Abstract
Vasovagal syncope (VVS) and postural tachycardia syndrome (POTS) are the main forms of orthostatic intolerance in pediatrics and both are underlying causes of neurally-mediated syncope. In recent years, increasing attention has been paid to the management of VVS and POTS in children and adolescents. A number of potential mechanisms are involved in their pathophysiology, but the leading cause of symptoms varies among patients. A few studies thus have focused on the individualized treatment of VVS or POTS based on selected hemodynamic parameters or biomarkers that can predict the therapeutic effect of certain therapies and improve their effectiveness. This review summarizes the latest developments in individualized treatment of VVS and POTS in children and indicates directions for further research in this field.
Keywords: Vasovagal syncope, Postural tachycardia syndrome, Individualized management, Pathophysiology, Children, Adolescents
Introduction
Orthostatic intolerance (OI) is common in the pediatric population and is defined as having difficulties in tolerating the upright posture because of several symptoms (e.g., lightheadedness, blurred vision, palpitations, tremor, fatigue, and even syncope) that are relieved by recumbency [1]. Vasovagal syncope (VVS) and postural tachycardia syndrome (POTS) are the main forms of OI in pediatric patients [1]. VVS is usually triggered by prolonged standing (acute OI) or exposure to mental stress and is manifested as sudden syncopal episodes accompanied by pallor, nausea, diaphoresis, and other symptoms associated with hypotension and/or bradycardia resulting from vagal excitation [2]. A diagnosis of VVS is made mainly based on the typical manifestations noted above, but sometimes a provocative test, the head-up tilt test (HUTT), should be performed to confirm the diagnosis, especially when the patient has atypical signs. A positive response in the HUTT that supports a diagnosis of VVS is that the patient suddenly faints or has pre-syncopal signs during the course of tilting combined with either hypotension (vasoinhibitory type) or bradycardia (cardioinhibitory type), or both (mixed type), and the cutoff values for hypotension and bradycardia vary with age [2]. POTS is a form of chronic OI characterized by an excessive increase in heart rate when moving from the supine to the upright position [2, 3]. The diagnostic criterion for “excessive increase of heart rate” in pediatric patients with POTS refers to an increase in heart rate ≥40 beats per min (bpm) from the supine to the upright position or the maximum upright heart rate reaches ≥130 bpm (in children aged 6 to 12 years) or ≥125 bpm (in adolescents aged 13 to 18 years) within the initial 10 min after standing up in the HUTT or standing test [4]. Both VVS and POTS are underlying causes of neurally-mediated syncope, which is defined as syncope associated with autonomic neural reflexes or autonomic nerve dysfunction and accounts for ~75% of pediatric syncope cases [2, 4]. The recurrent or sustained symptoms of OI, either frequent syncopal episodes or chronic systemic symptoms, may have negative effects on children’s physical and mental health and significantly lower the quality of the children’s and parents’ lives [1]. In recent years, increasing attention has been paid to the management of VVS and POTS in children and adolescents. The Heart Rhythm Society [4], Canadian Cardiovascular Society and Canadian Pediatric Cardiology Association [5], American College of Cardiology/American Heart Association (ACC/AHA) [2], European Society of Cardiology (ESC) [6], and Chinese Pediatric Cardiology Society (CPCS) [3] have successively published guidelines or expert consensus statements on the diagnosis and management of pediatric VVS or POTS. In addition, a number of studies have focused on the individualized treatment of VVS or POTS based on selected hemodynamic parameters or biomarkers that can predict the therapeutic effects of certain therapies to improve their effectiveness. In the following, we review the latest developments in individualized treatment for VVS and POTS in children for the purpose of improving the therapeutic strategy in further clinical studies in this field.
Pathophysiology and Individualized Management of VVS in Pediatric Patients
Pathophysiology and Theoretical Basis of Individualized Therapy for VVS
VVS is the most common form of reflex syncope, comprising postural syncope and emotional/stress syncope in response to various triggers. Currently, there are continuing disputes about the pathophysiology of VVS, indicating that the dominant mechanisms causing syncope vary among VVS patients.
When upright, it is a challenge for humans to maintain sufficient cerebral blood flow (CBF) because the heart is lower than the brain and 70% of the blood flow is below heart level. Adequate blood volume and the integrity of cardiovascular and autonomic compensatory mechanisms are essential for tolerance of orthostatic stress [7]. Any impairment in the orthostatic compensatory system can lead to decreased CBF and symptoms of OI. To a certain extent, OI is a “cost” of bipedalism.
Early researchers believed that the key pathological mechanism of VVS was the Bezold-Jarisch reflex, which is induced by an under-filled and hyper-contractile heart [8]. However, increasingly opposing opinions have emerged to challenge this classic theory [9, 10]. According to recent categorizations, the postural VVS patient typically experiences three stages during a syncopal episode based on the hemodynamic data with a positive response during the HUTT (Fig. 1).
Fig. 1.
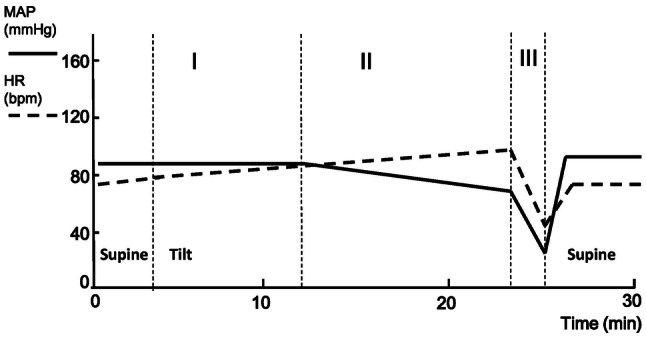
Illustration of the three stages of hemodynamic change in pediatric patients with classic postural vasovagal syncope (VVS) during the head-up tilt test (HUTT). Typical changes in mean arterial pressure (MAP; solid line) and heart rate (HR; dashed line) during the HUTT (vertical dashed lines, different stages during the test until a positive response). MAP is maintained relatively stable accompanied by increased HR in stage I. Then MAP decreases slowly while HR increases continuously in stage II. Both MAP and HR fall abruptly in stage III, indicating a positive response of the mixed type. The MAP and HR rise to normal levels soon after the patient returns to the supine position. bpm, beats per minute.
In stage I, the blood pressure (BP) is relatively stable. After standing up, a transient fall of BP called initial orthostatic hypotension occurs because of the decreased intrathoracic blood volume due to gravity, and the BP can recover in 30–60 s by reflex tachycardia. This initial fall of BP occurs in both healthy people and VVS patients. Then, the BP remains stable by means of increased reflex sympathetic activity and compensatory vasoconstriction, although the sympathetic activity, heart rate, and peripheral vascular resistance increase more in VVS patients than in healthy people. Patients with a relatively insufficient central volume have to exert more effort to compensate for the excessive reduction of inevitable cardiac preload. Patients may feel well or at most experience transient and slight dizziness as well as palpitation at this stage because of the relatively stable BP and mild tachycardia. During this stage, efforts to increase venous return, such as physical counter-pressure maneuvers and external compression on the lower limbs, can be helpful.
In stage II, the BP slowly decreases despite the more obvious tachycardia combined with continuously increasing sympathetic activity and peripheral vascular resistance, leading to the symptoms of pre-syncope. Patients begin to show prodromal manifestations such as weakness, pallor, blurred vision, severe lightheadedness, nausea, and abdominal pain due to the insufficient blood supply. Sweating may also appear as a result of sympathetic activation. The gradual decline of BP reflects the decreased cardiac output, which is believed to be related to the reduction of cardiac preload, vasodilation in selected peripheral vascular beds, and excessive tachycardia in this stage [11, 12]. The inappropriate vasodilation in specific peripheral vascular beds has been shown to play an important role at this stage [1, 13]. However, peripheral vasodilation is not evident in all VVS patients, and the extent of tachycardia can be mild to moderate in some patients, indicating that the predominant mechanism of the reduction in cardiac output differs among individuals.
In stage III, the CBF suddenly falls, combined with a sharply decreased BP and/or heart rate, leading to a loss of consciousness. A syncopal attack finally occurs at this stage because of a significant decrease in CBF. These abrupt hemodynamic changes have been attributed to the impairment of baroreflex integrity and cerebral autoregulation [14]. However, the exact mechanism is still unknown.
Not every patient experiences the three typical stages, implying that each patient has distinct major problems with the orthostatic regulatory mechanisms. For example, hypotension and bradycardia do not always occur simultaneously at stage III. Some patients exhibit only a decrease in BP without detectable bradycardia during the HUTT and are considered to have the vasoinhibitory type of VVS; in these patients, a deficiency in peripheral vasoconstriction is thought to be the main problem. In contrast, other patients experience convulsive syncope due to severe bradycardia or sudden asystole without the warning decline in BP (lacking stage II) and are classified as having the cardioinhibitory type of VVS [4]. In addition, in a recent study, VVS patients were divided into two groups according to their baseline supine BP. During the tilt test, low tyrosine hydroxylase levels and reduced norepinephrine (NE) synthesis were found in the low-pressure group, while increased NE transporter levels resulting in enhanced NE reuptake were found in the normal-pressure group [15]. Despite these variations among VVS patients, it is evident that the principles of management should be determined for each patient.
In summary, several potential mechanisms are involved in the pathophysiology of VVS, but the leading cause of symptoms in each patient varies. Any of the mechanisms, such as relatively insufficient central volume leading to an excessive reduction in cardiac preload, inappropriate peripheral vasodilation, abnormal NE metabolism, and a loss of baroreflex integrity, can play the major role and become the therapeutic target in different individuals. Although it is not fully understood why patients with VVS demonstrate the vasoinhibitory type, cardioinhibitory type, or mixed type, and why sometimes one type converts into another, there is a consensus that therapies against these pathophysiological mechanisms may abolish the abnormal orthostatic regulatory process at the first two stages to avoid syncope at stage III. Therefore, in theory, individualized therapy directed against the predominant pathophysiological mechanism can be expected to abort the subsequent symptoms and achieve a more satisfactory therapeutic effect.
