Abstract
While the coronavirus disease 2019 (COVID-19) pandemic advances, the scientific community continues to struggle in the search for treatments. Several improvements have been made, including discovery of the clinical efficacy of chloroquine (CQ) in patients with COVID-19, but effective treatment protocols remain elusive. In the search for novel treatment options, many scientists have used the in-silico approach to identify compounds that could interfere with the key molecules involved in entrance, replication or dissemination of severe acute respiratory syndrome coronavirus-2. However, most of the identified molecules are not available as pharmacological agents at present, and assessment of their safety and efficacy could take many months. This review took a different approach based on the proposed pharmacodynamic model of CQ in COVID-19. The main mechanism of action responsible for the favourable outcome of patients with COVID-19 treated with CQ seems to be related to a pH-modulation-mediated effect on endolysosomal trafficking, a characteristic of chemical compounds often called ‘lysosomotropic agents’ because of the physico-chemical properties that enable them to diffuse passively through the endosomal membrane and undergo protonation-based trapping in the lumen of the acidic vesicles. This review discusses lysosomotropic and lysosome targeting drugs that are already in clinical use and are characterized by good safety profiles, low cost and wide availability. Some of these drugs –particularly azithromycin and other macrolides, indomethacin and some other non-steroidal anti-inflammatory drugs, proton pump inhibitors and fluoxetine – could provide additional therapeutic benefits in addition to the potential antiviral effect that is still to be confirmed by well-controlled clinical trials. As some of these drugs have probably been used empirically in the treatment of COVID-19, it is hoped that colleagues worldwide will publish patient data to enable evaluation of the potential efficacy of these agents in the clinical context, and rapid implementation in therapeutic protocols if they are shown to have a beneficial effect on clinical outcome.
Keywords: COVID-19, Lysosomotropic agents, Endosome, Antiviral, Drug repurposing
1. Introduction
The main objective of all viruses is to hijack the intracellular machinery of the host cell for genome replication and assembly. To achieve this, the virus first has to bypass the cellular membrane barrier. The structure of some viruses enables direct fusion at the plasma membrane after recognition of the binding site [1]. However, most enveloped viruses depend on the endocytic pathway prior to fusion with the host cell [2]. Once endocytosis has been initiated, survival and replication of the virus depend on successful delivery of the viral nucleocapsid into the correct intracellular department, as different viral families target distinct compartments as primary replication sites [3]. Moreover, in order to survive, the virus has to escape from the endosomal compartment silently, without triggering the cellular defence mechanisms [4]. As a result, viral evolution involves highly complex escape strategies relying on different cues of endosomal maturation and trafficking [5]. Establishment of an acidic endosomal lumen is the key requirement for normal functioning of the endocytic pathway [6], so pH sensing has evolved as one of the fundamental strategies used by viruses to monitor endosomal maturation [7]. Two important sensing strategies are used by most viruses for induction of membrane fusion mechanisms: direct pH sensing mediated by a conformational change of the envelope glycoproteins, and endosomal protease-mediated proteolytic cleavage [7]. From a pharmacological perspective, this makes endosomal pH an attractive drug target. Neutralization of endosomal pH obstructs endosomal trafficking, impedes sorting mechanisms, and inhibits activity of the endosomal proteases [6]. As a consequence, pharmacological modulation of endolysosomal pH could prevent viral fusion and impair the formation of viral replication complexes. Moreover, as pH modulation is important for regulation of exocytosis, vesicular pH modulation could also affect viral dissemination [8]. Endosomal trafficking is necessary for normal cellular functioning, so long-term disruption of endosomal function could have considerable pathophysiological consequences [9]. Moreover, the infectivity of some viruses that use alternative strategies to enter the cell could be increased by pH-mediated inhibition of the endosomal pathway [10]. For this reason, most antiviral drug development strategies have focused on more specific fusion mechanisms rather than general inhibition of endocytosis. However, short-term inhibition of endocytosis could be a viable strategy for the management of viral infections with no alternative treatment options, such as coronavirus disease 2019 (COVID-19). Recent evidence suggests that chloroquine (CQ) and hydroxychloroquine (HCQ) may be effective therapeutic strategies for COVID-19 [11], [12], [13], [14]. Although the exact mechanism of action responsible for the antiviral activity of CQ and HCQ remains unclear, the elevation of lysosomal pH seems to play a key role [14]. This is in concordance with previous findings, as proteolytic processing of glycoprotein S by lysosomal proteases seems to be necessary for the fusion of coronavirus with the host cell [2]. Moreover, treatment of cells with glycoprotein S – the main protein responsible for viral fusion of severe acute respiratory syndrome coronavirus (SARS-CoV) and severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) – resulted in translocation of the main viral receptor, angiotensin-converting enzyme 2 (ACE-2), to endosomal vesicles [15]. Furthermore, the same effect was demonstrated for spike-bearing pseudoviruses, emphasizing the importance of glycoprotein S for viral fusion [15]. It is important to mention that although many groups have demonstrated that SARS-CoV enters the cells by receptor-mediated, pH-sensitive endocytosis [15], some groups have reported that viral fusion might also occur by pH-independent direct fusion with the plasma membrane [16]. Although the exact mechanism responsible for the entrance of SARS-CoV-2 into the host cell remains to be elucidated, the authors believe that data from clinical trials on CQ and HCQ support the importance of pH-dependent endocytosis, and that other widely available pharmacological agents with lysosomotropic properties may also demonstrate clinical effectiveness in the treatment of COVID-19. This review discusses commonly used pharmacological agents in the context of their lysosomotropic effects and pH-modulation-mediated interaction with cytoplasmic vesicle trafficking (Fig. 1 .). Although almost 5 million people had been diagnosed with COVID-19 at the time of writing (20th May 2020), organized clinical data that would facilitate the evaluation of potential antiviral effects of the drugs discussed below on SARS-CoV-2 are unavailable at present. Colleagues in possession of such data are urged to evaluate the possible benefits of these drugs, as they might offer new hope in terms of prevention and/or treatment of this devastating disease.
