Highlights
-
•
First reported case of Critical Illness-Associated Cerebral Microbleeds in Covid-19.
-
•
EEG plays a crucial role in Covid-19 patients in the ICU.
-
•
Acute neurological complications should be explored systematically.
Keywords: Covid-19, SARS-CoV2, Microbleeds, EEG, ICU, Cytokine, Brain, Inflammation, Coma
Abstract
Objectives
We describe a patient suffering from Covid19-related acute respiratory distress syndrome (ARDS), highlighting the diagnostic role of the EEG in ICU.
History
A Covid-19 patient undergoing mechanical ventilation due to related acute respiratory distress syndrome (ARDS), presented altered mental status in the ICU. Video-EEG revealed a focal monomorphic theta slowing in bilateral frontal-central regions. Concordant with the EEG localization, MRI showed abundant microbleeds located in bilateral white matter junction, various regions of corpus callosum and internal capsule, suggestive of Critical Illness-Associated Cerebral Microbleeds. CSF analysis excluded the presence of encephalitis, SARS-Cov2 RNA-PCR in CSF was negative. Clinical and biological picture was suggestive of cytokine release syndrome.
Conclusion
This is the first reported case of Critical Illness-Associated Cerebral Microbleeds in the context of Covid-19. Knowledge of Covid-19 is still partial and acute neurological complications should be explored systematically. In our case, EEG helped to rule out non-convulsive status epilepticus, but revealed focal dysfunction, justifying further investigations.
EEG plays a crucial role in these patients, allowing investigating the presence of focal or diffuse cerebral dysfunction. This is particularly helpful for Covid-19 patients in the ICU, where the neurological examination is challenging by the severity of the respiratory illness.
1. Introduction
It was recently documented that 36.4% of patients with Coronavirus Disease 2019 (Covid-19) develop neurological symptoms (Mao et al., 2020). Only one case of viral encephalitis has been reported so far where genome sequencing in the CSF confirmed the presence of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (Li et al., 2020).
In patients with mild or moderate respiratory illness, neurological symptoms are nonspecific, like headache or vertigo, or can be more specific but still not disabling, like hypogeusia and hyposmia (Lechien et al., 2020, Li et al., 2020, Mao et al., 2020, Wu et al., 20200). In more severe respiratory illness, patients can develop serious neurologic symptoms and disorders of consciousness, which have been associated either to acute toxic encephalitis due to systemic toxemia, metabolic disorders, and hypoxia (Li et al., 2020, Mao et al., 2020, Wu et al., 20200) or to inflammatory cerebrovascular injury, closely linked to the development of a systemic inflammatory response syndrome (SIRS) (Li et al., 2020, Mao et al., 2020, Wu et al., 20200). Recently, a case of Covid-19-associated acute necrotizing hemorrhagic encephalopathy has been reported. While direct viral invasion could not be demonstrated, intracranial cytokine storm was proposed as a key factor, resulting in blood–brain barrier break-down (Poyiadji et al., 2020).
Here we describe the case of a Covid-19 patient undergoing mechanical ventilation due to acute respiratory distress syndrome (ARDS), who developed neurological symptoms in the intensive care unit (ICU), for whom intensive EEG follow-up contributed the discovery of Critical Illness-Associated Cerebral Microbleeds.
2. Case report
A 56-year-old woman with a history of tobacco smoking-induced pulmonary emphysema and hypothyroidism, following symptoms of cough and fever was diagnosed positive to SARS-Cov2-PCR on nasopharyngeal swab test. Antibiotics were administered and patient recovered at home. After ten days, she developed a respiratory failure and she was admitted to ICU at the University Hospitals of Geneva. On admission, she was febrile (39.6 °C) and presented clinical and imaging signs of pneumonia. The rest of the physical examination was normal, in particular the neurological examination did not show any abnormalities. The patient was conscious and orientated, with no sensory or motor deficits. Blood test results showed a normal white blood cell count, with absolute lymphopenia (0.61 g/l) and reduction of CD4+ (212/microl) and particularly of CD8+ (101/microl), no anemia, a normal platelet count, elevated C-reactive protein level (52.6 mg/l), undetectable procalcitonin level, hypoalbuminemia (28 g/l) and hypoproteinemia (52 g/l). Fibrinogen was slightly elevated (5 g/l), PTT was 38.1 sec and Quick was normal. Hepatic and renal tests were normal. Heart function was not affected. ARDS related to Covid-19 was diagnosed, and the patient underwent endotracheal intubation with mechanical ventilation support, prophylactic antibiotic and anticoagulant therapies. The respiratory evolution was favorable and the patient was weaned from mechanical ventilation after 14 days.
