Abstract
Coronavirus disease 2019 (COVID‐19) represents a very heterogeneous disease. Some aspects of COVID‐19 pneumonia question the real nature of ground glass opacities and its consolidative lesions. It has been hypothesized that COVID‐19 lung involvement could represent not only a viral effect but also an immune response induced by the infection, causing epithelial/endothelial lesions and coagulation disorders. We report 3 cases of COVID‐19 pneumonia in which contrast‐enhanced ultrasound was suggestive of consolidations with perfusion defects, at least in part caused by ischemic or necrotic changes and not only by inflammatory or atelectasis events.
Keywords: contrast‐enhanced ultrasound, COVID‐19, lung ultrasound
Abbreviations
- ARDS
acute respiratory distress syndrome
- CEUS
contrast‐enhanced ultrasound
- COVID‐19
coronavirus disease 2019
- SARS
severe acute respiratory syndrome
- US
ultrasound
Coronavirus disease 2019 (COVID‐19) causes clusters of severe pneumonia, evolving into respiratory failure and death through bilateral pulmonary involvement. 1 Respiratory failure requiring intensive support is similar to ARDS. The wide and simultaneous spread of the infection, with the need for advanced health care support, makes this pandemic a real health emergency.
COVID‐19 is a heterogeneous disease. Not all people exposed are infected; not all people infected show important symptoms, and not all symptomatic patients develop respiratory failure. COVID‐19 can be divided into 3 stages: (1) an asymptomatic period, with or without 2019 novel coronavirus polymerase chain reaction swab positivity; (2) a nonsevere symptomatic period; and (3) a severe symptomatic stage with respiratory failure. 2 The first period is probably the most important from an epidemiologic point of view because the finding from the viral RNA swab can fail. The stage with severe respiratory failure seems to represent a time‐related spectrum within phenotypes. 3
Computed tomography is the reference standard for identifying lung involvement in patients with COVID‐19, with sensitivity of 97.2% at presentation, showing bilateral patchy lesions with a mainly peripheral parenchymal pattern. Ground glass opacities are present in the early stage, whereas consolidation and a mixed pattern (47.0%) are noted from the second week. Ground glass opacities plus interlobular septal thickening (crazy‐paving pattern) seem to be the main pattern in third week. 4 , 5
These phenomena may be attributed to the fact that consolidations may regress to small areas of irregular linear opacities. When the clinical course is favorable, the disease improves in the third week, When the disease progresses, lung infiltrates spread, and consolidative areas in the posterior segments of the lower lobes appear.
Although some features of COVID‐19 resemble those described for the usual viral pneumonia, some morphologic aspects of the lung involvement question the real nature of the consolidative lesions. This is particularly intriguing in light of the hypothesis that COVID‐19 could represent not only a viral effect but also an immune response induced by the infection. 6 In this series, we describe 3 cases of confirmed COVID‐19 with pulmonary consolidations in which studies with contrast‐enhanced ultrasound (CEUS) 7 showed large perfusion defects in the lung.
Case Descriptions
Three male patients were admitted for fever (>39°C), and symptoms compatible with COVID‐19. Severe acute respiratory syndrome (SARS) coronavirus 2 polymerase chain reaction nasal and pharyngeal swab results were positive, and hematologic findings were in accord with the diagnosis of COVID‐19. Some clinical characteristics of the patients at admission are summarized in Table 1. All patients underwent radiographic and chest ultrasound (US) examinations within 3 hours of admission, according to the guidelines approved at our center, and all signed an informed consent form. The chest US examination was performed with an RS85 system equipped with a 3.5–5‐MHz transducer (Samsung Medison, Seoul, Korea), exploring the patients in a sitting position, scanning the bases and the posterior and lateral regions of the chest. The anterior chest was explored in the supine position. The results of the chest radiographic and US examinations are summarized in Table 2.
Table 1.
