Abstract
Acute kidney injury (AKI) is associated with increased morbidity, prolonged hospitalization, and mortality, especially in high risk patients. Phosphodiesterase 5 inhibitors (PDE5Is), currently available as first-line therapy of erectile dysfunction in humans, have shown a beneficial potential of reno-protection through various reno-protective mechanisms. The aim of this work is to provide a comprehensive overview of the available literature on the reno-protective properties of PDE5Is in the various forms of AKI. Medline was systematically searched from 1946 to November 2019 to detect all relevant animal and human studies in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement. In total, 83 studies were included for qualitative synthesis. Sildenafil is the most widely investigated compound (42 studies), followed by tadalafil (20 studies), icariin (10 studies), vardenafil (7 studies), zaprinast (4 studies), and udenafil (2 studies). Even though data are limited, especially in humans with inconclusive or negative results of only two clinically relevant studies available at present, the results of animal studies are promising. The reno-protective action of PDE5Is was evident in the vast majority of studies, independently of the AKI type and the agent applied. PDE5Is appear to improve the renal functional/histopathological alternations of AKI through various mechanisms, mainly by affecting regional hemodynamics, cell expression, and mitochondrial response to oxidative stress and inflammation.
Keywords: acute kidney injury, avanafil, icariin, phosphodiesterase 5 inhibitors, renal insufficiency, sildenafil citrate, tadalafil, udenafil, vardenafil dihydrochloride, zaprinast
1. Introduction
AKI is considered a complex disorder with increased morbidity, prolonged hospitalization and mortality especially in high risk patients that may be attributed to various causes (pre-renal; renal, i.e., intrinsic to the renal parenchyma; and post-renal), including the use of nephrotoxic medications such as contrast media (CM), dehydration, sepsis, renal surgery, renal ischemia, ischemia–reperfusion (IR) renal injury, and urinary tract obstruction [1]. Criteria used for the diagnosis of AKI vary widely among studies in humans [2], including percent change in the baseline serum creatinine (sCr) levels (e.g., an increase of variously 25–50%) and absolute elevation from baseline sCr level (e.g., an increase of variously 0.5–2.0 mg/dL) [3]. These variable definitions have been addressed by two consensus groups, namely the Acute Dialysis Quality Initiative (ADQI) proposing the RIFLE (Risk, Injury, Failure, Loss and End-stage kidney disease) system [4] and more recently the Acute Kidney Injury Network (AKIN), which have attempted to standardize the diagnosis of AKI irrespective of etiology. According to the AKIN diagnostic criteria [5], AKI is an abrupt (within 48 h) reduction in human kidney function defined as occurrence of any of the following after a reno-toxic event: (a) absolute increase in sCr ≥ 0.3 mg/dL (≥ 26.4 μmol/L) or a percentage increase in sCr ≥ 50% (1.5-fold from baseline), which is known or presumed to have occurred within the prior seven days [6]; or (b) a reduction in urine output (documented oliguria of < 0.5 mL/kg/h for more than 6 h). This definition is in accordance with the current Clinical Practice Guideline for AKI by “Kidney Disease: Improving Global Outcomes” (KDIGO) [6]. Nevertheless, a recent systematic review evaluating the methods used to investigate AKI biomarkers showed that results are difficult to interpret, not comparable, and not consistently reproducible due to the impact of the variable AKI definitions still used to determine the outcome of interest in human studies (38.0% of the studies used the AKIN; 21.4% used the RIFLE; 20.3% used the KDIGO; and 20.3% used another definition) [2]. Similarly, variable definitions of AKI have been used in animal studies, a fact that has been recognized as an important limitation in translating preclinical findings in clinical studies [7,8] among others [9]. Several reviews of available animal models, including their advantages and disadvantages, have been discussed [10]; however, the types of models are often incomplete and many details, such as model techniques and modeling time, are not mentioned. Currently proposed AKI models include, among others: IR renal injury, including shock wave lithotripsy (SWL); injection of drugs, toxins, or endogenous toxins; ureteral obstruction, contrast-induced nephropathy (CIN); trauma such as burn; etc. [10,11,12,13,14,15,16].
Depending on the insult type, there are various mechanisms leading to renal damage such as renal vasoconstriction [17], vascular endothelial damage, cytokine expression [18], increase of IL-18, mediating acute tubular necrosis, caspase activity stimulation, p53 up-regulation [19], accumulation of toxic metabolites [20], mast cells/neutrophils activation, reactive oxygen species (ROS) generation causing lipid peroxidation that leads to cellular membrane destruction, excessive intracellular DNA breakdown, energy depletion, intracellular Ca2+ elevation, higher inducible nitric oxide (NO) synthase (iNOS) expression, NO deficiency, intra-parenchymal hemorrhage [21], fibrosis, direct cellular toxicity, tubular obstruction, vascular congestion, activation of angiotensin II axis [22], mitochondrial dysfunction [23], cell cycle arrest in G2 phase, ATPase activity inhibition, and cellular transport modification. ROS activate pro-apoptotic proteins eventually promoting Bax translocation (regulated by PI3K/Akt pathway) to the outer mitochondrial membrane, causing the release of cytochrome c in the cytosol [24]. Bax is also responsible for caspase 9 activation that activates caspase 3, triggering apoptosis. The tubular component of AKI consists of injured, necrotic/apoptotic cells falling into the lumen that cause obstruction/back leak of the filtrate to the interstitial space, inducing inflammation.
CIN is a real, albeit rare, entity in current clinical medical practice that represents a serious iatrogenic AKI form, occurring 24–72 h after administration of iodinated contrast media (CM) during angiographic or other procedures, such as urography [3,25]. The exact pathophysiology of CIN is not fully elucidated but oxidative stress is considered a major mechanism in CIN [26], and the identification of novel biomarkers that may more accurately detect renal function changes, reflect kidney damage, assist monitoring, and elucidate pathophysiology have attracted considerable scientific attention nowadays [27]. CM passing through the kidney results in an intense tubular transport that increases the activity in the thick ascending limb of Henle’s loop. This process increases oxygen consumption/metabolic activity of outer renal medulla, exacerbating the marginal hypoxic conditions. Prostanoids and NO are mainly responsible for the medullary vasodilatory response [28]. Therefore, any NO deficit may contribute to an additional hypoxic renal insult. CIN and IR renal injury share common pathways regarding the vasodilatory potential of NO. IR renal injury is a common complication during renal transplantation/artery angioplasty, partial nephrectomy, cardiopulmonary/aortic bypass surgery, and others [29]. In the IR renal injury setting, however, there are conflicting results reported, with some studies suggesting that NO induces cytotoxicity, and others showing that increased NOS activity is linked to increased renal blood flow in the ischemic region [30]. NOSs are a family of enzymes catalyzing the production of NO from L-arginine. There are three isoforms: the endothelial NOS (eNOS), the neuronal NOS (nNOS), and the iNOS involved in immune response. In the IR renal injury, endogenous NO is synthesized by eNOS and iNOS [31], while it is found that eNOS-mediated NO production plays a pivotal protective role in IR-induced AKI [1]. IR renal injury is also closely linked to ROS generation/apoptosis.
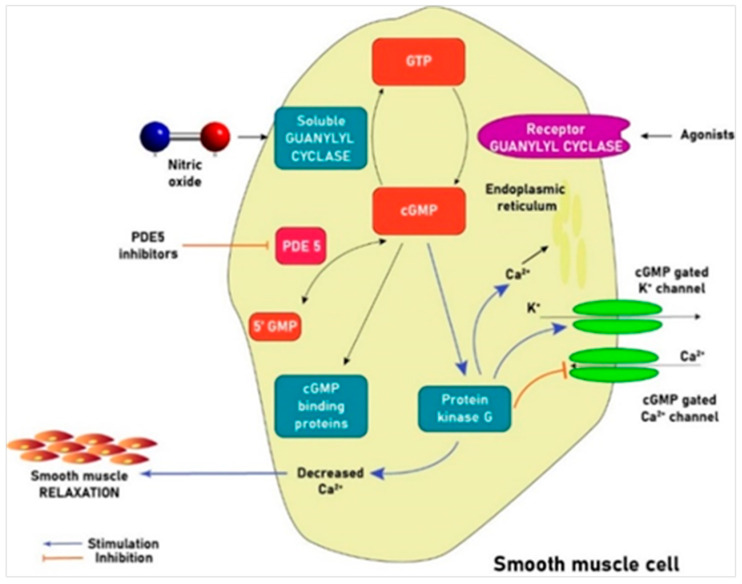
Prevention and/or management of the various AKI forms, such as CIN, is mainly supportive at present, consisting of intravenous hydration [32]. Even though the potential beneficial effects of many agents with antioxidant properties have been tested, the value of such substances other than sodium bicarbonate remains controversial [32,33]. Phosphodiesterase 5 (PDE5) inhibitors (PDE5Is) are currently recommended as first-line therapy of erectile dysfunction (ED) by enhancing the vasodilatory effects of NO [34]. Acting via the selective inhibition of cyclic guanosine monophosphate (cGMP)-specific PDE5 that metabolizes cGMP, the principal mediator of NO-induced smooth muscle relaxation, PDE5Is cause vasodilatation in the corpora cavernosa promoting erection (Figure 1). This class of drugs has shown beneficial potential through various mechanisms in some CIN animal models [33]. The aim of this paper is to provide a comprehensive overview of the available literature on the potential reno-protective properties of PDE5Is in the various forms of AKI.
Figure 1.
PDE5I-induced smooth muscle relaxation in the corpora cavernosa. cGMP is the principal mediator of NO-induced smooth muscle relaxation/vasodilation [35]. cGMP propels a series of intracellular changes including inhibition of Ca2+ entry into the cell, Ca2+ shift into the endoplasmic reticulum, activation of K+ channels leading to membrane hyperpolarization, and stimulation of a cGMP-dependent protein kinase that activates a myosin light chain phosphatase. All these actions promote smooth muscle relaxation. NO penetrates the cytoplasm of smooth muscle cells binding to guanylyl cyclase (sGC), which catalyzes the enzymatic conversion of GTP to cGMP. Elevation of cGMP stimulates cGMP-dependent protein kinase G leading to PDE5 phosphorylation/activation. PDE5 hydrolyzes cGMP in the cavernosal tissue. Inhibition of PDE5 results in smooth muscle relaxation with increased arterial blood flow, leading to compression of the sub-tunical venous plexus followed by penile erection [36].
2. Experimental Section
Medline (Ovid Medline Epub Ahead of Print, In-Process & Other Non-Indexed Citations, Ovid MEDLINE(R) Daily, and Ovid MEDLINE(R) 1946 to November 2019) was systematically searched to detect all relevant animal and human studies in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement [37], using the following keyword combinations (Medical Subject Headings; MeSH): PDE5i or avanafil or benzamidenafil or dasantafil or icariin or lodenafil or mirodenafil or sildenafil or tadalafil or udenafil or vardenafil or zaprinast combined with renal or kidney or nephrotoxicity or contrast or CIN or AKI or nephrotoxic or cisplatin or aminoglycoside or trauma or acute kidney injury or NSAIDS or non-steroidal or shock or sepsis or hypoperfusion or hypovolaemia or hypovolemia or renal artery stenosis or obstruction or acute tubular necrosis or glomerulonephritis or nephritis or renal failure or adenine or cyclosporine. The specific literature search strategy used is available in Appendix A. The reference lists of selected studies were screened for other potentially eligible studies. After excluding duplicates, citations in abstract form, and non-English citations, the titles/abstracts of full papers were screened for relevance, defined as original research focusing on the topic “nephropathy AND effects of phosphodiesterase 5 inhibitors”. Studies focusing on alterations of renal function and/or structure for >3 months (conventionally considered as following the KDIGO definition of chronic kidney disease (CKD) were excluded [6]). Two review authors (G.G. and IE.Z.) independently scanned the title and the abstract content, or both, of every record retrieved to determine which studies should be assessed further evaluated and extracted all data. Disagreements were resolved through consensus or by consultation with a third author (C.M.). A final draft of the manuscript was prepared after several revisions and approved by all authors.
3. Results
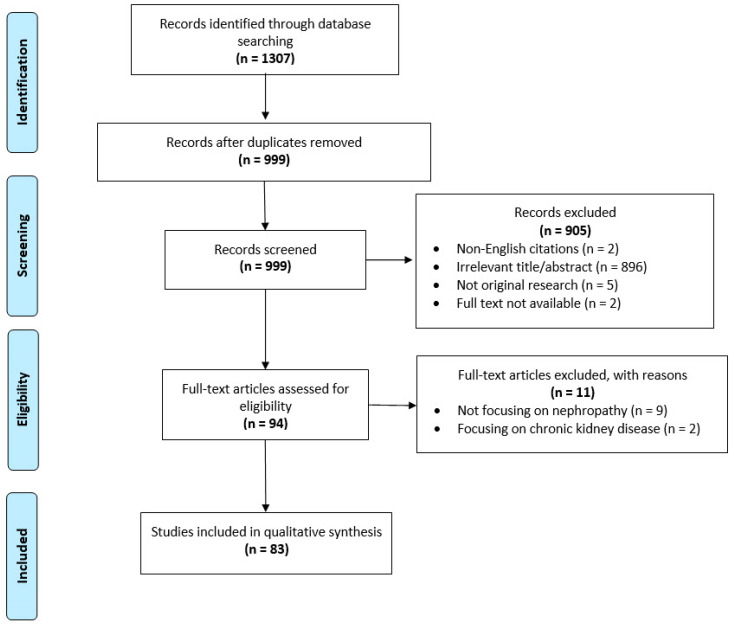
In total, 83 studies were included for qualitative synthesis (Figure 2). Among the 11 natural/synthetic agents currently available (avanafil, benzamidenafil, dasantafil, icariin, lodenafil, mirodenafil, sildenafil, tadalafil, udenafil, vardenafil, and zaprinast), sildenafil is the most widely investigated (n = 42 studies), followed by tadalafil (n = 20 studies), icariin (n = 10 studies), vardenafil (n = 7 studies), zaprinast (n = 4 studies), and udenafil (n = 2 studies). No studies on lodenafil, benzamidenafil, mirodenafil, avanafil, or dasantafil were detected. Most of the studies (n = 79) used animal models, including among others currently proposed AKI models (IR renal injury, including SWL; injection of drugs, toxins, or endogenous toxins; ureteral obstruction; CIN; trauma such as burn; etc.) [10,11,12,13,14,15,16] and variable definitions of AKI in line with the situation observed in human studies [2]. Only four human studies were detected: two preclinical studies utilizing human tissue [24,38] and two clinical trials [17,39].
Figure 2.
PRISMA flow chart showing the study selection procedure.
The reno-protective action of PDE5Is was evident in the vast majority of studies (n = 81), independently of the AKI type and the agent applied. Only one human study on sildenafil [39] and one animal study on zaprinast [40] failed to reveal any reno-protective action of PDE5Is, showing a neutral effect. PDE5Is appeared to be beneficial in AKI of various etiologies by improving renal functional/histopathological alternations through various mechanisms, such as affecting regional hemodynamics, cell expression, and mitochondrial response to oxidative stress and inflammation.
The main characteristics and results of the human studies evaluating the potential reno-protective effects of PDE5Is are summarized in Table 1 [17,24,38,39]. The main characteristics and results of the animal studies on currently proposed AKI models evaluating the potential reno-protective effects of sildenafil, tadalafil, icariin, vardenafil, zaprinast–udenafil are summarized in Table 2 [23,30,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61], Table 3 [29,35,45,49,62,63,64,65,66,67,68,69,70,71,72,73,74], Table 4 [18,75,76], Table 5 [45,77,78], and Table 6 [21,40,79,80], respectively. The main characteristics and results of the animal studies in the AKI-CKD transition spectrum (focusing on renal function and/or structure alterations for up to three months, not fulfilling the KDIGO definition for CKD [6]) evaluating the potential reno-protective effects of sildenafil, tadalafil, icariin, vardenafil, zaprinast–udenafil are summarized in Table A1 [19,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98], Table A2 [99,100], Table A3 [22,101,102,103,104], Table A4 [105,106,107,108], and Table A5 [109,110], respectively (Appendix B).
Table 1.
Human studies evaluating the potential reno-protective effects of phosphodiesterase 5 inhibitors.
| Reference Country/Year |
Type of Study | AKI Model | PDE5I Route | Timing | Sample | AKI Renal Effects | PDE5I Renal Effects | Outcome |
|---|---|---|---|---|---|---|---|---|
| [24]/ China/2019 |
Preclinical study on HEK-293 cell culture | Cisplatin Various doses Finally chosen 20 μΜ dose 24 h |
Icariin Various doses (0.25–2.0 μΜ) 24 h prior to cisplatin |
PRE | Centrifuged at 4 °C, 10,000 g, for 20 min | Reduced viability, ↑p-NF-Kb ↓GSH concentration ↑MDA levels,↑Bax, ↓Bcl-2 ↑ROS generation, ↑Caspace 3 ↑iNOS/TNF-a/IL-1β Nuclear fragmentation and cellular condensation |
Improved viability, ↓p-NF-kB ↑GSH concentration ↓MDA levels, ↓Bax, ↑Bcl-2 ↓ ROS generation, ↓Caspace 3 ↓iNOS/TNF-a/IL-1β Blunted apoptotic changes Antiapoptotic action (PI3K/Akt pathway) |
POS |
| [38]/ China/2017 |
Preclinical study using huMSCs in adult male Wistar rats | 2.5% Adenine Orally 4 weeks +4th generation huMSCs |
Icariin huMSCs were pretreated with 100 uM ICA for 1 week |
PRE | 3, 7, 14 days after treatment | ↑Urine outputm, ↑Urea, ↑Cr ↑Damage renal tissue, ↑TNF-a ↓SOD, ↑MDA, ↑IL-6, ↑IL-10 |
↓Urine output,↓Urea, ↓Cr ↓Damage renal tissue, ↓TNF-a ↑SOD, ↓MDA, ↓IL-6, ↓IL-10 ↑BMP-7, ↑bFGF |
POS |
| [17]/ Israel/2015 |
Clinical trial (non-RCT) |
PN with 20 min cold ischemia | Tadalafil Orally: 20 mg/day 1 day pre-operatively and 2 days postoperatively |
PRE and POST | Pre-op and at 1,3,8, 24, 48, 72 h post op | ↑NGAL, ↑KIM-1,↑sCr, ↓GFR | Attenuated all studied parameters | POS |
| [39]/ USA/2016 |
Clinical trial (RCT) |
RAPN | Sildenafil Orally 100 mg prior to RAPN |
PRE | ↓GFR | ↓GFR (No improvement) | NEUT |
Abbreviations: AKI, acute kidney injury; Bax, proapoptotic protein; Bcl-2, antiapoptotic gene; bFGF, basic fibroblast growth factor; BMP-7, bone morphogenetic protein-7; GSH, glutathione; HEK, human embryonic kidney cells; huMSCs, human umbilical cord mesenchymal stem cells; iNOS, inducible NOS; IL, interleukin; LY6G, MDA, malondialdehyde; NOX-4, NADPH oxidase 4; PDE5I, phosphodiesterase 5 inhibitor; p-NF-Kb, phosphorylation nuclear factor kappa-light-chain-enhancer of activated B cells; PN, partial nephrectomy; RAPN, Robot assisted partial nephrectomy; RCT, randomized controlled trial; ROS, reactive oxygen species; sCr, serum creatinine; SOD, superoxide dismutase; TNF-a, tumor necrosis factor a; ↓, reduced; ↑, increased.
