Abstract
Metastasis, a leading contributor to the morbidity of cancer patients, occurs through a multi-step process: invasion, intravasation, extravasation, colonization, and metastatic tumor formation. Each process is not only promoted by cancer cells themselves but is also affected by their microenvironment. Given this complexity, drug discovery for anti-metastatic drugs must consider the interaction between cancer cells and their microenvironments. The zebrafish is a suitable vertebrate animal model for in vivo high-throughput screening studies with physiological relevance to humans. This review covers the zebrafish model used to identify anti-metastatic drugs.
Keywords: zebrafish, metastasis, EMT, angiogenesis, phenotyping screening
1. Introduction
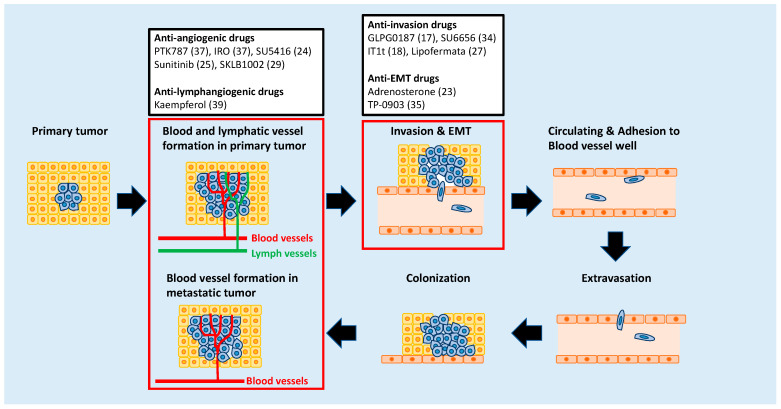
Overt metastases, the end result of malignant alteration in cancer cells, are responsible for approximately 90% of cancer-associated mortality. Metastasis consists of a multi-step process: invasion in which neoplastic epithelial cells invade into the adjacent tissue after they lose cell-cell adhesion; intravasation in which tumor cells penetrate through the endothelium of blood or lymphatic vessels to enter the systemic circulation; survival in the circulatory system in which certain circulating tumor cells appear able to survive in the bloodstream; extravasation in which cancer cells extravasate through the capillary endothelium at distal sites; colonization in which cancer cells proliferate in a new environment, and metastatic tumor formation in which cancer cells form a new tumor in secondary organs (Figure 1) [1,2,3]. Recent studies demonstrated that each of these processes is promoted not only by cancer cells but also by the tumor microenvironment [4,5]. Therefore, the interaction between cancer cells with their microenvironments must be considered while developing therapeutic strategies for metastasis.
Figure 1.
Anti-metastatic drugs identified through zebrafish-based screening that target different stages of metastasis. The numbers in parentheses indicate reference.
From the therapeutic point of view, the metastatic process can be divided into two phases: the early phase including tumor cell invasion and extravasation, and the late phase consisting of colonization and metastatic tumor formation. During the former phase, anti-metastatic treatments focus on inhibiting the spread of cancer cells to distant organs. Likewise, the goal of treatment during the latter phase is to inhibit the proliferation of disseminated cancer cells in distant organs [6].
Cancer research using zebrafish as a model has attracted attention because this model offers many unique advantages that are not readily provided by other animal models [7,8,9]. For example, (i) zebrafish have orthologues to 86% of 1318 human drug targets, 71% of human proteins, and 82% of disease-causing human proteins [10,11]; (ii) drugs are administered to zebrafish by dissolving them in water, except hydrophobic drugs that are administered to the zebrafish by microinjection [12,13]; (iii) the effects of chemicals may be evaluated through direct observation due to the transparency of zebrafish embryos; (iv) zebrafish generate large numbers of progeny, thus increasing the power of statistical analyses; (v) husbandry expenses associated with zebrafish are much lower than for mammals due to their low-maintenance nature. These advantages have made zebrafish a popular platform for drug screening.
However, there are several limitations associated with zebrafish. One limitation of zebrafish is that they are not mammals. Therefore, the effects of the drugs observed in zebrafish models must be validated in advanced preclinical models. Another limitation of using zebrafish for in vivo drug screening is that it is difficult to test drugs that are insoluble in water. A previous study that screened 23 drugs known to cause cardiotoxicity in humans yielded four out of five false-negative results in zebrafish due to poor absorption. The efficacy of drugs that yielded false negatives was confirmed by microinjection studies [13]. The other limitation associated with zebrafish is that the optimum temperature for breeding zebrafish is 28 °C; this temperature differs from that of the human body by 9 °C (37 °C). In xenograft experiments that involve the transplantation of human or mouse cells into zebrafish, the zebrafish maintained a temperature of 31–34 °C, since 28 °C is not optimal for the growth of the transplanted cells. However, previous studies demonstrate that human cancer cells that are inoculated into zebrafish embryos have a better proliferation index at 36 °C than at 34 °C. Therefore, 36 °C is considered the most suitable temperature for testing chemotherapeutic drugs such as 5-fluorouracil. Although the zebrafish is a poikilothermic and eurythermal animal, there was a 10% increase in the mortality rate of zebrafish maintained at 36 °C compared to that of zebrafish at 34 °C [14].
Several anti-metastatic drugs have been identified through studies using zebrafish models [15]. The transplantation of cancer cells into zebrafish embryos is most frequently used in these models. Briefly, human cancer cells labeled with a fluorescent marker such as enhanced green fluorescent protein (EGFP) are inoculated into larvae or embryos of zebrafish. This is because zebrafish embryos are transparent and do not have a fully developed adaptive immune system until 21 days post fertilization (dpf) [16]. Zebrafish are maintained for a few days with or without the drug. The effects of the drug are evaluated by observing the behavior of the cell in the zebrafish during tumor invasion, migration, extravasation, and angiogenesis (Figure 2). Drugs that have been shown to suppress metastatic processes in zebrafish xenograft models are listed in Table 1.
Figure 2.
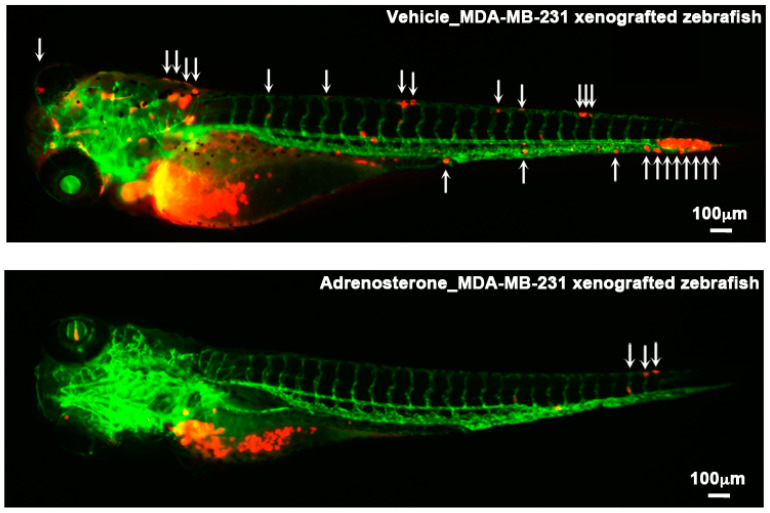
Examples of zebrafish xenograft models. Metastatic dissemination of red fluorescence protein (RFP)-labeled MDA-MB-231 cells in the vehicle-(top) or adrenosterone (bottom)-treated Tg(kdrl: EGFP) fish. The cells are inoculated into the duct of Cuvier of the fish at 48 hpf and then treated with either vehicle or adrenosterone for 24 h. White arrows indicate disseminated MDA-MB-231 cells. Images are shown in 4× magnification. Scale bar = 100 μm. Images are reprinted from [23].
