Abstract
Background:
Calcium-phosphate product is associated with mortality among patients with end-stage kidney disease on dialysis. However, clinical evidence among hospitalized patients is limited. The objective of this study was to investigate the relationship between admission calcium-phosphate product and 1-year mortality in hospitalized patients.
Materials and Methods:
All adult patients admitted to a tertiary referral hospital in 2009–2013 were studied. Patients who had both available serum calcium and phosphate measurement within 24 h of hospital admission were included. Admission calcium-phosphate product (calcium × phosphate) was stratified based on its distribution into six groups: <21, 21–<27, 27–<33, 33–<39, 39–<45, and ≥45 mg2/dL2. Multivariate cox proportional hazard analysis was performed to evaluate the association between admission calcium-phosphate product and 1-year mortality, using the calcium-phosphate product of 33–<39 mg2/dL2 as the reference group.
Results:
A total of 14,772 patients were included in this study. The mean admission calcium-phosphate product was 34.4 ± 11.3 mg2/dL2. Of these patients, 3194 (22%) died within 1 year of hospital admission. In adjusted analysis, admission calcium-phosphate product of ≥45 mg2/dL2 was significantly associated with increased 1-year mortality with hazard ratio of 1.41 (95% 95% confidence interval 1.25–1.67), whereas lower admission calcium-phosphate product was not significantly associated with 1-year mortality.
Conclusion:
Elevated calcium-phosphate product was significantly associated with increased 1-year mortality in hospitalized patients.
Keywords: Calcium phosphate, hospital mortality, patients
Introduction
Calcium and phosphate are essential elements for the human body and their derangements have adverse clinical effects.[1,2,3,4] Hypocalcemia and hypercalcemia are both associated with poor clinical outcomes including increased hospital mortality.[3,5,6] Elevated serum calcium may enhance vascular atherogenesis and tissue hypoxia via calcification and increased coagulability.[7] Increase in serum calcium is also associated with incident heart failure[7] and acute ischemic stroke.[8] Elevated serum phosphorus levels are associated with increased mortality across diverse population groups, regardless of chronic kidney disease (CKD) status.[9,10,11,12,13,14,15,16] Elevated serum phosphate is also an important risk factor for accelerated vascular calcification and cardiovascular events in patients with CKD and end-stage kidney disease (ESKD).[17]
Studies have demonstrated that elevated calcium-phosphate product (a multiplication of serum calcium and serum phosphate concentration) of >55 mg2/dL2 is associated with increased calcification of blood vessels and soft tissues and translates to poor cardiovascular and mortality outcomes,[9,18,19,20,21,22,23,24,25,26,27,28,29,30] especially in ESKD patients on maintenance hemodialysis.[9] While elevated serum calcium-phosphate product has correlated with poor short-term outcomes including acute kidney injury (AKI) and in-hospital mortality, little is known about the effects of calcium-phosphate product on 1-year mortality. This additional clinical knowledge would further impact the management of both calcium and phosphate derangement in different clinical settings.
Thus, we conducted this study to evaluate the impact of admission calcium-phosphate product on hospitalized patients' 1-year mortality.
Materials and Methods
Study population
This was a single-center cohort study conducted at Mayo Clinic Hospital, Rochester, Minnesota, USA. All hospitalized adult patients who had available admission serum calcium and phosphate between January 2009 and December 2013 were included in this study. In patients with multiple admissions during the study period, only the first hospitalization was included. The Mayo Clinic Institutional Review Board approved this study and exempted the need for informed consent due to the minimal risk nature of the study. All included patients provided research authorization for data use.
Data collection
Clinical characteristics, demographic information, and laboratory data were abstracted from the institutional electronic medical record system. The admission serum calcium and phosphate was obtained within 24 h of hospitalization. Serum calcium and phosphate levels were measured using a photometric method. This method was used throughout the study period. The admission calcium-phosphate product, which was obtained by multiplying total serum calcium and phosphate levels (calcium × phosphate), was the predictor of interest. Estimated glomerular filtration rate (eGFR) was calculated using the CKD Epidemiology Collaboration equation.[31] The Charlson comorbidity score was calculated to evaluate the burden of comorbidities at the time of admission.[32] Principal diagnoses were grouped based on admission ICD-9 codes.
