Abstract
Background:
Somatic inactivating mutations in ARID1A, a component of the SWI/SNF chromatin remodeling complex, are detected in various types of human malignancies. Loss of ARID1A compromises DNA damage repairs, and induced DNA damage burdens may increase the reliance on PARP-dependent DNA repairs of cancer cells to maintain genome integrity and renders cell susceptibility to PARP inhibitor therapy.
Design:
Isogenic ARID1A−/− and wild-type cell lines were used for assessing DNA damage response, DNA compactness and profiling global serine/threonine phosphoproteomic in vivo. A panel of inhibitors targeting DNA repair pathways was screened for a synergistic anti-tumor effect with irradiation in ARID1A−/− tumors.
Results:
ARID1A-deficient endometrial cells exhibited sustained levels in DNA damage response, a result further supported by in vivo phosphoproteomic analysis. Our results showed that ARID1A is essential for establishing an open chromatin state upon DNA damages, a process that is required for recruitment of 53BP1 and RIF1, key mediators of non-homologous end-joining (NHEJ) machinery, to DNA lesions. The inability of ARID1A−/− cells to mount NHEJ repair resulted in a partial cytotoxic response to radiation. Small-molecule compound screens indicated that PARP inhibitor acted synergistically with radiation to potentiate cytotoxicity in ARID1A−/− cells. Combination treatment with low-dose radiation and olaparib greatly improved anti-tumor efficacy, resulting in long-term remission in mice bearing ARID1A-deficient tumors.
Conclusion:
ARID1A-deficient cells acquire high sensitivity to PARP inhibition after exposure to exogenously induced DNA breaks such as ionizing radiation. Our findings suggest a novel biologically-informed strategy for treating ARID1A-deficient malignancies.
Keywords: Uterine cancer, Radiation therapy, ARID1A and DNA damage repair
Introduction
Altered chromatin structure, due to somatic mutations or epigenetic alterations of genes involved in chromatin remodeling, is a major contributor to tumor development (1). SWI/SNF chromatin remodeling complexes are an important class of epigenetic modulators, which regulate a plethora of basic biological functions including DNA replication, transcription, and DNA repair by remodeling chromatin configuration. Loss-of-function mutations in SWI/SNF genes have been identified in many types of human cancer, and an important example is the AT-rich interactive domain 1A (ARID1A) gene which is the most frequently mutated subunit in the SWI/SNF chromatin remodeling complex (2–5). ARID1A mutations are frequently detected in endometrium-derived cancers including ~50% of ovarian clear cell carcinomas, ~35% of uterine endometrioid carcinomas, and ~30% of ovarian endometrioid carcinomas (2,5–7). Mutations in ARID1A have also been reported in stomach, bladder, pancreas, and hepatocellular carcinomas among others (8–12). ARID1A is a tumor suppressor, and functions as a cell cycle checkpoint protein (3). Genetically engineered mice with ARID1A deletions in their ovarian surface epithelium are known to develop ovarian carcinomas in a background of PTEN inactivation or PIK3CA activation (13,14).
The finding of frequent ARID1A mutations in endometrium-derived cancers suggests that targeting the ARID1A pathway as an anti-cancer intervention has translational potential. However, it has become apparent that targeting tumor suppressor genes such as TP53 using standard pharmacological approach is highly challenging; in contrast, targeting gain-of-function oncogenes by pharmacological and antibody interventions has proven to be much promising. Nonetheless, loss-of-function mutations in tumor suppressors may result in acquisition of dependence of cancer cells on alternative compensatory pathway(s) or downstream molecular effector(s). This unique feature of loss-of-function mutations in tumor suppressor genes offers opportunities for targeting cancer cells by disrupting compensatory or alternative pathway(s)(15). A well-known example is the sensitivity of tumor cells with BRCA-inactivation to PARP inhibition, a consequence of their deficiency on homologous repair (HR) (16,17).
To extend this strategy to ARID1A−/− tumors, we sought first to understand the impact of inactivation of ARID1A or other SWI/SNF chromatin remodeling proteins in DNA damage repair pathways (18–20). In mammalian cells, DNA double-strand breaks (DSBs) are predominantly repaired by the NHEJ and HR pathways, each of which harness a unique set of molecular players. The balance between both pathways is essential for genome stability, and disturbance of the balance often leads to disease, including cancer. SWI/SNF chromatin remodeling has been reported to participate in the early phase (before strand intrusion phase) of DSB repair through rapid localization to the DSB sites, clearing local nucleosome occupancy, and physically facilitating recruitment of DNA repair enzymes and other modulators to the vicinity of DSBs (18–20). Thus, loss of ARID1A may disturb the balance of HR/NHEJ DNA repair efficacy and may render cells susceptible to specific genotoxic treatment. Indeed, two studies reported the involvement of ARID1A in NHEJ- and HR-mediated DSB repair, respectively (21,22). However, ARID1A inactivation-induced negative regulation of both NHEJ and HR repair pathways remains of interest, and has yet to be fully elucidated (23). It also remains to be determined which DNA repair mechanism or molecular pathway is employed by ARID1A-deficient tumors for survival and maintenance of DNA integrity in the face of endogenous stress and environmental challenge that result in DNA damages.
In this study, we first established that ARID1A deficiency led to a functional compromise in NHEJ repair and, to a lesser degree, in HR or alt-NHEJ. Similar to cells with NHEJ deficiency, ARID1A deficient cells were partially sensitive to radiation-induced DNA damage, likely due to sufficient HR DNA repair activity. We next performed a synthetic lethal screen to identify drugs affecting DNA repair that might act in concert with irradiation in ARID1A-deficient tumor cells. As a result, we identified PARP as a strong synthetic lethal interactor. Applying this treatment approach in animal models, we were able to induce long-term remission in ARID1A-deficient tumors, which persisted after completion of the treatment, whereas the same treatment was not effective in tumors with intact ARID1A function. Our findings indicate that disturbance of the DNA repair balance associated with ARID1A-deficiency can be exploited to develop highly specific and potent anticancer treatments.
Methods
Animal Studies and Tumor Xenografts
All animal-related procedures were approved by the Johns Hopkins University Animal Care and Use Committees. PAX8-rtTA/TetO-Cre mouse strains were acquired from Dr. Ronny Drapkin (13,24). Arid1aflox/flox mice were generated as previously described (13,24). For xenograft assays, 2×106 cells were injected subcutaneously with Matrigel (v/v) (BD Biosciences) into flanks of athymic nu/nu mice. Once the tumor volume reached ~100 mm3 (approximately 10–14 days), mice were randomized into four groups for further analysis.
Human Tissue Samples
Archived formalin-fixed and paraffin-embedded human tissues were provided by Seirei Mikatahara General Hospital, Japan. All specimens were de-identified and the study was approved by local ethical committee (IRB #14–46; 2014/12/15) for a waiver of informed consent of the subjects.