Improvement of Individualized Treatment of VVS in Children and Adolescents
The management of VVS in children includes conventional therapy, pharmacological therapy, and cardiac pacing. According to previous studies, conventional therapy usually involves tilt training, physical counter-pressure maneuvers, and increased intake of salt and water, while pharmacological therapy basically involves fludrocortisone, β-adrenergic receptor blockers, α-adrenergic receptor agonists, and serotonin transporter inhibitors [3]. These treatment options for VVS in children are based on the theories described above. For example, increased salt and water intake and fludrocortisone are recommended to raise the central volume, while midodrine (an α1-adrenergic receptor agonist) is prescribed against probable peripheral vasodilation. For treatment of the cardioinhibitory type of VVS, cardiac pacing should be cautiously considered for patients suffering from recurrent syncope related to reflex vagal asystole [2]. The disappearance of the recurrent syncope during follow-up as well as a negative response in the HUTT at the endpoint of follow-up are usually taken as effective responses. However, there are always non-responders to each therapy. Without individualized therapy, only 20%–29% of children with VVS benefit from conventional therapy, including health education and an increased intake of salt and water [16, 17]. Even with medication, only 43% of pediatric patients with VVS responded to metoprolol and 75% responded to midodrine, in two randomized controlled trials (RCTs) [16, 17]. The major problems in management strategies for VVS may be that the therapeutic options do not specifically target the mechanisms in patients with different primary pathogeneses, and there are no indicators for the various mechanisms to facilitate selection. Therefore, it is necessary to find predictors of the therapeutic effect in VVS patients that indicate the dominant pathophysiological mechanism in each patient, to identify individuals who will respond to the corresponding therapy and consequently improve the quality of life for VVS patients who are affected by recurrent syncope.
Acceleration Index as a Predictor of the Effectiveness of Orthostatic Training in Treating Children with VVS
Orthostatic training (also known as tilt training) is a protocol to practice standing for gradually prolonged durations. Children with VVS are usually instructed to stand 15 cm away from the foot of a wall with their upper body leaning against the wall for no more than 20 min [4]. This training is considered useful for improving the coordination of the autonomic and cardiovascular systems during orthostatic regulation and, in theory, to adapt the patient to the upright posture. One RCT in adults showed that well-controlled orthostatic training can significantly enhance baroreflex sensitivity and heart rate variability [18]. However, the symptom improvement rate was not satisfactory in earlier studies, and corresponding data in pediatric patients are limited [19, 20]. The acceleration index is the instantaneous rate of increase of a patient’s heart rate when they change from a supine to an upright position and is calculated from the intervals between adjacent R waves (RR intervals) in electrocardiograms during the process. In detail, the acceleration index is calculated as [(A − B)/A]×100, where A is the mean duration (in milliseconds) of the RR intervals during the last 15 s before changing to an upright position and B is the shortest RR interval within the period of 5 to 40 s after standing. This index has been positively correlated with plasma epinephrine levels and represents sympathetic activity [21]. One recent study revealed that VVS children with a lower baseline acceleration index have a better response to orthostatic training, suggesting that this index may predict the effectiveness of orthostatic training in this population. The index has a cutoff value of 26.77 with a sensitivity of 85% in predicting the therapeutic response [22], it is easy to measure, and it may become a useful indicator to improve the therapeutic efficacy of orthostatic training for children with VVS.
Predictors of the Effectiveness of β-Adrenergic Receptor Blockers in Treating Children with VVS
The use of β-adrenergic receptor blockers (β-blockers) as the treatment for patients with VVS has been disputed for years. The supposed therapeutic rationale for these agents is that β-blockers suppress the activation of ventricular mechanoreceptors by attenuating the excessive contractility of the heart, which is deemed to initiate the classic Bezold-Jarisch reflex, leading to vagal excitation in patients with VVS [23, 24]. Nevertheless, as noted above, the role of the Bezold-Jarisch reflex in the pathophysiology of VVS is debatable, and several randomized comparisons of β-blockers and placebo in the treatment of VVS in adults have not confirmed the superiority of the former [25–27]. Studies of VVS in the pediatric population also report conflicting results on the efficacy of β-blockers [16, 28, 29]. It is notable that a number of studies have revealed that β-blockers are beneficial in certain age groups [16, 28, 30]. These different results indicate that β-blockers may be effective for selected patients with VVS and that predictors of the responses to β-blockers are needed to rationalize their use.
Increase in Heart Rate before a Positive Response in the Head-Up Tilt Test
To identify predictors of the therapeutic efficacy of metoprolol (a β1-selective blocker) in treating children with VVS, Zhang et al. [31] focused on the hemodynamic features during the HUTT. In this process, reflex tachycardia occurs after a patient is tilted from the supine position. The investigators found that, during the period before a positive response in the HUTT, children with a greater increase in heart rate in the tilted position than in the supine position show a better therapeutic response to metoprolol during follow-up. Using a heart rate increase of 30 bpm in the HUTT as the threshold, the sensitivity of prediction of an effective response to metoprolol among children with VVS is 81%, indicating that the change in heart rate during the HUTT can be an ideal predictor.
Echocardiography-Derived Left Ventricular Ejection Fraction and Fractional Shortening
Previous studies have revealed that the plasma epinephrine levels in the supine position are mildly higher in patients with VVS than in the healthy population, whereas the plasma epinephrine increases significantly after the patient is tilted, and further increases to pre-syncope or syncope during the HUTT [32, 33]. The increased epinephrine is probably associated with excessive ventricular constriction that may evoke reflex hypotension and bradycardia in some patients with VVS, which rationalizes the use of β-blockers. Song et al. [34] explored some non-invasive indicators of standing baseline epinephrine to predict the therapeutic response to metoprolol and discovered that the left ventricular ejection fraction (LVEF) and fractional shortening (LVFS) measured by echocardiogram in the supine position can achieve this goal. During the 6-month follow-up, the responders to metoprolol had higher baseline LVEF and LVFS than the non-responders (LVEF: 72.5% ± 3.2% vs 64.6% ± 3.4%; LVFS: 40.9% ± 2.3% vs 34.9% ± 2.9%). With a cutoff value of 70.5%, the sensitivity of LVEF to predict a therapeutic response to metoprolol in children with VVS was 81.3% as determined by the receiver operating characteristic (ROC) curve analysis. The predictive sensitivity of LVFS reached 93.8% when using a borderline cutoff of 37.5%. However, the use of LVEF as a predictor for a therapeutic response to metoprolol is limited as the cut-off baseline LVEF value of >70.5% may exclude major populations of patients.
Twenty-Four-Hour Urine NE Level
Recently, Kong et al. investigated the 24-h urine NE level, which represents the functional status of the sympathetic nervous system to some extent, in children with VVS [35]. They found that the levels varied within the patient group and were positively correlated with the supine BP, implying that the baseline sympathetic activity differs among children with VVS. This result is consistent with the findings of Vaddadi et al. who reported that patients with VVS can be divided into normal-pressure and low-pressure groups according to their supine BP [15]. As expected, children with a better response to metoprolol had higher 24-h urine NE levels before treatment. With 34.84 µg/24 h as the cutoff value, the 24-h urine NE level can be used to predict the therapeutic effect of metoprolol therapy in children with VVS with a sensitivity of 70% and a specificity of 100%.
Flow-Mediated Vasodilation (FMD) as a Predictor of the Effectiveness of α1-Adrenergic Receptor Agonists in Treating Children with VVS
Excessive vasodilatation is known to be a vital pathophysiological mechanism underlying the reduced cardiac output in a group of patients with VVS [10, 11]. In RCTs, midodrine hydrochloride, an α1-adrenergic receptor agonist, has been shown to be effective in treating children with VVS [17, 36]. The effective rate of midodrine therapy is ~75% in non-selected children with VVS. In a study to search for an indicator of vascular tone to predict the therapeutic effect of vasoconstrictors such as midodrine, Zhang et al. measured FMD in pediatric patients with VVS before they received midodrine [37]. The FMD detected by vascular ultrasound represents endothelium-dependent vascular function. During follow-up for 10–18 months, the children with greater baseline FMD had a lower recurrence rate of syncope. The ROC for the predictive value of FMD showed a high sensitivity (90%) for a therapeutic response to midodrine when using an FMD of 8.85% as the cutoff. Thus, they concluded that FMD can be taken as an indicator for the selection of midodrine for the treatment of VVS in children.
Individualized Treatment with Cardiac Pacing in Children with VVS
Children diagnosed with the cardioinhibitory or mixed type of VVS may experience sudden asystole during episodes of syncope. Nevertheless, it is debatable to treat these children with cardiac pacing against the asystole triggered by the vagal reflex because they often have a benign prognosis. According to recent RCTs and meta-analyses, pacemaker therapy is recommended (Class IIa) by the guidelines from the ACC/AHA and ESC for patients aged ≥40 years with recurrent syncope and documented asystole ≥3 s related to syncope or spontaneous asystole ≥6 s without syncope recorded by an implantable loop recorder [2, 6, 38]. In pediatric patients, there is no strong evidence for the necessity of cardiac pacing in the management of VVS. A retrospective study investigated pacemaker implantation in 11 children 0.8–17 years of age who were diagnosed with cardioinhibitory VVS or pallid breath-holding spells (a specific type of VVS in infants) and in whom syncope was correlated with a documented asystole ≥10 s [39]. The results showed that implantation completely prevented the recurrence of syncope in 10 out of the 11 children during follow-up (median, 6.8 years) [39]. The ACC/AHA guideline states that pacemaker therapy might be considered in children with severe reflex syncope secondary to pallid breath-holding spells (Class IIb) [2]. According to the CPCS guidelines, cardiac pacing should be considered in pediatric VVS patients with a symptomatic prolonged asystolic pause ≥4 s on the advice of experienced pediatric cardiologists [4]. In brief, pacemaker therapy should be cautiously but not regularly considered in the management of VVS children with threatening asystole. The benefits and adverse complications of pacemaker implantation should be weighed before making a decision.
Pathophysiology and Individualized Management of Postural Tachycardia Syndrome in Pediatric Patients
Pathophysiology and Theoretical Basis for Individualized Therapy in POTS
Different from VVS, POTS is a form of chronic OI with heterogeneous etiologies. The identical major sign in patients with POTS is the sustained excessive tachycardia as a compensatory response to orthostatic posture, although their symptoms are diverse and can appear in any posture [40] (Fig. 2). Several mechanisms have been reported for patients with POTS, such as central hypovolemia, peripheral vascular dysfunction, and hyperadrenergic stimulation [1, 3]. There may be more than one mechanism in an individual patient with POTS; thus, it is necessary to reveal these mechanisms in order to prescribe a sensible therapeutic regimen.
Fig. 2.
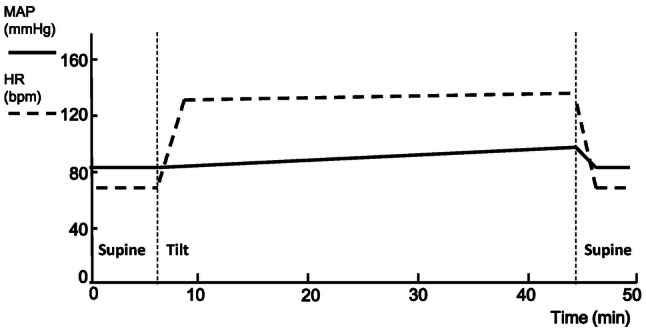
Illustration of typical hemodynamic changes in pediatric patients with POTS during the HUTT (solid line, MAP; dashed line, HR; vertical dashed lines, beginning and end of tilting). HR significantly increases while MAP increases slightly and remains relatively stable during the entire course. Both of MAP and HR fall to normal levels soon after the patient returns to the supine position. MAP, mean arterial pressure; HR, heart rate; POTS, postural tachycardia syndrome; HUTT, head-up tilt test; bpm, beats per minute.