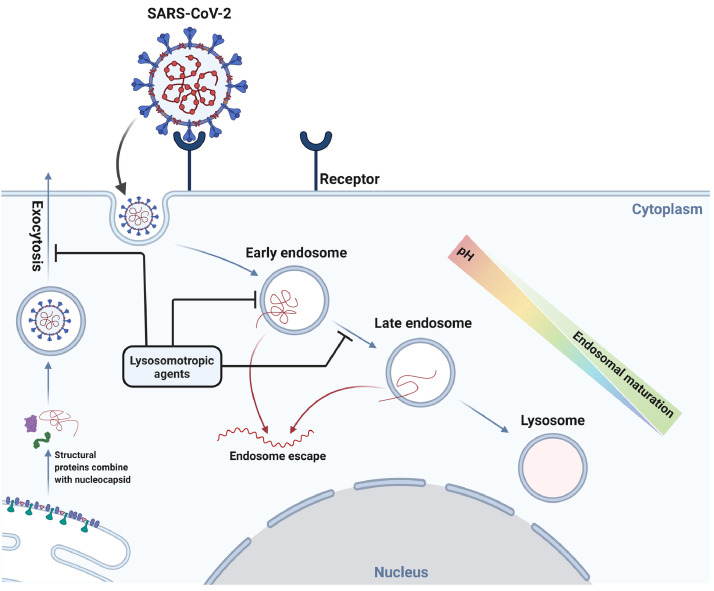
Fig. 1.
Possible interaction of lysosomotropic agents and lysosome targeting drugs with the replication cycle of severe acute respiratory syndrome corovavirus-2 (SARS-CoV-2) in a cell. Lysosome targeting drugs and lysosomotropic drugs, such as chloroquine, have the ability to interfere with endosomal trafficking, affecting both endosomal maturation and exocytotic pathways. Taking into consideration the dependence of viral replication on the normal functioning of endocytic trafficking, commonly used pharmacological agents that demonstrate lysosomotropic properties should be examined as a possible therapeutic option for the treatment of coronavirus disease 2019.
2. A brief introduction to the concept of lysosomotropism in pharmacology
The concept of lysosomotropism was first introduced by De Duve et al. in 1974 [17]. The term was originally proposed for all substances taken up by lysosomes, regardless of their chemical structure or mechanism of action. Interestingly, the importance of the potential antiviral properties of lysosomotropic agents was emphasized in the original publication [17]. Moreover, some of the original concepts of lysosomotropism, such as the possibility to make virtually any substance lysosomotropic by suitable coupling with an appropriate chemical carrier, are now considered to be common principles in drug design. In general, most substances with weakly basic and lipophilic properties are believed to demonstrate lysosomotropism to some extent. These properties enable lysosomotropic drugs to diffuse passively through the endosomal membrane and undergo protonation-based trapping in the lumen of the acidic vesicle. From a chemical standpoint, most drugs that demonstrate lysosomotropic properties are cationic substances that usually belong to the group of primary, secondary or tertiary amines; from a pharmacological point of view, these compounds are often described by very large volumes of distribution and long residual effects [18]. Moreover, lysosomotropic agents inhibit endosomal maturation and disrupt endolysosomal trafficking, and these effects are of particular interest in the context of viral infection, as described above. The exact mechanisms by which different lysosomotropic agents induce these changes are still not well understood; however, modulation of pH and interaction with different molecular systems involved in pH regulation of endolysosomal vesicles seem to be the most important. For deeper understanding of the current concept of lysosomotropism, the reader is referred to the informative review by Marceau et al. [18]. Antimalarial drugs, such as CQ, which have demonstrated clinical efficacy in the treatment of COVID-19 [11] are often referred to as classic lysosomotropic agents because their primary mechanism of action relies on their lysosomotropic properties. CQ works by sequestration in digestive vacuoles that resemble endolysosomal compartments, and increases their pH to stop the nutrient supply [18]. Another interesting example of lysosomotropic pharmaceuticals are drugs used for the treatment of tuberculosis, where the drug has to reach both extracellular bacilli in the interstitial compartments and caseum, and intracellular pathogens residing in phagolysosomes of immune cells [18,19]. Consequently, retention of antituberculosis drugs in infected phagolysosomes has been considered an important factor in maximization of their efficiency [19], and this property has been investigated actively in the course of development of novel antituberculosis agents [20,21]. The importance of phagolysosomal sequestration of lysosomotropic agents in the context of Mycobacterium tuberculosis infection is also reflected by the fact that lysosomal neutralization with bafilomycin A or ammonium chloride inhibits the tuberculostatic effect of some drugs [22]. Interestingly, moderate antituberculosis activity has been shown in vitro for some standard lysosomotropic drugs, such as fluoxetine and sertraline, and this effect was absent in their non-protonable analogues that cannot undergo ion trapping [22]. However, apart from drugs that are well-known lysosomotropic substances, many pharmacological agents from other therapeutic classes belong to this group of chemicals based on both their physico-chemical properties and their effect on endolysosomal function in vitro and in vivo. It is suggested that these drugs should be examined in the context of their potential use in COVID-19 treatment protocols, as most of them could potentially be used to target both viral replication and dissemination and symptoms of the disease. Moreover, some drugs can produce similar effects on the endolysosomal pathway by targeting endosomal compartment pH through different mechanisms such as inhibition of vacuolar-type H+-ATPase (V-ATPase). In the following text, these compounds are considered as potential candidate drugs with antiviral effects (Fig. 2 ). As many pharmacological agents have shown endolysosomal pH-modulating activity both in vitro and in vivo, this review focused solely on drugs that satisfied several criteria that are considered important in this context: existing evidence of endolysosomal pH modulation, good safety profile, wide availability, low cost, and additional therapeutic benefits apart from the potential preventive and therapeutic antiviral effect (Table 1 ).
