Abstract
Partial gene sequencing for the bovine coronavirus at the World Genebank is available for many countries, which are distributed unevenly in five continents, but so far, no sequencing of strains has been recorded in Iran. One hundred ninety‐four stool samples from calves with diarrhoea less than one‐month old were collected from five different geographical regions of country in order to detect coronavirus and characterize it if coronavirus was found. Samples were screened for the presence of BCoV by using a commercially available ELISA kit. Furthermore, RT‐PCR was carried out on positive samples for confirmation of the presence of N and S specific genes. Sequencing and phylogenetic analysis was carried out following RT‐PCR tests. 7.2% of samples, were positive for BCoV and all stool samples from the South‐West, Northeast and West regions of Iran were negative. The results showed that all the strains of coronavirus identified in Iran were completely in independent clusters and that they did not stand in the same cluster as any of the strains identified in other parts of the world. The strains from Iran were quite different from strains in other parts of the world but from the point of similarity these viruses showed some similarities to the European strains, such as those found in France, Croatia, Denmark and Sweden.
Keywords: bovine coronavirus, calf diarrhoea, Iran, Phylogenetic analysis, RT‐PCR
This is the first report of sequencing and phylogeneticic analysis of bovine coronavirus strains isolated from neonatal calf diarrhea in Iran. Sequencing and phylogenetic analysis of BCoV showed that coronaviruses in Iran were quite different from known Bovine coronaviruses (BCoV) in other parts of the world. The phylogenetic tree of gene N indicated that all the viruses identified in Iran were completely in independent clusters and that they did not stand in the same cluster with any of the coronaviruses identified in other parts of the world. Regarding the gene S phylogenetic tree, the coronaviruses were divided into four general groups, with all strains from Iran being grouped together which was different from BCoV isolated in other parts of the world.
1. INTRODUCTION
Bovine coronavirus (BCoV) is one of the viruses that can cause diarrhoea in beef and dairy calves during the neonatal periods. Diarrhoea associated with BCoV has a worldwide distribution and occurs from 1 day to 3 months of age but mostly between 1 and 2 weeks of age (Constable, Hinchcliff, Done, & Gruenberg, 2017). The risk of infection decreases with age. Disease leads to significant economic losses both in beef and dairy industry (Afshari Safavi, Mohammadi, Rad, & Naghibi, 2012; Bok et al., 2015). Bovine coronavirus belongs to the order of the Nidovirales of the Coronaviridae family in the genus Betacoronavirus and the species Betacoronavirus 1 (International committee on taxonomy of viruses, 2012). The virus can be shed in stool of up to 70% of adult cows, despite the presence of specific antibodies in their serum and faeces (Constable et al., 2017). Calves born from infected cows have a higher risk of diarrhoea (Oma, Tråvén, Alenius, Myrmel, & Stokstad, 2016). Faecal‐oral transmission is the primary infection method, but transmission through aerosols may also occur. Bovine coronavirus is detected in wild ruminants with diarrhoea (Tsunemitsu, el‐Kanawati, Smith, Reed, & Saif, 1995). The transmission of the virus to the herd can either be done directly through the arrival of the new cow or indirectly through personnel or contaminated equipment (Constable et al., 2017).
An important epidemiological characteristic of coronavirus infections in newborn calves is that protection against disease is dependent on the presence of specific colostral antibody in the lumen of the intestine of the newborn. Colostral antibody in serum does not directly protect but contributes to mucosal immunity through re‐secretion into the gut lumen. Protection against clinical disease depends on the amount of immunoglobulin in the lumen of the intestine (Constable et al., 2017). Protection from coronavirus infection occurs as long as there is colostrum antibody in the lumen of the intestine, and this explains why coronavirus diarrhoea commonly occurs between 1– 2 weeks of age. Survival from coronaviral diarrhoea in calves depends on high levels of serum antibody (Butaso et al., 1998; Godet, Grosclaude, Delmas, & Laude, 1994; Izzo et al., 2011). Bovine coronavirus is associated with three known syndromes in cattle: winter dysentery in adult cows, respiratory disease of different ages and diarrhoea in neonatal calves (MacLachlan & Dubovi, 2017; Oma et al., 2016). Epithelial cells in the respiratory tract, enterocytes in the distal small intestine and in colon are the primary sites of BCoV replication. It is hypothesized that initial replication of BCoV in the respiratory tract seeds virus to intestinal tissue after the swallowing of mucus containing virus. However, if there is a high viral load in the environment due to presence of large number of infected animals it could also be a direct source of infection for the digestive tract (Saif, Redman, Moorhead, & Theil, 1986). Diarrhoea is caused by the destruction of the mature absorbing cells of the intestinal villi as well as generating crypt cells in small intestine, and also the mucosal surface in the large intestine (MacLachlan & Dubovi, 2017).
Coronaviruses are large enveloped viral particles containing a positive sense single stranded RNA genome (26–30 kb), coding for several structural proteins, including nucleocapsid (N), membrane (M), haemagglutinin‐esterase (HE), spike (S) proteins and several non‐structural proteins (NSPs) like polymerase (Pol) (Gunn, Collins, O’Connell, & O’Shea, 2015).
The S glycoprotein harbours domains responsible for receptor binding and induction of neutralizing antibodies and is the most polymorphic viral protein among CoV species and also among strains of the same species. It is utilized for the molecular characterization of the isolates. The S glycoprotein consists of two subunits, S1 (N‐terminal) and S2 (C‐terminal). The S1 hypervariable region is useful for studying the variability and evolution of this virus (Bok et al., 2015; Brandão et al., 2006; Hasoksuz, Sreevatsan, Cho, Hoet, & Saif, 2002). The S1 subunit is responsible for virus binding to host‐cell receptors (Godet et al., 1994; Kubo, Yamada, & Taguchi, 1994), induction of neutralizing antibody (Yoo & Deregt, 2001) and haemagglutinating activity (Schultze, Gross, Brossmer, & Herrler, 1991). Its sequences are variable and mutations in this region have been associated with altered antigenicity and virus pathogenicity (Fazakerley, Parker, Bloom, & Buchmeier, 1992; Hingley, Gombold, Lavi, & Weiss, 1994). On the other hand, the sequences of the S2 subunit are conserved and are responsible for membrane fusion activity (Luo & Weiss, 1998; Yoo & Derget, 2001).