Table 2.
Animal studies evaluating the potential reno-protective effects of sildenafil.
| Reference /Country/Year |
Studied Animal | AKI Model | PDE5I Route | Timing | Sample | AKI Renal Effects | PDE5I Renal Effects | Outcome |
|---|---|---|---|---|---|---|---|---|
| [41]/ South Korea/2009 |
Male Sprague Dawley rats |
Cisplatin Single intraperitoneal injection 5 mg/kg |
Sildenafil Intraperitoneal 0.4 mg/kg Just after the injection of cisplatin |
POST | Left nephrectomy 96 h post cisplatin |
↑BUN, ↑sCr, ↑Bax/Bcl-2 ratio ↑Caspase 3 expression ↑TUNEL positive cells Loss of brush border Vacuolation/Desquamation |
↓sCr, ↓Bax/Bcl-2 ratio ↓Caspase 3 expression ↓TUNEL positive cells ↑eNOS and iNOS Significantly attenuated renal changes |
POS |
| [30]/ Korea/2009 |
Male Sprague Dawley rats |
IR renal injury model | Sildenafil Intraperitoneal 0.5 mg/kg 1 h prior to ischemia |
PRE | Depending on the group 0-168 h after reperfusion | ↑BUN, ↑sCr, ↑cGMP ↑Bax/Bcl-2 ratio, ↑Caspase 3 activity ↑TUNEL positive cells Loss of brush border Vacuolation/Desquamation |
↓BUN, ↓sCr, ↑↑ cGMP ↓Bax/Bcl-2 ratio, ↓Caspase 3 activity ↓TUNEL positive cells ↑↑ ERK activity Attenuated all histological changes |
POS |
| [42]/ Turkey/2010 |
Male Wistar albino rats | IR renal injury model | Sildenafil Orally 60 min pre-operatively |
PRE | Left nephrectomy either at 45 min post occlusion or at 105 min post occlusion and reperfusion injury | ↑MPO enzyme level and activity ↑TBARS Sclerosis of glomeruli Enlargement of Bowman space Loss of microvilli/Pyknotic nuclei Tubular necrosis/Interstitial edema Leucocyte infiltration Glomerular and tubular degeneration |
↓MPO enzyme level and activity ⇔TBARS Attenuated tubular damage Preserved normal morphology Significantly decreased neutrophil infiltration |
POS |
| [43]/ Brazil/2010 |
Wistar rats | IR renal injury model | Sildenafil Orally 1 mg/kg 60 min prior to ischemia |
PRE | At 24 h and 7 days scintigraphy and nephrectomy | Scintigraphy: functional deficit representing ATN No PDE5i: ↑ cellular necrosis Vacuolation Intratubular cast formation |
Reversed effect to normal split function PDE5i: just dilatation of tubular lumen No significant change in histology |
POS |
| [44]/ Oman/2011 |
Male Wistar rats | Cisplatin Single intraperitoneal injection 5 mg/kg |
Sildenafil Intraperitoneally 0.4 mg/kg for 5 days or Sildenafil Subcutaneously 10 mg/kg for 5 days |
POST | Blood samples and bilateral nephrectomy 5 days post treatment | ↓RBF, ↓BP, ↓Body weight ↑Urine output ↑BUN, ↑sCr, ↓CrCl ↑N-acetyl-β-D-glycosaminidase ↑TNF-a (plasma and renal) ↑Renal platinum concentration Acute Tubular Necrosis/Apoptotic cells |
↑RBF, ↑BP (i.p.) No improvement in b.w. and u.o. ↓BUN, ↓sCr, ↑CrCl (i.p.) ↓N-acetyl-β-D-glycosaminidase Minimal improvement in TNF-a No change in platinum concentration Improvement of histological changes |
POS |
| [45]/ Turkey/2011 |
Wistar albino rats | UUO model |
Sildenafil-orally-1 mg/day Vardenafil-orally-0.5 mg/day Tadalafil-orally-10 mg/72 h For 30 days |
POST | 30 days | ↑Tubular cell apoptosis ↑ eNOS ↑ iNOS |
↓ Tubular cell apoptosis ↓ eNOS ↓ iNOS Sildenafil better results |
POS |
| [46]/ Spain/2011 |
Minipigs | IR renal injury model | Sildenafil Intravenously 0.7 or 1.4 mg/kg 30 min prior to or during warm ischemia |
PRE OR SIM | Monitoring of hemodynamics up to 45 min following unclamping | ↓Systemic MAP (especially 1.4 mg/kg) ↑RVF (0.7 mg/kg) |
POS | |
| [47]/ Turkey/2011 |
Male Wistar rats | CLP model |
Sildenafil Orally 10 or 20 mg/kg After the procedure |
POST | 16 h after CLP | ↓SOD, ↓GSH, ↑MPO, ↑LPO ↑Mean inflammation score ↑TNF-a |
↑SOD, ↑GSH, ↓MPO, ↓LPO ↓Mean inflammation score ↓TNF-a |
POS |
| [48]/ United Kingdom |
Female Large White Landrace crossbred pigs | Cardiopulmonary bypass 2.5 h | Sildenafil Intravenously 10 mg in 50 mL saline 0.9% |
SIM | 90 min pre-op 90 min post-op 24 h post-op |
↓CCl, ↑Proteinuria, ↑IL-18 ↓ NO Pseudodilation of proximal tubules ↑iNOS ↑ cortical expression endothelin-1 Inflammatory cell infiltration |
↑CCl ↓Proteinuria ↓IL-18 Significantly increased RBF (24 h) ↑NO Prevented phenotypic changes in proximal tubular cells ↓cortical expression endothelin-1 Preserved eNOS ↓iNOS ↓ inflammatory cell infiltration |
POS |
| [49]/ Turkey/2012 |
Male Sprague Dawley rats |
IR renal injury model | Sildenafil Orally: 1 mg/kg 60 min pre-operatively Tadalafil Orally: 1 mg/kg 60 min pre-operatively |
PRE | Nephrectomy post procedure | ↑MPO levels ↑MDA levels ↑iNOS gen, ↑eNOS expression ↑ apoptotic cells ↑p53 positive cells Leucocyte migration Edema/Tubular dilatation |
MPO: no significant improvement ↓MDA (Sdf), ⇔ MDA (Tdf) levels ↓iNOS gen, ↓eNOS expression ↓apoptotic cell death ( Sdf > Tdf) ↓p53 positive cells All changes were attenuated |
POS |
| [50]/ Germany/2013 |
NO-GC1 KO mice C57Bl/6Rj |
UUO model | Sildenafil Orally 100 mg/kg In the 4th week post op |
POST | 4 weeks post op | ↓cGMP ↓NO-stimulated guanyle cyclase activity (KO mice) |
↑cGMP ↑NO sensitivity ↓SBP (more efficient in operated group rather than KO group) |
POS |
| [23]/ USA/2013 |
Female New Zealand white rabbits | Folic Acid Intraperitoneally Single dose 250 mg/kg |
Sildenafil Intraperitoneally 24 h after injury 0.3 mg/kg/day For 6 days |
POST | Blood samples and kidneys were harvested 24 h post treatment | ↓mRNA expression COX1 and Tfam ↓mtDNA copy number ↑KIM-1 |
↑mRNA expression COX1 and Tfam ↑mtDNA copy number ↓KIM-1 |
POS |
| [51]/ Egypt/2014 |
Sprague Dawley male rats | Cisplatin Intraperitoneally 6 mg/kg |
Sildenafil Intraperitoneally 2 mg/kg 1 h before and 24 h after cisplatin injection |
PRE and POST | 96 h after cisplatin injection | ↑BUN, ↑sCr, ↑MDA, ↑TNF-a ↑Caspase-3, ↓SOD ↓Nitrite/nitrate level Acute tubular necrosis |
↓BUN, ↓sCr, ↓MDA, ↓TNF-a ↓Caspase-3, ↑SOD ↑Nitrite/nitrate |
POS |
| [52]/ Turkey/2014 |
Adult female Wistar albino rats | Burn model | Sildenafil Orally 10 or 20 mg/kg just after burn |
POST | 24 h after the scald burn | Renal: ↑MDA, ↓Gpx, ↑VEGF ⇔ Flt-1, ⇔TAC, ⇔OSI, ⇔TOS Serum: ↑MDA, ↓Gpx, ⇔VEGF, ⇔Flt-1, ↓TAC, ⇔OSI, ↑TOS, ⇔Flt-1/VEGF ratio |
Renal: ↓MDA, ↑Gpx, ↓VEGF ⇔Flt-1 (T10), ⇔TAC, ⇔OSI, ⇔TOS(T20) Serum: ↓MDA, ↑Gpx, ⇔VEGF ⇔Flt-1, ↑TAC, ↓OSI (T10) ↑Flt-1/VEGF ratio (T10) ↓TOS (T10) ↓Histopathological scores (no significant difference in T20) |
POS |
| [53]/ Egypt/2014 |
Male Wistar rats | Gentamicin Intraperitoneally 100 mg/kg/day for 6 days |
Sildenafil Orally 5 mg/kg/day for 6 days 1 h before gentamycin |
PRE | 24 h after last gentamycin injection | ↑Cr, ↑Urea, ↑urinary albumin ↑MDA, ↑nitrite/nitrate levels ↓CAT (renal), ↓SOD, ↑iNOS, ↓eNOS Degeneration and necrobiosis in epithelial cells |
↓Cr, ↓Urea, ↓urinary albumin ↓MDA, ↓nitrite/nitrate levels ↑CAT (renal), ↑SOD ↓iNOS, ↑eNOS Reversed histological alterations |
POS |
| [54]/ USA/2014 |
Male wild-type (WT) littermates or PKG Tg mice | UUO model |
Sildenafil Subcutaneously 12 mg/kg twice daily for 14 days |
POST | 14 days | ↓Renal PKG activity Increase (↑) at Ang II, Collagen type I, III mRNA, α-SMA, E-cadherin, TNF-a, TGF-β1, pSmad2, ICAM-1 ↑Macrophage infiltration |
↑Renal PKG activity Decrease (↓) at Ang II, Collagen type I, III mRNA, α-SMA, E-cadherin, TNF-a, TGF-β1, pSmad2, ICAM-1 ↓Macrophage infiltration |
POS |
| [55]/ Brazil/2014 |
New Zealand white rabbits | CIN model | Sildenafil Orally 6 mg/kg before CM or 6 mg/kg before CM and 8 hourly for 48 h |
PRE and POST | 1/2/24/48 h |
No changes in kidney to body weight ratio ↑sCr ↓Na, ↑K Multifocal tubular necrosis Tubular degeneration Luminal protein casts |
No significant changes in kidney to body weight ratio ↓↓sCr (continuous) ↑Na, ↓K Continuous treatment blunted all changes |
POS |
| [56]/ Egypt/2015 |
Male Sprague-Dawley rats | IR renal injury model | Sildenafil Orally (1 mg/kg) 60 min before anesthesia |
PRE | Blood + urine samples (basal, at 2, 24, 48 h and 7 days) + Kidney tissue |
↑sCr, ↑BUN, ↓Bcl-2 ↓Nrf2/HO-1/NQO-1 (genes) ↑ Proinflammatory cytokine genes (TNF-a, ICAM-1, IL-β) ↓Nrf 2 protein expression Acute tubular necrosis, detachment of epithelial cells from basement membrane, intracellular cast formation, loss of brush border, neutrophil infiltration |
No improvement in BUN/sCr, ↑Bcl-2 ↑Nrf2/HO-1/NQO-1 (genes) ↓ Proinflamamtory cytokine genes (TNF-a, ICAM-1, IL-β) ↑ Nrf 2 protein expression Improved histological features of renal injury (mild tubular necrosis) |
POS |
| [57]/ Brazil/2016 |
Male Wistar rats | CIN model | Sildenafil Orally 50 mg/kg/d 7 days (started 5 days before CM) |
PRE and POST | 48 h after CM administration | ↑BUN, ↑sCr, ↑urine protein ↓GFR, ↓RPF, ↑RVR ↑superoxide anions production ↑H2O2 production ↑peroxynitrite and hydroxyl production ⇔ NO Reduced body weight Renal hypertrophy |
↓BUN, ↓sCr, ↓urine protein ↑GFR, ↑RPF, ↓RVR ⇔superoxide anions production ↓ H2O2 production ↓peroxynitriteand hydroxyl production ⇔ NO No effect of PDE5 on histological changes |
POS |
| [58]/ Egypt/2016 |
Male Wistar albino rats | IR renal injury model | Sildenafil Intraperitoneally (0.5 + 1.0 mg/kg) 1 h before ischemia |
PRE | Blood/kidney tissue samples 24 h after reperfusion | ↓CrCl, ↑ BUN, ↑Uric acid, ↑FeNa ↑Plasma potassium ↓GSH levels,↑TBARS, ↑SAG levels Glomerular damage, detachment of basement membrane, loss of brush border, tubular dilation, atroprhy, neutrophil accumulation |
↑CrCl, ↓BUN, ↓Uric acid ↓FeNa ↓Plasma potassium ↑GSH levels, ↓TBARS ↓ SAG levels ↓Renal tissue damage |
POS |
| [59]/ Turkey/2018 |
Female Wistar albino rats |
CIN model | Sildenafil Orally 50 mg/kg 48 h prior to CM |
PRE | 48 h after CM administration | ↑HIF-2a (serum and tissue) ↑ BUN, ↑Cr (serum and urine) Hemorrhage, shedding of brush border, tubular vacuolization, degeneration, inflammatory cell infiltration, intratubulat cast obstruction |
↓HIF-2a (serum and tissue) ↓ sCr Sildenafil improved all histological changes |
POS |
| [60]/ Egypt/2018 |
Male albino rats | Cisplatin 5 mg/kg Single dose intraperitoneally |
Combination Sildenafil, Orally 40 mg/kg Gemfibrozil-Orally– 100 mg/kg 14 days prior or after |
PRE OR POST | Day 17 | ↑sCr, ↓HO-1, ↓GSH ↓eNOS, ↓TNF-a ↑Tubular injury/tubular necrosis |
All changes improved with sildenafil and gemfibrozil especially in the group given after cisplatin | POS |
| [61]/ Egypt/2019 |
Mongrel dogs (aged 2-3 years) | IR renal injury model | Sildenafil Orally 1 mg/kg 1 h prior to operation or In the perfusion fluid 0.5 mg/kg during the operation |
PRE OR SIM | Prior and at the end of the experiment (Day 1,3,7,14) |
↑sCr, ↑BUN, ↓GFR ↑caspase 3, ↑Nrf2 ↑TNF-a, ↑ IL-1Β, ↑ICAM -1 ↓eNOS Renal degeneration Cortical and medullary interstitial fibrosis |
↓sCr, ↓BUN, ↑GFR ↓caspase 3, ↑↑Nrf2 ↓TNF-a, ↓IL-1Β, ↓ICAM -1 ↑eNOS Significantly improved all histological changes |
POS |
Abbreviations: AKI, acute kidney injury; Ang II, angiotensin II; Bax, proapoptotic protein; Bcl-2, antiapoptotic gene; BP, blood pressure; BUN, blood urea nitrogen; Ca2+, calcium; CAT, catalase; cGMP, cyclic guanosine monophosphate; CIN, contrast induced nephropathy; CLP, caecal ligation and puncture; COX, cyclo-oxygenase CrCl, creatinine clearance, eNOS, endothelial NOS, FeNa, fractional excretion of sodium, GFR, glomerular filtration rate; GPx, glutathione peroxidase; GSH, glutathione; HIF-2a, heterodimeric nuclear transcription factor-2 alpha; HO-1, heme oxygenase 1; IR, ischemia reperfusion; ICAM-1, intercellular adhesion molecule 1; IL, interleukin; iNOS, inducible NOS; K, potassium; KIM-1, kidney injury molecule-1; LPO, lipid peroxidation; MAP, mean arterial pressure; MDA, malondialdehyde; MPO, myeloperoxidase; Na, sodium; NO, nitric oxide; NRF2, nuclear erythroid related factor 2; OSI, oxidative stress index; P, phosphorus; PDE5I, phosphodiesterase 5 inhibitor; PKG, protein kinase G; pSmad2, antibody; RBF, renal blood flow; RPF, renal plasma flow; RRI, renal resistive index; RVF, renal vascular flow; RVR, renal vascular resistance; SAG, superoxide anion generation; sCr, serum creatinine; sFlt1, soluble fms-like tyrosine kinase-1; SOD, superoxide dismutase; SBP, systolic blood pressure; TAC, total antioxidant capacity; Tfam, mitochondrial transcription factor; TGF-β1, transforming growth factor beta 1; TBARS, thiobarbituric acid reactive substances; TNF-a, tumor necrosis factor a; TOS, total oxidant status; TUNEL, terminal deoxynucleotidyl transferase dUTP nick end labeling; UUO, unilateral ureteral obstruction; VEGF, vascular endothelia growth factor; ↓, reduced; ↑, increased ⇔, no change.
Table 3.