Cell lines
The immortalized normal human endometrial cell line, hEM3, previously established in our team was used for this study (25). Knockout of ARID1A was performed as previously described (25). Two pairs of isogenic cell lines, HCT116 and MCF10A (ARID1A+/+, ARID1A−/−), were purchased from Horizon Discovery Ltd. We used a previously established ARID1A-Tet-on OVISE cell line which originally lacks ARID1A expression due to inactivating mutation and re-expresses ARID1A upon tetracycline-induction (3). Other cancer cell lines (TOV21G1, RMG1, HEC151, ECC1) were grown as previously described (25).
Laser micro-irradiation assay
Cells were seeded in a Nunc glass-bottom dish (Thermo Scientific). After 5 min incubation with 2 μM Hoechst (Thermo Scientific), cells were mounted on a pre-heated (37°C) stage of a Zeiss LSM 710 confocal microscope equipped with a 405 nm laser source. To induce localized DSBs, the laser setting was set to 100% power output with 4 laser iterations. Image analysis was performed using Zeiss Zen 2010 software.
Statistical Analyses
Mann-Whitney test (two-tailed) was used to calculate the statistical significance between two experimental groups. A combination index was calculated as previously described (26), and synergistic drug interactions were analyzed by the method developed by Chou and Talalay (27). Two-way ANOVA with Bonferroni correction was used to calculate the statistical significance in experiments shown in Fig. 2C–E and Fig. S4. Statistical analyses were performed using GraphPad Prism 7 software (La Jolla, CA). All results are expressed as means ± SEM. Two-sided p ≤ 0.05 was considered significant.
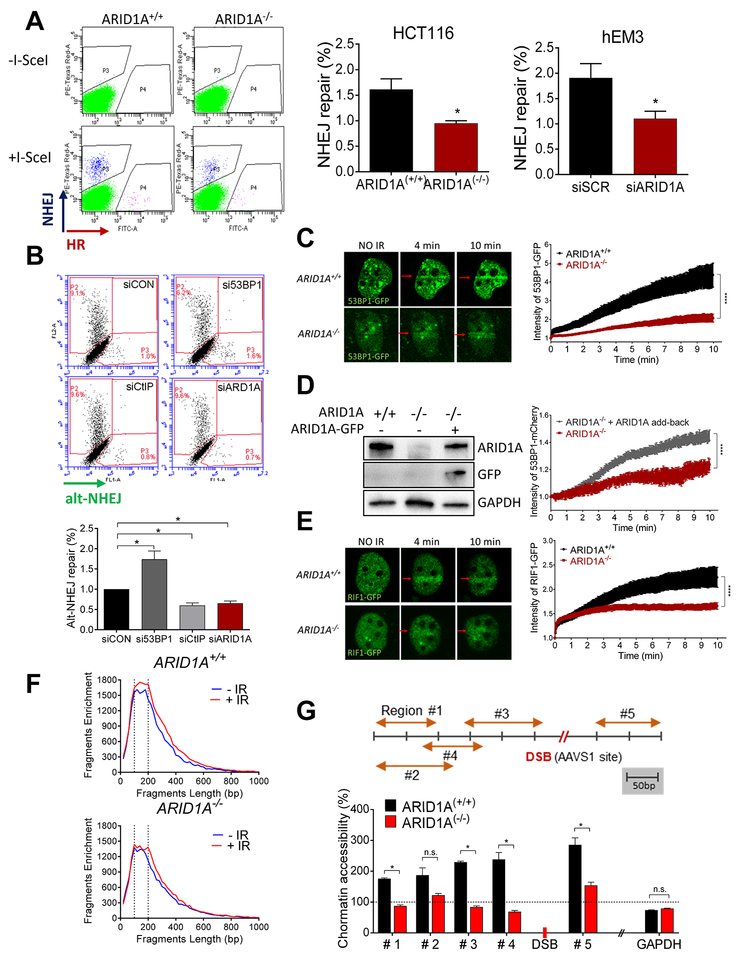
Fig. 2. ARID1A deficiency suppresses NHEJ-mediated repair of DSBs and chromatin accessibility.
(A) (left) Flow cytometric analysis of TLR expressing HCT116 ARID1A+/+ and ARID1A−/− cells performed 72 h after transduction with the I-Sec/GFP donor lentiviral construct. DSB repair mediated by HR is seen as GFP+, and repair mediated by NHEJ is seen as mCherry+. (right) Histograms show quantification of NHEJ repair in both isogenic HCT116 and gEM3 cells. Data are presented as mean ± SEM, n=4. Mann-Whitney test (two-tailed) was used to calculate statistical significance; *p<0.05. (B) Flow cytometric analysis of EJ2-expressing U2OS cells after transfection with specific siRNAs. Four days after transfection with I-Sec, alt-NHEJ repair ability, seen as a GFP+ signal, was quantified and plotted. Data are presented as mean ± SEM, n=4; *p<0.05. (C) (left): Representative live-cell images of ARID1A+/+ and ARID1A−/− cells expressing 53BP1-GFP at the indicated time points after microirradiation (red arrow). (right): Intensity of GFP at irradiated area (yellow box) was measured every 3 seconds and plotted after normalization. Data are presented as mean ± SEM; more than 7 cells were analyzed per group. (D) Immunoblots showing expression of ARID1A, GFP, and GAPDH. ARID1A−/− cells were co-transfected with 53BP1-mCherry and/or ARID1A-GFP. The intensity of mCherry at the micro-irradiated sites from the indicated cells was monitored over time. Data are presented as means ± SEM; more than 6 cells were analyzed per group. (E) (left): Representative live images of cells transfected with RIF1-GFP at the indicated time points after microirradiation (red arrow). (right): Intensity of GFP at irradiated area (yellow box) was measured every 3 seconds and plotted after normalization. Data were plotted as described in (C); more than 12 cells were analyzed per group. (C-E) Two-way ANOVA with Bonferroni correction was used to calculate the statistical significance; ***p<0.001. (F) ATAC-seq analysis was performed in ARID1A+/+ and ARID1A−/− cells before (blue) and 1 h after (red) irradiation (4 Gy). Fragment size (~146 bp) shown with dotted lines. (G) Schematic representation of the genomic locus of AAVS1 (chromosome 19) and regions amplified with specific primers (orange arrows). Following localized DSB at the AAVS1 site (red) (CRISPR/Cas9 transfected cells), chromatin was isolated from ARID1A+/+ and ARID1A−/− cells and was subjected to nuclease digestion or was untreated. Chromatin accessibility at specified regions was calculated and plotted after normalization to no-DSB control (CRISPR/Cas9 control plasmid transfection). GAPDH, which is located on a different chromosome (chromosome 12), was used as a control. Data are presented as mean ± SEM, n=4; *p<0.05.
Other methods can be found in Supplementary Materials.