Table 1.
A list of the anti-metastatic drugs identified in zebrafish xenograft models.
| Drug | Targeting Molecule | Targeting Molecular Event | Cancer Cell | Recipient Zebrafish | Inoculation Site | Reference |
|---|---|---|---|---|---|---|
| GLPG0187 | αv integrin | Invasion | MDA-MB-231 (Breast) | Tg(Fli1: EGFP) | Duct of Cuvier | [17] |
| IT1t | CXCR4 | Invasion | MDA-MB-231 (Breast) | Tg(Fli1: EGFP) | Duct of Cuvier | [18] |
| SB431542 | TGFβR1 | Invasion | MDA-MB-231 (Breast) | Tg(Fli1: EGFP) | Duct of Cuvier | [19] |
| SB525334 | TGFβR1 | Invasion | KIA (UPS) | Tg(Fli1: EGFP) | Yolk sac | [20] |
| 2-O-Bn-InsP5 | PDK1 | Invasion | MDA-MB-231 (Breast) | Tg(kdrl: HsHRAS-mCherry)s896 | Duct of Cuvier | [21] |
| Tenovin-6 | SIRT1/2 | Migration | TC252 (Ewing sarcoma) and A673 (Ewing sarcoma) | Tg(Fli1: EGFP) | Duct of Cuvier | [22] |
| Adrenosterone | HSD11β1 | EMT | HCCLM3 (Liver) and MDA-MB-231 (Breast) | Tg(kdrl: EGFP) | Duct of Cuvier | [23] |
| SU5416 | VEGFR | Angiogenesis | MDA-MB-435 (Skin) | Tg(Fli1: EGFP) | Peritoneal cavity | [24] |
| Sunitinib | VEGF | Angiogenesis | T241 (Thyroid) | Tg(Fli1: EGFP) | Perivitelline space | [25] |
| Sunitinib | VEGF | Invasion | SJmRBL-8 (Retinoblastoma) | Tg(Fli1: EGFP) | Eye | [26] |
| LY294002 | PI3-kinase | Invasion | MDA-MB-231 (Breast) | Tg(Fli1: EGFP) | Duct of Cuvier | [19] |
| GM6001 | MMPs | Invasion | MDA-MB-231 (Breast) | Tg(Fli1: EGFP) | Duct of Cuvier | [19] |
| Lipofermata | FATP | Invasion | ZMEL1 (Skin) | Casper | Subcutaneous tissue | [27] |
| DHS | Unknown | Invasion | LLC (Lung) | Tg(Fli1: EGFP) | Perivitelline cavity | [28] |
| SKLB1002 | VEGFR2 | Angiogenesis | B16-F10 (Skin) | Tg(Fli1: EGFP) | Perivitelline space | [29] |
| Osimertinib | EGFR | Angiogenesis | H1975 (Lung) | Tg(Fli1: EGFP) | Duct of Cuvier | [30] |
Another approach that is frequently used to assess anti-metastatic drugs is phenotype-based screening. Phenotype-based screening has been increasingly employed in biomedical and pharmaceutical research since the contributions of phenotypic screening to the discovery of first-in-class small-molecule drugs exceeded that of target-based approaches [31]. Diverse zebrafish-based screening experiments with various phenotypic readouts are conducted, which results in the identification of candidate substances that are further examined in preclinical and clinical trials [32,33]. The drugs that have been shown to suppress metastasis through phenotype-based screening models are listed in Table 2.
Table 2.
A list of the anti-metastatic drugs identified through phenotype-based screening using zebrafish.
| Drug | Targeting Molecule | Targeting Molecular Event | Screening Platform | # of Drugs Subjected to the Screen | Reference |
|---|---|---|---|---|---|
| SU6656 | Src | Migration | Tg(cldnb: EGFP) | 2960 compounds from the LOPAC1280 library, the NatProd library, and the GSK Published Kinase Inhibitor Set (PKIS) | [34] |
| Adrenosterone | HSD11β1 | EMT |
Tg(Fabp10A: mCherry-T2A-Twist1-ERT2) × Tg(fabp10a: TA; TRE:xmrk; krt4: GFP) |
68 chemicals from the Prestwick Chemical Library | [23] |
| TP-0903 | AXL receptor | EMT | Tg(snai1b: GFP) | Not applicable | [35] |
| PTK787 | VEGFR | Angiogenesis | WT | Not applicable | [36] |
| IRO | Unknown | Angiogenesis | Tg(VEGFR2: GRCFP) | 1280 chemicals from the LOPAC1280 library | [37] |
| F11 | PhKG1 | Angiogenesis | Tg (fli1: EGFP) | 288 chemicals | [38] |
| Kaempferol | VEGFR2/3 | Lymphangiogenesis | Tg(lyve1: EGFP) | 1120 chemicals from the Prestwick Chemical Library | [39] |
This review focuses on zebrafish models that have been used as screening platforms for the identification of anti-metastatic drugs. Figure 1 illustrates which of the identified drugs suppress each step of metastasis.
2. Targeting Metastatic Dissemination
Metastatic dissemination is the initial step of metastasis. During this step, cancer cells in the primary tumor site invade the neighboring tissue, enter the blood vessels, survive in the circulation, and exit the vessels to penetrate foreign tissues. Different genes and molecular mechanisms are responsible for each of these steps [40,41].
Integrins are cell-surface adhesion receptors consisting of α and β transmembrane protein subunits, which directly interact with extracellular matrix (ECM) components. Integrins also contribute to tumor development and metastasis progression [42]. The αv integrin antagonist GLPG0187 suppresses the metastatic dissemination of breast cancer cells in a zebrafish xenograft model that involves the inoculation of mCherry-labeled MDA-MB-231 cells into the duct of Cuvier of Tg(fli1: EGFP), which expresses EGFP in all blood vessels [43]. In this study, 56% of vehicle-treated zebrafish developed aggressive tumor lesions on their tail fins. In contrast, only 18% of GLPG0187-treated fish developed secondary tumors on the tail fin. Genetic inhibition of αv integrin shows the same effect as the pharmacological inhibition with GLPG0187, thus demonstrating that the anti-metastatic effects of GLPG0187 result from the inhibition of αv integrin rather than its off-target effects [17].