Patients’ Details
| Characteristic | Patient 1 | Patient 2 | Patient 3 |
|---|---|---|---|
| Sex | Male | Male | Male |
| Age | 56 | 38 | 65 |
| Time from onset, d | 10 | 4 | 4 |
| po 2, mm Hg | 46 | 20.3 | 90.2 |
| pco 2, mm Hg | 35.3 | 51.2 | 37.2 |
| White cells/μL | 13,800 | 2,420 | 10,080 |
| Lymphocytes, % | 6.3 | 0.5 | 8.4 |
| Lactate dehydrogenase, U/L | 569 | 299 | 299 |
| Alanine aminotransferase, IU/L | 48 | 26 | 38 |
| Aspartate aminotransferase, IU/L | 65 | 30 | 42 |
| C‐reactive protein, mg/dL | 24.28 | 4.26 | 21.98 |
| d‐Dimer, ng/mL | 779 | 766 | 1276 |
| Platelets/μL | 270,000 | 118,000 | 238,000 |
| Fibrinogen, mg/dL | >700 | >700 | >700 |
| INR | 1.15 | 1.13 | 1.4 |
Table 2.
Patient‐Specific Radiographic and US Findings
| Patient | Radiographic Findings | US Findings |
|---|---|---|
| 1 | Extended parenchymal densities in the upper and middle fields bilaterally and in the lower left field | Bilateral anterior and posterior patchy vertical artifacts (B‐lines), small subpleural consolidations, and large basal left consolidation with air bronchogram |
| 2 | Bilateral middle basal interstitial thickenings, more visible to the left, where they had a consolidative aspect | Inhomogeneous vertical patchy vertical artifacts and patchy white lung in lateral and posterobasal areas, small subpleural consolidations, bibasal large consolidations with air bronchograms, and minimal bilateral pleural effusion |
| 3 | Hiloperihilar bilateral interstitial densities and interstitial thickening in the middle and basal fields | Separate vertical artifacts anteriorly, confluent vertical artifacts and white lung areas in posterior fields, small subpleural consolidations, and large left posterobasal consolidation |
All patients showed radiographic interstitial and consolidative abnormalities. Ultrasound findings were characterized by vertical artifacts (B‐lines), often grouped and confluent in white lung areas, and with a patchy appearance. All patients had echographic parenchymal consolidations at the bases, in a gravitational position (Table 2), whereas only 1 subject had minimal bilateral pleural effusion. In each patient, there was more than 1 nonbasal subpleural consolidation, less than 2 cm in diameter, with a wedge‐shaped or angular aspect, containing central air artifacts. The US findings of COVID‐19 lung involvement were comparable with the descriptions found in the literature. 8 , 9
The patients required noninvasive respiratory support but had an uneventful recovery. The symptoms improved after combined treatment with sarilumab, azithromycin, and enoxaparin, allowing discharge of the patients in 10 to 12 days.
Because of the presence of small cuneiform lung consolidations with a central echogenic spot (a US sign described in patients with pulmonary embolism), the finding of high level of d‐dimer, and the impossibility of performing chest computed tomography with a contrast medium, a CEUS examination in accordance with European Federation of Societies for Ultrasound in Medicine and Biology guidelines 7 was performed at the bedside, after acquisition of written informed consent. The patients were in a supine position with the sides alternately raised to explore the rear bases. Video sequences were acquired in the dual mode (basal and CEUS protocol) after rapid intravenous administration of 2.4 mL of a suspension of sulfur hexafluoride microbubbles (SonoVue; Bracco SpA, Milan, Italy), followed by bolus of 5 mL of normal saline. The enhancement was continuously observed for 5 minutes.
After intravenous injection of SonoVue, the major consolidations showed abnormal early arterial enhancement within 9 seconds from the administration (Figure 2). The enhancement was partial, often inhomogeneous, without evidence of pulmonary arteries in a segmentary arrangement. In particular, incomplete enhancement of the major consolidations was achieved, especially in part of the pulmonary cortex, whereas little or no perfusion of large deep parts was observed. The boundaries between perfused and nonperfused areas were sharp. Consolidations of less than 2 cm did not show enhancement (Figure 3). Early wash‐out phenomena were not noticed.
Figure 2.
