Abstract
Background/Objectives
While daily hydration is best assessed in 24-h urine sample, spot sample is often used by health care professionals and researchers due to its practicality. However, urine output is subject to circadian variation, with urine being more concentrated in the morning. It has been demonstrated that afternoon spot urine samples are most likely to provide equivalent urine concentration to 24-h urine samples in adults. The aim of the present study was to examine whether urine osmolality (UOsm) assessed from a spot urine sample in specific time-windows was equivalent to 24-h UOsm in free-living healthy children.
Subjects/Methods
Among 541 healthy children (age: 3–13 years, female: 45%, 77% non-Hispanic white, BMI:17.7 ± 4.0 kg m−2), UOsm at specific time-windows [morning (0600–1159), early afternoon (1200–1559), late afternoon (1600–1959), evening (2000–2359), overnight (2400–0559), and first morning] was compared with UOsm from the corresponding pooled 24-h urine sample using an equivalence test.
Results
Late afternoon (1600–1959) spot urine sample UOsm value was equivalent to the 24-h UOsm value in children (P < 0.05; mean difference: 62 mmol kg−1; 95% CI: 45–78 mmol kg−1). The overall diagnostic ability of urine osmolality assessed at late afternoon (1600–1959) to diagnose elevated urine osmolality on the 24-h sample was good for both cutoffs of 800 mmol kg−1 [area under the curve (AUC): 87.4%; sensitivity: 72.6%; specificity: 90.5%; threshold: 814 mmol kg−1] and 500 mmol kg−1 (AUC: 83.5%; sensitivity: 75.0%; specificity: 80.0%; threshold: 633 mmol kg−1).
Conclusion
These data suggest that in free-living healthy children, 24-h urine concentration may be approximated from a late afternoon spot urine sample. This data will have practical implication for health care professionals and researchers.
Subject terms: Biomarkers, Health care
Introduction
About 55–70% of the human body consists of water, which plays a vital role as a structural component of cells, tissues, and organs [1–3]. Water is a solvent for protein synthesis [4], and it helps the hydrolytic reactions for macronutrients [5], waste elimination [6], and thermoregulation [7, 8]. Low water intake and underhydration are associated with various health issues such as increased risk of kidney disease [9, 10], diabetes [11], obesity [12], and impaired cognitive function [13–16]. Children may be more susceptible to the adverse effects of underhydration [17] and may be at greater risk. Adequate hydration or drinking water is reported to be associated with children’s health benefits. For instance, children with normal body weight seem to have better hydration compared with obese [18], while increasing water intake improves cognitive performance [19–23]. Interestingly, a large proportion of children in the United States exhibit elevated urine concentration (assessed via urine osmolality) suggesting that they might be underhydrated [24–26]. Thus, it is important for health care practitioners to incorporate hydration assessment into practice when evaluating overall children’s’ health.
Hydration can be assessed by using different blood (plasma osmolality, plasma copeptin, plasma sodium, urea nitrogen/creatinine ratio) and urine (urine osmolality, urine specific gravity, urine color, urine volume, void frequency) biomarkers, body mass changes, or thirst, considering body weight loss as gold standard marker during exercise [27]. A 24-h urine sample is recommended to assess daily hydration with urinary biomarkers [28] as it is valid, noninvasive method, and represents the behavioral and neuroendocrine responses throughout an entire day [29]. Moreover, urine osmolality provides an indication of arginine vasopressin (AVP) levels, the main regulator of water homeostasis [29, 30]. However, collecting 24-h urine samples for hydration assessment in real-life is methodologically difficult and burdensome [28], especially in children [31]. Consequently, many large-scale studies, including the National Health And Nutrition Examination Survey in the US, have used spot urine sampling [24, 26, 32]. Although many large-scale studies, including NHANES, have used spot urine sampling, it is likely that the accuracy of the spot sample in relation to the 24-h sample may be dependent upon the timing of sample collection, given the known circadian variability in urine production [28].
Urine volume and concentration fluctuate throughout the day due to the timing of fluid intake (mostly during the day, not at night) [33]. Water homeostasis via AVP secretion is also sensitive to changes in circadian rhythm phases [34], independent of fluid intake [33]. Specifically, investigation in rats and mice has demonstrated elevated AVP and subsequently elevated urine osmolality during the late sleep phase [34]. These responses serve as a mechanism to augment water conservation and prevent water loss during sleep. This elevation in AVP has been found to peak after completion of the late sleep phase, in the early wake phase [34]. Similar findings have been demonstrated in human adults, in which water conservation (decreased urine volume, elevated urine osmolality) was evident in overnight and morning voids [33]. Decreased urine volume and elevated urine osmolality have also been observed overnight in children independent of sex and puberty stage [35]. Recent studies examining the association between spot and 24-h urinary hydration biomarkers in free-living healthy adults have found that urine osmolality and urine specific gravity values obtained from the voids collected between 1400 and 2000 h (mid- to late-afternoon) were equivalent to those of the 24-h urine sample [33, 36]. Similarly, significantly elevated morning urine osmolality compared with 24-h urine osmolality has consistently been observed [33, 36, 37]. This contradicts common practice of using first-morning voids for hydration measurement [28, 38].