Hypovolemia
Nearly 70% of patients with POTS have a reduced blood volume [3]. It is apparent that a relatively insufficient blood volume can worsen central hypovolemia when patients are in a long-term upright position, leading to excessive tachycardia as compensation. In some patients, hypovolemia is secondary to specific underlying causes, such as dehydration and anemia. However, in most cases, patients with POTS do not have a definite history of dehydration despite the low blood volume [41, 42]. Raj et al. reported that a group of patients with POTS had paradoxically unchanged plasma renin activity and lower aldosterone compared with healthy controls regardless of the significant reduction in total blood volume [43], suggesting that disorders of the renin-aldosterone system contribute to the hypovolemia in some patients with POTS.
Peripheral Vascular Dysfunction
Dysfunction of the peripheral vascular system can cause a failure of vasoconstriction or excessive vasodilation in certain vascular beds, such as the lower limbs or splanchnic vasculature, resulting in blood volume pooling and distributive central hypovolemia in some patients with POTS, especially when in an upright position [44, 45]. Central hypovolemia inevitably leads to reduced cardiac output and compensatory orthostatic tachycardia.
Peripheral Autonomic Denervation
Peripheral autonomic denervation is one of the acknowledged causes of vascular dysfunction. Impaired adrenergic vasoconstriction in the lower limbs and splanchnic vascular bed has been found in patients with POTS [13, 46, 47], who were classified as having “neuropathic POTS”. The exact etiology of this autonomic neuropathy is not clear, although studies have indicated that autoimmune processes might contribute to the pathological mechanisms [48–50].
Imbalance Between Vasoconstrictors and Vasodilators
An imbalance between the vasoconstrictors and vasodilators in the peripheral circulation may be another important factor contributing to vascular dysfunction. Several vasodilators, such as nitric oxide (NO) [51], hydrogen sulfide (H2S) [52], and adrenomedullin (ADM) [53], are higher, whereas vasoconstrictors such as endothelin [54] and urotensin II [55] are lower in patients with POTS than in healthy controls. These changes in vasoactive factors may participate in the abnormal vascular tone in patients with POTS.
Structural Defects of Peripheral Blood Vessels
Rarely, structural defects of peripheral blood vessels can also predispose individuals to vascular dysfunction. The prevalence of Ehlers-Danlos syndrome, which is a heterogeneous group of heritable connective tissue disorders, is significantly higher in patients with POTS than in the general population [56]. The possible mechanism underlying this syndrome is that the vascular wall of peripheral veins is excessively stretched in response to the increased pressure caused by orthostatic posture, resulting in venous pooling [56]. By a similar mechanism, the symptoms of POTS can be seen in patients with a congenital absence of valves in the deep veins of the legs [57].
Hyperadrenergic Stimulation
While standing, high levels of circulating NE as well as increased sympathetic nerve activity have been reported in a group of patients with POTS, who are classified as having “hyperadrenergic POTS” [50, 58, 59]. These patients usually have manifestations such as pallor, tremor, palpitations, tachycardia, and hypertension, suggesting sympathetic activation. In some patients, the increased NE level is considered to be associated with autoantibodies to adrenergic receptors [60]. In addition, a mutation of the SLC6A2 gene encoding the NE transporter has been found in some patients showing significant sympathetic activation because of the decreased reuptake of synaptic NE [61].
Other Mechanisms
Apart from the classic mechanisms described above, several other functional disorders or diseases are thought to be involved in the pathophysiology of POTS.
Mechanisms Associated with Autoimmunity
A variety of phenomena support the hypothesis that the autoimmune response participates in the etiology of POTS. Patients with POTS may have experienced a viral infection, vaccination, operation or trauma before the onset of symptoms [62]. Moreover, some autoantibodies, such as those against adrenergic receptors, angiotensin II type I receptors, and ganglionic acetylcholine receptors, have been detected in the plasma of patients with POTS, of whom 20% have a history of autoimmune disease [49, 63–66]. Researchers have studied the roles of these autoantibodies in patients with POTS. Fedorowski et al. found that the autoantibodies from some POTS patients can activate α1- or β1/2-adrenergic receptors in transfected cells, and this activation can be suppressed by specific blockers of the corresponding receptors [64]. Furthermore, the upright heart rate of patients with POTS is correlated with an activated response of adrenergic receptors mediated by autoantibodies. On the basis of studies of plasma autoantibodies to adrenergic receptors, the orthostatic tachycardia in patients with POTS is probably attributable to the direct stimulatory effect of cardiac receptor-activating autoimmune antibodies on sinus rhythm control or to a compensatory response to the excessive vasodilation elicited by vascular receptor autoantibodies [60, 66].
Prolonged Bed Rest and Deconditioning
Some patients with POTS tend to stay in bed because of the symptoms of OI. However, gravitational deconditioning can appear with prolonged bed rest, leading to decreased blood volume, abnormal redistribution of blood volume, and failure of vasoconstriction as well as dysfunction of the skeletal muscle pump, which can aggravate the symptoms of OI [67, 68]. The prevalence of deconditioning in adult and adolescent patients with POTS is reported to be up to 93% and 68%, respectively [69, 70]. As a result, it is believed that the symptoms of POTS can be alleviated by physical activity rather than bed rest.
Improvement of Individualized Treatment of POTS in Children and Adolescents
Multiple mechanisms lead to the heterogeneity of POTS and pose a great challenge to management because the patients cannot be treated by a unified method. In addition to common measures such as education for patients and their parents, the advocacy of physical exercise, and psychological support, targeted therapy focusing on the major underlying pathological mechanisms can predictably achieve better therapeutic effects. For example, to attain the best therapeutic outcome, advocating salt and water intake is suitable for patients with hypovolemia; vasoconstrictors tend to be prescribed for patients with vascular dysfunction; and adrenergic receptor antagonists should be used for patients with hyperadrenergic stimulation. Furthermore, the most important thing for successful individualized therapy is to search for appropriate clinical signs or biomarkers that can indicate the major pathological mechanisms to predict the therapeutic effects of different treatments.
Predictors of the Effectiveness of Oral Rehydration Salts in the Treatment of Children with POTS
Early studies confirmed that increased fluid and salt intake can improve the symptoms of OI by increasing blood volume, although the extent of improvement varies [71, 72]. Oral rehydration salts (ORSs) are the most commonly used agents in children with POTS. Several markers reflecting hypovolemia have been reported to elevate the pertinence of the use of ORSs.
Twenty-Four-Hour Urinary Sodium Concentration
The loading of sodium in body fluids is known to be positively correlated with blood volume. Zhang et al. [73] aimed to explore the relationship between 24-h urinary sodium and ORS therapy in children with POTS. The ORS therapy resulted in a significant increase in urinary sodium excretion and responders to ORSs, whose OI symptom score was decreased by at least 2 after treatment with ORSs, had a lower baseline 24-h urinary sodium concentration than non-responders. The suggested condition for the prediction of response to ORS therapy is a 24-h urinary sodium concentration <124 mmol/L with a high predictive sensitivity of 93%. These results support the speculation that a low 24-h urinary sodium concentration, which is believed to be associated with decreased sodium loading as well as body fluid volume, can be taken as an indicator for using ORSs as a treatment option.
Body Mass Index
Another candidate predictor for the effectiveness of ORS therapy is body mass index (BMI), which is calculated from body height and weight. Stewart et al. revealed that BMI was correlated with blood flow in a group of young women with POTS [74]. Li et al. [75] reported in a pediatric population that children with POTS had a lower BMI than the controls in the same age group. These findings suggest that BMI reflects blood volume to a certain extent. Furthermore, this study found that pediatric POTS patients with a lower BMI respond better to ORS therapy than those with a higher BMI. Using a BMI <18 kg/m2 as the cutoff, the effectiveness of ORSs in the treatment of children with POTS is well predicted with a sensitivity up to 92% and specificity up to 82.8% [75]. It is easy to measure body height and weight. The results also implied a potential relationship between BMI and blood volume in accordance with previous studies. However, BMI can be influenced by many other factors in addition to blood volume, especially in children.
Mean Corpuscular Hemoglobin Concentration
The red blood cell (RBC) volume is correlated with blood volume. A previous study showed that RBC volume was significantly reduced in a group of patients with POTS [76]. Lu et al. [77] explored the items that reflect RBC volume included in a complete blood count in children with POTS to search for predictors of the therapeutic effect of ORS treatment. According to their findings, responders to ORSs had a lower mean corpuscular volume and a higher mean corpuscular hemoglobin concentration (MCHC) than non-responders, while other items such as hematocrit did not differ between the two groups. The MCHC also showed a moderate predictive value of a therapeutic effect of ORSs on POTS in children by ROC analysis, and the recommended borderline for predicting responders was an MCHC >347.5 g/L. To illuminate the potential reason that the responders to ORSs have an increased MCHC, the investigators explained that low blood volume may contribute to either insufficient plasma volume or reduced RBC volume or both, and ORSs directly replenish the plasma volume rather than the RBC volume. Therefore, patients with an increased MCHC are likely to have an insufficient plasma volume and benefit from ORS therapy. The complete blood count can be tested in hospitals at different levels, but further evaluation is needed because the sensitivity and specificity of prediction is just about 65%.
Baroreflex Sensitivity
The baroreflex plays an important role in the autonomic regulation of BP, and baroreflex sensitivity (BRS) reflects the functional status of the autonomic nervous system. Convertino et al. reported that decreased blood volume results in an enhanced BRS, implying an association between BRS and blood volume [78]. Li et al. [79] designed a study to determine the role of BRS in the prediction of the therapeutic effect of ORS in children with POTS. The BRS was measured supine using a continuous hemodynamic monitoring system. Children in the POTS group showed much greater BRS than the control group, and BRS was positively correlated with the change in heart rate from the supine to the upright posture in the POTS group. In addition, responders to ORS therapy had greater baseline BRS than non-responders. Both high predictive sensitivity (85.7%) and specificity (87.5%) can be achieved when a BRS >17.01 ms/mmHg is taken as a determinant of ORS therapeutic efficacy in pediatric POTS patients. As expected, an increased BRS was found in responders to ORSs who were supposed to have an insufficient blood volume with enhanced BRS. The BRS seems to be an ideal predictor in terms of the high predictive sensitivity and specificity, although the measurement is a little difficult for general use.
