Abstract
Background:
Preterm infants who receive differential red blood cell (RBC) transfusions at birth may show brain structure differences across development, including abnormalities in white matter (WM) structure and organization. This study investigated long-term outcomes of brain structure in female infants born preterm, at an average age of 13 years old, who received red blood cell (RBC) transfusions in the neonatal period according to a liberal or restrictive approach. Results from this study will increase understanding of the effects of transfusion on the developing brain.
Study design and methods:
This follow-up study included female preterm infants who participated in a clinical trial and had been randomized at birth to either a liberal or restrictive hematocrit threshold. Brain structures were measured in childhood using structural magnetic resonance imaging (MRI) scans. Due to the low number of females in the restrictive transfusion group at follow-up, additional females were recruited for inclusion. Main outcome measures included cerebral and subcortical brain region volumes.
Results:
Total intracranial volume was significantly decreased in females who were randomized to higher average hematocrit levels at birth. Infants in the liberal transfusion group had proportionately smaller volumes in all measures of regional cerebral WM and subcortical brain volumes, reaching significance for temporal lobe WM and caudate volumes.
Conclusion:
Female premature infants who received a liberal transfusion threshold at birth had decreased WM volumes, which suggests the potential long-term neurodevelopmental risks associated with liberal transfusion practices.
Keywords: Brain, brain structure, premature, preterm, red blood cell, transfusion
Introduction
While survival of very low birth weight (VLBW, <1500 g) infants has markedly improved with advances in perinatal medicine [1,2], these infants remain at risk for several long-term morbidities, including deficits in the motor, sensory, cognitive, and behavioral domains [3–13]. This suggests that brain development, in particular, is an area of vital importance in evaluating premature infant outcomes.
There are multiple risk factors associated with adverse neurodevelopmental outcomes in preterm infants (e.g., low birth weight, early gestational age, male sex, infectious processes and systemic inflammation, various prenatal and maternal factors), many of which may not be intervened upon clinically [14]. However, one possible etiology of altered brain development that can be targeted for intervention is neonatal anemia and its management. Preterm infants are particularly vulnerable to impaired tissue oxygen delivery as a consequence of their small blood volume, immature hematopoietic systems, iatrogenic phlebotomy losses, and ventilator requirements. Thus, red blood cell (RBC) transfusion is a common therapy for preterm infants in the first few weeks of life. It is estimated that 70–80% of VLBW infants born annually in the United States will receive at least one RBC transfusion during their hospital stay [15,16]. But RBC transfusions in the preterm infant are not without risk, and the mechanisms by which this therapy affects the developing preterm infant brain are not well understood [17]. It is not known whether minimizing anemia and maintaining a higher, but normal, hemoglobin level through transfusions (a liberal approach) or tolerating anemia and minimizing transfusions (a restrictive approach) is better for the developing brain [15,18–20]. Clinical practice has shifted towards a more restrictive approach due to growing awareness of transfusion risks [21–24]. Given the frequency of RBC transfusion and the dearth of evidence-based guidelines to direct this therapy, it is crucial to investigate both short- and long-term outcomes of transfusion strategies on the development of the premature infant’s brain.
One of the first randomized clinical trials to directly address differential transfusion policies and their outcomes for preterm infants was reported by Bell et al. [19], in which 100 preterm infants with birth weights of 500–1300 g were randomized into two transfusion groups based on hematocrit threshold (i.e., liberal or restrictive). For each treatment group, transfusion threshold levels consisted of three steps in hematocrit level, which became progressively lower as subjects advanced through three phases of better clinical condition based on their respiratory status. The results of this study demonstrated more frequent major adverse neurological events in the restrictive RBC transfusions group. As an extension to this original trial, Nopoulos et al. investigated long-term outcomes of brain structure at school age for preterm infants who had been randomly assigned to a liberal or restrictive transfusion strategy at birth, as compared to normal healthy controls [25]. Quantitative measures of brain structure were obtained from magnetic resonance imaging (MRI). In comparison to healthy full-term controls, the liberally transfused group showed greater abnormalities in brain structure with substantially lower measures of intracranial volume (ICV) and cerebral white matter (WM) volume; these differences were not significantly different in the restrictive group. Furthermore, within the liberally transfused group, a significant sex-by-group interaction for cerebral WM volume was demonstrated. Females in the liberally transfused group showed a diffuse WM volume decrement across all four cerebral lobes (frontal, temporal, parietal, and occipital), and a decreased volume of subcortical structures (caudate, putamen, and thalamus) compared to healthy controls. In contrast, compared to healthy control group, the males in the liberally transfused group showed only a volume decrement in the caudate and no significant decrement in cerebral WM. These findings contradicted previous implications of better neurodevelopmental outcomes in liberally transfused infants [19,20,26] and instead, suggested the possibility of long-term neurodevelopmental risks associated with liberal transfusion practices. Furthermore, it suggested that this risk was heightened, in particular, for females.