Results
ARID1A deficiency is associated with a sustained DNA damage response and DSBs
To study the role of ARID1A in DNA damage, we first quantified DNA damage and the damage response in complex atypical hyperplasia (CAH) of endometrium which exhibited focal and heterogeneous loss of ARID1A in the same tissue sections (left panels, Fig. 1A). Immunohistochemical analysis showed more intense phospho-H2AX (S139, γH2AX) immunoreactivity in ARID1A-negative tumor areas, compared to adjacent areas with ARID1A expression (middle panels, Fig. 1A and Fig. S1). To confirm that increased γH2AX staining reflected DSBs, we also stained tissues with phosphorylated ATM (S1981), a marker of an activated DNA damage response. We observed concordance between pATM and γH2AX staining (right panels, Fig. 1A), implying that unrepaired DSBs were more prevalent in ARID1A-deficient epithelial cells.
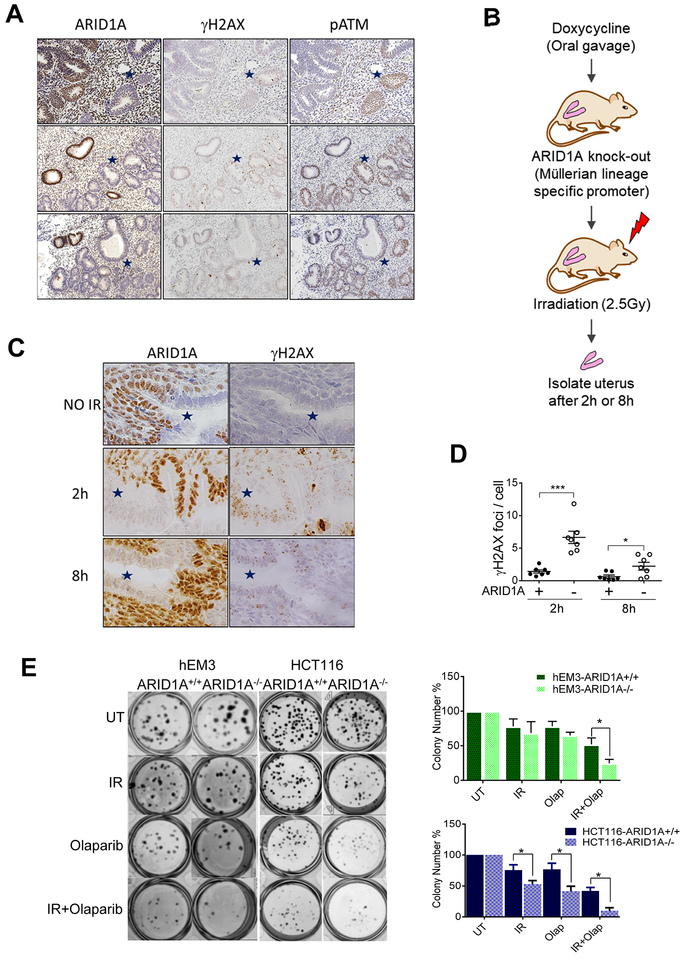
Fig. 1. ARID1A deficiency results in sustained DSBs and sensitizes cells to irradiation.
(A) Immunohistochemical staining patterns of ARID1A, γH2AX, and phosphorylated-ATM (s1981) in complex atypical hyperplasia (CAH) of endometrium. ARID1A-loss of expression areas are marked with blue stars. Representative images are shown. (B) Schematic illustration of the experimental procedure. (C) Immunohistochemical staining of ARID1A and γH2AX in endometrial tissue of ARID1Aflox/flox mice. Mice were treated with doxycycline for one week to induce deletion of ARID1A, and were sacrificed for analysis at the indicated time points after irradiation (2.5 Gy). ARID1A-loss areas are marked with blue stars. Representative images are shown. (D) Quantitation of γH2AX foci per endometrial epithelial cell. Data are presented as mean ± SEM; more than 500 cells were analyzed per group. Mann-Whitney test (two-tailed) was used to calculate significance; *p<0.05, ***p<0.001. (E) Clonogenic formation visualized by crystal violet staining on day 7 post-irradiation. Colony numbers were quantified and plotted (right panels). Data are presented as mean ± SEM, n=6; *p<0.05.
Although previous studies implicating chromatin remodeling proteins such as ARID1A in DNA repair, such findings have rarely been validated in vivo. To functionally establish the in vivo role of ARID1A in DSB repair, we assessed the status of DNA damage using a genetically engineered mouse model with conditional deletion of ARID1A. This model was established by crossing ARID1Aflox/flox mice with PAX8-Cre mice carrying doxycycline-inducible Cre recombinase under control of the Pax8 promoter (Fig. 1B). Pax8 is a transcription factor expressed in Mullerian epithelial cells lining the gynecologic tract, including endometrial epithelial cells; thus, doxycycline administration caused Pax8-driven, endometrial epithelium-specific deletion of ARID1A. In early stages of doxycycline administration, we observed a heterogeneous loss of ARID1A in murine uterine epithelium, shown by stretches of endometrial epithelium with ARID1A-loss (stars in left panels, Fig. 1C) alternating with stretches of ARID1A-expressing epithelium (Fig. 1C). This provided an opportunity to directly compare DNA damage in ARID1A expressing and non-expressing epithelium from the same animals. Whole-animal irradiation (2.5 Gy) was used to induce DSBs. Immunohistochemical staining revealed more prominent γH2AX punctate foci in ARID1A−/− cells at 2 h and 8 h post-irradiation compared to ARID1A-intact cells (right panels, Fig. 1C). Quantification of γH2AX foci per cell indicated that the increase in punctate foci in ARID1A−/− cells was significant (Fig. 1D).
To further assess the mechanistic role of ARID1A in DSB repair, we deleted ARID1A in an immortalized endometrial epithelial cell line, hEM3, using CRISPR/Cas9. Consistent with in vivo observations, irradiation resulted in higher levels of γH2AX in hEM3-ARID1A−/− than in hEM3-ARID1A+/+ cells at all time-points examined (top panel, Fig. S2A). We verified these results in parental hEM3 cells following siRNA-mediated silencing of ARID1A (bottom panel, Fig. S2A). Introduction of ARID1A in Tet-on OVISE diminished γH2AX levels (Fig. S2B). Results of a comet assay showed that ionizing radiation-induced DNA DSBs were exacerbated by siRNA-mediated ARID1A silencing in parental hEM3 cells, and were rescued by ARID1A re-expression in ARID1A-mutated OVISE cells (Fig. S2C–D).
We next performed a clonogenic assay to determine whether ARID1A-deficient cells were more sensitive to radiation-induced DNA damage. After irradiation, we observed a modest decrease in survival of HCT116-ARID1A−/− cells; however, we could not observe a different clonogenic survival after irradiation in hEM-ARID1A+/+ and ARID1A−/− cells (Fig. 1E). These results suggest that increased DNA damage in ARID1A-deficient cells could be rescued by a compensatory survival pathway, and suggest that an additional treatment combined with irradiation would be required for killing ARID1A-deficient cells.
