Significance Statement
Currently, the clinical presentation, optimal management strategy, and outcomes for patients with kidney transplants who develop COVID-19 infection remain unknown. The description of our cohort represents the first cohort of patients with kidney transplants and COVID-19 infection and includes clinical features, markers of inflammation, and a strategy for management that includes both immunosuppression reduction and the use of adjuvant therapy, including hydroxychloroquine, azithromycin, and tocilizumab. This approach appears to have resulted in favorable outcomes in our cohort of hospitalized kidney transplant patients and provides an effective treatment strategy for the management of these patients
Keywords: transplant outcomes, kidney transplantation, Epidemiology and outcomes, COVID-19
Abstract
Background
The novel SARS-CoV-2 virus has caused a global pandemic of coronavirus disease 2019 (COVID-19). Although immunosuppressed individuals are thought to be at an increased risk of severe disease, little is known about their clinical presentation, disease course, or outcomes.
Methods
We report 15 kidney transplant recipients from the Columbia University kidney transplant program who required hospitalization for confirmed COVID-19, and describe their management, clinical course, and outcomes.
Results
Patients presented most often with a fever (87%) and/or cough (67%). Initial chest x-ray most commonly showed bilateral infiltrates, but 33% had no acute radiographic findings. Patients were managed with immunosuppression reduction and the addition of hydroxychloroquine and azithromycin. Although 27% of our patients needed mechanical ventilation, over half were discharged home by the end of follow-up.
Conclusions
Kidney transplant recipients with COVID-19 have presentations that are similar to that of the general population. Our current treatment protocol appears to be associated with favorable outcomes, but longer follow-up of a larger cohort of patients is needed.
Coronavirus disease 2019 (COVID-19), caused by the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus, first spread to the United States in January 2020, with the first case in New York City diagnosed at the end of the following month.1,2 Since that time, the New York metropolitan area has become the epicenter of COVID-19 in the United States, with approximately 40% of confirmed COVID-19 cases and 25% of all reported COVID-19 deaths as of March 28, 2020. Given that the metropolitan area is home to nine major academic transplant centers, there is significant concern regarding the susceptibility, presentation, and ideal management of organ transplant recipients who develop COVID-19. International data regarding the management and prognosis for kidney transplant recipients with COVID-19 has been limited to case reports.3,4 Here, we describe 15 consecutive cases of COVID-19 among kidney transplant recipients at our center, Columbia University Medical Center, who required hospitalization through March 27, 2020.
Case Series
The 15 patients included in this series had a median age of 51 (interquartile range, 28–72) years and were predominantly men (65%) and deceased donor kidney recipients (80%), with a median time since transplant of 49 (interquartile range, 38–118; range, 0–232) months (Table 1). All but one patient were taking tacrolimus at the time of COVID-19 diagnosis, and most (80%) were also taking either mycophenolate mofetil or mycophenolic acid. Despite our status as an early steroid withdrawal center for most transplants, ten patients (67%) were taking prednisone at the time of COVID-19 diagnosis. The underlying cause of ESKD varied.
Table 1.
Characteristics of kidney transplant recipients with COVID-19
| Characteristics | All Patients, n=15 |
|---|---|
| Baseline characteristics | |
| Age, yr | 51 (IQR, 28–72; range, 21–78) |
| Female, n (%) | 5 (33) |
| Time since transplant, mo | 49 (IQR, 38–118; range, 0–232) |
| Deceased donor, n (%) | 12 (80) |
| Multiorgan recipient, n (%) | 2 (13) |
| Maintenance immunosuppression, n (%) | |
| Tacrolimus | 14 (93) |
| Mycophenolate mofetil or mycophenolic acid | 12 (80) |
| Belatacept | 2 (13) |
| Leflunomide | 1 (7) |
| Azathioprine | 1 (7) |
| Prednisone | 10 (67) |
| Clinical presentation, n (%) | |
| Fever | 13 (87) |
| Cough | 9 (60) |
| Fatigue/malaise | 4 (27) |
| Dyspnea (exertional or rest) | 4 (27) |
| Diarrhea | 3 (20) |
| Myalgia | 2 (13) |
| Hemoptysis | 1 (7) |
| Emesis | 1 (7) |
| Laboratory tests on diagnosisa | |
| White blood cell count, ×1000/μl (n=13) | 4.8 (range, 2.1–12.7) |
| Absolute lymphocyte count, /μl (n=11) | 800 (range, 110–1410) |
| Ferritin, ng/ml (n=12) | 471 (range, 93–1963) |
| Lactate dehydrogenase, U/L (n=12) | 275 (range, 113–450) |
| Procalcitonin, ng/ml (n=13) | 0.46 (range, 0.08–18.7) |
| Erythrocyte sedimentation rate, mm/h (n=12) | 40.5 (range, 0–75) |
| C-reactive protein, mg/L (n=13) | 104 (range, 0.3–232) |
| IL-6, pg/ml (n=12) | 24 (range, <5–120) |
| Initial chest x-ray, n (%) | |
| Multifocal/bilateral patchy opacities | 7 (47) |
| No acute findings | 5 (33) |
| Left lower lobe opacities | 1 (7) |
| Right lower lobe opacity | 1 (7) |
| Report not available | 1 (7) |
Data are displayed as n (%), median (IQR), or median (range). COVID-19, coronavirus disease 2019; IQR, interquartile range.