The major limitation of the brain structure study was that sex effects could only be evaluated in the liberal group, as there were only two females with high-quality MRI scans in the restrictive preterm group. Therefore, there remains the possibility that the structural deficits observed in the females of the liberal group are simply a sex effect, generalizable to all premature females compared to males, rather than a specific neurodevelopmental risk for the preterm females in the liberal group. The purpose of this study was to further investigate long-term brain structure outcomes in female preterm infants by augmenting the previous study sample with additional females who had been treated under a restrictive transfusion approach, allowing for a direct comparison in brain structure outcomes between preterm females. By directly investigating brain structure between two transfusion groups in a sex-specific manner, this study may lend further understanding of the relationship between RBC transfusions and long-term brain structure outcomes. We hypothesized that females in the liberally transfused group would show decreased ICV, and proportionately lower volumes of cerebral WM and subcortical structures compared to the females in the restrictive transfusion group.
Materials and methods
Participants
The follow-up brain structure study began in 2005, approximately 13 years following the start of the initial transfusion trial. The follow-up study and current study were approved by the Institutional Review Board. Participants were recruited from the 100 infants in the original transfusion trial who were randomized at birth to receive RBC transfusions based on either liberal or restrictive guidelines as determined by hematocrit (HCT) thresholds [19]. From the original sample of 100 preterm infants, a total of 55 returned for the follow-up study to complete a battery of neurocognitive, behavioral, and brain imaging measures. Reasons for not participating included: 3 were deceased, 17 declined to participate, and 25 could not be contacted. Of the 55 follow-up participants, 44 completed high-quality structural MRI scans. There were no significant differences in demographics between participants and nonparticipants [25].
There were 16 females from the liberal transfusion group but only two females from the restrictive transfusion group in the follow-up study. We recruited seven additional females who were born in the same hospital during the same period as the original transfusion trial and received routine care (similar to restrictive protocol). They met the same inclusion and exclusion criteria, but were not in the original trial (i.e., received no interventions). Details regarding inclusion and exclusion criteria were previously reported [19]. Review of transfusion and HCT measures were obtained from medical record chart review for these additional seven females.
Demographic and neonatal characteristics
Parents completed a demographic questionnaire. Assessment of parental socioeconomic status (SES) was conducted using a Hollingshead-based 5-point scale ranging from 1 (highest income, education, and social status) to 5 (lowest income, education, and social status) [27].
Neonatal characteristics were obtained from the original transfusion trial and from medical chart review for the additional seven females. Illness severity was assessed using the Score for Neonatal Acute Physiology (SNAP) [28] on the day of birth (SNAP0) and as an average for the first week of life (SNAPw1). The original transfusion trial had set transfusion threshold levels for each study group [19]. Hematocrit levels were obtained multiple times a week for all infants and a measure of total average HCT was calculated using each infant’s average weekly HCT, reflecting the mean HCT level over the course of his or her inpatient stay.
Image acquisition
All MRI data were acquired on a 3 T Siemens Trio scanner (Siemens, Erlangen, Germany). The protocol acquired a 3-dimensional T1-weighted magnetization-prepared rapid-acquisition gradient-echo sequence in the coronal plane with 1-mm isotropic resolution. A turbo spin-echo T2-weighted sequence was obtained in the coronal plane with 1-mm isotropic resolution. MRI data previously acquired for the participants of the original long-term follow-up study were reutilized for analyses in this study.
Scans were processed using the BRAINS AutoWorkup pipeline developed locally [29]. The automated pipeline includes spatial alignment of T1-weighted images along the AC-PC line and interhemispheric fissure, coregistration of the multimodal images with the AC-PC aligned T1, bias field correction, and inhomogeneity correction and intensity normalization to provide initial tissue classification and brain segmentation based on the Talairach atlas [30]. An atlas-based anatomical identification procedure was used for defining tissue volumes of gray matter (GM), white matter (WM), and cerebrospinal fluid of the four cerebral lobes (of both hemispheres) and subcortical structures [31]. An automated segmentation algorithm based on an artificial neural network [32] was used to further define subcortical and cerebellar regions of interest and obtain volumetric measurements.
Measures of interest included ICV and total brain tissue, which was further divided into cerebral volume and cerebellum volume. Cerebrum tissue was further divided into cerebral WM and cerebral GM volumes. Cerebral WM was segmented into the four regions of frontal lobe, parietal lobe, temporal lobe, and occipital lobe volumes. Finally, caudate, putamen and thalamus volumes were obtained.
