Abstract
There is a long history of personal protective equipment (PPE) used by the surgeon to minimize the transmission of various pathogens. In the context of the present coronavirus disease 2019 pandemic there is significant controversy as to what forms of PPE are appropriate or adequate. This review aims to describe the pathogenic mechanism and route of spread of the causative virus, severe acute respiratory syndrome coronavirus, as it pertains to accumulated published data from experienced centers globally. The various forms of PPE that are both available and appropriate are addressed. There are options in the form of eyewear, gloves, masks, respirators, and gowns. The logical and practical utilization of these should be data driven and evolve based on both experience and data. Last, situations specific to surgical populations are addressed. We aim to provide granular collective data that has thus far been published and that can be used as a reference for optimal PPE choices in the perioperative setting for surgical teams.
Keywords: coronavirus disease 2019, operation, SARS-CoV-2, surgery
The concept of personal protective equipment (PPE) for the surgeon has been in place for greater than 100 years. The aim has been to protect both the surgeon and the patient. As zoonotic respiratory pathogens emerge, surgeons, and their teams will, however, need to adapt quickly to what will adequately protect them and their patients. There has been a generalized call for more PPE; however, there is a wide array of PPE available. High-quality evidence related to the epidemiology of coronavirus disease 2019 (COVID-19) is rapidly evolving1; therefore, inferences based on existing data will need to be made to make decisions moving forward. Physicians need to be knowledgeable about transmission of the virus, as well as the capabilities and limitations of their respective PPE. This review aims to address both the mechanism of the contagion and the capabilities of PPE in the perioperative setting in the context of the current COVID-19 pandemic.
THE CORONAVIRUS SARS-COV-2 AND ROUTES OF PATHOGENESIS
Coronaviruses are spherically enveloped RNA viruses that have club-shaped spike projections emanating from the surface, giving them the appearance of a solar corona.2 Before the appearance of severe acute respiratory syndrome coronavirus (SARS-CoV) in 2003, these were primarily thought to be mild seasonal pathogens.3 The virus causing the COVID-19 pandemic is SARS-CoV-2,4 which has 79% homology with SARS-CoV, and a similar receptor-binding domain.5 The particle core diameter is 82 to 94 nm, and spikes extend another 19 nm, giving a total average size of 101 to 113 nm (0.1 μm).3 Spikes are essential for infectivity,6 which is thought to be via the angiotensin-converting enzyme-2 receptor expressed in type II alveolar cells.5,7 During initial infection, there are also high viral loads in the upper respiratory tract.8 Transmission is believed to be predominantly via inhalation of droplets. The distinction between droplet (10–100 μm) and aerosol (<10 μm) is based on diameter of the respiratory particles leading to transmission (Fig. 1).9 Droplet transmission occurs during an expiratory event (breathing, talking, coughing, sneezing) that deposits droplets onto a susceptible person's mucous membranes; because these gravitationally settle quickly, transmission is only through close contact. By comparison, aerosol transmission is through smaller respiratory droplets that can remain airborne.10 Although many respiratory pathogens can behave as “airborne” in a favorable environment, relatively few (notably, tuberculosis, measles, and varicella) are thought to have the potential to be transmitted by the long-range airborne route.9 Data related to transmission for COVID-19 is largely inferred from data related to SARS-CoV.11 Some reports also show aerosol transmission of SARS-CoV.12,13 Air samples from COVID-19 patients’ rooms have, however, been very low or negative for virus therefore supporting the idea of droplet-only transmission.14–16 During normal breathing, the falling distance of droplets is <2 cm, but coughing and sneezing produce larger and higher velocity droplets.17,18 A 30 μm droplet produced during a cough travels up to 2.5 m from the subject.18 Certain patient procedures are also considered to aerosolize respiratory secretions, including endotracheal intubation, cardiopulmonary resuscitation, bronchoscopy, open suction of the respiratory tract, sputum induction, use of nebulizer therapy, noninvasive positive pressure ventilation, and high frequency oscillatory ventilation.16,19 This is particularly relevant to surgical populations as nearly all of these procedures can be a part of standard perioperative care.
FIGURE 1.
