Abstract
Background
Current WHO guidelines (2018) recommend screening for cryptococcal antigen (CrAg) in HIV-infected persons with CD4+ T cell counts< 100 cells/μL, followed by pre-emptive antifungal therapy among CrAg positive (CrAg+) persons, to prevent cryptococcal meningitis related deaths. This strategy may also be considered for those persons with a CD4+ T cell count of < 200 cells/uL according the WHO guidelines. However, there is sparse evidence in the literature supporting CrAg screening and pre-emptive antifungal therapy in those HIV-infected persons with this CD4+ T cell counts< 200 cells/μL.
Method
We conducted a meta-analysis using data extracted from randomized controlled studies (RCTs) and cohort studies found in a search of Pubmed, Web of Science, the Cochrane Library and the EMBASE/MEDLINE database.
Results
The pooled prevalence of CrAg positivity in HIV-infected persons with CD4+ T cell counts< 200 cells/μL was 5% (95%CI: 2–7). The incidence of CM in CrAg+ persons was 3% (95%CI: 1–6). Among those CrAg+ persons who did not receive pre-emptive treatment, or those who received placebo, the incidence of CM was 5% (95%CI: 2–9), whereas the incidence of CM among those who received pre-emptive antifungal therapy was 3% (95%CI: 1–6), which is a statistically significant reduction in incidence of 40% (RR: 7.64, 95%CI: 2.96–19.73, p < 0.00001). As for persons with CD4+ T cell counts between 101 ~ 200 cells/μL, the risk ratio for the incidence of CM among those receiving placebo or no intervention was 1.15, compared to those receiving antifungal treatment (95%CI: 0.16–8.13).
Conclusions
In our meta-analysis the incidence of CM was significantly reduced by pre-emptive antifungal therapy in CrAg+ HIV-infected persons with CD4 < 200 cells/μL. However, more specific observational data in persons with CD4+ T cell counts between 101 ~ 200 cells/μL are required in order to emphasize specific benefit of CrAg screening and pre-emptive antifungal treating in CrAg+ persons with CD4+ T cell counts < 200 cells/μL.
Keywords: HIV, Cryptococcal antigenemia, Screening, CD4 + T cell count, Pre-emptive treatment, Meta-analysis
Background
Cryptococcal meningitis (CM) continues to cause significant mortality in HIV-infected individuals [1, 2], and results in 181,100 deaths globally each year [3]. In resource-limited regions such as sub-Saharan Africa, 15% of HIV-related deaths are due to CM [3]. However, it is possible to detect cryptococcal antigen (CrAg) in blood several weeks to months (22 days on average) before the onset of signs and symptoms of meningitis [4, 5], and thus, the presence of CrAg in blood may be used as a marker for initiation of pre-emptive antifungal therapy in HIV-infected individuals with low CD4+ T cell counts. Previous studies have emphasized that pre-emptive antifungal therapy in CrAg+ persons is imperative to prevent death [6–8]. The prevalence of CrAg positivity among HIV-infected individuals can be considerable, ranging between 1 to 16% in several African and Southeast Asian countries [9], and among persons with CD4+ T cells counts< 100 cells/μL, the prevalence of CrAg positivity averages 7%, with regional variations in prevalence [3]. CrAg positivity resulted in a 20% increase in mortality after antiretroviral therapy (ART) initiation [10] if fluconazole therapy was not initiated prior to ART initiation, and the risk of CM in CrAg+ persons may be as high as 25% during the first year of ART, when fluconazole pre-emptive therapy is not prescribed for these patients [11, 12].
According to the 2018 version of the WHO guidelines, routine CrAg screening and pre-emptive antifungal therapy are recommended in treatment-naive HIV persons with CD4+ T cell counts< 100 cells/μL [13]. The guidelines also state that these strategies may also be considered for HIV-infected persons with CD4+ T cell counts< 200 cells/μL [13]. We therefore conducted a meta-analysis to investigate the prevalence of CrAg positivity in HIV-infected patients, and the benefit of pre-emptive antifungal treatment in HIV-infected persons with CD4+ T cell counts< 200 cells/μL.
Method
Search strategy and article screening
We searched relevant English articles in Pubmed, Cochrane Library, MEDLINE/EMBASE and Web of Science from inception until the end of March 20th 2020. The search terms we used were as follows: “acquired immunodeficiency syndrome”, “HIV”, “AIDS”, “cryptococcosis”, and “prophylaxis”. We combined these terms by using “and” or “or”. To avoid missing significant articles, we also screened references of previous meta-analyses and their included studies for eligibility.