Statistical analyses
Analyses were performed with IBM SPSS Statistics 21.0 (IBM Corporation Software Group, Somers, NY, USA). All brain measures were initially analyzed for group comparisons using a dichotomous approach (restrictive versus liberal preterm groups) with the general linear models analysis of covariance (ANCOVA) procedure. Analyses of ICV included age and height as covariates. All general brain measures (including total tissue, cerebrum, cerebral tissue, cerebral gray, cerebral white, and cerebellum), regional WM volumes (including frontal, temporal, parietal, and occipital lobes), and neural net volumes (including caudate, putamen, and thalamus) were expressed as ratios to ICV. Age was used as a covariate in group analyses. Due to our a-priori hypothesis that the liberal group would show smaller volumes of ICV, WM and subcortical tissue, a one-tailed p value was used.
Correlational studies
Average HCT levels were used as a continuous measure and Pearson correlations were calculated to directly assess the relationship between brain volume morphology and HCT levels using both groups, combined. Age was included as a covariate. To minimize the number of tests, correlations were done only on the brain measures hypothesized to be abnormal (ICV, WM measures, and subcortical measures). Given our hypothesis that greater values of HCT are associated with decrements in ICV, WM, and subcortical region volumes, we utilized a one-tailed p value.
Results
Group demographics
To determine if the seven additional females were similar to the original restrictive group, demographics and neonatal characteristics were compared. Males and females were included for the original transfusion groups, as the low number of females (n = 2) in original restrictive group was inadequate for group comparisons. The seven additional females were older at the time of follow-up assessment but otherwise were comparable to the original restrictive group (see Supplementary Table 1). Figure 1 illustrates average hematocrit (HCT) by study group for all subjects (including males and females of the original study). HCT levels of the additional females were similar to HCT profiles of the restrictive group and were significantly lower than the mean of the liberally transfused group. This supported the assumption that the additional seven females were transfused by a “restrictive” approach. For all group analyses, the additional seven females were included with the two females of the original restrictive group, for a total of nine female participants in the restrictive transfusion group.
Figure 1.
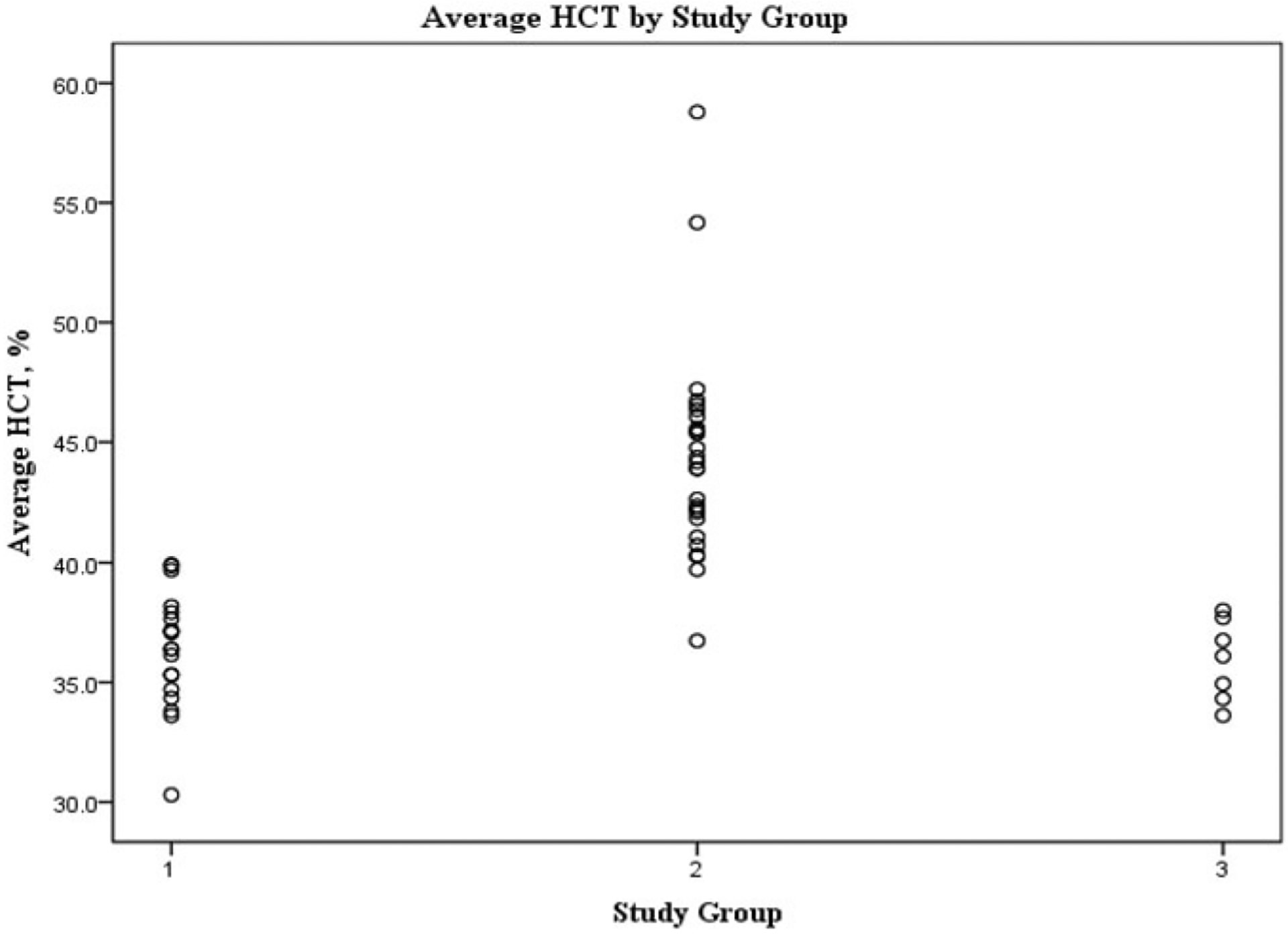
Average HCT by Study Group (restrictive, liberal, and additional females). Where: 1=Restrictive Transfusion Group 2=Liberal Transfusion Group 3=Additional Females