DNA repair and cell cycle regulatory proteins associated with ARID1A deficiency identified by global in vivo phosphoproteomic profiling
To explore the underlying mechanism of increased DNA damage signals in ARID1A depleted cells, we performed global in vivo serine/threonine phosphoproteomic profiling as an unbiased approach to interrogate differential cellular responses to irradiation between ARID1A−/− and ARID1A+/+ tumor xenografts (Supplementary Table 1). Network analysis showed enrichment of proteins with altered phosphorylation involved in DNA replication and repair, including ATM, 53BP1, TOP2A, and TOP2B, in ARID1A−/− tumors (Fig. S3A). The finding of enriched pATM expression by phosphoproteomic profiling orthogonally validated our immunostaining data. Using Ingenuity Pathway Analysis (IPA), we found enriched pathways including NHEJ and Base Excision Repair (BER) DNA repair pathways involving XRCC1, XRCC4, ATM, and PRIM1, as well as Cell Cycle Control of Chromosomal Replication pathway involving TOP2A, TOP2B, and CDC7 (Fig. S3B). Collectively, our data provide new evidence of ARID1A in DSB replication and repair regulation. The data also support a view that defects in these pathways resulting from ARID1A loss led to a prolonged DNA damage response and persistence of DSBs after DNA damage insults.
ARID1A-dependent chromatin regulation and recruitment of NHEJ repair proteins
To determine whether ARID1A was required for NHEJ and/or HR, we monitored both repair mechanisms simultaneously (Fig. 2A) (28). A significant decrease in NHEJ activity was observed in ARID1A−/− cells. HR activity was also slightly decreased in ARID1A−/− cells, although precise quantitation of HR was difficult due to the low incidence even in the setting of ARID1A-intact cells. The impact of ARID1A loss on NHEJ appears to be of biological significance, as a high proportion of cells rely on this pathway. We further characterized the impact of ARID1A in alt-NHEJ, a NHEJ sub-pathway(29,30). Knockdown of CtIP (effector) and 53BP1 (suppressor) served as controls. We found that alt-NHEJ was also decreased in ARID1A-depleted cells (Fig. 2B). Thus, the data indicate that ARID1A deficiency causes a generalized impairment in DSB repair, with NHEJ being the process most significantly impacted.
Since the activity of NHEJ and HR DNA repair pathways is cell-cycle dependent (31), we asked whether ARID1A-deletion caused cell cycle alterations, which may indirectly affect DNA repair. Cell cycle analysis of hEM3 isogenic cell lines indicated that ARID1A−/− cells displayed reduced S and G2/M fractions compared to ARID1A+/+ cells (Fig. S4). This result is consistent with that observed in a pair of isogeneic HCT116 cell lines (22). Because HR occurs primarily during S and G2/M phases, we postulate that reduced HR observed in ARID1A-deficient cells can likely be attributed to attenuation in S and G2/M phases.
To determine whether an ARID1A-dependent process is required for recruitment of NHEJ repair proteins, we monitored recruitment of 53BP1 to DNA damage lesions induced by laser micro‐irradiation. We found a significant delay in the recruitment of this key NHEJ repair factor to sites of laser-induced DNA damage in ARID1A-deficient cells (Fig. 2C). Restoration of ARID1A expression in ARID1A-deficient cells partially restored 53BP1 recruitment efficiency (Fig. 2D). Similar results were observed for RIF1, another crucial component of the NHEJ repair pathway acting downstream of 53BP1 (Fig. 2E). There was no evidence of reciprocal regulation of ARID1A recruitment to DSBs by 53BP1. Knockdown or knockout of ARID1A in mammalian cells did not affect 53BP1 recruitment to DBSs induced by micro-irradiation (Fig. S5). Collectively, our results show that ARID1A deficiency compromises NHEJ repair by preventing the recruitment of key NHEJ proteins, including 53BP1 and RIF1, to DSB sites.
Since SWI/SNF complexes function as master regulators of chromatin structure, we hypothesized that regulation of the chromatin landscape by ARID1A is a key step in response to DSBs. To assess genome-wide chromatin configuration (openness vs. compactness), we performed ATAC-seq in ARID1A+/+ and ARID1A−/− cells before, and one hour after, irradiation (Fig. 2F). Enrichment of 146 bp DNA fragments indicates an increased open chromatin state because these fragments correspond to mono-nucleosome occupancy. As compared to ARID1A−/− cells, a larger 146 bp DNA peak was observed in ARID1A−/− cells after irradiation, implicating a functional role of ARID1A in modulating the global chromatin state in response to ionizing radiation. We introduced site-specific DSBs near the AAVS1 locus (chromosome 19) and the GAPDH locus (chromosome 12) in ARID1A+/+ and ARID1A−/− cells using CRISPR/Cas9, and evaluated chromatin accessibility near the introduced DSBs as described in Supplementary Materials. As expected, we observed increased chromatin accessibility at genomic regions proximal to the AAVS1 locus in ARID1A+/+ cells compared to ARID1A−/− cells (Fig. 2G). We did not observe significant differences in chromatin accessibility at genomic regions near GAPDH in ARID1A+/+ cells compared to ARID1A−/− cells (Fig. 2G). Based on these data, we conclude that ARID1A-dependent alteration in chromatin configuration, which facilitates the recruitment of key effectors of the DNA repair machinery to the damaged sites, is a prerequisite step for initiating DSB repair.
Synergistic cytotoxicity of PARP inhibition and irradiation in ARID1A-deficient cells
Because ARID1A-deficient cells were only marginally more radiosensitive than ARID1A-intact cells, we sought to identify an approach to enhance the therapeutic index. Since platinum-based chemotherapy (for example, carboplatin), is routinely used for treatment of ovarian and endometrial carcinomas, and has been shown to increase sensitivity to radiation therapy (32,33), we tested the sensitivity of ARID1A-deficient cells to carboplatin, either as a single agent or in combination with radiation. Clonogenic assays were performed on isogenic pairs of ARID1A+/+ and ARID1A−/− cells derived from hEM3 and HCT116 parental cell lines. Although hEM3-ARID1A−/− cells were slightly more sensitive to carboplatin than ARID1A+/+ cells, differences were less apparent in the HCT116 isogenic model. For both cell types, carboplatin combined with irradiation did not result in appreciable improvements over single agent treatment (Fig. S6A&B). This negative result prompted us to screen a panel of additional chemotherapeutic drugs and inhibitors of DNA repair and of epigenetic regulators (>Fig. 3A, S7). Among these drugs screened, the PARP inhibitor, olaparib, emerged as the most potent and specific radiosensitizer for ARID1A−/− cells (Fig. 3A). ATR inhibitor, AZD-6783, also enhanced potency of radiation therapy for ARID1A−/− cells. PIK-587, a dual PI3K and mTOR kinase inhibitor, potently inhibited cell growth irrespective of ARID1A deletion status (Fig. S7). Next, we tested the efficacy of combining PARP inhibitor and irradiation in a 3D culture system on both HCT116 and hEM3 pairs of isogenic cell lines. In these experiments, we observed synergistic cytotoxicity in ARID1A−/− cells (Fig. 3B). The increased sensitivity to ionizing radiation plus PARP inhibitor was further verified in clonogenic cell survival analysis in both hEM3 and HCT116 isogenic cell lines (Fig. 1E).