The primary outcome was 1-year mortality. Patients' vital status was derived from our institutional registry and social security death index database.
Statistical analysis
All continuous variables were summarized as mean ± standard deviation. All categorical variables were summarized as counts with percentage. Patient characteristics were compared among admission serum calcium-phosphate product groups using ANOVA for continuous variables and Chi-square test for categorical variables. Admission calcium-phosphate product was categorized based on its distribution into six groups (<21, 21–<27, 27–<33, 33–<39, 39–<45, and ≥45 mg2/dL2). Given its lowest 1-year mortality, calcium-phosphate product of 33–<39 mg2/dL2 was selected as the reference group for outcome comparison. Patient survival was initiated at hospital admission and followed until death or the last inpatient/outpatient follow-up visit. One-year mortality risk was estimated using Kaplan–Meier plot and compared between admission serum calcium-phosphate product groups using a log-rank test. Multivariable cox proportional hazard analysis was constructed to adjust for priori-defined covariates when assessing the independent association between admission calcium-phosphate product and 1-year mortality. The hazard ratio (HR) was adjusted for age, sex, race, eGFR, principal diagnosis, Charlson score, history of coronary artery disease, congestive heart failure, peripheral vascular disease, stroke, diabetes mellitus, chronic obstructive pulmonary disease, cirrhosis, AKI, mechanical ventilation, vasopressor use at admission, calcium supplement use prior to admission, and year of hospitalization. A two-tailed P < 0.05 was considered statistically significant. All analyses were performed using JMP statistical software (version 10, SAS Institute, Cary, NC, USA).
Results
Clinical characteristics
A total of 14,772 patients were studied. About 54% were male. The mean age was 62 ± 18 years. The mean eGFR was 67 ± 35 ml/min/1.73 m2. The mean admission calcium-phosphate product was 34.4 ± 11.3 mg2/dL2. The distribution of admission calcium-phosphate product was as follows: 8% with calcium-phosphate product of <21 mg2/dL2, 15% with 21–<27 mg2/dL2, 26% with 27–<33 mg2/dL2, 25% with 33–<39 mg2/dL2, 14% with 39–<45 mg2/dL2, and 12% with ≥45 mg2/dL2. Table 1 summarizes patient clinical characteristics based on admission calcium-phosphate product levels.
Table 1.
Baseline clinical characteristics
| Variables | All | Calcium-phosphate product at hospital admission (mg2/dL2) | ||||||
|---|---|---|---|---|---|---|---|---|
| <21 | 21-<27 | 27-<33 | 33-<39 | 39-<45 | ≥45 | P | ||
| n | 14772 | 1162 | 2219 | 3875 | 3631 | 2046 | 1839 | |
| Age (years) | 62±18 | 60±18 | 63±17 | 63±17 | 61±18 | 60±18 | 60±18 | <0.001 |
| Male | 7937 (54) | 565 (49) | 1251 (56) | 2116 (55) | 1902 (52) | 1041 (51) | 1062 (58) | <0.001 |
| Race | ||||||||
| Caucasian | 13391 (91) | 1041 (90) | 2050 (92) | 3553 (92) | 3269 (90) | 1822 (89) | 1656 (90) | <0.001 |
| African American | 291 (2) | 26 (2) | 33 (1) | 64 (2) | 59 (2) | 64 (3) | 45 (2) | |
| Asian | 169 (1) | 19 (2) | 11 (1) | 39 (1) | 50 (1) | 25 (1) | 25 (1) | |
| Others | 921 (6) | 76 (7) | 125 (6) | 219 (6) | 253 (7) | 135 (7) | 113 (6) | |
| eGFR (ml/min/1.73 m2) | 67±35 | 78±32 | 77±30 | 73±30 | 70±33 | 62±38 | 36±36 | <0.001 |