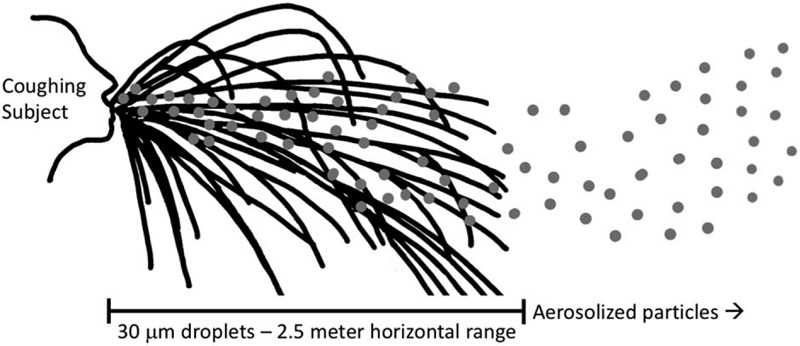
Schematic of droplet versus airborne transmission of respiratory particles produced from a cough. A coughing subject produces respiratory particles of various sizes; larger droplets tend to follow a ballistic trajectory (black lines), whereas aerosolized particles (gray dots) become suspended in air to varying degrees, extending their range.17
As data are forthcoming on the asymptomatic spread of this virus, each infected person on average can spread to another 2.2 people, and an epidemic will increase as long as this value is >1 person.20 Therefore the goal of PPE is to protect the individual and limit this spread in the asymptomatic healthcare worker to their colleagues and respective patients who are disease free.
THE RISK TO HEALTHCARE WORKERS
A recent report by the Chinese Center for Disease Control and Prevention showed that only 3.8% of all cases were healthcare personnel. Although 15% were classified as severe or critical, there was a 0.2% mortality rate.21 Earlier in the pandemic, infection of healthcare workers was as high as 29%,21 and this dramatically decreased thereafter. This decrease in infection rate is likely a reflection of the PPE measures put in place to appropriately protect healthcare workers.
TYPES OF PERSONAL PROTECTIVE EQUIPMENT AND SUPPORTING DATA
There are several different types of PPE, including eye protection, gowns, gloves, surgical face masks, nonpowered filtering facepiece respirators (FFRs), and powered air-purifying respirators. These are discussed in detail below. A summary of the types of PPE that healthcare workers are using with supporting data for their use is presented in Table 1.
TABLE 1.
Types of Personal Protective Equipment for Healthcare Workers

Eye Protection
On January 23, 2020, Dr. Wang Guangfa of Peking University First Hospital reported that he had COVID-19 and believed it was transmitted through his conjunctiva because he wore an N95 mask.37 There is evidence that ACE2 receptors are present in the aqueous humor of the eye,38 but conjunctivitis has not been described in patients with COVID-19,39,40 and no virus was identified on the conjunctival swabs of 114 patients with confirmed COVID-19 pneumonia.24 Laboratory studies also show that influenza can enter the nasopharynx with only ocular exposure,22 but rates of transmission of influenza are unchanged by wearing goggles.22 Another study, however, showed that uninfected SARS-CoV healthcare workers were more likely to use goggles.23 Thus, at this time, there is limited data that transmission of COVID-19 can occur through the mucous membranes of the eyes, or that eye protection beyond what is already routine is helpful in preventing transmission. Nevertheless, eye protection is recommended when caring for a patient with known or suspected COVID-19 infection by the United States Centers for Disease Control and Prevention (CDC),19 in Korea in a number of different patient care situations,41 and in Hong Kong for high-risk patient interactions.16
Gowns and Gloves
Because COVID-19 is thought to spread via droplets, less data regarding the transmission of COVID-19 are focused on gowns and gloves, which are the PPE components of contact precautions. Handwashing has been unequivocally recognized as an essential measure to slow contact transmission; its frequent practice is less reliant on new or additional healthcare resources and therefore will not be discussed in further detail here. Gowns are rated by American National Standards Institute/Association for the Advancement of Medical Instrumentation PB70:2012 standards level 1 to 4, with level 4 having the greatest protection against viruses.42 During the SARS epidemic, 2 studies found that uninfected healthcare workers in Hong Kong more often wore gowns as a part of their PPE when compared to infected healthcare workers11,23; one of these studies also reported a significant difference for wearing gloves.22 Both reported uninfected healthcare workers were more likely to wear all recommended PPE.11,23 It should be noted that in a laboratory environment, aerosolized SARS-CoV-2 has a median half-life of 5.6 hours on stainless steel, and 6.8 hours on plastic,25 and has also been detected on surfaces in patient rooms.16 Therefore, the use of gown and gloves may help prevent transmission from fomites to the healthcare worker's mucous membranes. The CDC recommends both gowns and gloves when caring for a patient with known or suspected COVID-19.19 Full body protective suits have been used in other viral epidemics that are bloodborne and spread primarily by contact (eg, Ebola virus43) but the authors found no data regarding their use for SARS-CoV or COVID-19.