Predictors of the Effectiveness of β-Adrenergic Receptor Blockers in the Treatment of Children with POTS
β-Blockers are prescribed to relieve symptoms of POTS in children based on the mechanism of hyperadrenergic stimulation and have been shown to be effective in children and adults with POTS [80, 81]. However, one controlled study on children with POTS reported that the effective rate of metoprolol therapy is only 57.9% [82]. Therefore, indicators of hyperadrenergic status that can be easily detected are needed to improve the therapeutic effect of β-blockers.
Orthostatic Plasma NE Level
To test the “hyperadrenergic stimulation” hypothesis, Zhang et al. [83] examined the orthostatic plasma NE level in children with POTS. The blood samples were collected after the patients remained standing for 5 min. The orthostatic plasma NE levels were positively correlated with the symptom score as well as the increase in heart rate during the HUTT. After treatment with metoprolol for 3 months, the responders who showed significant improvement in symptoms and upright heart rate had higher baseline orthostatic plasma NE levels than the non-responders. Based on the ROC analysis, the authors recommend an orthostatic plasma NE level >3.59 pg/mL as a reference for the selection of metoprolol as therapy for children with POTS. The predictive sensitivity is 77% and the specificity is 92%. As β-blockers can block the effect of increased NE levels, these results may be direct evidence for individualized use of metoprolol; however, it should be noted that the sample size was small.
Plasma C-Type Natriuretic Peptide
C-type natriuretic peptide (CNP) is a small bioactive peptide that is involved in the pathogenesis of several cardiovascular diseases; it may be an ideal biomarker because it is stable in the circulation and is easily detected [84]. Takekoshi et al. reported that CNP increases the synthesis of catecholamines by promoting the expression of tyrosine hydroxylase mRNA in vitro [85], while in isolated heart experiments Springer et al. reported that CNP increases heart rate by acting on the sinoatrial node through its receptors [86]. Based on these experimental findings, Lin et al. [87] assessed the plasma CNP in children diagnosed with POTS and explored its value in the prediction of a therapeutic effect of β-blockers. The plasma CNP levels were markedly higher in children with POTS than in healthy children, indicating a role of CNP in the pathogenesis of POTS. During follow-up, the researchers found that the responders to metoprolol had higher baseline plasma CNP levels than the non-responders. The recommended cutoff value of plasma CNP is 32.55 pg/mL to predict the efficacy of metoprolol in the treatment of children diagnosed with POTS, with high sensitivity (96%) and moderate specificity (70%). Based on this study, the increased plasma CNP levels in responders to metoprolol are in accordance with the results of previous studies showing that CNP promotes the synthesis of catecholamines. Therefore, CNP can be taken as an indicator of “hyperadrenergic POTS”.
Plasma Copeptin
Copeptin is a glycopeptide derived from the cleavage of the precursor of arginine vasopressin (AVP), which is sensitive to changes in osmotic pressure and blood volume but is unstable in the circulation [88]. Copeptin is equally released with AVP and is stable for detection as an ideal biomarker of AVP secretion. Franklin et al. demonstrated that copeptin is inhibited by high central catecholamine levels [89]. Zhao et al. [90] measured plasma copeptin in children with POTS and analyzed its value in the prediction of a therapeutic effect of metoprolol. The responders to metoprolol were found to have lower baseline plasma copeptin levels than the non-responders. A plasma copeptin level <10.2 pmol/L ensures a satisfactory predictive sensitivity reaching 90% and a specificity of 79%. This study revealed that reduced plasma copeptin levels are associated with an improved therapeutic effect of metoprolol, indirectly implying that plasma copeptin may have a negative correlation with catecholamines, although further studies are needed to test this hypothesis.
Predictors of the Effectiveness of α-Adrenergic Receptor Agonists in the Treatment of Children with POTS
Peripheral vascular tone plays a vital role in the maintenance of hemodynamic stability as well as sufficient cerebral blood flow in an orthostatic posture [1]. As noted above, although several mechanisms can account for the vascular dysfunction in the pathogenesis of POTS, the failure of vasoconstriction or excessive vasodilation, resulting in pooling in the peripheral circulation, is almost uniformly the direct cause of decreased central volume [91]. Based on this rationale, vasoconstrictors such as α-adrenergic receptor agonists are used to treat patients with POTS. Previous studies have demonstrated the efficacy of midodrine in treating children and adolescents with POTS [80, 82, 92], and a number of indicators of vascular dysfunction have been found to help make the decision to use midodrine [93].
Erythrocytic Hydrogen Sulfide Production
H2S has been confirmed as the third gaseous signaling molecule after NO and CO. H2S has significant regulatory effects on the cardiovascular system and is recognized as a strong vasodilator through several different pathways [94, 95]. The role of H2S in the prediction of the therapeutic response to midodrine in children with POTS was investigated by Yang and colleagues [52]. They revealed that children in the POTS group had higher erythrocytic H2S production than controls. Furthermore, after treatment with midodrine for 3 months, the OI symptom score declined in the POTS group, and the decreasing score was positively correlated with the original erythrocytic H2S production. Responders to midodrine had greater baseline erythrocytic H2S production than non-responders. The sensitivity and specificity for prediction are 79% and 78%, respectively, under the condition of an erythrocytic H2S production >27.1 nmol/min/108 RBCs. They concluded that children with POTS can be treated with midodrine in reference to this cutoff value. It seems that elevated erythrocytic H2S production can predict the therapeutic response to midodrine, a vasoconstrictor, basically because of the vasodilatory effect of H2S on various peripheral vascular beds. However, the exact mechanisms by which this α1-agonist acts on the vasoactive effects mediated by H2S still need further investigation.
Plasma Midregional Pro-adrenomedullin
ADM is a vasoactive peptide involved in several cardiovascular diseases and disorders in other systems [96, 97]. It is known to be a potent vasodilator, but it is difficult to measure its circulatory content [98]. Midregional pro-adrenomedullin (MR-proADM) is a stable fragment of pro-ADM and is produced in equimolar amounts with ADM. Zhang et al. [53] reported that children with POTS have significantly higher plasma MR-proADM levels than healthy controls. Children in the POTS group were treated with midodrine and were followed up for at least 3 months, and a symptom score was used to evaluate the therapeutic effect and identify the responders. As expected, the responders had higher plasma MR-proADM levels before treatment than the non-responders. Based on ROC analysis, when the plasma MR-proADM is >61.5 pg/mL, the sensitivity and specificity for the prediction of a therapeutic effect of midodrine are 100% and 72%, respectively. According to this study, the circulatory MR-proADM concentration, which correlates well with ADM production, can be used as an indicator to predict the therapeutic response to midodrine.
Plasma Copeptin
As noted above, copeptin can be used as a predictor of metoprolol therapy for the treatment of children with POTS. Zhao et al. [99] investigated plasma copeptin in another group of children with POTS and monitored the therapeutic effect of midodrine during follow-up. Increased plasma concentrations of copeptin, which in fact represent AVP concentrations, were found in the POTS group compared with the controls. AVP is known to be a strong vasoconstrictor; therefore, the elevated plasma copeptin levels might be explained by a compensatory response to vascular dysfunction in children with POTS. In addition, responders to midodrine in this study showed even higher plasma copeptin levels at the first visit than the non-responders. Applying a plasma copeptin concentration >10.5 pmol/L as the cutoff, the sensitivity and specificity for predicting the efficacy of midodrine in treating children with POTS reaches 86% and 76%, respectively. High levels of copeptin (or AVP) may indicate a state of relative hypovolemia. The authors explained that midodrine works better in patients with high copeptin levels probably because midodrine can abate the insufficient venous return in these patients by reducing the venous capacity through vasoconstriction.
Flow-Mediated Vasodilation
Endothelial function has been confirmed to play a critical role in modulating vascular tone. A previous study [51] explored endothelial function in children with POTS by measuring the plasma concentration of NO, the activity of NO synthase, and FMD. Both the plasma NO concentration and NO synthase activity were elevated, while FMD was greater in children diagnosed with POTS than in the control group, indicating that endothelial dysfunction most likely takes part in the pathogenesis of POTS. A subsequent study [100] revealed that baseline FMD is greater in responders to midodrine therapy than in non-responders during the follow-up of children with POTS. An FMD >9.85% can be taken as the referral determinant for the use of midodrine with moderate predictive value for treating such children. The sensitivity and specificity of prediction are 74% and 80%, respectively. This study also showed that the FMD of POTS patients decreases after treatment with midodrine, indicating that α1-adrenergic receptors may contribute to the regulation of endothelial function. The mechanism by which α1-adrenergic receptors interact with endothelium in POTS children is still unknown, although complex interactions have been reported between the endothelium and the autonomic nervous system [101].
Changes in Blood Pressure during the Standing Test
The orthostatic BP partly reflects the peripheral vascular tone during orthostasis. In a normal orthostatic response, the BP drops immediately after standing because of the gravitational redistribution of blood volume but recovers within 30 s due to the effect of the baroreflex [1]; then, the BP remains stable at a level almost equal to or no more than 10% above the supine BP [102]. It is worth considering focusing on the change in BP to distinguish different types of POTS. Patients with hyperadrenergic POTS may show a stronger increase in orthostatic BP and even orthostatic hypertension [3, 59]. In contrast, a slight decline in BP may appear in POTS patients with vascular dysfunction or hypovolemia [1]. Deng et al. [103] studied the difference in BP during the standing test between children diagnosed with POTS who were responsive and non-responsive to midodrine. In the baseline standing test before treatment, there was no significant difference between the two groups in supine BP, while the maximum upright systolic BP (SBP) and diastolic BP (DBP) as well as the increase in the SBP and DBP from the supine to the upright position were lower in responders than in non-responders. Based on further ROC analysis, effectiveness of midodrine in treating children with POTS can be expected by referring to the increase in SBP combined with an increase in DBP during the baseline standing test. The combined condition was an increase in SBP ≤0 mmHg (SBP declines) or an increase of DBP ≤6.5 mmHg. The predictive sensitivity and specificity are 72% and 88%, respectively, with this cutoff value. In other words, a decline in SBP or less of an increase in DBP during standing may indicate insufficient compensatory vasoconstriction or inappropriate vasodilatation and favor treatment with vasoconstrictors. This predictor is favorable because BP measurement during the diagnostic standing test is routine for children with POTS.
Individualized Management of POTS Based on Clinical Symptoms and Signs
Since POTS is a group of disorders with heterogeneous origins but similar clinical manifestations, researchers have made efforts to construct a proper classification to guide its management.