n<15 because of some patients being diagnosed at outside hospitals or as outpatients, or inconsistent laboratory test ordering upon admission.
Patients reported symptom onset ranging from 1 day to nearly 3 weeks before admission. The most common presenting symptom was fever, which was reported in 13 (87%) cases, followed by cough, which was present in nine (60%) cases (Table 1). Only one patient had neither fever nor cough as a presenting symptom, and instead reported exertional dyspnea and malaise. Three patients (20%) reported diarrhea, and only two patients (13%) reported myalgias. Two patients were in the hospital for 6 and 7 days, respectively, before developing a fever and being tested for COVID-19.
About half of our patients had bilateral/multifocal opacities noted on initial chest x-ray, whereas two patients (13%) had lobar opacities and five patients (33%) had unremarkable radiographs initially (Table 1). Among patients for whom laboratory data obtained at the time of diagnosis were available, median white blood cell count was 4.8×103/μl (range, 2.1–12.7) and median absolute lymphocyte count was 800/μl (range, 110–1410). We observed wide variation in admission values for ferritin (median, 471 ng/ml; range, 93–1963), lactate dehydrogenase (median, 275 U/L; range, 113–450), procalcitonin (median, 0.46 ng/ml; range, 0.08–18.70), erythrocyte sedimentation rate (median, 40.5 mm/h; range, 0–75), C-reactive protein (median, 104 mg/L; range, 0.3–232), and IL-6 (median, 24 pg/ml; range, <5–120).
The primary change in immunosuppression in the majority of patients was complete cessation of antimetabolites or leflunomide (ten out of 14, 71%) while continuing the tacrolimus (with a goal trough of 4–7 ng/ml) and the baseline prednisone in those individuals who were on maintenance prednisone (Table 2). One patient who was on a regimen of tacrolimus and high-dose prednisone was switched to a regimen of prednisone 20 mg only, as was a patient who had just completed a course of thymoglobulin induction therapy (6 mg/kg) after a deceased donor transplantation. Two patients were also on maintenance belatacept, including one patient who was on a four-drug regimen and a second patient whose dose was deferred because of their severe ongoing hypoxemia. (Tables 3 and 4).
Table 2.
Clinical management and outcomes of kidney transplant recipients with COVID-19
| Clinical Management and Outcomes | All Patients, n=15 (%) |
|---|---|
| Change in immunosuppression | |
| Discontinued only MMF/MPA/AZA/leflunomide | 10/14 (71) |
| Prednisone decreased | 1/10 (10) |
| Belatacept infusion postponed | 1/2 (50) |
| Discontinued all immunosuppression | 2 (14) |
| Replaced tacrolimus and MMF with prednisone | 1 (7) |
| No change | 1 (7) |
| Anti–COVID-19 therapies | |
| Hydroxychloroquine without azithromycin | 4 (27) |
| Hydroxychloroquine plus azithromycin | 9 (60) |
| Tocilizumab | 1 (7) |
| Outcomes | |
| AKI | 6 (40) |
| Intubation required | 4 (27) |
| Days between admission and intubation (n=4) | 5 (range, 0–9) |
| Hospitalization disposition | |
| Died | 2 (13) |
| Discharged | 8 (53) |
| Days between admission and discharge (n=8) | 4.5 (range, 0–9) |
| Hospitalization ongoing | 6 (40) |
| Days between admission and end of follow-up (n=6) | 7 (range, 3–11) |
Data are displayed as n (%) or median (range). COVID-19, coronavirus disease 2019; MMF, mycophenolate mofetil; MPA, mycophenolic acid; AZA, azathioprine.
Table 3.