On the 10th day in the ICU, a partial withdrawal of sedation was initiated, but the patient became agitated motivating the re-administration of sedation for another 4 days.
After sedative drugs and mechanical ventilation withdrawal, she was vigilant but not following simple orders. Vital signs were in the normal range. Neurological examination did not show any obvious sensitive or motor deficits. Hematologic profile showed leukocytosis (maximum 20.3 g/l) with persistent lymphopenia, anemia (minimum Hb 92 g/l) and thrombocytosis (maximum 602 g/l). Elevated C-reactive protein (161 mg/l) and ferritin (490 microg/l) and persistent hypoalbuminemia were also noted. Fibrinogen and D-dimer were increased (maximum fibrinogen 9 g/l and maximum D-dimer 2141 ng/ml), but Quick and PTT remained stable. Cardiovascular profile, electrolytes, renal and hepatic functions were stable. Procalcitonin remained undetectable.
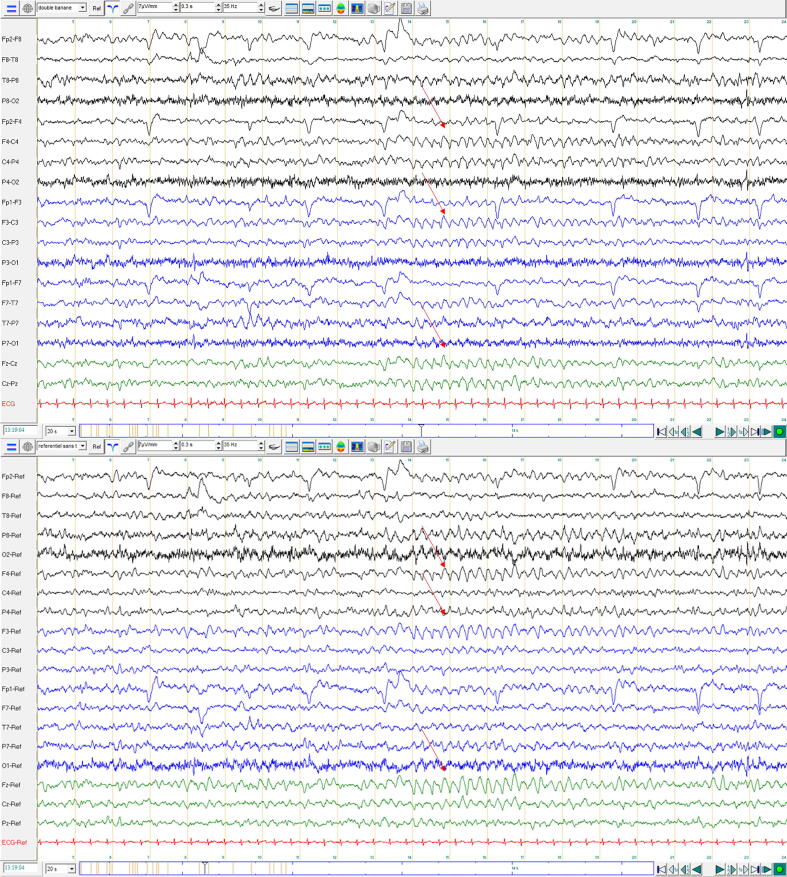
A first routine video-EEG on day 18 was performed, which revealed an intermittent onset of 4 Hz rhythms over the bilateral parasagittal regions (Fig. 1), lasting from 5 s to maximum 25 s. A benzodiazepine infusion, however, did not show either clinical or electroencephalographic improvement.
Fig. 1.
