Abstract
Antibodies against γ-aminobutyric acid B (GABAB) receptor are associated with limbic encephalitis (LE). It is estimated that ~1/2 of patients with LE have small-cell lung cancer. The present study analyzed the specific GABAB receptor antibodies in serum and cerebrospinal fluid (CSF) samples of 12 patients. The clinical manifestations, therapy and outcome were retrospectively compared. The median onset age was 65.1 years and all patients presented with new-onset seizures. In total, 11 (91.6%) patients had memory deficits, 7 (58.3%) patients had psychiatric problems and 4 (33.3%) patients had a disturbance of consciousness. Furthermore, lung cancer was detected in 7 patients (58.3%) by CT scan. Lymphocytic pleocytosis and protein concentration elevation in CSF were detected in 3 (25%) and 4 (33.3%) patients, respectively. Furthermore, MRI scan results identified 4 (33.3%) patients with abnormalities in the mesial temporal region. The lung cancer tissues of 3 patients were positively stained for anti-GABAB receptor on immunohistochemistry. All patients received antiepileptic drugs and immunotherapy. In total, 3 patients with lung cancer were subjected to tumor resection. Those patients without cancer exhibited neurological improvement at the follow-up. The present results suggested that seizures and memory deficits were the major manifestations in Chinese patients with anti-GABAB receptor antibodies who were responsive to immunotherapy. The lung cancer tissues from patients with anti-GABAB receptor antibodies were positively stained for anti-GABAB receptor. Collectively, the present results suggested that patients with underlying lung cancer have a relatively poor prognosis.
Keywords: γ-aminobutyric acid, encephalitis, Chinese, seizure, treatment
Introduction
Limbic encephalitis (LE) is characterized by autoimmune inflammation of structures of the limbic system. In the clinic, patients with LE present with mesial temporal lobe epilepsy, memory disturbance and neuropsychiatric symptoms (1-3). LE occurs in paraneoplastic and non-paraneoplastic settings (2). Furthermore, LE with autoantibodies against synaptic antigens includes leucine-rich glioma inactivated protein 1 (LGI1), α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor, metabotropic glutamate receptor 5 and γ-aminobutyric acid B (GABAB) receptors (1-3). Among these autoantibodies associated with LE, the autoantibody to GABAB receptor was first described by Lancaster et al (4).
Anti-GABAB receptor encephalitis is a relatively rare disease: Only 100 cases have been reported in the literature since 2010 (4-9). In addition, ~1/2 of patients with antibodies against GABAB receptor encephalitis have small cell lung cancer (SCLC). The majority of patients exhibit neurological improvement after immunotherapy and tumor therapy (4-9).
To date, only a small number of cases of positivity for antibodies against GABAB have been reported in the Asian population (7-9). Therefore, the present study investigated the clinical manifestations, therapy and outcomes in Chinese patients with GABAB receptor antibodies.
Materials and methods
Patients and methods
In total, 12 patients diagnosed with anti-GABAB receptor encephalitis at Qilu Hospital of Shandong University (Jinan, China) between August 2015 and December 2018 were included in the study. This study was approved by the Ethics Committee of Qilu Hospital of Shandong University (Jinan, China; no. KYLL-2017-550). Written informed consent was obtained from each patient or a relative serving as a legal representative. The diagnostic criteria were based on characteristic neurological syndromes suspected to be autoimmune-associated and the detection of specific GABAB receptor antibodies, as previously reported (10,11). All neurological syndromes, including LE and other neuropsychiatric manifestations, including ataxia, opsoclonus-myoclonus syndrome and brainstem encephalitis, were considered during patient selection. Detailed information, including clinical symptoms and results of laboratory examinations, cerebrospinal fluid (CSF) assay, electroencephalogram (EEG), radiologic examination (CT and MRI), as well as therapies and outcome information, were obtained.