Detailed description of individual cases, including individual cases and select laboratory tests at admission
| Case | Age, yr | Sex | Months since Transplant | Presenting Symptoms | Symptom Duration before Admission | Initial Chest X-Ray Findings | WBC Count, ×1000/ul | Absolute Lymphocyte Count | Ferritin, ng/mL | LDH, U/L | Procalcitonin, ng/mL | ESR, mm/hr | CRP, mg/L | IL-6, pg/mL |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 70 | M | 60 | Fever, cough, fatigue | 2–3 wk | No acute findings | 4.8 | 500 | 155 | 409 | 0.34 | 16 | 100 | 89.5 |
| 2 | 64 | M | 232 | Fever, cough, fatigue | 4 d | Bilateral mid and lower lung reticular opacities and hazy bibasilar opacities | ||||||||
| 3 | 28 | M | 42 | Fever, cough, myalgia | 1 d | Bilateral haziness and patchy opacities (left greater than right) | 11.7 | 860 | 187 | 193 | 17.05 | 60 | 173 | <5 |
| 4 | 51 | M | 118 | Fever, cough | 9 d | Bilateral multifocal patchy opacities | 2.8 | 370 | 1514 | 231 | 0.86 | 64 | 129 | 120 |
| 5 | 32 | F | 14 | Fever, dyspnea, diarrhea | Same day | Right lower lobe hazy opacity | 5.7 | 1160 | 173 | 338 | 0.26 | 27 | 134 | 13 |
| 6 | 21 | M | 46 | Fever, fatigue, diarrhea | 4 d | |||||||||
| 7 | 36 | M | 38 | Fever, myalgia | 2 d | Left lower lobe opacities | 3.4 | 850 | 879 | 113 | 0.15 | 75 | 11 | 8 |
| 8 | 72 | F | 49 | Fever, cough, dyspnea | 2–3 d | No acute findings | 3.6 | 790 | 0.08 | 0 | ||||
| 9 | 51 | F | 9 | Fever, cough | 1 d | Diffuse multifocal opacities | 3.8 | 110 | 760 | 450 | 3.66 | 0 | 55 | 51 |
| 10 | 76 | M | 136 | Fever, diarrhea | 1 d | No acute findings | 8.1 | 810 | 93 | 205 | 0.13 | 38 | 6 | 120 |
| 11 | 61 | M | 0 | Fever, cough | 1 d | No acute findings | 3.6 | 1963 | 419 | 18.69 | 43 | 130 | 34 | |
| 12 | 22 | M | 34 | Fever, exertional dyspnea | 2 d | No acute findings | 2.1 | 230 | 821 | 231 | 3.93 | 31 | 104 | 16 |
| 13 | 78 | M | 117 | Exertional dyspnea, malaise | 1 wk | Bilateral patchy opacities | 5.7 | 860 | 453 | 318 | 0.46 | 38 | 208 | 10 |
| 14 | 72 | F | 120 | Fever, cough, hemoptysis | 3–4 d | Diffuse interstitial airspace opacities with upper lobe predominance | 5.5 | 1410 | 467 | 330 | 0.14 | 50 | 74 | 32 |
| 15 | 25 | F | 80 | Cough, diarrhea, emesis | 1 wk | Bilateral hazy opacities | 12.7 | 390 | 476 | 224 | 0.62 | 52 | 232 | 5 |
WBC, white blood cell; LDH, lactate dehydrogenase; ESR, erythrocyte sedimentation rate; CRP, c-reactive protein; M, male; F, female.
Table 4.
