Abstract
Novel coronavirus disease 2019 (COVID‐19) caused by severe acute respiratory syndrome virus (SARS‐CoV‐2) has become a global health care crisis. The Centers for Disease Control and Prevention (CDC) lists immunocompromised patients, including those requiring immunosuppression following renal transplantation, as high risk for severe disease from SARS‐CoV‐2. Treatment for other viral infections in renal transplant recipients often includes a reduction in immunosuppression; however, no current guidelines are available recommending the optimal approach to managing immunosuppression in the patients who are infected with SARS‐CoV‐2. It is currently advised to avoid corticosteroids in the treatment of SARS‐CoV‐2 outside of critically ill patients. Recently published cases describing inpatient care of COVID‐19 in renal transplant recipients differ widely in disease severity, time from transplantation, baseline immunosuppressive therapy, and the modifications made to immunosuppression during COVID‐19 treatment. This review summarizes and compares inpatient immunosuppressant management strategies of recently published reports in the renal transplant population infected with SARS‐CoV‐2 and discusses the limitations of corticosteroids in managing immunosuppression in this patient population.
Keywords: COVID‐19, immunosuppression, SARS‐CoV‐2, coronavirus, renal transplant, corticosteroid
Novel coronavirus disease 2019 (COVID‐19), caused by severe acute respiratory syndrome virus (SARS‐CoV‐2), was first identified in Wuhan, China, in December 2019. 1 Since that time the disease has spread globally at an alarming rate, with the World Health Organization in March 2020 declaring the outbreak a pandemic and a major threat to international public health. In the United States, the first case of COVID‐19 was identified in January 2020; the spread of the virus since that time has been exponential with the number of cases in the United States surpassing that of all other countries. 2 Person‐to‐person spread of SARS‐CoV‐2 is highly efficient via close contact and respiratory droplets, with an incubation period that can extend from 2 to 14 days. Respiratory symptoms, which can range from mild to critically ill requiring mechanical ventilation, are the most common clinical feature of COVID‐19; however, patients who are immunocompromised may present atypically. 3 , 4
The Centers for Disease Control and Prevention (CDC) lists immunocompromised patients, including those requiring immunosuppressive therapy following organ transplantation, as high risk for severe disease from SARS‐CoV‐2. 5 Unlike some other RNA viruses, such as influenza or respiratory syncytial virus, no prophylactic agents, treatments, or vaccinations are approved for SARS‐CoV‐2. 6 At this time, supportive care is paramount to combating this virus in solid organ transplant recipients. Very little data are currently available regarding the optimal medical management of renal transplant patients testing positive for SARS‐CoV‐2 including strategies for reducing or modifying immunosuppression. 4 , 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 Corticosteroids are a cornerstone of many immunosuppressive regimens; however, their use in SARS‐CoV‐2 is controversial. 18 , 19 , 20 , 21 , 22 The purpose of this review is to summarize and compare inpatient immunosuppressant management strategies of recently published reports in the renal transplant population infected with SARS‐CoV‐2 and to discuss the limitations of corticosteroids in managing immunosuppression.
1. Methods
A literature review was performed using PubMed and Science Direct to identify relevant English‐language articles published through April 15, 2020. Search terms included coronavirus, severe acute respiratory syndrome coronavirus 2, SARS‐CoV‐2, SARS‐CoV, COVID‐19, COVID, renal transplantation, and kidney transplantation. The search resulted in 12 total articles reporting on patients who received inpatient treatment for SARS‐CoV‐2. Due to the lack of randomized controlled trials, we included case reports and case series. We independently reviewed the titles and abstracts for inclusion.
2. Review of Published Literature in Renal Transplant Recipients
Although no controlled trials currently exist, 40 published cases have demonstrated strategies for inpatient management of SARS‐CoV‐2 in renal transplant recipients (Table 1). Most patients were male, deceased‐donor recipients, with an average age of 55 years and receiving maintenance immunosuppression that included tacrolimus with mycophenolate and prednisone. Recipients described were between 1 month and 22 years post‐transplant with most patients presenting with severe respiratory symptoms requiring oxygen. Immunosuppressant management in 30 patients consisted of complete cessation of calcineurin inhibitor and antiproliferative therapy with reliance on corticosteroid monotherapy, typically with intravenous methylprednisolone. 4 , 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 Only three patients were managed without making any change in their baseline immunosuppressive regimen, and one of these patients was receiving a steroid‐sparing regimen at baseline. Of these three patients, none progressed to mechanical ventilation, and all had a shorter duration of symptoms than average, lasting ~2 weeks or less. 7 , 10 Only one other case reported a steroid‐sparing regimen at baseline; this patient’s immunosuppression was managed with cessation of antiproliferative therapy and dose reduction in tacrolimus; however, methylprednisolone 40 mg/day was also added for the duration of hospitalization. The patient fully recovered after 61 days of reported symptoms. 13
Table 1.
