Abstract
Background
In the literature, there are still controversies regarding the effect of phytosterol(PS) supplementation on fasting blood sugar (FBS), insulin levels and glycosylated hemoglobin (HbA1c) in humans. We aimed to assess the impact of PS supplementation on FBS, HbA1c and insulin levels by conducting a systematic review and meta-analysis of the available randomized controlled trials (RCTs).
Methods
A comprehensive search was conducted to identify all RCTs published up to May 2019 in the following databases: PubMed-MEDLINE, Web of Science, Cochrane Library and Scopus. The mean difference with 95% confidence intervals (CIs) was pooled using a random-effects model (DerSimonian-Laird method).
Results
Twenty-six arms from 20 RCTs were included in the present meta-analysis. Our findings show that PS supplementation decreases insulin levels (mean difference [MD]: −6.426 μU/ml, 95% CI: −7.187, −5.665, P- value = 0.000). However, PS supplementation did not have significant effects on FBS and HbA1c levels. Following PS supplementation, significant changes in FBS (mean difference [MD]: −1.942 mg/dl, 95% CI: −3.637, −0.246, P- value = 0.025) and HbA1c (mean difference [MD]: −0.059%, 95% CI: −0.114, −0.004, P- value = 0.035) based on PS dosage (mg/d) were recorded.
Conclusions
In patients with a baseline BMI <25 kg/m2, PS consumption significantly increased FBS levels. Patients who consumed 1–2 g/day of PS had a lower FBS and lower HbA1c levels.
Electronic supplementary material
The online version of this article (10.1007/s40200-020-00526-z) contains supplementary material, which is available to authorized users.
Keywords: Phytosterol, Fasting blood sugar, Glycosylated hemoglobin, Insulin
Introduction
Phytosterols (stanols and sterols) are a group of integral components of the cell membrane in plants. Usually, the human diet contains approximately 200–300 mg per day of plant sterols [1]. The main dietary sources of phytosterols (PS) are vegetable oils (corn, soybean, sunflower, and olive), nuts, fruits and seeds [2]. Since the early 1950s, PS have been shown to lower cholesterol absorption in animals [3]. PS are structurally similar to cholesterol with some slight differences in the side chain of the molecule [4]. It has been suggested that due to the chemical similarity with cholesterol, plant sterols could block cholesterol absorption. Several meta-analyses have focused on lipid-lowering, anti-obesity and anti-inflammatory effects of PS [5–7].
Over the last decade, the hypoglycemic activity of PS has attracted the attention of many researchers [8, 9]. One of first studies to provide evidence regarding this effect of PS has shown that, in hyperglycemic rats, glucose levels decreased and insulin levels increased after oral administration of ß-sitosterol [10]. It has been suggested that the uptake of glucose induced by PS is mediated by the adenine monophosphate-activated protein kinase (AMPK) [11]. Hyperglycemia and diabetes have become increasingly prevalent worldwide. In 2012, about 2.2 million deaths were attributed to high blood glucose [12]. In 2015, the global cost of diabetes was appreciated at 1.31 trillion dollars [13]. Numerous trials have investigated the effect of drugs, dietary interventions, supplements or physical exercise in the management of diabetes [14].
The impact of plant sterols/stanols on plasma glucose and insulin concentrations have been investigated by several clinical trials [9, 15–17] which have yielded conflicting results. Therefore, we performed a meta-analysis of the available randomized controlled trials regarding the effects of PS on fasting blood sugar (FBS), glycosylated hemoglobin (HbA1c) and insulin levels in order to clarify the impact of PS on these indices.
Materials and methods
Search strategy and selection criteria
We followed the preferred reporting items from the systematic reviews and meta-analysis (PRISMA statement) during the preparation of this systematic review [18]. Two study authors independently conducted a systematic search of the databases PubMed-MEDLINE, Web of Science, Cochrane Library and Scopus (all years to May 31st 2019).
The used keywords were relevant to the objectives of the study. The search strategy was conducted using MeSH and non-MeSH keywords without date or language restrictions. The terms used in the electronic search included: (“phytosterol”[Mesh]) OR “phytosterol”[Title/Abstract] “phytostanol”[Mesh] OR “phytostanol”[Title/Abstract] OR “plantsterol”[Mesh] OR”plantsterol”[Title/Abstract] OR “plant stanol”[Mesh] OR “plant stanol”[Title/Abstract] OR “sitosterol”[Mesh] OR “sitosterol”[Title/Abstract] OR “sitostanol”[Mesh] OR “sitostanol”[Title/Abstract] OR “campesterol”[Mesh] OR “campesterol”[Title/Abstract] OR “campestanol”[Mesh] OR “campestanol”[Title/Abstract] OR “stigmasterol”[Mesh] OR “stigmasterol”[Title/Abstract] OR “stigmastanol”[Mesh] OR “stigmastanol”[Title/Abstract] OR brassicasterol[Mesh] OR brassicasterol[Title/Abstract] AND “Intervention Studies”[Mesh] OR “intervention”[Title/Abstract] OR “controlled trial”[Title/Abstract] OR “randomized”[Title/Abstract] OR “randomised” [Title/Abstract]..