Evidently, there is a lack of consensus regarding the applicability and optimal timing of spot urine samples in hydration assessment. In addition, a validation study on the equivalence of urine osmolality from a spot urine sample to the 24-h sample has not been done in children, despite evidence of circadian rhythm regulations of water homeostasis. Further validation study in this pediatric population is needed as the total body water differs by age [1] and fluid regulating hormone can be affected by the growth hormones [39]. Thus, the purpose of this study was to identify the time-windows during which spot values of urine osmolality are equivalent to 24-h values, in healthy free-living children.
Methods
Participants
A total of 541 healthy children (3–13 years, 45% female, 77% non-Hispanic white) were recruited from the Northwest Arkansas in the US via online advertisement and flyers. The study was approved by the Institutional Review Board in accordance with the Helsinki Declaration of 1975 as revised in 1983. This analysis was drawn from a larger study registered at www.clinicialtrials.gov as NCT# 02937038. Signed consent was obtained from each subject’s legal guardian, and the children provided verbal assent indicating their willingness to participate.
A medical history and a behavioral questionnaire were used to exclude children if they: (1) used a diaper or if they had enuresis during the day or the night, (2) had metabolic, cardiovascular, hematologic, hepatic, gastrointestinal, renal, pulmonary, endocrine, or psychiatric diseases, based on the medical history questionnaire, (3) used medication that interferes with water metabolism, (4) had surgical operation on digestive tract, except appendectomy, (5) had regular drug treatment within 15 days prior to the start of the study.
Study protocol
During the first meeting, subjects and their parents were briefed on how to collect their urine samples in the urine containers. Body mass and height were recorded with minimal clothing and no shoes to the nearest 0.1 kg and 0.01 m, respectively. Body mass index (BMI) was calculated (BMI = weight (kg)/height (m)2), and it was categorized into ‘normal’ or ‘overweight/obese’ based on the BMI-for-age percentiles graph obtained from Centers for Disease Control and Prevention website. Nine numbered 1-L urine containers (Nalgene Wide-Mouth Bottle, Thermo Scientific, Waltham, MA, USA) were provided, with instructions to collect each void in a separate, numbered container. In case participants produced more than nine voids over the 24-h period, an additional 3.5-L urine container (24-h Urine Collection Containers, VWR®, Radnor, PA, USA) was provided to collect the rest of the urine produced over the 24-h period. Toilet hats (Medi-Pak Collection Hat, McKesson, San Francisco, CA, USA) were provided for girls to facilitate urine collection. Subjects’ parents were asked to record the time of each void on each container and in a provided form.
Data collection started on Sunday morning and subjects were instructed to refrain from strenuous exercise for the entire weekend. Participants were asked to perform a 24-h urine collection using the following procedure: on Sunday morning upon waking, participants voided and then discarded this first-morning void, as this reflects the hydration status and fluid/foods consumed on the day before [40]. They then collected all subsequent voids in the provided numbered urine containers, including voids produced during the night. On Monday morning upon awakening, participants collected their first-morning void, thus completing the 24-h collection. Urine samples were returned to the lab on Monday morning, and subjects were compensated with a prepaid gift card.
Laboratory protocol
A 10-mL sample from each urine bottle was aliquoted into a plastic tube and the rest of each bottle was combined in a 24-h urine container to create the 24-h sample. The aliquot amount of 10 mL was within similar range for each void which is around 5–10% of total void volume. All spot urine samples and 24-h urine samples were analyzed for osmolality directly upon retrieval or after storage at 4 °C within 4 days using a freezing point depression osmometer (3250 single-sample osmometer, Advanced Instrument, Norwood, MA, USA) [41].