The BCoV N protein is a 50–60 KD phosphoprotein that is bound to viral genomic RNA to form the helical nucleocapsid. N protein may play a role in replication of viral RNA since the antibody directed against the N protein inhibits the in vitro RNA polymerase chain reaction (Khalili, Morshedi, Keyvanfar, & Hemmatzadeh, 2006). The coronavirus nucleocapsid (N) is a structural protein that forms complexes with genomic RNA, interacts with the viral membrane protein during virion assembly and plays a critical role in enhancing the efficiency of virus transcription and assembly (Perlman & Netland, 2009).
Little epidemiological information is available regarding BCoV detection, incidence and its characterization in cattle in Iran. The aim of this study was to evaluate the molecular epidemiology of bovine coronaviruses in Iran, as well as to compare the two genes coding for N and S proteins, in a phylogenetic study of coronaviruses strains, and also to compare strains from Iran with other countries.
2. MATERIALS AND METHODS
2.1. Samples
A total of 194 stool samples were collected from calves less than one month old with diarrhoea during May 2014 to August 2016. Ninety‐six diarrheic stool samples from calves under seven days old and 98 samples from calves more than seven days old consisting of 48 samples between 8 to 14 days and 40 specimens between 15 to 30 days were obtained. The consistency of stool samples was recorded based on the scoring system 0–3, defined as follows: faecal score of 0 = normal consistency, 1 = semi‐formed or pasty, 2 = loose faeces and 3 = watery faeces; calves with a faecal score of 2 or 3 were classified as positive for diarrhoea (Graham, Renaud, Duffield, & Kelton, 2018). In this study a total of 27 industrial and traditional dairy farms located in five geographical regions of the country, based on the climate conditions and geographical distance were sampled. These five geographical regions including: North, West, Southwest, Center and Northeast of country which comprised of 10 provinces Tehran, Qom, Qazvin, Markazi (city of Saveh), Alborz, Khuzestan (city of Ahvaz), Khorasan Razavi (city of Mashhad), Golestan (Gorgan), Isfahan and Kermanshah (Figure 1).
Figure 1.
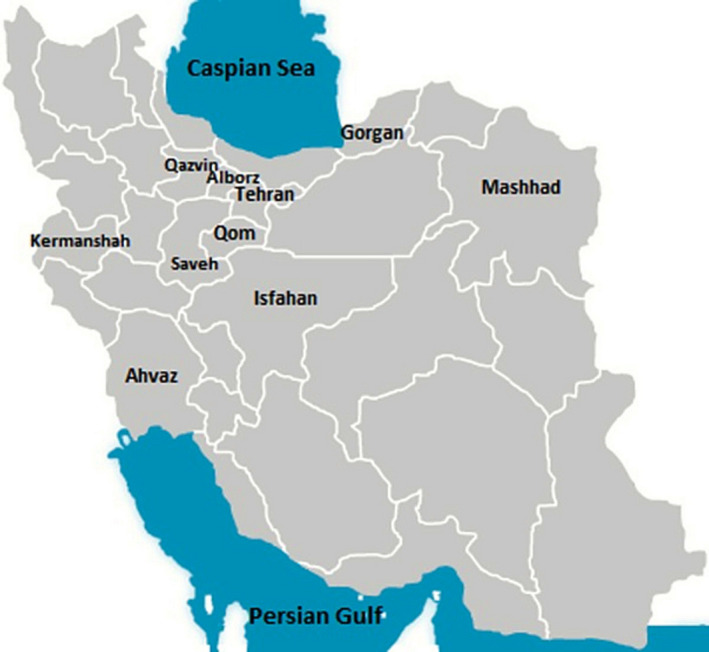
Map of Iran and 10 provinces were targeted for sampling based on their dairy farms’ density and geographical significance. Considering of climate condition and geographical distance, five distinct geographical areas were defined, and the provinces from which sampling occurred were divided into these areas; the North area namely the south of Alborz mountain ranges area (including Tehran, Alborz and Qazvin), the Northeast area (including Mashahd and Gorgan), the Central area of Iran (including Saveh, Qom and Isfahan), the Southwest area (Ahvaz) and finally the West area (including Kermanshah)
2.2. Laboratory tests
All samples were examined by using an antigen ELISA (antigen captured ELISA, IDEXX Rota‐Corona‐k99, USA) test. Positive samples in ELISA, were chosen for RT‐PCR and sequencing of the N and S genes. Finally, 7 and 5 positive samples from the PCR test were selected based on the differences in herds and geographical area, for phylogenetic analysis of the N and S genes.
2.3. Primers
Primers were used with a little change based on two papers by Hasoksuz et al., (2002) and also Liu et al., (2006).
Based on these papers, the oligonucleotide primers were designed from the S glycoprotein gene of the BCoV Mebus strain (GenBank accession No. M31053) used for DNA sequencing (Table 1), and for N gene, the oligonucleotide primers were designed from the published sequence of N gene of Mebus strain (GenBank accession No. U00735). The sequences of the primers are shown in Table 1.
Table 1.
The sequences of the primers designed for the S and N genes, the predicted RT‐PCR product size and their positions in genes S and N
| Target gene | Primer | Sequence (5′−3′) | Position | Product length | Reference no. |
|---|---|---|---|---|---|
| Nucleocapsid | BCV‐N‐f | GCCGATCAGTCCGACCAATC | 29475–29494 | 407 | Masaharu et al (2012) |
| Nucleocapsid | BCV‐N‐R | AGAATGTCAGCCGGGGTAT | 29881–29863 | ||
| S |
S1AF a |
5’‐ATG TTT TTG ATA CTT TTA ATT−3’ | 1–21 | 633 | Hasoksuz et al. (2002); Liu et al. (2006) |
| S | S1AR b | 5’‐AGT ACC ACC TTC TTG ATA AA−3’ | 654–635 |
The sequences of primers, the predicted RT‐PCR product size, the sequences of primers of gene S and its position in gene S.