C-X-C chemokine receptor type 4 (CXCR4) and its cognate ligand C-X-C motif chemokine 12 (CXC12), which is also known as stromal cell-derived factor 1, play a pivotal role in regulating physiological processes such as hematopoiesis, leukocyte trafficking and cell migration [44]. The binding of CXC12 to CXCR4 also contributes to tumor development and metastatic progression [45]. Furthermore, CXCR4-overexpressing cancer cells preferentially develop metastatic tumors in distant organs that produce high levels of CXCL12 in human specimens and murine models [46]. The same phenomenon was also confirmed in a zebrafish xenograft model; in this model, a sub-cell line of MDA-MB-231 cells that overexpresses CXCR4 (MDA-MB-231-B) showed more aggressive metastatic behavior compared to the parental MDA-MB-231 cells upon inoculation into the duct of Cuvier of Tg(kdrl:EGFP), which selectively expresses EGFP in endothelial cells. However, MDA-MB-231-B cells that are inoculated into cxcl12a−/−/cxcl12b−/− background Tg(kdrl:EGFP) zebrafish fail to induce invasion and the formation of micro-metastases in the tail fin. Moreover, pharmacological inhibition using IT1t, a potent CXCR4 antagonist, suppresses early metastatic events of the cells in vivo. Genetic inhibition of CXCR4 shows the same effect as treatment with IT1t [18].
Members of the proto-oncogenic Src family of non-receptor protein tyrosine kinases also play critical roles in several metastatic cellular signal transduction pathways. The activation of Src family kinases in human cancers may occur through a variety of mechanisms, and the activity of Src family kinases is often critical for tumor progression. Several studies demonstrate that Src-mediated signaling plays a critical role in promoting metastasis [47,48]. A phenotype screening study using Tg(cldnb: EGFP) to mark the migrating posterior lateral line primordium as a readout for inhibition of collective cell migration identified the Src kinase inhibitor SU6656 has as an anti-metastatic agent. A murine model of metastatic cancer using 4T1 murine mammary carcinoma cells confirms that SU6656-treated mice show significantly fewer surface metastases in the lung compared with vehicle-treated mice [34].
Besides the three aforementioned drugs, several other pharmacological agents suppress the metastatic dissemination of human cancer cells in zebrafish xenograft models. The transforming growth factor-β (TGF-β receptor inhibitors SB431542 or SB525334 suppress the metastatic dissemination of breast cancer cells or undifferentiated pleomorphic sarcoma (UPS) cells in zebrafish xenograft models, respectively [19,20]. The inhibition of the phosphoinositide-dependent kinase-1 (PDK1)/Phospholipase C gamma 1 (PLCγ1) complex with the small molecule inhibitor 2-O-Bn-InsP5 significantly reduces the metastatic dissemination of MDA-MB-231 cells in Tg(kdrl: HsHRAS- mCherry)s896 zebrafish embryos [21]. The sirtuin (SIRT) 1/2 inhibitor tenovin-6 suppresses the metastatic dissemination of TC252 Ewing sarcoma cells in Tg(fli1: EGFP) [22]. Other compounds that suppress the metastatic dissemination of cancer cells in zebrafish xenograft models are listed in Table 1.
Experimental studies demonstrate that cancer cells can disseminate systemically from the earliest epithelial alterations in HER-2 and PyMT transgenic mice [49]. Clinical studies also reveal that cancer cells disseminate during the earliest phase of metastasis and are detected in the bone marrow years before the development of overt metastases [50]. These findings suggest that targeting the metastatic dissemination of cancer is not an effective strategy for blocking metastasis. However, the metastatic dissemination of cancer cells is directly observed in the living zebrafish due to the transparency of zebrafish embryos. This is a unique advantage possessed by zebrafish models. Combining this advantage with new imaging technologies would allow for the identification of molecular mechanisms responsible for the metastatic dissemination of cancer cells. The insights yielded by these future findings may lead to the discovery of new anti-metastatic drugs.
3. Targeting the Epithelial-Mesenchymal Transition (EMT) Process
EMT plays a central role in early embryonic morphogenesis, its program enables various types of epithelial cells to convert into mesenchymal cells [51]. Experimental studies demonstrate that EMT also contributes to metastatic progression by increasing the invasiveness, motility, and resistance of cancer cells to chemotherapy/apoptosis [52]. Therefore, EMT would be an ideal therapeutic target for anti-metastatic drugs.
Two transgenic zebrafish models that offer a screening platform for the identification of anti-EMT drugs have been reported. One is a tamoxifen-controllable Twist1a-ERT2 transgenic zebrafish line Tg(fabp10a:mCherry-T2A-Twist1a-ERT2), which induces spontaneous metastatic dissemination of cancer cells through induction of EMT. The other is the snailb promoter-driven GFP transgenic zebrafish line that labels epithelial cells undergoing EMT in zebrafish embryos [23,35].
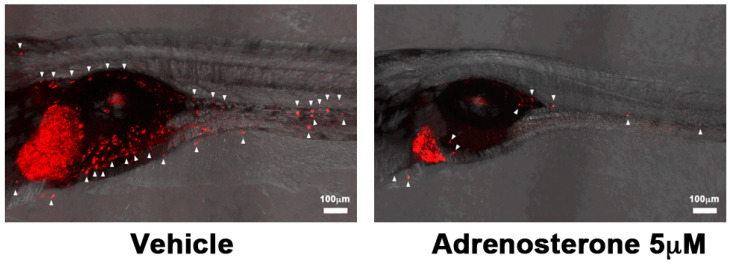
A tamoxifen-controllable Twist1a-ERT2 transgenic zebrafish serves as a platform for the discovery of anti-metastatic drugs. The activation of Twist1a-ERT2 following 48 h of tamoxifen treatment induces the conversion of epithelial cells into mesenchymal cells in the liver. By crossing this model with xmrk (a homolog of the hyperactive form of EGFR) transgenic zebrafish [53], which develops hepatocellular carcinoma, approximately 80% of the double-transgenic zebrafish showed spontaneous dissemination of mCherry-labeled hepatocytes from the liver to the entire abdomen region and the tail region within five days from the treatment initiation. FDA-approved drugs are subjected to in vivo screening using this model. Adrenosterone, an inhibitor of hydroxysteroid (11-beta) dehydrogenase 1 (HSD11β1), suppresses cellular dissemination in this model (Figure 3). This suppressor effect is validated in a zebrafish xenotransplantation model in which highly-metastatic human cell lines are inoculated into the duct of Cuvier of Tg(kdrl:EGFP) transgenic zebrafish. Genetic inhibition of HSD11β1 also suppresses the metastatic dissemination of these cell lines in a zebrafish xenotransplantation model. This suppression results from the re-expression of E-cadherin and other epithelial markers and lost partial expression of mesenchymal markers through the down-regulation of Snail and Slug [23].
Figure 3.
Examples of phenotype-based screening in zebrafish. Metastatic dissemination of mCherry-positive cells from the liver of vehicle (left) or adrenosterone (right)-treated Twist1-ERT2/xmrk double-transgenic fish. White arrows indicate disseminated mCherry-positive cells. Images are shown as Z-stack images using 100× magnification. Scale bar = 100 μm. Images are reprinted from [23].