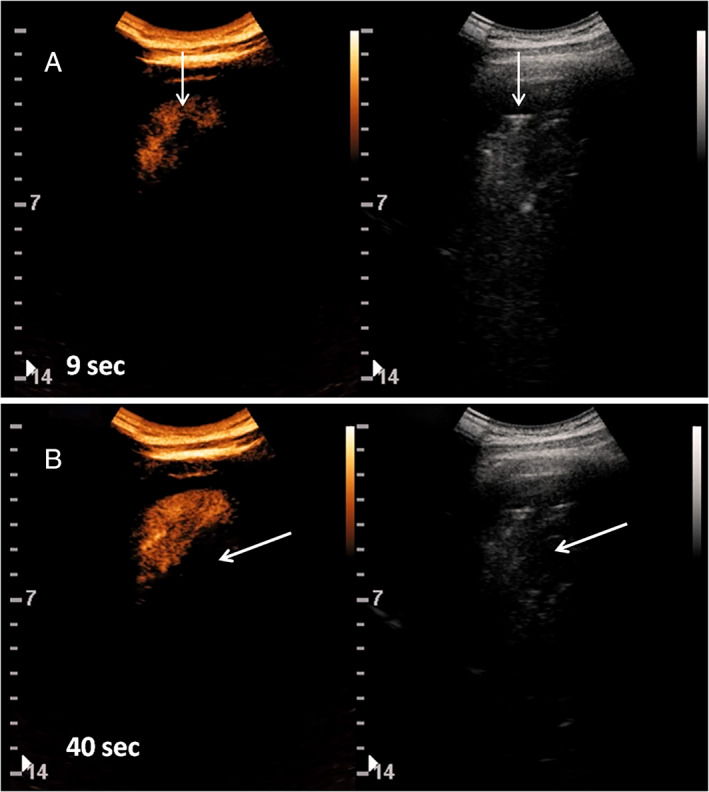
Contrast‐enhanced US (left) and grayscale B‐mode (right) images of left basal consolidation (arrows) in patient 2. A, Enhancement after 9 seconds from the injection of 2.4 mL of SonoVue. Consolidation shows partial and uneven perfusion. B, after 40 seconds, part of the coconsolidation shows a rather homogeneous parenchymal phase, whereas part of the consolidation does not show enhancement.
Figure 3.
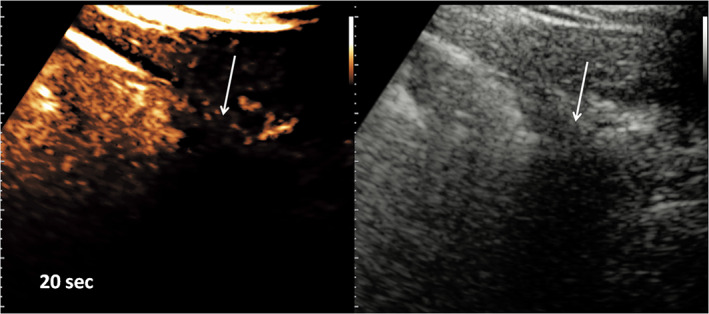
Detail of a small cuneiform subpleural consolidation (arrows). Contrast‐enhanced US (left) and grayscale B‐mode (right) images were acquired at 20 seconds from the injection of 2.4 mL of SonoVue. An obvious defect of perfusion of the lesion is shown.
Discussion
Contrast‐enhanced US imaging of the lung is proposed in the assessment of pulmonary consolidations. 7 Pneumonia shows ready and complete enhancement starting 8 seconds after the injection, with a treelike pulmonary arterial vascular pattern (Figure 1B), appearing as linear hyperechoic images with greater enhancement compared with the parenchyma. Compressive atelectasis behaves in a similar way (Figure 1E), whereas obstructive atelectasis can show avascular fluid channels (fluid bronchograms) inside homogeneous parenchymal enhancement. Embolic consolidations show absent or inhomogeneous enhancement. 10 In a case of long‐lasting atelectasis or pneumonia, perfuse patchy inhomogeneity, due to hypoxic vasoconstriction phenomena, may appear. 11
Figure 1.
