Abstract
Purpose:
Helicobacter pylori (H. pylori) is the leading cause of gastric cancer. High antibody levels to H. pylori virulence factors Vacuolating cytotoxin A (VacA) and Cytotoxin-associated gene A (CagA) have been suggested as gastric cancer risk markers. In the US, H. pylori sero-prevalence is two-fold higher in African Americans compared to whites. We sought to assess whether African Americans also exhibit higher antibody levels to VacA and CagA.
Methods:
Antibody responses to H. pylori proteins were measured by multiplex serology in 686 African Americans and whites of the Southern Community Cohort Study. Among VacA- and CagA-seropositives, we analyzed the association of race with antibody level using logistic regression models to produce odds ratios (OR) and 95% confidence intervals (CI).
Results:
Sero-positive African Americans had significantly higher mean antibody levels to both VacA and CagA, which resulted in increased odds for the highest quartile of antibody levels compared to sero-positive whites (VacA, OR: 6.08; 95% CI: 3.41, 10.86; CagA, OR: 3.77; 95% CI: 1.61, 8.84).
Conclusion:
Our findings support future studies to assess the association of differential antibody responses by race with risk of gastric cancer in the US, which could then aid in developing targeted H. pylori eradication strategies.
Keywords: Helicobacter pylori, serology, antibody level, disparity
INTRODUCTION
Helicobacter pylori (H. pylori) infection is the leading cause for gastric cancer. Infection with H. pylori causes gastritis, which may become chronic. This chronic inflammation in turn leads to morphological changes in the gastric epithelium transitioning from chronic atrophic gastritis to intestinal metaplasia, dysplasia, and, in 1-3% of H. pylori-infected individuals, to gastric cancer [1]. It is not fully resolved to date why only a proportion of infected individuals develop cancer and ongoing research targets biomarkers to identify individuals at increased gastric cancer risk early along the cascade of events leading to carcinogenesis. Serology provides an easy-to-apply non-invasive methodology and indeed H. pylori sero-positive individuals, and more specifically those sero-positive to virulence factors Vacuolating cytotoxin A (VacA) and Cytotoxin-associated gene A (CagA), were found to be at an approximately 2- to 4-fold increased risk of developing gastric cancer [2, 3]. Some studies have evaluated the antibody response to H. pylori quantitatively and results indicate that level of antibody response adds information beyond sero-positivity alone. High antibody levels were found to be associated with gastric mucosal inflammation, grade of histological gastritis, mucosal bacterial density, and gastric cancer risk [4-6].
H. pylori infects the stomach mucosa in approximately half of the world’s population with varying prevalence by geographic region and population [7]. In the US for instance, the overall H. pylori sero-prevalence was 31% among adults aged 20 years or older in a national survey from 1999-2000. However, the prevalence varied by race, with 52% among African Americans compared to 21% among whites [8]. This largely reflects the disparity in non-cardia gastric cancer incidence, the gastric cancer sub-type most strongly associated with H. pylori, for which the age-standardized incidence rate for 1995 to 2013 was 6.43 in African Americans and 2.16 per 100,000 person-years in whites [9]. Moreover, gastric cancer accounts for the greatest racial disparity in mortality from cancer, being over two-fold higher in African Americans compared to whites, suggesting the need for improved screening and prevention strategies [10, 11]. Despite the significantly higher H. pylori sero-prevalence in African Americans compared to whites there are no reports available whether African Americans also exhibit higher level of antibody response. In the present study we sought to assess whether African Americans are more likely to have higher antibody levels to VacA and CagA than whites in the Southeast US.