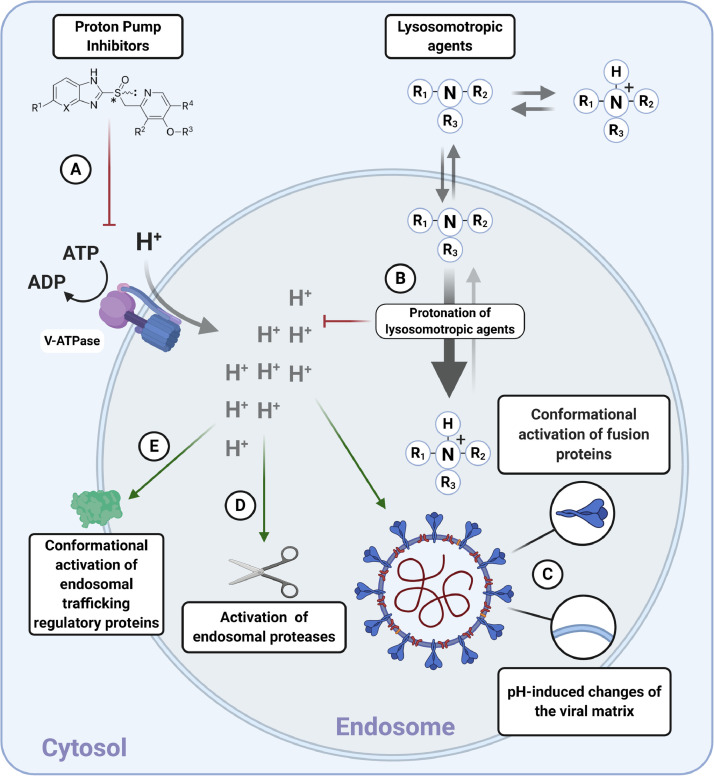
Fig. 2.
Possible mechanisms of endolysosomal pH-modulation-mediated effects on the structure and function of endolysosomal vesicles with potential consequences for viral fusion, trafficking and shedding. (A) Drugs affecting activity of vesicular acidification mechanisms neutralize endolysosomal compartments. The illustration shows the general structure of a proton pump inhibitor; this might neutralize endolysosomal compartments through inhibition of vacuolar-type H+-ATPase (V-ATPase). (B) Weakly basic and lipophilic properties of lysosomotropic compounds enable them to diffuse passively through the endosomal membrane and undergo protonation-based trapping and subsequent neutralization of acidic vesicles. (C) The acidic environment enables conformational changes that activate viral fusion proteins. Moreover, H+ interacts with the viral matrix and activates mechanisms implicated in later stages of viral fusion and/or uncoating. (D) Acidic pH of endolysosomal compartments activates proteases involved with viral fusion processes. (E) The acidic luminal environment activates endolysosomal trafficking regulatory proteins [56].
Table 1.
Drugs used in coronavirus disease 2019 (COVID-19) clinical trials with potential endolysosomal pH-mediated effect on severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). Clinicaltrials.gov was searched for COVID-19, and pharmaceutical intervention data were analysed on 20th May 2020. Both observational and interventional trials were included in the table.
| Drug Current ongoing clinical trials (COVID-19) [57] | Physico-chemical properties [58] |
Endolysosomal neutralization reported in the literature | Antiviral activity reported in the literature | Adverse effects | ||
|---|---|---|---|---|---|---|
| Partition coefficient (logP) | Acid dissociation constant (pKa) | |||||
| Macrolide antibiotics | ||||||
| Azithromycin | NCT04359095, NCT04359316, NCT04329832, NCT04390594, NCT04359953, NCT04381962, NCT04334382, NCT04344444, NCT04332094, NCT04370782, NCT04329572, NCT04363060, NCT04332107, NCT04363203, NCT04358068, NCT04336332, NCT04365231, NCT04371406, NCT04286503, NCT04339816, NCT04347512, NCT04395768, NCT04339426, NCT04328272, NCT04354428, NCT04348474, NCT04341870, NCT04392128, NCT04343092, NCT04349410, NCT04374552, NCT04341207, NCT04351919, NCT04334512, NCT04335552, NCT04338698, NCT04374019, NCT04371107, NCT04344379, NCT04361461, NCT04366089, NCT04381936, NCT04373733, NCT04368351, NCT04380818, NCT04394182, NCT04345861, NCT04349592, NCT04324463, NCT04382846, NCT04365582, NCT04322123, NCT04322396, NCT04321278, NCT04369365, NCT04383717, NCT04347031, NCT04390152, NCT04355052, NCT04341727, NCT04362189, NCT04374903, [Macrolide (NCT02735707)] | 2.44 | Strongest acidic: 12.43 Strongest basic: 9.57 |
Yes [29,59,60] | Yes [35,61] | Nausea, vomiting, abdominal pain, diarrhoea, allergy, antibiotic-associated colitis, cholestatic jaundice, prolongation of the QT interval [62] |
| Non-steroidal anti-inflammatory drugs | ||||||
| Naproxen | NCT04325633 | 2.99 | Strongest acidic: 4.19 Strongest basic: -4.8 |
None | Yes [63,64] | Gastrointestinal toxicity, renal impairment, increased risk of cardiovascular events, hypersensitivity reactions, fluid retention [62] |
| Ibuprofen |
NCT04334629, NCT04382768 |
3.84 | Strongest acidic: 4.85 | None | Yes [65] | |
| Indomethacin | NCT04344457 | 3.53 | Strongest acidic: 3.79 Strongest basic: -2.9 |
Yes [42] | Yes [43,44,66] | |
| Proton pump inhibitors | ||||||
| Omeprazole | NCT04333407 | 2.43 | Strongest acidic: 9.29 Strongest basic: 4.77 |
Yes [67] | Yes [68] | Gastrointestinal disturbances, headache, hypomagnesaemia [62] |
| Lansoprazole | NCT04325633 | 3.03 | Strongest acidic: 9.35 Strongest basic: 4.16 |
Yes [69] | Yes [70,71] | |
| Selective serotonin re-uptake inhibitors | ||||||
| Fluoxetine | NCT04377308 | 4.17 | Strongest basic: 9.8 | Yes [72], [73], [74] | Yes [75,76] | Gastrointestinal upset, appetite and weight changes, hypersensitivity reactions, hyponatraemia, suicidal thoughts, lower seizure threshold, prolongation of the QT interval, increased risk of bleeding, increased risk of serotonin syndrome [62] |
| Fluvoxamine |
NCT04383886, NCT04342663 |
2.8 | Strongest basic: 8.86 | Yes [73] | None | |
3. Antibiotics