Published Cases on COVID‐19 in Hospitalized Renal Transplant Recipients
| Age, yrs | Sex | Time from RTx, yrs | Type of RTx | Baseline IS | Change to IS | COVID severity | COVID treatment | Antibacterial treatment | Time from symptom onset to hosp., days | Time from sympton onset to recovery, days | Clinical outcome | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 16 | 70 | F | 17 | Unknown | CNI/mTORi | Cessation of all, MP 16 mg/day | Severe | HCQ, lopinavir/ritonavir | Yes, not specified | Unknown | Unknown | Recovery |
| 47 | F | 9 | Unknown | MMF, CNI, pred | Cessation of all, MP 16 mg/day | Critical | HCQ, lopinavir/ritonavir, tocilizumab | Yes, not specified | Unknown | Unknown | Inpatient at time of publication | |
| 71 | M | 13 | Unknown | MMF, CNI, pred | Cessation of all, MP 16 mg/day | Severe | HCQ, lopinavir/ritonavir | Yes, not specified | Unknown | Unknown | Expired | |
| 57 | M | 2 | Unknown | MMF, CNI, pred | Cessation of all, MP 16 mg/day | Critical | HCQ, lopinavir/ritonavir, tocilizumab | Yes, not specified | Unknown | Unknown | Expired | |
| 51 | M | 23 | Unknown | MMF, CNI | Cessation of all, MP 16 mg/day | Severe | HCQ, lopinavir/ritonavir, tocilizumab | Yes, not specified | Unknown | Unknown | Recovery | |
| 46 | M | 2 | Unknown | MMF, CNI | Cessation of all, MP 16 mg/day | Severe | HCQ, lopinavir/ritonavir | Yes, not specified | Unknown | Unknown | Recovery | |
| 59 | M | 5 | Unknown | MMF, CNI, pred | Cessation of all, MP 16 mg/day | Critical | HCQ, lopinavir/ritonavir | Yes, not specified | Unknown | Unknown | Expired | |
| 70 | F | 6 | Unknown | CNI, pred | Cessation of all, MP 16 mg/day | Critical | HCQ, lopinavir/ritonavir | Yes, not specified | Unknown | Unknown | Expired | |
| 60 | M | 8 | Unknown | MMF, CNI, pred | Cessation of all, MP 16 mg/day | Mild | HCQ, lopinavir/ritonavir | Yes, not specified | Unknown | Unknown | Inpatient at time of publication | |
| 73 | M | 6 | Unknown | MMF, CNI, pred | Cessation of all, MP 16 mg/day | Severe | HCQ, lopinavir/ritonavir | Yes, not specified | Unknown | Unknown | Inpatient at time of publication | |
| 59 | M | 10 | Unknown | MMF, pred | Cessation of all, MP 16 mg/day | Severe | HCQ, lopinavir/ritonavir, tocilizumab | Yes, not specified | Unknown | Unknown | Inpatient at time of publication | |
| 63 | M | 15 | Unknown | MMF, CNI | Cessation of all, MP 16 mg/day | Severe | HCQ, lopinavir/ritonavir, tocilizumab | Yes, not specified | Unknown | Unknown | Expired | |
| 49 | M | 2 | Unknown | MMF, CNI, pred | Cessation of all, MP 16 mg/day | Severe | HCQ, lopinavir/ritonavir, tocilizumab | Yes, not specified | Unknown | Unknown | Inpatient at time of publication | |
| 60 | F | 2 | Unknown | MMF, CNI, pred | Cessation of all, MP 16 mg/day | Severe | HCQ, lopinavir/ritonavir | Yes, not specified | Unknown | Unknown | Inpatient at time of publication | |
| 57 | M | 10 | Unknown | MMF, CNI | Cessation of all, MP 16 mg/day | Mild | HCQ, lopinavir/ritonavir | Yes, not specified | Unknown | Unknown | Inpatient at time of publication | |
| 54 | M | 17 | Unknown | CNI, pred | Cessation of all, MP 16 mg/day | Severe | HCQ, darunavir + ritonavir | Yes, not specified | Unknown | Unknown | Inpatient at time of publication | |
| 60 | M | 13 | Unknown | CNI | Cessation, MP 16 mg/day | Mild | HCQ, lopinavir/ritonavir | Yes, not specified | Unknown | Unknown | Inpatient at time of publication | |
| 50 | M | 9 | Unknown | MMF, CNI, pred | Cessation of all, MP 16 mg/day | Mild | HCQ, darunavir + ritonavir | Yes, not specified | Unknown | Unknown | Inpatient at time of publication | |
| 69 | M | 22 | Unknown | CNI, pred | Cessation of all, MP 16 mg/day | Mild | HCQ, darunavir + ritonavir | Yes, not specified | Unknown | Unknown | Inpatient at time of publication | |
| 44 | M | 14 | Unknown | CNI, mTORi | Cessation of all, MP 16 mg/day | Mild | HCQ, darunavir + ritonavir | Yes, not specified | Unknown | Unknown | Inpatient at time of publication | |
| 17 | 29 | M | 1 | LR | MMF, cyclosporine, MP | None | Mild | Lopinavir/ritonavir + IVIG | Moxifloxacin | 2 | 15 | Recovery |