METHODS
Study population
The SCCS is a prospective cohort study that recruited approximately 86,000 men and women aged 40 to 79 from 12 southeastern US states at community health centers (CHCs) (86%) or by mail (14%) between 2002 and 2009 [12]. Participants completed a survey and about half of those recruited at CHCs provided a blood sample at baseline. We retrieved stored blood samples from the original 2 x 2 x 3 x 3 factorial design, whereby 792 individuals were randomly selected from the 12,162 SCCS participants with a blood sample given at baseline who were enrolled from March 2002 to October 2004, with strata defined by self-reported race (African American, white), sex (male, female), smoking status (never, former, current), and body mass index (BMI, 18-24.9, 25-29.9, 30-45 kg/m2) [13]. Of these, 686 participants are included in the present analysis as the available serum was depleted from other assays performed on this group for 77 (9.7%), samples for 3 (0.4%) individuals were unusable because of serum handling issues, 3 individuals (0.4%) were missing information on antibiotic use, and the ancestry estimates for 23 (2.9%) were highly discordant with self-reported race, implying potential data entry errors. Fifty microliters of serum were aliquoted and sent to the German Cancer Research Center (DKFZ, Heidelberg, Germany) on dry ice for multiplex serology analysis.
H. pylori multiplex serology
Antibody responses to 15 H. pylori proteins (GroEl (strain G27), UreA, HP0231, NapA, HP0305, CagM, CagD, CagA, HpaA, Catalase, HyuA, VacA, Cad, HcpC, HP1564 (all strain 26695)) were measured by multiplex serology [13, 14]. H. pylori proteins were recombinantly expressed as GST-tag fusion proteins and affinity-purified on fluorescent polystyrene beads (Luminex Corp., Austin, TX, USA). A mixture of the differently labeled antigen-loaded beads was incubated with serum and the amount of bound serum antibody was quantified using a biotin-labelled secondary anti-human IgG/IgA/IgM antibody and subsequent labelling with streptavidin-R-phycoerythrin. A Luminex flow cytometer (Luminex Corp., Austin, TX, USA) then distinguished the bead-type and consequently the bound antigen as well as quantified the amount of bound serum antibody on at least 100 beads per type measured with the median fluorescence intensity (MFI) as output. Antigen-specific cut-offs were defined as described previously [13] and overall H. pylori sero-positivity was defined as being sero-positive to at least 4 of the 15 proteins included. Five replicate samples from each of two randomly selected individuals were analyzed for antibody responses to all 15 H. pylori proteins as quality control. We observed only one discordant result among the five replicate samples of one of the quality control individuals in sero-positivity to antigen HP0231. All other results replicated concordantly for both quality control individuals.
Statistical analysis
Differences in H. pylori sero-status and study characteristics between African Americans and whites were assessed using Chi-squared test for categorical variables and Wilcoxon rank-sum test for continuous variables.
Since antibody level to VacA and CagA were not normally distributed we calculated geometric means and 95% confidence intervals (CI) among H. pylori-antigen sero-positive individuals and compared these by race applying t-tests. In addition, antibody responses to VacA and CagA were divided into quartiles based on the distribution of these levels among all H. pylori-antigen-positive individuals (not race-specific). The association of race (African Americans compared to whites) with increasing antibody level quartile as outcome was then compared to the lowest quartile using unconditional logistic regression models to produce odds ratios (OR) and 95% CI Factors considered a priori as potentially relating to level of an individual’s immune response and therefore considered to be included in the model were original stratification factors sex, BMI, and smoking, as well as a medication history of regular aspirin intake and antibiotics use within one year prior to recruitment, and other socio-demographic factors including socio-economic status (education and household income) and the participant’s age [15-20]. Excluding the factors by which this population was stratified by (sex, BMI, and smoking status), all other factors except for regular aspirin intake were significantly different by race and were included in the model to test for the association of antibody level to VacA and CagA with adjustment for these factors. We finally present three models: a) unadjusted, b) adjusted for sex (male, female), BMI (18-24.9, 25-29.9, 30-45 kg/m2), and smoking status (never, former, current), c) additional to adjustment in model b) adjusted for age (continuous), education (<High school, High school/GED, >High school), household income (<$15,000, $15,000–<$25,000, ≥$25,000), and antibiotics use during the past year before recruitment (yes/no). Five participants were lacking information on household income and where therefore excluded from model c).