| Principal diagnosis | ||||||||
| Cardiovascular | 2787 (19) | 136 (12) | 336 (15) | 768 (20) | 802 (22) | 404 (20) | 341 (19) | <0.001 |
| Endocrine/metabolic | 864 (6) | 75 (6) | 132 (6) | 195 (5) | 189 (5) | 111 (5) | 162 (9) | |
| Gastrointestinal | 1820 (12) | 197 (17) | 395 (18) | 523 (13) | 375 (10) | 181 (9) | 149 (8) | |
| Hematology/oncology | 2412 (16) | 146 (13) | 357 (16) | 663 (17) | 637 (18) | 358 (17) | 251 (14) | |
| Infectious disease | 744 (5) | 158 (14) | 148 (7) | 166 (4) | 124 (3) | 70 (3) | 78 (4) | |
| Respiratory | 777 (5) | 75 (6) | 135 (6) | 249 (6) | 157 (4) | 89 (4) | 72 (4) | |
| Injury/poisoning | 2343 (16) | 178 (15) | 328 (15) | 633 (16) | 627 (17) | 333 (16) | 244 (13) | |
| Other | 3025 (20) | 197 (17) | 388 (17) | 678 (17) | 720 (20) | 500 (24) | 542 (29) | |
| Charlson score | 2.4±2.7 | 2.2±1.7 | 2.3±2.7 | 2.3±2.7 | 2.3±2.6 | 2.4±2.7 | 2.7±2.7 | <0.001 |
| Comorbidities | ||||||||
| Coronary artery disease | 3166 (21) | 185 (16) | 464 (21) | 841 (22) | 808 (22) | 446 (22) | 422 (23) | <0.001 |
| Congestive heart failure | 1316 (9) | 56 (5) | 148 (7) | 338 (9) | 352 (10) | 196 (10) | 226 (12) | <0.001 |
| Peripheral vascular disease | 569 (4) | 30 (3) | 63 (3) | 133 (3) | 154 (4) | 83 (4) | 106 (6) | <0.001 |
| Stroke | 1188 (8) | 86 (7) | 178 (8) | 330 (9) | 297 (8) | 163 (8) | 134 (7) | 0.65 |
| Diabetes mellitus | 3524 (24) | 221 (19) | 470 (21) | 862 (22) | 868 (24) | 544 (27) | 559 (30) | <0.001 |
| COPD | 1448 (10) | 90 (8) | 241 (11) | 407 (11) | 347 (10) | 181 (9) | 182 (10) | 0.02 |
| Cirrhosis | 648 (4) | 73 (6) | 105 (5) | 188 (5) | 125 (3) | 60 (3) | 97 (5) | <0.001 |
| AKI | 4378 (30) | 219 (19) | 412 (19) | 825 (21) | 951 (26) | 773 (38) | 1198 (65) | <0.001 |
| Mechanical ventilation | 998 (7) | 122 (10) | 175 (8) | 239 (6) | 191 (5) | 121 (6) | 150 (8) | <0.001 |
| Vasopressor | 698 (5) | 82 (7) | 88 (4) | 144 (4) | 148 (4) | 89 (4) | 147 (8) | <0.001 |
| Calcium supplement | 4590 (31) | 365 (31) | 661 (30) | 1198 (31) | 1115 (31) | 612 (30) | 639 (35) | 0.01 |
Continuous data are presented as mean±SD, categorical data are presented as count (%). SD: Standard deviation, AKI: Acute kidney injury, COPD: Chronic obstructive pulmonary disease, eGFR: Estimated glomerular filtration rate
Admission serum calcium phosphate levels and 1-year mortality
Of 14,772 patients, 3194 (22%) died within 1 year of hospital admission. Based on the Kaplan–Meier plot in Figure 1, the cumulative 1-year mortality was 24.7% with admission calcium-phosphate product of <21 mg2/dL2, 26.0% with 21–<27 mg2/dL2, 23.9% with 27–<33 mg2/dL2, 22.9% with 33–<39 mg2/dL2, 25.0% with 39–<45 mg2/dL2, and 29.3% with ≥45 mg2/dL2 (P < 0.001). In unadjusted analysis, admission calcium-phosphate product of 21–<27 and ≥45 mg2/dL2 was significantly associated with increased 1-year mortality. However, in adjusted analysis, only admission calcium-phosphate product of ≥45 mg2/dL2 was significantly associated with increased 1-year mortality with HR of 1.41 (95% confidence interval 1.25–1.67), while lower admission calcium-phosphate product was not significantly associated with 1-year mortality [Table 2]. A subgroup analysis of hospital survivors showed a similar association between admission calcium-phosphate product and 1-year mortality.
Figure 1.