| 4 | 50 | M | 4 | DD | Tac, everolimus, pred | Cessation of Tac and everolimus | Critical | Lopinavir/ritonavir + HCQ + Interferon beta | Ceftaroline and Meropenem | 6 | >18 | Remained intubated at time of publication submission |
| 12 | 52 | M | 12 | LR | Tac, MMF, pred | Cessation of Tac and MMF | Mild | Interferon alfa + IVIG | Biapenem | 7 | 21 | Recovery |
| 9 | 49 | M | 6 | DD | Tac, MMF, pred | Cessation of Tac and MMF, Pred changed to MP 20‐40 mg/day followed by taper | Moderate | Umifenovir + ribavirin + IVIG | Moxifloxacin | 15 | 22 | Recovery |
| 8 | 58 | M | 12 | Unknown | MMF, pred | Cessation of MMF and Pred; MP 80 mg/day | Severe | Lopinavir/ritonavir | No | 4 | 40 | Expired |
| 7 | 38 | M | 0.25 | DD | Tac, MMF, steroid | Cessation of MMF and reduced tac | Unknown | Oseltamivir or Arbidol | No | 15 | 17 | Recovery |
| 64 | M | 3 | DD | MMF, rapamycin, steroid | Cessation of MMF, discontinuation of steroids following MP burst for suspected rejection | Unknown | Oseltamivir or Arbidol + IVIG | Cefixime | 4 | 32 | Recovery requiring supplemental oxygen; remained hospitalized at time of publication | |
| 37 | F | .42 | DD | Tac, MMF, steroid | Cessation of MMF, Tac held and restarted at reduced dose | Unknown | Oseltamivir or Arbidol + IVIG | Cefixime | 1 | 12 | Recovery; remained hospitalized at time of publication | |
| 47 | M | 1 | DD | Tac, MMF, steroid | Cessation of all | Unknown | Oseltamivir or Arbidol | No | 4 | 19 | Recovery; remained hospitalized at time of publication | |
| 38 | M | 2 | DD | Tac, MMF, steroid | None | Unknown | Oseltamivir or Arbidol | No | 8 | 8 | Recovery | |
| 11 | 75 | M | 10 | DD | Tac, MMF, steroid | Cessation of tac and MMF | Severe | HCQ + lopinavir/ ritonavir | Yes | 3 | 8 | Expired |
| 52 | F | 0.67 | DD | Tac, MMF, steroid | Cessation of tac and MMF | Severe | HCQ + darunavir/ cobicistat | Yes | 1 | 9 | Recovery requiring supplementary oxygen at time of publication | |
| 14 | 58 | M | 2 | Unknown | Belatacept, MMF, pred | Hold of next scheduled belatacept, cessation of MMF low dose, CSA started before hospital discharge | Mild | None | Yes, not specified | 6 | 24 | Recovery |
| 15 | 67 | F | 1 | DD | Tac, MMF, pred | Cessation of MMF | Critical | none | Yes, not specified | Unknown | 12 | Died |
| 54 | F | 0.25 | DD | Tac, MMF, pred | Cessation of tac and MMF | Critical | Oseltamivir | Yes, not specified | Unknown | Unknown | Still intubated at time of publication | |
| 65 | M | 1 | DD | Tac, MMF, pred | Cessation of MMF | Moderate | Unknown | Unknown | Unknown | Unknown | Requiring supplemental oxygen; remained hospitalized at time of publication | |
| 69 | F | 0.08 | DD | Tac, MMF, pred | Cessation of MMF | Moderate | None | Doxycycline, piperacillin‐tazobactam | Unknown | Unknown | Required supplemental oxygen; remained hospitalized at time of publication | |
| 45 | M | 3 | Unknown | Tac, azathioprine, pred | Cessation of azathioprine, reduced tac, increase pred | Moderate | Unknown | Unknown | Unknown | Unknown | Requiring supplemental oxygen; remained hospitalized at time of publication | |
| 10 | 50 | M | 4 | Unknown | Tac, MMF | None | Mild | None | Ceftriaxone | 9 | 13 | Recovery |
| 13 | 48 | M | 17 | LR | Tac, MMF | Cessation of MMF, lower trough concentration of tac, MP 40 mg/day | Unknown | Oseltamivir, Arbidol, interferon alpha, IVIG | Moxifloxacin | 10 | 61 | Recovery |
| a | 57 | M | 0.67 | DD | Tac, MMF | Reduced dose MMF and lower trough concentration of tac | Moderate | HCQ | Cefepime and azithromycin | 8 | 23 | Recovery requiring supplemental oxygen at discharge |
CNI = calcineurin inhibitors; COVID severity: mild = room air; moderate = supplemental oxygen; severe = noninvasive ventilation; critical = mechanical ventilation; DD = deceased donor; HCQ = hydroxychloroquine; IS = immunosuppression; IVIG = intravenous immunoglobulin; LR = living related; MMF = mycophenolate; MP = methylprednisolone IV; mTORi = mTOR inhibitors; RTx = renal transplantation; Tac = tacrolimus.