Detailed description of treatment of individual cases and their outcomes at last follow-up
| Case | Admission Immunosuppression | Immunosuppression Change | Anti–COVID-19 Therapy | Mechanical Ventilation Required | AKI | AKI Outcome | RRT Required | Symptoms to Intubation | Hospitalization Outcome |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Belatacept, MPA, prednisone | Held MPA, postponed belatacept | Hydroxychloroquine, azithromycin | Yes | Resolved | Deceased | |||
| 2 | Tacrolimus, MMF, prednisone | Held MPA | Hydroxychloroquine, azithromycin | Discharged home | |||||
| 3 | Tacrolimus, azathioprine, prednisone, adalimumab | Held azathioprine | Hydroxychloroquine, azithromycin | Yes | Ongoing | Discharged home | |||
| 4 | Tacrolimus, MMF, prednisone | Held MPA | Hydroxychloroquine, azithromycin | Yes | Resolving | Discharged home | |||
| 5 | Tacrolimus, MMF, prednisone | Held MMF | Hydroxychloroquine | Discharged home | |||||
| 6 | Tacrolimus, MMF | No change | Discharged home | ||||||
| 7 | Belatacept, tacrolimus, MMF, prednisone | Held MMF | Hydroxychloroquine | Discharged home | |||||
| 8 | Tacrolimus, MPA | Held all immunosuppression | Hydroxychloroquine, azithromycin | Discharged home | |||||
| 9 | Tacrolimus, MMF, prednisone | Held MMF | Hydroxychloroquine | Yes | 6 | Remains intubated (day 8 of mechanical ventilation) | |||
| 10 | Tacrolimus, leflunomide | Held leflunomide | Hydroxychloroquine | Discharged home | |||||
| 11 | Tacrolimus, MMF | Held tacrolimus and MMF, started prednisone | Yes | Yes | Ongoing | yes | 5 | Remains intubated (day 6 of mechanical ventilation) | |
| 12 | Tacrolimus, prednisone | Decrease prednisone and tacrolimus | Hydroxychloroquine, azithromycin, tocilizumab | Yes | yes | 5 | Extubated on day 5 | ||
| 13 | Tacrolimus, MMF, prednisone | Held MMF | Hydroxychloroquine, azithromycin | Yes | Yes | Resolving | 7 | Deceased | |
| 14 | Tacrolimus, MMF | Held MMF | Hydroxychloroquine, azithromycin | Remains hospitalized | |||||
| 15 | Tacrolimus, MMF, prednisone | Held MMF | Hydroxychloroquine, azithromycin | Yes | Ongoing | Remains hospitalized |
Patients 2 and 6 were admitted to other hospitals, as a result, admission lab data are not available. The remaining empty cells in the table are missing information. COVID-19, coronavirus disease 2019; MPA, mycophenolic acid; MMF, mycophenolate mofetil.
As per the Columbia University COVID-19 protocol, in addition to holding the antimetabolite, 13 (87%) patients received hydroxychloroquine, including nine that received it with adjunctive azithromycin. Hydroxychloroquine therapy was not used for the recent transplant recipient and for the patient first admitted to another institution. A single dose of tocilizumab was given to the patient whose immunosuppression was reduced to prednisone alone shortly after intubation for acute respiratory distress syndrome. This patient has since been successfully been extubated on day 5 of mechanical ventilation.
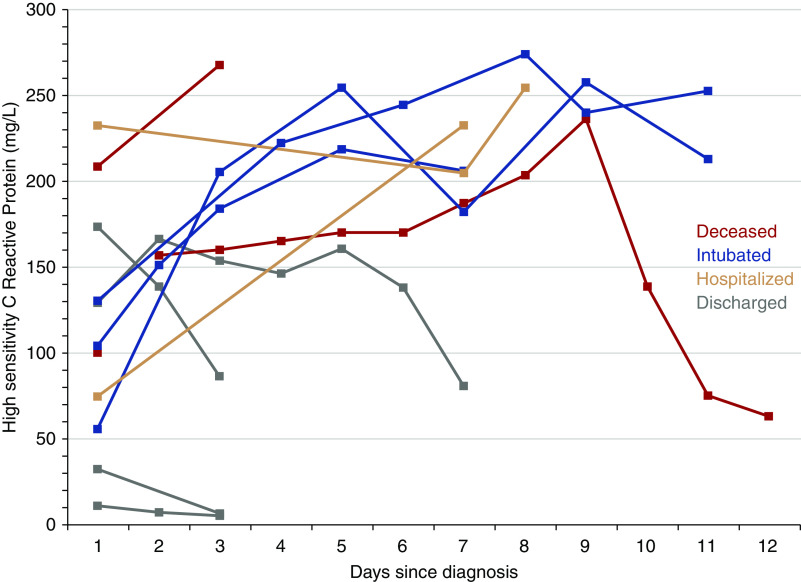
Six patients (40%) had AKI, although none had a kidney biopsy performed to determine the cause. Four patients (27%) required intubation and mechanical ventilation between 0 and 9 days after admission (Table 2), of whom three remain on mechanical ventilation and two died because of severe acute respiratory distress syndrome, including one who declined mechanical ventilation. Patients who required mechanical ventilation were intubated between days 5 and 7 after the onset of symptoms (Table 4). In addition, one patient developed symptoms during an inpatient stay while being treated for an acute antibody-mediated rejection with high-dose steroids and plasmapheresis, which were stopped, and another patient developed symptoms after completion of a course of thymoglobulin induction therapy after a deceased donor transplantation. Both of these patients, who are in their first-year post transplant, remain intubated and on mechanical ventilation at this time. Among the patients who developed AKI, only two patients, both of whom were intubated, required RRT. One of these patients was experiencing delayed graft function at the time of COVID-19 diagnosis, and the other patient had a failing allograft at the time of diagnosis. At the end of follow-up, an additional three patients remain hospitalized outside of the intensive care unit, and eight have been discharged home at a median of 4.5 (range, 0–9) days after admission. Available high-sensitivity C-reactive protein trends for patients are shown in Figure 1.