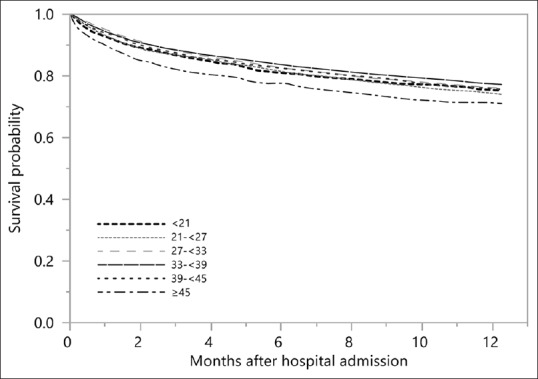
Kaplan–Meier plot showing 1-year mortality among patients with various admission serum calcium phosphate levels
Table 2.
The association between admission calcium-phosphate product levels and 1-year mortality
| Calcium-phosphate product at hospital admission (mg2/dL2) | 1-year mortality (%) | Univariate analysis | Multivariate analysis | ||
|---|---|---|---|---|---|
| HR (95% CI) | P | #Adjusted HR (95% CI) | P | ||
| <21 | 24.7 | 1.11 (0.96-1.29) | 0.14 | 1.09 (0.94-1.26) | 0.28 |
| 21-<27 | 26.0 | 1.16 (1.03-1.30) | 0.01 | 1.09 (0.97-1.23) | 0.13 |
| 27-<33 | 23.9 | 1.05 (0.95-1.16) | 0.34 | 0.99 (0.90-1.10) | 0.97 |
| 33-<39 | 22.9 | 1 (reference) | - | 1 (reference) | - |
| 39-<45 | 25.0 | 1.10 (0.98-1.24) | 0.10 | 1.10 (0.98-1.24) | 0.10 |
| ≥45 | 29.3 | 1.39 (1.24-1.56) | <0.001 | 1.41 (1.25-1.67) | <0.001 |
#Adjusted for age, sex, race, GFR, principal diagnosis, Charlson comorbidity score, coronary artery disease, congestive heart failure, peripheral vascular disease, stroke, diabetes mellitus, COPD, cirrhosis, AKI, mechanical ventilation, vasopressor use at admission, calcium supplement use prior to admission, and year of hospitalization. GFR: Glomerular filtration rate, HR: Hazard ratio, CI: Confidence interval, AKI: Acute kidney injury, COPD: Chronic obstructive pulmonary disease
Subgroup analysis based on the chronic kidney disease status
In a subgroup analysis of patients without CKD, calcium-phosphate product of <21, 21–< 27, and ≥45 mg2/dL2 was significantly associated with increased 1-year mortality [Table 3a]. In patients with CKD, only calcium-phosphate product of ≥45 mg2/dL2 was significantly associated with increased 1-year mortality [Table 3b]. There was an interaction between GFR and calcium-phosphate product on 1-year mortality (P for interaction = 0.001).
Table 3.
Subgroup analysis based on the chronic kidney disease
| Calcium-phosphate product at hospital admission (mg2/dL2) | 1-year mortality (%) | Univariate analysis | Multivariate analysis | ||
|---|---|---|---|---|---|
| HR (95% CI) | P | #Adjusted HR (95% CI) | P | ||
| a. eGFR ≥60 (n=8701) | |||||
| <21 | 24.4 | 1.38 (1.15-1.66) | 0.001 | 1.29 (1.07-1.56) | 0.01 |
| 21-<27 | 24.7 | 1.37 (1.18-1.60) | <0.001 | 1.25 (1.07-1.46) | 0.004 |
| 27-<33 | 21.2 | 1.15 (0.99-1.32) | 0.054 | 1.05 (0.92-1.21) | 0.46 |
| 33-<39 | 19.0 | 1 (reference) | - | 1 (reference) | - |
| 39-<45 | 21.6 | 1.16 (0.97-1.39) | 0.10 | 1.18 (0.99-1.41) | 0.06 |
| ≥45 | 23.1 | 1.29 (1.02-1.63) | 0.03 | 1.40 (1.11-1.78) | 0.005 |
| b. eGFR <60 (n=6071) | |||||
| <21 | 25.5 | 0.87 (0.67-1.11) | 0.26 | 0.79 (0.62-1.03) | 0.08 |
| 21-<27 | 28.8 | 0.99 (0.83-1.19) | 0.91 | 0.94 (0.79-1.14) | 0.54 |
| 27-<33 | 29.2 | 1.00 (0.86-1.16) | 0.96 | 0.97 (0.83-1.12) | 0.67 |
| 33-<39 | 28.8 | 1 (reference) | - | 1 (reference) | - |
| 39-<45 | 28.3 | 0.97 (0.82-1.14) | 0.69 | 1.02 (0.87-1.20) | 0.79 |
| ≥45 | 31.3 | 1.13 (0.98-1.30) | 0.09 | 1.27 (1.09-1.48) | 0.002 |
#Adjusted for age, sex, race, GFR, principal diagnosis, Charlson comorbidity score, coronary artery disease, congestive heart failure, peripheral vascular disease, stroke, diabetes mellitus, COPD, cirrhosis, AKI, mechanical ventilation, and vasopressor use at admission, calcium supplement use prior to admission, and year of hospitalization. GFR: Glomerular filtration rate, eGFR: Estimated GFR, HR: Hazard ratio, CI: Confidence interval, AKI: Acute kidney injury, COPD: Chronic obstructive pulmonary disease
Discussion
By analyzing 14,772 patients, we demonstrated that calcium-phosphate product ≥45 mg2/dL2 was significantly associated with a 1.4-fold increased risk of mortality within 1 year. The increased 1-year mortality risk remained significant after adjustment for multiple clinical variables and regardless of CKD status.