Statistical analysis
Void time was categorized into different time-windows: morning (0600–1159), early afternoon (1200–1559), late afternoon (1600–1959), evening (2000–2359), overnight (2400–0559), and first morning (0600–0959 the next day). Broad 4-h time-windows were selected to maximize the number of children producing a sample within each time-window. If a child produced more than one urine sample within the same time-window, only the first spot urine sample within each time-window was used for the analyses to equally weigh the data. Ultimately, sample size at each time-window was different, as each participant did not void during every time-window. Equivalence of urine osmolality between the first spot urine sample of each time-window and the 24-h urine sample was tested using the (1–2α) × 100% computed confidence interval (CI) between the spot- and 24-h urine sample. Spot urine sample at each time-window was considered equivalent if the CI lies between −ε and ε. On the other hand, they were considered not equivalent if one of the bounds of the CI is outside the interval −ε and ε. In addition, two one-sided tests were examined to evaluate the equivalence. A right one-sided t-test on the lower bound of the interval and a left one-sided t-test on the upper bound of the interval were defined by the user by obtaining P-values for both tests. The greatest of these P-values were taken to evaluate the equivalence [42]. The bound of ε = 80 mmol kg−1 was used in the present study, since it represents 10% of the urine osmolality value (800 mmol kg−1) that is used to determine underhydration [32]. Previous study in an adult population used the bound of ε = 100 mmol kg−1, however, a narrower bound was chosen for the current study as the distribution of fluid intake in children is not as large as the one in adults [43]. Lastly, the overall diagnostic ability of urine osmolality at the specific time-window, to identify elevated urine osmolality by using cutoffs of 800 mmol kg−1 and 500 mmol kg−1 based on the 24-h sample was performed by receivers operating characteristics (ROC) analysis [32]. Urine osmolality below 500 mmol kg−1 has been used as a physiological index of adequate water intake [29] and the European Food Safety Authority has based their water intake recommendations on urine osmolality values below 500 mmol kg−1 [44]. On the other side urine osmolality greater than 800 mmol kg−1 has been widely used as an index of underhydration where vasopressin in increased to maintain water homeostasis [24, 45, 46]. All the analyses were performed by using JMP Pro (version 14.0, SAS Inc., Gary, NC, USA). A value of P < 0.05 was regarded as statistically significant.
Results
Characteristics of the 541 children (age: 3–13 years, girls: 45%) appear in Table 1 and are reported by ages based on the grouping of the water dietary reference intakes established by the Institute of Medicine [47]. As expected, not all participants provided a urine sample within each time-window. For most of the time-windows, at least two-thirds of children provided a urine sample, except for the overnight window, during which only 13% of participants provided a urine sample. Mean [95% CI] spot urine osmolality for each time-window were as follows: morning, 766 [741, 791] mmol kg−1; early afternoon, 753 [726, 779] mmol kg−1; late afternoon, 757 [728, 785] mmol kg−1; evening, 784 [755, 784] mmol kg−1; overnight, 729 [660, 797] mmol kg−1; first morning, 800 [755, 846] mmol kg−1. Mean [95% CI] 24-h urine osmolality was 706 [686, 726] mmol kg−1. Results of the equivalence analysis are presented in Table 2, and the overall urine osmolality value in each time-window was higher compared with 24-h urine osmolality. Among all six time-windows, only the spot urine sample obtained at 1600–1959 was identified as equivalent to the 24-h urine sample (P < 0.05).
Table 1.
Subject description
| 3 years (n = 43) | 4–8 years (n = 281) | 9–13 years (n = 217) | Total (n = 541) | |
|---|---|---|---|---|
| Age (years) | 3.0 ± 0.0 | 5.9 ± 1.4a | 10.8 ± 1.4a,b | 7.6 ± 3.0 |
| Weight (kg) | 17.5 ± 9.0 | 23.2 ± 5.6a | 45.5 ± 16.5a,b | 31.6 ± 16.2 |
| Height (cm) | 99.3 ± 4.4 | 119.1 ± 11.2a | 148.7 ± 13.3a,b | 129.3 ± 20.4 |
| BMI (kg m−2) | 17.7 ± 8.8 | 16.2 ± 2.3 | 20.5 ± 9.5a,b | 18.0 ± 7.0 |
| Overweight/Obese (%) | 19.0 | 22.6 | 31.4 | 25.7 |
| 24-h UOsm (mmol kg−1) | 665 ± 209 | 732 ± 234 | 679 ± 245b | 706 ± 238 |
| 24-h UVol (mL day−1) | 441 ± 201 | 614 ± 295 | 926 ± 636a,b | 725 ± 489 |
| 24-h UVoid (number∙day−1) | 6.2 ± 2.0 | 6.8 ± 2.9 | 5.7 ± 2.5b | 6.4 ± 2.7 |
BMI body mass index, UOsm urine osmolality, UVol urine volume, UVoid urine void frequency
aDifference from 3 years (P < 0.05)
bDifference from 4–8 years (P < 0.05)
Table 2.