Tg(snailb: GFP), which provides a whole-animal EMT reporter system in zebrafish, is established for rapid drug screening. This model allows for the labeling of epithelial cells undergoing EMT to produce sox10-positive neural crest (NC) cells. Treating embryos of this model with candidate small-molecule EMT-inhibiting compounds previously demonstrated that TP-0903, an inhibitor of AXL receptor tyrosine kinase, blocked the delamination of cranial NC cells in both the lateral and medial populations. TP-0903 stimulates retinoic acid (RA) biosynthesis and RA-dependent transcription. These studies identified TP-0903 as an activator of RA in vivo and raised the possibility that its prior success in eliminating disseminated cancer cells depends upon the RA-dependent inhibition of EMT [35].
The activation of EMT endows epithelial carcinoma cells with mesenchymal traits, stem-like characteristics, increased drug resistance, invasiveness, and metastatic ability [54]. Furthermore, a recent study shows that EMT needs to be transient and reversible, and the transition from a fully mesenchymal phenotype to an epithelial-mesenchymal hybrid state or a fully epithelial phenotype is associated with malignant phenotypes [55]. Cortisol, which is produced by HSD11β1, is reported to promote the metastatic progression of breast cancer cells. The knockdown of the cortisol receptor in highly metastatic cells prevents them from metastasizing to distant organs [56]. TP-0903 is also reported to sensitize erlotinib-resistant non-small cell lung cancer cells to erlotinib by reversing the mesenchymal phenotype in preclinical tumor models [57]. This evidence supports that adrenosterone and TP-0903 would suppress the metastatic progression of human cancer cells.
4. Targeting Angiogenesis
Angiogenesis is the formation and remodeling of new blood vessels and capillaries from pre-existing blood vessels. The growth potential of avascular tumors is severely restricted due to the lack of blood supply. The formation of new blood vessels provides avascular tumors with nutrients, oxygen, and more efficient removal of waste products [58]. Although anti-angiogenic agents targeting vascular endothelial growth factor (VEGF) signaling are already used to effectively treat some cancers, there is a continued need for the development of new inhibitors of angiogenesis to circumvent resistance or reduce toxicity.
A pioneering study demonstrates that the treatment of zebrafish embryos with the VEGF receptor inhibitor PTK787/ZK222584 completely blocks the formation of all major blood vessels. Furthermore, the overexpression of the downstream effector AKT/PKB allows blood vessels to form in the presence of the drug [36]. Following this study, several studies performing high-throughput screening in zebrafish models are reported to identify new anti-angiogenic drugs [37].
A transgenic zebrafish that expresses green reef coral fluorescent protein (GRCFP) under the control of the VEGFR2 promoter Tg(VEGFR2: GRCFP) offers a high-throughput platform for the discovery of anti-angiogenic drugs. Previous in vivo drug screening studies using this model identified indirubin-3′-monoxime (IRO) as an inhibitor of angiogenesis that did not affect vasculogenic vessel development or preexisting vasculature. An in vitro assay using human umbilical vein endothelial cells (HUVECs) confirmed that IRO inhibits two major components of the angiogenic process—endothelial tube formation and cell proliferation. However, IRO does not significantly affect endothelial cell migration [37]. Another high-throughput screening study using Tg(fli: EGFP) also identified the PhK subunit G1 (PhKG1) inhibitor F11 as an anti-angiogenic drug in vivo and in vitro using cultured human endothelial cells [38].
A previous study using high-resolution confocal microscopy allows one to visualize the neovascularization and behavior of highly metastatic cells. In the study, the DsRed-labeled MDA-MB-435 human melanoma cell line was inoculated into the peritoneal cavity of Tg(fli1: EGFP) zebrafish at 2 dpf. The interactions between the DsRed-labeled cells and the GFP-labeled vessels during angiogenesis are visualized through three-dimensional reconstructions of the interaction. The images reveal that RhoC amplification in the DsRed cells induces the formation of dynamic membrane protrusions and blebs and that VEGF secreted by the DsRed cells increases vascular permeability. These molecules work cooperatively to facilitate the invasion and intravasation by regulating the cytoskeletal and vascular remodeling. Pharmacological inhibition of VEGFR using SU5416 restored the integrity of the vessel wall and decreased the size of the DsRed tumor from 25.5 to 16 μm [24].
Another zebrafish model of metastasis may be used to visualize cancer cell dissemination, invasion, and metastasis at the single-cell level. Using this model, DiI-labeled T241 murine fibrosarcoma cells are injected into the perivitelline space of Tg(fli1: EGFP) zebrafish at 48 hpf and the zebrafish is subsequently transferred to a hypoxia chamber. Under hypoxia, the cells disseminate from their primary sites, invade the neighboring tissue, and metastasize to distal parts of the body through the upregulation of VEGF, which is a hypoxia-regulated angiogenic factor. In contrast, significant dissemination of cells does not occur under normoxia. Sunitinib, a VEGF inhibitor, inhibits hypoxia-induced invasion, dissemination, and metastasis of T241 tumors in this model [25]. The other compounds that suppress angiogenesis are listed in Table 2.
The U.S. Food and Drug Administration (FDA) has approved several anti-angiogenic drugs to treat cancer. Most of the inhibitors target VEGF, its receptor, or other specific molecules involved in angiogenesis [59]. Among the drugs verified by zebrafish models, only sunitinib has been approved by the FDA. PTK787/ZK222584 (also known as vatalanib) has been extensively tested in Phase I, II, and III clinical trials [60,61,62]. Clinical trials for SU5416 were discontinued due to discouraging results [63,64,65]. The limited efficacy of anti-angiogenic drugs remains a continuing problem in their clinical use [66,67]. Unique advantages provided by zebrafish models that would be suitable for angiogenesis research including the discovery and evaluation of anti-angiogenic agents, the identification of novel targets for anti-angiogenesis, and the elucidation of molecular mechanisms responsible for angiogenesis. Therefore, new insights from these studies would contribute to improving the therapeutic benefits of anti-angiogenic strategies.
5. Targeting Lymphangiogenesis
The growth of new lymphatic vessels (lymphangiogenesis) in tumors is an integral part of metastasis. Cancer cells in the primary tumor site initially spread via lymphatic vessels to their regional lymph nodes [68]. Previous in vivo high-throughput screening studies using Tg(lyve1: EGFP), which expresses EGFP in the lymphatic vessels [69], identified that treatment with kaempferol, which inhibits VEGFR2/3, suppresses the migration of lymphatic precursors known as secondary sprouts from the posterior cardinal vein. Further, a murine in vivo lymphangiogenesis Matrigel plug assay confirmed that kaempferol has anti-lymphatic activity. A mouse xenograft model where luciferase-expressing MDA-MB-231 cells (MDA-MB-231-luc-D3H2LN) were inoculated into the mammary fat pad of nude mice demonstrated that kaempferol reduced lymph node metastases compared with vehicle treatment. However, bioluminescence imaging and hematoxylin and eosin (H&E) staining showed that 80% of kaempferol-treated mice had metastatic tumors in the pancreas and diaphragm in both the prevention and intervention regimens, compared with 10% of control mice. These results indicate that kaempferol is only effective at reducing lymph node metastases [39].
Kaempferol is widely distributed in the plant kingdom and is a common constituent of fruits and vegetables. Kaempferol and its derivatives have cardioprotective, neuroprotective, anti-inflammatory, antidiabetic, antioxidant, antimicrobial properties [70]. Past studies demonstrate that kaempferol suppresses the proliferation of various cancer cells derived from the lung, breast, pancreas, prostate, and colon [71]. Moreover, multi-ethnic cohort studies have demonstrated that flavonols such as kaempferol prevent the development of pancreatic cancer [72]. Therefore, the daily intake of kaempferol may prevent the progression of metastasis.