Detection of autoimmune antibodies
Cell-based indirect immunofluorescence tests were used to detect the following autoantibodies: Anti-N-methyl-D-aspartate receptor, anti-GABAB receptor, anti-AMPA receptor, anti-LGI1 and anti-contactin-associated protein-like 2, and paraneoplastic antibodies anti-Yo (anti-Purkinje cell antibody), anti-Hu (anti-neuronal nuclear antibody 1), anti-Ri (anti-neuronal nuclear antibody 2), anti-CV2 (collapsin response mediator protein 5), anti-amphiphysin in serum and CSF samples (Euroimmun AG; cat. nos. FA 112d-1, FA 1111-1). Diluted patient samples were reacted with 293 cells (Euroimmun AG) transfected with plasmids containing human target gene sequences, and FITC-labeled anti-human immunoglobulin (Ig)G (cat. no. ab97224; 1:500 dilution; Abcam) was used as the secondary antibody (8). Positive and negative reactions were determined based on the intensity of cytoplasmic immunofluorescence compared with positive and negative controls under a fluorescence microscope(Olympus IX-70; Olympus Corporation).
Immunohistochemical staining
Anti-GABAB receptor in the tumor tissues were evaluated by immunohistochemical staining with specific antibodies. After deparaffinization in xylene and graded alcohol concentrations, endogenous peroxidase was blocked in 0.3% hydrogen peroxide. Non-specific binding was blocked by incubation in 10% bovine serum albumin (Sigma Aldrich; Merck KGaA). Sections were incubated with primary polyclonal antibody against human GABAB receptor (cat. no. sc-393270; 1:200 dilution; Santa Cruz Biotechnology, Inc.). A horseradish peroxidase-labeled secondary antibody (cat. no. sc-2005; 1:500 dilution; Santa Cruz Biotechnology, Inc.) was then added. The slides were stained with diaminobenzidine and then counterstained with hematoxylin. The stained slides were dehydrated and observed under a microscope. In total, lung cancer tissues from three patients were stained. The lung cancer tissues from one other patient who had SCLC without the manifestations of anti-GABAB receptor encephalitis was also stained and used as a control.
Treatment and follow-up
Patients received antiepileptic drug therapy, immunotherapy and tumor therapy when required. The therapeutic effects were assessed using the modified Rankin Scale (mRS) (12).
Results
Clinical manifestations
In total, nine patients were male (75%) and three were female (25%). The age of symptom onset ranged from 54 to 74 years (median, 65.1 years). The time of symptom onset to diagnosis was from 1 to 36 weeks (median, 6.9 weeks). Seizures occurred in all 12 patients and nine (75%) patients presented with seizures as the initial symptoms. Furthermore, three patients developed status epilepticus prior to the treatments. No seizure was recorded during EEG exams. The seizure frequency prior to treatment ranged from 2 to 15 times per week (median, 4.2 times per week). Furthermore, memory deficits and psychiatric symptoms (including behavioral, mood and personality changes) were documented in 11 (91.6%) and seven (58.3%) patients, respectively. Awareness impairment was observed in four patients (33.3%) (Table I). However, none of the patients had the clinical manifestations of ataxia, opsoclonus-myoclonus syndrome and brainstem encephalitis.
Table I.
Clinical manifestations of encephalitis patients with anti-GABABR antibody.