Next, the demographic and neonatal characteristics for the original liberal female group (n = 16) were compared to the newly constructed female restrictive group (n = 9) (see Table 1). As expected, average HCT was significantly different between the transfusion groups. Age at follow-up was significantly different between groups, with the restrictive females being older at time of follow-up.
Table 1.
Independent samples T-test comparing demographic and neonatal characteristics between liberal and restrictive groups.
| Restrictive Females | Liberal Females |
|||||
|---|---|---|---|---|---|---|
| (n=9) | (n=16) | |||||
| Mean | (SD) | Mean | (SD) | F | P | |
| Age, yrs | 13.88 | .80 | 12.59 | 1.37 | 2.080 | 0.018* |
| Head Circumference, cm | 51.23 | 11.65 | 53.63 | 2.03 | 5.459 | 0.424 |
| GA,wks | 27.62 | 2.23 | 29.15 | 2.25 | 0.112 | 0.115 |
| BW,g | 967.22 | 266.34 | 946.94 | 192.07 | 2.807 | 0.827 |
| Height, cm | 157.63 | 5.10 | 150.14 | 8.20 | 1.880 | 0.021* |
| Overall Avg HCT, % | 35.74 | 2.84 | 44.30 | 5.46 | 1.221 | 0.000* |
| Week 1 Avg HCT, % | 45.18 | 4.40 | 51.17 | 7.29 | 1.264 | 0.035* |
| Lowest HCT, % | 24.11 | 2.37 | 32.25 | 7.01 | 2.646 | 0.003* |
| No. of Days on Oxygen | 54.67 | 38.57 | 33.38 | 50.14 | 0.014 | 0.283 |
| No. of Days on Ventilator | 14.67 | 18.34 | 14.13 | 21.08 | 0.003 | 0.949 |
| SNAP0 | 12.44 | 7.80 | 13.38 | 7.67 | 0.301 | 0.775 |
| SNAPw1 | 8.22 | 5.85 | 6.56 | 4.57 | 1.901 | 0.439 |
| Total No. of Transfusions | 3.00 | 2.45 | 3.38 | 3.01 | 0.510 | 0.763 |
| SES | 2.67 | 0.83 | 3.00 | 0.63 | 2.147 | 0.270 |
Note:
Significant at p<.05 level
Illness severity (Score for Neonatal Acute Physiology, SNAP22 ratings) are reported as an average of the daily ratings obtained beginning on the first day of life (SNAP0) and once daily for the first week of life (SNAPw1).
Note: Participant Age at time of MRI in years (Age), Head circumference in centimeters at time of follow-up, Gestational Age in weeks (GA), Birth Weight in grams (BW), Average Hematocrit in % (Avg HCT),Score for Neonatal Acute Physiology at birth and week 1 average (SNAP0, SNAPw1), Parental Socioeconomic Status (SES, ranging from 1 [highest income, education, and social prestige] to 5 [lowest education, income, and social prestige] so that the lower numbers reflect higher social class), Mother and Father Educational Achievement in years (Mom Ed, Dad Ed), intracranial volume in milliliters (ICV)
Group comparisons
Table 2 shows all general, regional white, and subcortical brain measures for restrictive and liberal females. Total ICV was significantly decreased in females of the liberal transfusion group (p = .042). After controlling for the global decrement in volume of the liberal female group, there were no significant differences in any general brain volume measure, though cerebral WM showed a trend towards decreased volume in the liberal transfusion group (p = .078). In all measures of regional cerebral WM and subcortical volumes, the liberal female group had proportionately smaller absolute volumes compared to the restrictive female group. These differences were statistically significant for temporal lobe WM (p = .019) and caudate (p = .033).