Case discussed in this article.
Investigational agents targeting SARS‐CoV‐2 were administered to 34 of 40 patients, with 12 different strategies trialed among patients. Additionally, broad‐spectrum antibiotic therapy was administered to 34 patients. The average duration of symptoms from those who reported was 21 days, with 18% of patients progressing to respiratory failure requiring mechanical ventilation. 4 , 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 Eight patients who had presented with severe or critical oxygen needs had died by the time of case publication; all eight patients had immunosuppressant therapy converted to corticosteroid monotherapy while hospitalized. 8 , 11 , 15 , 16
3. Discussion
Outside of supportive care, the optimal management of SARS‐CoV‐2 has not yet been established. This is especially true in solid organ transplant recipients where adjustments to immunosuppressive medications must be considered while balancing the potential for acute rejection and coinfection with bacterial or opportunistic pathogens. We have summarized recently published cases describing different immunosuppressant management strategies for renal transplant recipients with SARS‐CoV‐2. Although the details presented in these cases have many differences, most patients received corticosteroid monotherapy for maintaining immunosuppression, and all but two of the remaining cases also received a corticosteroid in combination with other agents.
3.1. A Case Supporting Corticosteroid‐sparing Immunosuppression Modifications
We present the case and outcomes of a renal transplant recipient with SAR‐CoV‐2 treated in our hospital whose immunosuppressive therapy was managed with only a modest reduction in calcineurin inhibitor target trough concentration and antiproliferative dose reduction. A 57‐year‐old African American man with a history of a deceased‐donor kidney transplant (DDKT) 8 months earlier, contacted the renal transplant team with complaints of poor oral intake, reported fever of 38.2°C, abdominal bloating, and back pain for the past 3 days. He was advised to go to the emergency department. He presented the following day with complaints of low‐grade fever, chills, decreased oral intake, and ongoing abdominal discomfort. In accordance with CDC recommendations and state and local health officials, the patient was considered low risk and did not meet criteria to be a person under investigation or for COVID‐19 testing at that time. During his visit, an abdominal radiograph showed no acute findings, and chest radiograph showed findings of congestive heart failure or positive fluid balance and left perihilar and basilar airspace opacity questionably due to pulmonary edema or superimposed pneumonia. Physical examination revealed body temperature of 37.2°C, blood pressure of 101/53 mm Hg, pulse of 79 beats per minute, respiratory rate of 18 breaths per minute, and oxygen saturation of 93% on room air. His serum creatinine (Scr) had increased to 3.2 from 2.0 mg/dl 6 days earlier, and white blood cell (WBC) count was 1.5 × 103/µl with absolute neutrophil count (ANC) of 0.7 × 103/µl. The patient improved with supportive care and was discharged with recommendations to follow up with the renal transplant office.
On day 8 of illness, the renal transplant team was contacted again with reported ongoing concerns of poor oral intake, and the patient presented to the clinic the same day for further evaluation. Symptoms included general malaise, fatigue, chills, myalgias, anorexia with poor oral intake, dyspnea while lying on the left side, diarrhea, abdominal bloating, and decreased urine output. Upon arrival, the patient’s physical examination revealed a blood pressure of 84/52 mm Hg and pulse of 60 beats per minute, dry mucous membranes, erythematous injected conjunctiva, bilateral pulmonary crackles, and poor capillary refill. Significant laboratory values included a further elevated Scr, 3.4 mg/dl, and continued neutropenia, ANC 1.2 × 103/µl, and leukopenia, WBC 2.1 × 103/µl. Following the results of chest computed tomography without contrast showing patchy ground‐glass opacities throughout both lungs, the patient was directly admitted to the hospital with differential diagnosis including dehydration, tacrolimus toxicity, renal transplant rejection, cytomegalovirus (CMV), COVID‐19, influenza, and community‐acquired pneumonia.