Equivalence test of urine osmolality at different time-windows with 24-h urine sample by using 80 mmol kg−1 as bound
| Time-window (h) | Mean spot UOsm (mmol kg−1) | Mean absolute difference (spot vs. 24-h UOsm) | N | 95 % CI of mean (mmol kg−1) | Equivalence test | |
|---|---|---|---|---|---|---|
| Lower limit | Upper limit | Bound 80 mmol kg−1 | ||||
| 0600–1159 | 766 | (+) 80 | 375 | 61 | 100 | N |
| 1200–1559 | 753 | (+) 65 | 461 | 48 | 83 | N |
| 1600–1959a | 757 | (+) 62 | 470 | 45 | 78 | Y |
| 2000–2359 | 784 | (+) 90 | 357 | 73 | 107 | N |
| Overnight | 729 | (+) 50 | 71 | 10 | 89 | N |
| First morning | 800 | (+) 88 | 414 | 74 | 102 | N |
aEquivalent to 24-h sample based on 80 mmol kg−1 bound (P < 0.05)
Overnight:2400–0559, first morning: 0600–0959 the next day
Bold values indicates time-window that was identified as equivalent to the 24-h urine sample (P < 0.05)
Based on the ROC analysis, the overall diagnostic ability of the spot urine osmolality at 1600–1959 to identify elevated 24-h urine osmolality (>800 mmol kg−1) was good with area under the curve (AUC) of 87.4% with sensitivity of 72.6%, specificity of 90.5%, and threshold of 814 mmol kg−1 (Fig. 1a). Similarly, the overall diagnostic ability of the spot urine sample, at the same time-window (1600–1959), to identify 24-h urine osmolality greater than 500 mmol kg−1 was also good (AUC: 83.5%, sensitivity: 75.0%, specificity: 80.0%, and threshold 633 mmol kg−1; Fig. 1b).
Fig. 1.
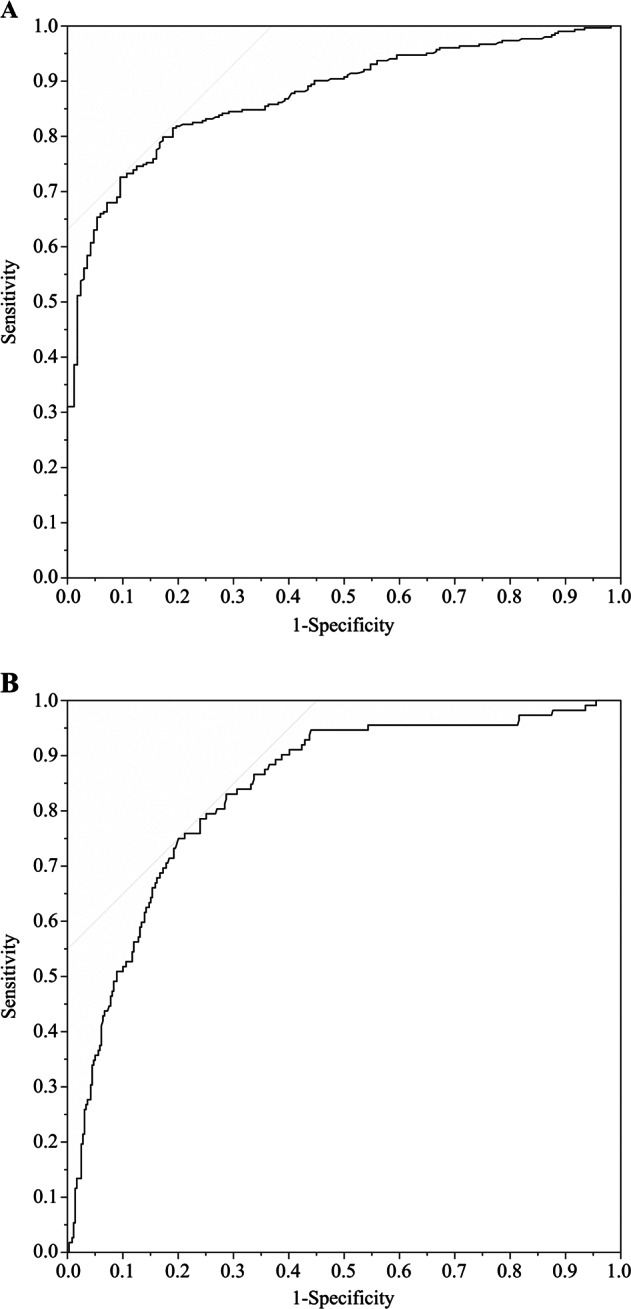
Receiver operating characteristic curve of the urine osmolality collected at 1600–1959 compared with 24-h urine osmolality cutoff of a 800 mmol kg−1 and b 500 mmol kg−1
Discussion
The aim of the study was to identify possible time-windows during which urine osmolality assessed from a spot urine sample was equivalent to 24-h urine osmolality in children. We found that the spot urine sample from late afternoon (1600–1959) was equivalent to urine osmolality from the 24-h urine sample. This supports previous research in adults, that also found equivalence between 24 h and late afternoon spot urine osmolality [33, 36]. In addition, slight fluctuation in spot urine osmolality throughout the day may demonstrate the circadian regulation of fluid balance, as similarly observed in previous studies [33, 35]. Urine osmolality from spot urine samples collected during other time-windows including morning ones were not equivalent to 24-h urine osmolality. In particular, mean urine osmolality was highest in the first-morning spot sample. This supports evidence from previous studies [33, 36, 37] concluding that the first-morning urine sample is not optimal for accurate hydration assessment. Curiously, spot urine osmolality at all time-windows overestimated the 24-h urine osmolality (Table 2). This is in accordance with previous research in adults that found all time-windows except 1400–1600 to overestimate the 24-h sample [36]. This likely resulted from unequivalent representation of subjects in each time-window, as not all children voided in each time-window. In addition, some subjects voided more than once in a time-window and only the first void was used for spot analysis, while all voids were included in 24-h analyses.