A manual search was carried out using the Google Scholar search engine and the references were cited in appropriate systematic reviews. The search in PubMed, Web of Science and Cochrane Library was restricted to humans and the English language, and for Scopus, it was restricted to article, the English language, humans, and medicine. We examined the titles and abstracts of the obtained studies. Finally, the full texts of selected studies were investigated according to the inclusion/exclusion criteria.
Eligibility criteria
Studies were included if they met the following criteria: 1) the study was a randomized clinical trial (RCT), 2) the subjects involved were given PS supplements 3) the authors reported sufficient information about FBS, HbA1c and Homeostatic Model Assessment for Insulin Resistance (HOMA-IR), 4) the studies were conducted on adult subjects (>18 years). Studies were excluded if they met the following exclusion criteria: 1) non-RCTs studies, 2) the studies were performed on children or animals, 3) the studies lacked a placebo group, 4) duplicate publications 5) the studies did not report on fasting blood sugar, HbA1c and HOMA-IR at baseline and at the end of the intervention.
Data extraction
Two authors independently extracted and coded all the data. Disagreements were resolved by consensus with the third author. The relevant data extracted included: study identification (author, year and country), study design (cross-over or parallel, level of blinding), duration, sample size of each group, participant characteristics (age, gender, BMI, health status), intervention characteristics, study location, supplement dosage. (Supplementary Table 1).
Quality assessment
Two independent research investigators (A.S. and H.K.) assessed the methodological quality of the selected full texts using the Cochrane criteria [19]. Thus, the quality of the selected studies was evaluated based on the allocation concealment, sequence generation sufficiency, blinding, clarification of failures (imperfect outcome data), selective reporting of the results, and other probable sources of bias. According to the Cochrane Handbook recommendation, the studies were stratified as low risk of bias, high risk of bias or unclear regarding each domain (Supplementary Table 2).
Statistical analysis
The mean change and standard deviation (SD) for FBS, HbA1c and HOMA-IR were used to estimate the overall effect size of the intervention. Estimates of effect sizes were expressed based on weighted mean difference (MD) and 95% CI from the random-effects model. We assessed the heterogeneity among the studies using the I2 test that determines the between-study variance and demonstrates whether the observed inconsistency among studies is real or by chance [20–22]. There was between-study heterogeneity if I2 > 50%. Publication bias was evaluated using visual assessment of funnel plots and Egger’s weighted regression tests. The nonlinear potential effects of PS dosage (g/day) and treatment duration (weeks) were examined using fractional polynominal modeling (polynomials). All statistical analyses were performed using the Stata software, version 12 (Stata Corp, College Station, Texas, USA) and a P value <0.05 was considered as significant.
Results
Study selection
Out of 3.965 articles identified in the PubMed-MEDLINE, Scopus, Cochrane Library and Web of Science databases, 381 duplicate articles were excluded. We further excluded 3.205 publications based on the title and abstract screening approach. We reviewed the remaining 379 articles by reading the full text and excluded 377 studies due to the following reasons: studies that included a co-intervention along with PS only in one group (n = 6), review articles (n = 7), studies reporting duplicate data (n = 4), studies included no data of interest (n = 360) (Fig. 1).
Fig. 1.
Flow diagram for the studies identified and included into the meta-analysis
Study characteristics
The characteristics of the eligible studies are summarized in Supplementary Table 2. The sample size of the included studies was between 21 and 244 participants. Out of the 20 included studies, six studies were performed in Europe, 4 in America and 10 studies in Asia. The duration of the trials was between 3 and 24 weeks. One study was conducted in women only [23], one in men only [24] and the rest of the eligible studies involved both genders. Seventeen studies had a single-arm parallel group whereas three studies had a crossover design [9, 24–37].