Two reviewers (Y L, Y Q) independently screened all obtained articles by titles and abstracts. After removing ineligible articles by referring to our inclusion and exclusion criteria, the remaining articles were further selected for full-text reviewing.
Inclusion and exclusion criteria
Inclusion criteria
Randomized-controlled studies (RCTs) or cohort studies,
Study subjects had baseline CD4+ T cell counts< 200 cells/μL.
CrAg serology was tested for study subjects.
Fluconazole or other azole medications were used as the intervention
Exclusion criteria
We excluded articles if: (1) all of the study subjects were with CD4+ T cell counts< 100 cells/μL; (2) all of the study subjects were diagnosed with CM or asymptomatic CM; (3) sample size was less than 50; or (4) the incidence of CM and all-cause mortality was unreported.
Data extraction and quality assessment
The data we extracted included first author, publication year, type of study, study duration, study location, total number of study subjects, baseline CD4+ T-cell counts, age, CrAg screening methods, diagnostic methods for CM, CM events, death events, adverse drug effects, and other opportunistic infections. The JBI (Joanna Briggs Institute) Critical Appraisal Checklist for Cohort Studies was used as a quality assessment tool for cohort studies [14]. The potential bias risk of RCTs was assessed using the Cochrane “risk of bias” tool [15].
Data analysis
Statistical analysis of data related to proportion of CrAg positivity, the incidence of CM, and all-cause mortality were performed by STATA 14 (Statacorp, Texas, USA) with a 95% confidence interval (95%CI). We used random-effects or fixed-effects models in Review manager 5.3 (The Nordic Cochrane Center, Copenhagen) to compare the incidence of CM and all-cause mortality in CrAg+ persons.
We evaluated statistical heterogeneity through visual inspection of forest plots. Statistical heterogeneity was also assessed using I2 statistics [16], which was considered non-negligible if I2 > 50%. Herein, random-model was applied if I2 > 50% and fixed-model was used when I2 < 50% [17]. Reporting bias was assessed by examining the asymmetry of funnel plots [16].
The study was registered at the International Prospective Register of Systematic Reviews (PROSPERO), and the registration number is CRD42018110980.
Results
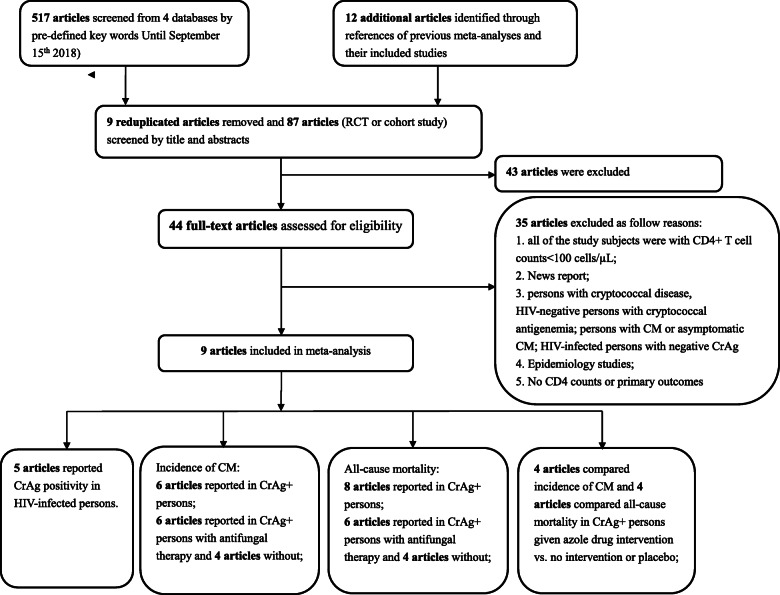
In total, 517 articles were obtained from 4 databases, among which 295 were from Pubmed, 111 were from Web of Science, 13 were from Cochrane Library, and 98 were from MEDLINE/EMBASE. Eighty-four of the 517 articles were RCTs or cohort studies. Additional 12 articles (RCTs or cohort studies) were extracted from references of previous meta-analyses and their included studies, as shown in Fig. 1.
Fig. 1.
Flow chart of the study selection process
All the 96 RCTs or cohort studies were included for screening. Initially, nine articles (six from Web of Science and three from MEDLINE/EMBASE) were found to be duplicated, and were therefore excluded from the 96 articles. After screening titles and abstracts, 43 of the remaining 87 articles were excluded. Subsequently, 35 articles were excluded from the remaining 44 articles after screening the full-text of each study, among which ten articles only included patients with CD4+ T-cell counts≤100 cells/μL, one article was a news report, three articles reported patients with cryptococcal disease, two articles reported HIV-negative patients with cryptococcal antigenemia, four articles reported data from patients with CM or asymptomatic CM, six articles reported data from HIV-infected patients with negative CrAg, one article reported on the epidemiology of cryptococcosis, and eight articles did not report CD4+ T cell counts or primary outcomes. Finally, a total of 9 articles were included in our meta-analysis.