F: upstream primer.
R: downstream primer.
2.4. RNA extraction and reverse transcription
RNA from faeces was extracted using RNX‐Plus solution for total RNA isolation (Sinaclon Bioscience, Iran) as instructed by the manufacturer. Then the complementary DNA (cDNA) was produced by using the Maxime RT premix kit (Intron biotechnology, Korea) as instructed by the manufacturer.
The PCR conditions used were based on the two papers by of Hasoksuz, Kayar, Dodurka, and Ilgaz (2005) as well as Liu et al., (2006) for the gene S and the study of Fukuda et al., (2012) for the gene N. Briefly after initial denaturation at 94 C for 5 min, cDNAs entered to 40 cycles, each one with three steps including denaturation at 94 C for 45 s, primer annealing at 52 C (for N gene) or 50 C (for S gene) for 45 s, extension at 72 C for 1 min that finally followed by final extension step at 72 C for 10 min.
The PCR products were visualized on 1.5% agarose gel stained with ethidium bromide. PCR the products 407 bp and 654 bp were detected for genes N and S, respectively.
2.5. Sequencing and phylogenetic analysis
An AccuPrep® PCR Purification Kit (Bioneer) was used for the purification of the PCR products. Sequencing was with the primers (both directions) that were used after PCR (Bioneer).
The data were then edited using CLC Sequence Viewer 6 software. Phylogenetic studies were also performed using MEGA7 (version 7.0.21; BioDesign Institute, Tempe, USA) software based on Neighbour‐joining (Kumar, Nei, Dudley, & Tamura, 2008; Tamura, Dudley, Nei, & Kumar, 2007).
Statistical evaluation was performed using SPSS 20.0 software. The Chi square test was used to examine the statistical differences between different variables.
Data of sequenced PCR products were submitted in the GenBank database with following accession numbers: MG386279 (IRI‐Tehran), MG859967 (IRI‐Isfahan‐2), MG859968 (IRI‐Isfahan‐1), MG859969 (IRI‐Alborz), MG859970 (IRI‐Qom), MG859971 (IRI‐Saveh), MG859972 (IRI‐Qazvin), MG386280 (IRI‐Saveh‐S), MG859973 (IRI‐Qom‐s), MG859974 (IRI‐Isfahan‐s), MG859975 (IRI‐Tehran‐S), MG859976 (IRI‐Alborz‐S).
A Maximum Likelihood tree based on partial length of the nucleocapsid gene, isolates collected from this study are represented in Figure 2. The evolutionary history was inferred using the Neighbour‐Joining method. The optimal tree with a sum of branch length = 0.13257831 is shown. A Maximum Likelihood tree based on the partial length of the spike gene, isolates collected from this study are represented in Figure 3. The evolutionary history was inferred using the Neighbour‐Joining method. The optimal tree with a sum of branch length = 0.13257831 is shown.
Figure 2.
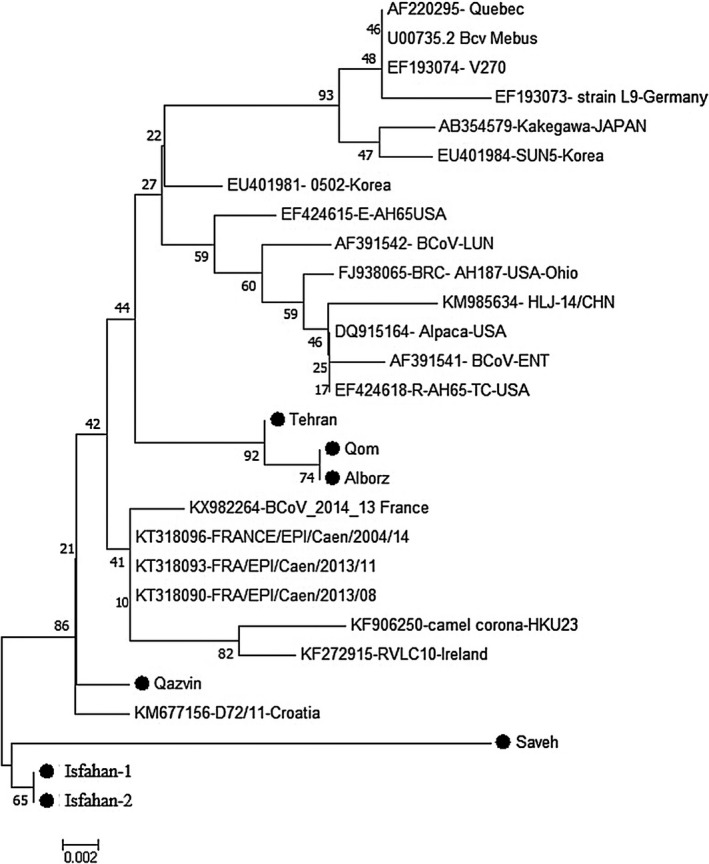
Evolutionary relationships of taxa, Maximum Likelihood tree based on partial length of the nocleucapsid gene, Isolates from this study are represented by a filled circle (●). The evolutionary history was inferred using the Neighbour‐Joining method. The optimal tree with the sum of branch length = 0.13257831 is shown. The percentage of replicate trees in which the associated taxa clustered together in the bootstrap test (1,000 replicates) are shown next to the branches
Figure 3.