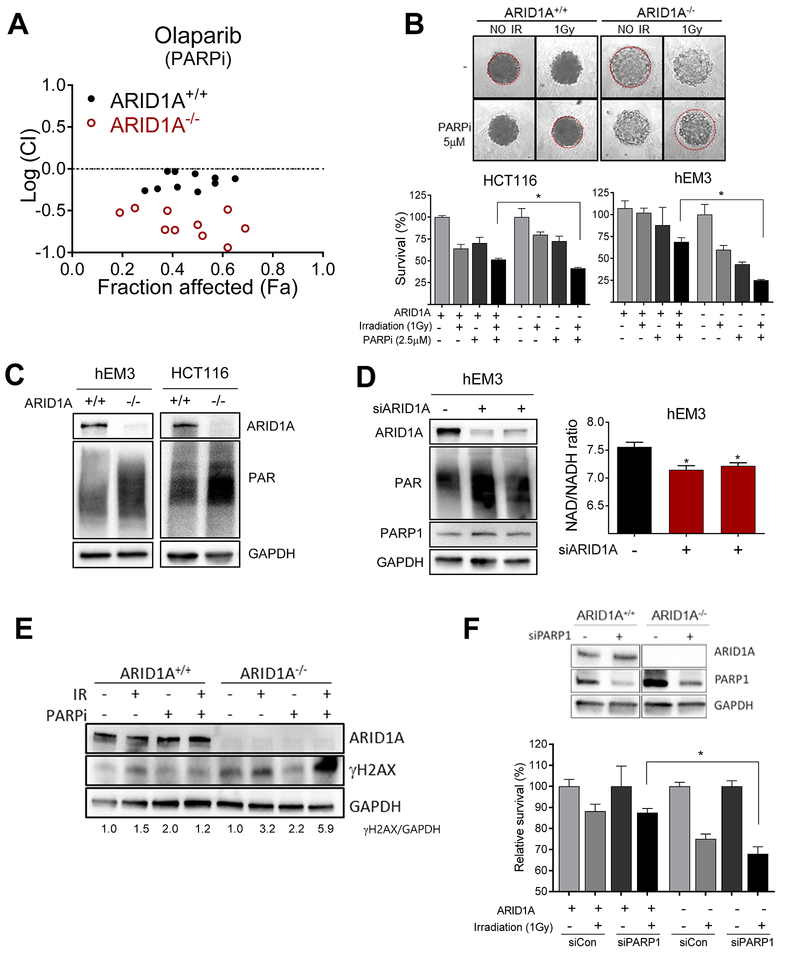
Fig. 3. Synergy between PARP inhibition and radiation in ARID1A-deficient cells.
(A) Logarithmic combination index (CI) plot of irradiation (1–2 Gy) in combination with PARP inhibitor, olaparib, over a range of concentrations in ARID1A+/+ and ARID1A−/− cells. The horizontal dashed line at Log (CI) = 0 separates synergy [Log (CI)<0] and antagonism [Log(CI)>0]. (B) Representative images of 3-D culture spheroids are shown. After treatment as indicated, cell survival was measured and plotted. Data are presented as mean ± SEM, n=4. Mann-Whitney test (two-tailed) was used to calculate the statistical significance between two comparison groups; *p<0.05. (C) Immunoblots for ARID1A, PARylation, and GAPDH of extracts from indicated cell lines. (D) (left) Immunoblots for ARID1A, PARylation, PARP1 and GAPDH of extracts from hEM3 cells transfected with ARID1A siRNA or scramble control siRNAs. (right) NAD/NADH ratio determined in hEM3 cells transfected with ARID1A or control siRNAs. Data are presented as mean ± SEM, n=4; *p<0.05. (E) Immunoblots for ARID1A, γH2AX, and GAPDH. The ratio of γH2AX/GAPDH is indicated at the bottom. (F) (top) Immunoblots showing efficiency of PARP1 silencing by siRNAs. (bottom) Effect of PARP1 knockdown on survival of ARID1A+/+ and ARID1A−/− tumor cells in the presence or absence of irradiation. Data are presented as mean ± SEM, n=4; *p<0.05. Mann-Whitney test (two-tailed).
To gain molecular insight into the synergistic effects, we compared PARP activity between ARID1A−/− and ARID1A+/+ cells. We observed elevated basal levels of PolyADP-ribosylation (PARylation) in ARID1A−/− cells (Fig. 3C). This result was confirmed by gene silencing using two different siRNAs (Fig. 3D). Since PARP-mediated PARylation occurs via consumption of NAD+, the relative level of NAD+ (NAD/NADH ratio) is another indicator of cellular PARP activity. We found that the NAD/NADH ratio was reduced in ARID1A knockdown hEM3 cells, confirming increased PARP activity in ARID1A-deficient cells (Fig. 3D). The extent of DNA breaks resulting from single or combined irradiation and PARP inhibitor exposure was assessed by measuring the levels of γH2AX (Fig. 3E). We found that combining PARP inhibition with irradiation led to significant increases in γH2AX levels in ARID1A−/− cells but not in isogenic ARID1A+/+ cells (Fig. 3E).
PARP inhibitors are thought to compromise DNA damage repair through two major mechanisms. First, they cause ‘PARP-trapping’ by preventing PARP dissociation from DNA. Another mechanism is through persistence of single-strand breaks, which ultimately progress to DSBs during replication. To determine which mechanisms are relevant to our model, we excluded a PARP-trapping effect by RNAi-mediated knockdown of PARP1 in hEM3-ARID1A−/− and ARID1A+/+ cells. When PARP1 was depleted, ARID1A−/− cells were still more sensitive to irradiation than ARID1A+/+ cells (Fig. 3F). These data are consistent with the notion that suppression of PARP1 catalytic activity is the primary mechanism underlying synergy between PARP inhibitor and irradiation.
Combining PARP inhibitor with irradiation is more effective than monotherapy for treating ARID1A-mutated tumors
In light of our observation that irradiation increased PARP inhibitor sensitivity in ARID1A−/− cells, we evaluated this treatment strategy in mice using isogenic HCT116 cell lines that differed only in ARID1A status (right panel, Fig. 4A). Tumor volumes and animal weights did not differ between different groups before treatment (Fig. S8A–B). Strikingly, irradiation combined with PARP inhibitor completely prevented the growth of ARID1A−/− tumors, demonstrating superior efficacy over either treatment alone (Fig. 4B). In contrast, the addition of PARP inhibitor did not enhance the effect of irradiation on reducing growth of ARID1A+/+ tumors — ARID1A+/+ tumors continued to progress insidiously (Fig. 4B). Immunoblotting of tumor lysates confirmed ARID1A expression and depletion of PARylated proteins by olaparib (Fig. S8C–D). To determine whether combination treatment led to long-lasting effects that persisted after treatment was terminated, we followed the animals that received combination therapy for four additional weeks after the final treatment. We found that ARID1A+/+ tumors regrew quickly after cessation of therapy, whereas ARID1A−/− tumors continued to shrink (Fig. 4C).