The characteristics of the 9 included studies were shown in Table 1. Our assessment of quality and potential risk bias in these studies indicated that the following factors could contribute to clinical and methodological heterogeneity, including: (1) the confounding factors or subject recruiting or incomplete follow-up in one of the 8 cohort studies, (2) the unclear risk of attrition in the RCT, and (3) the unclear risk of reporting and other bias in the RCT, as shown in Supplementary Table 1 and Supplementary Table 2. The reporting bias presented by funnel plots were shown in Supplementary Figure 2.
Table 1.
Characteristics of the 9 included studies
| Author, year (reference) | Number of participants | Study type | Study duration | Age (years) | CD4 (cells/μL) | CrAg screening methods | CM diagnostic methods | Location | Therapeutic regimens | Primary outcomes | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Incidence of CM | All-cause mortality | ||||||||||
| Chariyalertsak, 2002 [18] | 129 | Prospective study | 104 weeks | 18 ~ 60 | <200 | Not report | Fungal culture, a histopathological examination, or buffy coat smear | Thailand | 63 for oral itraconazole (200 mg/day) as group 1; 66 for matched placebo as group 2 | 0 in group 1; 7 in group 2 | 12 in group 1; 11 in group 2 |
| Manfredi, 1997 [19] | 249 | Retrospective study | 6 years | 22 ~ 59 | <200 | Not report | Specific polysaccharide antigen detection from body fluids | Italy | 128 for oral fluconazole (100 mg/d every third week) as group 1; 121 for no antifungal treatment as group 2 | 2 in group 1; 9 in group 2 | 12 in group 1; 13 in group 2 |
| Parkes-Ratanshi, 2011 [20] | 1519 | Prospective study | 42 months | Not report | <200 | Not report | CrAg titre> 1:8 on two occasions, or a positive CSF CrAg or Cryptococcus neoformans grown from blood or CSF culture | Uganda | 760 fluconazole 200 mg 3 times per week for minimum 12 weeks as group 1; 759 allocated to placebo as group 2 | 1 in group 1; 18 in group 2 | 0 in group 1; 7 in group 2 |
| McKinsey, 1999 [21] | 295 | Randomized, placebo-Controlled study | Not report | ≥13 | <150 | Not report | Fungal culture | Not report | 149 for itraconazole capsules (200 mg/day) as group 1; 146 for matched placebo as 2 | 1 in group 1; 8 in group 2 | 32 in group 1; 21 in group 2 |
| Meya, 2010 [22] | 584 | Prospective study | 30 months | ≥18 | <200 | Not report | Not report | Uganda | Fluconazole (200 ~ 400 mg) for 2 ~ 4 weeks | 3 in CrAg+ persons and 0 in CrAg- persons | 6 in CrAg+ persons and 0 in CrAg- persons |
| Kapoor, 2015 [23] | 72 | Retrospective study | 15 months | ≥18 | <200 | LFA | Positive CSF India ink | Sub-Saharan Africa | 800 mg fluconazole orally for 2 weeks, followed by 400 mg orally for 2 weeks | 0 in CrAg+ persons and 1 in CrAg- persons | 2 in CrAg+ persons and 8 in CrAg- persons |
| Govender, 2015a [1] | 1079 | Retrospective study | 19 months | Not report | < 200 | LA or the Latex-Cryptococcus antigen detection system | CrAg detected in CSF | South Africa | Fluconazole ranging from 400 to 800 mg per day for at least 3 months | unknow in persons with CD4 < 200 | unknow in persons with CD4 < 200 |
| Beyene, 2017 [10] | 783 | Prospective study | 18 months | > 14 | ≤150 | LFA | CSF CrAg | Ethiopia | Fluconazole 800 mg/day 2 weeks, followed by 400 mg/day 8 weeks | 2 in CrAg+ persons and 0 in CrAg- persons | 4 in CrAg+ persons and 0 in CrAg- persons |