6. Targeting Tumor Microenvironments
Cancer cells are surrounded by many different types of cells: vascular endothelial cells, immune cells, fibroblasts, adipocytes, and the extracellular matrix (ECM). Experimental studies demonstrate that crosstalk between cancer cells and the surrounding microenvironment promotes metastasis. Therefore, targeting the tumor microenvironment may be an effective therapeutic strategy for suppressing metastasis [73].
The highly metastatic zebrafish melanoma cell line, ZMEL1 is established from melanomas of Tg(mitfa: BRAFV600E; p53−/−) fish, which develop melanoma by four months old [74]. After subcutaneously inoculating the cells into the ventral flank of adult casper recipient zebrafish, 83% (n = 25/30) of the fish showed widespread spontaneous metastases in the anterior region just behind the gill structure. Other metastases were observed on the posterior tail musculature and eye two weeks after the inoculation [75]. Studies using this model show that the interaction between disseminated melanoma cells and their microenvironment plays a critical role in promoting the formation of metastatic tumors at distant sites. Adipocytes increase the proliferation and invasion of adjacent ZMEL1 cells by transferring lipids to the cells. The lipids are incorporated into the cells via the fatty acid transport protein (FATP)/SLC27A family of lipid transporters, which are overexpressed by the cells. Pharmacological inhibition of FATPs with the small-molecule inhibitor lipofermata reduces the proliferation and invasion of ZMEL1 cells by interfering with lipid transport [27]. Also, endothelin-3 (EDN3), which is expressed in the microenvironment surrounding ZMEL1 cells, promotes a differentiated and proliferative state through the up-regulation of MYC, premelanosome protein (PMEL), tyrosinase-related protein 1 (TYRP1), and tyrosinase (TRY). The ZMEL1 cells that are transplanted into EDN3-deficient zebrafish decrease the formation of metastatic tumors compared to those in the wild-type fish [76].
Due to the phenotypic plasticity of cancer cells that emerge from interactions between cancer cells and their microenvironment, cancer cells have developed resistance to anti-cancer drugs, which have been identified from in vitro screens using two-dimensional cell culture systems. Therefore, drug discovery needs to also consider the phenotypic plasticity of cancer cells. Zebrafish models can be used for in vivo drug screening to evaluate the effect of chemicals on cancer cells while accounting for the tumor microenvironment. Lipofermata, which targets the tumor microenvironment, is one example of an anti-metastatic drug that has been identified by using a zebrafish model [27]. A recent study also reports that the pharmacological inhibition of FATP2 with lipofermata abrogates the activity of pathologically-activated neutrophils, which play a critical role in regulating the immune response during cancer progression, and substantially delays tumor progression [77]. This evidence supports that targeting the microenvironments that surround cancer cells would be an effective strategy for blocking metastasis.
Acknowledgments
We sincerely appreciate Prof. Zhiyuan Gong (National University of Singapore) and American Association for Cancer Research (AACR) for allowing us to reprint the images in Figure 2 and Figure 3 into this review. The images are reprinted from a previous study of ours [23]. The study was funded by National Medical Research Council of Singapore (R-154000547511) and Ministry of Education of Singapore (R-154000A23112) to Z. Gong.
Funding
This research received no external funding.
Conflicts of Interest
J.N. and H.M. declare no conflict of interest.
References
- 1.Fidler I. The pathogenesis of cancer metastasis: The ’seed and soil’ hypothesis revisited. Nat. Rev. Cancer. 2003;3:453–458. doi: 10.1038/nrc1098. [DOI] [PubMed] [Google Scholar]
- 2.Steeg P.S. Metastasis suppressors alter the signal transduction of cancer cells. Nat. Rev. Cancer. 2003;3:55–63. doi: 10.1038/nrc967. [DOI] [PubMed] [Google Scholar]
- 3.Chambers A.F., Groom A.C., MacDonald I.C. Dissemination and growth of cancer cells in metastatic sites. Nat. Rev. Cancer. 2002;2:563. doi: 10.1038/nrc865. [DOI] [PubMed] [Google Scholar]
- 4.Pollard J.W. Tumour-educated macrophages promote tumour progression and metastasis. Nat. Rev. Cancer. 2004;4:71–78. doi: 10.1038/nrc1256. [DOI] [PubMed] [Google Scholar]
- 5.Karnoub A.E., Dash A.B., Vo A.P., Sullivan A., Brooks M.W., Bell G.W., Richardson A.L., Polyak K., Tubo R., Weinberg R.A. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature. 2007;449:557–563. doi: 10.1038/nature06188. [DOI] [PubMed] [Google Scholar]
- 6.Chaffer C.L., Weinberg R.A. A perspective on cancer cell metastasis. Science. 2011;331:1559–1564. doi: 10.1126/science.1203543. [DOI] [PubMed] [Google Scholar]
- 7.White R., Rose K., Zon L. Zebrafish cancer: The state of the art and the path forward. Nat. Rev. Cancer. 2013;13:624–636. doi: 10.1038/nrc3589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zon L.I., Peterson R.T. In vivo drug discovery in the zebrafish. Nat. Rev. Drug Discov. 2005;4:35–44. doi: 10.1038/nrd1606. [DOI] [PubMed] [Google Scholar]
- 9.Letrado P., de Miguel I., Lamberto I., Díez-Martínez R., Oyarzabal J. Zebrafish: Speeding Up the Cancer Drug Discovery Process. Cancer Res. 2018;78:6048–6058. doi: 10.1158/0008-5472.CAN-18-1029. [DOI] [PubMed] [Google Scholar]
- 10.Gunnarsson L., Jauhiainen A., Kristiansson E., Nerman O., Larsson D.G.J. Evolutionary conservation of human drug targets in organisms used for environmental risk assessments. Environ. Sci. Technol. 2008;42:5807–5813. doi: 10.1021/es8005173. [DOI] [PubMed] [Google Scholar]
- 11.Howe K., Clark M.D., Torroja C.F. The zebrafish reference genome sequence and its relationship to the human genome. Nature. 2013;496:498–503. doi: 10.1038/nature12111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yao Y., Sun S., Wang J., Fei F., Dong Z., Ke A., He R., Wang L., Zhang L., Ji M., et al. Canonical Wnt Signaling Remodels Lipid Metabolism in Zebrafish Hepatocytes following Ras Oncogenic Insult. Cancer Res. 2018;78:5548–5560. doi: 10.1158/0008-5472.CAN-17-3964. [DOI] [PubMed] [Google Scholar]