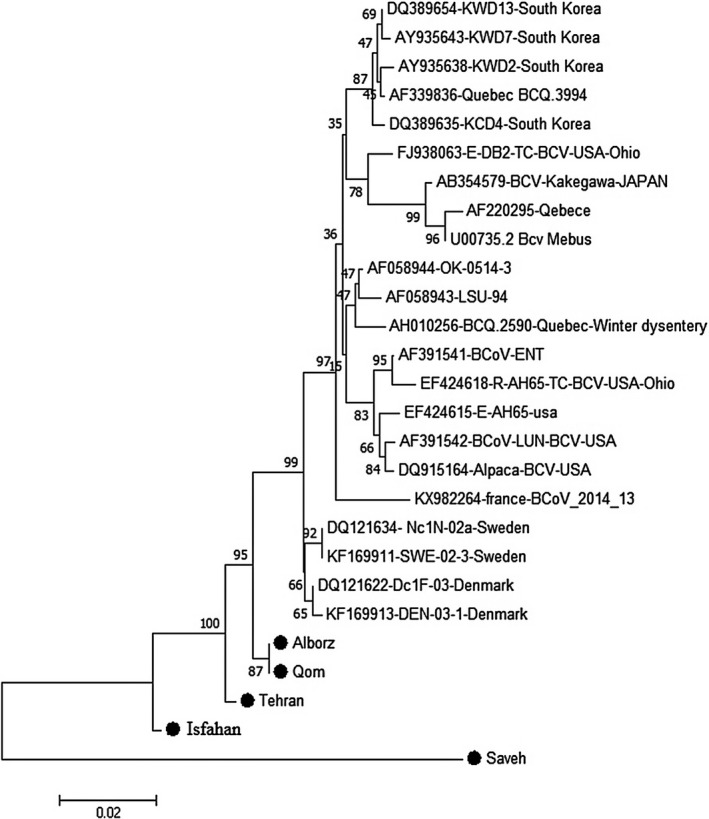
Evolutionary relationships of taxa, Maximum Likelihood tree based on partial length of the spike gene, Isolates from this study are represented by a filled circle (●). The evolutionary history was inferred using the Neighbour‐Joining method. The optimal tree with the sum of branch length = 0.13257831 is shown. The percentage of replicate trees in which the associated taxa clustered together in the bootstrap test (1,000 replicates) are shown next to the branches
3. RESULTS
From 194 faecal samples tested with ELISA, 14 samples (7.2%) were positive for the presence of coronavirus antigens. The results of the experiments are shown in Table 2. The incidences of coronavirus in cities with positive samples were as follows: Tehran 7.5%, Qom, Qazvin and Karaj 20%, Isfahan 10%, and the provinces of Markazi (Saveh) and Khuzestan (Ahvaz) 8.3%. The mean age of coronavirus positive calves was 9 days, from which 36% were males and 64% were females. Of all sampled calves (194 samples), 10.2% (10 samples of 98 samples) of the calves ≥ 7 days of age, were positive for BCoV, whereas only 4.2% (4 samples of 96 samples) of the calves with age of 0–7 days were positive for BCoV. Statistical analysis using student's t‐test showed that coronavirus positive cases were significantly higher in calves over seven days old than those under seven days old (p < .05). Mean value of faecal score in the group of calves with positive ELISA result for BCoV (14 samples) was 2.5 and for calves with a negative result for BCoV (180 samples) was 2.1.
Table 2.
Results of ELISA test on stool samples of diarrheic calves in different cities, sex and days of age
| City | No | Positive | Negative | Female | Male | ≤7 days age | ≥7 days age | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No | % | No | % | Positive | Negative | Positive | Negative | Positive | Negative | Positive | Negative | ||
| Tehran | 40 | 3 | 7.5 | 37 | 92.5 | 1 | 24 | 2 | 13 | 1 | 19 | 2 | 18 |
| Isfahan | 30 | 3 | 10 | 27 | 90 | 1 | 17 | 2 | 10 | 1 | 12 | 2 | 15 |
| Karaj | 10 | 2 | 20 | 8 | 80 | 0 | 5 | 2 | 3 | 1 | 4 | 1 | 4 |
| Qom | 10 | 2 | 20 | 8 | 80 | 1 | 4 | 1 | 4 | 0 | 3 | 2 | 5 |
| Qazvin | 10 | 2 | 20 | 8 | 80 | 1 | 4 | 1 | 4 | 0 | 4 | 2 | 4 |
| Saveh | 12 | 1 | 8.3 | 11 | 91.6 | 0 | 6 | 1 | 5 | 1 | 2 | 0 | 9 |
| Ahvaz | 12 | 1 | 8.3 | 11 | 91.6 | 1 | 5 | 0 | 6 | 0 | 4 | 1 | 7 |
| Other cities | 70 | 0 | 0 | 70 | 100 | 0 | 33 | 0 | 37 | 0 | 44 | 0 | 26 |
| Total | 194 | 14 | 7.21 | 180 | 92.18 | 5 | 98 | 9 | 82 | 4 | 92 | 10 | 88 |
The products of 407 bp and 654 bp were detected for gene N and S, respectively.
The evolution trees of the coronavirus positive samples identified in this study are based on the sequences of the N gene and S genes, in comparison with sequences derived from GeneBank, which are constructed using a neighbouring nucleotide sequence based on a sorted nucleotide sequence (Figures 2 and 3). The results of the phylogenetic tree for gene N (Figure 2) indicate that all the strains identified in Iran were completely in independent clusters and that they did not cluster with any of the strains identified in other parts of the world and or with by reference strains such as the Mebus and Quebec. Regarding the gene S phylogenetic tree (Figure 3), the coronaviruses were divided into four general groups, with all strains from Iran being grouped together and separate from BCoV isolated in other parts of the world.
Based on the distance matrix analysis, the least distance in gene N belonged to Tehran isolate and Qom and Alborz (0.003), subsequently Qazvin and French isolates (0.006), and then between Isfahan and Qazvin (0.009), and Isfahan and French isolates (0.009). and base on gene S, the least distance was detected between Qom and Sweden, then Denmark isolates (0.017 and 0.015 respectively) and at the next level, between USA‐Ohio isolates and Qom, Tehran and Alborz isolates (0.03).