| Borges, 2019 [24] | 214 | Prospective study | 36 months | > 17 | < 200 | LFA | India ink microscopy on the CSF, CSF CrAg test and fungal culture | Brazil | Fluconazole 900 mg for 2 weeks, 450 mg for 8 to 10 weeks and a subsequent maintenance dose of 150–300 mg | 1 in CrAg+ persons with antifungal treatment and 0 in CrAg+ persons without intervention | 2 in CrAg+ persons |
“No” means “no data”; “Yes” means “data exists”; OIs means: other opportunity infections
LFA Lateral flow assay, LA Latex Agglutination, LP Lumbar puncture
a“Govender, 2015” study was included for evaluating the prevalence of CrAg positivity. Only the data in persons with CD4 < 200 was used
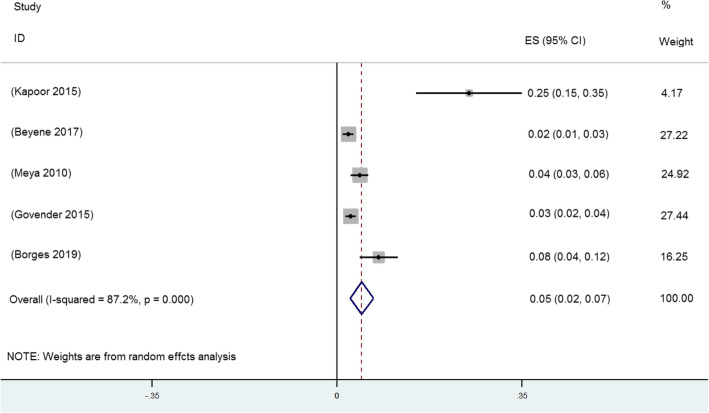
Five of the 9 included studies reported the prevalence of CrAg positivity (1949 persons with CD4+ T cell counts< 200 cells/μL in four studies; 783 persons with CD4+ T cell counts< 150 cells/μL in one study). The pooled CrAg positivity prevalence in 2732 HIV-infected persons with CD4+ T cell counts < 200 cells/μL was 5% (95%CI: 2–7, I2 = 87.2%), as shown in Fig. 2.
Fig. 2.
Prevalence of CrAg positivity among HIV-infected persons with CD4+ T cells< 200 cells/μL. Abbreviations: ES = effect size
Six studies reported the incidence of CM among CrAg+ persons (1806 persons with CD4+ T cell counts< 200 cells/μL in four studies; 312 persons with CD4+ T cell counts< 150 cells/μL in two study). The incidence of CM in 2118 CrAg+ persons was 3% (95%CI: 1–5; P = 0.021; I2 = 62.3%), as shown in Table 2 and Supplementary Figure 1a.
Table 2.
The incidence of CM and all-cause mortality among CrAg+ and CrAg- persons, and among persons with and without antifungal therapy
| Number of reported studies | Number of persons | ES (95%CI) | I2 | P | |
|---|---|---|---|---|---|
| Incidence of CM among CrAg + persons | |||||
| 1.1 Incidence of CM among CrAg+ persons | 6 | 2118 | 0.03 (0.01, 0.05) | 62.3% | 0.021 |
| Incidence of CM among persons with and without antifungal therapy | |||||
| 2.1 Incidence of CM among persons with antifungal therapy | 6 | 1088 | 0.03 (0.01, 0.06) | 57.7% | 0.037 |
| 2.2 Incidence of CM among persons without antifungal therapy | 4 | 1092 | 0.05 (0.02, 0.09) | 71.5% | 0.015 |
| All-cause mortality in CrAg+ persons | |||||
| 1.1 All-cause mortality among CrAg+ persons | 8 | 2265 | 0.14 (0.06, 0.22) | 94.5% | 0.000 |
| All-cause mortality among persons who with and without antifungal therapy | |||||
| 2.1 All-cause mortality among persons with antifungal therapy | 6 | 396 | 0.17 (0.11, 0.24) | 58.3% | 0.035 |
| 2.2 All-cause mortality among persons without antifungal therapy | 4 | 1092 | 0.10 (0.01, 0.19) | 93.2% | 0.000 |
Six studies reported the incidence of CM among persons who received antifungal therapy (922 persons with CD4+ T cell counts< 200 cells/μL in four studies; 166 persons with CD4+ T cell counts< 150 cells/μL in two studies) and four studies reported the incidence of CM among persons who received placebo or no intervention (946 persons with CD4+ T cell counts< 200 cells/μL in three studies; 146 persons with CD4+ T cell counts< 150 cells/μL in one studies). The incidence of CM of 1088 persons receiving antifungal therapy was 3% (95%CI: 1–6; P = 0.037; I2 = 57.7%), whereas the incidence of CM of 1092 persons in nine studies who received placebo or no intervention was 5%, which equates to a 40% reduction in CM incidence in persons receiving antifungal therapy (95%CI: 2–9; P = 0.015; I2 = 71.5%), as shown in Table 2 and Supplementary Figure 1c and d.