| Case1 | Case2 | Case3 | Case4 | Case5 | Case6 | Case7 | Case8 | Case9 | Case10 | Case11 | Case12 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sex | M | M | M | M | M | F | F | M | M | M | M | F |
| Age (years) | 62 | 70 | 64 | 54 | 66 | 62 | 69 | 74 | 67 | 65 | 69 | 60 |
| TOSD (weeks) | 8 | 3 | 2 | 2 | 7 | 2 | 8 | 9 | 2 | 3 | 1 | 36 |
| Psychiatric symptoms | - | + | + | + | + | - | + | + | - | - | + | - |
| Memory deficits | + | + | + | + | + | + | - | + | + | + | + | + |
| Awareness impairment | + | + | + | - | - | - | - | - | - | - | - | + |
| Seizures | + | + | + | + | + | + | + | + | + | + | + | + |
| Status epilepticus | + | - | - | + | - | - | - | - | - | - | + | - |
| Seizure frequency prior to therapy (times/week) | 3 | 5 | 4 | 15 | 3 | 3 | 2 | 3 | 4 | 2 | 3 | 3 |
| Anti-GABABR antibody (grading), serum/CSF | +/+++ | ++/+++ | +++/+++ | ++/++ | ++/++ | ++/+ | +++/++ | ++/+ | ++/++ | ++/+ | ++/+ | +++/++ |
| Anti-GABABR antibody (grading), lung cancer tissue | + | ND | ND | ND | ND | + | ND | ND | ND | ND | ND | ND |
| Other positive autoimmune antibodies | TG;TPO | - | - | - | - | - | TG;TPO | - | - | - | Hu | Hu; TG; TPO |
| Serum sodium (mmol/l) | 144 | 138 | 123 | 140 | 146 | 142 | 141 | 142 | 147 | 139 | 135 | 140 |
| CSF WC (/mm3; RR:0-6) | 2 | 130 | 4 | 62 | 2 | 6 | 4 | 1 | 34 | 1 | 6 | 4 |
| CSF protein (g/l; RR:0-0.45) | 0.74 | 0.84 | 0.26 | 0.24 | 0.33 | 0.35 | 0.70 | 0.45 | 0.42 | 0.44 | 0.62 | 0.42 |
| CSF OB | + | - | - | - | ND | - | - | ND | - | + | - | + |
| Lung tumor (CT scan) | + | + | - | + | + | + | - | - | - | + | - | + |
| Tumor tissue pathology | Aden | ND | - | ND | ND | SCLC | - | ND | ND | - | ND | SCLC |
| Brain MRI limbic lobes abnormality | + | - | + | - | + | + | - | - | - | - | - | - |
| Cortical atrophy | + | - | - | - | - | + | - | - | - | - | - | - |
| EEG generalized slow waves | + | + | + | ND | + | - | + | + | + | + | + | + |
| Epileptic waves | - | - | + | ND | + | - | - | + | + | - | + | + |
| Immunotherapy drugs | IVIg+Dex | Mpd | IVIg+Dex | IVIg+Dex | Mpd | IVIg | IVIg+Dex | IVIg+Mpd | IVIg+Mpd | IVIg | IVIg+Dex | IVIg |
| Anti-epileptic drugs | LEV | LEV | LEV+VPA | LEV | OXA+LEV | LEV | LEV | LEV+VPA | LEV+VPA | VPA | LEV+VAP | LEV+TOP |
| Tumor treatment mRS | Yes | No | ND | No | No | Yes | ND | ND | ND | No | No | No |
| (before/after treatments) | 4/2 | 4/4 | 4/2 | 3/3 | 3/3 | 3/2 | 3/1 | 3/2 | 3/1 | 3/3 | 3/2 | 4/4 |
| Follow-up duration (Months) | 30 | 6 | 18 | 4 | 5 | 8 | 18 | 6 | 20 | 10 | 8 | 3 |
| Follow-up results | No relapse | Died | No relapse | Died | Died | Died | No relapse | No relapse | No relapse | Died | Died | Died |
Grading: (+), 1:1-1:10; (++), 1:10-1:100; (+++), >1:100. GABABR, γ-aminobutyric acid B receptor; M, male; F, female; TOSD, time of onset of symptoms to diagnosis; CSF, cerebrospinal fluid; ND, not done or no date available; TG, anti-thyroglobulin antibody; TPO, anti-thyroid peroxidase antibody; WC, white cell; RR, reference range; OB, oligoclonal bands; SCLC, small cell lung cancer; Aden, adenocarcinoma; CT, computer tomography; MRI, magnetic resonance imaging; EEG, electroencephalogram; Mpd, methylprednisolone; IVIg, intravenous immunoglobulin; Dex, dexamethasone; LEV, levetiracetam; VPA, sodium valproate; OXA, oxcarbazepine; TOP, topiramate; mRS, modified Rankin scale.