Meta-analysis results
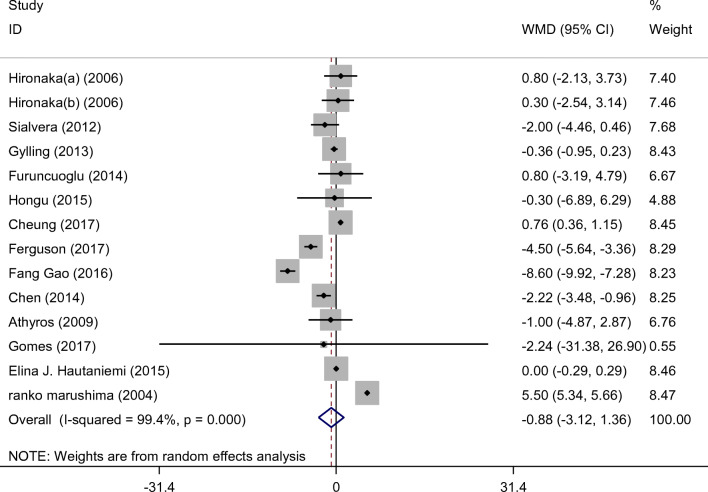
Fourteen studies including a total of 1306 participants (case = 655, and control = 651) reported FBS as an outcome measure. Combined results from the random-effects model indicated that FBS did not change significantly following PS consumption (mean difference (MD): −0.878 mg/dl, 95% CI: −3.115, 1.359, P value = 0.442) with significant heterogeneity among the studies (I2 = 99.4%, P value = 0.000) (Fig. 2).
Fig. 2.
Forest plot presenting mean difference (WMD) and 95% confidence intervals for the impact of Phytosterols Supplementation on FBS
Four studies including a total of 504 participants (case = 251, and control = 253) reported insulin levels as an outcome measure. Overall results from the random-effects model indicated that PS consumption did result in significant change in insulin levels (mean difference (MD): −6.426 μU/ml, 95% CI: −7.187, −5.665, P value = 0.000). There was no significant heterogeneity among these studies (I2 = 0.0%, P value = 0.540) (Fig. 3).
Fig. 3.
Forest plot presenting mean difference (WMD) and 95% confidence intervals for the impact of Phytosterols Supplementation on insulin level
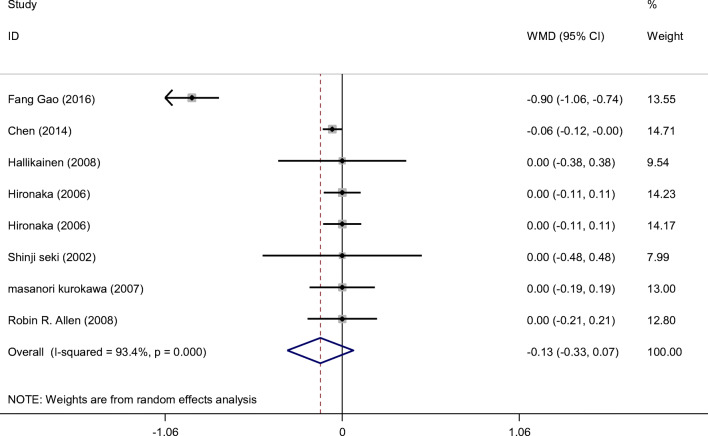
Among the eligible studies, five studies including a total of 634 participants (case = 325, and control = 309) reported an association between PS consumption and HbA1c levels. PS consumption did not alter HbA1c levels significantly (mean difference (MD): −0.131%, 95% CI: −0.329, 0.067, P value = 0.195). There was a significant heterogeneity among these studies (I2 = 93.4%, P value = 0.000) (Fig. 4).
Fig. 4.
Forest plot presenting mean difference (WMD) and 95% confidence intervals for the impact of Phytosterols Supplementation on HbA1c
Subgroup analysis
The results of the subgroup analyses are provided in Table 1. We stratified the studies based on BMI (<25 kg/m2, ≥25 kg/m2), PS dosage (g/day), trial duration (weeks) and type of study population. The subgroup analysis showed that PS supplementation in participants with a baseline BMI <25 kg/m2 significantly increased FBS (mean difference (MD): 0.748 mg/dl, 95% CI: 0.364, 1.113, P value = 0.000). Moreover, FBS was significantly reduced in the subgroup given 1–2 g of PS (mean difference (MD): −1.942 mg/dl, 95% CI: −3.637, −0.246, P value = 0.025). In addition, HbA1c was significantly reduced in the subgroup receiving 1–2 g of PS (mean difference (MD): −0.059%, 95% CI: −0.114, −0.004, P value = 0.035).
Table 1.