4. DISCUSSION
In this study, the occurrence of coronavirus in neonatal calf diarrhoea was 7.2%. The nearly same results have been documented in some studies. For example, Pérez et al., (1998) in Costa Rica using DOT‐ELISA, found that coronavirus was present in stool samples of 7% of scouring calves. In another study in India from 45 necropsied calves, four (8.88%) cases were found to be positive for coronavirus according to RT‐PCR (Singh et al., 2020). Castell et al., (2019) in Uruguay using real‐time polymerase chain reaction (PCR) targeted the S gene, detected BCoV in 7.8% (64 of 824 samples) of faeces (763) and intestinal contents (61) from beef and dairy cattle.
In contrast, the occurrence of coronavirus in some other studies is higher than in our study, which includes studies in the countries of: France (16.5% and is significantly associated with diarrhoea) (Bendali, Bichet, Schelcher, & Sanaa, 1999), Brazil (15.6% by using semi‐nested (SN) PCR) (Stipp et al., 2009), India (14% with ELISA and about 20% by RT‐PCR) (Hansa, Rai, Yaqoob Wani, & Dhama, 2012), Australia (21.6% in dairy calves with diarrhoea by using RT‐PCR) (Izzo et al., 2011), South Korea ( 15.6% of diarrhoea samples using RT‐PCR and nested PCR) (Park et al., 2007), and Turkey (37.1% in 0–30 days old calves by indirect antigen‐capture ELISA) (Hasoksuz et al., 2005). Bok et al. (2015) in a study in Argentina using an ELISA test, reported that the virus was present in 12.13% (29/239) of diarrhoea cases of dairy calves.
Some other studies also suggested an even lower prevalence than in the current study, as reported by Ali Khan et al., (2009) in Pakistan (2%). Bartels, Holzhauer, Jorritsma, Swart, and Lam (2010) tested 424 calves with non‐normal faeces (‘custard‐like’‐yellowish‐coloured with custard consistency or diarrhoea) from 108 Dutch dairy herds for shedding coronavirus using a tetrakit assay which had 88.9% sensitivity and 98.7% specificity relative to an ELISA test. The prevalence of coronavirus in their samples was 2.8% (1.6%‐ 5.2%). In another study in Japan, Kirisawa, Takeyama, Koiwa, and Iwai (2007) used nested RT‐PCR, and observed that virus presented in 2 samples out of 99 from calves with diarrhoea. Singh et al., (2019) tested a total of 816 infected dairy calves with diarrhoea below 3 months of age in Central and North India by using sandwich antigen ELISA. A total of 7 (0.85%) cases tested positive for BCoV.
In the present study, with increasing consistency of stool score, the incidence of coronavirus was increased, so that the mean score of coronavirus positive and negative samples was 2.5 and 2.1, respectively, although this difference was not statistically significant (p > .05). In some studies, (Bjorkman, Svensson, Christensson, & Verdier, 2003; Busato et al., 1998; Erdogan, Unver, Gunes, & Citil, 2003), there was not any significant correlation between the presence of coronavirus in faeces and clinical diarrhoea; in contrast, in some other studies, a significant correlation between these two parameters was reported (Bendali et al., 1999; Pérez et al., 1998; Reynolds et al., 1986; Stipp et al., 2009).
Of the samples testing positive for coronavirus (14) the prevalence of BCoV 28.5% were in calves less than 7 days old (4 samples out of 14 positive samples), and 71.4% (10 samples out of 14 positive) were in calves more than 7 days old. In a previous study the highest percentage of positive samples was found in 15–30 day old calves (29%) (Stipp et al., 2009).
Gene partial sequencing (not complete) for the bovine coronavirus at the World Genebank is available for new countries, which are distributed unevenly in five continents (Kin et al., 2016), but so far, no sequencing of strains has been recorded in Iran and the current study is the first comprehensive study in this field.
In the present study, the results of the phylogenetic tree of gene N (Figure 2) indicate that all the strains identified in Iran were completely in independent clusters and that they did not stand in the same cluster as any of the strains identified in other parts of the world or with reference strains such as the Mebus and Quebec. But in terms of proximity and similarity, it can be said that the virus identified from Qazvin province is close to a virus detected in Croatia (Figure 2). However, the viruses identified in Isfahan and Saveh are not close to any other bovine coronaviruses in the world. Amongst the viruses identified in Iran, it can be said that the strain identified in Saveh has the most difference from other strains identified in Iran and other countries, but among the Iranian strains in this study it most closely resembles the Isfahan strain. The Qom, Alborz, and Tehran strains are also in one cluster, and this is also logical in regional terms.
Regarding the gene S phylogenetic tree (Figure 3), the coronaviruses are divided into four general groups, all strains of Iran being grouped together with strains from Switzerland and Denmark being in a separate group. The reference strains and South Korea strains are also in a separate group and strains from the United States constitute another cluster. Liu et al., in 2006, showed a correlation between Swedish and Danish samples from calves with respiratory symptoms and/ or diarrhoea based on the phylogenetic analysis of the S gene. They showed that some strains of the virus had been established in the herds for a long time, while in other herds, samples taken at different time points revealed different groups of viruses which are the evidence for the introduction new viruses into the herd (Liu et al., 2006). The same results were recorded in our study, and we found that strains of Iran were very close to the strains of the Sweden and Denmark.
In a study conducted by Nathalie et al., N‐genes in the United States and South Korea were roughly contained in a cluster, reference strains such as Mebus and Quebec were in a cluster and strains from France were also located in a separate cluster. The same results were recorded in the present study, and Iranian strains form a separate cluster, but were more closely aligned with the strains studied in France (Natalie et al., 2016).
In a study conducted in Croatia, bovine coronavirus was detected in 78.8% of stool samples of cattle with clinical symptoms. The sequencing of the N and S genes in that study showed the proximity and stratification of Croatian strains to the strains of Denmark and Italy (Lojkić et al., 2015). In our study, there was a proximity of the N gene between Iranian and Croatian strains as well as Denmark strains.