End‐stage renal disease attributed to diabetic nephropathy and prior nephrotic syndrome led to DDKT (kidney donor profile index 66%, recipient calculated panel reactive antibodies [C‐PRA] 0%, estimated posttransplant survival score 79%, CMV donor positive/recipient negative). In the 3 months before admission, BK virus polymerase chain reaction (PCR) and CMV PCR were negative, and Luminex was negative for donor‐specific antibodies. The patient had previously completed 6 months of CMV prophylaxis with valganciclovir. Immunosuppression at the time of presentation included tacrolimus extended release (Envarsus XR) 7 mg/day boosted with ketoconazole 100 mg/day and mycophenolic acid (MPA) 540 mg twice/day.
Upon hospitalization, the patient’s immunosuppression regimen was continued with the exception of MPA that was reduced to 360 mg twice/day. Cefepime and azithromycin were initiated, and the Infectious Disease service was consulted. The COVID‐19 PCR test (performed by NxGen MDx Lab), respiratory viral panel, blood cultures, CMV PCR, interleukin (IL)‐6 level, and tacrolimus levels were obtained.
On day 9 of illness (hospitalization day 2), the patient required 3 L of oxygen via nasal cannula, was afebrile, and continued to deny cough or other respiratory symptoms. Infectious disease obtained a sputum culture, legionella urinary antigen, and invasive fungal work‐up (fungal beta‐d‐glucan, aspergillus galactomannan antigen, histoplasma antigen, fungal antibodies). His Scr remained elevated at 3.0 mg/dl, and sodium bicarbonate infusion was maintained for metabolic acidosis (Figure 1).
Figure 1.
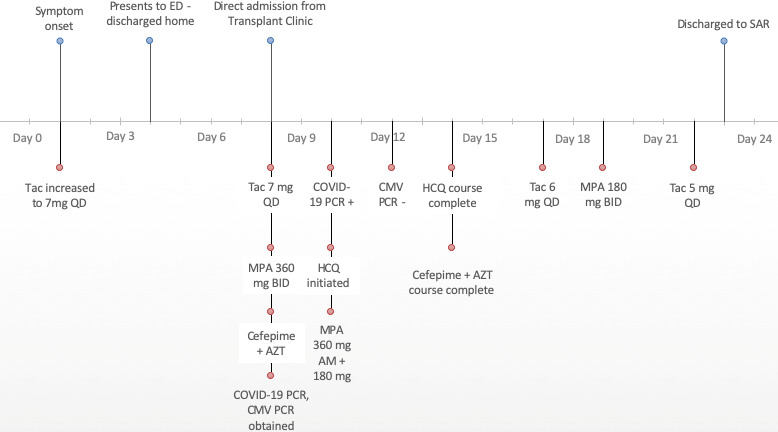
Course of SARS‐CoV‐2 illness in a renal transplant recipient. AZT = azithromycin; CMV = cytomegalovirus; HCQ = hydroxychloroquine; MPA = mycophenolic acid; PCR = polymerase chain reaction; SAR = subacute rehabilitation; Tac = tacrolimus.
On day 10 of illness (hospital day 3), COVID‐19 PCR was confirmed positive, and Infectious Disease initiated hydroxychloroquine 400 mg twice/day for 1 day followed by 200 mg/day for 4 days. Baseline QTc was obtained (436 ms) due to concern for QTc prolongation with the combination of hydroxychloroquine, tacrolimus, and azithromycin. The patient continued to be neutropenic. The MPA was further reduced to 360 mg am and 180 mg pm. Tacrolimus dose was maintained because levels were therapeutic (Table 1). The Scr and metabolic acidosis improved, allowing for transition to oral sodium bicarbonate.
Bacterial, CMV, and fungal work‐up were nonsignificant, and the patient completed a 7‐day course of cefepime and azithromycin. The IL‐6 level was 5 pg/ml, indicating the patient was unlikely to benefit from administration of an IL‐6 inhibitor. Despite the patient reporting feeling improved, he remained on 3–4 L of oxygen. On day 17 of illness (hospital day 10), pulmonology was consulted due to the ongoing oxygen needs and recommended the patient be discharged home on oxygen allowing time for recovery from the acute lung injury. On day 19 of illness, MPA was further reduced to 180 mg twice/day due to continued low lymphocyte count, and tacrolimus was reduced to 5 mg/day due to a supratherapeutic level. The patient was readied for discharge on day 23 of illness with a plan for subacute rehabilitation to continue recovery on supplemental oxygen (4 L).