Figure 1.
Heterogeneity in high-sensitivity C-reactive protein measurements at admission and subsequently during the course of hospitalization.
Discussion
The sudden and rapid spread of COVID-19 throughout the globe has resulted in early uncertainty in the identification and management of this disease. Although the general understanding of the clinical presentation of COVID-19 is improving, information about select patient groups who may warrant special consideration, such as transplant recipients, remains limited. Here, we present a series of 15 cases of COVID-19 in kidney transplant recipients at our center who required hospitalization.
The most common presenting symptom we observed was fever, followed by cough, similar to larger reports from general population cohorts.1,5 Clinical presentation in our patients was consistent with those seen in case reports of kidney transplant recipients from Spain and Wuhan, where patients presented with fever that preceded a dry cough by several days, or cough, dyspnea, and chest tightness.3,4 We should note that the majority of the patients in our cohort were on prednisone at the time of diagnosis despite our status as an early steroid withdrawal center for most kidney transplant recipients, perhaps suggesting that the greater immunosuppression associated with the use of an immunosuppressive three-drug regimen may predispose patients to a more severe infection requiring hospitalization.
The ideal treatment for kidney transplant recipients with COVID-19 remains uncertain at present. Although the Columbia University COVID-19 protocol is to withdraw the antimetabolite and introduce hydroxychloroquine (with azithromycin in the absence of QT interval prolongation), the true efficacy of this approach remains unclear given the relatively small number of adverse outcomes to date. In both previously published case reports of COVID-19 in kidney transplant recipients, maintenance immunosuppression was also reduced. The patient reported from Wuhan recovered after cessation of immunosuppression and treatment with methylprednisolone, intravenous Ig, and IFNα.4 For the Spanish patient, maintenance tacrolimus and everolimus were both stopped, and treatment was initiated with lopinavir/ritonavir and hydroxychloroquine.3 IFNβ was eventually added when the patient’s hypoxia worsened and they subsequently required intubation.
Unfortunately, there are many questions pertaining to the management of kidney transplant patients with COVID-19 that currently rely on expert opinion because of the paucity of evidence. The management of transplant recipients with mild symptoms as outpatients via telemedicine is a strategy that we have used on a case-by-case basis. However, attempting to identify patients who are likely to progress is currently a challenge, and relies on the relatively insensitive subjective assessment of worsening dyspnea on exertion and self-reported vital signs, along with the usual recommendations for self-isolation. This approach is not without risks, particularly in those individuals thought to be at significantly increased risk, given the rapid nature of decompensation seen among patients who eventually go on to develop acute respiratory distress syndrome, and needs further study to determine the optimal strategy.6
Although more than half of our patients have been successfully discharged, the optimal timing of the reintroduction of immunosuppressive agents is not yet clear. Current estimates are that viral shedding can occur for up to 2 weeks or more after improvement of symptoms, but there is also considerable variation (maximum observed shedding of 37 days).7–9 There is also an association between severity of illness and peak viral loads, which may in turn influence the duration of subsequent viral shedding.10 Thus, given the present uncertainty, our current clinical practice has been to delay reintroduction of these agents for up to 2 weeks after discharge, recognizing that prolonged reduction of immunosuppression increases the risk of allograft rejection.
We observed that 27% of our cases required intubation, a proportion that is similar for cases in New York City overall. Over half of the patients in our series were discharged home by the time this manuscript was prepared, and only two had died. Despite concerns about a possible biphasic nature of the illness, to date, none of the discharged patients have been readmitted with worsening disease. Although these findings are encouraging regarding the prognosis of transplant recipients who develop COVID-19 and require hospitalization, the large number of patients who remain hospitalized makes it impossible to draw any conclusions in this patient population regarding the overall rate of respiratory failure requiring intubation, or death. In addition to these 15 hospitalized patients, at least eight additional kidney transplant recipients in our program have tested positive for SARS-CoV-2 infection, but to date have not required hospitalization. Additionally, the limited availability of COVID-19 testing in New York City makes it likely that mild cases of the disease have remained unrecognized among other transplant recipients at our center.