Surgical Face Masks
In the United States, surgical masks are rated by ASTM (ASTM International) F2100-19 as level 1, 2, or 3 based on their permeability to synthetic blood, bacteria (Staphylococcus aureus), particles (latex spheres), and flammability. Level 1 masks must have >95% filtration efficiency to bacteria and air particles 0.1 to 5.0 μm. For levels 2 and 3, this increases to >98%. Level 3 surgical masks do not have additional filtration efficiency but do have greater resistance to synthetic blood. Very few reports or guidelines distinguish between the different types of surgical masks though. Furthermore, these ratings apply to the materials used in manufacturing. Because surgical masks fit loosely, they cannot reliably prevent inhalation of all airborne particles,26 but they do help to a degree. One study found that a level 1 mask reduced passage of 0.02 to 1 μm particles 2.7-fold.44
Surgical masks appear to be helpful in decreasing the transmission of influenza, SARS-CoV, and COVID-19. Two randomized controlled trials have been performed to compare surgical masks (level 3)26 or unspecified27 to N95 masks regarding transmission of influenza to healthcare workers. The results showed no difference in the influenza infection rates between the surgical mask and N95 groups.26,27 Another study by Yen et al28 compared hospitals with and without SARS-CoV healthcare worker infections. These authors found that zero healthcare worker infections were more likely in hospitals where surgical masks were worn upon entering the hospital, on the wards, and in outpatient clinics.28 This is the same plan of action currently being used in Hong Kong, Singapore, and Taiwan.16,45,46 A recent study from Singapore of 41 healthcare workers in close contact (10+ min, <2 m away) with a critically ill COVID-19 patient showed zero transmissions.47 Of these healthcare workers, 35 of 41 (85%) were using a surgical mask for respiratory protection.47 Thus, since surgical face masks filter particles larger than 0.1 to 5.0 μm and the droplets that carry Sars-CoV-2 are larger, it stands to reason that there should be adequate protection from transmission of COVID-19 in low risk circumstances. These findings have led many institutions to promote use of surgical face masks in lower risk patient interactions, and advocate for the futility of N95 respirator masks especially during times of scarcity. It should be noted, however, that the CDC recommends use of a surgical face mask if a respirator is not available when caring for a patient with known or suspected COVID-19.19
Filtering Facepiece Respirators—Nonpowered
FFRs are PPE designed to protect against respirable particulate matter (irrespective of whether they are inert or biologic particles). The most commonly used FFR do not require electricity (nonpowered), and function simply as a mask. They are classified by the percentage of challenge particles >0.3 μm filtered out during testing (95%, 99%, 99.9%) and by the type of particles filtered out. “R” rated FFR are somewhat resistant to oil, “P” rated FFR are more strongly resistant to oil, whereas “N” rated FFR are not resistant to oil. FFRs have an assigned protection factor (APF) of 10 meaning they can reduce aerosol concentration to one tenth of the ambient air (ie, blocking 90% of airborne particles). They must meet certification tests established by National Institute of Occupational Safety and Health; however, the FDA does not perform independent testing of any FFR other than surgical N95 masks.48 Surgical N95 masks are constructed to prevent contamination of the sterile field while providing the same protection of conventional N95s. The FDA publishes certified vendors of surgical N95s as a resource for individuals and for purchasing institutions.49 The different types of FFRs are presented in Table 2.
TABLE 2.
Comparison of Nonpowered Filtering Facepiece Respirators That Cover the Mouth and Nose

FFRs have been tested against ultrafine viral particles (0.02–0.5 μm).50 Results varied slightly by the type of viral particle, but with airflow rates for nonstrenuous breathing, N95 penetration was 0.23% to 1.68%, and N99 penetration was 0.96% to 1.03%. Of note, viral penetration did increase with moderate airflow (simulating strenuous breathing), up to approximately 5% for N99 and N95 FFRs.49 Another study compared viral penetration of N95 masks to surgical masks, and showed that while N95 penetration was 5%, level 2 surgical mask penetration was 21%, and level 1 surgical mask penetration was 85% for moderate airflow.30 The extrapolation of these data to infection by droplet transmission may be flawed, however, due to the larger size of the droplets (10–100 μm) that carry SARS-CoV-2 viral particles in a nonexperimental setting. An example of this is the similar transmission rates of influenza to healthcare workers using N95 or surgical masks described above.26,27