- 13.Milan D.J., Peterson T.A., Ruskin J.N., Peterson R.T., MacRae C.A. Drugs That Induce Repolarization Abnormalities Cause Bradycardia in Zebrafish. Circulation. 2003;107:1355–1358. doi: 10.1161/01.CIR.0000061912.88753.87. [DOI] [PubMed] [Google Scholar]
- 14.Cabezas-Sainz P., Guerra-Varela J., Carreira M., Mariscal J., Roel M., Rubiolo J., Sciara A., Botana L., López R., Sánchez L. Improving Zebrafish Embryo Xenotransplantation Conditions by Increasing Incubation Temperature and Establishing a Proliferation Index With ZFtool. BMC Cancer. 2018;18:3. doi: 10.1186/s12885-017-3919-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Brown H.K., Schiavone K., Tazzyman S., Heymann D., Chico T.J. Zebrafish xenograft models of cancer and metastasis for drug discovery. Expert Opin. Drug Discov. 2017;12:379–389. doi: 10.1080/17460441.2017.1297416. [DOI] [PubMed] [Google Scholar]
- 16.Lieschke G.J., Trede N.S. Fish immunology. Curr. Biol. 2009;19:R678–R682. doi: 10.1016/j.cub.2009.06.068. [DOI] [PubMed] [Google Scholar]
- 17.Li Y., Drabsch Y., Pujuguet P., Ren J., van Laar T., Zhang L., van Dam H., Clément-Lacroix P., Ten D.P. Genetic depletion and pharmacological targeting of αv integrin in breast cancer cells impairs metastasis in zebrafish and mouse xenograft models. Breast Cancer Res. 2015;25:28. doi: 10.1186/s13058-015-0537-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tulotta C., Stefanescu C., Beletkaia E., Bussmann J., Tarbashevich K., Schmidt T., Snaar-Jagalska B. Inhibition of signaling between human CXCR4 and zebrafish ligands by the small molecule IT1t impairs the formation of triple-negative breast cancer early metastases in a zebrafish xenograft model. Dis. Model. Mech. 2016;9:141. doi: 10.1242/dmm.023275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Drabsch Y., He S., Zhang L., Snaar-Jagalska B., Ten D.P. Transforming growth factor-β signalling controls human breast cancer metastasis in a zebrafish xenograft model. Breast Cancer Res. 2013;15:R106. doi: 10.1186/bcr3573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ye S., Liu Y., Fuller A.M., Katti R., Ciotti G.E., Chor S., Alam M.Z., Devalaraja S., Lorent K., Weber K., et al. TGFb and Hippo Pathways Cooperate to Enhance Sarcomagenesis and Metastasis through the Hyaluronan-Mediated Motility Receptor (HMMR) Mol. Cancer Res. 2020;18:560–573. doi: 10.1158/1541-7786.MCR-19-0877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Raimondi C., Calleja V., Ferro R., Fantin A., Riley A.M., Potter B.V., Brennan C.H., Maffucci T., Larijani B., Falasca M. A Small Molecule Inhibitor of PDK1/ PLCγ1 Interaction Blocks Breast and Melanoma Cancer Cell Invasion. Sci. Rep. 2016;20:26142. doi: 10.1038/srep26142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ban J., Aryee D., Fourtouna A., Van der Ent W., Kauer M., Niedan S., Machado I., Rodriguez-Galindo C., Tirado O., Schwentner R., et al. Suppression of deacetylase SIRT1 mediates tumor-suppressive NOTCH response and offers a novel treatment option in metastatic Ewing sarcoma. Cancer Res. 2014;74:6578–6588. doi: 10.1158/0008-5472.CAN-14-1736. [DOI] [PubMed] [Google Scholar]
- 23.Nakayama J., Lu J., Makinoshima H., Gong Z. A Novel Zebrafish Model of Metastasis Identifies the HSD11β1 Inhibitor Adrenosterone as a Suppressor of Epithelial-Mesenchymal Transition and Metastatic Dissemination. Mol. Cancer Res. 2020;18:477–487. doi: 10.1158/1541-7786.MCR-19-0759. [DOI] [PubMed] [Google Scholar]
- 24.Stoletov K., Montel V., Lester R.D., Gonias S.L., Klemke R. High-resolution imaging of the dynamic tumor cell–vascular interface in transparent zebrafish. Proc. Natl. Acad. Sci. USA. 2007;104:17406–17411. doi: 10.1073/pnas.0703446104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lee S., Rouh I.P., Dahl Jensen L., Zhang D., Ji H., Hauptmann G., Ingham P., Cao Y. Hypoxia-induced pathological angiogenesis mediates tumor cell dissemination, invasion, and metastasis in a zebrafish tumor model. Proc. Natl. Acad. Sci. USA. 2009;106:19485. doi: 10.1073/pnas.0909228106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chen X., Wang J., Cao Z., Hosaka K., Jensen L., Yang H., Sun Y., Zhuang R., Liu Y., Cao Y. Invasiveness and metastasis of retinoblastoma in an orthotopic zebrafish tumor model. Sci. Rep. 2015;14:10351. doi: 10.1038/srep10351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang M., Di Martino J., Bowman R.L., Campbell N.R., Baksh S.C., Simon-Vermot T., Kim I.S., Haldeman P., Mondal C., Yong-Gonzales V., et al. Adipocyte-Derived Lipids Mediate Melanoma Progression via FATP Proteins. Cancer Discov. 2018;8:1006–1025. doi: 10.1158/2159-8290.CD-17-1371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Savio M., Ferraro D., Maccario C., Vaccarone R., Jensen L.D., Corana F., Mannucci B., Bianchi L., Cao Y., Stivala L.A. Resveratrol analogue 4,4’-dihydroxy-trans-stilbene potently inhibits cancer invasion and metastasis. Sci. Rep. 2016;1:19973. doi: 10.1038/srep19973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhang S., Cao Z., Tian H., Shen G., Ma Y., Xie H., Liu Y., Zhao C., Deng S., Yang Y., et al. SKLB1002, a novel potent inhibitor of VEGF receptor 2 signaling, inhibits angiogenesis and tumor growth in vivo. Clin. Cancer Res. 2011;17:4439–4450. doi: 10.1158/1078-0432.CCR-10-3109. [DOI] [PubMed] [Google Scholar]
- 30.Li X.Y., Huang L.T., Wu J.Q., He M.F., Zhu S.H., Zhan P., Lv T.F., Song Y. Zebrafish Xenograft Model of Human Lung Cancer for Evaluating Osimertinib Resistance. Biomed. Res. Int. 2019;27:3129748. doi: 10.1155/2019/3129748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Swinney D.C., Anthony J. How were new medicines discovered? Nat. Rev. Drug Discov. 2011;10:507–519. doi: 10.1038/nrd3480. [DOI] [PubMed] [Google Scholar]