3.2. Rationale for Corticosteroid‐sparing Regimens
We have described the case of a renal transplant recipient who was successfully treated for COVID‐19 with supportive care along with steroid‐sparing immunosuppression regimen changes that included dose‐reduced antiproliferative therapy and a modest decrease in tacrolimus target trough level. Different from most of the previously described cases, our patient was not receiving a corticosteroid‐containing maintenance immunosuppressive regimen before admission, which is typical for most patients who are transplanted and managed within our institution. For several reasons we chose to make only modest maintenance immunosuppressant modifications for our patient and not to rely on corticosteroid therapy to maintain immunosuppression. First, the data regarding outcomes following corticosteroid administration in COVID‐19 are mixed. The World Health Organization, CDC, Infectious Diseases Society of America, and Society of Critical Care Medicine currently recommend against the routine use of corticosteroids for managing respiratory distress in patients with SARS‐CoV‐2 unless they are indicated for another reason (e.g., asthma), the patient is experiencing refractory septic shock, or for acute respiratory distress syndrome. 18 , 19 , 20 , 21 , 22 , 23 It is also thought that corticosteroids may inhibit immune response, reduce pathogen clearance, and increase viral shedding if administered early during the clinical course of SARS‐CoV‐2 or with mild disease. 22 , 23 , 24 Finally, currently published cases of SARS‐CoV‐2 in renal transplant recipients have demonstrated variable results in progression of respiratory disease and survival when substituting higher doses of corticosteroids for complete cessation of maintenance calcineurin inhibitor and antiproliferative therapy. 4 , 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17
One group noted that of two transplant cases managed with this strategy, one renal transplant and the other a bone marrow transplant recipient, both patients developed nosocomial bacterial infections. The authors cited that the use of corticosteroids to maintain immunosuppression in these patients likely increased the risk for these infections and warrants caution with the use of corticosteroids in maintaining immunosuppression for transplant patients with SARS‐CoV‐2. 8 Because of these risks coupled with our patient’s 0% cPRA before transplant, indicating the patient was not highly sensitized to HLA antigens, we believed he could be managed safely with modest reductions in immunosuppression without substituting corticosteroids (Table 2).
Table 2.
Pertinent Laboratory Values Over Course of SARS‐CoV‐2 Illness in a Renal Transplant Recipient
| Day of illness | Scr, mg/dl | WBC, 103/µl | Lymphocytes, 103/µl | ANC, 103/µl | QTC, ms | Oxygen, L | Oxygen, sat, % | Tacro level, ng/ml |
|---|---|---|---|---|---|---|---|---|
| 4 | 3.2 | 1.5 | 0.3 | |||||
| 8 | 3.4 | 2.1 | 1.2 | 423 | 4 | 96 | ||
| 9 | 3 | 2.2 | 0.3 | 1.4 | 4 | 91–96 | 6.1 | |
| 10 | 2.4 | 2.2 | 0.3 | 1.4 | 407 | 3 | 91 | 5 |
| 11 | 2.1 | 2.2 | 0.2 | 1.4 | 436 | 2 | 92–94 | 5.5 |
| 12 | 1.8 | 0.3 | 429 | 3 | 91–93 | 5.3 | ||
| 13 | 1.7 | 2.7 | 0.2 | 2.0 | 439 | 2 | 93–95 | |
| 14 | 1.8 | 3.0 | 0.3 | 2.3 | 424 | 3 | 91–92 | |
| 15 | 1.9 | 4.6 | 0.4 | 3.6 | 438 | 4 | 85–91 | 5.3 |
| 16 | 2.0 | 4.6 | 0.4 | 3.5 | 443 | 3 | 93–99 | |
| 17 | 1.9 | 5.7 | 0.4 | 4.3 | 5 | 90–92 | 7.6 | |
| 18 | 1.9 | 7.6 | 0.4 | 5.9 | 5 | 87–96 | ||
| 19 | 1.9 | 6.9 | 0.3 | 5.3 | 4 | 90–98 | 4.4 | |
| 20 | 1.7 | 6.4 | 0.8 | 5.1 | 4 | 90–98 | ||
| 21 | 1.8 | 7.2 | 0.1 | 5.8 | 5 | 82–100 | ||
| 22 | 1.8 | 8.1 | 0.5 | 6..3 | 427 | 5 | 87–96 | 7.4 |
ANC = absolute neutrophil count; Scr = serum creatinine; WBC = white blood cells.