Subgroup analysis
| Sub grouped by | No. of trials | WMD195% CI | P Value | P for heterogeneity | I2 (%) | P for between subgroup heterogeneity |
|---|---|---|---|---|---|---|
| FBS | ||||||
| Baseline BMI | 0.000 | |||||
| <25 kg/m2 | 3 | 0.748 0.364 1.133 | 0.000 | 0.952 | 0.0 | |
| ≥25 kg/m2 | 11 | −1.329 -4.141 1.483 | 0.354 | 0.000 | 99.5 | |
| Dosage | 0.000 | |||||
| <1 g | 2 | 3.386 -1.196 7.969 | 0.145 | 0.002 | 89.9 | |
| 1–2 g | 10 | −1.942 -3.637 -0.246 | 0.025 | 0.000 | 96.3 | |
| ˃2 g | 2 | −0.725 -2.063 0.612 | 0.288 | 0.205 | 37.8 | |
| Intervention duration (weeks) | 0.000 | |||||
| ≤8 | 4 | −1.961 -5.360 1.439 | 0.258 | 0.008 | 96.9 | |
| 8–15 | 5 | 0.972 -2.721 4.666 | 0.606 | 0.433 | 99.6 | |
| ≥16 | 5 | −1.857 -6.153 2.439 | 0.397 | 0.941 | 96.5 | |
| Type of Study Population | ||||||
| Healthy | 4 | 1.634 -2.048 5.315 | 0.385 | 0.000 | 99.6 | 0.000 |
| Hyperlimidemic Patients | 5 | −0.924 -3.788 1.941 | 0.527 | 0.000 | 82.4 | |
| Metabolic syndrome | 3 | −3.535 -9.671 2.602 | 0.259 | 0.000 | 98.7 | |
| HbA1c | ||||||
| Baseline BMI | 0.002 | |||||
| <25 kg/m2 | 3 | 0.000 -0.071 0.071 | 1.00 | 1.000 | 0.0 | |
| ≥25 kg/m2 | 5 | −0.320 -0.863 0.223 | 0.248 | 0.000 | 97.9 | |
| Dosage | 0.013 | |||||
| <1 g | 2 | 0.000 -0.096 0.096 | 1.000 | 1.000 | 0.0 | |
| 1–2 g | 4 | −0.059 -0.114 -0.004 | 0.035 | 0.806 | 0.0 | |
| Intervention duration (weeks) | ||||||
| ˂8 | 2 | −0.248 -0.616 0.120 | 0.189 | 0.000 | 97.0 | 0.051 |
| 8–15 | 3 | 0.000 -0.133 0.133 | 1.000 | 1.000 | 0.0 | |
| ≥16 | 3 | −0.297 -0.804 0.211 | 0.252 | 0.000 | 97.9 | |
| Type of Study population | 0.000 | |||||
| Diabetes | 2 | −0.468 -1.349 0.414 | 0.298 | 0.000 | 94.4 | |
| Hyperlipidemic Patients | 4 | 0.000 -0.069 0.069 | 1.000 | 1.000 | 0.0 | |
BMI = Body Mass Index; FBS = fasting blood sugar; HbA1c = glycosylated hemoglobin
Publication bias
The evaluation of the publication bias by visual inspection of the funnel plot demonstrated no evidence of publication bias in the meta-analysis of PS supplementation on FBS, insulin levels and HbA1c (Supplementary Fig. 1). The Egger’s linear regression test also revealed the same result (FBS: P value =0.053, insulin levels: P value = 0.198), and HbA1c: P value = 0.690). The sensitivity analyses did not revealed any effect of a single study on the combined results.
Discussion
Using systematic review and meta-analytical methods, we pooled the evidence from the 14 published RCTs regarding the effect of PS on FBS, HbA1c and insulin levels. The pooled effect estimates for change showed that the consumption of PS did not had a significant effect on FBS and HbA1c. The pooled estimate from five studies showed that PS consumption was associated with a decline in insulin levels. In patients with a baseline BMI <25 kg/m2, PS consumption significantly increased FBS levels. The serum levels of FBS and HbA1c was significantly decreased in the subjects who consumed 1–2 g/day of PS.
A good control of FBS, HbA1c and insulin levels are of major importance and would be of interest in the management of conditions such as insulin resistance and diabetes. In the therapy of these afflictions, a simple dietary intervention such as PS supplementation could be beneficial. Moreover, several meta-analyses have concluded that PS supplementation leads to a decline in obesity indices [38], improves atherogenic and anti-Atherogenic apolipoproteins [39], reduces serum lipoprotein a (Lp(a)) and free fatty acid (FFA) [40] and improves the lipid profile in humans [41].
PS are natural compounds found in foods (such as vegetable oils, cereals, fruit and vegetables) which are safe and effective in lowering cholesterol levels [42–44] . Currently, various marketed food products contain PS: milk, orange juice, yoghurt, snack bars and enriched margarine. Although the lipid-lowering effect of PS has already been presented in several studies, [45, 46] the role of PS in reducing FBS-, HbA1c and insulin levels is yet to be established. Although PS- enriched foods have been shown to exhibit beneficial effects in diabetic patients, the exact mechanism of action explaining the perceived favorable effects of PS in diabetes is still unclear. A plausible mechanisms regarding the effect of PS on these indices might be related to the expression and translocation of glucose transporter 4 in the skeletal muscle, liver and white adipose tissue, which have been reported to increase after PS supplementation [43]. In vitro studies have also mentioned that PS can stimulate the AMPK pathway. AMPK activators are virtuous candidates in the treatment of obesity, metabolic syndrome, hypercholesterolemia and type 2 diabetes mellitus [47–49]. β-sitosterol stimulates the phosphorylation of AMPK whose activation enhances the glucose transporter 4 translocation and expression [11, 50]. In addition, PS could increment adiponectin concentrations which could activate AMPK in situ [52] and can enhance the activity of glucose transporters [51, 52].