RESULTS
In univariate analysis African Americans were significantly younger than whites and more likely to have a lower educational level and household income per year but less likely to having taken antibiotics within the past year before recruitment (Table 1). With respect to H. pylori, African Americans were more likely to be overall H. pylori sero-positive than whites (89% versus 69%, respectively) and among H. pylori sero-positives the sero-prevalence of VacA- and CagA was also higher among African Americans than among whites (VacA sero-positivity 94% vs. 88%, respectively; CagA sero-positivity 75% vs. 36%, respectively) (Table 1). Among these H. pylori-antigen sero-positive study participants, African Americans had significantly higher average antibody levels than whites to VacA (African Americans, geometric mean MFI (95% CI): 2,330 (2,079, 2,611); whites, geometric mean MFI (95% CI): 1,165 (998, 1,359), P < 0.001) and CagA (African Americans, geometric mean MFI (95% CI): 18,228 (17,044, 19,494); whites geometric mean MFI (95% CI): 12,783 (10,941, 14,831), P < 0.001) (Table 1).
Table 1:
Study characteristics of whites and African Americans in the Southern Community Cohort Study factorial design.
| Whites | African Americans | |||
|---|---|---|---|---|
| Age, [years], median (IQR)a | 52 | (45, 60) | 49 | (45, 57) |
| Education, n (%)b | ||||
| <High school | 90 | (26) | 120 | (35) |
| High school/GED | 139 | (41) | 141 | (41) |
| >High school | 111 | (33) | 85 | (25) |
| Household income [$], n (%)b | ||||
| <15,000 | 205 | (61) | 208 | (61) |
| 15,000- <25,000 | 62 | (18) | 84 | (24) |
| ≥25,000 | 71 | (21) | 51 | (15) |
| Medication use | ||||
| Antibiotics within last year yes, n (%)b | 187 | (55) | 148 | (43) |
| Regular Aspirin intake yes, n (%)b | 278 | (82) | 301 | (87) |
| H. pylori sero-status | ||||
| Overall H. pylori+, n (%)b | 234 | (69) | 308 | (89) |
| VacA+ among H. pylori+, n (%)b | 206 | (88) | 291 | (94) |
| Antibody level [MFI], geometric mean (95% CI)c | 1,165 | (998, 1,359) | 2,330 | (2,079, 2,611) |
| CagA+ among H. pylori+, n (%)b | 85 | (36) | 232 | (75) |
| Antibody level [MFI], geometric mean (95% CI)c | 12,738 | (10,941, 14,831) | 18,228 | (17,044, 19,494) |
Abbreviations: +, sero-positive; BMI, body mass index; CagA, cytotoxin-associated gene A; CI: confidence interval; GED, General Education Development; IQR, interquartile range; VacA, Vacuolating cytotoxin A
P<0.05 in Wilcoxon rank-sum test
P<0.05 in Chi-Square test
P<0.05 in Student’s t-test
When analyzed in quartiles this resulted in significantly increased odds for high antibody levels in H. pylori-antigen-positive African Americans compared to whites (Q4 versus Q1: VacA, OR: 5.40; 95% CI: 3.14, 9.29; Q4 versus Q1: CagA, OR: 4.34; 95% CI: 2.04, 9.26). These associations were significant above and beyond adjustment for matching variables BMI, smoking and sex (Q4 versus Q1: VacA, OR: 5.81; 95% CI: 3.32, 10.18; Q4 versus Q1: CagA, OR: 4.24; 95% CI: 1.93, 9.31) as well as other socio-demographic and medical history factors that might affect the immune response to pathogens further including age, socio-economic status and antibiotics use prior to recruitment (Q4 versus Q1: VacA, OR: 6.08; 95% CI: 3.41, 10.86; Q4 versus Q1: CagA, OR: 3.77; 95% CI: 1.61, 8.84) (Figure 1).