There are several studies that examine N95 use as it relates to transmission of SARS-CoV and SARS-CoV-2. As mentioned above, Yen et al28 compared hospitals with and without infected healthcare workers during the SARS-CoV epidemic in Taiwan and found that 100% of hospitals with zero SARS-CoV healthcare worker infections used N95 masks in “zones of risk.”28 A case-matched study of healthcare workers infected with SARS-CoV compared their PPE use to healthcare workers who were not infected.23 Nearly 100% of the participants used either a surgical mask or an N95 mask, and there was no difference in the type of mask used when comparing infected and noninfected healthcare workers.23 A study specifically examining critical care nurses also found no differences in the rates of becoming infected when using a surgical mask compared to an N95 mask.29 Wang et al31 published healthcare worker infection rates from Wuhan University from December 2019 and showed that none of the 278 staff members who wore N95 masks became infected. Doctors and nurses on surgical floors who did not interact with COVID-19 patients were considered low risk and did not wear masks, and 10 of 215 (4.6%) of these individuals ultimately became infected.31 Currently, the CDC recommends use of an N95 mask when caring for a patient with known or suspected COVID-19 if available and recommends an N95 or higher respirator when performing or present for aerosol-generating procedures.19 In Korea, N95 masks at minimum have been recommended for all hospital interactions except disposal of medical waste.23 In Hong Kong, they are recommended for staff working in triage, with confirmed COVID-19 cases, and when performing aerosol generating procedures.16
Because of the COVID-19 pandemic, resource scarcity has raised the prospect of reuse of FFRs. Extended single use of FFR is favored over limited reuse because of the risk of self-inoculation.31 For extended single use, this is most practical when patients are cohorted (patients with a common diagnosis in the same unit).51 It has been shown that donning an FFR up to 20 times maintains good fit; however, quality of fit decreases with subsequent uses and best fit was seen after the first 5 donnings.52 A Veterans Administration study also revealed the median tolerance time for FFR masks to be 6 to 7 hours due to heat and facial discomfort, suggesting that healthcare workers cannot be expected to work prolonged shifts (≥12 hours) in an environment which requires continuous FFR.32 It should also be noted that bleach and microwave decontamination of FFRs may damage or render masks unsafe.53 Conversely, ultraviolet irradiation and ethylene oxide may be more suitable for decontamination.53 Researchers at Duke University recently published a verifiable decontamination protocol for N95 respirators using hydrogen peroxide vapor.54 Unpublished data from Stanford University suggests that heating N95 masks to 70°C for 30 minutes is sufficient to destroy coronavirus particles and does not diminish filtration efficiency.55
Alternate types of nonpowered FFR include elastomeric facepiece respirators (EFR, either half- or full-face versions) commonly known as “gas masks.” EFRs employ a reusable facepiece which covers the mouth and nose (half-face) or mouth, nose, and eyes (full-face) with disposable filter cartridges which can filter particulates as well as vapors and gasses (when using appropriate filters). Advantages of EFR include reusability and expanded applicability; however, they require adequate cleaning and are known to significantly limit verbal communication between healthcare providers.33
Filtering Facepiece Respirators—Powered Air-purifying Respirators
Powered air-purifying respirators (PAPR) and atmosphere supplying respirators provide additional protection through fan powered and filtered airflow coupled with complete enclosure of the head. One specific advantage of PAPRs is they may be used by individuals with facial hair or those who fail standard fit tests for FFR. They also provide low-resistance breathing and often do not touch the face which may be more comfortable. They have components which can be reused, and an APF of 25.34 More advanced forms of PPE used in industry and firefighting include supplied air respirators (APF = 1000) and self-contained breathing apparatus (APF = 10,000).34 The authors could not find specific data regarding these types of respirators as they pertain to the protection or transmission of SARS-CoV or SARS-CoV-2 in healthcare workers.
COVID-19 AND SITUATIONS SPECIFIC TO SURGERY
As of March 30, 2020 the authors were unable to find published cases of transmission of SARS-CoV or SARS-CoV-2 from patients to surgeons or operating room staff, but news sources that have reported this during transsphenoidal surgery.56 All protocols for PPE use in the operating room must therefore be inferred. Operating rooms and airborne infectious isolation rooms must have a minimum of 20 air changes per hour and do not recirculate to other rooms, which translates into 99.9% efficiency for the removal of air in 21 minutes.57 By comparison, nonisolation patient rooms have a minimum of 4 air changes per hour and may recirculate air.57 It is known that aerosol transmission of infectious diseases can be influenced by ventilation,9,58 which suggests that transmission of SARS-CoV-2 may be lower in an operating room. This must be weighed, however, against the fact that aerosolizing procedures are much more likely to take place in an operating room.