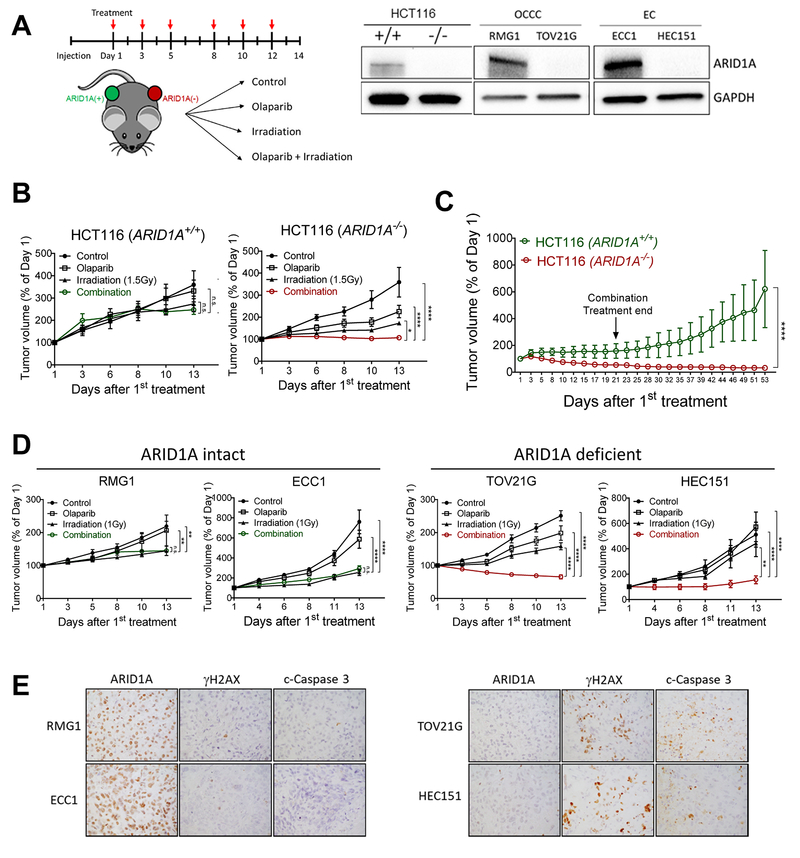
Fig. 4. Combination of PARP inhibitor and ionizing radiation inhibits growth and induces apoptosis in ARID1A-deficient tumor xenografts.
(A) (left) Schematic of treatment regimen in mice. Immunocompromised athymic nu/nu mice were inoculated with indicated human tumor cells, and 10–14 days later, mice were randomly stratified into 4 treatment groups. Olaparib (50 mg/kg) and/or irradiation (1–2 Gy) were administrated 3 times per week. (Right) Immunoblots performed to assess ARID1A protein expression in each cell line. (B) Tumor volume of HCT116-ARID1A+/+ and ARID1A−/− xenografts monitored for 2 weeks. Data are presented as mean ± SEM, n=5. Mann-Whitney test (two-tailed) was used to calculate the statistical significance between two comparison groups on day 13; *p<0.05, ****p<0.0001. (C) Relative tumor volume measured in mice inoculated with HCT116 cancer cells and treated with olaparib and ionizing radiation (2 Gy) combined therapy for three weeks. Tumor growth was monitored until day 53. Data are presented as mean ± SEM, n=4; ****p<0.0001. (D) Tumor volume measured in different groups. RMG1 and ECC1 are ARID1A wild-type and express ARID1A protein while TOV21G and HEC151 harbor inactivating mutations and lose ARID1A protein expression. Data are presented as mean ± SEM, n=5; *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001. (E) Immunohistochemical staining of ARID1A, γH2AX, and cleaved caspase-3 in tumor xenografts excised from mice treated with olaparib and ionizing radiation (2 Gy).
To determine the generalizability of these findings, we evaluated the same treatment regimen in additional xenograft models: ovarian clear cell carcinoma cell lines (RMG-1 and TOV21G) and uterine endometrioid cancer cell lines (ECC1 and HEC151). ECC1 and RMG-1 are ARID1A wildtype and express ARID1A protein, while HEC151 and TOV21G lose expression of ARID1A due to inactivation mutations (right panel, Fig. 4A). In mice bearing ARID1A-expressing tumors (ECC1 and RMG-1), PARP inhibition did not confer additional anti-tumor benefits over irradiation, whereas combination therapy was more effective than irradiation or PARP inhibitor monotherapy in mice bearing ARID1A-deficient tumors (HEC151 or TOV21G) (Fig. 4D). Immunohistochemical staining for γH2AX and cleaved caspase-3 revealed significantly higher levels of DSBs and cell death in ARID1A-deficient tumors than in ARID1A-expressing tumors following combination treatment (Fig. 4E). We continued monitoring the mice for another 3 weeks after treatment. While long-term remissions were achieved in mice bearing ARID1A-deficient tumors (HEC151 and TOV21G), early recurrence and tumor regrowth was observed in mice with ARID1A-expressing tumors (Fig. S8E).
Discussion
This investigation of mechanisms underlying the biological functions of ARID1A in NHEJ repair yielded novel biological and translational implications. Of particular importance, we discovered that combining radiation with PARP inhibitor therapy was highly effective in eradicating ARID1A-mutated tumors. The effect of PARP inhibition on ARID1A-mutated tumors was mechanistically distinct from effects on HR-deficient tumors, such as those harboring deleterious BRCA-mutations, which are intrinsically sensitive to PARP inhibitor monotherapy due to their loss of ability to repair PARP inhibitor-induced replication fork collapse (34,35). As to why PARP inhibitor monotherapy was less effective in ARID1A-deficient tumors, we speculate that the HR machinery in ARID1A-deficient cells remains intact, and the activity is sufficient to repair DSBs resulting from replication fork stalling, including those induced by PARP trapping.