We found good overall diagnostic ability of urine osmolality measured from a spot sample at 1600–1959 to identify elevated urine osmolality based on the 24-h sample with two different cutoffs; 800 mmol kg−1 and 500 mmol kg−1. Accordingly, a concentrated spot sample, measured in the afternoon, would indicate a high probability of underhydration as indicated by concentrated 24-h urine. Based on this data, future studies examining hydration in children could probably use an afternoon spot urine sample when 24-h urine sampling is not feasible [24, 32, 48]. Furthermore, an afternoon spot sample may reduce the burden of urine collection on children—especially younger ones for whom a 24-h collection may be more difficult—and increase the likelihood of routine hydration assessment in children. Routine hydration assessment is crucial because inadequate water intake and underhydration have been reported to be associated with various health issues in both children and adults [6, 12, 13, 17]. Furthermore, dietary habits acquired early in life can be sustained during adulthood; therefore, acquiring healthy hydration habits from the youngest age may also be important for health in later life [49–51].
The current study is the very first to examine the equivalence of a hydration biomarker between spot and 24-h urine sample in a pediatric population. With the large sample size of 541, we were able to identify that the spot urine osmolality at 1600–1959 was equivalent to 24-h urine osmolality. The current study supports and builds upon previous research establishing equivalence between afternoon spot urine and 24-h urine osmolality in adults with wide-ranging fluid intake behaviors [36], extending this to children aged 3–13 years old. This could be useful in helping clinicians and researchers in interpreting urinary biomarkers in existing and future research and in clinical practice. Some limitations of the study include the unequal distribution of children within age categories, as well as the unequal number of samples analyzed in each of the six time-windows as not all children voided during all time-windows. The aliquot amount of 10 mL from each void for sample analysis was around 5–10% of total void volume, however, this may be influenced the 24-h urine osmolality as the extracted proportion of volume was not the same. Lastly, equivalence was demonstrated for samples obtained from healthy free-living children around or after the usual midday meal time in this study, but not for samples obtained in the morning. These results may not be extrapolated to those children who have different daily rhythm, meal times, and eating and drinking patterns such as in athletic children.
In conclusion, a spot urine sample collected during late afternoon (1600 and 1959 h) may provide a good alternative to a 24-h urine collection for assessing hydration in healthy children, useful to health care practitioners.
Acknowledgements
The authors would like to thank Michael Berry, Jesus De La Sancha, Rebecca Eberle, Reid Fawcet, Julia Hurtz, Nicole Huettner, Nicholas Lane, Brennen Long, Michael McCullough, Julia McGill, Zoe McKinney, Jordan Means, Nicole Medina, Katherine Montgomery, Mackenzie Ray, Emily Rolling, Audrey Smith, Whitney Stone, and Olivia Taylor for their help during data collection. This study was funded by Danone Research.
Author contributions
SAK, JHB, and ETP designed the research study; HS, LGS, and ADS, conducted data collection and sample analysis; HS, AM, and SAK analyzed the data; HS and ATC wrote the paper. SAK was the principal investigator and had primary responsibility for the final content. All authors read, critically revised, and approved the final paper.