Intubation is thought to be a particularly high-risk aerosolizing procedure. Viral loads of SARS-CoV-2 are known to be high in the upper respiratory tract of infected patients,59 and a study of critical care nurses from Toronto during the SARS-CoV epidemic found that these nurses were more likely to become infected when present during intubations.29 For this reason, specific recommendations regarding PPE for intubation of patients with COVID-19 have been made.60 SARS-CoV-2 is also present in bodily tissues; although nearly all patients (93%) have evidence of virus in their sputum, some (29%) also have virus in the stool.61 Only 1% have evidence of virus in the blood.61 Taken together, this suggests that pulmonary and upper respiratory surgeries are likely to be higher risk, but that colorectal and gastrointestinal surgeries may also have greater risk of transmission. Questions have been raised regarding risk during laparoscopic surgery, and if cauterization can aerosolize SARS-CoV-2.62 There is evidence that hepatitis B virus and human papillomavirus may become aerosolized during electrocautery,63,64 although they both are DNA viruses that have different mechanisms of transmission and sites of infection compared to SARS-CoV-2, challenging this inference. Nevertheless, given the paucity of data, and since it is known that virus RNA may be within the stool, every attempt should be made to minimize the evacuation of cautery smoke into the operating room per current guidelines from the Society of American Gastrointestinal and Endoscopic Surgeons.65 This can be done using devices that filter released carbon dioxide.65
A study of 3 patients with SARS-CoV who underwent caesarean sections reported that no healthcare workers in the operating room were infected when using the following PPE: powered air-purifying respirator with a hood covering the face and shoulders, goggles, gown, gloves, and cap.66 Of note, 2 out of 3 of these patients developed wound infections, but their being treated with high-dose steroids may have been a confounder.66 Before the COVID-19 pandemic, use of PAPRs in combination with body suits was already popular in orthopedic surgery. A randomized controlled trial that accrued from 1974 to 1979 demonstrated that orthopedic surgeons who used PAPRs had significantly decreased aerosolized bacterial counts in the operating room and decreased incidence of deep joint sepsis35; the application of this study to modern operating rooms, however, has come into question.36 A study of simulated joint surgery using fluorescein demonstrated that spread of respirable particulate matter to the proceduralist was eliminated with a powered air-purifying respirator and a level 4 gown.67 A similar study also showed that individuals wearing surgical masks without a PAPR had evidence of particulate matter within their nostrils, eyebrows, and eyelashes.68 Although certainly not definitive, these studies suggest that PAPR may be effective in reducing aerosolized transmission of COVID-19.
Protocols for running an operating room during the COVID-19 pandemic have emerged in the last month.60,69,70 Wong et al70 recommended reviewing the patient, inducing anesthesia, operating, and recovering all within the operating room itself. In contrast, Ti et al69 recommended anesthesia inductions in a separate negative pressure room. Both authors recommend OR staff wear at minimum N95 masks, goggles, or face shield, gown, and boot covers, with a powered air-purifying respirator for intubation and aerosol-generating procedures.69,70 Ti et al69 recommended a runner be stationed outside the operating room to get supplies and deliver them to a trolley which is pushed into an anteroom, that is then accessed by a member of the operating room team. This may limit the amount of PPE used with every entry and exit from the operating room.60 It has also been recommended that postoperatively all team members shower.69 A COVID-19 operating room coordinator and a team huddle before surgery may be used to facilitate smooth transitions of care and appropriate use of PPE.60,70 We have compiled a list of recommendations from our institution regarding operating room PPE use during the COVID-19 pandemic (Table 3).
TABLE 3.
Our Hospital's Recommendations Regarding Operating Room Personal Protective Equipment During the COVID-19 Pandemic

There is a paucity of data on the optimal postoperative management of patients with suspected or confirmed COVID-19 infection. We were unable to find reliable guidance on the risks of deep breathing, incentive spirometry, ambulation outside the room, or other maneuvers as part of pulmonary hygiene. A single report, in press, describes 3 patients who underwent elective operations (incisional hernia repair, cholecystectomy, hysterectomy), and were found to have COVID-19 in the postoperative period; 2 out of these 3 patients died.71 Although this small series cannot truly tell what a patient's risk is if they contract COVID-19 in the perioperative period, it suggests that strong consideration should be taken for nonemergency operations at this time.
If elective or semielective surgery is not being performed on patients with COVID-19, this is an additional modality to protect surgeons and operating room teams. To this end, many institutions, including ours, are utilizing multidisciplinary teams to determine which procedures can be delayed. Protocols are evolving to allow for preoperative testing of patients in the semielective surgical setting to ascertain the safest scenarios both for the healthcare team and for the patient. As resources are made available (specifically testing mechanisms) this approach will certainly be adopted by mirroring the approach of countries such as China, who were first to experience this pandemic. Ultimately, the most effective mitigation of risk for the entirety of the population will be the generation of viable effective vaccines that will provide protection for the at-risk population.
CONCLUSIONS
Use of PPE is but one of myriad factors in the effort to mitigate the transmission of hospital acquired infections. Other factors in transmission, some mentioned above, include aerosol and droplet dynamics, infectious particle settling rate, ventilation and air distribution patterns, humidity and temperature, number of infected, number of susceptible, length of exposure, type and degree of invasive procedures, UV light and chemical exposure, and persistence of pathogens within a host.58 Use of PPE and to what extent PPE are used, however, can often be individually decided upon by physicians. The premise of this review was not so much to make recommendations, because many hospitals, organizations, and societies have already done so. It was rather to explain the data that exist, and we presume, how these recommendations came to be made. Furthermore, this body of work can serve as a reference for decision making regarding how and when PPE are used when healthcare providers feel they have a choice, or when they are requesting more PPE. Additional data will be needed to better allocate these now very valuable resources.
Footnotes
Y.F. has the following disclosures, all unrelated to the writing of this manuscript: Scientific consultant for Intuitive, Medtronic, Johnson & Johnson, and Olympus. C.L.S., L.W.T., D.J.D., and L.G.M. have no conflicts of interest to declare.