Animal studies evaluating the potential reno-protective effects of tadalafil.
| Reference/ Country/Year |
Studied Animal | AKI Model | PDE5I Route | Timing | Sample | AKI Renal Effects | PDE5I Renal Effects | Outcome |
|---|---|---|---|---|---|---|---|---|
| [62]/ Turkey/2019 |
New Zealand rabbits | UUO model |
Tadalafil Orally 10 mg/72 h for 30 days prior to obstruction |
PRE | 4th hour and 1st and 3rd day | ↑Resistivity index ↑Pulsatility index |
↓Resistivity index ↓Pulsatility index In the non-obstructed kidney reduced resistivity index at 4th hour then normal |
POS |
| [63]/ Turkey/2011 |
Male Sprague Dawley rats |
IR renal injury model | Tadalafil Orally 1 mg/kg 60 min pre-operatively |
PRE | At 45 min post occlusion or at 105 min post occlusion and reperfusion injury | Sclerosis of glomeruli Enlargement of Bowman space Loss of microvilli/Tubular necrosis Interstitial edema/Leucocyte infiltration Hyaline degeneration |
Attenuated histological changes and decreased neutrophil infiltration | POS |
| [45]/ Turkey/2011 |
Wistar albino rats | UUO model |
Sildenafil-orally-1 mg/day Vardenafil-orally-0.5 mg/day Tadalafil-orally-10 mg/72 h For 30 days |
POST | 30 days | ↑Tubular cell apoptosis ↑eNOS ↑iNOS |
↓Tubular cell apoptosis ↓eNOS ↓iNOS Sildenafil better results |
POS |
| [64]/ Turkey/2011 |
Male Wistar albino rats | IR renal injury model | Tadalafil Orally 10 mg/kg 60 min pre-operatively |
PRE | Left nephrectomy at 120 min post-operatively | ↑Total oxidant status Tubular necrosis/Vacuolization Congestion/Mononuclear cell infiltration |
↑ Total antioxidant status Reduced all injuries to the renal tissue. |
POS |
| [65]/ USA/2012 |
Adult female pigs | IR renal injury model | Tadalafil 40 mg Two doses (12 h before and just prior to surgery) |
PRE | Induction and Days 1, 3, 7 post occlusion |
↑Creatinine after nephrectomy ↑↑ Creatinine Day 1 post ischemia |
↓Creatinine after nephrectomy No significant change in creatinine Day 1 post ischemia |
POS |
| [49]/ Turkey/2012 |
Male Sprague Dawley rats |
IR renal injury model | Sildenafil Orally: 1 mg/kg 60 min pre-operatively Tadalafil Orally: 1 mg/kg 60 min pre-operatively |
PRE | Nephrectomy post procedure | ↑ MPO levels ↑MDA levels ↑iNOS gen, ↑eNOS expression ↑apoptotic cells ↑p53 positive cells Leucocyte migration Edema/Tubular dilatation |
MPO: no significant improvement ↓MDA (Sdf), ⇔MDA (Tdf) levels ↓iNOS gen, ↓eNOS expression ↓apoptotic cell death (Sdf > Tdf) ↓p53 positive cells All changes were attenuated |
POS |
| [66]/ Israel/2013 |
Male Sprague Dawley rats |
IR renal injury model | Tadalafil Orally 10 mg/kg 24-hr prior to ischemia |
PRE | 30/60 min after nephrectomy 60/120/180/240 min after clamping |
↑V, ↑UNaV, ↑FeNa, ↓GFR, ⇔RPF, ↑NGAL, ↑KIM-1 Tubular dilatation/Loss of brush border Necrosis and cast formation |
↓V, ↓UNaV, ↓FeNa, ↑GFR, ↑RPF, ↓NGAL, ↓KIM-1 Blunted all changes |
POS |
| [67]/ China/2014 |
Male Wistar rats | Sepsis model | Tadalafil Orally 10 mg/kg 24 h prior to procedure for 28 days |
PRE and POST | Nephrectomy and samples at: 8 days post treatment and 6 weeks post treatment | ↑Systolic and diastolic BP, ↑NO ↑BUN, ↑sCr, ↑MDA levels ↓SOD, ↑TGF-β |
↓Systolic and diastolic BP, ↓NO, ↓BUN, ↓sCr, ↓MDA levels, ↑SOD ↑IL-10, ↓TNF-a, ↓IL-1β, ↓TGF-β ↓RANTES, ↓MIP-1β, ↓MCP-1 |
POS |
| [68]/ Turkey/2015 |
Female Wistar albino rats | IR renal injury model | Tadalafil Orally 10 mg/kg 24 h prior to procedure |
PRE | Cardiac blood samples and nephrectomy after reperfusion injury | No significant difference Severe tubular dilatation degeneration and necrosis/Enlargement of Bowman capsule |
in IMA/NO/MDA levels Blunted all changes |
POS |
| [69]/Turkey/2015 | Wistar albino rats | IR renal injury model | Tadalafil Intraperitoneally 10 mg/kg Immediately prior to procedure |
PRE | Blood samples and nephrectomy following 60 min of reperfusion injury | ↑MDA levels (serum/renal) ↓TAC levels (serum/renal) ↑APAF-1, ↑iNOS, ↑eNOS Loss of nucleus/Cellular edema Vacuolization/Brush border loss Tubular dilatation/edema Interstitial congestion |
⇔MDA (renal), ↓MDA (serum) ⇔TAC (renal), ↑TAC (serum) ↓APAF-1, ↓iNOS, ↓eNOS Damage was significantly less after tadalafil treatment |
POS |
| [35]/Turkey/2015 | Female Wistar albino rats | CIN model | Tadalafil Orally 10 mg/kg immediately after contrast |
POST | 48 h after CM administration | Significant weight loss after dehydration ↑Serum cystatin C ↑BUN, ↑sCr, ↑MDA Medullary congestion |
Significant weight loss after dehydration ↓Serum cystatin C ↓BUN, ↓sCr, ↓MDA Similar histological findings |
POS |
| [29]/ Egypt/2016 |
Adult male albino rats | IR renal injury model | Tadalafil Orally (5 mg/kg) Pre-treatment |
PRE | Blood/kidney tissue samples 6 h after reperfusion | ↑sCr, ↑BUΝ, ↑MDA levels ↓SOD activity, ↑MPO activity ↑ICAM-1, ↑TNF-a, ↑IL-1β ↑Caspase-3 activity Congestion and interstitial hemorrhage, proximal and tubular necrosis |
↓sCr, ↓BUΝ, ↓MDA levels ↑SOD activity, ↓MPO activity ↓ICAM-1, ↓TNF-a, ↓IL-1β ↓Caspase-3 activity Dilated proximal, distal, and collecting tubules and interstitial connection |
POS |
| [70]/ Nigeria/2016 |
Male Wistar rats | Cisplatin Intraperitoneal 5 mg/kg |
Tadalafil Orally: 2 or 5 mg/kg for 7 days pretreatment |
PRE | Blood samples and renal tissue obtained 3 days post cisplatin | ↓Na/K/HCO3/Ca2+/P ↑BUN, ↑sCr, ↑MDA/GPx ↓GSH/SOD/CAT (renal) |
Significant attenuation of all histological and biochemical alterations | POS |
| [71]/ Israel/2017 |
Male albino Wistar rats | CLP model | Tadalafil Orally 5 or 10 mg/kg End of the procedure |
POST | Left nephrectomy + Blood samples 16 h postoperatively |
↓CAT, ↓SOD, ↑IL-6, ↑sCr, ↑MPO, ↑MDA, ↑Cystatin C ↑Mac387 antibody ↑Tubular injury, glomerulus deformities ↑Inflammatory cell infiltration |
↑CAT, ↑SOD, ↓IL-6, ↓sCr, ↓MPO, ↓MDA, ↓Cystatin C ↓Mac387 antibody ↓Tubular injury, glomerulus deformities ↓Inflammatory cell infiltration |
POS |
| [72]/ Brazil/2017 |
Male Wistar rats | IR renal injury model | Tadalafil Orally 10 mg/kg 1 h pre-procedure |
PRE | After nephrectomy | Interstitial Leucocyte accumulation | Successful reversal by tadalafil | POS |
| [73]/ Brazil/2017 |
Male Wistar rats | IR renal injury model | Tadalafil Orally: 10 mg/kg 1 h before ischemia |
PRE | Fluorescence imaging (ICG) Blood samples |
↓ICG signal, ↑TNF-a, ↑IL-1β ↑IL-6 ↑BUN, ↑sCr, ↑CRP |
↑ICG signal, ↓TNF-a, ↓IL-1β ↓IL-6 ↓BUN, ↓sCr, ↓CRP |
POS |
| [74]/ Turkey/2019 |
Male Sprague Dawley rats | UUO model | Tadalafil Orally 10 mg/72 h |
--- | 15 days post ligation | ↑aSMA, ↑TGF-β Partial: inflammatory cell infiltration/severe epithelial atrophy/edema of epithelial cells/vacuolation Complete: macrophage infiltration/hemorrhage/irregular dark nuclei/thinner epithelium/denuded epithelial cells |
↓aSMA, ↓TGF-β Attenuation of all changes with tadalafil |
POS |
Abbreviation: AKI, acute kidney injury; APAF-1, apoptotic protease activating factor 1; aSMA, α-smooth muscle actin; BUN, blood urea nitrogen; Ca2+, calcium; CAT, catalase; CIN, contrast induced nephropathy; CLP, caecal ligation and puncture; CRP, c-reactive protein; eNOS, endothelial NOS; FeNa, fractional excretion of sodium; GFR, glomerular filtration rate; GPx, glutathione peroxidase; GSH, glutathione; HCO3−, bicarbonate; IR, ischemia/reperfusion; ICAM-1, intercellular adhesion molecule 1; IL, interleukin; ICG, indocyanine green; IMA, ischemia modified albumin; iNOS, inducible NOS; K, potassium; KIM-1, kidney injury molecule-1; Mac387, Macrophage antibody; MCP-1, monocyte chemoattractant protein 1; MDA, malondialdehyde; MIP-1β, macrophage inflammatory protein-1β; MPO, myeloperoxidase; Na, sodium; NGAL, neutrophil gelatinase-associated lipocalin; NO, nitric oxide; P, phosphorus; PDE5I, phosphodiesterase 5 inhibitor; RANTES, Regulated upon Activation Normal T-cell Expressed, and Secreted; RPF, renal plasma flow; sCr, serum creatinine; Sdf, sildenafil; SOD, superoxide dismutase; TAC, total antioxidant capacity; Tdf, tadalafil; TGF-β1, transforming growth factor beta 1; TNF-a, tumor necrosis factor a; UNaV, urine sodium volume; UUO, unilateral ureteral obstruction; V, urine volume; ↓, reduced; ↑, increased ⇔, no change.
Table 4.
Animal studies evaluating the potential reno-protective effects of icariin.
| Reference/ Country/Year |
Studied Animal | AKI Model | PDE5I Route | Timing | Sample | AKI Renal Effects | PDE5I Renal Effects | Outcome |
|---|---|---|---|---|---|---|---|---|
| [75]/ China/2015 |
Male BALB/c mice | Cisplatin 15 mg/kg Intraperitoneal |
Icariin Orally 30 or 60 mg/kg/day For 6 days |
PRE | At 6 days | ↑BUN, ↑sCr, ↑MDA ↓GSH concentration, ↓Catalase ↓SOD activity, ↑TNF-a, ↑NF-Kb ↑TUNEL positive cells ↑Caspase-3, ↓Bcl-2 Tubular congestion/edema Loss of brush border/Tubular cell flattening and necrosis/nuclear pyknosis Severe invasion of inflammatory cells |
↓BUN, ↓sCr,↓MDA ↑GSH concentration, ↑Catalase ↑SOD activity, ↓TNF-a, ↓NF-kB ↓TUNEL positive cells ↓Caspase-3, ↑Bcl-2 Partial improvement of the features (dose dependent) |
POS |
| [18]/ China/2018 |
Male C57BL/6N mice | CLP model | Icariin Orally 30 or 60 mg/kg 3 days prior to surgery |
PRE | Observed for 5 days | ↑BUN, ↑sCr, ↑MDA levels ↑IL-1β/IL-6/TNF-a ↑ NF-κB ↓ GSH concentration ↓Catalase, ↓SOD activity ↑TUNEL +ve cells ↑Renal vascular permeability ↑Bax,↓Bcl-2, ↑Caspase 3 Extensive tubular necrosis/Loss of brush border |
↓BUN, ↓sCr, ↓MDA levels ↓IL-1β/IL-6/TNF-a, ↓ NF-κB ↑GSH concentration ↑Catalase, ↑SOD activity ↓TUNEL +ve cells (60>30) ↓Renal vascular permeability ↓Bax, ↑Bcl-2, ↓Caspase 3 ↑Survival (both doses) Improvement in all histological features |
POS |
| [76]/ Taiwan/2019 |
Adult C57BL/6J | UUO model | Icariin Orally 20 mg/kg/day For 3 days prior and 3, 7, or 14 days after |
PRE and POST | 3, 7, or 14 days post ligation | ↑TGF-β, ↑α-SMA ↑fibronectin ↑NOX-4, ↓E-cadherin, ↓SOD-1 ↓Catalase, ↑CTGF, ↑Ly6G ↑F4/80, ↑phosphorylation IL-1β ↑Phosphorylation COX-2/NF-κΒ-65 Tubular dilatation/interstitial cell proliferation/inflammatory cell infiltration/tuft to capsule glomerular adhesions/collagen deposition |
↓TGF-β, ↓α-SMA, ↓fibronectin ↓NOX-4,↑E-cadherin, ↑SOD-1 ↑Catalase, ↓CTGF, ↓Ly6G ↓F4/80, ↓phosphorylation IL-1β ↓Phosphorylation COX-2/NF-κΒ-65 Non-significant moderate reversal by icariin |
POS |
Abbreviations: AKI, acute kidney injury; Bcl-2, antiapoptotic gene; BUN, blood urea nitrogen; CLP, caecal ligation and puncture; COX, cyclo-oxygenase; CTGF, connective tissue growth factor; F4/80, macrophage marker; GSH, glutathione; IL, interleukin; LY6G, neutrophil marker; MDA, malondialdehyde; NF-κB, nuclear factor kappa-like chain-enhancer of activated B cells; NOX-4, NADPH oxidase 4; PDE5I, phosphodiesterase 5 inhibitor; sCr, serum creatinine; SOD, superoxide dismutase; TGF-β1, transforming growth factor beta 1; TNF-a, tumor necrosis factor a; TUNEL, Terminal deoxynucleotidyl transferase dUTP nick end labeling; UUO, unilateral ureteral obstruction; ↓, reduced; ↑, increased.
Table 5.
Animal studies evaluating the potential reno-protective effects of vardenafil.
| Reference/ Country/Year |
Studied Animal | AKI Model | PDE5I Route | Timing | Sample | AKI Renal Effects | PDE5I Renal Effects | Outcome |
|---|---|---|---|---|---|---|---|---|
| [45]/ Turkey/2011 |
Wistar albino rats | UUO model |
Sildenafil-orally-1 mg/day Vardenafil-orally-0.5 mg/day Tadalafil-orally-10 mg/72 h For 30 days |
POST | 30 days | ↑Tubular cell apoptosis ↑eNOS ↑ iNOS |
↓Tubular cell apoptosis ↓ eNOS ↓iNOS Sildenafil better results |
POS |
| [77]/ Greece/2013 |
Male Wistar rats | IR renal injury model |
Vardenafil Intravenously 0.02, 0.2, 2, 20 μg/kg 1 h pre-operatively or 2μg/kg 45 min post occlusion |
PRE or POST | Blood samples and right nephrectomy 4 h post ischemia | Edema Loss of brush border Nuclear condensation |
↓sCr (0.2, 2, 20 μg/kg) No change when given post-ischemia ↓FENa, ↑Renal uptake of tracer ↑cGMP, ↑ERK 1/2 phosphorylation Renoprotection (in scintigraphy) Significant improvement in all histo-logical changes irrespectively of dose |
POS |
| [78]/ Brazil/2015 |
Male Wistar rats | IR renal injury model | Vardenafil Solution in a probe (1 mg/mL in 10 mg/kg) 1 h prior the ligation |
PRE | Left nephrectomy Cytophotometry 24 h after reperfusion |
↑Cleaved caspase-3 ↑sCr ↑Vacuolar degeneration |
↓ Cleaved caspase-3 ↓ Vacuolar degeneration |
POS |
Abbreviations: AKI, acute kidney injury; cGMP, cyclic guanosine monophosphate; eNOS, endothelial NOS; ERK, extracellular signal-regulated kinase; FeNa, fractional excretion of sodium; IR, ischemia/reperfusion; iNOS, inducible NOS; PDE5I, phosphodiesterase 5 inhibitor; sCr, serum creatinine; UUO, unilateral ureteral obstruction; ↓, reduced; ↑, increased.
Table 6.
Animal studies evaluating the potential reno-protective effects of zaprinast and udenafil.
| Reference/ Country/Year |
Studied Animal | AKI Model | PDE5I Route | Timing | Sample | AKI Renal Effects | PDE5I Renal Effects | Outcome |
|---|---|---|---|---|---|---|---|---|
| [79]/ USA/1995 |
Male Sprague-Dawley rats | IR renal injury model | Zaprinast Intravenously 0.03 and 0.3 mg/kg/min 24 h after ischemia |
POST | During clamping, PDE5i infusion, up to 6 days following ischemia | ↑sCr, ↓GFR | ↓sCr, ↑GFR, ↓Low MAP ↑UNaV, ↑Urinary cGMP ↑Cortical and medullary blood flow |
POS |
| [40]/ USA/2013 |
Female Sprague-Dawley rats | IR renal injury model | Zaprinast Intraperitoneally 10 mg/kg or 20 mg/kg Single dose 30 min pre-operatively |
PRE | 24 h post operatively blood samples and left nephrectomy |
No statistically significant differrences in either BUN levels or sCr levels or histologic scores or TUNEL positive cells | NEUT | |
| [80]/ Germany/2017 |
6-8-week-old mice | UUO model | Zaprinast, Intraperitoneally 10 mg/kg/day for 7 days |
POST | After 7 days | ↑cGMP, ↑sCr | ↑↑cGMP, ↑MMP9, ↑TGF-β ⇔sCr, ↓Collagen |
POS |
| [21]/Turkey/2017 | Female Wistar albino rats | IR renal injury model | Udenafil Orally: 10 mg/kg 1 h prior to clamping |
PRE | 60 min and 24 h after reperfusion |
↑BUN, ↑sCr ↑MDA, ↑NGAL |
↓BUN, ↓sCr ↓MDA, ↓NGAL Lowest pathological damage rates |
POS |
Abbreviations: AKI, acute kidney injury; BUN, blood urea nitrogen; cGMP, cyclic guanosine monophosphate; GFR, glomerular filtration rate; IR renal, ischemia/reperfusion; MAP, mean arterial pressure; MDA, malondialdehyde; MMP9, Matrix metallopeptidase 9; NGAL, neutrophil gelatinase-associated lipocalin; PDE5I, phosphodiesterase 5 inhibitor; sCr, serum creatinine; TUNEL, terminal deoxynucleotidyl transferase dUTP nick end labeling; UNaV, urinary sodium excretion; UUO, unilateral ureteral obstruction; ↓, reduced; ↑, increased ⇔, no change.
4. Discussion
PDE5Is have received a lot of attention since the first drugs were launched in the market. Four potent selective agents (avanafil, sildenafil, tadalafil, and vardenafil) have been approved by the European Medicines Agency (EMA) and the Food and Drug Administration (FDA) for the treatment of ED [111,112]. ED can be managed successfully with currently available treatment options, but it cannot be cured and most patients will be treated without cause-specific options, such as the use of PDE5Is [34]. Exceptions are psychogenic, post-traumatic arteriogenic in young patients, and hormonal causes (e.g., hypogonadism) of ED, which are potentially curable with specific treatments that might be employed first, when such causes are detected [34]. Consequently, treatment strategy of ED should be tailored depending on invasiveness, efficacy, safety, cost, and patient preference of the currently available options; in the context of this strategy, PDE5Is are currently recommended strongly as first-line treatment option given that lifestyle changes are initiated/risk factors are modified prior to or at the same time as initiating ED treatment [34].
