev N., Polonskaia A., Darnell J.C., Darnell R.B., Patel D.J., and Serganov A.. 2015. Crystal structure reveals specific recognition of a G-quadruplex RNA by a β-turn in the RGG motif of FMRP. Proc. Natl. Acad. Sci. USA. 112:E5391–E5400. 10.1073/pnas.1515737112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voigt F., Zhang H., Cui X.A., Triebold D., Liu A.X., Eglinger J., Lee E.S., Chao J.A., and Palazzo A.F.. 2017. Single-Molecule Quantification of Translation-Dependent Association of mRNAs with the Endoplasmic Reticulum. Cell Rep. 21:3740–3753. 10.1016/j.celrep.2017.12.008 [DOI] [PubMed] [Google Scholar]
- Volta V., Beugnet A., Gallo S., Magri L., Brina D., Pesce E., Calamita P., Sanvito F., and Biffo S.. 2013. RACK1 depletion in a mouse model causes lethality, pigmentation deficits and reduction in protein synthesis efficiency. Cell. Mol. Life Sci. 70:1439–1450. 10.1007/s00018-012-1215-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang M., and Lemos B.. 2017. Ribosomal DNA copy number amplification and loss in human cancers is linked to tumor genetic context, nucleolus activity, and proliferation. PLoS Genet. 13:e1006994 10.1371/journal.pgen.1006994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warner J.R. 1999. The economics of ribosome biosynthesis in yeast. Trends Biochem. Sci. 24:437–440. 10.1016/S0968-0004(99)01460-7 [DOI] [PubMed] [Google Scholar]
- Warner J.R., and McIntosh K.B.. 2009. How common are extraribosomal functions of ribosomal proteins? Mol. Cell. 34:3–11. 10.1016/j.molcel.2009.03.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watanabe-Susaki K., Takada H., Enomoto K., Miwata K., Ishimine H., Intoh A., Ohtaka M., Nakanishi M., Sugino H., Asashima M., and Kurisaki A.. 2014. Biosynthesis of ribosomal RNA in nucleoli regulates pluripotency and differentiation ability of pluripotent stem cells. Stem Cells. 32:3099–3111. 10.1002/stem.1825 [DOI] [PubMed] [Google Scholar]
- Wong Q.W.L., Li J., Ng S.R., Lim S.G., Yang H., and Vardy L.A.. 2014. RPL39L is an example of a recently evolved ribosomal protein paralog that shows highly specific tissue expression patterns and is upregulated in ESCs and HCC tumors. RNA Biol. 11:33–41. 10.4161/rna.27427 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woolnough J.L., Atwood B.L., Liu Z., Zhao R., and Giles K.E.. 2016. The regulation of rRNA gene transcription during directed differentiation of human embryonic stem cells. PLoS One. 11:e0157276 10.1371/journal.pone.0157276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue S., Tian S., Fujii K., Kladwang W., Das R., and Barna M.. 2015. RNA regulons in Hox 5′ UTRs confer ribosome specificity to gene regulation. Nature. 517:33–38. 10.1038/nature14010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanagitani K., Juszkiewicz S., and Hegde R.S.. 2017. UBE2O is a quality control factor for orphans of multiprotein complexes. Science. 357:472–475. 10.1126/science.aan0178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanagiya A., Delbes G., Svitkin Y.V., Robaire B., and Sonenberg N.. 2010. The poly(A)-binding protein partner Paip2a controls translation during late spermiogenesis in mice. J. Clin. Invest. 120:3389–3400. 10.1172/JCI43350 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yano K., Wada T., Suzuki S., Tagami K., Matsumoto T., Shiwa Y., Ishige T., Kawaguchi Y., Masuda K., Akanuma G., et al. . 2013. Multiple rRNA operons are essential for efficient cell growth and sporulation as well as outgrowth in Bacillus subtilis. Microbiology. 159:2225–2236. 10.1099/mic.0.067025-0 [DOI] [PubMed] [Google Scholar]
- Yonashiro R., Tahara E.B., Bengtson M.H., Khokhrina M., Lorenz H., Chen K.C., Kigoshi-Tansho Y., Savas J.N., Yates J.R., Kay S.A., et al. . 2016. The Rqc2/Tae2 subunit of the ribosome-associated quality control (RQC) complex marks ribosome-stalled nascent polypeptide chains for aggregation. eLife. 5:e11794 10.7554/eLife.11794 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon A., Peng G., Brandenburg Y., Zollo O., Xu W., Rego E., and Ruggero D.. 2006. Impaired control of IRES-mediated translation in X-linked dyskeratosis congenita. Science. 312:902–906. 10.1126/science.1123835 [DOI] [PubMed] [Google Scholar]
- You K.T., Park J., and Kim V.N.. 2015. Role of the small subunit processome in the maintenance of pluripotent stem cells. Genes Dev. 29:2004–2009. 10.1101/gad.267112.115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu S., and Lemos B.. 2016. A portrait of ribosomal DNA contacts with Hi-C reveals 5S and 45S rDNA anchoring points in the folded human genome. Genome Biol. Evol. 8:3545–3558. 10.1093/gbe/evw257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y., Duc A.C.E., Rao S., Sun X.L., Bilbee A.N., Rhodes M., Li Q., Kappes D.J., Rhodes J., and Wiest D.L.. 2013. Control of hematopoietic stem cell emergence by antagonistic functions of ribosomal protein paralogs. Dev. Cell. 24:411–425. 10.1016/j.devcel.2013.01.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Q., Shalaby N.A., and Buszczak M.. 2014. Changes in rRNA transcription influence proliferation and cell fate within a stem cell lineage. Science. 343:298–301. 10.1126/science.1246384 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang M., Zhu X., Zhang Y., Cai Y., Chen J., Sivaprakasam S., Gurav A., Pi W., Makala L., Wu J., et al. . 2015. RCAD/Ufl1, a Ufm1 E3 ligase, is essential for hematopoietic stem cell function and murine hematopoiesis. Cell Death Differ. 22:1922–1934. 10.1038/cdd.2015.51 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y., O’Leary M.N., Peri S., Wang M., Zha J., Melov S., Kappes D.J., Feng Q., Rhodes J., Amieux P.S., et al. . 2017. Ribosomal Proteins Rpl22 and Rpl22l1 Control Morphogenesis by Regulating Pre-mRNA Splicing. Cell Rep. 18:545–556. 10.1016/j.celrep.2016.12.034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou X., Liao W.J., Liao J.M., Liao P., and Lu H.. 2015. Ribosomal proteins: functions beyond the ribosome. J. Mol. Cell Biol. 7:92–104. 10.1093/jmcb/mjv014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zismanov V., Chichkov V., Colangelo V., Jamet S., Wang S., Syme A., Koromilas A.E., and Crist C.. 2016. Phosphorylation of eIF2α Is a Translational Control Mechanism Regulating Muscle Stem Cell Quiescence and Self-Renewal. Cell Stem Cell. 18:79–90. 10.1016/j.stem.2015.09.020 [DOI] [PubMed] [Google Scholar]