Diagnostic examinations
No changes in the routine hematological and biochemical examinations among patients were identified. Only two patients (16.7%) presented with mild hyponatremia (<137 mmol/l). Antibodies against the GABAB receptor were detected in serum and CSF samples of all patients and titers ranged from 1:1 to 1:100. In addition, patients were screened for paraneoplastic antibodies, revealing that two patients (16.7%) had positive Hu antibodies in their serum and CSF. Furthermore, anti-thyroglobulin and anti-thyroperoxidase antibodies were detected in the serum samples of three patients (25%).
All patients underwent CSF analysis. It was demonstrated that white blood cells were elevated (>6 cells/mm3) in three cases (25%; range, 1-130/mm3). Furthermore, the protein concentration of CSF was elevated in four patients (33.3%; >0.45 g/l; range, 0.24-0.84 g/l). In total, three out of the 10 patients tested for CSF oligoclonal bands had positive results (30%; Table I).
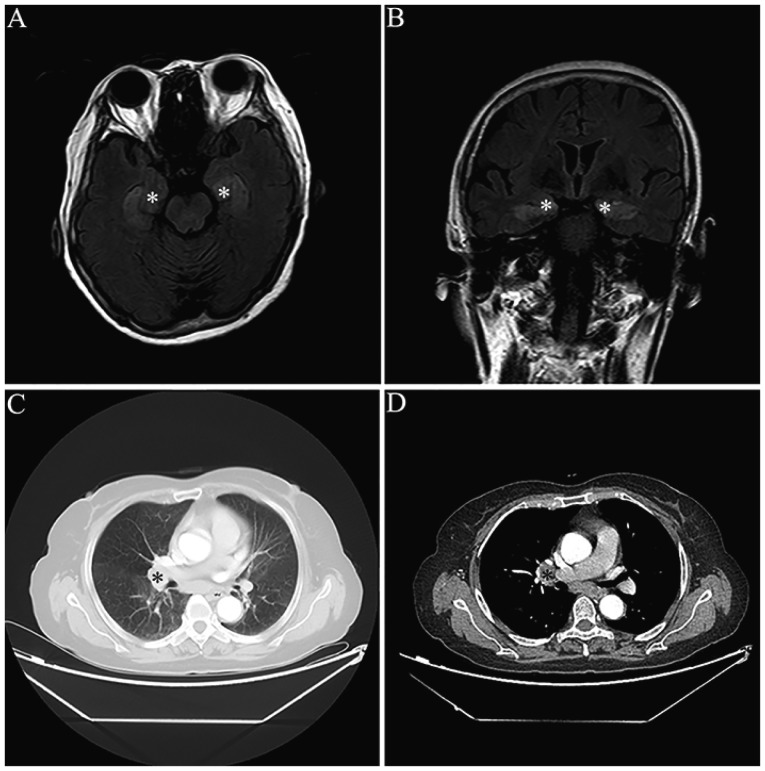
MRI scans demonstrated abnormalities in mesial temporal regions on T2-weighted and fluid-attenuated inversion recovery MRI sequences in four patients (33.3%) (Fig. 1). Furthermore, three patients exhibited bilateral abnormalities and one patient had unilateral abnormalities. It was identified that two patients (16.7%) had diffused cortical atrophy. In addition, EEG examination results were available for 11 patients. It was demonstrated that there were temporal lobe epileptic activities in six patients (6/11; 54.5%) and general slow waves in 10 patients (10/11; 90.9%; Table I).
Figure 1.
Images of one limbic encephalitis patient with anti-γ-aminobutyric acid B receptor antibody. (A) Transverse and (B) coronal sections of MRI fluid-attenuated inversion recovery sequence indicate increased signals in the bilateral mesial temporal lobes (white asterisks). (C and D) Thorax CT revealing one mass in the right lung hilus on (C) pulmonary and (D) mediastinal window image (black asterisks).