Among patients with ESKD patients on maintenance hemodialysis, data from the US Renal Data System has suggested that an elevated calcium-phosphate product level was associated with increased mortality risk.[9] This risk was more pronounced in those with a calcium phosphorus product ≥72 mg2/dL2.[9] In line with our study, we have further shown that high calcium-phosphate product is detrimental in both CKD and non-CKD patients. However, our study contradicts the findings obtained from the Modification of Diet in Renal Disease (MDRD) Study cohort where elevated serum calcium-phosphate product had a trend toward increased cardiovascular disease (CVD)-related mortality in CKD patients but did not reach statistical significance after adjustment for GFR and other confounders.[33] However, the MDRD cohort was underpowered with a sample size of 840 patients. Furthermore, our patient cohort may represent a population at greater medical risk to disturbances of calcium and phosphorus balance, as indicated by the need for hospitalization. Our study comprising a large patient cohort across several years demonstrates that elevated calcium-phosphate product in hospitalized patients should warrant increased clinical attention and management.
The pathophysiologic mechanism for an elevation of serum calcium-phosphate product on adverse 1-year mortality outcome is not fully clear and likely multifactorial. High serum calcium and phosphate levels are associated with accelerated aortic and coronary calcification, especially in patients with CKD and ESKD.[9,18,19,20,21,22,23,24,25,26,27,28,29,30,34] This calcification predisposes patients to increased CVD-related mortality.[35] From an in vitro study, Reynolds et al. found that human vascular smooth muscle cells underwent vesicle-mediated calcification in response to changes in extracellular calcium and phosphate concentrations.[36] Prior studies have also revealed that elevated admission serum calcium-phosphate product is an independent risk factor for AKI in hospitalized patients,[29] and AKI per se is an independent risk factor itself for increased mortality with an adjusted relative risk varying from 1.6–3.9.[37,38]
It is worth noting that non-CKD patients with low admission calcium-phosphate product <27 mg2/dL2 also had an elevated risk of death within 1 year. We hypothesize that the underlying mechanism is related to the effects of hypocalcemia and hypophosphatemia. Hypocalcemia causes prolonged QT interval and other arrhythmias, which predispose patients to increased incidence of cardiac death.[3,39] Likewise, hypophosphatemia is associated with ventricular ectopic activity,[40] rhabdomyolysis, metabolic encephalopathy, and respiratory failure.[41] However, we did not observe the statistical significance of low serum calcium-phosphate product and mortality rate in CKD patients. This could be due to the lack of statistical power from a small sample size.
Our study has several limitations. First, this is a single-center, retrospective cohort study. A causal relationship between calcium-phosphate product and mortality cannot be established. Second, the study population was predominantly Caucasian. Therefore, generalizability of this study may be limited. Third, although we extensively adjusted for potential confounders, this association may remain confounded by unmeasured or unknown factors. For example, information regarding the severity of illness and cause of calcium-phosphate product disturbance was lacking. The cause of death was also unknown, and this limits the ability to understand the pathological consequence of an abnormal calcium-phosphate product. Future studies consisting of a more diverse population and increased clinical data are needed to further assess this association.