Compliance with ethical standards
Conflict of interest
SAK has served as scientific consultant for Quest Diagnostics, Standard Process and Danone Research. SAK has active grants with Standard Process and Danone Research. ADS is a scientific consultant for Gatorade Sports Science Institute. JHB and ETP are employees of Danone Research, France. The remaining authors declare that they have no conflict of interest.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Petraccia L, Liberati G, Giuseppe Masciullo S, Grassi M, Fraioli A. Water, mineral waters and health. Clin Nutr. 2006;25:377–85. doi: 10.1016/j.clnu.2005.10.002. [DOI] [PubMed] [Google Scholar]
- 2.Sawka MN, Cheuvront SN, Carter R. Human Water Needs. Nutr Rev. 2005;11:S30–S39. doi: 10.1111/j.1753-4887.2005.tb00152.x. [DOI] [PubMed] [Google Scholar]
- 3.Lang F, Busch LG, Ritter M, Volkl H, Waldegger S, Gulbins E, et al. Functional significance of cell volume regulatory mechanisms. Physiol Rev. 1998;78:247–306. doi: 10.1152/physrev.1998.78.1.247. [DOI] [PubMed] [Google Scholar]
- 4.Haussinger D. The role of cellular hydration in the regulation of cell function. Biochem j. 1996;313:697–710. doi: 10.1042/bj3130697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jéquier E, Constant F. Water as an essential nutrient: the physiological basis of hydration. Eur J Clin Nutr. 2010;64:115–23. doi: 10.1038/ejcn.2009.111. [DOI] [PubMed] [Google Scholar]
- 6.Popkin BM, Rosenberg IH. Water, hydration and health. Nutr Rev. 2010;68:439–58. doi: 10.1111/j.1753-4887.2010.00304.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.McKinley MJ, Martelli D, Pennington GL, Trevaks D, McAllen RM. Integrating competing demands of osmoregulatory and thermoregulatory homeostasis. Physiology. 2018;33:170–81. doi: 10.1152/physiol.00037.2017. [DOI] [PubMed] [Google Scholar]
- 8.Sawka MN, Cheuvront SN, Kenefick RW. Hypohydration and human performance: impact of environment and physiological mechanisms. Sport Med. 2015;45:51–60. doi: 10.1007/s40279-015-0395-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sontrop JM, Dixon NS, Garg AX, Buendia-Jimenez I, Dohein O, Huang SS, et al. Association between water intake, chronic kidney disease, and cardiovascular disease: a cross-sectional analysis of NHANES data. Am J Nephrol. 2013;37:434–42. doi: 10.1159/000350377. [DOI] [PubMed] [Google Scholar]
- 10.Strippoli GFM, Jonathan CC, Rochtchina E, Flood VM, Wang JJ, Mitchell P. Fluid and nutrient intake and risk of chronic kidney disease. Nephrology. 2011;16:326–34. doi: 10.1111/j.1440-1797.2010.01415.x. [DOI] [PubMed] [Google Scholar]
- 11.Roussel R, Fezeu L, Bouby N, Balkau B, Lantieri O, Alhenc-Gelas F, Marre M, Bankir L. Low water intake and risk for new-onset hyperglycemia. Diabetes Care. 2011;34:2551–4. doi: 10.2337/dc11-0652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chang T, Ravi N, Plegue MA, Sonneville KR, Davis MM. Inadequate hydration, BMI, and obesity among US adults: NHANES 2009–12. Ann Fam Med. 2016;14:320–4. doi: 10.1370/afm.1951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Grandjean AC, Grandjean NR. Dehydration and cognitive performance. J Am Coll Nutr. 2007;26:549S–554S. doi: 10.1080/07315724.2007.10719657. [DOI] [PubMed] [Google Scholar]
- 14.Szinnai G, Schachinger H, Arnaud MJ, Linder L, Keller U. Effect of water deprivation on cognitive-motor performance in healthy men and women. Am J Physiol. 2005;289:275–80. doi: 10.1152/ajpregu.00501.2004. [DOI] [PubMed] [Google Scholar]
- 15.Pross N, Demazieres A, Girard N, Barnouin R, Metzger D, Klein A, et al. Effects of changes in water intake on mood of high and low drinkers. PLoS ONE. 2014;9:e94754.. doi: 10.1371/journal.pone.0094754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Muñoz CX, Johnson EC, McKenzie AL, Guelinckx I, Graverholt G, Douglas JC, et al. Habitual total water intake and dimensions of mood in healthy young women. Appetite. 2015;92:81–6. doi: 10.1016/j.appet.2015.05.002. [DOI] [PubMed] [Google Scholar]
- 17.Maughan RJ. Hydration, morbidity, and mortality in vulnerable populations. Nutr Rev. 2012;70(Suppl 2):S152-5.. doi: 10.1111/j.1753-4887.2012.00531.x. [DOI] [PubMed] [Google Scholar]