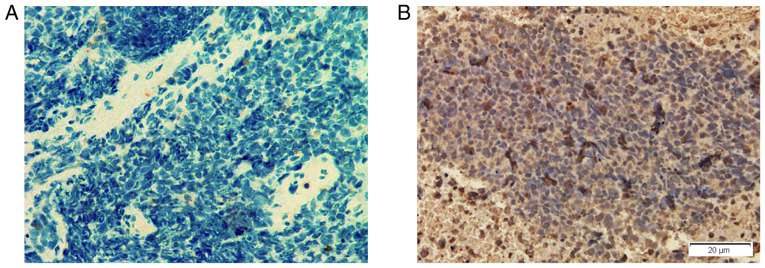
All patients received tumor screening by CT scans. Lung cancer was detected in seven patients (58.3%) (Fig. 1). Furthermore, tissue pathology exams indicated that two patients had SCLC and one patient had neuroendocrine adenocarcinoma (Table I). It was revealed that the lung cancer tissues of these three patients were positively stained for anti-GABAB receptors by immunohistochemistry (Fig. 2). However, the other four patients with tumors refused further pathological examinations and surgical treatments.
Figure 2.
GABAB receptors are expressed in the tumor tissue of a patient with small cell lung cancer and anti-GABAB receptor encephalitis. (A) No nuclei with brown staining are detected in the lung tumor tissues from one patient with small cell lung cancer without the manifestations of anti-GABAB receptor encephalitis on immunohistochemistry stain. (B) Numerous cells with brown colored nuclei were observed in lung tumor tissues of one patient (Case 6) with small cell lung cancer and anti-GABAB receptor encephalitis on immunohistochemistry stain. GABAB, γ-aminobutyric acid B.
Treatment and outcome assessment
All patients received antiepileptic drug treatments, including oxcarbazepine, sodium valproate, topiramate and levetiracetam. In addition, all patients received immunotherapy, which included intravenous immunoglobulin and/or the steroid hormones methylprednisolone or dexamethasone. In total, three patients with lung cancer received tumor resection and chemotherapy. The neurological function scores evaluated by mRS and the scores of the patients were 3.37±0.52 (range, 3-4) prior to therapy and 2.38±0.92 (range, 1-4) after therapy. The mean follow-up duration was 11.3 months (range, 3-30 months). Mortality occurred in seven patients at follow-up. Furthermore, after therapy, patients without tumors exhibited neurological improvement, including seizure control, and had no relapse at follow-up (range, 3-24 months; Table I).
Discussion
The present study assessed a number of Chinese patients with anti-GABAB receptor encephalitis. This rare disease primarily affects middle-aged and aged males who have a high risk of receptor encephalitis, usually manifesting as LE, and has symptoms including seizures, memory deficits, psychosis and altered consciousness (4-9). Furthermore, seizures are frequently the initial and most prominent symptom, which are usually refractory to anti-epileptic drugs but exhibit a response to immunotherapy (4-9,13). In the present study, all patients had seizures as the major symptom. Consistent with previous studies, manifestations including memory deficits, psychiatric changes and confusion were observed in the present study (4-9). The GABAB receptor is a G protein-coupled receptor for the inhibitory neurotransmitter GABA. The GABAB receptor is able to mediate pre-synaptic and post-synaptic GABAergic inhibition and suppress high activity states. Autoantibodies binding to GABAB receptor may promote synaptic activity states with excessive synchronization in neuronal networks, which leads to epileptic seizures (1-3,14). It has been previously demonstrated that mice with GABAB receptor dysfunction developed seizures and learning difficulties (15).