Early studies attempted to divide patients with POTS into three types according to their calf blood flow measured by venous occlusion strain gauge plethysmography [104, 105]. Low-flow POTS is considered to be related to mild absolute hypovolemia or abnormal regulation of local blood flow [106]. Patients of this type are characterized by cool, pallid extremities and acrocyanosis and are believed to benefit from therapy with volume loading by increased uptake of salt and water or drugs such as fludrocortisone or erythropoietin. Patients with high-flow POTS, manifesting with edema of the lower extremities, have reduced peripheral vascular resistance [46] and relatively enhanced blood volume. Their symptoms can be alleviated by vasoconstrictors such as α-adrenergic receptor agonists. Patients with normal-flow POTS may have splanchnic regional pooling [13] and can be treated with midodrine or a splanchnic-selective vasoconstrictor such as octreotide. Signs of hypermobility syndrome may occur in this type of patient [107]. These studies mainly involved adolescents and young adults and were aimed at individualized management for patients with POTS based on the characteristic underlying hemodynamic features. However, the optimal therapy for treating each type of POTS is based on theory, and better-designed controlled studies are needed to confirm the efficacy of various therapies in the treatment of the different subgroups.
A more recent study enrolled 708 children and adolescents with POTS and evaluated the effectiveness of available drugs in controlling symptoms including lightheadedness, headache, nausea, gastrointestinal dysmotility, pain, and insomnia [108]. All the drugs were effective in relieving specific symptoms to varying degrees. For example, fludrocortisone, midodrine, and desmopressin were used to control lightheadedness, and the effective rates were 42.8%, 33.9%, and 38.9%, respectively. For headache, the therapeutic effects of five drugs (cyproheptadine, verapamil, metoprolol, atenolol, and nebivolol) were evaluated, and they ranged from 14.2% for verapamil to 44.8% for metoprolol. For controlling nausea, ondansetron, scopolamine, and meclizine were effective in only 37.9%, 17%, and 8.7% of the patients, respectively. For gastrointestinal dysmotility, the remission rate was 36.7% with pyridostigmine and 32.7% with erythromycin. Duloxetine and pregabalin demonstrated efficacy in 41.2% and 34.3% of the patients suffering from pain symptoms. Finally, four drugs used for insomnia (clonidine, trazodone, zolpidem, and eszopiclone) alleviated the symptoms in 17.9% to 30.7% of patients. These results suggest that symptoms should be taken as essential references when making the choice of therapy for children with POTS, but treatment simply guided by symptoms does not achieve high effective rates.
Priorities of Reported Predictors in the Management of VVS and POTS
To date, several predictors of therapeutic effects have been reported to guide the individualized management of VVS and POTS in children. Physicians should cautiously evaluate the clinical manifestations and refer to one or more proper predictors before making the treatment plan for a specific patient. In clinical practice, comprehensive factors should be taken into account in the decision on therapeutic modalities. First, more sensitivity and specificity for prediction usually means more certainty of successful treatment. A sensitivity or specificity >90% indicates a high predictive value. Second, methods to determine the value of predictors can also impact the decision. For example, predictors drawn from the HUTT or standing test avoid additional examinations because the test is performed for diagnostic purposes. Predictors detected by ultrasound or 24-h urine collection are easily accepted by children and their parents because of their non-invasiveness. Predictors in items of routine blood tests are more favorable than those detectable only in some large testing institutions. We compare the predictive sensitivity and specificity as well as demands for detection among the reported predictors in Table 1. However, it is difficult to confirm the absolute priorities of these predictors based on the current information. It will be helpful to conduct studies that compare the therapeutic efficacy of individualized therapy based on the different predictors to determine their value in clinical practice and finally provide recommended priorities of the predictors.
Table 1.
Predictors of effectiveness of different therapies on VVS and POTS in children.
| Therapies | Predictors | Cut-off value | Sensitivity (%) | Specificity (%) | Tests or samples required |
|---|---|---|---|---|---|
| Research on children with VVS | |||||
| Orthostatic training | Acceleration index [22] | < 26.77 | 85 | 69.2 | HUTT |
| Metoprolol | ΔHR in HUTT [31] | > 30 bpm | 81 | 80 | HUTT |
| LVEF [34] | > 70.5% | 80 | 100 | UCG | |
| LVFS [34] | > 37.5% | 90 | 90 | UCG | |
| 24 h urine NE [35] | > 34.84 μg/24 h | 70 | 100 | 24-h urine | |
| Midodrine hydrochloride | FMD [37] | > 8.85% | 90 | 80 | Vascular ultrasound |
| Research on children with POTS | |||||
| ORS | 24 h urine sodium [73] | < 124 mmol/L | 93 | 77 | 24-h urine |
| BMI [75] | < 18 kg/m2 | 92 | 82.8 | General measurement | |
| MCHC [77] | > 347.5 g/L | 68.8 | 63.2 | Blood routine | |
| BRS [79] | > 17.01 ms/mmHg | 85.7 | 87.5 | CHMS | |
| Metoprolol | Orthostatic plasma NE [83] | > 3.59 pg/mL | 77 | 92 | Blood sample |
| Plasma CNP [87] | > 32.55 pg/mL | 96 | 70 | Blood sample | |
| Plasma copeptin [90] | < 10.2 pmol/L | 90 | 79 | Blood sample | |
| Midodrine hydrochloride | Plasma MR-proADM [53] | > 61.5 pg/mL | 100 | 72 | Blood sample |
| Ery H2S production [52] | > 27.1 nmol/min/108 RBC | 79 | 78 | Blood sample | |
| FMD [100] | > 9.85% | 74 | 80 | Vascular ultrasound | |
| Plasma copeptin [99] | > 10.5 pmol/L | 86 | 76 | Blood sample | |
| ΔBP in standing test [103] | ΔSBP ≤ 0 mmHg or ΔDBP ≤ 6.5 mmHg | 72 | 88 | Standing test | |
VVS, vasovagal syncope; POTS, posture tachycardia syndrome; HUTT, head-up tilt test; ΔHR in HUTT, the increase of heart rate before positive response in head-up tilt test compared with supine heart rate; LVEF, left ventricular ejection fraction; LVFS, left ventricular fractional shortening; UCG, ultrasound cardiogram; 24 h urine NE, 24-hour urine norepinephrine level; FMD, flow-mediated vasodilation; ORS, oral rehydration salt; BMI, body mass index; MCHC, mean corpuscular hemoglobin concentration; BRS, baroreflex sensitivity; NE, norepinephrine; CNP, Ctype natriuretic peptide; MRproADM, midregional proadrenomedullin; Ery H2S production 24 h urine sodium twentyfourhour urinary sodium concentration, erythrocytic hydrogen sulfide production; ΔBP, changes of blood pressure; ΔSBP, the increase of systolic blood pressure from supine to upright position; ΔDBP, the increase of diastolic blood pressure from supine to upright position; bpm, beats per minute; CHMS, Continuous hemodynamic monitoring system.
Perspectives for Individualized Management of VVS and POTS in Children and Adolescents
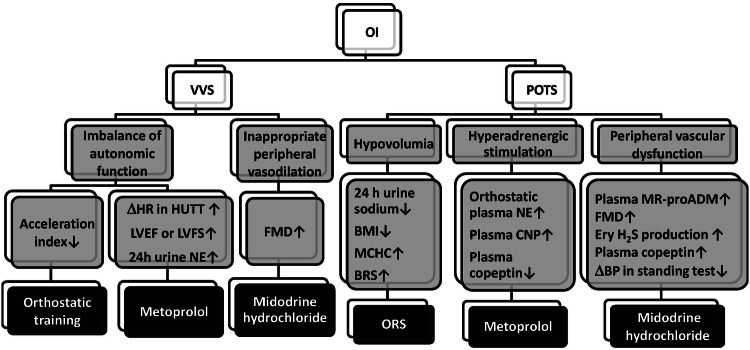
In summary, researchers have made great efforts to achieve individualized management of VVS and POTS in children (Table 1). Treatment strategies based on clinical features and pathogenesis are being established. To a certain extent, clinical symptoms can indicate the pathogenesis of children with OI. However, the same clinical manifestations can be induced by distinct underlying mechanisms; hence, the subjective symptoms can be taken as important references but not the main evidence for individualized therapeutic approaches. Conversely, objective indexes, such as hemodynamic parameters or biomarkers, can reflect the potential pathogenesis of pediatric VVS or POTS more exactly and are more suitable predictors of the therapeutic effect of different treatment approaches (Fig. 3). It is noteworthy that measures are more preferable if they are uncomplicated and non-invasive or at least minimally invasive for pediatric patients, as noted above. Therefore, a favorable individualized treatment protocol should combine subjective symptoms with objective predictors. We suggest that the relationship between the external symptom spectrum and the intrinsic pathogenesis of VVS or POTS in children should be defined – for example, by establishing a symptom scoring system indicating different mechanisms. In addition, more sensitive predictors of the therapeutic effect of different therapies with favorable detection methods should be identified in multicenter-based clinical studies and large-sample studies to realize the ideal individualized management for children and adolescents with VVS and POTS.
Fig. 3.
Schematic of the pathophysiology, corresponding indicators, and therapeutic options for vasovagal syncope and postural tachycardia syndrome in children. Grey boxes, potential pathophysiological mechanisms and changes in corresponding indicators; black boxes, individual therapeutic options based on the corresponding mechanisms; ↑, increase; ↓, decrease; OI, orthostatic intolerance; VVS, vasovagal syncope; POTS, postural tachycardia syndrome; ΔHR in HUTT, increase of heart rate before positive response in the head-up tilt test compared with supine heart rate; LVEF, left ventricular ejection fraction; LVFS, left ventricular fractional shortening; 24 h urine NE, twenty-four-hour urine norepinephrine level; FMD, flow-mediated vasodilation; 24 h urine sodium, twenty-four-hour urinary sodium concentration; BMI, body mass index; MCHC, mean corpuscular hemoglobin concentration; BRS, baroreflex sensitivity; ORS, oral rehydration salt; NE, norepinephrine; CNP, C-type natriuretic peptide; MR-proADM, midregional pro-adrenomedullin; Ery H2S production, erythrocytic hydrogen sulfide production; ΔBP, changes of blood pressure.
Acknowledgements
This review was supported by Peking University Clinical Scientist Program (BMU2019LCKXJ001, Beijing) and Fundamental Research Funds for the Central Universities of China.
Conflict of interest
The authors claim that there are no conflicts of interest.