Studies in South Korea in 2004 and 2005 showed that although the sampled animals had different clinical symptoms (winter dysentery, respiratory symptoms and calves’ diarrhoea), the isolated coronaviruses were highly genetically related and are classified in very well‐defined groups in comparison to reference strains from the United States and Canada. They concluded that circulating BCoV in South Korea evolved from a different evolutionary pathway in comparison to the reference strains (Park et al., 2007).
Takiuchi, Alfieri, and Alfieri (2008) showed that all S1‐encoding genes of Brazilian bovine coronaviruses differ from the Mebus strain (97.8% identity for nucleotides and 96.8% identity for amino acids) and were most similar to American BCoV‐ ENT, strain 182NS and other Canadian strains (98.7% for nucleotides and 98.7% for amino acids).
According to a study conducted in Argentina from seven selected samples, it has been shown that BCoV strains circulating in the farms are from a different cluster from other geographical egions. Interestingly, the Argentine strains are far away from the BCoV strain of the historical reference (at both the nucleotide and amino acid levels) (Bok et al., 2015). The spike gene's phylogenetic analysis showed that the Irish strain cluster is in the same group with new strains of BCoV from Europe, which probably represents a similar evolutionary pathway. Direct sequencing analysis showed the amino acid changes in the spike protein that were specific to the Irish strains (Gunn et al., 2015). The results of the present study are similar and the strain of Ireland is very close to the strain of France and is also close to the strains of Iran.
5. CONCLUSION
It can be concluded that Iranian BCoV strains are different (divergent) from strains found throughout the world, but the closest strains to Iranian strains are the European strains according to the phylogenetic tree. From the point of similarity Iranian strains are most similar to those from France, Croatia, Denmark and Sweden, which may be due to the import of Iranian cow from these countries. But in general, there is a need for more epidemiological study to determine the exact clinical epidemiology of the disease, other clinical symptoms and the pathogenesis of the disease. On the other hand, due to the many differences between the isolates of Iran and other countries, molecular epidemiology research, is needed to isolate the virus and to cultivate it, and to record the strains.
CONFLICT OF INTEREST
There is no conflict of interest to declare.
AUTHOR CONTRIBUTION
Samad Lotfollahzadeh: Conceptualization; Data curation; Funding acquisition; Project administration; Validation; Writing‐original draft; Writing‐review & editing. Omid Madadgar: Data curation; Formal analysis; Methodology; Validation; Writing‐original draft. Mohammad Reza Mohebbi: Data curation; Writing‐original draft. Mohammad Reza Mokhber Dezfoulie: Formal analysis; Software; Visualization. David George Watson: Writing‐review & editing.
ETHICAL APPROVAL AND CONSENT TO PARTICIPATE
An informed approval was sought from dairy farmers and farm managers before commencement of sampling.
CONSENT TO PUBLICATION
Not applicable
ACKNOWLEDGEMENTS
The authors acknowledge dairy farmers, resident veterinarians and managers of dairy farms for their help with sample collection.
Lotfollahzadeh S, Madadgar O, Reza Mohebbi M, Reza Mokhber Dezfouli M, George Watson D. Bovine coronavirus in neonatal calf diarrhoea in Iran. Vet Med Sci. 2020;6:686–694. 10.1002/vms3.277
Contributor Information
Samad Lotfollahzadeh, Email: samadlzadeh@ut.ac.ir.
Omid Madadgar, Email: samadlzadeh@ut.ac.ir.
REFERENCES
- Afshari Safavi, E. A. , Mohammadi, G. R. , Rad, M. , & Naghibi, A. (2012). A case‐control study of association between diarrhea in newborn calves and infection with rotavirus and coronavirus in some industrial dairy herds of Mashhad Aarea, Iran in 2008. Archives of Razi Institute, 67, 35–41. [Google Scholar]
- Ali Khan, J. , Sarwar Khan, M. , Arif Khan, M. , Avais, M. , Maqbool, A. , Salman, M. , & Rehman, Z. (2009). Epidemiology of major bacterial and viral causes of diarrheoa in buffalo calves in three districts of the punjab province of pakistan. Pakistanian Journal of Zoology, Supplementary Series, 9, 187–193. [Google Scholar]
- Bartels, C. J. , Holzhauer, M. , Jorritsma, R. , Swart, W. A. , & Lam, T. J. (2010). Prevalence, prediction and risk factors of enteropathogenes in normal and non‐normal faeces of yang Dutch dairy calves. Preventive Veterinary Medicine, 93, 162–169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bendali, F. , Bichet, H. , Schelcher, F. , & Sanaa, M. (1999). Pattern of diarrhea in newborn beef calves in southwest France. Research in Veterinary Science, 30, 61–74. [PubMed] [Google Scholar]
- Bjorkman, C. , Svensson, C. , Christensson, B. , & De Verdier, K. (2003). Cryptosporidium parvum and Giardia intestinalis in calf diarrhoea in Sweden. Acta Veterinaria Scandinavica, 44, 145–152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bok, M. , Miño, S. , Rodriguez, D. , Badaracco, A. , Nuñes, I. , Souza, S. P. , … Parreño, V. (2015). Molecular and antigenic characterization of bovine Coronavirus circulating in Argentinean cattle during 1994–2010. Veterinary Microbiology, 181, 221–229. 10.1016/j.vetmic.2015.10.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brandão, P. E. , Gregori, F. , Richtzenhain, L. J. , Rosales, C. A. R. , Villarreal, L. Y. B. , & Jerez, J. A. (2006). Molecular analysis of Brazilian strains of bovine coronavirus (BCoV) reveals a deletion within the hypervariable region of the S1 subunit of the spike glycoprotein also found in human coronavirus OC43. Archives of Virology, 151, 1735–1748. 10.1007/s00705-006-0752-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Busato, A. , Lentze, T. , Hofer, D. , Burnens, A. , Hentrich, B. , & Gaillard, C. (1998). A case control study of potential enteric pathogens for calves raised in cow–calf herds. Journal of Veterinary Medicine B, 45, 519–528. 10.1111/j.1439-0450.1998.tb00823.x [DOI] [PubMed] [Google Scholar]