Figure 1: Association of race with antibody level to VacA and CagA among H. pylori-antigen-positive individuals.
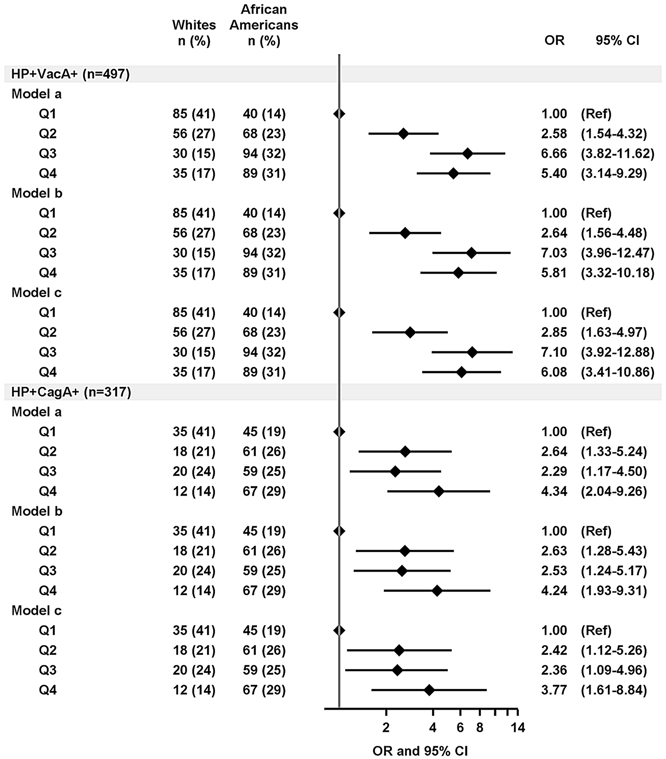
The association of African American race compared to whites with being in the higher antibody quartile (Q2, Q3, and Q4) in reference to Q1 was determined using logistic regression models to compute odds ratios (OR) and 95% confidence intervals (CI). Model (a): unadjusted; model (b): adjusted for sex (male, female), BMI (18-24.9, 25-29.9, 30-45 kg/m2), and smoking status (never, former, current); model (c): additional adjustment for age (continuous), education (<High school, High school/GED, >High school), household income ($, <15,000, 15,000- <25,000, ≥25,000), and antibiotics use during the past year before recruitment (yes/no). Quartile borders (Q1, Q2, Q3, Q4) were based on the overall sample set among H. pylori and antigen –positives (VacA: Q1: 200-712 MFI, Q2: >712-1,915 MFI, Q3: >1,915-4,063 MFI, Q4: >4,063 MFI; CagA: Q1: 3,091-14,475, Q2: >14,475-21,079, Q3: >21,079-26,219, Q4: >26,219).
DISCUSSION
In the present study, we report that not only sero-prevalence to H. pylori significantly differs by race in a predominantly low-income population in the southern US but that also the level of antibody response to virulence factors CagA and VacA among sero-positives is higher in African Americans than whites in this population. This association remained significant even after adjustment for factors potentially interfering with the host’s immune response, including smoking, BMI, sex, socio-economic status as a measure of stress, antibiotic use, and the participant’s age [15-20].
To our knowledge this is the first study in African Americans compared to whites to look at the level of humoral immune response to H. pylori, and more specifically its virulence factors VacA and CagA, and our findings may lead to further research questions in the interest of examining and remediating racial health disparities. It has been previously described that the inflammatory response to certain pathogens is stronger in African Americans than in whites [21] and that level of circulating pro-inflammatory cytokines also varies by race [22], which may have implications for inflammation-related outcomes like gastric cancer. Therefore, our findings warrant clarification of whether increased antibody levels in African Americans also relate to stronger host inflammatory responses being an important factor in gastric carcinogenesis. Bacterial factors that should be considered in this respect include bacterial load, and the isoform of virulence factor (e.g. allotypes s1m1, s1m2, and s2m2 for VacA and different CagA EPIYA motifs) as well as the amount of virulence factor expressed. Ultimately, it needs to be elucidated whether these factors may contribute to the racial disparity in gastric cancer incidence and mortality in the US [10].