Other EMA/FDA approved indications of PDE5Is include pulmonary arterial hypertension (PAH) (sildenafil and tadalafil) and management of men with moderate to severe LUTS secondary to benign prostatic obstruction with or without ED (tadalafil) [34,113,114,115]. Besides the aforementioned agents, there are other non-EMA/FDA approved PDE5Is including benzamidenafil, dasantafil, lodenafil, mirodenafil, and udenafil, some of which are commercially available in a few countries (lodenafil in Brazil; mirodenafil in South Korea; and udenafil in South Korea, Russia, and Philippines) [113]. Other agents with weak PDE5I properties include icariin and zaprinast [116]. Icariin, a prenylated flavonol glycoside extracted from plants of the Epimedium genus, has demonstrated PDE5I activity in vitro, enhancement of NO, and antioxidant activity [116]. It has been widely used in Chinese traditional medicine. It shows peak concentration levels at 1 h and should be avoided in patients with bleeding disorders, hypotension, arrhythmias, and hormone-sensitive cancers (breast, ovarian, or prostate). Zaprinast is an inhibitor of PDE5, PDE6, PDE9, and PDE11. In the past, it has been used for the treatment of PAH and inhibition of malaria parasites. Zaprinast activates the G-protein coupled receptor, GPR35, that plays a crucial role in cardiovascular disease, pain, regulation of inflammation, hypertension, diabetes, and irritable bowel disease [117,118]. The main characteristics of PDE5Is are summarized in Table 7 [34,112,113,119,120,121,122,123,124,125].
Table 7.
Main characteristics of phosphodiesterase 5 inhibitors.
| PDE5i | FDA Approved | Launch Date | Pharmacokinetics | Recommended Dosage | Indications | Side Effects | Contraindications | Emerging and Other Off-Label Therapeutic Applications |
|---|---|---|---|---|---|---|---|---|
| Sildenafil | Yes | 1998 | Cmax = 560 µg/L Tmax = 0.8–1 h T1/2 = 2.6–3.7 h Affected by heavy/fatty meals |
ED: 25–100 mg OD PAH: 5–20 mg TDS |
ED PAH |
Headache: 12.8% Flushing: 10.4% Dyspepsia: 4.6% Nasal congestion: 1.1% Dizziness: 1.2% Abnormal vision: 1.9% |
Absolute:
Relative:
|
|
| Tadalafil | Yes | 2003 | Cmax = 378 µg/L Tmax = 2 h T1/2 = 17.5 h Not affected by heavy/fatty meals |
ED: 10-20 mg on demand ED: 5 mg OD LUTS: 5 mg OD PAH: 40 mg |
ED PAH LUTS |
Headache: 14.5% Flushing: 4.1% Dyspepsia: 12.3% Nasal congestion: 4.3% Dizziness: 2.3% Back pain: 6.5% Myalgia: 5.7% |
||
| Vardenafil | Yes | 2003 | Cmax = 18.7 µg/L Tmax = 0.9 h T1/2 = 3.9 h Affected by heavy/fatty meals |
ED: 5–20 mg on demand |
ED | Headache: 16% Flushing: 12% Dyspepsia: 4% Nasal congestion: 10% Dizziness: 2% Abnormal vision: < 2% |
||
| Avanafil | Yes | 2013 | Cmax = 5.2 µg/L Tmax = 0.5–0.75 h T1/2 = 6–17 h Affected by heavy/fatty meals |
ED: 50–200 mg on demand |
ED | Headache: 9.3% Flushing: 3.7% Dyspepsia: uncommon Nasal congestion 1.9% Dizziness: 0.6% Back pain: < 2% Myalgia: < 2% |
||
| Udenafil | No | 2005 | Cmax = 1137 µg/L Tmax = 0.76 h T1/2 = 9.88 h |
ED: 100 mg on demand |
ED | Headache: 2–9% Flushing: 11–23% Dyspepsia: uncommon Nasal congestion: 4–7% Red eye: 4–7% Chest discomfort: 0–5% |
||
| Lodenafil | No | 2007 | Cmax = 157 µg/L Tmax = 1.2 h T1/2 = 2.4 h |
ED: 80 mg on demand |
ED | Headache: 15–22% Flushing: 5–6% Dyspepsia: 5–22% Nasal congestion: 5–11% Abnormal vision: 5–6% |
||
| Mirodenafil | No | 2011 | Cmax = 2989 µg/L Tmax = 1.4 h T1/2 = 2.5 h |
ED: 80 mg on demand |
ED | Headache: 8–11% Flushing: 10–16% Dyspepsia: 3% Red eye: 3–4% Chest discomfort: 0–3% |
||
| Benzamidenafil | No | - | ID | ID | ID | ID | ID | ID |
| Dasantafil | No | - | ID | ID | ID | ID | ID | ID |
| Icariin | No | - | ID | ID | ID | ID | ID | ID |
| Zaprinast | No | - | ID | ID | ID | ID | ID | ID |
Abbreviations: AKI, acute kidney injury; BP, blood pressure; Cmax, serum maximum concentration; CHF, chronic heart failure; CKD, chronic kidney disease; CVD, cardiovascular disease; ED, erectile dysfunction; ID, insufficient data; NO, nitric oxide; NYHA, New York Heart Association; OD, once daily; PAH, pulmonary arterial hypertension; PDE5I, phosphodiesterase 5 inhibitor; Tmax, transport maximum.
PDE5Is interfere selectively with cGMP hydrolysis by PDE5, increasing intracellular cGMP, which results in smooth muscle relaxation/raised arterial blood flow improving penile erection. PDE5 belongs to a superfamily of enzymes that convert intracellular cAMP/cGMP into the consonant nucleotides. It is a cytosolic protein with three isoforms expressed in various organs apart from the penis (corpora cavernosa), including kidney (vessels, glomeruli, inner medullary collecting ducts, and cortical tubules) that specifically degrades cGMP [66]. In particular, PDE5A1 and PDE5A2 are widely expressed in tubular epithelial cells of the renal proximal tubule and medullary collecting duct, as well as in vascular smooth muscle cells, platelets, brain, and lung, while PDE5A3 is only expressed in vascular smooth muscle cells [126].
Cyclic nucleotide signal transduction pathways represent an emerging research field in kidney disease, with selective PDE5 inhibition attracting ongoing interest nowadays [127]. Current evidence supports the notion that regulation of the cGMP -dependent protein kinase 1-PDE signaling pathway may be reno-protective and that its regulation might provide novel, therapeutic strategies for chronic kidney disease with selective PDE5Is having shown potential in treating kidney fibrosis, while possessing antithrombotic and anticancer activity [128]. In this respect, PDE5Is represent a potential but still understudied/controversial option against various forms of AKI such as CIN [28], given that NO/cGMP are crucial mediators in renal vasculature and NO is an essential endogenous vasodilator for medullary oxygenation [33].
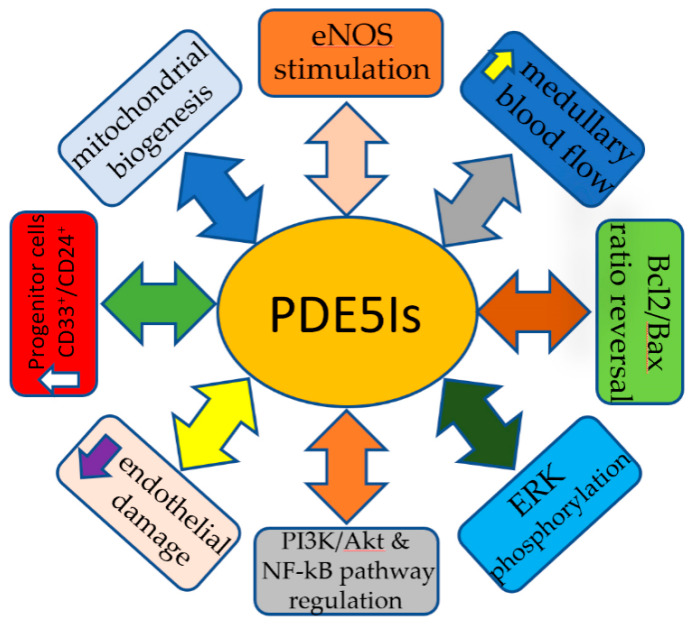
The mechanism of action of PDE5Is in the prevention and management of AKI is still not fully elucidated. Multiple mechanisms have been proposed to play a role in counteracting the cascade of changes caused by the renal injury. Stimulation of NO production through NOS, medullary blood flow improvement, protection against vascular endothelial damage, Bcl2/Bax ratio reversal, ERK phosphorylation, mitochondrial biogenesis activation, renal progenitor cell upregulation, and the regulation of multiple signaling pathways such as insulin/IGF1, T17/Treg, PI3K/Akt, and NF-kB [75] are the most well-described mechanisms through which PDE5Is offer protection. The increased ERK phosphorylation boosts NOS activity and subsequent rapid NO release [30]. The repairing process following any renal injury requires energy provided by the cellular mitochondria. Mitochondria are continuously regenerated but cellular injury such as sepsis and hypoxia induce rapid biogenesis. This process is mediated by a transcriptional co-activator, peroxisome proliferator-activated receptor γ co-activator 1a (PGC-1a). PGC-1a activates the nuclear respiratory factors 1 and 2, which eventually activate mitochondrial transcription factor A that is responsible for the transcription of mitochondrial DNA [23,67]. An alternative process that PDE5Is activate to promote recovery from renal injury is the renal progenitor cell stimulation. PDE5Is, more specifically icariin, upregulates HGF, WT-1, and BMP-7, which lead to an increased number of CD133+ and CD24+, cells that are capable of self-renew and also differentiate into podocytes or tubular cells [57,103]. In addition to the aforementioned actions, PDE5Is are likely to exert their protective effect through an alternative pathway. PDE5Is increase cGMP, which activates protein kinase G that opens mitochondrial KATP channels that induce depolarization of the mitochondrial inner membrane and Mg2+ release. The depolarized membrane results in reduced Ca2+ influx; therefore, suppressed cellular death and increased Mg2+ concentration reduces ROS and lessens p38 MAPK activation, which is responsible for apoptosis [30,88,129]. The most common reno-protective mechanisms of PDE5Is are summarized in Figure 3.
Figure 3.
Reno-protective mechanisms of PDE5Is.
To the best of our knowledge, this is the first review that attempts in a systematic way to define the reno-protective potential of PDE5Is in the various forms of AKI. Based on our results, it appears that sildenafil is the most widely PDE5I studied in AKI among the 11 natural/synthetic agents currently available (avanafil, benzamidenafil, dasantafil, icariin, iodenafil, mirodenafil, sildenafil, tadalafil, udenafil, vardenafil, and zaprinast).
The reno-protective effects of PDE5Is have been evaluated in four human studies to date (preclinical studies using human cells: n = 2 [24,38]; clinical studies: n = 2 [17,39]) (Table 1). In one study, human umbilical cord mesenchymal stem cells (huMSC), which have a high self-renewal/multi-directional differentiation potential, were treated with icariin and administered in an animal model of renal injury induced by adenine [38]. Blood urea nitrogen/sCr analysis showed amelioration of functional parameters. Icariin-treated huMSC increased the number of cells in injured renal tissues, reduced fibrosis, oxidative damage, inflammatory responses, and promoted expression of growth factors protecting injured renal tissue. In another study, cisplatin was added to human embryonic kidney (HEK)-293 renal cell cultures pre-treated with icariin [24]. The authors concluded that icariin prevents cisplatin-induced HEK-293 cell injury by inhibiting oxidative stress, inflammatory response, and cellular apoptosis partly via regulating nuclear factor kappa-like chain-enhancer of activated B cells (NF-κB) and PI3K/Akt signaling pathways. In a non-randomized clinical trial, 49 patients with renal tumors were submitted to open nephron-sparing surgery after renal artery clamping [17]. Twenty-two patients were pre-treated with tadalafil one day pre- and two days post-operatively and 27 patients underwent the same surgery without receiving tadalafil. Renal artery clamping induced kidney dysfunction reflected by increases in urinary NGAL and KIM-1 (two novel biomarkers for AKI) in all participants. Tadalafil reduced the urinary excretion of KIM-1, but not of NGAL. The incidence of AKI was comparable between groups but sCr elevation was significantly attenuated in the tadalafil-treated group compared to controls. It was concluded that tadalafil exerts reno-protective effects in AKI following nephron-sparing surgery. In a randomized placebo-controlled trial, 40 patients were submitted to robot-assisted partial nephrectomy after hilar clamping. The reno-protective effect of a single 100 mg oral dose of sildenafil immediately prior to clamping was evaluated [39]. GFR was similarly decreased between arms during the immediate postoperative period and at an intermediate-term follow-up of six months; the reno-protective effect of sildenafil was not evident in this study (neutral effect).
All animal studies investigating the potential reno-protective effect of sildenafil (n = 41) manifested a beneficial effect, irrespectively of the mechanism of AKI; almost all parameters evaluated (biochemical or morphological) were reported to improve (Table 2 and Table A1). Similarly, all animal studies investigating the potential reno-protective effect of tadalafil (n = 19) revealed beneficial outcomes (attenuated histopathological changes/improved biochemical profile; Table 3 and Table A2). Two studies provided comparative results for sildenafil and tadalafil, demonstrating the superiority of the former against tubular cell apoptosis, oxidative status, lipid peroxidation and NOS alterations [45,49]. Unique proteins, cells, and genes have been utilized to investigate the aftermath following icariin’s administration as a reno-protective agent, such as connective tissue growth factor, TUNEL positive cells, nephrin, α-smooth muscle actin, E-cadherin, LY6G, F4/80, NLRP3, NF-κΒ, etc. All available animal studies evaluating icariin (n = 8) showed a beneficial effect (oxidative injury reversal, obliteration of renal function impairment, and improvement of renal hemodynamics/NO sensitivity; Table 4 and Table A3). Similar to sildenafil/tadalafil, icariin suspends the inflammatory response initiation as well as the alteration of the cellular phase and preserves renal morphology. Finally, vardenafil, zaprinast, and udenafil have been investigated in a limited number of studies (n = 7, n = 4, and n = 2, respectively), almost all of which show antioxidant, anti-inflammatory, and reno-protective effects (Table 5, Table 6, Table A4, and Table A5). In one study, vardenafil was compared to sildenafil and tadalafil in an animal model of partial unilateral ureteric obstruction, reporting that all agents were beneficial with sildenafil showing best results [45]. Only one study failed to demonstrate any beneficial effect from zaprinast pre-treatment in an animal model of nephrectomy and concomitant contra-lateral renal hilar occlusion [40]. Even though data are still limited, especially in humans with inconclusive or negative results of only two clinically relevant studies available at present, the results of animal studies are promising and have already fueled clinical research, which is on-going with results expected to come out in the near future [122]. Nevertheless, the potential reno-protective capacity of PDE5Is in AKI warrants further investigation in clinical trials.
5. Conclusions
PDE5Is appear to be beneficial in AKI of various etiologies by improving renal functional/histopathological alternations through various mechanisms, such as by affecting regional hemodynamics, cell expression, and mitochondrial response to oxidative stress and inflammation. The reno-protective action of PDE5Is was evident in the vast majority of the studies, independently of the AKI type and the agent applied. Even though data are still limited, especially in humans with inconclusive or negative results of only two clinically relevant studies available at present, the results of animal studies are promising. The potential reno-protective capacity of PDE5Is in AKI warrants further investigation in clinical trials.
Acknowledgments
The authors would like to thank D. Pantartzi, Scientific Secretary of the Clinical Trial Office of the Department of Urology, University of Crete, Medical School, Heraklion, Crete, Greece, for the administrative and technical support.
Abbreviations
ADQI: Acute Dialysis Quality Initiative; AKI, acute kidney injury; AKIN, Acute Kidney Injury Network; Anti-dsDNA, antibody to double stranded DNA; Bax, proapoptotic protein; Bcl-2, antiapoptotic gene; BP, blood pressure; bFGF, basic fibroblast growth factor; Cmax, serum maximum concentration; BMP-7, bone morphogenetic protein-7; cAMP, cyclic adenosine monophosphate; cGMP, cyclic guanosine monophosphate; CHF, chronic heart failure; CIN, contrast-induced nephropathy; CKD, chronic kidney disease; CM, contrast media; COX, cyclo-oxygenase; CVD, cardiovascular disease; CYP3A4, cytochrome P450 3A4; DNA, deoxyribonucleic acid; DOCA, deoxycorticosterone acetate; ED, erectile dysfunction; EMA, European Medicines Agency; eNOS, endothelial nitric oxide synthase; ERK, extracellular signal-regulated kinases; FDA, Food and Drug Administration; GTP, guanosine-5′-triphosphate; GC, guanylyl cyclase; GFR, glomerular filtration rate; GSH, glutathione; HEK, human embryonic kidney cells; HGF, hepatocyte growth factor; HIF-2a, hypoxia induced factor 2a; HO-1, heme oxygenase 1; HSP-70, Heat shock protein 70, huMSCs, human umbilical cord mesenchymal stem cells; ICAM-1, intercellular adhesion molecule-1; ID, insufficient data; IgG, immunoglobulin G; IL, interleukin; iNOS, inducible nitric oxide synthase; IR, ischemia reperfusion; KDIGO, Kidney Disease, Improving Global Outcomes; KIM-1, kidney injury molecule-1; KO, knockout; LNAME, N(ω)-nitro-L-arginine methyl ester; MeSH, medical subject headings; Mac387, macrophage antibody; MDA, malondialdehyde; NAC, N-acetylcysteine; NF-Kb, nuclear factor kappa light-chain-enhancer of activated B cells; NGAL, neutrophil gelatinase-associated lipocalin; NO, nitric oxide; NOS, nitric oxide synthase; Nrf-2, nuclear factor erythroid 2-related factor-2; NQO1, NADPH quinine oxidoreductase 1; NYHA, New York Heart Association; OD, once daily; PAH, pulmonary arterial hypertension; PDE5, phosphodiesterase 5; PDE5Is, phosphodiesterase 5 inhibitors; PGC-1a, peroxisome proliferator-activated receptor γ coactivator 1a; PKG, protein kinase G; PRISMA, preferred reporting items for systematic reviews and meta-analyses; RIFLE, risk, injury, failure, loss and end-stage kidney disease; ROS, reactive oxygen species; SBP, systolic blood pressure; sCr, serum creatinine; Tfam, mitochondrial transcription factor; TGF-β1, transforming growth factor beta; Tmax, transport maximum; TSP, thrombospondin; TUNEL, terminal deoxynucleotidyl transferase dUTP nick end labeling; TNF-a, tumor necrosis factor -a; VEGF, vascular endothelial growth factor; WT-1, Wilms’ tumor 1 gene.
Appendix A. Literature Search Strategy
Database: Ovid Medline Epub Ahead of Print, In-Process & Other Non-Indexed Citations, Ovid MEDLINE(R) Daily and Ovid MEDLINE(R) 1946 to November 2019.