Conclusion
This study comprises a large cohort demonstrating that an elevation of calcium-phosphate product ≥45 mg2/dL2 was significantly associated with increased 1-year mortality, whereas lower admission calcium-phosphate product was not significantly associated with 1-year mortality. Our results suggest that calcium-phosphate product should be calculated and taken into consideration for patient care.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- 1.Kelly C, Gunn IR, Gaffney D, Devgun MS. Serum calcium, urine calcium and polymorphisms of the calcium sensing receptor gene. Ann Clin Biochem. 2006;43:503–6. doi: 10.1258/000456306778904632. [DOI] [PubMed] [Google Scholar]
- 2.Lederer E. Regulation of serum phosphate. J Physiol. 2014;592:3985–95. doi: 10.1113/jphysiol.2014.273979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cheungpasitporn W, Thongprayoon C, Mao MA, Kittanamongkolchai W, Sakhuja A, Erickson SB. Impact of admission serum calcium levels on mortality in hospitalized patients. Endocr Res. 2018;43:116–23. doi: 10.1080/07435800.2018.1433200. [DOI] [PubMed] [Google Scholar]
- 4.Thongprayoon C, Cheungpasitporn W, Mao MA, Sakhuja A, Erickson SB. Admission calcium levels and risk of acute kidney injury in hospitalised patients. Int J Clin Pract. 2018;72:e13057. doi: 10.1111/ijcp.13057. [DOI] [PubMed] [Google Scholar]
- 5.Miller JE, Kovesdy CP, Norris KC, Mehrotra R, Nissenson AR, Kopple JD, et al. Association of cumulatively low or high serum calcium levels with mortality in long-term hemodialysis patients. Am J Nephrol. 2010;32:403–13. doi: 10.1159/000319861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Akirov A, Gorshtein A, Shraga-Slutzky I, Shimon I. Calcium levels on admission and before discharge are associated with mortality risk in hospitalized patients. Endocrine. 2017;57:344–51. doi: 10.1007/s12020-017-1353-y. [DOI] [PubMed] [Google Scholar]
- 7.Lutsey PL, Alonso A, Michos ED, Loehr LR, Astor BC, Coresh J, et al. Serum magnesium, phosphorus, and calcium are associated with risk of incident heart failure: The Atherosclerosis Risk in Communities (ARIC) Study. Am J Clin Nutr. 2014;100:756–64. doi: 10.3945/ajcn.114.085167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chung JW, Ryu WS, Kim BJ, Yoon BW. Elevated calcium after acute ischemic stroke: Association with a poor short-term outcome and long-term mortality. J Stroke. 2015;17:54–9. doi: 10.5853/jos.2015.17.1.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Block GA, Hulbert-Shearon TE, Levin NW, Port FK. Association of serum phosphorus and calciumxphosphate product with mortality risk in chronic hemodialysis patients: A national study. Am J Kidney Dis. 1998;31:607–17. doi: 10.1053/ajkd.1998.v31.pm9531176. [DOI] [PubMed] [Google Scholar]
- 10.Kuo G, Lee CC, Yang SY, Hsiao YC, Chuang SS, Chang SW, et al. Hyperphosphatemia is associated with high mortality in severe burns. PLoS One. 2018;13:e0190978. doi: 10.1371/journal.pone.0190978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Haider DG, Lindner G, Wolzt M, Ahmad SS, Sauter T, Leichtle AB, et al. Hyperphosphatemia Is an independent risk factor for mortality in critically Ill patients: Results from a cross-sectional study. PLoS One. 2015;10:e0133426. doi: 10.1371/journal.pone.0133426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Askar AM. Hyperphosphatemia. The hidden killer in chronic kidney disease. Saudi Med J. 2015;36:13–9. doi: 10.15537/smj.2015.1.9843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cozzolino M, Ciceri P, Galassi A. Hyperphosphatemia: A novel risk factor for mortality in chronic kidney disease. Ann Transl Med. 2019;7:55. doi: 10.21037/atm.2018.06.50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cheungpasitporn W, Thongprayoon C, Mao MA, Kittanamongkolchai W, Sakhuja A, Erickson SB. Admission serum phosphate levels predict hospital mortality. Hosp Pract (1995) 2018;46:121–7. doi: 10.1080/21548331.2018.1483172. [DOI] [PubMed] [Google Scholar]