References
- 1.Stewart JM, Boris JR, Chelimsky G, Fischer PR, Fortunato JE, Grubb BP, et al. Pediatric disorders of orthostatic intolerance. Pediatrics 2018, 141. pii: e20171673. [DOI] [PMC free article] [PubMed]
- 2.Shen WK, Sheldon RS, Benditt DG, Cohen MI, Forman DE, Goldberger ZD, et al. 2017 ACC/AHA/HRS guideline for the evaluation and management of patients with syncope: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Heart Rhythm. 2017;14:e155–e217. doi: 10.1016/j.hrthm.2017.03.004. [DOI] [PubMed] [Google Scholar]
- 3.Sheldon RS, Grubb BP, Olshansky B, Shen WK, Calkins H, Brignole M, et al. 2015 Heart Rhythm Society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm. 2015;12:e41–e63. doi: 10.1016/j.hrthm.2015.03.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wang C, Li Y, Liao Y, Tian H, Huang M, Dong XY, et al. Chinese Pediatric Cardiology Society (CPCS) guideline for diagnosis and treatment of syncope in children and adolescents. Sci Bull. 2018;63:1558–1564. doi: 10.1016/j.scib.2018.09.019. [DOI] [PubMed] [Google Scholar]
- 5.Sanatani S, Chau V, Fournier A, Dixon A, Blondin R, Sheldon RS, et al. Canadian Cardiovascular Society and Canadian Pediatric Cardiology Association Position Statement on the approach to syncope in the pediatric patient. Can J Cardiol. 2017;33:189–198. doi: 10.1016/j.cjca.2016.09.006. [DOI] [PubMed] [Google Scholar]
- 6.Brignole M, Moya A, de Lange FJ, Deharo JC, Elliott PM, Fanciulli A, et al. 2018 ESC Guidelines for the diagnosis and management of syncope. Eur Heart J. 2018;39:1883–1948. doi: 10.1093/eurheartj/ehy037. [DOI] [PubMed] [Google Scholar]
- 7.Tao C, Tang C, Chen S, Jin H, Du J. Autonomic nervous function in vasovagal syncope of children and adolescents. Neurosci Bull. 2019;35:937–940. doi: 10.1007/s12264-019-00383-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Aviado DM, Guevara Aviado D. The Bezold-Jarisch reflex. A historical perspective of cardiopulmonary reflexes. Ann NY Acad Sci 2001, 940: 48–58. [PubMed]
- 9.Scherrer U, Vissing S, Morgan BJ, Hanson P, Victor RG. Vasovagal syncope after infusion of a vasodilator in a heart-transplant recipient. N Engl J Med. 1990;322:602–604. doi: 10.1056/NEJM199003013220906. [DOI] [PubMed] [Google Scholar]
- 10.Liu JE, Hahn RT, Stein KM, Markowitz SM, Okin PM, Devereux RB, et al. Left ventricular geometry and function preceding neurally mediated syncope. Circulation. 2000;101:777–783. doi: 10.1161/01.cir.101.7.777. [DOI] [PubMed] [Google Scholar]
- 11.Fu Q, Levine BD. Pathophysiology of neurally mediated syncope: Role of cardiac output and total peripheral resistance. Auton Neurosci. 2014;184:24–26. doi: 10.1016/j.autneu.2014.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li H, Liao Y, Han Z, Wang Y, Liu P, Zhang C, et al. Head-up tilt test provokes dynamic alterations in total peripheral resistance and cardiac output in children with vasovagal syncope. Acta Paediatr. 2018;107:1786–1791. doi: 10.1111/apa.14342. [DOI] [PubMed] [Google Scholar]
- 13.Stewart JM, Medow MS, Glover JL, Montgomery LD. Persistent splanchnic hyperemia during upright tilt in postural tachycardia syndrome. Am J Physiol Heart Circ Physiol. 2006;290:H665–H673. doi: 10.1152/ajpheart.00784.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ocon AJ, Medow MS, Taneja I, Stewart JM. Respiration drives phase synchronization between blood pressure and RR interval following loss of cardiovagal baroreflex during vasovagal syncope. Am J Physiol Heart Circ Physiol. 2011;300:H527–H540. doi: 10.1152/ajpheart.00257.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vaddadi G, Guo L, Esler M, Socratous F, Schlaich M, Chopra R, et al. Recurrent postural vasovagal syncope: sympathetic nervous system phenotypes. Circ Arrhythm Electrophysiol. 2011;4:711–718. doi: 10.1161/CIRCEP.111.962332. [DOI] [PubMed] [Google Scholar]
- 16.Zhang Q, Jin H, Wang L, Chen J, Tang C, Du J. Randomized comparison of metoprolol versus conventional treatment in preventing recurrence of vasovagal syncope in children and adolescents. Med Sci Monit 2008, 14: CR199–203. [PubMed]
- 17.Qingyou Z, Junbao D, Chaoshu T. The efficacy of midodrine hydrochloride in the treatment of children with vasovagal syncope. J Pediatr. 2006;149:777–780. doi: 10.1016/j.jpeds.2006.07.031. [DOI] [PubMed] [Google Scholar]
- 18.Tan MP, Newton JL, Chadwick TJ, Gray JC, Nath S, Parry SW. Home orthostatic training in vasovagal syncope modifies autonomic tone: results of a randomized, placebo-controlled pilot study. Europace. 2010;12:240–246. doi: 10.1093/europace/eup368. [DOI] [PubMed] [Google Scholar]
- 19.On YK, Park J, Huh J, Kim JS. Is home orthostatic self-training effective in preventing neurally mediated syncope? Pacing Clin Electrophysiol. 2007;30:638–643. doi: 10.1111/j.1540-8159.2007.00725.x. [DOI] [PubMed] [Google Scholar]
- 20.Duygu H, Zoghi M, Turk U, Akyuz S, Ozerkan F, Akilli A, et al. The role of tilt training in preventing recurrent syncope in patients with vasovagal syncope: a prospective and randomized study. Pacing Clin Electrophysiol 2008, 31: 592–596. [DOI] [PubMed]
- 21.Bergström B, Manhem P, Bramnert M, Lilja B, Sundkvist G. Impaired responses of plasma catecholamines to exercise in diabetic patients with abnormal heart rate reactions to tilt. Clin Physiol. 1989;9:259–267. doi: 10.1111/j.1475-097x.1989.tb00978.x. [DOI] [PubMed] [Google Scholar]
- 22.Tao C, Li X, Tang C, Jin H, Du J. Acceleration index predicts efficacy of orthostatic training on vasovagal syncope in children. J Pediatr. 2019;207:54–58. doi: 10.1016/j.jpeds.2018.10.063. [DOI] [PubMed] [Google Scholar]
- 23.Chen LY, Shen WK. Neurocardiogenic syncope: latest pharmacological therapies. Expert Opin Pharmacother. 2006;7:1151–1162. doi: 10.1517/14656566.7.9.1151. [DOI] [PubMed] [Google Scholar]
- 24.Sra JS, Murthy VS, Jazayeri MR, Shen YH, Troup PJ, Avitall B, et al. Use of intravenous esmolol to predict efficacy of oral beta-adrenergic blocker therapy in patients with neurocardiogenic syncope. J Am Coll Cardiol. 1992;19:402–408. doi: 10.1016/0735-1097(92)90498-c. [DOI] [PubMed] [Google Scholar]
- 25.Madrid AH, Ortega J, Rebollo JG, Manzano JG, Segovia JG, Sanchez A, et al. Lack of efficacy of atenolol for the prevention of neurally mediated syncope in a highly symptomatic population: aprospective, double-blind, randomized and placebo-controlled study. J Am Coll Cardiol 2001, 37: 554–559. [DOI] [PubMed]
- 26.Flevari P, Livanis EG, Theodorakis GN, Zarvalis E, Mesiskli T, Kremastinos DT. Vasovagal syncope: a prospective, randomized, crossover evaluation of the effect of propranolol, nadolol and placebo on syncope recurrence and patients’ well-being. J Am Coll Cardiol. 2002;40:499–504. doi: 10.1016/s0735-1097(02)01974-5. [DOI] [PubMed] [Google Scholar]
- 27.Sheldon R, Connolly S, Rose S, Klingenheben T, Krahn A, Morillo C, et al. Prevention of Syncope trial (POST): a randomized, placebo-controlled study of metoprolol in the prevention of vasovagal syncope. Circulation. 2006;113:1164–1170. doi: 10.1161/CIRCULATIONAHA.105.535161. [DOI] [PubMed] [Google Scholar]
- 28.Müller G, Deal BJ, Strasburger JF, Benson DW., Jr Usefulness of metoprolol for unexplained syncope and positive response to tilt testing in young persons. Am J Cardiol. 1993;71:592–595. doi: 10.1016/0002-9149(93)90517-g. [DOI] [PubMed] [Google Scholar]
- 29.Scott WA, Pongiglione G, Bromberg BI, Schaffer MS, Deal BJ, Fish FA, et al. Randomized comparison of atenolol and fludrocortisone acetate in the treatment of pediatric neurally mediated syncope. Am J Cardiol. 1995;76:400–402. doi: 10.1016/s0002-9149(99)80110-6. [DOI] [PubMed] [Google Scholar]
- 30.Sheldon RS, Morillo CA, Klingenheben T, Krahn AD, Sheldon A, Rose MS. Age-dependent effect of beta-blockers in preventing vasovagal syncope. Circ Arrhythm Electrophysiol. 2012;5:920–926. doi: 10.1161/CIRCEP.112.974386. [DOI] [PubMed] [Google Scholar]
- 31.Zhang QY, Du JB, Zhen JL, Li WZ, Wang YL. Hemodynamic changes during head-up tilt test and predictive value thereof in predicting the efficacy of metoprolol therapy in children with vasovagal syncope. Zhonghua Yi Xue Za Zhi 2007, 87: 1260–1262. [Article in Chinese]. [PubMed]
- 32.Bondanelli M, Alboni P, Margutti A, Franceschetti P, Dinelli M, Gruppillo P, et al. Plasma galanin response to head-up tilt in normal subjects and patients with recurrent vasovagal syncope. Metabolism. 2003;52:315–321. doi: 10.1053/meta.2003.50051. [DOI] [PubMed] [Google Scholar]
- 33.Alboni P, Dinelli M, Gruppillo P, Bondanelli M, Bettiol K, Marchi P, et al. Haemodynamic changes early in prodromal symptoms of vasovagal syncope. Europace. 2002;4:333–338. doi: 10.1053/eupc.2002.0241. [DOI] [PubMed] [Google Scholar]
- 34.Song J, Li H, Wang Y, Liu P, Li X, Tang C, et al. Left ventricular ejection fraction and fractional shortening are useful for the prediction of the therapeutic response to metoprolol in children with vasovagal syncope. Pediatr Cardiol. 2018;39:1366–1372. doi: 10.1007/s00246-018-1904-x. [DOI] [PubMed] [Google Scholar]
- 35.Kong Q, Yang X, Cai Z, Pan Y, Wang M, Liu M, et al. Twenty-four-hour urine NE level as a predictor of the therapeutic response to metoprolol in children with recurrent vasovagal syncope. Ir J Med Sci. 2019;188:1279–1287. doi: 10.1007/s11845-019-01979-9. [DOI] [PubMed] [Google Scholar]
- 36.Strieper MJ, Campbell RM. Efficacy of alpha-adrenergic agonist therapy for prevention of pediatric neurocardiogenic syncope. J Am Coll Cardiol. 1993;22:594–597. doi: 10.1016/0735-1097(93)90070-h. [DOI] [PubMed] [Google Scholar]
- 37.Zhang FW, Liao Y, Li XY, Chen L, Jin HF, Du JB. The predictive value of flow-mediated vasodilation on therapeutic efficacy of midorine hydrochloride for vasovagal syncope in children. Zhongguo Shi Yong Er Ke Za Zhi 2012, 27: 102–105. [Article in Chinese].