Search Strategy:
((((((((((((PDE5i[Title/Abstract]) OR avanafil[Title/Abstract]) OR benzamidenafil[Title/Abstract]) OR dasantafil[Title/Abstract]) OR icariin[Title/Abstract]) OR lodenafil[Title/Abstract]) OR mirodenafil[Title/Abstract]) OR sildenafil[Title/Abstract]) OR tadalafil[Title/Abstract]) OR vardenafil[Title/Abstract]) OR udenafil[Title/Abstract]) OR zaprinast[Title/Abstract]) AND (((((((((((((((((((((((((renal[Title/Abstract]) OR kidney[Title/Abstract]) OR nephrotoxicity[Title/Abstract]) OR contrast[Title/Abstract]) OR CIN[Title/Abstract]) OR AKI[Title/Abstract]) OR nephrotoxic[Title/Abstract]) OR cisplatin[Title/Abstract]) OR aminoglycoside[Title/Abstract]) OR trauma[Title/Abstract]) OR acute kidney injury[Title/Abstract]) OR nsaids[Title/Abstract]) OR non steroidal[Title/Abstract]) OR shock[Title/Abstract]) OR sepsis[Title/Abstract]) OR hypoperfusion[Title/Abstract]) OR hypovolaemia[Title/Abstract]) OR renal artery stenosis[Title/Abstract]) OR obstruction[Title/Abstract]) OR acute tubular necrosis[Title/Abstract]) OR glomerulonephritis[Title/Abstract]) OR nephritis[Title/Abstract]) OR renal failure[Title/Abstract]) OR cyclosporine[Title/Abstract]) OR adenine[Title/Abstract]).
Appendix B. Animal Studies in the AKI-CKD Transition Spectrum (Focusing on Renal Function and/or Structure Alterations for up to Three Months, Not Fulfilling the KDIGO Definition for CKD [6])
Table A1.
Animal studies evaluating the potential reno-protective effects of sildenafil.
| Reference /Country/Year |
Studied Animal | Model | PDE5I Route | Timing | Sample | Renal Injury Effects | PDE5I Renal Effects | Outcome |
|---|---|---|---|---|---|---|---|---|
| [81]/ Venezuela/2005 |
Male Sprague Dawley rats | 5/6 nephrectomy | Sildenafil Orally 2.5 mg/kg/day Either immediately after nephrectomy for 8 weeks Or 4 weeks after nephrectomy for 4 weeks |
POST | 8 weeks |
↑sCr, ↑SBP, ↑proteinuria ↓urinary NOX, ↓cGMP Glomerulosclerosis Tubulo-interstitial damage Macrophage accumulation Increased number of apoptotic cells |
↓sCr, ↓SBP, ↓proteinuria ↑ urinary NOX, ↑cGMP All changes improved especially if PDE5i was given early |
POS |
| [82]/ Spain/2007 |
Laboratory large-white pigs | Right single nephrectomy after 45 min of vascular clamping + Auto-transplantation + Left nephrectomy |
Sildenafil Orally 100 mg, 2 h pre-op |
PRE | 0/15/30/45/60 min after unclamping |
↓RVF, ↑RVR, ↓NO Minimal differences in tubular and endothelial structure |
↑↓RVF, ↓RVR, ↑NO Minimal differences in tubular and endothelial structure |
POS |
| [83]/ Egypt/2008 |
Adult male Wistar albino rats | L-NAME Orally 50 mg/kg/day for 4 weeks |
Sildenafil Orally 5 mg/kg/day 2 weeks after L-NAME for 2 weeks |
POST | At 4 weeks |
↓NO, ↓cGMP Glomerular collapse/mesangial matrix expansion with minimal cellular proliferation |
↑NO, ↑cGMP Improvement in histological alterations |
POS |
| [84]/ Spain/2009 |
Laboratory minipigs | Right single nephrectomy after 45 min of vascular clamping +Auto-transplantation +Left nephrectomy |
Sildenafil Orally 100 mg, 1.5 h pre-op |
PRE | 0/15/30/45/60 min after unclamping |
↓RVF, ↓RVR |
↑RVF, ↓ RVR, ↑NO Reduced tubular edema, Improved endothelial cell integrity and mitochondrial ultrastructure |
POS |
| [85]/ Korea/2009 |
Male Sprague Dawley | Streptozotocin Single intravenous dose 60 mg/kg |
Sildenafil Orally 3 mg/kg/day For 8 weeks |
POST | At 8 weeks | ↑glucose, ↑urine output ↑urine 8-OH dG ↑urine albumin ↑Kidney/BW ratio, ↑iNOS ↑Nitrotyrosine, ↑MCP-1 ↑ED-1 |
↑glucose, ↓urine output ↓urine 8-OH dG ↓urine albumin ↓Kidney/BW ratio, ↓iNOS ↓Nitrotyrosine, ↓MCP-1, ↓ED-1 |
POS |
| [86]/ Korea/2012 |
Male Sprague Dawley rats | Unilateral Nephrectomy and 1 week later DOCA strip 200 mg/kg implantation | Sildenafil Orally 50 mg/kg/day 2 weeks after DOCA for 2 weeks |
POST | At 4 weeks | ↑mortality, ↑SBP, ↑ Kidney weight ↓CrCl, ↑sCr, ↑FeNa, ↑ACR ↑ED-1, ↑BAX, ↓Bcl2, ↑aSMA, ↑TGF-b1 ↑TUNEL +ve, ↑fibronectin ↑mRNA TGF-β1/MCP-1 ↑ICAM 1t Tubular casts/Tubular obstruction/Vessel dilatation/Glomerulosclerosis/interstitial expansion |
↓mortality, ⇔ SBP, ↓Kindey weight ↑CrCl, ↓sCr, ↓FeNa, ↓ACR ↓aSMA ↓ED-1, ↓BAX, ↑Bcl2, ↓TGF-b1 ↓TUNEL +ve, ↓fibronectin ↓mRNA TGF-β1/MCP-1/↓ mRNA ICAM1 Reversed all renal injuries |
POS |
| [87]/ Venezuela/2012 |
Male Wistar rats | 5/6 nephrectomy | Sildenafil Orally 5 mg/kg/day for 60 days 24 h after nephrectomy |
POST | Every 2 weeks | ↑Kidney hypertrophy ↑Proteinuria, ↓NO2/NO3 ↓GMP (urine) ↑Nitrotyrosine |
↓Kidney hypertrophy ↓Proteinuria, ↑NO2/NO3 ↑cGMP (urine) ↓Nitrotyrosine |
POS |
| [19]/ Egypt/2013 |
Male Wistar rats | Cyclosporine A Subcutaneously 20 mg/kg/day 21 days |
Sildenafil Orally 5 mg/kg/day 21 days |
POST | At 21st day–urine sample/blood sample/renal tissue excision | ↑BUN, ↑sCr, ↑MDA levels ↑Urine Albumin/Cr ↓GSH/NO/catalase activity ↑iNOS, TNF-a, Caspase 3 activity Tubular degeneration and dilation and necrosis/Glomerulat damage/Congestion Dilated Bowman’s space/Hemorrhage |
↓BUN, ↓sCr, ↓MDA levels ↓Urine Albumin/Cr, ↑eNOS ↑GSH/NO/catalase activity ↓iNOS, TNF-a, Caspase 3 activity Improved all histological changes |
POS |
| [88]/ United Kingdom/2014 |
Porcine kidneys | 20 min warm ischemia followed by 2 or 18 h of cold storage | Sildenafil Intravenously 1.4 mg/kg 10 min prior to injury and 20min after reperfusion |
PRE and POST | Samples during reperfusion | ↓RBF, ↑intrarenal resistance ↓Urine cGMP ↑ sCr Steady increase of K+ ↑Tubular injury No difference in groups Tubular dilatation and debris and interstitial edema/Ischemic changes |
↑RBF, ↓intrarenal resistance ↑Urine cGMP, ↓sCr No significant difference in K+ No effect on tubular injury regarding GAL/Endothelin1 Slight improvement of histological |
POS |
| [89]/ Brazil/2014 [90]/ Brazil/2014 |
C57BL/6 mice | Left renal artery clamping for 2 weeks | Sildenafil Orally 40 mg/kg/day 2 weeks post op for 2 weeks |
POST | 4 weeks | Left kidney atrophy (clipped) Right kidney hypertrophy ↓BW, ↑SBP, ↑HR ↑Intrarenal angiotensin I/II ⇔Plasma angiotensin I/II/1-7 ↓NO, ↑ONOO-, ↑O2- Impaired vasodilation response to Ach |
↓Left kidney atrophy ↓Right kidney hypertrophy Normal BW, ↓SBP, ↓HR ↓Intrarenal angiotensin I/II ↑Plasma angiotensin 1-7 ↑NO, ↓ONOO-, ↓O2- Normal vasodilation response to Ach |
POS |
| [91]/ Egypt/2016 |
White albino male rats | Streptozocin Single intraperitoneal dose 55 mg/kg |
Sildenafil Orally 3 mg/kg/day For 8 weeks after Diabetic nephropathy |
POST | 8 weeks |
↓SOD, ↑TGF-β1, ↓NO, ↑sCr ↑BUN, ↑proteinuria ↑Kidney IL-1β ↑Advanced glycation end products (AGEPs) |
↑SOD, ↓TGF-β1, ↑NO, ↓sCr ↓BUN, ↓proteinuria ↓Kidney IL-1β ↓Advanced glycation end products (AGEPs) |
POS |
| [92]/ India/2016 |
Sprague-Dawley rats | Streptozocin Single intraperitoneal dose 60 mg/kg |
Sildenafil Orally 2.5 mg/kg/day for 6 weeks after 28 days |
POST | At 28th and 70th day | ↑sCr, ↑BUN, ↓CrCl ↑Total protein excretion ↑albumin (urine) Bowman’s capsule thickening, glomerular sclerosis |
↓sCr, ↓BUN, ↑CrCl ↓Total protein excretion ↓albumin (urine) Histopathology improvement |
POS |
| [93]/ Italy/2017 |
Male CD-1 mice | Streptozotocin Single intraperitoneal dose 150 mg/kg |
Sildenafil Intraperitoneally 1.6 mg/kg 3 days after STZ, for 4 weeks |
POST | ↑Glucose (urine), ↑MAP, ↓GFR ↑urinary ACR, ↑NGAL, ↑RRI ↓Renal volume, ↓BMP7 ↑suPAR, ↑Vascular leakage ↑FITC-dextran extravasation Reduced glomerular diameter/focal and segmental hyperplasia with diffuse mesangial proliferation/increased mesangial matrix deposition/acute tubular degeneration/eosinophilia/proximal tubule basal membrane thickening |
↓Urine glucose, ↓MAP, ↑GFR ↓urinary ACR, ↓NGAL, ↓RRI ↑Renal volume, ↑BMP7, ↓miR-22 ↓suPAR, ↓Vascular leakage ↓FITC-dextran extravasation Reduced mesangial matrix deposition |
POS | |
| [94]/ Egypt/2017 |
Adult male Sprague-Dawley rats | Doxorubicin Intraperitoneally 3.5 mg/kg Twice weekly for 3 weeks |
Sildenafil Orally 5 mg/kg/day for 21 days |
POST | ↑Urea, ↑sCr, ↑uric acid ↑MDA, ↓GSH, ↑TNF-a ↑caspase-3 Eosinophilic casts, tubule degeneration, vacuolization, endothelial cell edema |
↓Urea, ↓sCr, ↓uric acid ↓MDA, ↑GSH, ↓TNF-a ↓caspase-3 Histological improvement |
POS | |
| [95]/ South Africa/2017 |
Nulliparous pregnant female Sprague-Dawley rats | L-NAME Orally 0.3 g/L (drinking water) 4-8 days for EOPE + 8-14 days for LOPE |
Sildenafil Orally 10 mg/kg 4-8 days for EOPE 8-14 days for LOPE |
POST | Gestational Day 19 | ↑BP, ↑Urine excretion ↑Urinary nephrin mRNA ↑Podocin (urine), ↑sFlt-1(mRNA) ↓VEGF (mRNA), ↓PIGF Glomerular and tubular damage and mononuclear cell infiltration |
↓BP ↓Urinary nephrin mRNA ↓Podocin (urine) ↓sFlt-1 (mRNA) ↑VEGF (mRNA), ↑PIGF levels Attenuated histopathological changes |
POS |
| [96]/ Netherlands/2017 |
Rats | Adriamycin Or Streptozocin |
Sildenafil Orally 5 mg/kg/day for 6 weeks |
POST | Immortalized mouse podocytes + Mouse kidney cortex |
↑TRPC6 expression ↓Nephrin, ↑Glomerular desmin ↑Urinary albumin ↑Glomerular lesions |
↓TRPC6 expression, ↓Ca2+ influx ↑Nephrin ↓Glomerular desmin ↓Urinary albumin |
POS |
| [97]/ Oman/2018 |
Male Sprague-Dawley rats |
Adenine (0.25% w/w) orally Daily for 5 weeks |
Sildenafil Orally (0.1, 0.5 or 2.5 mg/kg) Daily for 5 weeks (alone or concomitantly with adenine) |
SIM | At Day 5 |
↑BUN, sCr, uric acid, P, NGAL, ↑total NO, IS, Caspase 3 +ve cells ↑Albumin, NAG activity ↓Osmolality, CrCl in urine ↓CAT, glutathione reductase, SOD ↓TAC, ↑MAPK ↑Fibrosis ↑Adiponectin, cystatin-C, TNF-a ↓Sclerostin, ↑MDA Tubular necrosis, tubular dilatation, tubular cast formation, necrotic nuclei, tubular cells apoptosis, cellular shedding, mononuclear infiltration |
↓BUN, sCr, uric acid, ↓P, NGAL, ↓total NO, IS, ↓Caspase 3 +ve cells ↓Albumin, ↓ NAG activity ↑Osmolality, ↓CrCl in urine ↑CAT, SOD ↓glutathione reductase, ↑TAC, ↓MAPK, ↓Fibrosis ↓Adiponectin, cystatin-C, TNF-a ↑Sclerostin (not 0.1 mg/kg) ↓MDA Improved tubular necrosis, tubular dilatation, tubular cast formation, mononuclear infiltration |
POS |
| [98]/ Egypt/2018 |
Male albino rats Sprague-Dawley | Streptozotocin Single intraperitoneal dose 45 mg/kg |
Sildenafil Orally: 3 mg/kg/Day 3 weeks after STZ for 15 days |
POST | Day 16 after initiation of Sildenafil | ↑sCr, ↑BUN ↑fasting and post prandial glucose ↓insulin levels ↑insulin resistance ↑MDA, ↓GSH, ↓CAT, ↓GPx, ↓SOD, ↓TAC |
↓sCr, ↓BUN ↓fasting and post prandial glucose ↑insulin levels ↓insulin resistance (insignificant) ↓MDA, ↑GSH, ↑CAT, ↑GPx, ↑SOD, ↑TAC |
POS |
Abbreviations: AKI, acute kidney injury; ACR, albumin-creatinine ratio; aSMA, α-smooth muscle actin; Bax, proapoptotic protein; Bcl-2, antiapoptotic gene; BP, blood pressure; BUN, blood urea nitrogen; BW, body weight; Ca2+, calcium; CAT, catalase; cGMP, cyclic guanosine monophosphate; CrCl, creatinine clearance, ED-1, monoclonal antibody, eNOS, endothelial NOS, FeNa, fractional excretion of sodium, FITC, fluorescein isothiocyanate, GFR, glomerular filtration rate; GPx, glutathione peroxidase; GSH, glutathione; HR, heart rate; ICAM-1, intercellular adhesion molecule 1; IL, interleukin; IS, indoxyl sulfate; iNOS, inducible NOS; K, potassium; MAP, mean arterial pressure; MAPK, mitogen-activated protein kinase; MCP-1, monocyte chemoattractant protein 1; MDA, malondialdehyde; Na, sodium; NAG, N-acetyl-beta-D-glucosaminidase; NGAL, neutrophil gelatinase-associated lipocalin; NO, nitric oxide; NOX, NADPH oxidase; P, phosphorus; PDE5I, phosphodiesterase 5 inhibitor; PIGF, placenta growth factor; RRI, renal resistive index; RVF, renal vascular flow; RVR, renal vascular resistance; sCr, serum creatinine; sFlt1, soluble fms-like tyrosine kinase-1; SOD, superoxide dismutase; SBP, systolic blood pressure; suPAR, soluble urokinase-type plasminogen activator receptor; TAC, total antioxidant capacity; TGF-β1, transforming growth factor beta 1; TRPC6, transient receptor potential cation channel 6; TNF-a, tumor necrosis factor a; TUNEL, terminal deoxynucleotidyl transferase dUTP nick end labeling; VEGF, vascular endothelia growth factor; 8-OH dG, 8-hydroxydeoxyguanosine; ↓, reduced; ↑, increased.
Table A2.
Animal studies evaluating the potential reno-protective effects of tadalafil.
| Reference/ Country/Year |
Studied Animal | Model | PDE5I Route | Timing | Sample | Renal Injury Effects | PDE5I Renal Effects | Outcome |
|---|---|---|---|---|---|---|---|---|
| [99]/ Turkey/2013 |
Male Sprague Dawley rats |
SWL model | Tadalafil Orally: 1 mg/kg Single dose 150 min prior to SWL |
PRE | Nephrectomy at Day 1/3/7 | Loss of micro-villi Tubular degeneration and necrosis Interstitial edema and fibrosis ↑ HSP-70 |
Reduced all histological damage ↓HSP-70 |
POS |
| [100]/ Turkey/2017 |
Male Sprague Dawley | SWL model | Tadalafil Orally: 1 mg/kg 60 min prior to SWL |
PRE | Bilateral nephrectomy 7 days post SWL | Renal tubular damage Peritubular fibrosis/Loss of microvilli ↑HSP-70 |
Significantly less tissue damage ⇔HSP-70 (glomerular) ↓HSP-70 (medullar/cortical) |
POS |
Abbreviation: AKI, acute kidney injury; HSP-70, heat shock protein 70; PDE5I, phosphodiesterase 5 inhibitor; SWL, shock wave lithotripsy; ↓, reduced; ↑, increased ⇔, no change.
Table A3.