- 32.North T.E., Goessling W., Walkley C.R., Lengerke C., Kopani K.R., Lord A.M., Weber G.J., Bowman T.V., Jang I.H., Grosser T., et al. Prostaglandin E2 regulates vertebrate haematopoietic stem cell homeostasis. Nature. 2007;447:1007–1011. doi: 10.1038/nature05883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.White R.M., Cech J., Ratanasirintrawoot S., Lin C.Y., Rahl P.B., Burke C.J., Langdon E., Tomlinson M.L., Mosher J., Kaufman C., et al. DHODH modulates transcriptional elongation in the neural crest and melanoma. Nature. 2011;471:518–522. doi: 10.1038/nature09882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gallardo V.E., Varshney G.K., Lee M., Bupp S., Xu L., Shinn P., Crawford N.P., Inglese J., Burgess S.M. Phenotype-driven chemical screening in zebrafish for compounds that inhibit collective cell migration identifies multiple pathways potentially involved in metastatic invasion. Dis. Model. Mech. 2015;8:565–576. doi: 10.1242/dmm.018689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jimenez L., Wang J., Morrison M.A., Whatcott C., Soh K.K., Warner S., Bearss D., Jette C.A., Stewart R.A. Phenotypic chemical screening using a zebrafish neural crest EMT reporter identifies retinoic acid as an inhibitor of epithelial morphogenesis. Dis. Model. Mech. 2016;9:389–400. doi: 10.1242/dmm.021790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chan J., Bayliss P.E., Wood J.M., Roberts T.M. Dissection of angiogenic signaling in zebrafish using a chemical genetic approach. Cancer Cell. 2002;1:257–267. doi: 10.1016/S1535-6108(02)00042-9. [DOI] [PubMed] [Google Scholar]
- 37.Tran T.C., Sneed B., Haider J., Blavo D., White A., Aiyejorun T., Baranowski T., Rubinstein A.L., Doan T.N., Dingledine R., et al. Automated, Quantitative Screening Assay for Antiangiogenic Compounds Using Transgenic Zebrafish. Cancer Res. 2007;67:11386. doi: 10.1158/0008-5472.CAN-07-3126. [DOI] [PubMed] [Google Scholar]
- 38.Camus S., Quevedo C., Menéndez S., Paramonov I., Stouten P.F., Janssen R.A., Rueb S., He S., Snaar-Jagalska B.E., Laricchia-Robbio L., et al. Identification of phosphorylase kinase as a novel therapeutic target through high-throughput screening for anti-angiogenesis compounds in zebrafish. Oncogene. 2012;31:4333–4342. doi: 10.1038/onc.2011.594. [DOI] [PubMed] [Google Scholar]
- 39.Astin J.W., Jamieson S.M., Eng T.C., Flores M.V., Misa J.P., Chien A., Crosier K.E., Crosier P.S. An in vivo antilymphatic screen in zebrafish identifies novel inhibitors of mammalian lymphangiogenesis and lymphatic-mediated metastasis. Mol. Cancer Ther. 2014;13:2450–2462. doi: 10.1158/1535-7163.MCT-14-0469-T. [DOI] [PubMed] [Google Scholar]
- 40.Chiang A.C., Massagué J. Molecular Basis of Metastasis. N. Engl. J. Med. 2008;359:2814–2823. doi: 10.1056/NEJMra0805239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Steeg P.S. Targeting metastasis. Nat. Rev. Cancer. 2016;16:201–218. doi: 10.1038/nrc.2016.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hamidi H., Ivaska J. Every step of the way: Integrins in cancer progression and metastasis. Nat. Rev. Cancer. 2018;18:533–548. doi: 10.1038/s41568-018-0038-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lawson N.D., Weinstein B.M. In vivo imaging of embryonic vascular development using transgenic zebrafish. Dev. Biol. 2002;248:307. doi: 10.1006/dbio.2002.0711. [DOI] [PubMed] [Google Scholar]
- 44.Domanska U.M., Kruizinga R.C., Nagengast W.B., Timmer-Bosscha H., Huls G., de Vries E.G., Walenkamp A.M. A review on CXCR4/CXCL12 axis in oncology: No place to hide. Eur. J. Cancer. 2013;49:219–230. doi: 10.1016/j.ejca.2012.05.005. [DOI] [PubMed] [Google Scholar]
- 45.Orimo A., Gupta P.B., Sgroi D.C., Arenzana-Seisdedos F., Delaunay T., Naeem R., Carey V.J., Richardson A.L., Weinberg R.A. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell. 2005;121:335–348. doi: 10.1016/j.cell.2005.02.034. [DOI] [PubMed] [Google Scholar]
- 46.Müller A., Homey B., Soto H., Ge N., Catron D., Buchanan M., McClanahan T., Murphy E., Yuan W., Wagner S., et al. Involvement of chemokine receptors in breast cancer metastasis. Nature. 2001;410:50. doi: 10.1038/35065016. [DOI] [PubMed] [Google Scholar]
- 47.Zhang X.H., Wang Q., Gerald W., Hudis C.A., Norton L., Smid M., Foekens J.A., Massagué J. Latent bone metastasis in breast cancer tied to Src-dependent survival signals. Cancer Cell. 2009;16:67–78. doi: 10.1016/j.ccr.2009.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Boyer B., Bourgeois Y., Poupon M.F. Src kinase contributes to the metastatic spread of carcinoma cells. Oncogene. 2002;21:2347–2356. doi: 10.1038/sj.onc.1205298. [DOI] [PubMed] [Google Scholar]
- 49.Husemann Y., Geigl J.B., Schubert F., Musiani P., Meyer M., Burghart E., Forni G., Eils R., Fehm T., Riethmuller G., et al. Systemic spread is an early step in breast cancer. Cancer Cell. 2008;13:58–68. doi: 10.1016/j.ccr.2007.12.003. [DOI] [PubMed] [Google Scholar]
- 50.Braun S., Vogl F.D., Naume B., Janni W., Osborne M.P., Coombes R.C., Schlimok G., Diel I.J., Gerber B., Gebauer G., et al. A pooled analysis of bone marrow micrometastasis in breast cancer. N. Engl. J. Med. 2005;353:793–802. doi: 10.1056/NEJMoa050434. [DOI] [PubMed] [Google Scholar]
- 51.Thiery J.P., Acloque H., Huang R.Y., Nieto M.A. Epithelial-mesenchymal transitions in development and disease. Cell. 2009;139:871–890. doi: 10.1016/j.cell.2009.11.007. [DOI] [PubMed] [Google Scholar]
- 52.Tsai J.H., Yang J. Epithelial-mesenchymal plasticity in carcinoma metastasis. Genes Dev. 2013;27:2192–2206. doi: 10.1101/gad.225334.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Li Z., Huang X., Zhan H., Zeng Z., Li C., Spitsbergen J.M., Meierjohann S., Schartl M., Gong Z. Inducible and repressable oncogene-addicted hepatocellular carcinoma in Tet-on xmrk transgenic zebrafish. J. Hepatol. 2012;56:419–425. doi: 10.1016/j.jhep.2011.07.025. [DOI] [PubMed] [Google Scholar]
- 54.Brabletz T. To differentiate or not—Routes towards metastasis. Nat. Rev. Cancer. 2012;12:425–436. doi: 10.1038/nrc3265. [DOI] [PubMed] [Google Scholar]