- 15.Thongprayoon C, Cheungpasitporn W, Chewcharat A, Mao MA, Thirunavukkarasu S, Kashani KB. Admission serum phosphate levels and the risk of respiratory failure. Int J Clin Pract. 2020;74:e13461. doi: 10.1111/ijcp.13461. [DOI] [PubMed] [Google Scholar]
- 16.Thongprayoon C, Cheungpasitporn W, Mao MA, Sakhuja A, Erickson SB. Admission hyperphosphatemia increases the risk of acute kidney injury in hospitalized patients. J Nephrol. 2018;31:241–7. doi: 10.1007/s40620-017-0442-6. [DOI] [PubMed] [Google Scholar]
- 17.Lau WL, Festing MH, Giachelli CM. Phosphate and vascular calcification: Emerging role of the sodium-dependent phosphate co-transporter PiT-1. Thromb Haemost. 2010;104:464–70. doi: 10.1160/TH09-12-0814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Heine GH, Nangaku M, Fliser D. Calcium and phosphate impact cardiovascular risk. Eur Heart J. 2013;34:1112–21. doi: 10.1093/eurheartj/ehs353. [DOI] [PubMed] [Google Scholar]
- 19.Foley RN, Collins AJ, Ishani A, Kalra PA. Calcium-phosphate levels and cardiovascular disease in community-dwelling adults: The Atherosclerosis Risk in Communities (ARIC) Study. Am Heart J. 2008;156:556–63. doi: 10.1016/j.ahj.2008.05.016. [DOI] [PubMed] [Google Scholar]
- 20.Hioki H, Miyashita Y, Shiraki T, Iida O, Uematsu M, Miura T, et al. Impact of deteriorated calcium-phosphate homeostasis on amputation-free survival after endovascular revascularization in patients with critical limb ischemia on hemodialysis. Vasc Med. 2016;21:137–43. doi: 10.1177/1358863X15619241. [DOI] [PubMed] [Google Scholar]
- 21.Komaba H, Fukagawa M. Phosphate-a poison for humans? Kidney Int. 2016;90:753–63. doi: 10.1016/j.kint.2016.03.039. [DOI] [PubMed] [Google Scholar]
- 22.Shin S, Kim KJ, Chang HJ, Cho I, Kim YJ, Choi BW, et al. Impact of serum calcium and phosphate on coronary atherosclerosis detected by cardiac computed tomography. Eur Heart J. 2012;33:2873–81. doi: 10.1093/eurheartj/ehs152. [DOI] [PubMed] [Google Scholar]
- 23.Shroff RC, McNair R, Skepper JN, Figg N, Schurgers LJ, Deanfield J, et al. Chronic mineral dysregulation promotes vascular smooth muscle cell adaptation and extracellular matrix calcification. J Am Soc Nephrol. 2010;21:103–12. doi: 10.1681/ASN.2009060640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yasin A, Liu D, Chau L, Madrenas J, Filler G. Fibroblast growth factor-23 and calcium phosphate product in young chronic kidney disease patients: A cross-sectional study. BMC Nephrol. 2013;14:39. doi: 10.1186/1471-2369-14-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lim S, Gun NT. Secondary hyperparathyroidism and calcium phosphate control in a hemodialysis population. Acta Med Indones. 2007;39:71–4. [PubMed] [Google Scholar]
- 26.Bailie GR, Massry SG National Kidney Foundation. Clinical practice guidelines for bone metabolism and disease in chronic kidney disease: An overview. Pharmacotherapy. 2005;25:1687–707. doi: 10.1592/phco.2005.25.12.1687. [DOI] [PubMed] [Google Scholar]
- 27.Ribeiro S, Ramos A, Brandão A, Rebelo JR, Guerra A, Resina C, et al. Cardiac valve calcification in haemodialysis patients: Role of calcium-phosphate metabolism. Nephrol Dial Transplant. 1998;13:2037–40. doi: 10.1093/ndt/13.8.2037. [DOI] [PubMed] [Google Scholar]