Initial reports showed that more than 90% of patients with COVID-19 received antibiotics [24]; however, reports on how patient outcome depends on the use of different antibiotics are scarce. Several groups of antibiotics are known to be lysosomotropic; however, the effect is observed most consistently for macrolides. Therefore, this review will focus on macrolides as a potentially effective group of antibiotics in cases of COVID-19, as numerous studies have shown that macrolides possess anti-inflammatory, antibacterial and antiviral properties. The anti-inflammatory effects [25] are of particular interest, as it has been suggested that a cytokine storm plays a central role in the development of acute respiratory distress syndrome following measurement of a high concentration of pro-inflammatory interleukins in the blood of deceased patients [26]. Another reason to consider macrolides is their antiviral effect, particularly for, but not limited to, clarithromycin, bafilomycin, erythromycin and azithromycin [27,28]. Although the effects of macrolides on the expression of cytokines and membrane receptors contribute to the antiviral effects, the exact mechanism remains unknown [27,28]. As several authors have reported that macrolides can affect lysosomal trafficking and lysosomal pH [29,30], it is hypothesized that this could be one of several mechanisms of antiviral activity of macrolides, and is particularly interesting in COVID-19. Although the antiviral effects of macrolides have been known for a long time, the use of these drugs in patients has not yielded groundbreaking results [27,31]. Although early clinical findings suggested the effectiveness of azithromycin in combination with HCQ [32], some recent observations have challenged the original conclusions [33,34]. Thus, well-controlled, randomized, large-scale clinical trials are needed to draw conclusions regarding the use of azithromycin for COVID-19 [34,35]. Hopefully, the 60 ongoing clinical trials listed on clinicaltrials.gov with azithromycin included in the interventional design will provide sufficient evidence to settle the ongoing debate on its potential effectiveness [23]. Apart from the potential antiviral and anti-inflammatory potential, other very important properties of macrolides should be mentioned, namely their ability to inhibit the proliferation of fibroblasts, production of collagen, and release of matrix protease and pro-inflammatory cytokines [36]. These are particularly interesting and important properties of macrolides in the context of long-term consequences, as it has been debated that COVID-19 may cause lung fibrosis [37]. Considering everything mentioned above and the recent in-silico finding that clarithromycin and erythromycin could have a direct antiviral effect by interfering with COVID-19 protease [38], macrolides should be investigated further as a treatment option, including the route of administration, with inhalatory macrolides being of particular interest. Unfortunately, inhalatory macrolides are not available for clinical use at present. The physico-chemical properties of these drugs suggest that they are suitable for aerosolization, and in-vivo experiments have demonstrated that inhalation leads to a rapid increase in concentration in alveolar macrophages and airway epithelial cells in the case of azithromycin [36].
4. Non-steroidal anti-inflammatory drugs
The safety of non-steroidal anti-inflammatory drugs (NSAIDs) in patients with COVID-19 has been debated since the discovery that ACE-2 is an entry receptor for SARS-CoV-2 [39]. Fang et al. hypothesized that ibuprofen increases the risk of developing severe and fatal COVID-19 as it is known that it can increase the expression of ACE-2 receptors [40]. However, this has not been supported by any clinical study, and cannot be confirmed post hoc given the common practice of not making raw patient data available. Nevertheless, taking into account the above-mentioned relationship between endosomal trafficking and viral replication, NSAIDs could provide additional benefits in the treatment of COVID-19 as a result of their inhibitory effect on autophagic flux [41]. Indomethacin is particularly interesting in this context. Several studies have shown that indomethacin can increase the pH of lysosomes [41,42]. Moreover, some results have suggested that indomethacin can block viral RNA synthesis independently of its effect on cyclo-oxygenase inhibition, and this effect was confirmed both in vitro and in vivo on dogs infected with canine coronavirus [43,44]. Furthermore, it has been shown that indomethacin enhances the inhibitory effect of CQ on autophagy, suggesting that NSAIDs and CQ could have a synergistic effect on viral replication [42]. In conclusion, at this point, there are not enough studies to demystify the potential role of NSAIDs in COVID-19; however, in addition to standard anti-inflammatory and antipyretic properties, other factors should be taken into account, such as the possible effect of NSAIDs on the expression of ACE-2, interference with autophagic flux and possible direct antiviral activity. The authors believe that more attention should be directed towards these potential effects, and structured clinical data should be collected in order to examine if some NSAIDs should be considered better than others for this indication.