First EEG findings. 20 s of 19-channel EEG, referenced to bipolar (upper window) and average (lower window) reference, showing intermittent 4 Hz activity in the bilateral parasagittal region (as shown). Background posterior activity at 8 Hz is unstable but symmetric. Black lines: recordings from the right hemisphere. Blue lines: recordings from the left hemisphere. Green lines: midlines traces. Distance between orange vertical bars: 1 s. Patient was awake during this period of recording (exploring the space, not speaking, not answering to simple verbal orders). (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
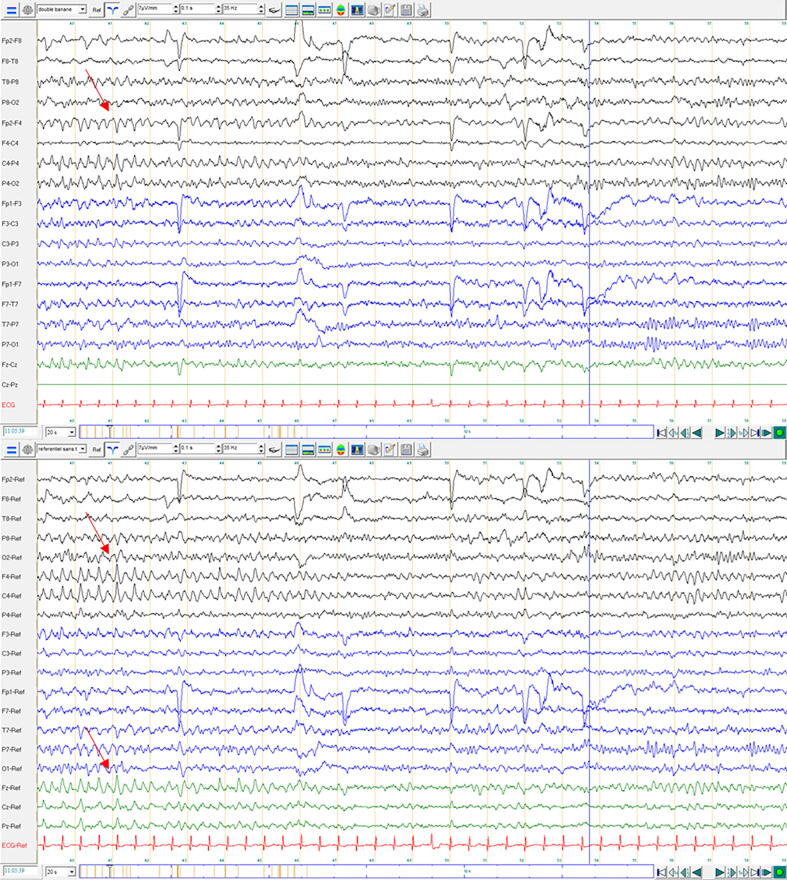
In order to determine the evolution of this pattern, a second EEG one day later was performed. It showed the same pattern with preserved background rhythms, suggesting focal injury in the absence of encephalopathy (Fig. 2).
Fig. 2.
EEG findings at day 19 (second EEG): 20 s per page, 19-channel EEG referenced to bipolar (upper window) and average (lower window) reference, showing intermittent, 4 Hz theta activity in the central-frontal region here with right more than left predominance. Background activity showing symmetric synchronous 10 Hz. Black lines: recordings from the right hemisphere. Blue lines: recordings from the left hemisphere. Green lines: midlines traces. Distance between orange vertical bars: 1 s. Electrical bridge artifact between Cz-Pz. Patient was awake during this period of recording but unresponsive (eyes open, exploring the space, not speaking, not following simple verbal orders). (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
Three days later, a third and last EEG did not find any significant changes. Patient presented normal sleep pattern (up to stage II) without any additional abnormalities.