Hyponatremia was detected in two patients. One patient (Case 3) with obvious hyponatremia (123 mmol/l) had a symptom of vomiting. No malignant tumor was detected in this patient during the follow-up. CSF cytology of patients with anti-GABAB receptor encephalitis has no specific features compared with that of other types of autoimmune or viral encephalitis. Consistent with previous studies, certain patients in the present study had lymphocytic pleocytosis and a mildly elevated protein concentration (4-9). Furthermore, in the majority of patients, EEG exam results indicated slow or epileptic activity in the temporal lobes. In addition, MRI scans identified that 1/3 of patients had hyperintense signals in the mesial temporal lobes, which was consistent with the results of previous studies (4-9). It has been reported that brain MRI scans may exhibit dynamic changes in volume and signal intensity in the amygdala and hippocampus, which indicates considerable inflammation and subsequent degeneration (4-9). Furthermore, brain 18-fluoro-deoxyglucose positron emission tomography hypermetabolism has been identified in certain patients (7). In addition, MRI changes in patients cannot provide specific information for the diagnosis of anti-GABAB receptor encephalitis. Therefore, negative brain MRI scan results may not exclude the diagnosis of this disease.
Antibodies against GABAB receptors are mainly from the IgG1 subclass, which may induce neuronal damage directly via complement activation and antibody-dependent cell-mediated cytotoxicity (2). In the central nervous system, the GABAB receptor is primarily expressed in the hippocampus, amygdala, thalamus and cerebellum (1-3). Furthermore, ~1/2 of patients with anti-GABAB receptor encephalitis have a paraneoplastic etiology, which is usually SCLC and is frequently identified after the development of neurologic symptoms (4-9). Thymus carcinoid, melanoma and gastric adenocarcinoma are also reported in patients with anti-GABAB receptor encephalitis and patients with SCLC usually have a poor prognosis after immunotherapy (4-9,16,17). Similarly, at the follow-up for the present study, high mortality was reported in patients with lung cancer. In addition, the present immunohistochemistry results indicated that GABAB receptor was expressed in lung cancer tissues; to the best of our knowledge, this has not been previously reported. Pulmonary neuroendocrine cells may produce GABA and GABAB receptors are expressed in airway epithelium (18). Therefore, the present results supported the hypothesis that the ectopic expression of neuronal proteins by the tumor reduces immune tolerance for these proteins, which then contributes to the development of the autoimmune encephalitis (19). The GABAB receptors become autoimmune antigens, which leads to extensive infiltration of cytotoxic T cells and neuronal degeneration. This effect also triggers B-cell immune responses, thus leading to the production of autoantibodies with neuronal functional alterations (1-3).
Autoantibodies recognizing the extracellular domain of the GABAB receptor may be detected in serum and CSF of patients with GABAB receptor encephalitis (1). These patients may also have other autoantibodies, including anti-Hu, anti-voltage-gated calcium channel and anti-thyroid antibodies (4-9). Furthermore, co-existence of anti-GABAB receptor antibodies and onconeuronal antibodies in patients with SCLC are frequently associated with poor prognosis (4-9). In the present study, two patients were also determined to have anti-Hu antibodies. As the diagnosis of tumors is established after the diagnosis of anti-GABAB receptor encephalitis, screening for cancer is important once the clinical diagnosis is confirmed. Furthermore, it has been suggested that tumor screening should be performed after the encephalitis diagnosis (20).
For the treatment of GABAB receptor encephalitis with malignancy, immunotherapy and tumor treatment are necessary (4-9). The first line of immunotherapy includes corticosteroids, Igs and plasmapheresis, either alone or in combination (11). Furthermore, it is strongly recommended that the therapy should be started once the anti-GABAB receptor encephalitis is diagnosed. Seizures caused by anti-GABAB receptor encephalitis are frequently refractory to any antiepileptic drugs, but respond well to immunotherapy (21). In line with this, the present results suggested that patients without cancer also responded well to immunotherapy.
In conclusion, it was indicated that seizures and memory deficits are the major manifestations of anti-GABAB receptor encephalitis in Chinese patients. Therefore, testing for anti-GABAB receptor antibodies may be used for elderly patients with LE or new-onset refractory seizures. Most patients with anti-GABAB receptor encephalitis without cancer responded well to immunotherapy. However, patients with underlying lung cancer had a relatively poor prognosis.