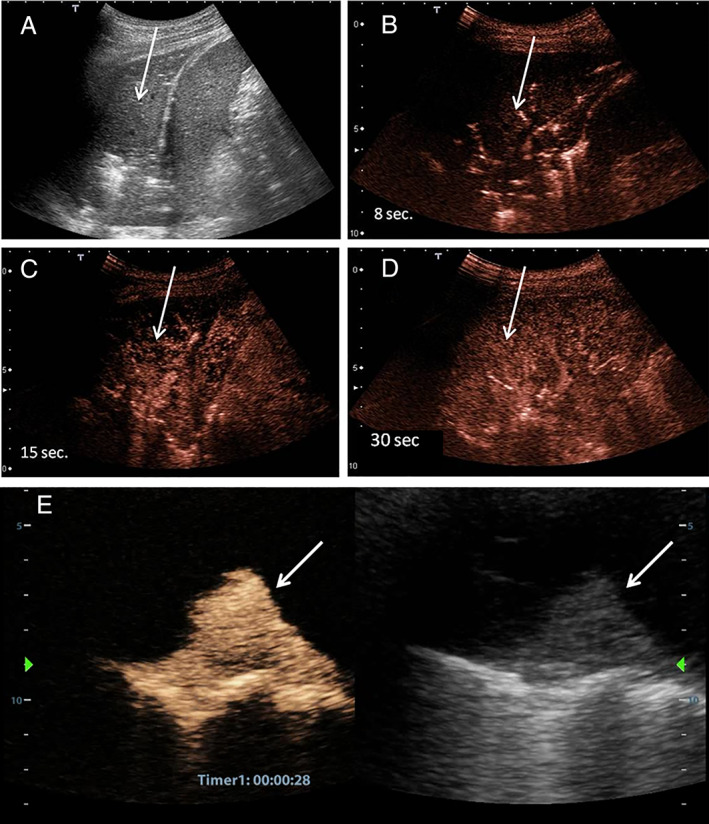
A, Ultrasound image of left basal pneumonia (arrows). The parenchyma of the lower lung lobe is consolidated. B, Contrast‐enhanced US 8 seconds from the injection of 2.4 mL of SonoVue. Arterial enhancement with a segmental appearance is shown. C, Initial parenchymal phase. D, Perfusional parenchymal phase. The whole lobe is enhanced. E, Right, Ultrasound image of compressive pulmonary atelectasis. Left, Same image at 30 seconds from the injection of 2.4 mL of SonoVue. The compressed and airless parenchyma is strongly enhanced.
Our patients showed large perfusion defects, delimited with respect to the perfused parenchyma, which often appeared only as a shell of enhanced tissue. This pattern is not typical of pneumonia and atelectasis. Inhomogeneous enhancement in the first 10 to 15 seconds was noted. The small cuneiform consolidations showed no enhancement, thus behaving as ischemic or infarct regions.
Figure 1 illustrates the perfusion stages of pneumonia and the CEUS appearance of a compressive atelectasis within a pleural effusion. Figure 2 shows some phases of the CEUS enhancement of basal macroconsolidations related to patient 2. Figure 3 shows the perfusion pattern of a small subpleural consolidation in patient 1.
Our findings may have importance in the pathogenetic interpretation of the consolidations in COVID‐19 and, consequently, in the ventilatory and medical treatments of patients with COVID‐19.
Among patients with COVID‐19 studied in Wuhan, China, about 15% progressed to severe cases. 12 Therefore, an important question is why only a subgroup of infected patients develop severe pulmonary impairment and others not. It is conceivable that in many patients with COVID‐19, during the initial stages of the disease, an adaptive immune response to limit the progression to severe stages is achieved. However, when the protective immune response is impaired, or, in particular, when aberrant inflammation mediated by proinflammatory macrophages, granulocytes, and cytokines is activated, secondary organ damage is possible. The cytokine release syndrome is a form of a systemic inflammatory response featured by interferons, interleukin 2, interleukin 6, and other mediators. 13 This overreaction was described in SARS, Middle East respiratory syndrome, and severe influenza (H1N1 and H5N1). The cytokine storm causes severe capillary damage and organ dysfunction. In the lung, diffuse alveolar damage, hyaline membrane formation, interstitial infiltration of inflammatory cells, fibrin exudate, and fibrotic changes were observed. Thrombosis of venules was noted in some cases, with infarction, hemorrhage, and fibrin thrombi. Systemic vasculitis was observed in one report of SARS. 14 Histopathologic findings in COVD‐19 are limited, but its features resemble those seen in SARS and Middle East respiratory syndrome. 15 , 16 Microscopic findings are described as nonspecific and include edema, pneumocyte hyperplasia, cellular infiltrates, multinucleate giant cell formation, and hemorrhagic necrosis.