In conclusion, among 15 kidney transplant recipients at our center with COVID-19, overall presentation was similar to that reported for the general population. Although many of our patients experienced a favorable outcome with our current treatment strategy, the small cohort and varied additional therapies makes it difficult to draw any conclusions beyond that of short-term safety and tolerability of our protocol. Longer-term follow-up is required to better understand the prognosis and sequelae of COVID-19 in immunosuppressed kidney transplant recipients.
Disclosures
Dr. Hardy reports grants from NHLBI, outside the submitted work. Dr. Husain reports grants from NCATS, during the conduct of the study. Dr. Ratner reports personal fees from Natera, personal fees from CSL Behring, personal fees from Sanofi, outside the submitted work; and I own a small amount of stock in Hansa BioPharma. Dr. Cohen reports personal fees from Natera, other from Alexion Pharmaceuticals, outside the submitted work. Dr. Mohan reports grants from NIDDK/NIH, during the conduct of the study; grants and other from Angion Pharmaceuticals, personal fees from Kidney International Reports, outside the submitted work.
Funding
Dr. Husainis supported by National Center for Advancing Translational Sciences grant KL2-TR001874. Dr. Mohan is supported by National Institute of Diabetes and Digestive and Kidney Diseases grants R01-DK114893 and U01-DK116066, and National Institute on Minority Health and Health Disparities grant R01-MD14161. Dr. Hardy is supported by National Heart, Lung, and Blood Institute grant T32-HL007854-21.
Supplementary Material
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
See related editorial, “The Immunocompromised Transplant Recipient and SARS-CoV-2 Infection,” on pages 1147–1149.
Contributor Information
Collaborators: S. Ali Husain, Jae H. Chang, David J. Cohen, R. John Crew, Geoffrey K. Dube, Hilda E. Fernandez, Heather K. Morris, Justin G. Aaron, Benjamin A. Miko, Marcus R. Pereira, Mark A. Hardy, Kasi R. McCune, Lloyd E. Ratner, P. Rodrigo Sandoval, Joshua Weiner, Demetra Tsapepas, and Sumit Mohan
References
- 1.Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al.: Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395: 497–506, 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Holshue ML, DeBolt C, Lindquist S, Lofy KH, Wiesman J, Bruce H, et al.; Washington State 2019-nCoV Case Investigation Team: First case of 2019 novel coronavirus in the United States. N Engl J Med 382: 929–936, 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Guillen E, Pineiro GJ, Revuelta I, Rodriguez D, Bodro M, Moreno A, et al.: Case report of COVID-19 in a kidney transplant recipient: Does immunosuppression alter the clinical presentation? [published online ahead of print Mar 20, 2020]. Am J Transplant doi:10.1111/ajt.15874 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhu L, Xu X, Ma K, Yang J, Guan H, Chen S, et al.: Successful recovery of COVID-19 pneumonia in a renal transplant recipient with long-term immunosuppression [published online ahead of print Mar 17, 2020]. Am J Transplant doi:10.1111/ajt.15869 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. ; China medical treatment expert group for Covid-19: Clinical characteristics of coronavirus disease 2019 in China [published online ahead of print Feb 28, 2020]. N Engl J Med doi:10.1056/NEJMoa2002032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Centers for Disease Control and Prevention: Interim Clinical Guidance for Management of Patients with Confirmed Coronavirus Disease (COVID-19), 2020. Available at: https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-guidance-management-patients.html. Accessed March 30, 2020.
- 7.Chu C-M, Poon LL, Cheng VC, Chan K-S, Hung IF, Wong MM, et al.: Initial viral load and the outcomes of SARS. CMAJ 171: 1349–1352, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chang, Mo G, Yuan X, Tao Y, Peng X, Wang F, et al. : Time kinetics of viral clearance and resolution of symptoms in novel coronavirus infection [published online ahead of print Mar 23, 2020]. Am J Respir Crit Care Med doi:10.1164/rccm.202003-0524LE [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al.: Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study [published correction appears in Lancet 395: 1038, 2020]. Lancet 395: 1054–1062, 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Liu Y, Yan L-M, Wan L, Xiang T-X, Le A, Liu J-M, et al.: Viral dynamics in mild and severe cases of COVID-19 [published online ahead of print Mar 19, 2020]. Lancet Infect Dis doi:10.1016/S1473-3099(20)30232-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.