Table 2.
BRAINS AutoWorkup general, regional white, & subcortical brain measures compared between female preterm transfusion groups.a
| Estimated Means of Volume, mL | ||||||
|---|---|---|---|---|---|---|
| Variable (Brain Area) | RESTRICTIVE (n=9) | LIBERAL (n=16) | F | (One-Tailed) P Value |
Effect Size | |
| ICV∞ | 1289.50 | 1198.25 | 3.316 | .042* | .889 | |
| [Brain Area] Volume % of ICVb | ||||||
| Variable (Brain Area) | RESTRICTIVE (n=9) | LIBERAL (n=16) | F | (One-Tailed) P Value |
Effect Size | |
| Total Tissue | 91.43 | 91.88 | .136 | .358 | .175 | |
| Cerebellum | 9.05 | 9.35 | .576 | .228 | .359 | |
| Cerebrum | 80.08 | 80.10 | .000 | .494 | .007 | |
| Cerebral Gray | 51.04 | 52.12 | 1.371 | .127 | .554 | |
| Cerebral White | 29.04 | 27.98 | 2.154 | .078 | .694 | |
| Frontal White | 11.24 | 10.98 | .5232 | .239 | .342 | |
| Temporal White | 4.85 | 4.49 | 4.9198 | .019* | 1.049 | |
| Parietal White | 6.91 | 6.89 | .0116 | .458 | .051 | |
| Occipital White | 2.52 | 2.31 | 1.5986 | .110 | .598 | |
| Caudate | .5516 | .5192 | 3.766 | .033* | .918 | |
| Putamen | .8104 | .7986 | .123 | .365 | .166 | |
| Thalamus | .9019 | .8970 | .024 | .440 | .073 | |
Note:
All general, regional white, & subcortical brain measures are adjusted for age;
Volumes (cc’s) to ICV ratios (shown as a percent of ICV); this accounts for the global decrement in volume of the liberal female group, by performing the comparison on the proportional measure; general linear models analysis of covariance (ANCOVA) procedure used for all analyses.
Controlling for age & height;
Significant at p < .05 level
Correlational analyses
Correlations were calculated to describe the relationship of average HCT with each brain measure that showed significant differences in the group comparisons (i.e., total ICV, temporal white, and caudate), as well as with all WM brain measures (see Supplementary Table 2). Every correlation was negative, indicating that higher HCT was associated with lower volumes of brain tissue. The correlations between average HCT and every WM measure all reached statistical significance (cerebral white: r = −0.545, p = .003; frontal white: r = −0.392, p = .029; parietal white: r = −0.368, p =.038; occipital white: r = −0.469, p = .010) as well as for both the caudate and thalamus (r = −0.416, p = .022; r = −0.425, p = .019 respectively) Figure 2 significant illustrates negative correlation of cerebral WM with average HCT.
Figure 2.

Correlation of cerebral white matter with overall mean hematocrit levels. Pearson correlation; r2 = .297; 95% confidence intervals shown Where: ◆Liberal ○Restrictive
Discussion
The current investigation is an extension of previous studies that investigated outcomes in preterm infants after differential transfusion strategies [19,25,33,34], and provides further evidence of potential long-term risks, specifically deficits of white matter (WM) brain structure, associated with liberal transfusion practice in the female premature infant. The group comparisons show that liberally transfused females have significantly smaller ICVs. After controlling for the global deficit in ICV, regional volumes of temporal lobe WM and caudate structure were significantly decreased in females in the liberal group. Other general and regional WM analyses at the level of the cerebral lobes and individual subcortical nuclei volumes did not demonstrate significant differences between groups, although a majority of these brain areas were descriptively lower in the liberal group. All WM volumes, caudate, and thalamic volumes demonstrated significant negative correlations with average hematocrit levels, indicating that the brain volume abnormalities observed in the female group comparisons were directly related to the hematocrit level. This pattern of decreased WM volumes is consistent with results of structural and neurocognitive studies demonstrating worse outcomes by the liberal group, particularly females [25,33,34].