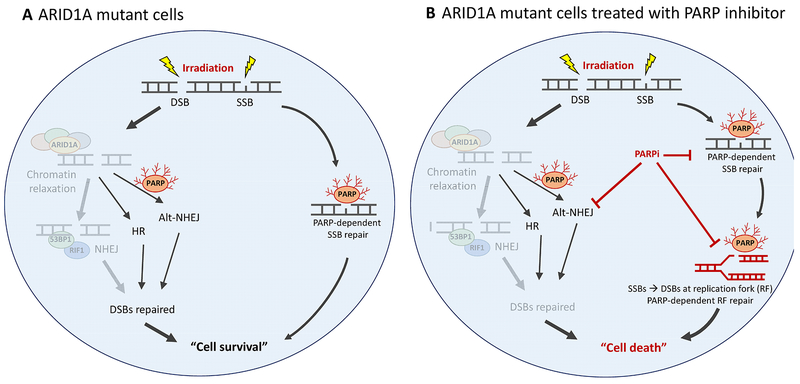
NHEJ is the primary repair mechanism for ionizing radiation-induced DSBs and the repair is active at all stages of cell cycle including G1 (36). We hypothesize that, because of the reduced NHEJ repair capacity in ARID1A-deficient cells, radiation-induced DNA breaks are not effectively repaired. As a result, ARID1A-deficient cells depend on other PARP-dependent DNA repair systems including HR, alt-NHEJ, and replication fork repairs to maintain DNA integrity. Accordingly, ionizing radiation-induced DNA breaks cause ARID1A-deficient cells to become profoundly dependent on these PARP-dependent repair pathways, which renders them highly sensitive to PARP inhibitors (Fig. 5)(35). If this hypothesis is correct, DNA-damaging agents that induce similar types of DNA breaks should exhibit synergy with PARP inhibition in ARID1A-mutated tumors, including human tumors. Indeed, our unpublished data using the alkylating agent, temozolomide, supports this view.
Fig. 5. ARID1A-deficiency renders cells dependent on compensatory DNA repair pathways involving PARP for repairing radiation-induced DNA breaks.
(A) Schematic models of the DNA repair pathways in ARID1A-mutant cells following irradiation. (B) ARID1A-mutated cells with irradiation and PARP inhibitor combined treatment.
A treatment regimen of combined ionizing radiation and PARP inhibition could, in principle, mediate effective killing of ARID1A-mutated tumor cells while sparing normal tissues, which retain ARID1A expression and functionality for efficient DNA repair. While the synthetic lethality of PARP inhibition in HR-defective BRCA mutated ovarian carcinomas is well-documented, the current study provides evidence in a new arena, demonstrating compromised DNA repair efficiency as an Achilles’ heel in ARID1A-mutated malignancies.
Radiotherapy is often a consideration in the management of locally-advanced and recurrent endometrial cancers, particularly for patients who are too frail for surgery (37,38). The anatomic distribution of clear cell and endometrioid carcinomas, which are characteristically confined to the pelvis, but which may metastasize to lymph nodes, renders these tumor types amenable to radiotherapy. ARID1A loss, which occurs in a significant proportion of cases, also confers intrinsic sensitivity to radiation. Importantly, our pre-clinical data support enhancement of efficacy by the combination of fractionated radiation with PARP inhibition, and should be tested in late stage, recurrent uterine endometrioid and clear cell carcinomas. With improvements in delivery of targeted radiation, there is renewed interest for applications of radiation therapy for ovarian cancer (39), and future studies should consider the addition of a PARP inhibitor to treat chemo-resistant ovarian endometrioid and clear cell carcinomas.
Importantly, identifying molecular genetic alterations predicting clinical response to radiation therapy remains an unmet need. Future clinical trials to test the proposed combination treatment strategy might consider accruing patients based on ARID1A mutation status, with the intent to evaluate ARID1A mutation or inactivation as a potential biomarker for predicting treatment outcome. A relevant question to be addressed is whether tumors with mutations in other subunits of the ARID1A chromatin remodeling complex display a similar phenotype. Lastly, efforts to optimize dosing schedules and safety monitoring will be necessary for translation of our preclinical findings to human trials.
Supplementary Material
Statement of translational relevance.
The role of PARP inhibitors beyond the treatment of homologous recombination-deficient cancers remains to be explored. ARID1A-deficient tumors, which are characterized by an attenuated capacity to carry out non-homologous end-joining (NHEJ) DNA repair, are only partially sensitive to PARP inhibitors. Here, we provide evidence that ARID1A-deficient tumors can become highly sensitive to PARP inhibitors following treatments, such as irradiation, that exogenously induce DNA breaks. Our findings provide pre-clinical evidence supporting a novel strategy for targeted treatment of ARID1A-deficient cancers and provide insight into the contribution of ARID1A in mediating DNA repair and replication.
Acknowledgements
The study is supported by NIH/NCI R21CA165807, RO1CA129080, P50CA228991, RO1CA215483, Gray Foundation, and the Richard W. TeLinde Endowment Fund from the Department of Gynecology and Obstetrics, Johns Hopkins University. The authors also thank Dr. Tae Magomi’s contribution to the preliminary work.
Footnotes
The authors declare no potential conflicts of interest.
References
- 1.Wilson BG, Roberts CW. SWI/SNF nucleosome remodellers and cancer. Nature reviews Cancer 2011;11:481–92 [DOI] [PubMed] [Google Scholar]
- 2.Jones S, Wang TL, Shih Ie M, Mao TL, Nakayama K, Roden R, et al. Frequent mutations of chromatin remodeling gene ARID1A in ovarian clear cell carcinoma. Science 2010;330:228–31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Guan B, Wang TL, Shih Ie M. ARID1A, a factor that promotes formation of SWI/SNF-mediated chromatin remodeling, is a tumor suppressor in gynecologic cancers. Cancer research 2011;71:6718–27 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Guan B, Mao TL, Panuganti PK, Kuhn E, Kurman RJ, Maeda D, et al. Mutation and loss of expression of ARID1A in uterine low-grade endometrioid carcinoma. The American journal of surgical pathology 2011;35:625–32 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wiegand KC, Shah SP, Al-Agha OM, Zhao Y, Tse K, Zeng T, et al. ARID1A mutations in endometriosis-associated ovarian carcinomas. N Engl J Med 2010;363:1532–43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cancer Genome Atlas Research N, Kandoth C, Schultz N, Cherniack AD, Akbani R, Liu Y, et al. Integrated genomic characterization of endometrial carcinoma. Nature 2013;497:67–73 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jones S, Stransky N, McCord CL, Cerami E, Lagowski J, Kelly D, et al. Genomic analyses of gynaecologic carcinosarcomas reveal frequent mutations in chromatin remodelling genes. Nat Commun 2014;5:5006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wu RC, Wang TL, Shih Ie M. The emerging roles of ARID1A in tumor suppression. Cancer Biol Ther 2014;15:655–64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bailey P, Chang DK, Nones K, Johns AL, Patch AM, Gingras MC, et al. Genomic analyses identify molecular subtypes of pancreatic cancer. Nature 2016;531:47–52 [DOI] [PubMed] [Google Scholar]