This is the first systematic review and meta-analysis of RCTs examining the influence of PS supplementation on FBS, insulin and HbA1c indices. However, there are some limitations that must be recognized, such as the different durations of the intervention, the preparation method and the formula of supplementation. PS existent in our foods are mixed with other dietary compounds. Therefore, other nutritional factors may also modulate the effects of PS in humans. We did not adjust for certain confounding elements (for instance, the dietary consumption of vegetables, whole grains and fruits), which may have influenced the outcomes regarding the association between PS consumption and the investigated variables. Moreover, there was a significant heterogeneity among the included studies and the random effect model was used to derive WMD. The different populations included in the primary RCTs, their baseline characteristics and other factors may have contributed to the heterogeneity [53, 54].
In our meta-analysis, the significant reduction in the FBS and HbA1c levels was recorded at a dose of 1–2 g/day of PS, which is a high dose for the usual diet of humans and hard to achieve in habitual diets [55].
Conclusion
The present meta-analysis revealed that PS consumption was associated with a decline in insulin levels. In subgroup analysis, the patients with a baseline BMI <25 kg/m2, PS consumption significantly increased FBS levels, whereas in subjects consuming 1–2 g/day of PS, FBS and HbA1c levels declined.
Electronic supplementary material
(DOCX 31 kb)
Acknowledgments
This study is related to the project NO. 1397/69281 From Student Research Committee, Shahid Beheshti University of Medical Sciences (SBMU), Tehran, Iran. We also appreciate the Student Research Committee and Research & Technology Chancellor in SBMU for their financial support of this study.
Compliance with ethical standards
Conflict of interest
None.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Azita Hekmatdoost, Email: a_hekmat2000@yahoo.com.
Parvin Mirmiran, Email: parvin.mirmiran@gmail.com.
References
- 1.Cabral CE, Klein MRST. Phytosterols in the treatment of hypercholesterolemia and prevention of cardiovascular diseases. Arq Bras Cardiol. 2017;109(5):475–482. doi: 10.5935/abc.20170158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ostlund RE., Jr Phytosterols in human nutrition. Annu Rev Nutr. 2002;22(1):533–549. doi: 10.1146/annurev.nutr.22.020702.075220. [DOI] [PubMed] [Google Scholar]
- 3.De Smet E, Mensink RP, Plat J. Effects of plant sterols and stanols on intestinal cholesterol metabolism: suggested mechanisms from past to present. Mol Nutr Food Res. 2012;56(7):1058–1072. doi: 10.1002/mnfr.201100722. [DOI] [PubMed] [Google Scholar]
- 4.Gupta A, Savopoulos C, Ahuja J, Hatzitolios A. Role of phytosterols in lipid-lowering: current perspectives. QJM: An International Journal of Medicine. 2011;104(4):301–308. doi: 10.1093/qjmed/hcr007. [DOI] [PubMed] [Google Scholar]
- 5.Shaghaghi MA, Abumweis SS, Jones PJ. Cholesterol-lowering efficacy of plant sterols/stanols provided in capsule and tablet formats: results of a systematic review and meta-analysis. J Acad Nutr Diet. 2013;113(11):1494–1503. doi: 10.1016/j.jand.2013.07.006. [DOI] [PubMed] [Google Scholar]
- 6.Wester I. Cholesterol-lowering effect of plant sterols. Eur J Lipid Sci Technol. 2000;102(1):37–44. doi: 10.1002/(SICI)1438-9312(200001)102:1<37::AID-EJLT37>3.0.CO;2-1. [DOI] [Google Scholar]
- 7.Demonty I, Ras RT, van der Knaap HC, Duchateau GS, Meijer L, Zock PL, Geleijnse JM, Trautwein EA. Continuous dose-response relationship of the LDL-cholesterol–lowering effect of phytosterol intake. J Nutr. 2008;139(2):271–284. doi: 10.3945/jn.108.095125. [DOI] [PubMed] [Google Scholar]
- 8.Sialvera T, Pounis G, Koutelidakis A, Richter D, Yfanti G, Kapsokefalou M, et al. Phytosterols supplementation decreases plasma small and dense LDL levels in metabolic syndrome patients on a westernized type diet. Nutr Metab Cardiovasc Dis. 2012;22(10):843–848. doi: 10.1016/j.numecd.2010.12.004. [DOI] [PubMed] [Google Scholar]