Animal studies evaluating the potential reno-protective effects of icariin.
| Reference/ Country/Year |
Studied Animal | Model | PDE5I Route | Timing | Sample | Renal Injury Effects | PDE5I Renal Effects | Outcome |
|---|---|---|---|---|---|---|---|---|
| [101]/ China/2011 |
Male Sprague–Dawley rats | Streptozotocin Single Dose Intravenously 40 mg/kg |
Icariin Orally 80 mg/kg For 8 weeks From 5th to 20th week post streptozotocin |
POST | Day 7 and Week 13 |
↑sCr, ↑BUN, ↑Glucose, ↑MDA, ↑Hyp, ↓SOD, ↑Collagen IV, ↑TGF-β1 Glomerular Hypertrophy Expansion of mesangial area and ECM |
↓sCr, ↓BUN, ↓MDA, ↓Hyp ↑SOD, ↓Collagen IV, ↓TGF-β1 Inhibited these changes |
POS |
| [102]/ Chiana/2014 |
Male Sprague-Dawley rats | 5/6 right nephrectomy model |
Icariin Orally 20 + 40 mg/kg/day 1 week after AKI for 12 weeks |
POST | 24 h before AKI and at Week 12 |
↑BUN,↑sCr, ↑ urinary protein ↑Apoptotic rate, ↑Bcl-2, ↑Bax ↓G0/G1 phase cells ↑S phase cells |
↓BUN, ↓sCr, ↓urinary protein ↓Apoptotic rate, ↓Bcl-2, ↓Bax ⇔G0/G1 phase cells, ↑G2/M phase ↓ S phase cells |
POS |
| [103]/ China/2015 |
Male Sprague Dawley rats | 1st stage: Partial nephrectomy 2nd stage: Right renal ligation |
Icariin Orally 40 mg/kg/day 8 weeks |
POST | At 8 weeks | ↑BUN, ↑sCr, ↑uric acid Mesangial expansion/Edema Basement membrane thickening and capillary compression/occlusion. Glomerular sclerosis/fibrosis Inflammatory cell infiltration |
↓BUN, ↓sCr, ↓uric acid ↑Renal progenitor cell proliferation ↓TGF-β1 Significantly improved glomerular lesions and blunted rest of the changes |
POS |
| [22]/ China/2017 |
Female Wistar rats | Pregnancy induced hypertension L-NAME 0.5 g/L from Day 12 of gestation |
Icariin Orally 10/50/100 mg/kg 18 days of gestation |
POST | BP: Days 1 and 18 Kidney tissue: Day 18 |
↑SBP (Day 18), ↑ BUN, ↑sCr ↑Proteinuria, ↓Pup weight ↓Nephrinm, ↑Ang II, ↑AGT Mesangial expansion Basement membrane thickening |
↓SBP (high dose), ↓BUN, ↓sCr ↓Proteinuria (medium/high dose) No difference in pup weight ↑Nephrin, ↓Ang II, ↓AGT Markdely reduced severity of lesions |
POS |
| [104]/ China/2018 |
MRL/lpr mice | K/O mice | Icariin Orally 10 mg/kg/day 8 weeks |
POST | Every 2 weeks |
↑Urine protein,↑IgG deposit ↑sCr, ↑BUN, ↑TNF-a ↑Serum anti-dsDNA ↑Translocation and phosphorylation of NF-kBp65 ↑F4/80, ↑NLRP3, ↑caspase 1p20 Increased glomerular proliferation/sclerosis/peripheral inflammation |
↓Urine protein, ↓IgG deposit ↓sCr, ↓BUN, ↓TNF-a ↓Serum anti-dsDNA ↓Translocation and phosphorylation of NF-kBp65 ↓F4/80, ↓NLRP3, ↓caspase 1p20 Improved all changes |
POS |
Abbreviations: AKI, acute kidney injury; Ang II, angiotensin II; Anti-dsDNA, antibody to double stranded DNA; AGT, angiotensinogen; Bax, proapoptotic protein; Bcl-2, antiapoptotic gene; BP, blood pressure; BUN, blood urea nitrogen; F4/80, macrophage marker; Hyp, hydroxyproline; IgG, immunoglobulin G; MDA, malondialdehyde; PDE5I, phosphodiesterase 5 inhibitor; SBP, systolic blood pressure; sCr, serum creatinine; SOD, superoxide dismutase; TGF-β1, transforming growth factor beta 1; TNF-a, tumor necrosis factor a; ↓, reduced; ↑, increased.
Table A4.
Animal studies evaluating the potential reno-protective effects of vardenafil.
| Reference/ Country/Year |
Studied Animal | Model | PDE5I Route | Timing | Sample | Renal Injury Effects | PDE5I Renal Effects | Outcome |
|---|---|---|---|---|---|---|---|---|
| [105]/ Germany/2008 |
Sprague Dawley rats | Mouse monoclonal anti-Thy 1 antibody ER-4 Single injection, 1 mg/kg |
Vardenafil Orally 20 mg/kg within 18 h and 10 mg/kg/day for 48 h |
PRE and POST | 24-h urine collection on Days 2 and 6 Blood sample: Day 6 |
↑PDE5-A, ⇔sCr ↑proteinuria Mesangial proliferation |
↑cGMP, ↓TSP-1, ⇔sCr ↓proliferation/cell number(glomerular) ↓collagen IV/fibronectin (glomerular) ↓TGF-β activation ⇔proteinuria |
POS |
| [106]/ China/2009 |
New Zealand Rabbits | Invagination of ureter in renal pelvis | Vardenafil Orally 0.3 mg/kg/day For 8 weeks post op |
POST | 8 weeks | Dilated renal pelvises Fibrotic PUJ ↑TGF-β1 ↓nNOS |
Dilated renal pelvises Less fibrotic PUJ ↓TGF-β1 ↑nNOS |
POS |
| [107]/ Hungary/2013 |
Sprague Dawley male rats | Streptozotocin Single intraperitoneal dose 60/mg/kg |
Vardenafil Orally 10 mg/kg/day for 8 weeks 72 h post STZ |
POST | 8 weeks after AKI | ↓cGMP, NCS elevated Urea levels Decreased body weight No difference in MAP ↑Urine protein/creatinine ratio ↑Fibronectin, ↑TGF-β1, ↑desmin, ↓nephrin, ↑Nitrotyrosine, ↑NOS Glomerular hypertrophy Mesangial expansion Adhesions to Bowman’s capsule Tubular dilatation and atrophy Mononuclear cell infiltration |
↑cGMP Developed kidney hypertrophy No difference in MAP ↓Urine protein/creatinine ratio ↓Fibronectin, ↓TGF-β1 ↓desmin, ↑nephrin No difference Attenuated all changes |
POS |
| [108]/ Turkey/2015 |
Male Swiss albino mice | Cyclosporine A 30 mg/kg Subcutaneously Daily for 28 days |
Vardenafil Orally 30 mg/kg/day For 28 days |
PRE | At 28 days |
↓Kidney weight ↑BUN, ↑sCr, ↑TOS levels ↓TAS levels,↓tissue NO ↓COX-1, ↓COX-2, ↓TGF-β1 ↓Pgp levels, ↓PDGF-A, ↓PDGF-C Histological changes: cortex/outer medulla |
No change in kidney weight ↓BUN, ↓sCr, ↓TOS levels ↑TAS levels, ↑tissue NO ↑COX-1, ↑COX-2, ⇔TGF-β1 ↑Pgp levels, ↑PDGF-A, ↑PDGF-C Normal histopathological appearances |
POS |
Abbreviations: AKI, acute kidney injury; BUN, blood urea nitrogen; cGMP, cyclic guanosine monophosphate; COX, cyclo-oxygenase; FeNa, fractional excretion of sodium; MAP, mean arterial pressure; NCS, not clinically significant; NO, nitric oxide, nNOS, neuronal NOS; PDE5I, phosphodiesterase 5 inhibitor; PDGF, platelet-derived growth factor; Pgp, P glycoprotein; PUJ, pelvic ureteric junction; sCr, serum creatinine; TAS, total antioxidant status; TGF-β1, transforming growth factor beta 1; TOS, total oxidant status; TSP-1, thrombospondin -1; ↓, reduced; ↑, increased ⇔, no change.
Table A5.
Animal studies evaluating the potential reno-protective effects of zaprinast and udenafil.
| Reference/ Country/Year |
Studied Animal | Model | PDE5I Route | Timing | Sample | Renal Injury Effects | PDE5I Renal Effects | Outcome |
|---|---|---|---|---|---|---|---|---|
| [109]/ Japan/1998 |
Mongrel dogs | Cut left renal nerves and electrostimulation of left renal bundle (distal end) | Zaprinast Intra-renal arterial infusion 10 or 100 μg/kg/min |
SIM | Simultaneously | ↓Urine flow, ↓UNaV, ↓FeNa ⇔RBF, ⇔GFR |
↑Urine flow, ↑UNaV, ↑FeNa ⇔ RBF, ⇔GFR, ↓RVR ↑Renal venous cGMP |
POS |
| [110]/ Korea/2010 |
10-week-old male Sprague-Dawley | Right nephrectomy + Left renal artery clamping for 45 min and Cyclosporine A 15 mg/kg subcutaneously |
Udenafil Orally: 10 mg/kg For 28 days after the procedure |
SIM and POST | On Day 28 blood samples and left nephrectomy | ↑BUN, ↑sCr, ↓eNOS, ⇔VEGF Decreased thickness of the proximal tubules and nuclei, vacuolization of the cytoplasm, altered cellular shape, fewer nuclei |
↓BUN, ↓sCr, ↑eNOS, ⇔VEGF ↓VEGF mRNA |
POS |
Abbreviations: AKI, acute kidney injury; BUN, blood urea nitrogen; cGMP, cyclic guanosine monophosphate; eNOS, endothelial NOS; FeNa, fractional excretion of sodium; GFR, glomerular filtration rate; PDE5I, phosphodiesterase 5 inhibitor; RBF, renal blood flow; RVR, renal vascular resistance; sCr, serum creatinine; UNaV, urinary sodium excretion; VEGF, vascular endothelia growth factor; ↓, reduced; ↑, increased ⇔, no change.
Author Contributions
Conceptualization, C.M. (Charalampos Mamoulakis); methodology, G.G., I.-E.Z., and C.M. (Charalampos Mamoulakis); validation, A.O.D., K.T., C.T., D.C., and A.T.; formal analysis, G.G. and I.-E.Z.; investigation, G.G. and I.-E.Z.; data curation, A.O.D., K.T., I.F., C.M. (Charalampos Mavridis), M.K., S.S., K.S., C.T., D.C., N.S., A.T., and C.M. (Charalampos Mamoulakis); writing—original draft preparation, G.G. and I.-E.Z.; writing—review and editing, A.O.D., K.T., I.F., C.M. (Charalampos Mamoulakis), M.K., S.S., K.S., C.T., D.C., N.S., A.T., and C.M. (Charalampos Mamoulakis); visualization, G.G., I.-E.Z., A.O.D., K.T., C.T., and D.C.; supervision, C.M. (Charalampos Mamoulakis); and funding acquisition, C.M. (Charalampos Mamoulakis). All authors have read and agreed to the published version of the manuscript.
Funding
The authors would like to thank the Special Research Account of the University of Crete for supporting the publication of this study (ELKE No 3550).
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Makris K., Spanou L. Acute Kidney Injury: Definition, Pathophysiology and Clinical Phenotypes. Clin. Biochem. Rev. 2016;37:85–98. [PMC free article] [PubMed] [Google Scholar]
- 2.Codorniu A., Lemasle L., Legrand M., Blet A., Mebazaa A., Gayat E. Methods used to assess the performance of biomarkers for the diagnosis of acute kidney injury: A systematic review and meta-analysis. Biomarkers. 2018;23:766–772. doi: 10.1080/1354750X.2018.1493616. [DOI] [PubMed] [Google Scholar]
- 3.ACR Committee on Drugs and Contrast Media . ACR Manual on Contrast Media Version 10.3. ACR; Reston, VA, USA: 2020. [Google Scholar]
- 4.Bellomo R., Ronco C., Kellum J.A., Mehta R.L., Palevsky P., Acute Dialysis Quality Initiative workgroup Acute renal failure—Definition, outcome measures, animal models, fluid therapy and information technology needs: The Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit. Care. 2004;8:R204. doi: 10.1186/cc2872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mehta R.L., Kellum J.A., Shah S.V., Molitoris B.A., Ronco C., Warnock D.G., Levin A., Acute Kidney Injury Network Acute Kidney Injury Network: Report of an initiative to improve outcomes in acute kidney injury. Crit. Care. 2007;11:R31. doi: 10.1186/cc5713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.KDIGO KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney Int. Suppl. 2012;2:1–138. doi: 10.1038/kisup.2012.1. [DOI] [Google Scholar]
- 7.Fiorentino M., Kellum J.A. Improving Translation from Preclinical Studies to Clinical Trials in Acute Kidney Injury. Nephron. 2018;140:81–85. doi: 10.1159/000489576. [DOI] [PubMed] [Google Scholar]
- 8.Zarjou A., Sanders P.W., Mehta R.L., Agarwal A. Enabling innovative translational research in acute kidney injury. Clin. Transl. Sci. 2012;5:93–101. doi: 10.1111/j.1752-8062.2011.00302.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Skrypnyk N.I., Siskind L.J., Faubel S., de Caestecker M.P. Bridging translation for acute kidney injury with better preclinical modeling of human disease. Am. J. Physiol. Renal. Physiol. 2016;310:F972–F984. doi: 10.1152/ajprenal.00552.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ortiz A., Sanchez-Nino M.D., Izquierdo M.C., Martin-Cleary C., Garcia-Bermejo L., Moreno J.A., Ruiz-Ortega M., Draibe J., Cruzado J.M., Garcia-Gonzalez M.A., et al. Translational value of animal models of kidney failure. Eur. J. Pharmacol. 2015;759:205–220. doi: 10.1016/j.ejphar.2015.03.026. [DOI] [PubMed] [Google Scholar]
- 11.Bao Y.W., Yuan Y., Chen J.H., Lin W.Q. Kidney disease models: Tools to identify mechanisms and potential therapeutic targets. Zool. Res. 2018;39:72–86. doi: 10.24272/j.issn.2095-8137.2017.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Singh A.P., Junemann A., Muthuraman A., Jaggi A.S., Singh N., Grover K., Dhawan R. Animal models of acute renal failure. Pharmacol. Rep. 2012;64:31–44. doi: 10.1016/S1734-1140(12)70728-4. [DOI] [PubMed] [Google Scholar]
- 13.Burmeister D.M., Gomez B.I., Dubick M.A. Molecular mechanisms of trauma-induced acute kidney injury: Inflammatory and metabolic insights from animal models. Biochim. Biophys. Acta Mol. Basis Dis. 2017;1863:2661–2671. doi: 10.1016/j.bbadis.2017.04.011. [DOI] [PubMed] [Google Scholar]
- 14.Iordache A.M., Docea A.O., Buga A.M., Zlatian O., Ciurea M.E., Rogoveanu O.C., Burada F., Sosoi S., Mitrut R., Mamoulakis C., et al. Sildenafil and tadalafil reduce the risk of contrast-induced nephropathy by modulating the oxidant/antioxidant balance in a murine model. Food Chem. Toxicol. 2020;135:111038. doi: 10.1016/j.fct.2019.111038. [DOI] [PubMed] [Google Scholar]
- 15.Sanz A.B., Sanchez-Nino M.D., Martin-Cleary C., Ortiz A., Ramos A.M. Progress in the development of animal models of acute kidney injury and its impact on drug discovery. Expert Opin. Drug Discov. 2013;8:879–895. doi: 10.1517/17460441.2013.793667. [DOI] [PubMed] [Google Scholar]
- 16.Sarica K., Yencilek F. Prevention of shockwave induced functional and morphological alterations: An overview. Arch. Ital. Urol. Androl. 2008;80:27–33. [PubMed] [Google Scholar]
- 17.Lahoud Y., Hussein O., Shalabi A., Nativ O., Awad H., Khamaisi M., Matar I., Nativ O., Abassi Z. Effects of phosphodiesterase-5 inhibitor on ischemic kidney injury during nephron sparing surgery: Quantitative assessment by NGAL and KIM-1. World J. Urol. 2015;33:2053–2062. doi: 10.1007/s00345-015-1579-3. [DOI] [PubMed] [Google Scholar]
- 18.Xie C., Liu L., Wang Z., Xie H., Feng Y., Suo J., Wang M., Shang W., Feng G. Icariin Improves Sepsis-Induced Mortality and Acute Kidney Injury. Pharmacology. 2018;102:196–205. doi: 10.1159/000487955. [DOI] [PubMed] [Google Scholar]
- 19.Abdel-latif R.G., Morsy M.A., El-Moselhy M.A., Khalifa M.A. Sildenafil protects against nitric oxide deficiency-related nephrotoxicity in cyclosporine A treated rats. Eur. J. Pharmacol. 2013;705:126–134. doi: 10.1016/j.ejphar.2013.02.039. [DOI] [PubMed] [Google Scholar]
- 20.Georgiadis G., Mavridis C., Belantis C., Zisis I.E., Skamagkas I., Fragkiadoulaki I., Heretis I., Tzortzis V., Psathakis K., Tsatsakis A., et al. Nephrotoxicity issues of organophosphates. Toxicology. 2018;406–407:129–136. doi: 10.1016/j.tox.2018.07.019. [DOI] [PubMed] [Google Scholar]
- 21.Ozlulerden Y., Toktas C., Aybek H., Kucukatay V., Sen Turk N., Zumrutbas A.E. The renoprotective effects of mannitol and udenafil in renal ischemia-reperfusion injury model. Investig. Clin. Urol. 2017;58:289–295. doi: 10.4111/icu.2017.58.4.289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang W., Yuan W., Xu N., Li J., Chang W. Icariin improves acute kidney injury and proteinuria in a rat model of pregnancyinduced hypertension. Mol. Med. Rep. 2017;16:7398–7404. doi: 10.3892/mmr.2017.7513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Whitaker R.M., Wills L.P., Stallons L.J., Schnellmann R.G. cGMP-selective phosphodiesterase inhibitors stimulate mitochondrial biogenesis and promote recovery from acute kidney injury. J. Pharmacol. Exp. Ther. 2013;347:626–634. doi: 10.1124/jpet.113.208017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhou Y.D., Hou J.G., Yang G., Jiang S., Chen C., Wang Z., Liu Y.Y., Ren S., Li W. Icariin ameliorates cisplatin-induced cytotoxicity in human embryonic kidney 293 cells by suppressing ROS-mediated PI3K/Akt pathway. Biomed. Pharmacother. 2019;109:2309–2317. doi: 10.1016/j.biopha.2018.11.108. [DOI] [PubMed] [Google Scholar]