- 10.Cancer Genome Atlas Research N. Comprehensive molecular characterization of urothelial bladder carcinoma. Nature 2014;507:315–22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schulze K, Imbeaud S, Letouze E, Alexandrov LB, Calderaro J, Rebouissou S, et al. Exome sequencing of hepatocellular carcinomas identifies new mutational signatures and potential therapeutic targets. Nat Genet 2015;47:505–11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang K, Kan J, Yuen ST, Shi ST, Chu KM, Law S, et al. Exome sequencing identifies frequent mutation of ARID1A in molecular subtypes of gastric cancer. Nat Genet 2011;43:1219–23 [DOI] [PubMed] [Google Scholar]
- 13.Guan B, Rahmanto YS, Wu RC, Wang Y, Wang Z, Wang TL, et al. Roles of deletion of Arid1a, a tumor suppressor, in mouse ovarian tumorigenesis. Journal of the National Cancer Institute 2014;106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chandler RL, Damrauer JS, Raab JR, Schisler JC, Wilkerson MD, Didion JP, et al. Coexistent ARID1A-PIK3CA mutations promote ovarian clear-cell tumorigenesis through pro-tumorigenic inflammatory cytokine signalling. Nature communications 2015;6:6118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Muller FL, Aquilanti EA, DePinho RA. Collateral Lethality: A new therapeutic strategy in oncology. Trends in cancer 2015;1:161–73 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dedes KJ, Wilkerson PM, Wetterskog D, Weigelt B, Ashworth A, Reis-Filho JS. Synthetic lethality of PARP inhibition in cancers lacking BRCA1 and BRCA2 mutations. Cell Cycle 2011;10:1192–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fong PC, Boss DS, Yap TA, Tutt A, Wu P, Mergui-Roelvink M, et al. Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. The New England journal of medicine 2009;361:123–34 [DOI] [PubMed] [Google Scholar]
- 18.Chai B, Huang J, Cairns BR, Laurent BC. Distinct roles for the RSC and Swi/Snf ATP-dependent chromatin remodelers in DNA double-strand break repair. Genes & development 2005;19:1656–61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Densham RM, Garvin AJ, Stone HR, Strachan J, Baldock RA, Daza-Martin M, et al. Human BRCA1-BARD1 ubiquitin ligase activity counteracts chromatin barriers to DNA resection. Nat Struct Mol Biol 2016;23:647–55 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Aydin OZ, Marteijn JA, Ribeiro-Silva C, Rodriguez Lopez A, Wijgers N, Smeenk G, et al. Human ISWI complexes are targeted by SMARCA5 ATPase and SLIDE domains to help resolve lesion-stalled transcription. Nucleic Acids Res 2014;42:8473–85 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Watanabe R, Ui A, Kanno S, Ogiwara H, Nagase T, Kohno T, et al. SWI/SNF factors required for cellular resistance to DNA damage include ARID1A and ARID1B and show interdependent protein stability. Cancer research 2014;74:2465–75 [DOI] [PubMed] [Google Scholar]
- 22.Shen J, Peng Y, Wei L, Zhang W, Yang L, Lan L, et al. ARID1 A Deficiency Impairs the DNA Damage Checkpoint and Sensitizes Cells to PARP Inhibitors. Cancer discovery 2015;5:752–67 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lu H, Shamanna RA, de Freitas JK, Okur M, Khadka P, Kulikowicz T, et al. Cell cycle-dependent phosphorylation regulates RECQL4 pathway choice and ubiquitination in DNA double-strand break repair. Nature communications 2017;8:2039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Perets R, Wyant GA, Muto KW, Bijron JG, Poole BB, Chin KT, et al. Transformation of the fallopian tube secretory epithelium leads to high-grade serous ovarian cancer in Brca;Tp53;Pten models. Cancer Cell 2013;24:751–65 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Suryo Rahmanto Y, Jung JG, Wu RC, Kobayashi Y, Heaphy CM, Meeker AK, et al. Inactivating ARID1A Tumor Suppressor Enhances TERT Transcription and Maintains Telomere Length in Cancer Cells. J Biol Chem 2016;291:9690–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yu Y, Gaillard S, Phillip JM, Huang TC, Pinto SM, Tessarollo NG, et al. Inhibition of Spleen Tyrosine Kinase Potentiates Paclitaxel-Induced Cytotoxicity in Ovarian Cancer Cells by Stabilizing Microtubules. Cancer Cell 2015;28:82–96 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chou TC. Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer research 2010;70:440–6 [DOI] [PubMed] [Google Scholar]
- 28.Certo MT, Ryu BY, Annis JE, Garibov M, Jarjour J, Rawlings DJ, et al. Tracking genome engineering outcome at individual DNA breakpoints. Nat Methods 2011;8:671–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chiruvella KK, Liang Z, Wilson TE. Repair of double-strand breaks by end joining. Cold Spring Harb Perspect Biol 2013;5:a012757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Howard SM, Yanez DA, Stark JM. DNA damage response factors from diverse pathways, including DNA crosslink repair, mediate alternative end joining. PLoS Genet 2015;11:e1004943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Branzei D, Foiani M. Regulation of DNA repair throughout the cell cycle. Nat Rev Mol Cell Biol 2008;9:297–308 [DOI] [PubMed] [Google Scholar]
- 32.Atagi S, Kawahara M, Yokoyama A, Okamoto H, Yamamoto N, Ohe Y, et al. Thoracic radiotherapy with or without daily low-dose carboplatin in elderly patients with non-small-cell lung cancer: a randomised, controlled, phase 3 trial by the Japan Clinical Oncology Group (JCOG0301). Lancet Oncol 2012;13:671–8 [DOI] [PubMed] [Google Scholar]
- 33.Douple EB, Richmond RC, O’Hara JA, Coughlin CT. Carboplatin as a potentiator of radiation therapy. Cancer Treat Rev 1985;12 Suppl A:111–24 [DOI] [PubMed] [Google Scholar]
- 34.Konecny GE, Kristeleit RS. PARP inhibitors for BRCA1/2-mutated and sporadic ovarian cancer: current practice and future directions. Br J Cancer 2016;115:1157–73 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Murai J, Huang SY, Das BB, Renaud A, Zhang Y, Doroshow JH, et al. Trapping of PARP1 and PARP2 by Clinical PARP Inhibitors. Cancer research 2012;72:5588–99 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mahaney BL, Meek K, Lees-Miller SP. Repair of ionizing radiation-induced DNA double-strand breaks by non-homologous end-joining. Biochem J 2009;417:639–50 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Suidan RS, He W, Sun CC, Zhao H, Smith GL, Klopp AH, et al. National trends, outcomes, and costs of radiation therapy in the management of low- and high-intermediate risk endometrial cancer. Gynecologic oncology 2019;152:439–44 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.van der Steen-Banasik E, Christiaens M, Shash E, Coens C, Casado A, Herrera FG, et al. Systemic review: Radiation therapy alone in medical non-operable endometrial carcinoma. European journal of cancer (Oxford, England: 1990) 2016;65:172–81 [DOI] [PubMed] [Google Scholar]
- 39.Fields EC, McGuire WP, Lin L, Temkin SM. Radiation Treatment in Women with Ovarian Cancer: Past, Present, and Future. Frontiers in oncology 2017;7:177. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.