- Castells, M. , Giannitti, F. , Caffarena, R. D. , Casaux, M. L. , Schild, C. , Castells, D. , … Colina, R. (2019). Bovine coronavirus in Uruguay: genetic diversity, risk factors and transboundary introductions from neighboring countries. Archives of Virology, 164, 2715–2724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Constable, P. D. , Hinchcliff, K. W. , Done, S. , & Gruenberg, W. (2017). Veterinary Medicine, A textbook of the diseases of cattle, horses, sheep, pigs and goats, 11th ed. Philadelphia: Saunders Ltd. [Google Scholar]
- Erdogan, H. M. , Unver, A. , Gunes, V. , & Citil, M. (2003). Frequency of rotavirus and coronavirus in neonatal calves in Kars district. Kafkas Üniversitesi, Veteriner Fakültesi Derg, 9, 65–68. [Google Scholar]
- Fazakerley, J. K. , Parker, S. E. , Bloom, F. , & Buchmeier, M. J. (1992). The V5A13.1 envelope glycoprotein deletion mutant of mouse hepatitis virus type 4 is neuroattenuated by its reduced rate of spread in the central nervous system. Virology, 187, 178–188. 10.1016/0042-6822(92)90306-A [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukuda, M. , Kuga, K. , Miyazaki, A. , Suzuki, T. , Tasei, K. , Aita, T. , … Tsunemitsu, H. (2012). Development and application of one‐step multiplex reverse transcription PCR for simultaneous detection of five diarrheal viruses in adult cattle. Archive in Virology, 157, 1063–1069. 10.1007/s00705-012-1271-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Godet, M. , Grosclaude, J. , Delmas, B. , & Laude, H. (1994). Major receptor‐binding and neutralization determinants are located within the same domain of the transmissible gastroenteritis virus (coronavirus) spike protein. Journal of Virology, 68, 8008–8016. 10.1128/JVI.68.12.8008-8016.1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham, A. N. , Renaud, D. L. , Duffield, T. F. , & Kelton, D. F. (2018). Short communication: Calf cleanliness does not predict diarrhea upon arrival at a veal calf facility. Journal of Dairy Science, 101, 3363–3366. 10.3168/jds.2017-14113 [DOI] [PubMed] [Google Scholar]
- Gunn, L. , Collins, P. J. , O’Connell, M. J. , & O’Shea, H. (2015). Phylogenetic investigation of enteric bovine coronavirus in Ireland reveals partitioning between European and global strains. Irish Veterinary Journal, 68, 31 10.1186/s13620-015-0060-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansa, A. , Rai, R. B. , Yaqoob Wani, M. , & Dhama, K. (2012). ELISA and RT‐PPCR based detection of bovine coronavirus in north India. Asian Journal of Animal and Veterinary Advances, 7, 1120–1129. [Google Scholar]
- Hasoksuz, M. , Kayar, A. , Dodurka, T. , & Ilgaz, A. (2005). Detection of respiratory and enteric shedding of bovine coronaviruses in cattle in Northwestern Turkey. Acta Veterenaria Hungarica, 53, 137–146. 10.1556/AVet.53.2005.1.13 [DOI] [PubMed] [Google Scholar]
- Hasoksuz, M. , Sreevatsan, S. , Cho, K. O. , Hoet, A. E. , & Saif, L. J. (2002). Molecular analysis of the S1 subunit of the spike glycoprotein of respiratory and enteric bovine coronavirus isolates. Virus Research, 84, 101–109. 10.1016/S0168-1702(02)00004-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hingley, S. T. , Gombold, J. L. , Lavi, E. , & Weiss, S. R. (1994). MHV–A59 fusion mutants are attenuated and display altered hepatotropism. Virolgy, 200, 1–10. 10.1006/viro.1994.1156 [DOI] [PubMed] [Google Scholar]
- International committee on taxonomy of viruses . (2012). Virus taxonomy: classification and nomenclature of viruses: ninth report of the international committee on taxonomy of viruses. London: academic press. (Immunology and microbiology 2011. ISBN 9780123846846 (alk. paper).
- Izzo, M. M. , Kirkland, P. D. , Mohler, V. L. , Perkins, N. R. , Gunn, A. A. , & House, J. K. (2011). Prevalence of major enteric pathogens in Australian dairy calves with diarrhea. Australian Veterinary Journal, 89, 167–173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khalili, M. , Morshedi, A. , Keyvanfar, H. , & Hemmatzadeh, F. (2006). Detection of bovine coronavirus by RT‐PCR in a field study. Vetrinarski Arhiv, 76, 291–329. [Google Scholar]
- Kin, N. , Miszczak, F. , Diancourt, L. , Caro, V. , Moutou, F. , Vabret, A. , & Ar Gouilha, M. (2016). Comparative molecular epidemiology of two closely related coronaviruses, bovine coronavirus (BCoV) and human coronavirus OC43 (HCoV‐OC43), reveals a different evolutionary pattern. Infection, Genetics and Evolution, 40, 186–191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirisawa, R. , Takeyama, A. , Koiwa, M. , & Iwai, H. (2007). Detection of bovine torovirus in fecal specimens of calves with diarrhea in Japan. Journal of Veterinary Medical Science, 69, 471–476. 10.1292/jvms.69.471 [DOI] [PubMed] [Google Scholar]