Acknowledgements
Not applicable.
Funding
This work was supported by grants from the Natural Science Foundation of China (grant no. 81873786), the Natural Science Foundation of Shandong Province (grant no. ZR2017MH082), Innovative Research Project of Resident Standardization Training of Qilu Hospital, Shandong University (grant no. ZPZX2019A04) and Undergraduate Teaching Reform and Research Project of Cheeloo College of Medicine, Shandong University (grant no. qlyxjy-201917).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
XZ was responsible for the analysis of the data and the drafting of the manuscript. XY was responsible for the autoimmune antibody detection experiments and immunohistochemistry staining. XL was responsible for the analysis of the radiology data and the revision of the manuscript. SW was responsible for the design, data analysis, critical revision and final approval of the manuscript. All authors read and approved the final version of the manuscript.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Qilu Hospital of Shandong University (Jinan, China; no. KYLL-2017-550). Written informed consent was obtained from each patient or a relative serving as a legal representative.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Lancaster E, Dalmau J. Neuronal autoantigens--pathogenesis, associated disorders and antibody testing. Nat Rev Neurol. 2012;8:380–390. doi: 10.1038/nrneurol.2012.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Melzer N, Meuth SG, Wiendl H. Paraneoplastic and nonparaneoplastic autoimmunity to neurons in the central nervous system. J Neurol. 2013;260:1215–1233. doi: 10.1007/s00415-012-6657-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dalmau J, Geis C, Graus F. Autoantibodies to synaptic receptors and neuronal cell surface proteins in autoimmune diseases of the central nervous system. Physiol Rev. 2017;97:839–887. doi: 10.1152/physrev.00010.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lancaster E, Lai M, Peng X, Hughes E, Constantinescu R, Raizer J, Friedman D, Skeen MB, Grisold W, Kimura A, et al. Antibodies to the GABA(B) receptor in limbic encephalitis with seizures: Case series and characterisation of the antigen. Lancet Neurol. 2010;9:67–76. doi: 10.1016/S1474-4422(09)70324-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Höftberger R, Titulaer MJ, Sabater L, Dome B, Rózsás A, Hegedus B, Hoda MA, Laszlo V, Ankersmit HJ, Harms L, et al. Encephalitis and GABAB receptor antibodies: Novel findings in a new case series of 20 patients. Neurology. 2013;81:1500–1506. doi: 10.1212/WNL.0b013e3182a9585f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dogan Onugoren M, Deuretzbacher D, Haensch CA, Hagedorn HJ, Halve S, Isenmann S, Kramme C, Lohner H, Melzer N, Monotti R, et al. Limbic encephalitis due to GABAB and AMPA receptor antibodies: A case series. J Neurol Neurosurg Psychiatry. 2015;86:965–972. doi: 10.1136/jnnp-2014-308814. [DOI] [PubMed] [Google Scholar]
- 7.Kim TJ, Lee ST, Shin JW, Moon J, Lim JA, Byun JI, Shin YW, Lee KJ, Jung KH, Kim YS, et al. Clinical manifestations and outcomes of the treatment of patients with GABAB encephalitis. J Neuroimmunol. 2014;270:45–50. doi: 10.1016/j.jneuroim.2014.02.011. [DOI] [PubMed] [Google Scholar]