- 25.van der Molen A.J., Reimer P., Dekkers I.A., Bongartz G., Bellin M.F., Bertolotto M., Clement O., Heinz-Peer G., Stacul F., Webb J.A.W., et al. Post-contrast acute kidney injury—Part 1: Definition, clinical features, incidence, role of contrast medium and risk factors: Recommendations for updated ESUR Contrast Medium Safety Committee guidelines. Eur. Radiol. 2018;28:2845–2855. doi: 10.1007/s00330-017-5246-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tsarouhas K., Tsitsimpikou C., Papantoni X., Lazaridou D., Koutouzis M., Mazzaris S., Rezaee R., Mamoulakis C., Georgoulias P., Nepka C., et al. Oxidative stress and kidney injury in trans-radial catheterization. Biomed. Rep. 2018;8:417–425. doi: 10.3892/br.2018.1071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mamoulakis C., Fragkiadoulaki I., Karkala P., Georgiadis G., Zisis I.E., Stivaktakis P., Kalogeraki A., Tsiaoussis I., Burykina T., Lazopoulos G., et al. Contrast-induced nephropathy in an animal model: Evaluation of novel biomarkers in blood and tissue samples. Toxicol. Rep. 2019;6:395–400. doi: 10.1016/j.toxrep.2019.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Morcos S.K. Can selective inhibitors of cyclic guanosine monophosphate (cGMP)-specific phosphadiesterase type 5 (PDE 5) offer protection against contrast induced nephropathy? Quant. Imaging Med. Surg. 2014;4:214–215. doi: 10.3978/j.issn.2223-4292.2014.06.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.El-Sisi A.E., Sokar S.S., Abu-Risha S.E., Ibrahim H.A. Combination of tadalafil and diltiazem attenuates renal ischemia reperfusion-induced acute renal failure in rats. Biomed. Pharmacother. 2016;84:861–869. doi: 10.1016/j.biopha.2016.10.009. [DOI] [PubMed] [Google Scholar]
- 30.Choi D.E., Jeong J.Y., Lim B.J., Chung S., Chang Y.K., Lee S.J., Na K.R., Kim S.Y., Shin Y.T., Lee K.W. Pretreatment of sildenafil attenuates ischemia-reperfusion renal injury in rats. Am. J. Physiol. Renal. Physiol. 2009;297:F362–F370. doi: 10.1152/ajprenal.90609.2008. [DOI] [PubMed] [Google Scholar]
- 31.Novak J.E., Handa R. Contrast Nephropathy Associated with Percutaneous Coronary Angiography and Intervention. Cardiol. Clin. 2019;37:287–296. doi: 10.1016/j.ccl.2019.04.004. [DOI] [PubMed] [Google Scholar]
- 32.van der Molen A.J., Reimer P., Dekkers I.A., Bongartz G., Bellin M.F., Bertolotto M., Clement O., Heinz-Peer G., Stacul F., Webb J.A.W., et al. Post-contrast acute kidney injury. Part 2: Risk stratification, role of hydration and other prophylactic measures, patients taking metformin and chronic dialysis patients: Recommendations for updated ESUR Contrast Medium Safety Committee guidelines. Eur. Radiol. 2018;28:2856–2869. doi: 10.1007/s00330-017-5247-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mamoulakis C., Tsarouhas K., Fragkiadoulaki I., Heretis I., Wilks M.F., Spandidos D.A., Tsitsimpikou C., Tsatsakis A. Contrast-induced nephropathy: Basic concepts, pathophysiological implications and prevention strategies. Pharmacol. Ther. 2017;180:99–112. doi: 10.1016/j.pharmthera.2017.06.009. [DOI] [PubMed] [Google Scholar]
- 34.Salonia A., Bettocchi C., Carvalho J., Corona G., Jones T.H., Kadioglu A., Martinez-Salamanca I., Minhas S., Serefoǧlu E.C., Verze P., et al. EAU Guidelines on Sexual and Reproductive Health. Edn. Presented at the EAU Annual Congress Amsterdam 2020. EAU Guidelines Office; Arnhem, The Netherlands: 2020. [Google Scholar]
- 35.Ozbek K., Ceyhan K., Koc F., Sogut E., Altunkas F., Karayakali M., Celik A., Kadi H., Koseoglu R.D., Onalan O. The protective effect of single dose tadalafil in contrast-induced nephropathy: An experimental study. Anatol. J. Cardiol. 2015;15:306–310. doi: 10.5152/akd.2014.5380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lue T.F. Erectile dysfunction. N. Engl. J. Med. 2000;342:1802–1813. doi: 10.1056/NEJM200006153422407. [DOI] [PubMed] [Google Scholar]
- 37.Moher D., Liberati A., Tetzlaff J., Altman D.G., Group P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Int. J. Surg. 2010;8:336–341. doi: 10.1016/j.ijsu.2010.02.007. [DOI] [PubMed] [Google Scholar]
- 38.Li W., Wang L., Chu X., Cui H., Bian Y. Icariin combined with human umbilical cord mesenchymal stem cells significantly improve the impaired kidney function in chronic renal failure. Mol. Cell. Biochem. 2017;428:203–212. doi: 10.1007/s11010-016-2930-8. [DOI] [PubMed] [Google Scholar]
- 39.Krane L.S., Peyton C.C., Olympio M.A., Hemal A.K. A randomized double blinded placebo controlled trial of sildenafil for renoprotection prior to hilar clamping in patients undergoing robotic assisted laparoscopic partial nephrectomy. J. Surg. Oncol. 2016;114:785–788. doi: 10.1002/jso.24419. [DOI] [PubMed] [Google Scholar]
- 40.Polcari A.J., Farooq A.V., Woods M.E., Ripsch M.S., Picken M., Turk T.M., White F.A. Effect of the phosphodiesterase-5 inhibitor zaprinast on ischemia-reperfusion injury in rats. J. Endourol. 2013;27:338–342. doi: 10.1089/end.2012.0198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lee K.W., Jeong J.Y., Lim B.J., Chang Y.K., Lee S.J., Na K.R., Shin Y.T., Choi D.E. Sildenafil attenuates renal injury in an experimental model of rat cisplatin-induced nephrotoxicity. Toxicology. 2009;257:137–143. doi: 10.1016/j.tox.2008.12.017. [DOI] [PubMed] [Google Scholar]
- 42.Oruc O., Inci K., Aki F.T., Zeybek D., Muftuoglu S.F., Kilinc K., Ergen A. Sildenafil attenuates renal ischemia reperfusion injury by decreasing leukocyte infiltration. Acta Histochem. 2010;112:337–344. doi: 10.1016/j.acthis.2009.02.005. [DOI] [PubMed] [Google Scholar]
- 43.Medeiros P.J., Villarim Neto A., Lima F.P., Azevedo I.M., Leao L.R., Medeiros A.C. Effect of sildenafil in renal ischemia/reperfusion injury in rats. Acta Cir. Bras. 2010;25:490–495. doi: 10.1590/S0102-86502010000600006. [DOI] [PubMed] [Google Scholar]
- 44.Ali B.H., Abdelrahman A.M., Al-Salam S., Sudhadevi M., AlMahruqi A.S., Al-Husseni I.S., Beegam S., Dhanasekaran S., Nemmar A., Al-Moundhri M. The effect of sildenafil on cisplatin nephrotoxicity in rats. Basic Clin. Pharmacol. Toxicol. 2011;109:300–308. doi: 10.1111/j.1742-7843.2011.00724.x. [DOI] [PubMed] [Google Scholar]
- 45.Akgul T., Huri E., Yagmurdur H., Ayyildiz A., Ustun H., Germiyanoglu C. Phosphodiesterase 5 inhibitors attenuate renal tubular apoptosis after partial unilateral ureteral obstruction: An experimental study. Kaohsiung J. Med. Sci. 2011;27:15–19. doi: 10.1016/j.kjms.2010.03.001. [DOI] [PubMed] [Google Scholar]
- 46.Lledo-Garcia E., Subira-Rios D., Ogaya-Pinies G., Tejedor-Jorge A., Canizo-Lopez J.F., Hernandez-Fernandez C. Intravenous sildenafil as a preconditioning drug against hemodynamic consequences of warm ischemia-reperfusion on the kidney. J. Urol. 2011;186:331–333. doi: 10.1016/j.juro.2011.03.036. [DOI] [PubMed] [Google Scholar]
- 47.Cadirci E., Halici Z., Odabasoglu F., Albayrak A., Karakus E., Unal D., Atalay F., Ferah I., Unal B. Sildenafil treatment attenuates lung and kidney injury due to overproduction of oxidant activity in a rat model of sepsis: A biochemical and histopathological study. Clin. Exp. Immunol. 2011;166:374–384. doi: 10.1111/j.1365-2249.2011.04483.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Patel N.N., Lin H., Toth T., Jones C., Ray P., Welsh G.I., Satchell S.C., Sleeman P., Angelini G.D., Murphy G.J. Phosphodiesterase-5 inhibition prevents postcardiopulmonary bypass acute kidney injury in swine. Ann. Thorac. Surg. 2011;92:2168–2176. doi: 10.1016/j.athoracsur.2011.07.002. [DOI] [PubMed] [Google Scholar]
- 49.Kucuk A., Yucel M., Erkasap N., Tosun M., Koken T., Ozkurt M., Erkasap S. The effects of PDE5 inhibitory drugs on renal ischemia/reperfusion injury in rats. Mol. Biol. Rep. 2012;39:9775–9782. doi: 10.1007/s11033-012-1843-1. [DOI] [PubMed] [Google Scholar]
- 50.Stegbauer J., Friedrich S., Potthoff S.A., Broekmans K., Cortese-Krott M.M., Quack I., Rump L.C., Koesling D., Mergia E. Phosphodiesterase 5 attenuates the vasodilatory response in renovascular hypertension. PLoS ONE. 2013;8:e80674. doi: 10.1371/journal.pone.0080674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Helmy M.W., Helmy M.M., Abd Allah D.M., Abo Zaid A.M., Mohy El-Din M.M. Role of nitrergic and endothelin pathways modulations in cisplatin-induced nephrotoxicity in male rats. J. Physiol. Pharmacol. 2014;65:393–399. [PubMed] [Google Scholar]
- 52.Gokakin A.K., Atabey M., Deveci K., Sancakdar E., Tuzcu M., Duger C., Topcu O. The effects of sildenafil in liver and kidney injury in a rat model of severe scald burn: A biochemical and histopathological study. Ulus Travma Acil Cerrahi Derg. 2014;20:319–327. doi: 10.5505/tjtes.2014.39586. [DOI] [PubMed] [Google Scholar]
- 53.Morsy M.A., Ibrahim S.A., Amin E.F., Kamel M.Y., Rifaai R.A., Hassan M.K. Sildenafil Ameliorates Gentamicin-Induced Nephrotoxicity in Rats: Role of iNOS and eNOS. J. Toxicol. 2014;2014:489382. doi: 10.1155/2014/489382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Cui W., Maimaitiyiming H., Qi X., Norman H., Zhou Q., Wang X., Fu J., Wang S. Increasing cGMP-dependent protein kinase activity attenuates unilateral ureteral obstruction-induced renal fibrosis. Am. J. Physiol. Renal. Physiol. 2014;306:F996–F1007. doi: 10.1152/ajprenal.00657.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lauver D.A., Carey E.G., Bergin I.L., Lucchesi B.R., Gurm H.S. Sildenafil citrate for prophylaxis of nephropathy in an animal model of contrast-induced acute kidney injury. PLoS ONE. 2014;9:e113598. doi: 10.1371/journal.pone.0113598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Zahran M.H., Hussein A.M., Barakat N., Awadalla A., Khater S., Harraz A., Shokeir A.A. Sildenafil activates antioxidant and antiapoptotic genes and inhibits proinflammatory cytokine genes in a rat model of renal ischemia/reperfusion injury. Int. Urol. Nephrol. 2015;47:1907–1915. doi: 10.1007/s11255-015-1099-5. [DOI] [PubMed] [Google Scholar]
- 57.de Almeida L.S., Barboza J.R., Freitas F.P., Porto M.L., Vasquez E.C., Meyrelles S.S., Gava A.L., Pereira T.M. Sildenafil prevents renal dysfunction in contrast media-induced nephropathy in Wistar rats. Hum. Exp. Toxicol. 2016;35:1194–1202. doi: 10.1177/0960327115626582. [DOI] [PubMed] [Google Scholar]
- 58.Mohey V., Singh M., Puri N., Kaur T., Pathak D., Singh A.P. Sildenafil obviates ischemia-reperfusion injury-induced acute kidney injury through peroxisome proliferator-activated receptor gamma agonism in rats. J. Surg. Res. 2016;201:69–75. doi: 10.1016/j.jss.2015.09.035. [DOI] [PubMed] [Google Scholar]
- 59.Altintop I., Tatli M., Karakukcu C., Soyer Sarica Z., Hanim Yay A., Balcioglu E., Ozturk A. Serum and Tissue HIF-2 Alpha Expression in CIN, N-Acetyl Cysteine, and Sildenafil-Treated Rat Models: An Experimental Study. Medicina. 2018;54:54. doi: 10.3390/medicina54040054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Behiry S., Rabie A., Kora M., Ismail W., Sabry D., Zahran A. Effect of combination sildenafil and gemfibrozil on cisplatin-induced nephrotoxicity; role of heme oxygenase-1. Ren. Fail. 2018;40:371–378. doi: 10.1080/0886022X.2018.1455596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zahran M.H., Barakat N., Khater S., Awadalla A., Mosbah A., Nabeeh A., Hussein A.M., Shokeir A.A. Renoprotective effect of local sildenafil administration in renal ischaemia-reperfusion injury: A randomised controlled canine study. Arab J. Urol. 2019;17:150–159. doi: 10.1080/2090598X.2019.1600995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ayyildiz A., Kaya M., Karaguzel E., Bumin A., Akgul T., Aklan Z., Germiyanoglu C. Effect of tadalafil on renal resistivity and pulsatility index in partial ureteral obstruction. Urol. Int. 2009;83:75–79. doi: 10.1159/000224873. [DOI] [PubMed] [Google Scholar]
- 63.Gasanov F., Aytac B., Vuruskan H. The effects of tadalafil on renal ischemia reperfusion injury: An experimental study. Bosn. J. Basic Med. Sci. 2011;11:158–162. doi: 10.17305/bjbms.2011.2567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Guzeloglu M., Yalcinkaya F., Atmaca S., Bagriyanik A., Oktar S., Yuksel O., Fansa I., Hazan E. The beneficial effects of tadalafil on renal ischemia-reperfusion injury in rats. Urol. Int. 2011;86:197–203. doi: 10.1159/000321927. [DOI] [PubMed] [Google Scholar]
- 65.Faddegon S., Best S.L., Olweny E.O., Tan Y.K., Park S.K., Mir S.A., Cadeddu J.A. Tadalafil for prevention of renal dysfunction secondary to renal ischemia. Can. J. Urol. 2012;19:6274–6279. [PubMed] [Google Scholar]
- 66.Sohotnik R., Nativ O., Abbasi A., Awad H., Frajewicki V., Bishara B., Sukhotnik I., Armaly Z., Aronson D., Heyman S.N., et al. Phosphodiesterase-5 inhibition attenuates early renal ischemia-reperfusion-induced acute kidney injury: Assessment by quantitative measurement of urinary NGAL and KIM-1. Am. J. Physiol. Renal. Physiol. 2013;304:F1099–F1104. doi: 10.1152/ajprenal.00649.2012. [DOI] [PubMed] [Google Scholar]
- 67.Zhu C.Y., Liu M., Liu Y.Z., Li W., Zhai W., Che J.P., Yan Y., Wang G.C., Zheng J.H. Preventive effect of phosphodiesterase 5 inhibitor tadalafil on experimental post-pyelonephritic renal injury in rats. J. Surg. Res. 2014;186:253–261. doi: 10.1016/j.jss.2013.07.056. [DOI] [PubMed] [Google Scholar]
- 68.Amasyali A.S., Akkurt A., Kazan E., Yilmaz M., Erol B., Yildiz Y., Erol H. The protective effect of tadalafil on IMA (ischemia modified albumin) levels in experimental renal ischemia-reperfusion injury. Int. J. Clin. Exp. Med. 2015;8:15766–15772. [PMC free article] [PubMed] [Google Scholar]
- 69.Erol B., Turker T., Tok A., Bektas S., Mungan G., Ozkanli S., Karakas B., Tokgoz H., Akduman B., Mungan A. The protective effects of tadalafil on renal damage following ischemia reperfusion injury in rats. Kaohsiung J. Med. Sci. 2015;31:454–462. doi: 10.1016/j.kjms.2015.06.005. [DOI] [PubMed] [Google Scholar]
- 70.Adeneye A.A., Benebo A.S. Chemopreventive Effect of Tadalafil in Cisplatin-Induced Nephrotoxicity in Rats. Niger. J. Physiol. Sci. 2016;31:1–10. [PubMed] [Google Scholar]
- 71.Benli E., Ayyildiz S.N., Cirrik S., Kokturk S., Cirakoglu A., Noyan T., Ayyildiz A., Germiyanoglu C. The effect of tadalafil therapy on kidney damage caused by sepsis in a polymicrobial septic model induced in rats: A biochemical and histopathological study. Int. Braz. J. Urol. 2017;43:345–355. doi: 10.1590/s1677-5538.ibju.2016.0075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wietzikoski E.G.G., Foiatto J.C., Czeczko N.G., Malafaia O., Koleski F.C., Mierzwa T.C., Gomes R.P.X. Tadalafil protector effect during ischemia-reperfusion in rats. Acta Cir. Bras. 2017;32:973–983. doi: 10.1590/s0102-865020170110000009. [DOI] [PubMed] [Google Scholar]
- 73.Medeiros V.F., Azevedo I.M., Carvalho M.D., Oliveira C.N., Egito E.S., Medeiros A.C. The renoprotective effect of oral Tadalafil pretreatment on ischemia/reperfusion injury in rats. Acta Cir. Bras. 2017;32:90–97. doi: 10.1590/s0102-865020170201. [DOI] [PubMed] [Google Scholar]
- 74.Kokturk S., Benli E., Ayyildiz A., Cirrik S., Cetinkol Y., Ayyildiz S.N., Noyan T. Positive outcomes of phosphodiesterase type 5 inhibitor on histopathologic and biochemical changes induced by ureteral obstruction. Rev. Assoc. Med. Bras. (1992) 2019;65:388–393. doi: 10.1590/1806-9282.65.3.388. [DOI] [PubMed] [Google Scholar]
- 75.Ma P., Zhang S., Su X., Qiu G., Wu Z. Protective effects of icariin on cisplatin-induced acute renal injury in mice. Am. J. Transl. Res. 2015;7:2105–2114. [PMC free article] [PubMed] [Google Scholar]
- 76.Chen H.A., Chen C.M., Guan S.S., Chiang C.K., Wu C.T., Liu S.H. The antifibrotic and anti-inflammatory effects of icariin on the kidney in a unilateral ureteral obstruction mouse model. Phytomedicine. 2019;59:152917. doi: 10.1016/j.phymed.2019.152917. [DOI] [PubMed] [Google Scholar]
- 77.Kyriazis I., Kagadis G.C., Kallidonis P., Georgiopoulos I., Marazioti A., Geronasiou A., Liourdi D., Loudos G., Schinas V., Apostolopoulos D., et al. PDE5 inhibition against acute renal ischemia reperfusion injury in rats: Does vardenafil offer protection? World J. Urol. 2013;31:597–602. doi: 10.1007/s00345-012-0980-4. [DOI] [PubMed] [Google Scholar]