As with a significant portion of patients presenting with SARS‐CoV‐2, our patient’s lymphocyte count at presentation was below the normal range. 25 , 26 Our patient’s neutrophils were also below normal range at the time of admission, which is uncommon in most cases; however, this was likely the result of his baseline immunosuppressive regimen being steroid sparing. 25 Due to the potential risks of corticosteroids and because our patient presented with only moderate respiratory symptoms, we chose to focus on reducing antiproliferative therapy as our main strategy for reducing immunosuppression as well as maintaining prehospitalization tacrolimus dose and making a modest reduction in goal trough from 6–8 ng/ml to 4–6 ng/ml. Following these changes, the patient’s neutrophil count gradually increased; however, his lymphocyte count remained low. Further reduction in antiproliferative therapy was made to try to target lymphocyte improvement. Ultimately, the patient experienced a long clinical course, similar to many of the previously published cases in renal transplant recipients, and he was able to be successfully readied for discharge and rehabilitated with continued supplemental oxygen.
3.3. Limitations and Need for Future Study
Several limitations are associated with making comparisons, generalizations, or drawing conclusions from the currently reported cases of renal transplant patients treated for SARS‐CoV‐2. The currently published cases present a large variation in transplant and clinical characteristics as well as SARS‐CoV‐2 and immunosuppressant management. 4 , 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 Time from transplantation differed greatly between patients, which could indicate that immunosuppressive intensity at baseline also varied widely among the different cases. Unfortunately, most of the currently published cases do not describe medication doses or target tacrolimus trough concentrations in detail to allow for comparison. Additionally, very limited data regarding patient transplant matching characteristics and risk factors for poor prognosis have been presented, making it difficult to assess baseline immunosuppressive therapy, appropriateness of modifications, and risk for poor outcomes. Limited to no detail is given in some cases for important clinical variables such as oxygen requirements, also making the comparison of clinical courses difficult. Finally, most patients received at least one investigational agent targeting SARS‐CoV‐2 as well as different empirical antibiotic therapies targeting a wide range of pathogens, including our patient who received a 5‐day course of hydroxychloroquine plus 7 days of antibacterial therapy. Although the efficacy of these investigational agents is unknown, and there is thought to be low likelihood of bacterial coinfection, these treatments may confound generalizability of the data. 27 Furthermore, some investigational agents may have influenced the changes made to immunosuppression. For example, in some cases where lopinavir/ritonavir was administered, the calcineurin inhibitor may have been purposefully targeted for substitution due to CYP3A4 and p‐glycoprotein drug‐drug interactions. 8 , 11
4. Conclusion
It is difficult to compare and draw conclusions regarding optimal immunosuppressant management in renal transplant recipients treated for SARS‐CoV‐2 from the limited data presented in currently published cases along with significant confounding variables. Most of the cases have relied on corticosteroid monotherapy for maintaining immunosuppression while treating SARS‐CoV‐2 in renal transplant recipients; however, the routine use of corticosteroids to treat patients with SARS‐CoV‐2 is not recommended. Renal transplant recipients with moderate oxygen requirements may be able to be managed successfully with steroid‐sparing modifications to immunosuppression including modest reductions in calcineurin inhibitor trough concentrations and antiproliferative dosing. Further data are needed to determine optimal immunosuppressant management in this patient population, including if a corticosteroid‐sparing strategy is viable in patients who present with severe clinical disease, such as those requiring ventilator support, or for those who are on steroid‐containing regimens at baseline.
Conflict of interest: The authors have declared no conflicts of interest for this article.