WM injury is known to be the most common injury to the preterm infant’s brain. Damage to the brain’s WM structure may underlie the cognitive impairments observed both early and in adolescence by infants born prematurely [7,35–38]. In one study of early brain development in preterm infants at term-equivalent age, several complex and bidirectional associations were demonstrated between degree of prematurity and specific regional brain volumes, including bilateral cerebral WM [39]. Two major pathologic hallmarks of WM injury are activation of microglia and depletion of “premyelinating” oligodendrocytes [40]. Premyelinating oligodendrocytes are highly vulnerable to injury by excitotoxicity, free radicals, and proinflammatory cytokines. In preterm infants, increased proinflammatory cytokine production and endothelial activation has been shown to be associated with transfusion-associated necrotizing enterocolitis [41]. Thus, there may be a role for inflammation caused by transfusion in the pathogenesis of brain injury in these preterm infants (i.e., female preterm infants who receive more RBC transfusions in the neonatal period may potentially have the largest inflammatory reaction and subsequently, the most structural WM abnormality). Another possible etiology of brain injury is the suppression of erythropoietin in the liberally transfused females (i.e., higher hemoglobin thresholds). Erythropoietin has been shown to be neuroprotective in animal models and human trials, with a range of actions postulated to underlie these neuroprotective effects such as anti-inflammatory, antiapoptotic, antioxidant, and neurotrophic [42–47]. However, future studies investigating the role of erythropoietin suppression in response to transfusion are essential.
An important limitation to the current study was the inclusion of the seven additional females for supplementation to the original restrictive group from the original quantitative brain outcome study [25]. The original transfusion trial was not initially designed as a long-term follow-up study. Participants were invited to return after roughly 12 years with no interim contact maintained, resulting in lower and unequal number of participants per transfusion and sex groups. As noted in the previous long-term outcome study [25], there was significant loss to follow-up from the original transfusion trial. Nopoulos et al. previously evaluated for potential bias regarding neonatal characteristics in the participants (n = 55) compared with nonparticipants (n = 45), and found there were no differences between groups [25]. Because the current standard of care in transfusion strategy is closer to the restrictive threshold used in these studies, the additional females recruited for inclusion in this study were similar to the original restrictive females in neonatal and demographics characteristics, with only age at the time of study being significantly different. However, age was used as a covariate in all structural brain volume analyses in order to reduce its potentially confounding effects. The additional seven infants also had more days on oxygen, more RBC transfusions, and lower SNAP scores than infants from the original trial. While not statistically significant, these may be potential clinically important considerations. Although the number of transfusions was also descriptively higher in the additional recruited females, a better marker for clinical status is average hematocrit, which was used in this study (and similar between groups). Another significant limitation is the small sample size and use of one-tailed statistical tests. While the statistical power may have been insufficient to find differences, the effect sizes for some of the differences, such as for the group differences in temporal WM (Cohen’s d = 1.05) and caudate volume (Cohen’s d = 0.918), were high. Finally, this study represents a cross-sectional view of the preterm infant’s brain volumes at a single time point in development. Future studies are necessary to truly elucidate the specific maturational insults to the developing neonatal brain, as well as developmental trajectories and aberrations associated with liberal RBC transfusion.
Given the high frequency of RBC transfusions for treatment of anemia in premature infants [16], it is crucial to investigate the long-term neurodevelopmental outcomes of this intervention. The results of the current study lend further support to the notion of adverse long-term outcomes in premature infants who received liberal transfusions during the neonatal period, specifically the risk for abnormal brain structural development by affecting the white matter of preterm females.
Supplementary Material
Financial support
This study was supported by NIH grant 2P01HL046925, National Institute for Heart, Lung and Blood (NHLBI). Preterm Transfusions: Brain Structure and Function Outcomes.
Footnotes
Supplemental data for this article can be accessed here https://www.tandfonline.com/doi/full/10.1080/14767058.2019.1683157.
Disclosure statement
There are no conflicts of interest for any author.