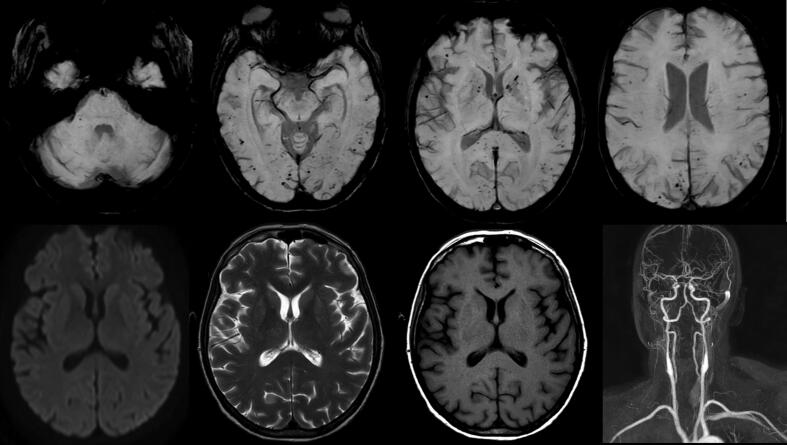
A brain MRI was performed at day 19. Susceptibility-weighed imaging (SWI)/T* sequences revealed numerous punctiform signal voids in bilateral juxtacortical white matter, corpus callosum, and internal capsule, compatible with cerebral microbleeds, without any ischemic or necrotizing lesion. Cervical and cerebral-MRI angiography was normal with no sign of stenosis or vasculitis (Fig. 3).
Fig. 3.
MRI findings. Upper row: Transverse SWI/T*showing diffuse microbleeds in bilateral juxtacortical white matter, corpus callosum, and internal capsule. Lower row from left to right: Diffusion weighted imaging, T2-weighted and T1-weighted, not showing any parenchymal lesion. 3D angiogram reconstruction not showing any focal stenosis of the extracranial and intracranial vessels. Right hemisphere is on the left side of the image.
We completed investigations with a lumbar puncture. CSF analysis showed normal cell count but revealed an increased protein level (1.31 g/l) and Immunoglobulins, with IgG oligoclonal bands type IV (identical in CSF and serum without intrathecal production). SARS-Cov2 RNA-PCR in CSF was negative.
Cognition and vigilance improved after 10 days from the first EEG with normalization of orientation and language, but persistent slight executive dysfunction.
3. Discussion
Cerebral microbleeds are MRI-visible small (generally 2–5 mm in diameter, up to 10 mm) focal areas of signal void caused by perivascular collections of hemosiderin deposits resulting from tiny blood leakages from damaged arteriolar walls (Pantoni 2010). Even though they can rarely be associated with diffuse axonal injury, high-altitude exposure, or brain radiation therapy, the most common etiologies are cerebral amyloid angiopathy and chronic hypertension (which our patient did not suffer from). In the former case, microbleeds are typically cortical; in the latter, they affect the basal ganglia and thalamus and the periventricular white matter (Pantoni 2010).
Recently, cases of Critical Illness-Associated Cerebral Microbleeds have been documented in patients undergoing mechanical ventilation because of respiratory failure (Fanou et al., 2017, Kuo et al., 2019), in particular if suffering from systemic diseases (neoplastic or infectious, including viral). Here, microhemorrhages did not involve periventricular white matter, cortex, basal ganglia and thalami, but were instead predominant in the juxtacortical white matter and corpus callosum (Riech et al., 2015, Fanou et al., 2017).
The pathogenesis of the condition remains unclear, but two mechanisms are proposed. The first suggests that hypoxemia leads to hydrostatic or chemical changes in the blood–brain barrier, which result in extravasation of erythrocytes. The second mechanism involves cerebrovascular inflammation, due to either the increase of cytokine levels or to endothelial dysfunction in the setting of intravascular coagulation (Pantoni 2010). The relationship between cerebral microbleeds and SIRS has been confirmed by the fact that multifocal hemorrhages feature in the neurotoxicity due to CD19 CAR-T cell therapy for B-cell malignancies (Gust et al., 2017).
In our patient, a direct infectious injury was ruled out by the CSF analysis, which excluded encephalitis and showed no hint of the presence of SARS-CoV-2, but increased protein and oligoclonal bands type IV suggestive of increase in permeability of the blood–brain barrier.
We conclude that the radiological picture in our patient, resembling Critical Illness–Associated Cerebral Microbleeds, is probably related to a cytokine release syndrome, together with alterations of brain blood barrier permeability caused by hypoxia (Fanou et al. 2017). Most likely, this has been promoted by inefficient mechanisms of adaptive immunity as suggested by her lymphopenia with reduction of both CD4+ and CD8+. A disseminated intravascular coagulation was excluded due to a normal PTT and lack of increase of fibrinogen despite the increase of D-dimer.