REFERENCES
- 1.Lipsitch M, Swerdlow DL, Finelli L. Defining the epidemiology of covid-19—studies needed. N Engl J Med 2020; 382:1194–1196. [DOI] [PubMed] [Google Scholar]
- 2.Fehr AR, Perlman S. Coronaviruses: an overview of their replication and pathogenesis. Methods Mol Biol 2015; 1282:1–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Neuman BW, Adair BD, Yoshioka C, et al. Supramolecular architecture of severe acute respiratory syndrome coronavirus revealed by electron cryomicroscopy. J Virol 2006; 80:7918–7928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Del Rio C, Malani PN. COVID-19—new insights on a rapidly changing epidemic. JAMA 2020; [Epub ahead of print]. [DOI] [PubMed] [Google Scholar]
- 5.Lu R, Zhao X, Li J, et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet 2020; 395:565–574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Neuman BW, Kiss G, Kunding AH, et al. A structural analysis of M protein in coronavirus assembly and morphology. J Struct Biol 2011; 174:11–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhao Y, Zhao Z, Wang Y, et al. Single-cell RNA expression profiling of ACE2, the putative receptor of Wuhan 2019-nCov. bioRxiv 2020; [Epub ahead of print]. [Google Scholar]
- 8.Woelfel R, Corman VM, Guggemos W, et al. Clinical presentation and virological assessment of hospitalized cases of coronavirus disease 2019 in a travel-associated transmission cluster. medRxiv 2020; [Epub ahead of print]. [Google Scholar]
- 9.Tang JW, Li Y, Eames I, et al. Factors involved in the aerosol transmission of infection and control of ventilation in healthcare premises. J Hosp Infect 2006; 64:100–114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Stilianakis NI, Drossinos Y. Dynamics of infectious disease transmission by inhalable respiratory droplets. J R Soc Interface 2010; 7:1355–1366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Seto WH, Tsang D, Yung RW, et al. Effectiveness of precautions against droplets and contact in prevention of nosocomial transmission of severe acute respiratory syndrome (SARS). Lancet 2003; 361:1519–1520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Booth TF, Kournikakis B, Bastien N, et al. Detection of airborne severe acute respiratory syndrome (SARS) coronavirus and environmental contamination in SARS outbreak units. J Infect Dis 2005; 191:1472–1477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yu IT, Li Y, Wong TW, et al. Evidence of airborne transmission of the severe acute respiratory syndrome virus. N Engl J Med 2004; 350:1731–1739. [DOI] [PubMed] [Google Scholar]
- 14.Liu Y, Ning Z, Chen Y, et al. Aerodynamic characteristics and RNA concentration of SARS-CoV-2 aerosol in Wuhan Hospitals during COVID-19 Outbreak. bioRxiv 2020; [Epub ahead of print]. [Google Scholar]
- 15.Ong SWX, Tan YK, Chia PY, et al. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from a symptomatic patient. JAMA 2020; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cheng V, Wong S, Chen J, et al. Escalating infection control response to the rapidly evolving epidemiology of the coronavirus disease 2019 (COVID-19) due to SARS-CoV-2 in Hong Kong. Infect Control Hosp Epidemiol 2020; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wang B, Zhang A, Sun JL, et al. Study of SARS transmission via liquid droplets in air. J Biomech Eng 2005; 127:32–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bourouiba L, Dehandschoewercker E, Bush JW. Violent expiratory events: on coughing and sneezing. J Fluid Mech 2014; 745:537–563. [Google Scholar]
- 19. Interim infection prevention and control recommendations for patients with suspected or confirmed Coronavirus disease 19 (COVID-19) in healthcare settings. [US Centers for Disease Control and Prevention Coronavirus Disease 2019]. March 19, 2020. Available at: https://www.cdc.gov/coronavirus/2019-ncov/infection-control/control-recommendations.html. Accessed March 30, 2020. [Google Scholar]
- 20.Li Q, Guan X, Wu P, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N Engl J Med 2020; 382:1199–1207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wu Z, McGoogan JM. Characteristics of and important lessons from the Coronavirus Disease 2019 (COVID-19) outbreak in China: summary of a report of 72,314 cases from the Chinese Center for Disease Control and Prevention. JAMA 2020; [Epub ahead of print]. [DOI] [PubMed] [Google Scholar]
- 22.Bischoff W, Reid T, Russell GB, et al. Transocular entry of seasonal influenza–attenuated virus aerosols and the efficacy of N95 respirators, surgical masks, and eye protection in humans. J Infect Dis 2011; 204:193–199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lau JT, Fung KS, Wong TW, et al. SARS transmission among hospital workers in Hong Kong. Emerg Infect Dis 2004; 10:280–286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Deng C, Yang Y, Chen H, et al. Ocular detection of SARS-CoV-2 in 114 cases of COVID-19 pneumonia in Wuhan, China: an observational study. Lancet 2020; [Epub ahead of print]. [Google Scholar]