Viral diseases are associated with coagulation disorders. Thrombosis, disseminated intravascular coagulation, and hemorrhage can occur. These pathologic findings and microembolism are complications described in influenza A infections. 17 Recently Tang et al 18 showed that in 183 patients with confirmed COVID‐19, the nonsurvivors had significantly higher d‐dimer and fibrin degradation product levels compared with survivors. In 2013, it was suggested that an abnormal urokinase pathway during SARS progression could contribute to more severe lung lesions. 19 Finally, COVID‐19 cases complicated by acute pulmonary embolism were described. 20 According to these reports, consolidations with perfusion defects observed in our patients could be supported, at least in part, by ischemic and necrotic changes and not only by inflammatory or atelectasis events. Moreover, small subpleural consolidations could be small embolic or thrombotic events.
Recently, Magro et al 21 examined skin and lung tissue from 5 patients with COVID‐19 who had respiratory failure and a purpuric skin rash. Histopathologic patterns were characterized by pauci‐inflammatory septal capillary injury but substantial fibrin deposition inside the capillary lumen. These vascular findings were accompanied by vascular deposits of terminal complement components (C5b9, C4d, and mannan‐binding lectin serine protease 2), suggesting systemic activation of the alternative and lectin‐based complement pathways. It is interesting to note that excessive complement activation may lead to the activation of a clotting pathway and diffuse thrombotic microangiopathy and is responsible for a massive local release of proinflammatory cytokines. 22 , 23
Although consolidations are commonly observed in patients with acute respiratory distress syndrome (ARDS), not all patients with severe respiratory failure from COVID‐19 have the characteristics of ARDS. Gattinoni et al 3 noted that patients with COVID‐19 who had respiratory failure showed severe hypoxemia associated with near‐normal respiratory system compliance: a combination rarely seen in ARDS. This pattern is seen at the beginning of the lung involvement, and it may improve, increase, or remain unchanged. It has been hypothesized that the worsening of these patients represents an evolution toward a picture similar to ARDS, characterized by low compliance and a decrease in the pulmonary gas volume. Our and other observations 22 , 23 suggest that there also may be an ulterior pattern of COVID‐19 lung involvement in which the consolidations do not represent atelectasis or easily recruitable areas but tissue with large perfusion defects. This possibility could be important for managing the ventilatory support of patients with respiratory failure. Ventilatory parameters could be rationally modulated according to the pattern of pulmonary impairment initially estimated with bedside lung US and CEUS.
Our findings require further confirmatory studies. However, if confirmed, they should lead to rethinking of the therapeutic approaches to COVID‐19, including the immunomodulatory therapy for the cytokine storm, but, above all, a direct intervention on the mechanisms of complement activation 23 and, consequently, on the coagulation/fibrinolysis cascade.
All of the authors of this article have reported no disclosures.