- 9.Li Q, Xing B. A phytosterol-enriched spread improves lipid profile and insulin resistance of women with gestational diabetes mellitus: a randomized, placebo-controlled double-blind clinical trial. Diabetes Technol Ther. 2016;18(8):499–504. doi: 10.1089/dia.2016.0103. [DOI] [PubMed] [Google Scholar]
- 10.Ivorra M, D'ocon M, Paya M, Villar A. Antihyperglycemic and insulin-releasing effects of beta-sitosterol 3-beta-D-glucoside and its aglycone, beta-sitosterol. Arch Int Pharmacodyn Ther. 1988;296:224–231. [PubMed] [Google Scholar]
- 11.Hwang S-L, Kim H-N, Jung H-H, Kim J-E, Choi D-K, Hur J-M, Lee JY, Song H, Song KS, Huh TL. Beneficial effects of β-sitosterol on glucose and lipid metabolism in L6 myotube cells are mediated by AMP-activated protein kinase. Biochem Biophys Res Commun. 2008;377(4):1253–1258. doi: 10.1016/j.bbrc.2008.10.136. [DOI] [PubMed] [Google Scholar]
- 12.Hirsh SP, Pons M, Joyal SV, Swick AG. Avoiding holiday seasonal weight gain with nutrient-supported intermittent energy restriction: a pilot study. Journal of Nutritional Science. 2019;8:e11. doi: 10.1017/jns.2019.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bommer C, Heesemann E, Sagalova V, Manne-Goehler J, Atun R, Bärnighausen T, Vollmer S. The global economic burden of diabetes in adults aged 20–79 years: a cost-of-illness study. The lancet Diabetes & endocrinology. 2017;5(6):423–430. doi: 10.1016/S2213-8587(17)30097-9. [DOI] [PubMed] [Google Scholar]
- 14.Birdee GS, Yeh G. Complementary and alternative medicine therapies for diabetes: a clinical review. Clinical Diabetes. 2010;28(4):147–155. doi: 10.2337/diaclin.28.4.147. [DOI] [Google Scholar]
- 15.Becker DJ, French B, Morris PB, Silvent E, Gordon RY. Phytosterols, red yeast rice, and lifestyle changes instead of statins: a randomized, double-blinded, placebo-controlled trial. American heart journal. 2013;166(1):187–96. e2. [DOI] [PubMed]
- 16.Cheung C-L, Ho DK-C, Sing C-W, Tsoi M-F, Cheng VK-F, Lee GK-Y, Ho YN, Cheung BMY. Randomized controlled trial of the effect of phytosterols-enriched low-fat milk on lipid profile in Chinese. Sci Rep. 2017;7:41084. doi: 10.1038/srep41084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yoshida M, Vanstone C, Parsons W, Zawistowski J, Jones P. Effect of plant sterols and glucomannan on lipids in individuals with and without type II diabetes. Eur J Clin Nutr. 2006;60(4):529–537. doi: 10.1038/sj.ejcn.1602347. [DOI] [PubMed] [Google Scholar]
- 18.Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC. Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. 2009;6(7):e1000100. doi: 10.1371/journal.pmed.1000100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Higgins JP, Green S. Cochrane handbook for systematic reviews of interventions. 2008.
- 20.Lipsey MW, Wilson DB. Practical meta-analysis: SAGE publications, Inc. 2001. [Google Scholar]
- 21.Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. 2002;21(11):1539–1558. doi: 10.1002/sim.1186. [DOI] [PubMed] [Google Scholar]
- 22.Harris RJ, Deeks JJ, Altman DG, Bradburn MJ, Harbord RM, JAJTSJ S. Metan: fixed-and random-effects meta-analysis. 2008;8(1):3–28. [Google Scholar]
- 23.Li Q, Xing BJDT, therapeutics. A phytosterol-enriched spread improves lipid profile and insulin resistance of women with gestational diabetes mellitus: a randomized, placebo-controlled double-blind clinical trial 2016;18(8):499–504. [DOI] [PubMed]
- 24.Gao F, Wang G, Wang L, Guo NJF, function. Phytosterol nutritional supplement improves pregnancy and neonatal complications of gestational diabetes mellitus in a double-blind and placebo-controlled clinical study 2017;8(1):424–8. [DOI] [PubMed]
- 25.Sialvera T, Pounis G, Koutelidakis A, Richter D, Yfanti G, Kapsokefalou M, et al. Phytosterols supplementation decreases plasma small and dense LDL levels in metabolic syndrome patients on a westernized type diet 2012;22(10):843–8. [DOI] [PubMed]
- 26.Gylling H, Halonen J, Lindholm H, Konttinen J, Simonen P, Nissinen MJ, et al. The effects of plant stanol ester consumption on arterial stiffness and endothelial function in adults: a randomised controlled clinical trial 2013;13(1):50. [DOI] [PMC free article] [PubMed]
- 27.Furuncuoglu Y, Basar M, Alıcı S, Sengul CJEJ. Effects of a stanol-enriched yogurt on plasma cholesterol levels 2014;11(4):230–4.