- 25.Van Doremalen N, Bushmaker T, Morris DH, et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N Engl J Med 2020; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Radonovich LJ, Simberkoff MS, Bessesen MT, et al. N95 respirators vs medical masks for preventing influenza among health care personnel: a randomized clinical trial. JAMA 2019; 322:824–833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Loeb M, Dafoe N, Mahony J, et al. Surgical mask vs N95 respirator for preventing influenza among health care workers: a randomized trial. JAMA 2009; 302:1865–1871. [DOI] [PubMed] [Google Scholar]
- 28.Yen MY, Lin YE, Lee CH, et al. Taiwan's traffic control bundle and the elimination of nosocomial severe acute respiratory syndrome among healthcare workers. J Hosp Infect 2011; 77:332–337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Loeb M, McGeer A, Henry B, et al. SARS among critical care nurses, Toronto. Emerg Infect Dis 2004; 10:251–255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bałazy A, Toivola M, Adhikari A, et al. Do N95 respirators provide 95% protection level against airborne viruses, and how adequate are surgical masks? Am J Infect Control 2006; 34:51–57. [DOI] [PubMed] [Google Scholar]
- 31.Wang X, Pan Z, Cheng Z. Association between 2019-nCoV transmission and N95 respirator use. J Hosp Infect 2020; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Radonovich LJ. Respirator tolerance in health care workers. JAMA 2009; 301:36–38. [DOI] [PubMed] [Google Scholar]
- 33.Hines SE, Brown C, Oliver M, et al. User acceptance of reusable respirators in health care. Am J Infect Control 2019; 47:648–655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. [1910.134] - Respiratory Protection. [United States Department of Labor, Occupational Safety and Health Administration]. June 8, 2011. Available at: https://www.osha.gov/laws-regs/regulations/standardnumber/1910/1910.134. Accessed March 29, 2020. [Google Scholar]
- 35.Lidwell OM, Lowbury EJ, Whyte W, et al. Effect of ultraclean air in operating rooms on deep sepsis in the joint after total hip or knee replacement: a randomised study. Br Med J (Clin Res Ed) 1982; 285:10–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Bohn WW, McKinsey DS, Dykstra M, et al. The effect of a portable HEPA-filtered body exhaust system on airborne microbial contamination in a conventional operating room. Infect Control Hosp Epidemiol 1996; 17:419–422. [DOI] [PubMed] [Google Scholar]
- 37.Lu CW, Liu XF, Jia ZF. nCoV transmission through the ocular surface must not be ignored. Lancet 2020; 395:e39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Holappa M, Valjakka J, Vaajanen A. Angiotensin(1-7) and ACE2, “The hot spots” of renin-angiotensin system, detected in the human aqueous humor. Open Ophthalmol J 2015; 9:28–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 2020; 395:507–513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wang D, Hu B, Hu C, et al. Clinical Characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. JAMA 2020; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Huh S. How to train health personnel to protect themselves from SARS-CoV-2 (novel coronavirus) infection when caring for a patient or suspected case. J Educ Eval Health Prof 2020; 17:10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Verbeek JH, Ijaz S, Mischke C, et al. Personal protective equipment for preventing highly infectious diseases due to exposure to contaminated body fluids in healthcare staff. Cochrane Database Syst Rev 2016; 4:CD011621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Rewar S, Mirdha D. Transmission of Ebola virus disease: an overview. Ann Glob Health 2014; 80:444–451. [DOI] [PubMed] [Google Scholar]
- 44.Derrick JL, Gomersall CD. Protecting healthcare staff from severe acute respiratory syndrome: filtration capacity of multiple surgical masks. J Hosp Infect 2005; 59:365–368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Gawande A. Keeping the coronavirus from infecting health-care workers: what Singapore's and Hong Kong's success is teaching us about the pandemics [The New Yorker]. March 21, 2020. Available at: https://www.newyorker.com/news/news-desk/keeping-the-coronavirus-from-infecting-health-care-workers. Accessed March 29, 2020. [Google Scholar]
- 46.Schwartz J, King CC, Yen MY. Protecting health care workers during the COVID-19 Coronavirus outbreak –Lessons from Taiwan's SARS response. Clin Infect Dis 2020; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ng K, Poon BH, Kiat Puar TH, et al. COVID-19 and the risk to health care workers: a case report. Ann Intern Med 2020; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. D’Alessandro MM, Cichowicz JK. Proper N95 respirator use for respiratory protection preparedness. US Centers for Disease Control and Prevention. 2020. Available at: https://blogs.cdc.gov/niosh-science-blog/2020/03/16/n95-preparedness/. Accessed March 28, 2020. [Google Scholar]