- 18.Maffeis C, Tommasi M, Tomasselli F, Spinelli J, Fornari E, Scattolo N, et al. Fluid intake and hydration status in obese vs normal weight children. Eur J Clin Nutr. 2016;70:560–5. doi: 10.1038/ejcn.2015.170. [DOI] [PubMed] [Google Scholar]
- 19.Edmonds CJ, Crosbie L, Fatima F, Hussain M, Jacob N, Gardner M. Dose-response effects of water supplementation on cognitive performance and mood in children and adults. Appetite. 2017;108:464–70. doi: 10.1016/j.appet.2016.11.011. [DOI] [PubMed] [Google Scholar]
- 20.Benton D, Burgess N. The effect of the consumption of water on the memory and attention of children. Appetite. 2009;53:143–6. doi: 10.1016/j.appet.2009.05.006. [DOI] [PubMed] [Google Scholar]
- 21.Perry CS, Rapinett G, Glaser NS, Ghetti S. Hydration status moderates the effects of drinking water on children’s cognitive performance. Appetite. 2015;95:520–7. doi: 10.1016/j.appet.2015.08.006. [DOI] [PubMed] [Google Scholar]
- 22.Fadda R, Rapinett G, Grathwohl D, Parisi M, Fanari R, Calo CM, et al. Effects of drinking supplementary water at school on cognitive performance in children. Appetite. 2012;59:730–7. doi: 10.1016/j.appet.2012.07.005. [DOI] [PubMed] [Google Scholar]
- 23.D’Anci KED, Constant F, Rosenberg IH, D’Anci KE. Hydration and cognitive function in children. Nutr Rev. 2006;64:457–64. doi: 10.1301/nr.2006.oct.457-464. [DOI] [PubMed] [Google Scholar]
- 24.Kenney EL, Long MW, Cradock AL, Gortmaker SL. Prevalence of inadequate hydration among US children and disparities by gender and race/ethnicity: National Health and Nutrition Examination Survey, 2009–2012. Am J Public Health. 2015;105:e113–e118. doi: 10.2105/AJPH.2015.302572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Suh H, Kavouras SA. Water intake and hydration state in children. Eur J Nutr. 2018;0:1–22. doi: 10.1007/s00394-018-1869-9. [DOI] [PubMed] [Google Scholar]
- 26.Stookey JD, Brass B, Holliday A, Arieff A. What is the cell hydration status of healthy children in the USA? Preliminary data on urine osmolality and water intake. Public Health Nutr. 2012;15:2148–56. doi: 10.1017/S1368980011003648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cheuvront SN, Kenefick RW. Dehydration: physiology assessment, and performance. Compr Physiol. 2014;4:257–85. doi: 10.1002/cphy.c130017. [DOI] [PubMed] [Google Scholar]
- 28.Cheuvront SN, Kenefick RW, Zambraski EJ. Spot Urine concentrations should not be used for hydration assessment: a methodology review. Int J Sport Nutr Exerc Metab. 2015;25:293–7. doi: 10.1123/ijsnem.2014-0138. [DOI] [PubMed] [Google Scholar]
- 29.Perrier ET, Buendia-Jimenez I, Vecchio M, Armstrong LE, Tack I, Klein A, et al. Twenty-four-hour urine osmolality as a physiological index of adequate water intake. Dis Markers. 2015;2015:231063.. doi: 10.1155/2015/231063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Armstrong LE, Johnson EC. Water intake, water balance, and the elusive daily water requirement. Nutrients. 2018;10:1–25. doi: 10.3390/nu10121928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rios-Leyvraz M, Bovet P, Tabin R, Genin B, Russo M, Rossier MF, et al. Urine spot samples can be used to estimate 24-hour urinary sodium excretion in children. J Nutr. 2018;148:1946–53. doi: 10.1093/jn/nxy211. [DOI] [PubMed] [Google Scholar]
- 32.Bar-David Y, Urkin J, Landau D, Bar-David Z, Pilpel D. Voluntary dehydration among elementary school children residing in a hot arid environment. J Hum Nutr Diet. 2009;22:455–60. doi: 10.1111/j.1365-277X.2009.00960.x. [DOI] [PubMed] [Google Scholar]
- 33.Perrier E, Demazieres A, Girard N, Pross N, Osbild D, Metzger D, et al. Circadian variation and responsiveness of hydration biomarkers to changes in daily water intake. Eur J Appl Physiol. 2013;113:2143–51. doi: 10.1007/s00421-013-2649-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gizowski C, Trudel E, Bourque CW, Mcgill J. Central and peripheral roles of vasopressin in the circadian defense of body hydration Integrated Program in Neurosciences. Best Pract Res Clin Endocrinol Metab. 2017;31:535–46. doi: 10.1016/j.beem.2017.11.001. [DOI] [PubMed] [Google Scholar]