References
- [1].Murphy SL, Mathews TJ, Martin JA, et al. Annual summary of vital statistics: 2013–2014. Pediatrics. 2017; 139(6):e20163239. [DOI] [PubMed] [Google Scholar]
- [2].Grisaru-Granovsky S, Reichman B, Lerner-Geva L, et al. Population-based trends in mortality and neonatal morbidities among singleton, very preterm, very low birth weight infants over 16 years. Early Hum Dev. 2014;90(12):821–827. [DOI] [PubMed] [Google Scholar]
- [3].Wilson-Costello D Is there evidence that long-term outcomes have improved with intensive care? Semin Fetal Neonat Med. 2007;12(5):344–354. [DOI] [PubMed] [Google Scholar]
- [4].Stoll BJ, Hansen NI, Bell EF, et al. Trends in care practices, morbidity, and mortality of extremely preterm neonates, 1993–2012. JAMA. 2015;314(10):1039–1051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Stoll BJ, Hansen NI, Bell EF, et al. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics. 2010;126(3): 443–456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Horbar JD, Carpenter JH, Badger GJ, et al. Mortality and neonatal morbidity among infants 501 to 1500 grams from 2000 to 2009. Pediatrics. 2012;129(6): 1019–1026. [DOI] [PubMed] [Google Scholar]
- [7].Mukerji A, Shah V, Shah PS. Periventricular/intraventricular hemorrhage and neurodevelopmental outcomes: a meta-analysis. Pediatrics. 2015;136(6): 1132–1143. [DOI] [PubMed] [Google Scholar]
- [8].Saigal S, Doyle LW. An overview of mortality and sequelae of preterm birth from infancy to adulthood. Lancet. 2008;371(9608):261–269. [DOI] [PubMed] [Google Scholar]
- [9].Orchinik LJ, Taylor HG, Espy KA, et al. Cognitive outcomes for extremely preterm/extremely low birth weight children in kindergarten. J Int Neuropsychol Soc. 2011;17(6):1067–1079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Aarnoudse-Moens CS, Weisglas-Kuperus N, van Goudoever JB, et al. Meta-analysis of neurobehavioral outcomes in very preterm and/or very low birth weight children. Pediatrics. 2009;124(2):717–728. [DOI] [PubMed] [Google Scholar]
- [11].Anderson P, Doyle LW, Victorian Infant Collaborative Study Group. Neurobehavioral outcomes of school-age children born extremely low birth weight or very preterm in the 1990s. JAMA. 2003;289(24): 3264–3272. [DOI] [PubMed] [Google Scholar]
- [12].Bhutta AT, Cleves MA, Casey PH, et al. Cognitive and behavioral outcomes of school-aged children who were born preterm: a meta-analysis. JAMA. 2002; 288(6):728–737. [DOI] [PubMed] [Google Scholar]
- [13].Johnson S, Marlow N. Early and long-term outcome of infants born extremely preterm. Arch Dis Child. 2017;102(1):97–102. [DOI] [PubMed] [Google Scholar]
- [14].Xiong T, Gonzalez F, Mu DZ. An overview of risk factors for poor neurodevelopmental outcome associated with prematurity. World J Pediatr. 2012;8(4): 293–300. [DOI] [PubMed] [Google Scholar]
- [15].Patel RM, Knezevic A, Shenvi N, et al. Association of red blood cell transfusion, anemia, and necrotizing enterocolitis in very low-birth-weight infants. JAMA. 2016;315(9):889–897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Ekhaguere OA, Morriss FH Jr., Bell EF, et al. Predictive factors and practice trends in red blood cell transfusions for very-low-birth-weight infants. Pediatr Res. 2016;79(5):736–741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Howarth C, Banerjee J, Aladangady N. Red blood cell transfusion in preterm infants: current evidence and controversies. Neonatology. 2018;114(1):7–16. [DOI] [PubMed] [Google Scholar]
- [18].Wang YC, Chan OW, Chiang MC, et al. Red blood cell transfusion and clinical outcomes in extremely low birth weight preterm infants. Pediatr Neonatol. 2017; 58(3):216–222. [DOI] [PubMed] [Google Scholar]
- [19].Bell EF, Strauss RG, Widness JA, et al. Randomized trial of liberal versus restrictive guidelines for red blood cell transfusion in preterm infants. Pediatrics. 2005;115(6):1685–1691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Kirpalani H, Whyte RK, Andersen C, et al. The Premature Infants in Need of Transfusion (PINT) study: a randomized, controlled trial of a restrictive (low) versus liberal (high) transfusion threshold for extremely low birth weight infants. J Pediatr. 2006; 149(3):301–307. [DOI] [PubMed] [Google Scholar]
- [21].Maier RF, Sonntag J, Walka MM, et al. Changing practices of red blood cell transfusions in infants with birth weights less than 1000 g. J Pediatr. 2000;136(2): 220–224. [DOI] [PubMed] [Google Scholar]
- [22].Widness JA, Seward VJ, Kromer IJ, et al. Changing patterns of red blood cell transfusion in very low birth weight infants. J Pediatr. 1996;129(5):680–687. [DOI] [PubMed] [Google Scholar]