- Kubo, H. , Yamada, Y. K. , & Taguchi, F. (1994). Localization of neutralizing epitopes and the receptor‐binding site within the amino‐terminal 330 amino acids of the murine coronavirus spike protein. Journal of Virology, 68, 5403–5410. 10.1128/JVI.68.9.5403-5410.1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar, S. , Nei, M. , Dudley, J. , & Tamura, K. (2008). A biologist‐centric software for evolutionary analysis of DNA and protein sequences. Briefing in Bioinformatics, 9, 299–306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu, L. H. , Hägglund, S. , Hakhverdyan, M. , Alenius, S. , Larsen, L. E. , & Belák, S. (2006). Molecular epidemiology of bovine coronavirus on the basis of comparative analyses of the S gene. Journal of Clinical Microbiology, 44, 957–960. 10.1128/JCM.44.3.957-960.2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lojkić, I. , Kresic, N. , Kresic, N. , Šimić, I. , Šimić, I. , Bedeković, T. , & Bedeković, T. (2015). Detection and molecular characterisation of bovine corona and toroviruses from Croatian cattle. BMC Veterinary Research, 11, 202–207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo, Z. , & Weiss, S. R. (1998). Roles in cell‐to‐cell fusion of two conserved hydrophobic regions in the murine coronavirus spike protein. Virology, 244, 483–494. 10.1006/viro.1998.9121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacLachlan, N. J. , & Dubovi, E. J. (2017). Fenner’s Veterinary Virology, 5th ed. Boston: Elsevier. [Google Scholar]
- Masaharu, F. , Kazufumi, K. , Ayako, M. , Tohru, S. , Keito, T. , Tsunehiko, A. , … Hiroshi, T. (2012). Development and application of one‐step multiplex reverse transcription PCR for simultaneous detection of five diarrheal viruses in adult cattle. Archive of Virology, 157, 1063–1069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nathalie, K. , Fabien, M. , Laure, D. , Valérie, C. , François, M. , Astrid, V. , & Meriadeg Ar, G. (2016). Comparative molecular epidemiology of two closely related coronaviruses, bovine coronavirus (BCoV) and human coronavirus OC43 (HCoV‐OC43), reveals a different evolutionary pattern. Infection, Genetics and Evolution, 40, 186–191. 10.1016/j.meegid.2016.03.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oma, V. S. , Tråvén, M. , Alenius, S. , Myrmel, M. , & Stokstad, M. (2016). Bovine coronavirus in naturally and experimentally exposed calves; viral shedding and the potential for transmission. Virology Journal, 13, 100 10.1186/s12985-016-0555-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park, S. J. , Lim, G. K. , Park, S. I. , Kim, H. H. , Koh, H. B. , & Cho, K. O. (2007). Detection and molecular characterization of calf diarrhoea bovine corona viruses circulating in south Korea during 2004–2005. Zoonoses and Public Health, 54, 223–230. 10.1111/j.1863-2378.2007.01045.x [DOI] [PubMed] [Google Scholar]
- Perez, E. , Kummeling, A. , Janssen, M. M. , Jimenez, C. , Alvarado, R. , & Cabal, M. (1998). Infectious agents associated with diarrhea of calves in the canton of Tilarán, Costa Rica. Preventive Veterinary Medicine, 33, 195–205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perlman, S. , & Netland, J. (2009). Review Coronaviruses post‐SARS: Update on replication and pathogenesis. Nature Reviews Microbiology, 7, 439–450. 10.1038/nrmicro2147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reynolds, D. J. , Morgan, J. H. , Chanter, N. , Jones, P. W. , Bridger, J. C. , & Debney, T. G. (1986). Microbiology of calf diarrhea in southern Britain. The Veterinary Record, 119, 34–39. [DOI] [PubMed] [Google Scholar]
- Saif, L. J. , Redman, D. R. , Moorhead, P. D. , & Theil, K. W. (1986). Experimentally induced coronavirus infections in calves: Viral replication in the respiratory and intestinal tracts. Journal of Veterinary Research, 47, 1426–1432. [PubMed] [Google Scholar]
- Schultze, B. , Gross, H. J. , Brossmer, R. , & Herrler, G. (1991). b. The S protein of bovine coronavirus is a hemagglutinin recognizing 9‐0‐acetylated sialic acid as receptor determinant. Journal of Virology, 65, 6232–6237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh, S. , Singh, R. , Singh, K. P. , Singh, V. , Malik, Y. P. S. , Kamdi, B. , … Kashyap, G. (2019). Prevalence of bovine coronavirus infection in organized dairy farms of Central and North regions, India. Biological Rhythm Research, 22, 1–7. 10.1080/09291016.2019.1629093 [DOI] [Google Scholar]
- Singh, S. , Singh, R. , Singh, K. P. , Singh, V. , Malik, Y. P. S. , Kamdi, B. , … Kashyap, G. (2020). Immunohistochemical and molecular detection of natural cases of bovine rotavirus and coronavirus infection causing enteritis in dairy calves. Microbial Pathogenesis, 138, 103814 10.1016/j.micpath.2019.103814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stipp, D. T. , Barry, A. F. , Alfieri, A. F. , Takiuchi, E. , Amude, A. M. , & Alfieri, A. A. (2009). Frequency of BCoV detection by a semi‐nested PCR assay in feces of calves from Brazilian cattle herds. Tropical Animal Health and Production, 41, 1563–1567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takiuchi, E. , Alfieri, A. F. , & Alfieri, A. A. (2008). Molecular analysis of the bovine coronavirus S1 gene by direct sequencing of diarrheic fecal specimens. Brazilian Journal of Medical and Biological Research, 41, 277–282. 10.1590/S0100-879X2008000400004 [DOI] [PubMed] [Google Scholar]
- Tamura, K. , Dudley, J. , Nei, M. , & Kumar, S. (2007). MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Molecular Biology and Evolution, 24, 1596–1599. 10.1093/molbev/msm092 [DOI] [PubMed] [Google Scholar]
- Tsunemitsu, H. , el‐Kanawati, Z. R. , Smith, D. R. , Reed, H. H. , & Saif, L. J. (1995). Isolation of coronaviruses antigenically indistinguishable from bovine coronavirus from wild ruminants with diarrhea. Journal of Clinical Microbiology, 33, 3264–3269. 10.1128/JCM.33.12.3264-3269.1995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoo, D. , & Deregt, D. (2001). A single amino acid change within antigenic domain II of the spike protein of bovine coronavirus confers resistance to virus neutralization. Clinical and Diagnostic Laboratory Immunology, 8, 297–302. 10.1128/CDLI.8.2.297-302.2001 [DOI] [PMC free article] [PubMed] [Google Scholar]