- 8.Guan HZ, Ren HT, Yang XZ, Lu Q, Peng B, Zhu YC, Shao XQ, Hu YQ, Zhou D, Cui LY. Limbic encephalitis associated with Anti-γ-aminobutyric Acid B receptor antibodies: A case series from china. Chin Med J (Engl) 2015;128:3023–3028. doi: 10.4103/0366-6999.168989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Qiao S, Zhang YX, Zhang BJ, Lu RY, Lai QL, Chen LH, Wu J. Clinical, imaging, and follow-up observations of patients with anti-GABAB receptor encephalitis. Int J Neurosci. 2017;127:379–385. doi: 10.1080/00207454.2016.1176922. [DOI] [PubMed] [Google Scholar]
- 10.Zuliani L, Graus F, Giometto B, Bien C, Vincent A. Central nervous system neuronal surface antibody associated syndromes: Review and guidelines for recognition. J Neurol Neurosurg Psychiatry. 2012;83:638–645. doi: 10.1136/jnnp-2011-301237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Graus F, Titulaer MJ, Balu R, Benseler S, Bien CG, Cellucci T, Cortese I, Dale RC, Gelfand JM, Geschwind M, et al. A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol. 2016;15:391–404. doi: 10.1016/S1474-4422(15)00401-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.van Swieten JC, Koudstaal PJ, Visser MC, Schouten HJ, van Gijn J. Interobserver agreement for the assessment of handicap in stroke patients. Stroke. 1988;19:604–607. doi: 10.1161/01.str.19.5.604. [DOI] [PubMed] [Google Scholar]
- 13.Hainsworth JB, Shishido A, Theeler BJ, Carroll CG, Fasano RE. Treatment responsive GABA(B)-receptor limbic encephalitis presenting as new-onset super refractory status epilepticus (NORSE) in a deployed U.S. soldier. Epileptic Disord. 2014;16:486–493. doi: 10.1684/epd.2014.0702. [DOI] [PubMed] [Google Scholar]
- 14.Emson PC. GABA(B) receptors: Structure and function. Prog Brain Res. 2007;160:43–57. doi: 10.1016/S0079-6123(06)60004-6. [DOI] [PubMed] [Google Scholar]
- 15.Prosser HM, Gill CH, Hirst WD, Grau E, Robbins M, Calver A, Soffin EM, Farmer CE, Lanneau C, Gray J, et al. Epileptogenesis and enhanced prepulse inhibition in GABA(B1)-deficient mice. Mol Cell Neurosci. 2001;17:1059–1070. doi: 10.1006/mcne.2001.0995. [DOI] [PubMed] [Google Scholar]
- 16.Boronat A, Sabater L, Saiz A, Dalmau J, Graus F. GABA(B) receptor antibodies in limbic encephalitis and anti-GAD associated neurologic disorders. Neurology. 2011;76:795–800. doi: 10.1212/WNL.0b013e31820e7b8d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jia XT, Pan Y, Di Z, Gu N, Liu Z, Kang YM. Anti-GABAB receptor encephalitis in a patient with gastric adenocarcinoma. Neurol Sci. 2018;39:1981–1984. doi: 10.1007/s10072-018-3536-6. [DOI] [PubMed] [Google Scholar]
- 18.Mizuta K, Osawa Y, Mizuta F, Xu D, Emala CW. Functional expression of GABAB receptors in airway epithelium. Am J Respir Cell Mol Biol. 2018;39:1981–1984. doi: 10.1165/rcmb.2007-0414OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.DeLuca I, Blachère NE, Santomasso B, Darnell RB. Tolerance to the neuron-specific paraneoplastic HuD antigen. PLoS One. 2009;4(e5739) doi: 10.1371/journal.pone.0005739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Titulaer MJ, Soffietti R, Dalmau J, Gilhus NE, Giometto B, Graus F, Grisold W, Honnorat J, Sillevis Smitt PA, Tanasescu R, et al. Screening for tumours in paraneoplastic syndromes: Report of an EFNS task force. Eur J Neurol. 2011;18:e19–e3. doi: 10.1111/j.1468-1331.2010.03220.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dubey D, Samudra N, Gupta P, Agostini M, Ding K, Van Ness PC, Vernino S, Hays R. Retrospective case series of the clinical features, management and outcomes of patients with autoimmune epilepsy. Seizure. 2015;29:143–147. doi: 10.1016/j.seizure.2015.04.007. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.