5. Proton pump inhibitors
To alleviate the side-effects of NSAIDs, proton pump inhibitors (PPIs) are often given concomitantly. It is important to note that PPIs can inhibit V-ATP-ase, the enzyme responsible for maintaining pH in endosomes. As a consequence, PPIs can induce cytosolic acidification and lysosomal and endosomal alkalinization [45,46]. As discussed above, this effect could be beneficial in patients with COVID-19. In the context of the direct and indirect antiviral effects of indomethacin discussed above, and its well-known gastrointestinal side-effects, a combination of indomethacin and PPIs should be investigated further as a potential therapeutic strategy in COVID-19. Additionally, ACE-2, which acts as a primary receptor for SARS-CoV-2 host cell entry, is a well-known intestinal transporter involved in amino acid absorption throughout the human small intestine [47], and gastrointestinal symptoms are reported to be part of the clinical presentation of COVID-19 [48,49]. Therefore, targeting the gastrointestinal system could have a dual benefit as lysosomotropic agents could provide symptomatic relief and reduce viral dissemination. The possibility that the faecal-oral transmission route is involved in viral spread, in combination with the recent finding that viral RNA remained positive in faeces even after negative conversion of viral RNA in the respiratory system, further supports the idea that lysosomotropic agents that primarily target the gastrointestinal system should be considered as part of the treatment of COVID-19 [48].
6. Selective serotonin re-uptake inhibitors
Many central nervous system drugs have lysosomotropic properties. Some lysosomotropic compounds used in experimental work on endolysosomal trafficking and function include the tricyclic antidepressants, imipramine and amitriptyline; the antipsychotic, chlorpromazine; and the selective serotonin re-uptake inhibitor, fluoxetine [50]. If further research provides clinical evidence for the antiviral effect of fluoxetine, it could be considered as part of the solution in addition to virtual psychotherapy for quarantine-induced anxiety. This might be more important than it seems, as findings published after the previous SARS epidemic suggest that the psychological effects of quarantine are underestimated [51], [52], [53], and the rapid response of psychologists and psychiatrists during the ongoing pandemic confirms that early intervention is important [54,55]. Moreover, maximal compliance is needed to implement successful quarantine and social isolation measures, and considering the information in the global media, anxiolysis is more important than ever in order to stop viral spread.
7. Conclusion
In conclusion, many pharmacological agents that are widely used in clinical practice demonstrate endolysosomal pH-modulating properties both in vitro and in vivo, and should thus be considered as potential therapeutic agents for COVID-19 in light of the recent findings on the clinical efficacy of CQ. Most of the drugs discussed in this review have good safety profiles, low cost and are widely available, which makes them interesting in the context of the rapidly advancing SARS-CoV-2 pandemic. As some of these drugs have probably been used empirically in the treatment of COVID-19, it is hoped that colleagues worldwide will publish patient data to enable evaluation of the potential efficacy of these agents. As effective treatment strategies for COVID-19 are still lacking, repurposing of drugs based on their lysosomotropic and endolysosomal pH-modulating effects could provide the additional preventive and therapeutic options that are desperately needed.
Acknowledgements
BioRender was used to create a schematic representation of the viral cycle.
Funding: None.
Competing interests: None declared.
Ethical approval: Not required.
References
- 1.Agelidis AM, Shukla D. Cell entry mechanisms of HSV: what we have learned in recent years. Fut Virol. 2015;10:1145–1154. doi: 10.2217/fvl.15.85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Burkard C, Verheije MH, Wicht O, van Kasteren SI, van Kuppeveld FJ, Haagmans BL, et al. Coronavirus cell entry occurs through the endo-/lysosomal pathway in a proteolysis-dependent manner. PLoS Pathog. 2014;10 doi: 10.1371/journal.ppat.1004502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Novoa RR, Calderita G, Arranz R, Fontana J, Granzow H, Risco C. Virus factories: associations of cell organelles for viral replication and morphogenesis. Biol Cell. 2005;97:147–172. doi: 10.1042/BC20040058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Staring J, Raaben M, Brummelkamp TR. Viral escape from endosomes and host detection at a glance. J Cell Sci. 2018;131(15):131. doi: 10.1242/jcs.216259. [DOI] [PubMed] [Google Scholar]
- 5.Marsh M, Helenius A. Virus entry: open sesame. Cell. 2006;124:729–740. doi: 10.1016/j.cell.2006.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Scott CC, Gruenberg J. Ion flux and the function of endosomes and lysosomes: pH is just the start: the flux of ions across endosomal membranes influences endosome function not only through regulation of the luminal pH. Bioessays. 2011;33:103–110. doi: 10.1002/bies.201000108. [DOI] [PubMed] [Google Scholar]
- 7.Mazzon M, Marsh M. Targeting viral entry as a strategy for broad-spectrum antivirals. F1000Res. 2019;8:1628. doi: 10.12688/f1000research.19694.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Marshansky V, Futai M. The V-type H+-ATPase in vesicular trafficking: targeting, regulation and function. Curr Opin Cell Biol. 2008;20:415–426. doi: 10.1016/j.ceb.2008.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hu Y-B, Dammer EB, Ren R-J, Wang G. The endosomal–lysosomal system: from acidification and cargo sorting to neurodegeneration. Transl Neurodegener. 2015;4:18. doi: 10.1186/s40035-015-0041-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fredericksen BL, Wei BL, Yao J, Luo T, Garcia JV. Inhibition of endosomal/lysosomal degradation increases the infectivity of human immunodeficiency virus. J Virol. 2002;76:11440–11446. doi: 10.1128/JVI.76.22.11440-11446.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gao J, Tian Z, Yang X. Breakthrough: chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies. Biosci Trends. 2020;14:72–73. doi: 10.5582/bst.2020.01047. [DOI] [PubMed] [Google Scholar]