- 28.Cozzolino M, Dusso AS, Slatopolsky E. Role of calcium-phosphate product and bone-associated proteins on vascular calcification in renal failure. J Am Soc Nephrol. 2001;12:2511–6. doi: 10.1681/ASN.V12112511. [DOI] [PubMed] [Google Scholar]
- 29.Thongprayoon C, Cheungpasitporn W, Mao MA, Harrison AM, Erickson SB. Elevated admission serum calcium phosphate product as an independent risk factor for acute kidney injury in hospitalized patients. Hosp Pract (1995) 2019;47:73–9. doi: 10.1080/21548331.2019.1568719. [DOI] [PubMed] [Google Scholar]
- 30.Thongprayoon C, Cheungpasitporn W, Mao MA, Erickson SB. Calcium-phosphate product and its impact on mortality in hospitalized patients. Nephrology (Carlton) 2020;25:22–8. doi: 10.1111/nep.13603. [DOI] [PubMed] [Google Scholar]
- 31.Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF, 3rd, Feldman HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604–12. doi: 10.7326/0003-4819-150-9-200905050-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47:1245–51. doi: 10.1016/0895-4356(94)90129-5. [DOI] [PubMed] [Google Scholar]
- 33.Menon V, Greene T, Pereira AA, Wang X, Beck GJ, Kusek JW, et al. Relationship of phosphorus and calcium-phosphorus product with mortality in CKD. Am J Kidney Dis. 2005;46:455–63. doi: 10.1053/j.ajkd.2005.05.025. [DOI] [PubMed] [Google Scholar]
- 34.Raggi P, Boulay A, Chasan-Taber S, Amin N, Dillon M, Burke SK, et al. Cardiac calcification in adult hemodialysis patients. A link between end-stage renal disease and cardiovascular disease? J Am Coll Cardiol. 2002;39:695–701. doi: 10.1016/s0735-1097(01)01781-8. [DOI] [PubMed] [Google Scholar]
- 35.Goodman WG, Goldin J, Kuizon BD, Yoon C, Gales B, Sider D, et al. Coronary-artery calcification in young adults with end-stage renal disease who are undergoing dialysis. N Engl J Med. 2000;342:1478–83. doi: 10.1056/NEJM200005183422003. [DOI] [PubMed] [Google Scholar]
- 36.Reynolds JL, Joannides AJ, Skepper JN, McNair R, Schurgers LJ, Proudfoot D, et al. Human vascular smooth muscle cells undergo vesicle-mediated calcification in response to changes in extracellular calcium and phosphate concentrations: A potential mechanism for accelerated vascular calcification in ESRD. J Am Soc Nephrol. 2004;15:2857–67. doi: 10.1097/01.ASN.0000141960.01035.28. [DOI] [PubMed] [Google Scholar]
- 37.Wei Q, Liu H, Tu Y, Tang RN, Wang YL, Pan MM, et al. The characteristics and mortality risk factors for acute kidney injury in different age groups in China-a cross sectional study. Ren Fail. 2016;38:1413–7. doi: 10.1080/0886022X.2016.1227618. [DOI] [PubMed] [Google Scholar]
- 38.Coca SG, Yusuf B, Shlipak MG, Garg AX, Parikh CR. Long-term risk of mortality and other adverse outcomes after acute kidney injury: A systematic review and meta-analysis. Am J Kidney Dis. 2009;53:961–73. doi: 10.1053/j.ajkd.2008.11.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Foley RN, Parfrey PS, Harnett JD, Kent GM, Hu L, O'Dea R, et al. Hypocalcemia, morbidity, and mortality in end-stage renal disease. Am J Nephrol. 1996;16:386–93. doi: 10.1159/000169030. [DOI] [PubMed] [Google Scholar]
- 40.Venditti FJ, Marotta C, Panezai FR, Oldewurtel HA, Regan TJ. Hypophosphatemia and cardiac arrhythmias. Miner Electrolyte Metab. 1987;13:19–25. [PubMed] [Google Scholar]
- 41.Subramanian R, Khardori R. Severe hypophosphatemia. Pathophysiologic implications, clinical presentations, and treatment. Medicine (Baltimore) 2000;79:1–8. doi: 10.1097/00005792-200001000-00001. [DOI] [PubMed] [Google Scholar]