- 38.Brignole M, Deharo JC, Menozzi C, Moya A, Sutton R, Tomaino M, et al. The benefit of pacemaker therapy in patients with neurally mediated syncope and documented asystole: a meta-analysis of implantable loop recorder studies. Europace. 2018;20:1362–1366. doi: 10.1093/europace/eux321. [DOI] [PubMed] [Google Scholar]
- 39.Paech C, Wagner F, Mensch S, Antonin Gebauer R. Cardiac pacing in cardioinhibitory syncope in children. Congenit Heart Dis. 2018;13:1064–1068. doi: 10.1111/chd.12682. [DOI] [PubMed] [Google Scholar]
- 40.Boris JR, Bernadzikowski T. Demographics of a large paediatric Postural Orthostatic Tachycardia Syndrome Program. Cardiol Young. 2018;28:668–674. doi: 10.1017/S1047951117002888. [DOI] [PubMed] [Google Scholar]
- 41.Boris JR. Postural orthostatic tachycardia syndrome in children and adolescents. Auton Neurosci. 2018;215:97–101. doi: 10.1016/j.autneu.2018.05.004. [DOI] [PubMed] [Google Scholar]
- 42.Li J, Liao Y, Du J, Zhang Q. Relationship between 24 hour urinary sodium and renin-angiotensin-aldosterone system in children with postural tachycardia syndrome. Natl Med J China 2015, 95: 2928–2932. [Article in Chinese]. [PubMed]
- 43.Raj SR, Biaggioni I, Yamhure PC, Black BK, Paranjape SY, Byrne DW, et al. Renin-aldosterone paradox and perturbed blood volume regulation underlying postural tachycardia syndrome. Circulation. 2005;111:1574–1582. doi: 10.1161/01.CIR.0000160356.97313.5D. [DOI] [PubMed] [Google Scholar]
- 44.Bai W, Chen SY, Jin HF, Du JB. Vascular dysfunction of postural tachycardia syndrome in children. World J Pediatr. 2018;14:13–17. doi: 10.1007/s12519-017-0104-8. [DOI] [PubMed] [Google Scholar]
- 45.Jacob G, Costa F, Shannon JR, Robertson RM, Wathen M, Stein M, et al. The neuropathic postural tachycardia syndrome. N Engl J Med 2000, 343: 1008–1014. [DOI] [PubMed]
- 46.Stewart JM. Pooling in chronic orthostatic intolerance: arterial vasoconstrictive but not venous compliance defects. Circulation. 2002;105:2274–2281. doi: 10.1161/01.cir.0000016348.55378.c4. [DOI] [PubMed] [Google Scholar]
- 47.Stewart JM, Weldon A. Reflex vascular defects in the orthostatic tachycardia syndrome of adolescents. J Appl Physiol. 2001;90:2025–2032. doi: 10.1152/jappl.2001.90.6.2025. [DOI] [PubMed] [Google Scholar]
- 48.Klein CM, Vernino S, Lennon VA, Sandroni P, Fealey RD, Benrud-Larson L, et al. The spectrum of autoimmune autonomic neuropathies. Ann Neurol. 2003;53:752–758. doi: 10.1002/ana.10556. [DOI] [PubMed] [Google Scholar]
- 49.Blitshteyn S. Autoimmune markers and autoimmune disorders in patients with postural tachycardia syndrome (POTS) Lupus. 2015;24:1364–1369. doi: 10.1177/0961203315587566. [DOI] [PubMed] [Google Scholar]
- 50.Fedorowski A. Postural orthostatic tachycardia syndrome: clinical presentation, aetiology and management. J Intern Med. 2019;285:352–366. doi: 10.1111/joim.12852. [DOI] [PubMed] [Google Scholar]
- 51.Liao Y, Chen S, Liu X, Zhang Q, Ai Y, Wang Y, et al. Flowmediated vasodilation and endothelium function in children with postural orthostatic tachycardia syndrome. Am J Cardiol. 2010;106:378–382. doi: 10.1016/j.amjcard.2010.03.034. [DOI] [PubMed] [Google Scholar]
- 52.Yang J, Zhao J, Du S, Liu D, Fu C, Li X, et al. Postural orthostatic tachycardia syndrome with increased erythrocytic hydrogen sulfide and response to midodrine hydrochloride. J Pediatr. 2013;163(1169–1173):e2. doi: 10.1016/j.jpeds.2013.04.039. [DOI] [PubMed] [Google Scholar]
- 53.Zhang F, Li X, Ochs T, Chen L, Liao Y, Tang C, et al. Midregional pro-adrenomedullin as a predictor for therapeutic response to midodrine hydrochloride in children with postural orthostatic tachycardia syndrome. J Am Coll Cardiol. 2012;60:315–320. doi: 10.1016/j.jacc.2012.04.025. [DOI] [PubMed] [Google Scholar]
- 54.Grubb BP, Kanjwal Y, Kosinski DJ. The postural tachycardia syndrome: a concise guide to diagnosis and management. J Cardiovasc Electrophysiol. 2006;17:108–112. doi: 10.1111/j.1540-8167.2005.00318.x. [DOI] [PubMed] [Google Scholar]
- 55.Liao Y, Du JB, Tang CS, Jin HF. Change and significance in the levels of plasma urotensin II and catestatin in children with postural orthostatic tachycardia syndrome. Beijing Da Xue Xue Bao 2011, 43: 436–439. [Article in Chinese]. [PubMed]
- 56.Wallman D, Weinberg J, Hohler AD. Ehlers-Danlos Syndrome and postural tachycardia syndrome: a relationship study. J Neurol Sci. 2014;340:99–102. doi: 10.1016/j.jns.2014.03.002. [DOI] [PubMed] [Google Scholar]
- 57.Bevegard S, Lodin A. Postural circulatory changes at rest and during exercise in five patients with congenital absence of valves in the deep veins of the legs. Acta Med Scand. 1962;172:21–29. doi: 10.1111/j.0954-6820.1962.tb07124.x. [DOI] [PubMed] [Google Scholar]
- 58.Bonyhay I, Freeman R. Sympathetic nerve activity in response to hypotensive stress in the postural tachycardia syndrome. Circulation. 2004;110:3193–3198. doi: 10.1161/01.CIR.0000147280.90339.E9. [DOI] [PubMed] [Google Scholar]
- 59.Grubb BP. Postural tachycardia syndrome. Circulation. 2008;117:2814–2817. doi: 10.1161/CIRCULATIONAHA.107.761643. [DOI] [PubMed] [Google Scholar]
- 60.Li H, Yu X, Liles C, Khan M, Vanderlinde-Wood M, Galloway A, et al. Autoimmune basis for postural tachycardia syndrome. J Am Heart Assoc. 2014;3:e000755. doi: 10.1161/JAHA.113.000755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Shannon JR, Flattem NL, Jordan J, Jacob G, Black BK, Biaggioni I, et al. Orthostatic intolerance and tachycardia associated with norepinephrinetransporter deficiency. N Engl J Med. 2000;342:541–549. doi: 10.1056/NEJM200002243420803. [DOI] [PubMed] [Google Scholar]
- 62.Dahan S, Tomljenovic L, Shoenfeld Y. Postural orthostatic tachycardia syndrome (POTS)–A novel member of the autoimmune family. Lupus. 2016;25:339–342. doi: 10.1177/0961203316629558. [DOI] [PubMed] [Google Scholar]
- 63.Watari M, Nakane S, Mukaino A, Nakajima M, Mori Y, Maeda Y, et al. Autoimmune postural orthostatic tachycardia syndrome. Ann Clin Transl Neurol. 2018;5:486–492. doi: 10.1002/acn3.524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Fedorowski A, Li H, Yu X, Koelsch KA, Harris VM, Liles C, et al. Antiadrenergic autoimmunity in postural tachycardia syndrome. Europace. 2017;19:1211–1219. doi: 10.1093/europace/euw154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ruzieh M, Batizy L, Dasa O, Oostra C, Grubb B. The role of autoantibodies in the syndromes of orthostatic intolerance: a systematic review. Scand Cardiovasc J. 2017;51:243–247. doi: 10.1080/14017431.2017.1355068. [DOI] [PubMed] [Google Scholar]
- 66.Vernino S, Stiles LE. Autoimmunity in postural orthostatic tachycardia syndrome: Current understanding. Auton Neurosci. 2018;215:78–82. doi: 10.1016/j.autneu.2018.04.005. [DOI] [PubMed] [Google Scholar]
- 67.Meck JV, Dreyer SA, Warren LE. Longduration head-down bed rest: project overview, vital signs, and fluid balance. Aviat Space Environ Med. 2009;80:A1–A8. doi: 10.3357/asem.br01.2009. [DOI] [PubMed] [Google Scholar]
- 68.Arbeille P, Kerbeci P, Mattar L, Shoemaker JK, Hughson R. Insufficient flow reduction during LBNP in both splanchnic and lower limb areas is associated with orthostatic intolerance after bedrest. Am J Physiol Heart Circ Physiol. 2008;295:H1846–H1854. doi: 10.1152/ajpheart.509.2008. [DOI] [PubMed] [Google Scholar]
- 69.Parsaik A, Allison TG, Singer W, Sletten DM, Joyner MJ, Benarroch EE, et al. Deconditioning in patients with orthostatic intolerance. Neurology. 2012;79:1435–1439. doi: 10.1212/WNL.0b013e31826d5f95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Burkhardt BE, Fischer PR, Brands CK, Sletten DM, Joyner MJ, Benarroch EE, et al. Exercise performance in adolescents with autonomic dysfunction. J Pediatr. 2011;158:15–19. doi: 10.1016/j.jpeds.2010.07.020. [DOI] [PubMed] [Google Scholar]
- 71.El-Sayed H, Hainsworth R. Salt supplement increases plasma volume and orthostatic tolerance in patients with unexplained syncope. Heart. 1996;75:134–140. doi: 10.1136/hrt.75.2.134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Hampton JL, Parry SW, Kenny RA, Newton JL. Lower 24 hour urinary sodium concentrations are associated with more severe symptoms in subjects with vasovagal syncope. Heart. 2004;90:687–688. doi: 10.1136/hrt.2003.020115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Zhang Q, Liao Y, Tang C, Du J, Jin H. Twenty-four-hour urinary sodium excretion and postural orthostatic tachycardia syndrome. J Pediatr. 2012;161:281–284. doi: 10.1016/j.jpeds.2012.01.054. [DOI] [PubMed] [Google Scholar]