- [23].Wallace AH, Dalziel SR, Cowan BR, et al. Long-term cardiovascular outcome following fetal anaemia and intrauterine transfusion: a cohort study. Arch Dis Child. 2017;102(1):40–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Cunningham KE, Okolo FC, Baker R, et al. Red blood cell transfusion in premature infants leads to worse necrotizing enterocolitis outcomes. J Surg Res. 2017; 213:158–165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Nopoulos PC, Conrad AL, Bell EF, et al. Long-term outcome of brain structure in premature infants: effects of liberal vs restricted red blood cell transfusions. Arch Pediatr Adolesc Med. 2011;165(5):443–450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Whyte RK, Kirpalani H, Asztalos EV, et al. Neurodevelopmental outcome of extremely low birth weight infants randomly assigned to restrictive or liberal hemoglobin thresholds for blood transfusion. Pediatrics. 2009;123(1):207–213. [DOI] [PubMed] [Google Scholar]
- [27].Hollingshead AB. Four factor index of social status (Unpublished manuscript). New Haven (CT): Yale University. 1975. [Google Scholar]
- [28].Richardson DK, Gray JE, McCormick MC, et al. Score for Neonatal Acute Physiology: a physiologic severity index for neonatal intensive care. Pediatrics. 1993; 91(3):617–623. [PubMed] [Google Scholar]
- [29].Pierson R, Johnson H, Harris G, et al. Fully automated analysis using BRAINS: AutoWorkup. NeuroImage. 2011;54(1):328–336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Talairach JT. Co-planar stereotaxic atlas of the human brain. New York (NY): Thieme Medical Publishers, Inc; 1988. [Google Scholar]
- [31].Andreasen NC, Rajarethinam R, Cizadlo T, et al. Automatic atlas-based volume estimation of human brain regions from MR images. J Comput Assist Tomogr. 1996;20(1):98–106. [DOI] [PubMed] [Google Scholar]
- [32].Powell S, Magnotta VA, Johnson H, et al. Registration and machine learning-based automated segmentation of subcortical and cerebellar brain structures. NeuroImage. 2008;39(1):238–247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].McCoy TE, Conrad AL, Richman LC, et al. The relationship between brain structure and cognition in transfused preterm children at school age. Dev Neuropsychol. 2014;39(3):226–232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].McCoy TE, Conrad AL, Richman LC, et al. Neurocognitive profiles of preterm infants randomly assigned to lower or higher hematocrit thresholds for transfusion. Child Neuropsychol J Norm Abnorm Dev Child Adolesc. 2011;17(4):347–367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Soria-Pastor S, Gimenez M, Narberhaus A, et al. Patterns of cerebral white matter damage and cognitive impairment in adolescents born very preterm. Int J Dev Neurosci. 2008;26(7):647–654. [DOI] [PubMed] [Google Scholar]
- [36].Dyet LE, Kennea N, Counsell SJ, et al. Natural history of brain lesions in extremely preterm infants studied with serial magnetic resonance imaging from birth and neurodevelopmental assessment. Pediatrics. 2006;118(2):536–548. [DOI] [PubMed] [Google Scholar]
- [37].Thompson DK, Lee KJ, Egan GF, et al. Regional white matter microstructure in very preterm infants: predictors and 7 year outcomes. Cortex. 2014;52:60–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Rogers CE, Smyser T, Smyser CD, et al. Regional white matter development in very preterm infants: perinatal predictors and early developmental outcomes. Pediatr Res. 2016;79(1):87–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Rogers EE, Hintz SR. Early neurodevelopmental outcomes of extremely preterm infants. Semin Perinatol. 2016;40(8):497–509. [DOI] [PubMed] [Google Scholar]
- [40].Kaindl AM, Favrais G, Gressens P. Molecular mechanisms involved in injury to the preterm brain. J Child Neurol. 2009;24(9):1112–1118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Dani C, Poggi C, Gozzini E, et al. Red blood cell transfusions can induce proinflammatory cytokines in preterm infants. EDITORIAL. 2017;57(5):1304–1310. [DOI] [PubMed] [Google Scholar]
- [42].Bierer R, Peceny MC, Hartenberger CH, et al. Erythropoietin concentrations and neurodevelopmental outcome in preterm infants. Pediatrics. 2006; 118(3):e635–e640. [DOI] [PubMed] [Google Scholar]
- [43].Neubauer AP, Voss W, Wachtendorf M, et al. Erythropoietin improves neurodevelopmental outcome of extremely preterm infants. Ann Neurol. 2010; 67(5):657–666. [DOI] [PubMed] [Google Scholar]
- [44].Rees S, Hale N, De Matteo R, et al. Erythropoietin is neuroprotective in a preterm ovine model of endotoxin-induced brain injury. J Neuropathol Exp Neurol. 2010;69(3):306–319. [DOI] [PubMed] [Google Scholar]
- [45].Zhu C, Kang W, Xu F, et al. Erythropoietin improved neurologic outcomes in newborns with hypoxicischemic encephalopathy. Pediatrics. 2009;124(2): e218–e226. [DOI] [PubMed] [Google Scholar]
- [46].Brown MS, Eichorst D, Lala-Black B, et al. Higher cumulative doses of erythropoietin and developmental outcomes in preterm infants. Pediatrics. 2009; 124(4):e681–e687. [DOI] [PubMed] [Google Scholar]
- [47].Liu W, Shen Y, Plane JM, et al. Neuroprotective potential of erythropoietin and its derivative carbamylated erythropoietin in periventricular leukomalacia. Exp Neurol. 2011;230(2):227–239. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