- 28.Hongu N, Kitts DD, Zawistowski J, Dossett CM, Kopeć A, Pope BT, et al. Pigmented rice bran and plant sterol combination reduces serum lipids in overweight and obese adults 2014;33(3):231–8. [DOI] [PMC free article] [PubMed]
- 29.Cheung C-L, Ho DK-C, Sing C-W, Tsoi M-F, Cheng VK-F, Lee GK-Y, et al. Randomized controlled trial of the effect of phytosterols-enriched low-fat milk on lipid profile in Chinese 2017;7:41084. [DOI] [PMC free article] [PubMed]
- 30.Ferguson JJ, Stojanovski E, MacDonald-Wicks L, Garg MLJM. Curcumin potentiates cholesterol-lowering effects of phytosterols in hypercholesterolaemic individuals. A randomised controlled trial. 2018;82:22–35. doi: 10.1016/j.metabol.2017.12.009. [DOI] [PubMed] [Google Scholar]
- 31.Hautaniemi EJ, Tikkakoski AJ, Tahvanainen A, Nordhausen K, Kähönen M, Mattsson T, et al. Effect of fermented milk product containing lactotripeptides and plant sterol esters on haemodynamics in subjects with the metabolic syndrome–a randomised, double-blind, placebo-controlled study 2015;114(3):376–86. [DOI] [PubMed]
- 32.Chen D-L, Huang P-H, Chiang C-H, Leu H-B, Chen J-W, Lin SJ. Phytosterols increase circulating endothelial progenitor cells and insulin-like growth factor-1 levels in patients with nonalcoholic fatty liver disease: A randomized crossover study. 2015;13:148–57.
- 33.Athyros VG, Kakafika AI, Papageorgiou AA, Tziomalos K, Peletidou A, Vosikis C, et al. Effect of a plant stanol ester-containing spread, placebo spread, or Mediterranean diet on estimated cardiovascular risk and lipid, inflammatory and haemostatic factors 2011;21(3):213–21. [DOI] [PubMed]
- 34.Allen RR, Carson L, Kwik-Uribe C, Evans EM, Erdman J. Daily consumption of a dark chocolate containing flavanols and added sterol esters affects cardiovascular risk factors in a normotensive population with elevated cholesterol 2008;138(4):725–31. [DOI] [PubMed]
- 35.Gomes GB, Zazula AD, Shigueoka LS, Fedato RA, da Costa ABBA, Guarita-Souza LC, et al. A randomized open-label trial to assess the effect of plant sterols associated with ezetimibe in low-density lipoprotein levels in patients with coronary artery disease on statin therapy 2017;20(1):30–6. [DOI] [PubMed]
- 36.Ribas S, Sichieri R, Moreira A, Souza D, Cabral C, Gianinni D, et al. Phytosterol-enriched milk lowers LDL-cholesterol levels in Brazilian children and adolescents: double-blind, cross-over trial 2017;27(11):971–7. [DOI] [PubMed]
- 37.Hallikainen M, Lyyra-Laitinen T, Laitinen T, Moilanen L, Miettinen TA, Gylling HJA. Effects of plant stanol esters on serum cholesterol concentrations, relative markers of cholesterol metabolism and endothelial function in type 1 diabetes. 2008;199(2):432–9. [DOI] [PubMed]
- 38.Ghaedi E, Varkaneh HK, Rahmani J, Mousavi SM, Mohammadi H, Fatahi S, Pantovic A, Darooghegi Mofrad M, Zhang Y. Possible anti-obesity effects of phytosterols and phytostanols supplementation in humans: a systematic review and dose–response meta-analysis of randomized controlled trials. Phytother Res. 2019;33(5):1246–1257. doi: 10.1002/ptr.6319. [DOI] [PubMed] [Google Scholar]
- 39.Ghaedi E, Kord-Varkaneh H, Mohammadi H, Askarpour M, Miraghajani M. Phytosterol supplementation could improve atherogenic and anti-Atherogenic apolipoproteins: a systematic review and dose–response meta-analysis of randomized controlled trials. J Am Coll Nutr. 2019:1–11. [DOI] [PubMed]
- 40.Fatahi S, Kord-Varkaneh H, Talaei S, Mardali F, Rahmani J, Ghaedi E, Tan SC, Shidfar F. Impact of phytosterol supplementation on plasma lipoprotein (a) and free fatty acid (FFA) concentrations: a systematic review and meta-analysis of randomized controlled trials. Nutr Metab Cardiovasc Dis. 2019;29(11):1168–1175. doi: 10.1016/j.numecd.2019.07.011. [DOI] [PubMed] [Google Scholar]