- 55.Kröger C., Afeyan A., Mraz J., Eaton E.N., Reinhardt F., Khodor Y.L., Thiru P., Bierie B., Ye X., Burge C.B., et al. Acquisition of a hybrid E/M state is essential for tumorigenicity of basal breast cancer cells. Proc. Natl. Acad. Sci. USA. 2019;116:7353–7362. doi: 10.1073/pnas.1812876116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Obradović M.M.S., Hamelin B., Manevski N., Couto J.P., Sethi A., Coissieux M.M., Münst S., Okamoto R., Kohler H., Schmidt A., et al. Glucocorticoids promote breast cancer metastasis. Nature. 2019;567:540–544. doi: 10.1038/s41586-019-1019-4. [DOI] [PubMed] [Google Scholar]
- 57.Soh K.K., Kim W., Lee Y.S., Peterson P., Siddiqui-Jain A., Warner S.L., Bearss D.J., Whatcott C.J. Abstract 235: AXL inhibition leads to a reversal of a mesenchymal phenotype sensitizing cancer cells to targeted agents and immuno-oncology therapies; Proceedings of the AACR 107th Annual Meeting 2016; New Orleans, LA, USA. 16–20 April 2016. [Google Scholar]
- 58.Folkman J. Role of angiogenesis in tumor growth and metastasis. Semin. Oncol. 2002;29:15–18. doi: 10.1053/sonc.2002.37263. [DOI] [PubMed] [Google Scholar]
- 59.Rajabi M., Mousa S.A. The Role of Angiogenesis in Cancer Treatment. Biomedicines. 2017;5:34. doi: 10.3390/biomedicines5020034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Scott E.N., Meinhardt G., Jacques C., Laurent D., Thomas A.L. Vatalanib: The clinical development of a tyrosine kinase inhibitor of angiogenesis in solid tumours. Expert Opin Investig Drugs. 2007;16:367–379. doi: 10.1517/13543784.16.3.367. [DOI] [PubMed] [Google Scholar]
- 61.Jost L.M., Gschwind H.P., Jalava T., Wang Y., Guenther C., Souppart C., Rottmann A., Denner K., Waldmeier F., Gross G., et al. Metabolism and disposition of vatalanib (PTK787/ZK-222584) in cancer patients. Drug Metab. Dispos. 2006;34:1817–1828. doi: 10.1124/dmd.106.009944. [DOI] [PubMed] [Google Scholar]
- 62.Los M., Roodhart J.M., Voest E.E. Target practice: Lessons from phase III trials with bevacizumab and vatalanib in the treatment of advanced colorectal cancer. Oncologist. 2007;12:443–450. doi: 10.1634/theoncologist.12-4-443. [DOI] [PubMed] [Google Scholar]
- 63.O’Donnell A., Padhani A., Hayes C., Kakkar A.J., Leach M., Trigo J.M., Scurr M., Raynaud F., Phillips S., Aherne W., et al. A Phase I Study of the Angiogenesis Inhibitor SU5416 (Semaxanib) in Solid Tumours, Incorporating Dynamic Contrast MR Pharmacodynamic End Points. Br. J. Cancer. 2005;93:876–883. doi: 10.1038/sj.bjc.6602797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Heymach J.V., Desai J., Manola J., Davis D.W., McConkey D.J., Harmon D., Ryan D.P., Goss G., Quigley T., Van den Abbeele A.D., et al. Phase II study of the antiangiogenic agent SU5416 in patients with advanced soft tissue sarcomas. Clin. Cancer Res. 2004;10:5732–5740. doi: 10.1158/1078-0432.CCR-04-0157. [DOI] [PubMed] [Google Scholar]
- 65.Lockhart A.C., Cropp G.F., Berlin J.D., Donnelly E., Schumaker R.D., Schaaf L.J., Hande K.R., Fleischer A.C., Hannah A.L., Rothenberg M.L. Phase I/pilot study of SU5416 (semaxinib) in combination with irinotecan/bolus 5-FU/LV (IFL) in patients with metastatic colorectal cancer. Am. J. Clin. Oncol. 2006;29:109–115. doi: 10.1097/01.coc.0000199882.53545.ac. [DOI] [PubMed] [Google Scholar]
- 66.Carmeliet P., Jain R.K. Molecular mechanisms and clinical applications of angiogenesis. Nature. 2011;473:298–307. doi: 10.1038/nature10144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Hayden E.C. Cutting off cancer’s supply lines. Nature. 2009;458:686–687. doi: 10.1038/458686b. [DOI] [PubMed] [Google Scholar]
- 68.Viviane M., Michael D. Lymphangiogenesis and cancer metastasis. J. Cell. Mol. Med. 2009;13:1405–1416. doi: 10.1111/j.1582-4934.2009.00834.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Okuda K.S., Astin J.W., Misa J.P., Flores M.V., Crosier K.E., Crosier P.S. lyve1 expression reveals novel lymphatic vessels and new mechanisms for lymphatic vessel development in zebrafish. Development. 2012;139:2381–2391. doi: 10.1242/dev.077701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Calderón-Montaño J.M., Burgos-Morón E., Pérez-Guerrero C., López-Lázaro M. A review on the dietary flavonoid kaempferol. Mini Rev. Med. Chem. 2011;11:298–344. doi: 10.2174/138955711795305335. [DOI] [PubMed] [Google Scholar]
- 71.Muhammad I., Bahare S., Javad S.-R., Tanweer A.G., Farhan S., Ali I., Muhammad S., Fokou P.V.T., Arshad M.U., Haroon K., et al. Kaempferol: A Key Emphasis to Its Anticancer Potential. Molecules. 2019;24:2277. doi: 10.3390/molecules24122277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Nöthlings U., Murphy S.P., Wilkens L.R., Henderson B.E., Kolonel L.N. Flavonols and pancreatic cancer risk: The multiethnic cohort study. Am. J. Epidemiol. 2007;166:924–931. doi: 10.1093/aje/kwm172. [DOI] [PubMed] [Google Scholar]
- 73.Joyce J.A. Therapeutic targeting of the tumor microenvironment. Cancer Cell. 2005;7:513–520. doi: 10.1016/j.ccr.2005.05.024. [DOI] [PubMed] [Google Scholar]
- 74.Patton E.E., Widlund H.R., Kutok J.L., Kopani K.R., Amatruda J.F., Murphey R.D., Berghmans S., Mayhall E.A., Traver D., Fletcher C.D., et al. BRAF mutations are sufficient to promote nevi formation and cooperate with p53 in the genesis of melanoma. Curr. Biol. 2005;15:249–254. doi: 10.1016/j.cub.2005.01.031. [DOI] [PubMed] [Google Scholar]
- 75.Heilmann S., Ratnakumar K., Langdon E., Kansler E., Kim I., Campbell N.R., Perry E., McMahon A., Kaufman C., van Rooijen E., et al. A Quantitative System for Studying Metastasis Using Transparent Zebrafish. Cancer Res. 2015;75:4272–4282. doi: 10.1158/0008-5472.CAN-14-3319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Kim I.S., Heilmann S., Kansler E., Zhang Y., Zimmer M., Ratnakumar K., Bowman R.L., Simon-Vermot T., Fennell M., Garippa R., et al. Microenvironment-derived factors driving metastatic plasticity in melanoma. Nat. Commun. 2017;9:14343. doi: 10.1038/ncomms14343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Veglia F., Tyurin V.A., Blasi M., De Leo A., Kossenkov A.V., Donthireddy L., To T.K.J., Schug Z., Basu S., Wang F., et al. Fatty acid transport protein 2 reprograms neutrophils in cancer. Nature. 2019;569:73–78. doi: 10.1038/s41586-019-1118-2. [DOI] [PMC free article] [PubMed] [Google Scholar]