- 12.Cortegiani A, Ingoglia G, Ippolito M, Giarratano A, Einav S. A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J Crit Care. 2020;57:279–283. doi: 10.1016/j.jcrc.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dong L, Hu S, Gao J. Discovering drugs to treat coronavirus disease 2019 (COVID-19) Drug Discov Ther. 2020;14:58–60. doi: 10.5582/ddt.2020.01012. [DOI] [PubMed] [Google Scholar]
- 14.Liu J, Cao R, Xu M, Wang X, Zhang H, Hu H, et al. Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro. Cell Discov. 2020;6:269. doi: 10.1038/s41421-020-0156-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wang H, Yang P, Liu K, Guo F, Zhang Y, Zhang G, et al. SARS coronavirus entry into host cells through a novel clathrin- and caveolae-independent endocytic pathway. Cell Res. 2008;18:290–301. doi: 10.1038/cr.2008.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Simmons G, Reeves JD, Rennekamp AJ, Amberg SM, Piefer AJ, Bates P. Characterization of severe acute respiratory syndrome-associated coronavirus (SARS-CoV) spike glycoprotein-mediated viral entry. Proc Natl Acad Sci USA. 2004;101:4240–4245. doi: 10.1073/pnas.0306446101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.De Duve C, De Barsy T, Poole B, Trouet A, Tulkens P, Van Hoof F. Lysosomotropic agents. Biochem Pharmacol. 1974;23:2495–2531. doi: 10.1016/0006-2952(74)90174-9. [DOI] [PubMed] [Google Scholar]
- 18.Marceau F, Bawolak M-T, Lodge R, Bouthillier J, Gagné-Henley A, Gaudreault RC, et al. Cation trapping by cellular acidic compartments: beyond the concept of lysosomotropic drugs. Toxicol Appl Pharmacol. 2012;259:1–12. doi: 10.1016/j.taap.2011.12.004. [DOI] [PubMed] [Google Scholar]
- 19.Dartois V. The path of anti-tuberculosis drugs: from blood to lesions to mycobacterial cells. Nat Rev Microbiol. 2014;12:159–167. doi: 10.1038/nrmicro3200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jia L, Tomaszewski JE, Hanrahan C, Coward L, Noker P, Gorman G, et al. Pharmacodynamics and pharmacokinetics of SQ109, a new diamine-based antitubercular drug. Br J Pharmacol. 2005;144:80–87. doi: 10.1038/sj.bjp.0705984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Shkurupii VA, Kozyaev MA, Nadeev AP. Ultrastructural characteristics of type A epithelioid cells during BCG-granulomatosis and treatment with lysosomotropic isoniazid. Bull Exp Biol Med. 2006;141:487–490. doi: 10.1007/s10517-006-0206-0. [DOI] [PubMed] [Google Scholar]
- 22.Schump MD, Fox DM, Bertozzi CR, Riley LW. Subcellular partitioning and intramacrophage selectivity of antimicrobial compounds against Mycobacterium tuberculosis. Antimicrob Agents Chemother. 2017;61(3) doi: 10.1128/AAC.01639-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.ClinicalTrials.gov. Available at:https://clinicaltrials.gov/[last accessed 8 June 2020].
- 24.Lai C-C, Shih T-P, Ko W-C, Tang H-J, Hsueh P-R. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): the epidemic and the challenges. Int J Antimicrob Agents. 2020;55 doi: 10.1016/j.ijantimicag.2020.105924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zarogoulidis P, Papanas N, Kioumis I, Chatzaki E, Maltezos E, Zarogoulidis K. Macrolides: from in vitro anti-inflammatory and immunomodulatory properties to clinical practice in respiratory diseases. Eur J Clin Pharmacol. 2012;68:479–503. doi: 10.1007/s00228-011-1161-x. [DOI] [PubMed] [Google Scholar]
- 26.Ruan Q, Yang K, Wang W, Jiang L, Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46:846–848. doi: 10.1007/s00134-020-05991-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Min J-Y, Jang YJ. Macrolide therapy in respiratory viral infections. Mediators Inflamm. 2012;2012 doi: 10.1155/2012/649570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Asada M, Yoshida M, Suzuki T, Hatachi Y, Sasaki T, Yasuda H, et al. Macrolide antibiotics inhibit respiratory syncytial virus infection in human airway epithelial cells. Antiviral Res. 2009;83:191–200. doi: 10.1016/j.antiviral.2009.05.003. [DOI] [PubMed] [Google Scholar]
- 29.Renna M, Schaffner C, Brown K, Shang S, Tamayo MH, Hegyi K, et al. Azithromycin blocks autophagy and may predispose cystic fibrosis patients to mycobacterial infection. J Clin Invest. 2011;121:3554–3563. doi: 10.1172/JCI46095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Huss M, Wieczorek H. Inhibitors of V-ATPases: old and new players. J Exp Biol. 2009;212:341–346. doi: 10.1242/jeb.024067. [DOI] [PubMed] [Google Scholar]
- 31.Wong EHC, Porter JD, Edwards MR, Johnston SL. The role of macrolides in asthma: current evidence and future directions. Lancet Respir Med. 2014;2:657–670. doi: 10.1016/S2213-2600(14)70107-9. [DOI] [PubMed] [Google Scholar]
- 32.Gautret P, Lagier J-C, Parola P, Hoang VT, Meddeb L, Mailhe M, et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents2020:105949. [DOI] [PMC free article] [PubMed] [Retracted]
- 33.Molina JM, Delaugerre C, Le Goff J, Mela-Lima B, Ponscarme D, Goldwirt L, et al. No evidence of rapid antiviral clearance or clinical benefit with the combination of hydroxychloroquine and azithromycin in patients with severe COVID-19 infection. Med Mal Infect. 2020;50:384. doi: 10.1016/j.medmal.2020.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Geleris J, Sun Y, Platt J, Zucker J, Baldwin M, Hripcsak G, et al. Observational study of hydroxychloroquine in hospitalized patients with Covid-19. N Engl J Med. 2020 doi: 10.1056/NEJMoa2012410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Damle B, Vourvahis M, Wang E, Leaney J, Corrigan B. Clinical pharmacology perspectives on the antiviral activity of azithromycin and use in COVID-19. Clin Pharmacol Ther. 2020 doi: 10.1002/cpt.1857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Siekmeier R, Hofmann T, Scheuch G. Inhalation of macrolides: a novel approach to treatment of pulmonary infections. Adv Exp Med Biol. 2015;839:13–24. doi: 10.1007/5584_2014_50. [DOI] [PubMed] [Google Scholar]
- 37.Shi H, Han X, Jiang N, Cao Y, Alwalid O, Gu J, et al. Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: a descriptive study. Lancet Infect Dis. 2020;20(4):425–434. doi: 10.1016/S1473-3099(20)30086-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Dayer MR. Old drugs for newly emerging viral disease, COVID-19: bioinformatic prospective. arXiv:2003.04524.