- 41.Wu T, Fu J, Yang Y, Zhang L, Han J. The effects of phytosterols/stanols on blood lipid profiles: a systematic review with meta-analysis. Asia Pac J Clin Nutr. 2009;18(2):179. [PubMed] [Google Scholar]
- 42.Berger A, Jones PJ, Abumweis SS. Plant sterols: factors affecting their efficacy and safety as functional food ingredients. Lipids Health Dis. 2004;3(1):5. doi: 10.1186/1476-511X-3-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Subash-Babu P, Ignacimuthu S, Alshatwi A. Nymphayol increases glucose-stimulated insulin secretion by RIN-5F cells and GLUT4-mediated insulin sensitization in type 2 diabetic rat liver. Chem Biol Interact. 2015;226:72–81. doi: 10.1016/j.cbi.2014.12.011. [DOI] [PubMed] [Google Scholar]
- 44.Patel MD, Thompson PD. Phytosterols and vascular disease. Atherosclerosis. 2006;186(1):12–19. doi: 10.1016/j.atherosclerosis.2005.10.026. [DOI] [PubMed] [Google Scholar]
- 45.Pollak O. Reduction of blood cholesterol in man. Circulation. 1953;7(5):702–706. doi: 10.1161/01.CIR.7.5.702. [DOI] [PubMed] [Google Scholar]
- 46.Best MM, Duncan CH, Van Loon EJ, Wathen JD. Lowering of serum cholesterol by the administration of a plant sterol. Circulation. 1954;10(2):201–206. doi: 10.1161/01.CIR.10.2.201. [DOI] [PubMed] [Google Scholar]
- 47.Drynan L, Patti A, Zammit VA. Flux control exerted by mitochondrial outer membrane carnitine palmitoyltransferase over β-oxidation, ketogenesis and tricarboxylic acid cycle activity in hepatocytes isolated from rats in different metabolic states. Biochem J. 1996;317(3):791–795. doi: 10.1042/bj3170791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hardie DG, Hawley SA. AMP-activated protein kinase: the energy charge hypothesis revisited. Bioessays. 2001;23(12):1112–1119. doi: 10.1002/bies.10009. [DOI] [PubMed] [Google Scholar]
- 49.Merrill G, Kurth E, Hardie D, Winder W. AICA riboside increases AMP-activated protein kinase, fatty acid oxidation, and glucose uptake in rat muscle. American Journal of Physiology-Endocrinology And Metabolism. 1997;273(6):E1107–E1E12. doi: 10.1152/ajpendo.1997.273.6.E1107. [DOI] [PubMed] [Google Scholar]
- 50.Kurth-Kraczek EJ, Hirshman MF, Goodyear LJ, Winder WW. 5'AMP-activated protein kinase activation causes GLUT4 translocation in skeletal muscle. Diabetes. 1999;48(8):1667–1671. doi: 10.2337/diabetes.48.8.1667. [DOI] [PubMed] [Google Scholar]
- 51.Collins M, Varady KA, Jones PJ. Modulation of apolipoprotein A1 and B, adiponectin, ghrelin, and growth hormone concentrations by plant sterols and exercise in previously sedentary humans. Can J Physiol Pharmacol. 2007;85(9):903–910. doi: 10.1139/Y07-078. [DOI] [PubMed] [Google Scholar]
- 52.Carling D. The AMP-activated protein kinase cascade–a unifying system for energy control. Trends Biochem Sci. 2004;29(1):18–24. doi: 10.1016/j.tibs.2003.11.005. [DOI] [PubMed] [Google Scholar]
- 53.Singh A, Hussain S, Najmi AK. Number of studies, heterogeneity, generalisability, and the choice of method for meta-analysis. J Neurol Sci. 2017;381:347. doi: 10.1016/j.jns.2017.09.026. [DOI] [PubMed] [Google Scholar]
- 54.Fletcher J. What is heterogeneity and is it important? Bmj. 2007;334(7584):94–96. doi: 10.1136/bmj.39057.406644.68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Li Y-C, Li C-L, Li R, Chen Y, Zhang M, Guo P-P, et al. Associations of dietary phytosterols with blood lipid profiles and prevalence of obesity in Chinese adults, a cross-sectional study. Lipids in health and disease. 2018;17(1):54-. [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX 31 kb)