References
- 1. Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID‐19) outbreak in China. JAMA 2020;323:1239. [DOI] [PubMed] [Google Scholar]
- 2. Centers for Disease Control and Prevention . First travel‐related case of 2019 novel coronavirus detected in the United States. https://www.cdc.gov/media/releases/2020/p0121‐novel‐coronavirus‐travel‐case.html Accessed April 6, 2020
- 3. Centers of Disease Control and Prevention . Interim infection prevention and control recommendations for patients with suspected or confirmed coronavirus disease 2019 (COVID‐19) in healthcare settings. Updated April 1, 2020. https://www.cdc.gov/coronavirus/2019‐ncov/infection‐control/controlrecommendations.html Accessed April 6, 2020
- 4. Guillen E, Pineiro GJ, Revuelta I, et al. Case report of COVID‐19 in a kidney transplant recipient: does immunosuppression alter the clinical presentation? Am J Transplant 2020; 10.1111/ajt.15874 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Centers for Disease Control and Prevention . Groups at higher risk for severe illness. https://www.cdc.gov/coronavirus/2019‐ncov/need‐extra‐precautions/groups‐at‐higher‐risk.html Accessed April 6, 2020
- 6. Manuel O, Estabrook M. RNA respiratory viral infections in solid organ transplant recipients: Guidelines from the American Society of Transplantation Infectious Diseases Community of Practice. Clin Transplant. 2019;33(9):e13511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Zhang H, Chen Y, Yuan Q, et al. Identification of kidney transplant recipients with Coronavirus Disease 2019. Eur Urol 2020. 10.1016/j.eururo.2020.03.030 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Huang J, Lin H, Wu Y, et al. COVID‐19 in post‐transplantation patients—report of two cases. Am J Transplant 2020. 10.1111/ajt.15896 [Epub ahead of print]. [DOI] [Google Scholar]
- 9. Chen S, Yin Q, Shi H, et al. A familial cluster, including a kidney transplant recipient, of Coronavirus Disease 2019 (COVID‐19) in Wuhan, China. Am J Transplant 2020. 10.1111/ajt.15903 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Seminari E, Colaneri M, Sambo M, et al. SARS CoV2 infection in a renal transplanted patient: a case report. Am J Transplant 2020. 10.1111/ajt.15902 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Gandolfini I, Delsante M, Fiaccadori E, et al. COVID‐19 in kidney transplant recipients. Am J Transplant 2020. 10.1111/ajt.15891 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Zhu L, Xu X, Ma K, et al. Successful recovery of COVID‐19 pneumonia in a renal transplant recipient with long‐term immunosuppression. Am J Transplant 2020. 10.1111/ajt.15869 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Zhong Z, Zhang Q, Xia H, et al. Clinical characteristics and immunosuppressants management of coronavirus disease 2019 in solid organ transplant recipients. Am J Transplant 2020. 10.1111/ajt.15928 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Marx D, Moulin B, Fafi‐Kremer S, et al. First case of COVID‐19 in a kidney transplant recipient treated with belatacept. Am J Transplant 2020. 10.1111/ajt.15919 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Banerjee D, Popoola J, Shah S, et al. COVID‐19 infection in kidney transplant recipients. Kidney Int 2020. 10.1016/j.kint.2020.03.018 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Alberici F, Delbarba E, Manenti C, et al. A single center observational study of the clinical characteristics and short‐term outcome of 20 kidney transplant patients admitted for SARS‐CoV2 pneumonia. Kidney Int 2020. 10.1016/j.kint.2020.04.002 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Ning L, Liu L, Li W, et al. Novel coronavirus (SARS‐CoV‐2) infection in a renal transplant recipient: case report. Am J Transplant 2020. 10.1111/ajt.15897 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. World Health Organization . Clinical management of severe acute respiratory infection (SARI) when COVID‐19 disease is suspected. Interim guidance. https://www.who.int/publications‐detail/clinical‐management‐of‐severe‐acute‐respiratory‐infection‐when‐novel‐coronavirus‐(ncov)‐infection‐is‐suspected Accessed April 11, 2020
- 19. Centers for Disease Control . Healthcare professionals: Frequently asked questions and answers. https://www.cdc.gov/coronavirus/2019‐ncov/hcp/faq.htmlAccessed April 9, 2020
- 20. Alhazzani W, Møller MH, Arabi YM, et al. Surviving sepsis campaign: Guidelines on the management of critically ill adults with coronavirus disease 2019 (COVID‐19). Crit Care Med. 2020. 10.1097/CCM.0000000000004363 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Bhimraj A, Morgan RL, Shumaker AH, et al. Infectious Diseases Society of America Guidelines on the treatment and management of patients with COVID‐19. Clin Infect Dis 2020. 10.1093/cid/ciaa478 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Wu C, Chen X, Cai Y, et al. Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Int Med 2020. 10.1001/jamainternmed.2020.0994 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Russell CD, Millar JE, Baillie JK. Clinical evidence does not support corticosteroid treatment for 2019‐CoV lung injury. Lancet 2020;395(10223):473–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Farkas J. Internet Book of Critical Care. EMCrit Project. Available from https://emcrit.org/ibcc/COVID19/. Accessed April 7, 2020.
- 25. Han R, Huang L, Jiang H, Dong J, Peng H, Zhang D. Early Clinical and CT Manifestations of coronavirus disease 2019 (COVID‐19) pneumonia. Am J Roentgenol 2020. 10.2214/AJR.20.22961 [Epub ahead of print]. [DOI] [PubMed] [Google Scholar]
- 26. Xu YH, Dong JH, An WM, et al. Clinical and computed tomographic imaging features of novel coronavirus pneumonia caused by SARS‐CoV‐2. J Infect 2020;80(4):394–400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Kim D, Quinn J, Pinsky B, et al. Rates of co‐infection between SARS‐CoV‐2 and other respiratory pathogens. JAMA 2020. 10.1001/jama.2020.6266 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]