- 39.Zhang H, Penninger JM, Li Y, Zhong N, Slutsky AS. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020;46:586–590. doi: 10.1007/s00134-020-05985-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fang L, Karakiulakis G, Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir Med. 2020;8(4):e21. doi: 10.1016/S2213-2600(20)30116-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chamoun-Emanuelli AM, Bryan LK, Cohen ND, Tetrault TL, Szule JA, Barhoumi R, et al. NSAIDs disrupt intestinal homeostasis by suppressing macroautophagy in intestinal epithelial cells. Sci Rep. 2019;9:1–15. doi: 10.1038/s41598-019-51067-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Vallecillo-Hernández J, Barrachina MD, Ortiz-Masiá D, Coll S, Esplugues JV, Calatayud S, et al. Indomethacin disrupts autophagic flux by inducing lysosomal dysfunction in gastric cancer cells and increases their sensitivity to cytotoxic drugs. Sci Rep. 2018;8:1–10. doi: 10.1038/s41598-018-21455-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Amici C, Di Caro A, Ciucci A, Chiappa L, Castilletti C, Martella V, et al. Indomethacin has a potent antiviral activity against SARS coronavirus. Antivir Ther. 2006;11:1021–1030. [PubMed] [Google Scholar]
- 44.Amici C, La Frazia S, Brunelli C, Balsamo M, Angelini M, Santoro MG. Inhibition of viral protein translation by indomethacin in vesicular stomatitis virus infection: role of eIF2α kinase PKR. Cell Microbiol. 2015;17:1391–1404. doi: 10.1111/cmi.12446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lee Y-Y, Jeon H-K, Hong JE, Cho YJ, Ryu JY, Choi J-J, et al. Proton pump inhibitors enhance the effects of cytotoxic agents in chemoresistant epithelial ovarian carcinoma. Oncotarget. 2015;6:35040–35050. doi: 10.18632/oncotarget.5319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lu Z-N, Tian B, Guo X-L. Repositioning of proton pump inhibitors in cancer therapy. Cancer Chemother Pharmacol. 2017;80:925–937. doi: 10.1007/s00280-017-3426-2. [DOI] [PubMed] [Google Scholar]
- 47.Vuille-dit-Bille RN, Camargo SM, Emmenegger L, Sasse T, Kummer E, Jando J, et al. Human intestine luminal ACE2 and amino acid transporter expression increased by ACE-inhibitors. Amino Acids. 2015;47:693–705. doi: 10.1007/s00726-014-1889-6. [DOI] [PubMed] [Google Scholar]
- 48.Xiao F, Tang M, Zheng X, Liu Y, Li X, Shan H. Evidence for gastrointestinal infection of SARS-CoV-2. Gastroenterology. 2020;158(6):1831–1833.e3. doi: 10.1053/j.gastro.2020.02.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Song Y, Liu P, Shi XL, Chu YL, Zhang J, Xia J, et al. SARS-CoV-2 induced diarrhoea as onset symptom in patient with COVID-19. Gut. 2020;69(6):1143–1144. doi: 10.1136/gutjnl-2020-320891. [DOI] [PubMed] [Google Scholar]
- 50.Lu S, Sung T, Lin N, Abraham RT, Jessen BA. Lysosomal adaptation: how cells respond to lysosomotropic compounds. PLoS One. 2017;12 doi: 10.1371/journal.pone.0173771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Hawryluck L, Gold WL, Robinson S, Pogorski S, Galea S, Styra R. SARS control and psychological effects of quarantine, Toronto, Canada. Emerg Infect Dis. 2004;10:1206–1212. doi: 10.3201/eid1007.030703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wu P, Fang Y, Guan Z, Fan B, Kong J, Yao Z, et al. The psychological impact of the SARS epidemic on hospital employees in China: exposure, risk perception, and altruistic acceptance of risk. Can J Psychiatry. 2009;54:302–311. doi: 10.1177/070674370905400504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Liu X, Kakade M, Fuller CJ, Fan B, Fang Y, Kong J, et al. Depression after exposure to stressful events: lessons learned from the severe acute respiratory syndrome epidemic. Compr Psychiatry. 2012;53:15–23. doi: 10.1016/j.comppsych.2011.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Park S-C, Park YC. Mental health care measures in response to the 2019 novel coronavirus outbreak in Korea. Psychiatry Investig. 2020;17:85–86. doi: 10.30773/pi.2020.0058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Xiao C. A novel approach of consultation on 2019 novel coronavirus (COVID-19)-related psychological and mental problems: structured letter therapy. Psychiatry Investig. 2020;17:175–176. doi: 10.30773/pi.2020.0047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.White JM, Whittaker GR. Fusion of enveloped viruses in endosomes. Traffic. 2016;17:593–614. doi: 10.1111/tra.12389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.ClinicalTrials.gov. Available at:https://clinicaltrials.gov/[accessed 8 June 2020].