- 78.Sousa R.C., Moreira Neto A.A., Capelozzi V.L., Ab’Saber A.M., Rodrigues O.R. Effects of vardenafil on the kidney of Wistar rats submitted to acute ischemia and reperfusion. Acta Cir. Bras. 2015;30:339–344. doi: 10.1590/S0102-865020150050000005. [DOI] [PubMed] [Google Scholar]
- 79.Guan Z., Miller S.B., Greenwald J.E. Zaprinast accelerates recovery from established acute renal failure in the rat. Kidney Int. 1995;47:1569–1575. doi: 10.1038/ki.1995.220. [DOI] [PubMed] [Google Scholar]
- 80.Wetzl V., Schinner E., Kees F., Faerber L., Schlossmann J. Differences in the renal antifibrotic cGMP/cGKI-dependent signaling of serelaxin, zaprinast, and their combination. Naunyn. Schmiedebergs. Arch. Pharmacol. 2017;390:939–948. doi: 10.1007/s00210-017-1394-z. [DOI] [PubMed] [Google Scholar]
- 81.Rodriguez-Iturbe B., Ferrebuz A., Vanegas V., Quiroz Y., Espinoza F., Pons H., Vaziri N.D. Early treatment with cGMP phosphodiesterase inhibitor ameliorates progression of renal damage. Kidney Int. 2005;68:2131–2142. doi: 10.1111/j.1523-1755.2005.00669.x. [DOI] [PubMed] [Google Scholar]
- 82.Lledo-Garcia E., Rodriguez-Martinez D., Cabello-Benavente R., Moncada-Iribarren I., Tejedor-Jorge A., Dulin E., Hernandez-Fernandez C., Del Canizo-Lopez J.F. Sildenafil improves immediate posttransplant parameters in warm-ischemic kidney transplants: Experimental study. Transplant. Proc. 2007;39:1354–1356. doi: 10.1016/j.transproceed.2007.01.082. [DOI] [PubMed] [Google Scholar]
- 83.Aboutabl M.E., Raafat M., Maklad Y.A., Kenawy S.A., El Din A.G. Sildenafil augments the beneficial hemodynamic and histopathological effects of amlodipine in nitric oxide-deficient hypertensive rats: Role of nitric oxide-cyclic GMP pathway. Pharmacol. Res. 2008;57:456–463. doi: 10.1016/j.phrs.2008.05.003. [DOI] [PubMed] [Google Scholar]
- 84.Lledo-Garcia E., Subira-Rios D., Rodriguez-Martinez D., Dulin E., Alvarez-Fernandez E., Hernandez-Fernandez C., del Canizo-Lopez J.F. Sildenafil as a protecting drug for warm ischemic kidney transplants: Experimental results. J. Urol. 2009;182:1222–1225. doi: 10.1016/j.juro.2009.05.006. [DOI] [PubMed] [Google Scholar]
- 85.Jeong K.H., Lee T.W., Ihm C.G., Lee S.H., Moon J.Y., Lim S.J. Effects of sildenafil on oxidative and inflammatory injuries of the kidney in streptozotocin-induced diabetic rats. Am. J. Nephrol. 2009;29:274–282. doi: 10.1159/000158635. [DOI] [PubMed] [Google Scholar]
- 86.Bae E.H., Kim I.J., Joo S.Y., Kim E.Y., Kim C.S., Choi J.S., Ma S.K., Kim S.H., Lee J.U., Kim S.W. Renoprotective effects of sildenafil in DOCA-salt hypertensive rats. Kidney Blood Press. Res. 2012;36:248–257. doi: 10.1159/000343414. [DOI] [PubMed] [Google Scholar]
- 87.Tapia E., Sanchez-Lozada L.G., Soto V., Manrique A.M., Ortiz-Vega K.M., Santamaria J., Medina-Campos O.N., Cristobal M., Avila-Casado C., Pedraza-Chaverri J., et al. Sildenafil treatment prevents glomerular hypertension and hyperfiltration in rats with renal ablation. Kidney Blood Press. Res. 2012;35:273–280. doi: 10.1159/000334952. [DOI] [PubMed] [Google Scholar]
- 88.Hosgood S.A., Randle L.V., Patel M., Watson C.J., Bradley J.A., Nicholson M.L. Sildenafil citrate in a donation after circulatory death experimental model of renal ischemia-reperfusion injury. Transplantation. 2014;98:612–617. doi: 10.1097/TP.0000000000000283. [DOI] [PubMed] [Google Scholar]
- 89.Dias A.T., Rodrigues B.P., Porto M.L., Gava A.L., Balarini C.M., Freitas F.P., Palomino Z., Casarini D.E., Campagnaro B.P., Pereira T.M., et al. Sildenafil ameliorates oxidative stress and DNA damage in the stenotic kidneys in mice with renovascular hypertension. J. Transl. Med. 2014;12:35. doi: 10.1186/1479-5876-12-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Dias A.T., Cintra A.S., Frossard J.C., Palomino Z., Casarini D.E., Gomes I.B., Balarini C.M., Gava A.L., Campagnaro B.P., Pereira T.M., et al. Inhibition of phosphodiesterase 5 restores endothelial function in renovascular hypertension. J. Transl. Med. 2014;12:250. doi: 10.1186/s12967-014-0250-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.El-Mahdy N.A., El-Sayad M.E., El-Kadem A.H. Combination of telmisartan with sildenafil ameliorate progression of diabetic nephropathy in streptozotocin-induced diabetic model. Biomed. Pharmacother. 2016;81:136–144. doi: 10.1016/j.biopha.2016.04.001. [DOI] [PubMed] [Google Scholar]
- 92.Tripathi A.S., Mazumder P.M., Chandewar A.V. Sildenafil, a phosphodiesterase type 5 inhibitor, attenuates diabetic nephropathy in STZ-induced diabetic rats. J. Basic Clin. Physiol. Pharmacol. 2016;27:57–62. doi: 10.1515/jbcpp-2015-0035. [DOI] [PubMed] [Google Scholar]
- 93.Pofi R., Fiore D., De Gaetano R., Panio G., Gianfrilli D., Pozza C., Barbagallo F., Xiang Y.K., Giannakakis K., Morano S., et al. Phosphodiesterase-5 inhibition preserves renal hemodynamics and function in mice with diabetic kidney disease by modulating miR-22 and BMP7. Sci. Rep. 2017;7:44584. doi: 10.1038/srep44584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Khames A., Khalaf M.M., Gad A.M., Abd El-Raouf O.M. Ameliorative effects of sildenafil and/or febuxostat on doxorubicin-induced nephrotoxicity in rats. Eur. J. Pharmacol. 2017;805:118–124. doi: 10.1016/j.ejphar.2017.02.046. [DOI] [PubMed] [Google Scholar]
- 95.Baijnath S., Murugesan S., Mackraj I., Gathiram P., Moodley J. The effects of sildenafil citrate on urinary podocin and nephrin mRNA expression in an L-NAME model of pre-eclampsia. Mol. Cell. Biochem. 2017;427:59–67. doi: 10.1007/s11010-016-2897-5. [DOI] [PubMed] [Google Scholar]
- 96.Sonneveld R., Hoenderop J.G., Isidori A.M., Henique C., Dijkman H.B., Berden J.H., Tharaux P.L., van der Vlag J., Nijenhuis T. Sildenafil Prevents Podocyte Injury via PPAR-gamma-Mediated TRPC6 Inhibition. J. Am. Soc. Nephrol. 2017;28:1491–1505. doi: 10.1681/ASN.2015080885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Ali B.H., Al Za’abi M., Adham S.A., Al Suleimani Y., Karaca T., Manoj P., Al Kalbani J., Yasin J., Nemmar A. The effect of sildenafil on rats with adenine-Induced chronic kidney disease. Biomed. Pharmacother. 2018;108:391–402. doi: 10.1016/j.biopha.2018.09.061. [DOI] [PubMed] [Google Scholar]
- 98.Mehanna O.M., El Askary A., Al-Shehri S., El-Esawy B. Effect of phosphodiesterase inhibitors on renal functions and oxidant/antioxidant parameters in streptozocin-induced diabetic rats. Arch. Physiol. Biochem. 2018;124:424–429. doi: 10.1080/13813455.2017.1419267. [DOI] [PubMed] [Google Scholar]
- 99.Danisoglu M.E., Aytac B., Kilicaslan H., Dogan S., Vuruskan H. Reduction of shock wave lithotripsy-induced renal tubular injury by tadalafil. Bratisl. Lek. Listy. 2013;114:616–620. doi: 10.4149/BLL_2013_131. [DOI] [PubMed] [Google Scholar]
- 100.Ozmerdiven G., Vuruskan B.A., Kaygisiz O., Vuruskan H. Protective effects of diltiazem and tadalafil on shock wave-induced kidney injury in rats. Bratisl. Lek. Listy. 2017;118:228–232. doi: 10.4149/BLL_2017_045. [DOI] [PubMed] [Google Scholar]
- 101.Qi M.Y., Kai C., Liu H.R., Su Y.H., Yu S.Q. Protective effect of Icariin on the early stage of experimental diabetic nephropathy induced by streptozotocin via modulating transforming growth factor beta1 and type IV collagen expression in rats. J. Ethnopharmacol. 2011;138:731–736. doi: 10.1016/j.jep.2011.10.015. [DOI] [PubMed] [Google Scholar]
- 102.Liang S.R., Bi J.W., Guo Z.L., Bai Y., Hu Z. Protective effect of icariin on kidney in 5/6 nephrectomized rats and its mechanism. Genet. Mol. Res. 2014;13:6466–6471. doi: 10.4238/2014.August.25.10. [DOI] [PubMed] [Google Scholar]
- 103.Huang Z., He L., Huang D., Lei S., Gao J. Icariin protects rats against 5/6 nephrectomy-induced chronic kidney failure by increasing the number of renal stem cells. BMC Complement. Altern. Med. 2015;15:378. doi: 10.1186/s12906-015-0909-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Su B., Ye H., You X., Ni H., Chen X., Li L. Icariin alleviates murine lupus nephritis via inhibiting NF-kappaB activation pathway and NLRP3 inflammasome. Life Sci. 2018;208:26–32. doi: 10.1016/j.lfs.2018.07.009. [DOI] [PubMed] [Google Scholar]
- 105.Hohenstein B., Daniel C., Wittmann S., Hugo C. PDE-5 inhibition impedes TSP-1 expression, TGF-beta activation and matrix accumulation in experimental glomerulonephritis. Nephrol. Dial. Transplant. 2008;23:3427–3436. doi: 10.1093/ndt/gfn319. [DOI] [PubMed] [Google Scholar]
- 106.Wang H.G., Yan X.Y., Cai Y.M., Wang H.G., Chen J.Y. The effect of phosphodiesterase isoenzyme 5 inhibitors on ureteropelvic junction obstruction in rabbits. Urol. Int. 2009;83:316–322. doi: 10.1159/000241675. [DOI] [PubMed] [Google Scholar]
- 107.Fang L., Radovits T., Szabo G., Mozes M.M., Rosivall L., Kokeny G. Selective phosphodiesterase-5 (PDE-5) inhibitor vardenafil ameliorates renal damage in type 1 diabetic rats by restoring cyclic 3′,5′ guanosine monophosphate (cGMP) level in podocytes. Nephrol. Dial. Transplant. 2013;28:1751–1761. doi: 10.1093/ndt/gfs391. [DOI] [PubMed] [Google Scholar]
- 108.Essiz D., Sozmen M., Sudagidan M., Devrim A.K. Phosphodiesterase type 5 inhibition attenuates cyclosporine A induced nephrotoxicity in mice. Biotech. Histochem. 2015;90:167–178. doi: 10.3109/10520295.2014.976270. [DOI] [PubMed] [Google Scholar]
- 109.Sekizawa T., Shima Y., Yoshida K., Tanahashi M., Yoshida M., Suzuki-Kusaba M., Hisa H., Satoh S. Effects of zaprinast on renal nerve stimulation-induced anti-natriuresis in anaesthetized dogs. Clin Exp Pharmacol. Physiol. 1998;25:1008–1012. doi: 10.1111/j.1440-1681.1998.tb02175.x. [DOI] [PubMed] [Google Scholar]
- 110.Yang J.W., Han S.T., Kim Y.S., Song S.H., Kim B.R., Eom M.S., Jung S.H., Choi S.O., Han B.G. Effects of a cGMP-specific phosphodiesterase inhibitor on expression of endothelial nitric oxide synthase and vascular endothelial growth factor in rats with cyclosporine-induced nephrotoxicity. Transplant. Proc. 2010;42:4625–4632. doi: 10.1016/j.transproceed.2010.09.167. [DOI] [PubMed] [Google Scholar]
- 111.Yuan J., Zhang R., Yang Z., Lee J., Liu Y., Tian J., Qin X., Ren Z., Ding H., Chen Q., et al. Comparative effectiveness and safety of oral phosphodiesterase type 5 inhibitors for erectile dysfunction: A systematic review and network meta-analysis. Eur. Urol. 2013;63:902–912. doi: 10.1016/j.eururo.2013.01.012. [DOI] [PubMed] [Google Scholar]
- 112.Dhaliwal A., Gupta M. PDE5 Inhibitor. StatPearls; Treasure Island, FL, USA: 2020. [Google Scholar]
- 113.Tzoumas N., Farrah T.E., Dhaun N., Webb D.J. Established and emerging therapeutic uses of PDE type 5 inhibitors in cardiovascular disease. Br. J. Pharmacol. 2019 doi: 10.1111/bph.14920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Oelke M., Weiss J.P., Mamoulakis C., Cox D., Ruff D., Viktrup L. Effects of tadalafil on nighttime voiding (nocturia) in men with lower urinary tract symptoms suggestive of benign prostatic hyperplasia: A post hoc analysis of pooled data from four randomized, placebo-controlled clinical studies. World J. Urol. 2014;32:1127–1132. doi: 10.1007/s00345-014-1255-z. [DOI] [PubMed] [Google Scholar]
- 115.Sakalis V.I., Karavitakis M., Bedretdinova D., Bach T., Bosch J., Gacci M., Gratzke C., Herrmann T.R., Madersbacher S., Mamoulakis C., et al. Medical Treatment of Nocturia in Men with Lower Urinary Tract Symptoms: Systematic Review by the European Association of Urology Guidelines Panel for Male Lower Urinary Tract Symptoms. Eur. Urol. 2017;72:757–769. doi: 10.1016/j.eururo.2017.06.010. [DOI] [PubMed] [Google Scholar]
- 116.Dell’Agli M., Galli G.V., Dal Cero E., Belluti F., Matera R., Zironi E., Pagliuca G., Bosisio E. Potent inhibition of human phosphodiesterase-5 by icariin derivatives. J. Nat. Prod. 2008;71:1513–1517. doi: 10.1021/np800049y. [DOI] [PubMed] [Google Scholar]
- 117.Mackenzie A.E., Lappin J.E., Taylor D.L., Nicklin S.A., Milligan G. GPR35 as a Novel Therapeutic Target. Front. Endocrinol. (Lausanne) 2011;2:68. doi: 10.3389/fendo.2011.00068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Keswani A.N., Peyton K.J., Durante W., Schafer A.I., Tulis D.A. The cyclic GMP modulators YC-1 and zaprinast reduce vessel remodeling through antiproliferative and proapoptotic effects. J. Cardiovasc. Pharmacol. Ther. 2009;14:116–124. doi: 10.1177/1074248409333266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Zhang W.H., Zhang X.H. Clinical and preclinical treatment of urologic diseases with phosphodiesterase isoenzymes 5 inhibitors: An update. Asian J. Androl. 2016;18:723–731. doi: 10.4103/1008-682X.167721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Kang S.G., Kim J.J. Udenafil: Efficacy and tolerability in the management of erectile dysfunction. Ther. Adv. Urol. 2013;5:101–110. doi: 10.1177/1756287212470019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Aujla H., Kumar T., Wozniak M., Dott W., Sullo N., Joel-David L., Morris T., Brookes C., Barber S., Murphy G.J. Effect of sildenafil (Revatio) on postcardiac surgery acute kidney injury: A randomised, placebo-controlled clinical trial: The REVAKI-2 trial protocol. Open Heart. 2018;5:e000838. doi: 10.1136/openhrt-2018-000838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Paick J.S., Ahn T.Y., Choi H.K., Chung W.S., Kim J.J., Kim S.C., Kim S.W., Lee S.W., Min K.S., Moon K.H., et al. Efficacy and safety of mirodenafil, a new oral phosphodiesterase type 5 inhibitor, for treatment of erectile dysfunction. J. Sex. Med. 2008;5:2672–2680. doi: 10.1111/j.1743-6109.2008.00945.x. [DOI] [PubMed] [Google Scholar]
- 123.Glina S., Fonseca G.N., Bertero E.B., Damiao R., Rocha L.C., Jardim C.R., Cairoli C.E., Teloken C., Torres L.O., Faria G.E., et al. Efficacy and tolerability of lodenafil carbonate for oral therapy of erectile dysfunction: A phase III clinical trial. J. Sex. Med. 2010;7:1928–1936. doi: 10.1111/j.1743-6109.2010.01711.x. [DOI] [PubMed] [Google Scholar]
- 124.Tzortzis V., Mitrakas L., Gravas S., Mamoulakis C., Meissner A., Kyriakou D., Melekos M.D. Oral phosphodiesterase type 5 inhibitors alleviate recurrent priapism complicating thalassemia intermedia: A case report. J. Sex. Med. 2009;6:2068–2071. doi: 10.1111/j.1743-6109.2009.01285.x. [DOI] [PubMed] [Google Scholar]
- 125.Sofikitis N., Kaltsas A., Dimitriadis F., Rassweiler J., Grivas N., Zachariou A., Kaponis A., Tsounapi P., Paterakis N., Karagiannis A., et al. The Effect of PDE5 Inhibitors on the Male Reproductive Tract. Curr. Pharm. Des. 2020 doi: 10.2174/1381612826666200226121510. [DOI] [PubMed] [Google Scholar]
- 126.Bender A.T., Beavo J.A. Cyclic nucleotide phosphodiesterases: Molecular regulation to clinical use. Pharmacol. Rev. 2006;58:488–520. doi: 10.1124/pr.58.3.5. [DOI] [PubMed] [Google Scholar]
- 127.Afsar B., Ortiz A., Covic A., Gaipov A., Esen T., Goldsmith D., Kanbay M. Phosphodiesterase type 5 inhibitors and kidney disease. Int. Urol. Nephrol. 2015;47:1521–1528. doi: 10.1007/s11255-015-1071-4. [DOI] [PubMed] [Google Scholar]
- 128.Shen K., Johnson D.W., Gobe G.C. The role of cGMP and its signaling pathways in kidney disease. Am. J. Physiol. Renal. Physiol. 2016;311:F671–F681. doi: 10.1152/ajprenal.00042.2016. [DOI] [PubMed] [Google Scholar]
- 129.Yamanaka R., Shindo Y., Hotta K., Suzuki K., Oka K. NO/cGMP/PKG signaling pathway induces magnesium release mediated by mitoKATP channel opening in rat hippocampal neurons. FEBS Lett. 2013;587:2643–2648. doi: 10.1016/j.febslet.2013.06.049. [DOI] [PubMed] [Google Scholar]