- 49. Respirator Trusted -Source Information – Surgical N95 Respirators. [US Centers for Disease Control and Prevention]. March 2, 2020. Available at: https://www.cdc.gov/niosh/npptl/topics/respirators/disp_part/respsource3surgicaln95.html. Accessed March 30, 2020. [Google Scholar]
- 50.Eninger RM, Honda T, Adhikari A, et al. Filter performance of N99 and N95 facepiece respirators against viruses and ultrafine particles. Ann Occup Hyg 2008; 52:385–396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fisher EM, Shaffer RE. Considerations for recommending extended use and limited reuse of filtering facepiece respirators in health care settings. J Occup Environ Hyg 2014; 11:D115–128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Roberge R, Niezgoda G, Benson S. Analysis of forces generated by N95 filtering facepiece respirator tethering devices: a pilot study. J Occup Environ Hyg 2012; 9:517–523. [DOI] [PubMed] [Google Scholar]
- 53.Viscusi DJ, Bergman MS, Eimer BC, et al. evaluation of five decontamination methods for filtering facepiece respirators. Ann Occup Hyg 2009; 53:815–827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Schwartz A, Stiegel M, Greeson N, et al. Decontamination and reuse of N95 respirators with hydrogen peroxide vapor to address worldwide personal protective equipment shortages during the SARS - CoV - 2 (COVID - 19) Pandemic. Appl Biosaf 2020; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Price A, Chu LF. Addressing COVID-19 Face Mask Shortages (v 1.3). [Learnly Anesthesia/Stanford AIM Lab COVID-19 Evidence Service, Stanford, California USA]. March 25, 2020. Available at: https://stanfordmedicine.app.box.com/v/covid19-PPE-1-2. Accessed March 30, 2020. [Google Scholar]
- 56. Otolaryngologists may contract COVID-19 during surgery. [ENTtoday]. March 20, 2020. Available at: https://www.enttoday.org/article/otolaryngologists-may-contract-covid-19-during-surgery/. Accessed April 2, 2020. [Google Scholar]
- 57. Chapter 9 - Health care facilities. In: Kennedy H, ed, 2019 ASHRAE Handbook – HVAC Applications. Atlanta, GA: ASHRAE; 2019:9.1–9.20. [Google Scholar]
- 58.Memarzadeh F, Xu W. Role of air changes per hour (ACH) in possible transmission of airborne infections. Build Simul 2012; 5:15–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zou L, Ruan F, Huang M, et al. SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N Engl J Med 2020; 382:1177–1179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wen X, Li Y. Anesthesia procedure of emergency operation for patients with suspected or confirmed COVID-19. Surg Infect (Larchmt) 2020; 21:299. [DOI] [PubMed] [Google Scholar]
- 61.Wang W, Xu Y, Gao R, et al. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA 2020; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Brat G, Heresey S, Chhabra K, et al. Protecting surgical teams during the COVID-19 Outbreak: a narrative review and clinical considerations. Ann Surg 2020; in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kwak HD, Kim SH, Seo YS, et al. Detecting hepatitis B virus in surgical smoke emitted during laparoscopic surgery. Occup Environ Med 2016; 73:857–863. [DOI] [PubMed] [Google Scholar]
- 64.Zhou Q, Hu X, Zhou J, et al. Human papillomavirus DNA in surgical smoke during cervical loop electrosurgical excision procedures and its impact on the surgeon. Cancer Manag Res 2019; 11:3643–3654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Pryor A. SAGES and EAES recommendations regarding surgical response to COVID-19 crisis. [Society of American Gastrointestinal and Endoscopic Surgeons]. March 29, 2020. Available at: https://www.sages.org/recommendations-surgical-response-covid-19/. Accessed April 2, 2020. [Google Scholar]
- 66.Wong SF, Chow KM, Shek CC, et al. Measures to prevent healthcare workers from contracting severe acute respiratory syndrome during high-risk surgical procedures. Eur J Clin Microbiol Infect Dis 2004; 23:131–133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Wendlandt R, Thomas M, Kienast B, et al. In-vitro evaluation of surgical helmet systems for protecting surgeons from droplets generated during orthopaedic procedures. J Hosp Infect 2016; 94:75–79. [DOI] [PubMed] [Google Scholar]
- 68.Makovicka JL, Bingham JS, Patel KA, et al. Surgeon personal protection: an underappreciated benefit of positive-pressure exhaust suits. Clin Orthop Relat Res 2018; 476:1341–1348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ti LK, Ang LS, Foong TW, et al. What we do when a COVID-19 patient needs an operation: operating room preparation and guidance. Can J Anesth/J Can Anesth 2020; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Wong J, Goh QY, Tan Z, et al. Preparing for a COVID-19 pandemic: a review of operating room outbreak response measures in a large tertiary hospital in Singapore. Can J Anesth/J Can Anesth 2020; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Aminian A, Safari S, Razeghian-Jahromi A, et al. COVID-19 Outbreak and surgical practice: unexpected fatality in peri-operative period. Ann Surg 2020; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]