- 58.DrugBank. Available at:https://www.drugbank.ca/[accessed 8 June 2020].
- 59.Persson HL, Vainikka LK, Sege M, Wennerström U, Dam-Larsen S, Persson J. Leaky lysosomes in lung transplant macrophages: azithromycin prevents oxidative damage. Respir Res. 2012;13:83. doi: 10.1186/1465-9921-13-83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Nujić K, Banjanac M, Munić V, Polančec D, Eraković Haber V. Impairment of lysosomal functions by azithromycin and chloroquine contributes to anti-inflammatory phenotype. Cell Immunol. 2012;279:78–86. doi: 10.1016/j.cellimm.2012.09.007. [DOI] [PubMed] [Google Scholar]
- 61.Schögler A, Kopf BS, Edwards MR, Johnston SL, Casaulta C, Kieninger E, et al. Novel antiviral properties of azithromycin in cystic fibrosis airway epithelial cells. Eur Respir J. 2015;45:428–439. doi: 10.1183/09031936.00102014. [DOI] [PubMed] [Google Scholar]
- 62.Hitchings A, Longsdale D, Burrage D, Baker E. Elsevier; Amsterdam: 2019. The top 100 drugs: clinical pharmacology and practical prescribing. [Google Scholar]
- 63.Zheng W, Fan W, Zhang S, Jiao P, Shang Y, Cui L, et al. Naproxen exhibits broad anti-influenza virus activity in mice by impeding viral nucleoprotein nuclear export. Cell Rep. 2019;27:1875–1885. doi: 10.1016/j.celrep.2019.04.053. [DOI] [PubMed] [Google Scholar]
- 64.Terrier O, Dilly S, Pizzorno A, Henri J, Berenbaum F, Lina B, et al. Broad-spectrum antiviral activity of naproxen: from influenza A to SARS-CoV-2 coronavirus. BioRxiv2020:7202.
- 65.Paessler S, Huang C, Sencanski M, Veljkovic N, Perovic V, Glisic S, et al. Ibuprofen as a template molecule for drug design against Ebola virus. Front Biosci. 2018;23:947–953. doi: 10.2741/4627. [DOI] [PubMed] [Google Scholar]
- 66.Xu T, Gao X, Wu Z, Selinger DW, Zhou Z. Indomethacin has a potent antiviral activity against SARS CoV-2 in vitro and canine coronavirus in vivo. BioRxiv2020:8.
- 67.Luciani F, Spada M, De Milito A, Molinari A, Rivoltini L, Montinaro A, et al. Effect of proton pump inhibitor pretreatment on resistance of solid tumors to cytotoxic drugs. J Natl Cancer Inst. 2004;96:1702–1713. doi: 10.1093/jnci/djh305. [DOI] [PubMed] [Google Scholar]
- 68.Long J, Wright E, Molesti E, Temperton N, Barclay W. Antiviral therapies against Ebola and other emerging viral diseases using existing medicines that block virus entry. F1000Res. 2015;4:30. doi: 10.12688/f1000research.6085.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Yu M, Lee C, Wang M, Tannock IF. Influence of the proton pump inhibitor lansoprazole on distribution and activity of doxorubicin in solid tumors. Cancer Sci. 2015;106:1438–1447. doi: 10.1111/cas.12756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Sasaki T, Yamaya M, Yasuda H, Inoue D, Yamada M, Kubo H, et al. The proton pump inhibitor lansoprazole inhibits rhinovirus infection in cultured human tracheal epithelial cells. Eur J Pharmacol. 2005;509:201–210. doi: 10.1016/j.ejphar.2004.12.042. [DOI] [PubMed] [Google Scholar]
- 71.Sasaki T, Nakayama K, Yasuda H, Yamaya M. A new strategy with proton pump inhibitors for the prevention of acute exacerbations in COPD. Ther Adv Respir Dis. 2011;5:91–103. doi: 10.1177/1753465810392264. [DOI] [PubMed] [Google Scholar]
- 72.Nadanaciva S, Lu S, Gebhard DF, Jessen BA, Pennie WD, Will Y. A high content screening assay for identifying lysosomotropic compounds. Toxicol In Vitro. 2011;25:715–723. doi: 10.1016/j.tiv.2010.12.010. [DOI] [PubMed] [Google Scholar]
- 73.Hallifax D, Houston JB. Saturable uptake of lipophilic amine drugs into isolated hepatocytes: mechanisms and consequences for quantitative clearance prediction. Drug Metab Dispos. 2007;35:1325–1332. doi: 10.1124/dmd.107.015131. [DOI] [PubMed] [Google Scholar]
- 74.Kazmi F, Hensley T, Pope C, Funk RS, Loewen GJ, Buckley DB, et al. Lysosomal sequestration (trapping) of lipophilic amine (cationic amphiphilic) drugs in immortalized human hepatocytes (Fa2N-4 cells) Drug Metab Dispos. 2013;41:897–905. doi: 10.1124/dmd.112.050054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Zuo J, Quinn KK, Kye S, Cooper P, Damoiseaux R, Krogstad P. Fluoxetine is a potent inhibitor of coxsackievirus replication. Antimicrob Agents Chemother. 2012;56:4838–4844. doi: 10.1128/AAC.00983-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Bauer L, Manganaro R, Zonsics B, Strating JRPM, El Kazzi P, Lorenzo Lopez M, et al. Fluoxetine inhibits enterovirus replication by targeting the viral 2c protein in a stereospecific manner. ACS Infect Dis. 2019;5:1609–1623. doi: 10.1021/acsinfecdis.9b00179. [DOI] [PMC free article] [PubMed] [Google Scholar]