Although the present study’s design with matching on race, sex, BMI and smoking status allowed us to assess the association of antibody level to H. pylori virulence factors VacA and CagA and controlled for factors that might impact the host’s immune response, the study also had limitations. The measure of antibiotic treatment within the past year before recruitment was self-reported and the influence of antibiotic treatment on antibody level therefore might have been underestimated in this study. Moreover, the H. pylori sero-prevalence in the investigated population was above the national average for African Americans (89% versus 52%) and whites (69% versus 21%), likely due to the predominantly low socio-economic status of the SCCS cohort, and results may therefore not be generalizable [8]. These findings therefore would need to be validated in an independent study. Additionally, differences in access to care may relate to differences in antibody levels, as racial differences in appropriate testing for H. pylori among symptomatic patients have been found [23], however as the vast majority of individuals with chronic H. pylori infection are asymptomatic, we do not think this would account for the significant results found.
In conclusion, we found that among H. pylori sero-positive individuals in the SCCS, African Americans had significantly higher levels of antibody response to both VacA and CagA compared to whites. Future studies will need to assess the association of the observed differential antibody response to the main gastric cancer related H. pylori virulence factors CagA and VacA with an increased risk of developing gastric cancer in the US, which could then aid in developing targeted H. pylori eradication strategies for populations with increased gastric cancer risk.
Acknowledgments
Funding
The Southern Community Cohort Study (SCCS) is funded by grant U01 CA202979 from the National Cancer Institute at the National Institutes of Health. SCCS data collection was performed by the Survey and Biospecimen Shared Resource which is supported in part by the Vanderbilt-Ingram Cancer Center (P30 CA68485).
Footnotes
Publisher's Disclaimer: This Author Accepted Manuscript is a PDF file of an unedited peer-reviewed manuscript that has been accepted for publication but has not been copyedited or corrected. The official version of record that is published in the journal is kept up to date and so may therefore differ from this version.
REFERENCES
- 1.Correa P, Houghton J. (2007) Carcinogenesis of Helicobacter pylori. Gastroenterology. 133: 659–72. [DOI] [PubMed] [Google Scholar]
- 2.Epplein M, Zheng W, Xiang YB, et al. (2012) Prospective study of Helicobacter pylori biomarkers for gastric cancer risk among Chinese men. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 21: 2185–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gao L, Michel A, Weck MN, Arndt V, Pawlita M, Brenner H. (2009) Helicobacter pylori infection and gastric cancer risk: evaluation of 15 H. pylori proteins determined by novel multiplex serology. Cancer research. 69: 6164–70. [DOI] [PubMed] [Google Scholar]
- 4.Shiota S, Murakami K, Okimoto T, Kodama M, Yamaoka Y. (2014) Serum Helicobacter pylori CagA antibody titer as a useful marker for advanced inflammation in the stomach in Japan. Journal of gastroenterology and hepatology. 29: 67–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tu H, Sun L, Dong X, et al. (2014) Serum anti-Helicobacter pylori immunoglobulin G titer correlates with grade of histological gastritis, mucosal bacterial density, and levels of serum biomarkers. Scandinavian journal of gastroenterology. 49: 259–66. [DOI] [PubMed] [Google Scholar]
- 6.Yoshida T, Kato J, Inoue I, et al. (2014) Cancer development based on chronic active gastritis and resulting gastric atrophy as assessed by serum levels of pepsinogen and Helicobacter pylori antibody titer. International journal of cancer. Journal international du cancer. 134: 1445–57. [DOI] [PubMed] [Google Scholar]