- 74.Stewart JM, Taneja I, Medow MS. Reduced body mass index is associated with increased angiotensin II in young women with postural tachycardia syndrome. Clin Sci (Lond) 2007;113:449–457. doi: 10.1042/CS20070104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Li H, Wang Y, Liu P, Chen Y, Feng X, Tang C, et al. Body mass index (BMI) is associated with the therapeutic response to oral rehydration solution in children with postural tachycardia syndrome. Pediatr Cardiol. 2016;37:1313–1318. doi: 10.1007/s00246-016-1436-1. [DOI] [PubMed] [Google Scholar]
- 76.Lin CJ, Chu YK, Chern CM. RBC volume deficiency in patients with excessive orthostatic decrease in cerebral blood flow velocity. J Chin Med Assoc. 2014;77:174–178. doi: 10.1016/j.jcma.2014.01.005. [DOI] [PubMed] [Google Scholar]
- 77.Lu W, Yan H, Wu S, Xu W, Jin H, Du J. Hemocytometric measures predict the efficacy of oral rehydration for children with postural tachycardia syndrome. J Pediatr. 2017;187:220–224. doi: 10.1016/j.jpeds.2017.04.034. [DOI] [PubMed] [Google Scholar]
- 78.Convertino VA, Baumgartner N. Effects of hypovolemia on aortic baroreflex control of heart rate in humans. Aviat Space Environ Med. 1997;68:838. [PubMed] [Google Scholar]
- 79.Li H, Liao Y, Wang Y, Liu P, Sun C, Chen Y, et al. Baroreflex sensitivity predicts short-term outcome of postural tachycardia syndrome in Children. PLoS One. 2016;11:e0167525. doi: 10.1371/journal.pone.0167525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lai CC, Fischer PR, Brands CK, Fisher JL, Porter CB, Driscoll SW, et al. Outcomes in adolescents with postural orthostatic tachycardia syndrome treated with midodrine and beta-blockers. Pacing Clin Electrophysiol. 2009;32:234–238. doi: 10.1111/j.1540-8159.2008.02207.x. [DOI] [PubMed] [Google Scholar]
- 81.Arnold AC, Okamoto LE, Diedrich A, Paranjape SY, Raj SR, Biaggioni I, et al. Low-dose propranolol and exercise capacity in postural tachycardia syndrome: a randomized study. Neurology. 2013;80:1927–1933. doi: 10.1212/WNL.0b013e318293e310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Chen L, Wang L, Sun J, Qin J, Tang C, Jin H, et al. Midodrine hydrochloride is effective in the treatment of children with postural orthostatic tachycardia syndrome. Circ J. 2011;75:927–931. doi: 10.1253/circj.cj-10-0514. [DOI] [PubMed] [Google Scholar]
- 83.Zhang Q, Chen X, Li J, Du J. Orthostatic plasma norepinephrine level as a predictor for therapeutic response to metoprolol in children with postural tachycardia syndrome. J Transl Med. 2014;12:249. doi: 10.1186/s12967-014-0249-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Moyes AJ, Hobbs AJ. C-type natriuretic peptide: A multifaceted paracrine regulator in the heart and vasculature. Int J Mol Sci 2019, 20. pii: E2281. [DOI] [PMC free article] [PubMed]
- 85.Takekoshi K, Ishii K, Isobe K, Nomura F, Nammoku T, Nakai T. Effects of natriuretic peptides (ANP, BNP, CNP) on catecholamine synthesis and TH mRNA levels in PC12 cells. Life Sci 2000, 66: PL303–311. [DOI] [PubMed]
- 86.Springer J, Azer J, Hua R, Robbins C, Adamczyk A, McBoyle S, et al. The natriuretic peptides BNP and CNP increase heart rate and electrical conduction by stimulating ionic currents in the sinoatrial node and atrial myocardium following activation of guanylyl cyclase-linked natriuretic peptide receptors. J Mol Cell Cardiol. 2012;52:1122–1134. doi: 10.1016/j.yjmcc.2012.01.018. [DOI] [PubMed] [Google Scholar]
- 87.Lin J, Han Z, Li H, Chen SY, Li X, Liu P, et al. Plasma C-type natriuretic peptide as a predictor for therapeutic response to metoprolol in children with postural tachycardia syndrome. PLoS One. 2015;10:e0121913. doi: 10.1371/journal.pone.0121913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Afsar B. Pathophysiology of copeptin in kidney disease and hypertension. Clin Hypertens. 2017;23:13. doi: 10.1186/s40885-017-0068-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Franklin L, Bauce L, Pittman QJ. Depletion of central catecholamines reduce pressor responses to arginine vasopressin. Brain Res. 1988;438:295–298. doi: 10.1016/0006-8993(88)91350-9. [DOI] [PubMed] [Google Scholar]
- 90.Zhao J, Du S, Yang J, Lin J, Tang C, Du J, et al. Usefulness of plasma copeptin as a biomarker to predict the therapeutic effectiveness of metoprolol for postural tachycardia syndrome in children. Am J Cardiol. 2014;114:601–605. doi: 10.1016/j.amjcard.2014.05.039. [DOI] [PubMed] [Google Scholar]
- 91.Tao C, Liu X, Zhang C, Chen Y, Huang Y. Comments on 2018 CPCS guideline for diagnosis and treatment of syncope in children and adolescents. Sci Bull. 2019;64:291–292. doi: 10.1016/j.scib.2019.01.008. [DOI] [PubMed] [Google Scholar]
- 92.Li J, Zhang Q, Hao H, Jin H, Du J. Clinical features and management of postural tachycardia syndrome in children: a single-center experience. Chin Med J (Engl) 2014;127:3684–3689. [PubMed] [Google Scholar]
- 93.Xu WR, Wang TY. Diagnosis and treatment of syncope in pediatric patients: a new guideline. Sci Bull. 2019;64:357. doi: 10.1016/j.scib.2019.01.024. [DOI] [PubMed] [Google Scholar]
- 94.Li Z, Polhemus DJ, Lefer DJ. Evolution of hydrogen sulfide therapeutics to treat cardiovascular disease. Circ Res. 2018;123:590–600. doi: 10.1161/CIRCRESAHA.118.311134. [DOI] [PubMed] [Google Scholar]
- 95.Zhao W, Zhang J, Lu Y, Wang R. The vasorelaxant effect of H(2)S as a novel endogenous gaseous K(ATP) channel opener. EMBO J. 2001;20:6008–6016. doi: 10.1093/emboj/20.21.6008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Lyngbakken MN, Myhre PL, Røsjø H, Omland T. Novel biomarkers of cardiovascular disease: Applications in clinical practice. Crit Rev Clin Lab Sci. 2019;56:33–60. doi: 10.1080/10408363.2018.1525335. [DOI] [PubMed] [Google Scholar]
- 97.Önal U, Valenzuela-Sánchez F, Vandana KE, Rello J. Mid-regional pro-adrenomedullin (MR-proADM) as a biomarker for sepsis and septic shock: narrative review. Healthcare (Basel) 2018, 6. pii: E110. [DOI] [PMC free article] [PubMed]
- 98.Voors AA, Kremer D, Geven C, Ter Maaten JM, Struck J, Bergmann A, et al. Adrenomedullin in heart failure: pathophysiology and therapeutic application. Eur J Heart Fail. 2019;21:163–171. doi: 10.1002/ejhf.1366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Zhao J, Tang C, Jin H, Du J. Plasma copeptin and therapeutic effectiveness of midodrine hydrochloride on postural tachycardia syndrome in children. J Pediatr. 2014;165(290–294):e1. doi: 10.1016/j.jpeds.2014.04.032. [DOI] [PubMed] [Google Scholar]
- 100.Liao Y, Yang J, Zhang F, Chen S, Liu X, Zhang Q, et al. Flow-mediated vasodilation as a predictor of therapeutic response to midodrine hydrochloride in children with postural orthostatic tachycardia syndrome. Am J Cardiol. 2013;112:816–820. doi: 10.1016/j.amjcard.2013.05.008. [DOI] [PubMed] [Google Scholar]
- 101.Hearon CM, Jr, Kirby BS, Luckasen GJ, Larson DG, Dinenno FA. Endothelium-dependent vasodilatory signalling modulates α1-adrenergic vasoconstriction in contracting skeletal muscle of humans. J Physiol. 2016;594:7435–7453. doi: 10.1113/JP272829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Brignole M, Moya A, de Lange FJ, Deharo JC, Elliott PM, Fanciulli A, et al. Practical Instructions for the 2018 ESC Guidelines for the diagnosis and management of syncope. Eur Heart J. 2018;39:e43–e80. doi: 10.1093/eurheartj/ehy071. [DOI] [PubMed] [Google Scholar]
- 103.Deng W, Liu Y, Liu AD, Holmberg L, Ochs T, Li X, et al. Difference between supine and upright blood pressure associates to the efficacy of midodrine on postural orthostatic tachycardia syndrome (POTS) in children. Pediatr Cardiol. 2014;35:719–725. doi: 10.1007/s00246-013-0843-9. [DOI] [PubMed] [Google Scholar]
- 104.Stewart JM, Montgomery LD. Regional blood volume and peripheral blood flow in postural tachycardia syndrome. Am J Physiol Heart Circ Physiol. 2004;287:H1319–H1327. doi: 10.1152/ajpheart.00086.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Stewart JM. Chronic orthostatic intolerance and the postural tachycardia syndrome (POTS) J Pediatr. 2004;145:725–730. doi: 10.1016/j.jpeds.2004.06.084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Stewart JM, Medow MS, Montgomery LD. Local vascular responses affecting blood flow in postural tachycardia syndrome. Am J Physiol Heart Circ Physiol. 2003;285:H2749–H2756. doi: 10.1152/ajpheart.00429.2003. [DOI] [PubMed] [Google Scholar]
- 107.Gazit Y, Nahir AM, Grahame R, Jacob G. Dysautonomia in the joint hypermobility syndrome. Am J Med. 2003;115:33–40. doi: 10.1016/s0002-9343(03)00235-3. [DOI] [PubMed] [Google Scholar]
- 108.Boris JR, Bernadzikowski T. Utilisation of medications to reduce symptoms in children with postural orthostatic tachycardia syndrome. Cardiol Young. 2018;28:1386–1392. doi: 10.1017/S1047951118001373. [DOI] [PubMed] [Google Scholar]