4. Conclusions
To our best knowledge, this is the first reported case of Critical Illness-Associated Cerebral Microbleeds related to SARS-CoV-2 infection. Knowledge of Covid-19 is still partial and acute neurological complications should be explored systematically.
In our case, EEG helped to rule out non-convulsive status epilepticus, but revealed focal dysfunction, justifying further investigations.
We suggest that EEG should be requested systematically for these patients. This is particularly helpful for Covid-19 patients in the ICU, who often present an altered mental status.
Acknowledgments
Acknowledgements
Pia De Stefano is supported by the SNF grant No 163398. PM is supported by an Ambizione fellowship from the SNF (PZ00P3_167836).
Conflicts of interest and financial disclosures
PDS, UN, LDS, PM, MS report no conflicts of interest.
PDS, UN, LDS, PM, MS report no disclosures.
Contributor Information
Pia De Stefano, Email: Pia.destefano@hcuge.ch.
Umberto Nencha, Email: umberto.nencha@hcuge.ch.
Ludovico De Stefano, Email: ludovico.destefano01@universitadipavia.it.
Pierre Mégevand, Email: pierre.megevand@hcuge.ch.
Margitta Seeck, Email: margitta.seeck@hcuge.ch.
References
- Fanou E.M., Coutinho J.M., Shannon P., Kiehl T.R., Levi M.M., Wilcox M.E. Critical illness-associated cerebral microbleeds. Stroke. 2017;48:1085–1087. doi: 10.1161/STROKEAHA.116.016289. [DOI] [PubMed] [Google Scholar]
- Gust J., Hay K.A., Hanafi L.-A., Li D., Myerson D., Gonzalez-Cuyar L.F. Endothelial activation and blood-brain barrier disruption in neurotoxicity after adoptive immunotherapy with CD19 CAR-T cells. Cancer Discov. 2017;7:1404–1419. doi: 10.1158/2159-8290.CD-17-0698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo K.H., Bin Yu.Y., Lai T.H. Cerebral microbleeds associated with critical illness. Acta Neurol. Taiwan. 2019;28:25–26. [PubMed] [Google Scholar]
- Lechien J.R., Chiesa-Estomba C.M., De Siati D.R., Horoi M., Le Bon S.D., Rodriguez A. Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): a multicenter European study. Eur. Arch. Otorhinolaryngol. 2020;6:1–11. doi: 10.1007/s00405-020-05965-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Z., Huang Y., Guo X. The brain, another potential target organ, needs early protection from SARS-CoV-2 neuroinvasion. Sci. China Life Sci. 2020;63:771–773. doi: 10.1007/s11427-020-1690-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mao L., Jin H., Wang M., Hu Y., Chen S., He Q. Neurologic manifestations of hospitalized patients with coronavirus Disease 2019 in Wuhan, China. JAMA Neurol. 2020;10 doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pantoni L. Cerebral small vessel disease: from pathogenesis and clinical characteristics to therapeutic challenges. Lancet Neurol. 2010;9:689–701. doi: 10.1016/S1474-4422(10)70104-6. [DOI] [PubMed] [Google Scholar]
- Poyiadji N., Shahin G., Noujaim D., Stone M., Patel S., Griffith B. COVID-19–associated acute hemorrhagic necrotizing encephalopathy: CT and MRI features. Radiology. 2020;2 doi: 10.1148/radiol.2020201187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riech S., Kallenberg K., Moerer O., Hellen P., Bärtsch P., Quintel M. The pattern of brain microhemorrhages after severe lung failure resembles the one seen in high-altitude cerebral edema. Crit. Care Med. 2015;43:e386–e389. doi: 10.1097/CCM.0000000000001150. [DOI] [PubMed] [Google Scholar]
- Wu Y., Xu X., Chen Z., Duan J., Hashimoto K., Yang L. Nervous system involvement after infection with COVID-19 and other coronaviruses. Brain Behav. Immun. 2020;S0889–1591(20):30357–30363. doi: 10.1016/j.bbi.2020.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]