- 7.Hooi JKY, Lai WY, Ng WK, et al. (2017) Global Prevalence of Helicobacter pylori Infection: Systematic Review and Meta-Analysis. Gastroenterology. 153: 420–9. [DOI] [PubMed] [Google Scholar]
- 8.Grad YH, Lipsitch M, Aiello AE. (2012) Secular trends in Helicobacter pylori seroprevalence in adults in the United States: evidence for sustained race/ethnic disparities. American journal of epidemiology. 175: 54–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Anderson WF, Rabkin CS, Turner N, Fraumeni JF Jr., Rosenberg PS, Camargo MC. (2018) The Changing Face of Noncardia Gastric Cancer Incidence Among US Non-Hispanic Whites. Journal of the National Cancer Institute. 110: 608–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ashktorab H, Kupfer SS, Brim H, Carethers JM. (2017) Racial Disparity in Gastrointestinal Cancer Risk. Gastroenterology. 153: 910–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.DeSantis CE, Miller KD, Goding Sauer A, Jemal A, Siegel RL. (2019) Cancer statistics for African Americans, 2019. CA Cancer J Clin. 69: 211–33. [DOI] [PubMed] [Google Scholar]
- 12.Signorello LB, Hargreaves MK, Steinwandel MD, et al. (2005) Southern community cohort study: establishing a cohort to investigate health disparities. Journal of the National Medical Association. 97: 972–9. [PMC free article] [PubMed] [Google Scholar]
- 13.Epplein M, Signorello LB, Zheng W, et al. (2011) Race, African ancestry, and Helicobacter pylori infection in a low-income United States population. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 20: 826–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Michel A, Waterboer T, Kist M, Pawlita M. (2009) Helicobacter pylori multiplex serology. Helicobacter. 14: 525–35. [DOI] [PubMed] [Google Scholar]
- 15.Padgett DA, Glaser R. (2003) How stress influences the immune response. Trends Immunol. 24: 444–8. [DOI] [PubMed] [Google Scholar]
- 16.Scepanovic P, Alanio C, Hammer C, et al. (2018) Human genetic variants and age are the strongest predictors of humoral immune responses to common pathogens and vaccines. Genome medicine. 10: 59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sopori M (2002) Effects of cigarette smoke on the immune system. Nat Rev Immunol. 2: 372–7. [DOI] [PubMed] [Google Scholar]
- 18.Stepanikova I, Bateman LB, Oates GR. (2017) Systemic Inflammation in Midlife: Race, Socioeconomic Status, and Perceived Discrimination. Am J Prev Med. 52: S63–S76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Struve J, Aronsson B, Frenning B, Granath F, von Sydow M, Weiland O. (1992) Intramuscular versus intradermal administration of a recombinant hepatitis B vaccine: a comparison of response rates and analysis of factors influencing the antibody response. Scandinavian journal of infectious diseases. 24: 423–9. [DOI] [PubMed] [Google Scholar]
- 20.Weber DJ, Rutala WA, Samsa GP, Santimaw JE, Lemon SM. (1985) Obesity as a predictor of poor antibody response to hepatitis B plasma vaccine. JAMA : the journal of the American Medical Association. 254: 3187–9. [PubMed] [Google Scholar]
- 21.Nedelec Y, Sanz J, Baharian G, et al. (2016) Genetic Ancestry and Natural Selection Drive Population Differences in Immune Responses to Pathogens. Cell. 167: 657–69 e21. [DOI] [PubMed] [Google Scholar]
- 22.Yao S, Hong CC, Ruiz-Narvaez EA, et al. (2018) Genetic ancestry and population differences in levels of inflammatory cytokines in women: Role for evolutionary selection and environmental factors. PLoS Genet. 14: e1007368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Florea A, Brown HE, Harris RB, Oren E. (2019) Ethnic Disparities in Gastric Cancer Presentation and Screening Practice in the United States: Analysis of 1997–2010 Surveillance, Epidemiology, and End Results-Medicare Data. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 28: 659–65. [DOI] [PMC free article] [PubMed] [Google Scholar]