References
- 1. Zhu N, Zhang D, Wang W, Li X, et al. A novel coronavirus from patients with pneumonia in China. N Engl J Med 2020; 382:727–733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Huang C, Wang Y, Li X, Ren L, et al. Clinical features of patients infected with novel 2019 coronavirus in Wuhan, China. Lancet 2020; 395:497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Gattinoni L, Chiumello D, Caironi P, et al. COVID‐19 pneumonia: different respiratory tratment for different phenotypes [published online ahead of print April 14, 2020]. Intensive Care Med. 10.1007/s00134-020-06033-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Ye Z, Zhang Y, Wang Y, Huang Z, Song B. Chest CT manifestations of new coronavirus disease 2019 (COVID‐19): a pictorial review [published online ahead of print March 19, 2020]. Eur Radiol. 10.1007/s00330-020-06801-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Salehi S, Abedi A, Balakrishnan S, Gholamrezanezhad A. Coronavirus disease 2019 (COVID‐19): a systematic review of imaging findings in 919 patients. AJR Am J Roentgenol 2020; 14:1–7. [DOI] [PubMed] [Google Scholar]
- 6. Liu Q, Zhou Y, Yang ZQ. The cytokine storm of severe influenza and development of immunomodulatory therapy. Cell Mol Immunol 2016; 13:3–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Sidhu PS, Cantisani V, Dietrich CF, et al. The EFSUMB guidelines and recommendations for the clinical practice of contrast‐enhanced ultrasound (CEUS) in non‐hepatic applications: update 2017. Ultraschall Med 2018; 39:e2–e44. [DOI] [PubMed] [Google Scholar]
- 8. Peng QY, Zhang LN, Chinese Critical Care Ultrasound Study Group (CCUSG) . Findings of lung ultrasonography of novel corona virus pneumonia during the 2019–2020 epidemic [published online ahead of print March 12, 2020]. Intensive Care Med. 10.1007/s00134-020-05996-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Soldati G, Smargiassi A, Inchingolo R, et al. Is there a role for lung ultrasound during the COVID‐19 pandemic [published online ahead of print March 20, 2020]. J Ultrasound Med. https://onlinelibrary.wiley.com/doi/abs/10.1002/jum.15284 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Sartrori S, Postorivo S, Di Vece F, Ermili F, Tassinari D, Tombesi P. Contrast enhanced ultrasonography in peripheral consolidations: what's its actual role? World J Radiol 2013; 5:372–380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Caremani M, Benci A, Lapini L, et al. Contrast enhanced ultrasonography (CEUS) in peripheral lung lesions: a study of 60 cases. J Ultrasound 2018; 11:89–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Guan WJ, Ni ZH, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020; 382:1708–1720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Tisoncik JR, Korth MJ, Simmons CP, Farrar J, Martin TR, Katze MG. Into the eye of the cytokine storm. Microbiol Mol Biol Rev 2012; 76:16–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Franks TJ, Chong PY, Chui P, et al. Lung pathology of severe acute respiratory syndrome (SARS): a study of eight autopsy cases from Singapore. Hum Pathol 2003; 34:743–748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Tian S, Hu W, Niu L, Liu H, Xu H, Xiao SY. Pulmonary pathology of early phase 2019 novel coronavirus (COVID‐19) pneumonia in two patients with lung cancer. J Thorac Oncol 2020; 15:700–704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Ng DL, Al Hosani F, Keating MK, et al. Clinicopathologic, immunohistochemical, and ultrastructural findings of a fatal case of Middle East respiratory syndrome coronavirus infection in the United Arab Emirates, April 2014. Am J Pathol 2016; 186:652–658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Yang Y, Tang H. Aberrant coagulation causes a hyper‐inflammatory response in severe influenza pneumonia. Cell Mol Immunol 2016; 13:432–442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Tang N, Dengju L, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost 2020; 18:844–847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Gralinski LE, Bankhead A, Jeng S, et al. Mechanisms of severe acute respiratory syndrome coronavirus‐induced acute lung injury. mBio 2013; 4:e00271–e00213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Xie Y, Wang X, Yang P, Zhang S. COVID‐19 complicated by acute pulmonary embolism [published online ahead of print March 16, 2020]. Radiol Cardiothorac Imaging. 10.1148/ryct.2020200067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Magro C, Mulvey JJ, Berlin D, et al. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID‐19 infection: a report of five cases [published online ahead of print April 15, 2020]. Transl Res. 10.1016/j.transl.2010.04.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Ciceri F, Beretta L, Scandroglio AM, et al. Microvascular COVID‐19 lung vessel obstructive thromboinflammatory syndrome (MicroCLOTS): an atypical acute respiratory distress syndrome working hypothesis [published online ahead of print April 15, 2020]. Crit Care Resusc. PMID32294809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Campbell CM, Kahwash R. Will complement inhibition be the new target in treating COVID‐19 [published online ahead of print April 9, 2020]. Circulation. 10.1161/CIRCULATIONAHA.120.047419 [DOI] [PubMed] [Google Scholar]