- 35.Mahler B, Kamperis K, Ankarberg-Lindgren C, Frøkiӕr J, Djurhuus JC, Rittig S. Puberty alters renal water handling. Am J Physiol Physiol. 2013;305:F1728–F1735. doi: 10.1152/ajprenal.00283.2013. [DOI] [PubMed] [Google Scholar]
- 36.Bottin JH, Lemetais G, Poupin M, Jimenez L, Perrier ET. Equivalence of afternoon spot and 24-h urinary hydration biomarkers in free-living healthy adults. Eur J Clin Nutr. 2016;70:904–7. doi: 10.1038/ejcn.2015.217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Armstrong LE, Pumerantz AC, Fiala KA, Roti MW, Kavouras SA, Casa DJ, et al. Human hydration indices: acute and longitudinal reference values. Int J Sport Nutr Exerc Metab. 2010;20:145–53. doi: 10.1123/ijsnem.20.2.145. [DOI] [PubMed] [Google Scholar]
- 38.McDermott BP, Anderson SA, Armstrong LE, Casa DJ, Cheuvront SN, Cooper L, et al. National athletic trainers’ association position statement: fluid replacement for the physically active. J Athl Train. 2017;52:877–95. doi: 10.4085/1062-6050-52.9.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Dimke H, Flyvbjerg A, Bourgeois S, Thomsen K, Frøkiӕr J, Houillier P, et al. Acute growth hormone administration induces antidiuretic and antinatriuretic effects and increases phosphorylation of NKCC2. Am J Physiol Physiol. 2007;292:F723–F735. doi: 10.1152/ajprenal.00276.2006. [DOI] [PubMed] [Google Scholar]
- 40.Iglesia I, Santaliestra-Pasias AM, Bel-Serrat S, Sadalla-Collese T, Miguel-Berges ML, Moreno LA. Fluid consumption, total water intake and first morning urine osmolality in Spanish adolescents from Zaragoza: data from the HELENA study. Eur J Clin Nutr. 2016;70:541–7. doi: 10.1038/ejcn.2015.203. [DOI] [PubMed] [Google Scholar]
- 41.Adams JD, Kavouras SA, Johnson EC, Jansen LT, Capitan-Jimenez C, Robillard JI, et al. The effect of storing temperature and duration on urinary hydration markers. Int J Sport Nutr Exerc Metab. 2017;27:18–24. doi: 10.1123/ijsnem.2016-0098. [DOI] [PubMed] [Google Scholar]
- 42.Schuirmann DJ. A comparison of the two one-sided tests procedure and the power approach for assessing the equivalence of average bioavailability. J Pharmacokinet Biopharm. 1987;15:657–80. doi: 10.1007/BF01068419. [DOI] [PubMed] [Google Scholar]
- 43.Özen AE, del Mar Bibiloni M, Pons A, Tur JA. Fluid intake from beverages across age groups: a systematic review. J Hum Nutr Diet. 2015;28:417–42. doi: 10.1111/jhn.12250. [DOI] [PubMed] [Google Scholar]
- 44.EFSA Panel on Dietetic Products Nutrition and Allergies (NDA) Scientific opinion on dietary reference values for water. EFSA J. 2010;8:1459. [Google Scholar]
- 45.Kavouras SA. Hydration, dehydration, underhydration, optimal hydration: are we barking up the wrong tree? Eur J Nutr. 2019;58:471–3. doi: 10.1007/s00394-018-01889-z. [DOI] [PubMed] [Google Scholar]
- 46.Kavouras SA, Johnson EC, Bougatsas D, Arnaoutis G, Panagiotakos DB, Perrier E, et al. Validation of a urine color scale for assessment of urine osmolality in healthy children. Eur J Nutr. 2016;55:907–15. doi: 10.1007/s00394-015-0905-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Food and Nutrition Board (FNB) of the Institute of Medicine. Panel on dietary reference intakes for electrolytes, water: DRI, dietary reference intakes for water, potassium, sodium, chloride, and sulfate. Natl Acad. 2005;73–185.
- 48.Manz F, Wentz A. 24-h hydration status: parameters, epidemiology and recommendations. Eur J Clin Nutr. 2003;57:10–9. doi: 10.1038/sj.ejcn.1601896. [DOI] [PubMed] [Google Scholar]
- 49.Emmett PM, Jones LR. Diet, growth, and obesity development throughout childhood in the Avon Longitudinal Study of Parents and Children. Nutr Rev. 2015;73:175–206. doi: 10.1093/nutrit/nuv054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Fiorito LM, Marini M, Mitchell DC, Smiciklas-Wright H, Birch LL. Girls’ early sweetened carbonated beverage intake predicts different patterns of beverage and nutrient intake across childhood and adolescence. J Am Diet Assoc. 2010;110:543–50. doi: 10.1016/j.jada.2009.12.027. [DOI] [PubMed] [Google Scholar]
- 51.Park S, Pan L, Sherry B, Li R. The Association of sugar-sweetened beverage intake during infancy with sugar-sweetened beverage intake at 6 years of age. Pediatrics. 2014;134:S56–S62. doi: 10.1542/peds.2014-0646J. [DOI] [PMC free article] [PubMed] [Google Scholar]