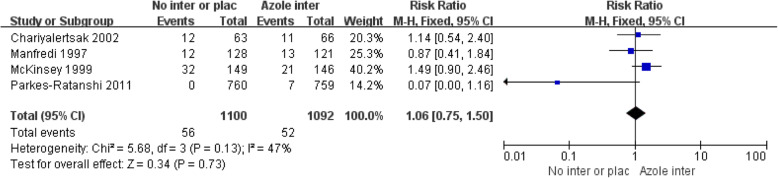
Four studies compared the incidence of CM between 1030 persons receiving azoles and 1050 persons receiving placebo or no intervention (1785 persons with CD4+ T cell counts< 200 cells/μL in three studies; 295 persons with CD4+ T cell counts< 150 cells/μL in one study). We found that the risk ratio of CM events among persons who received placebo or no intervention was 7.64 times higher than that of those who received antifungal therapy (95%CI: 2.96–19.73; P < 0.00001; I2 = 0%), as shown in Fig. 3.
Fig. 3.
Forest plots of incidence of CM among CrAg + persons receiving azole vs. no intervention or placebo. Abbreviations: M-H, Mantel Haenszel; CI, confidence interval. (“Azole inter” means “Azole drug intervention”, “No inter or plac” mean “No intervention or placebo”)
Eight studies reported all-cause mortality among CrAg+ persons (1953 persons with CD4+ T cell counts< 200 cells/μL in six studies; 312 persons with CD < 150 cells/μL in two studies). The all-cause mortality of 2265 CrAg+ persons was 14% (95%CI: 6–22; P = 0.000; I2 = 93.6%), as shown in Table 2 and Supplementary Figure 1b.
Six studies reported all-cause mortality in persons who received antifungal therapy (230 persons with CD4+ T cell counts< 200 cells/μL in four studies; 166 persons with CD4+ T cell counts< 150 cells/μ in two studies), four studies reported all-cause mortality in persons receiving placebo or no intervention (946 persons with CD4+ T cell counts< 200 cells/μL in three studies; 146 persons with CD4+ T cell counts< 150 cells/μL in one study). The all-cause mortality of 396 persons receiving antifungal therapy was 17% (95%CI: 11–24; P = 0.035; I2 = 58.3%), whereas the all-cause mortality of 1092 CrAg+ persons receiving placebo or no intervention was10%, (95%CI: 1–19; P = 0.000; I2 = 93.2%). Details are shown in Table 2 and Supplementary Figure 1e and f.
Four studies (1897 persons with CD4+ T cell counts< 200 cells/μL in three studies; 295 persons with CD4+ T cell counts< 150 cells/μL in one study) compared all-cause mortality between persons who received azole antifungal therapy and persons who received placebo or no intervention. No significant difference was found in all-cause mortality (risk ratio: 1.06, 95%CI: 0.75–1.50; P = 0.73; I2 = 47%) between 1100 CrAg+ persons who received an azole drug and 1092 CrAg+ persons who received placebo or no intervention (Fig. 4).
Fig. 4.
Forest plots of all-cause mortality among CrAg + persons receiving azole vs. no intervention or placebo. Abbreviations: M-H, Mantel Haenszel; CI, confidence interval. (“Azole inter” means “Azole drug intervention”, “No inter or plac” mean “No intervention or placebo”)
In addition, we estimated and compared the prevalence of CrAg positivity, the incidence of CM and all-cause mortality between persons with CD4+ T cell counts< 100 and persons with CD4+ T cell counts between 100 and 200 cells/μL. The results showed that the risk ratio of CrAg positivity prevalence among HIV-infected persons with CD4+ T cell counts< 100 cells/μL was 1.82 times that of those with 100–200 cells/μL (95%CI: 0.77–4.30; p = 0.007, I2 = 63%; three studies; 1886 persons). The risk ratio of the incidence of CM among HIV-infected persons with CD4+ T cell counts< 100 cells/μL was 2.53 times that of those with CD4+ T cell counts between 100 and 200 cells/μL (95%CI: 0.50–12.71, p = 0.26, I2 = 59%; four studies, 1960 persons). The risk ratio of the all-cause mortality among HIV-infected persons with CD4+ T cell counts < 100 cells/μL was 4.15 times that of those with CD4+ T cell counts between 100 and 200 cells/μL (95%CI: 0.89–19.42, p = 0.07, I2 = 0%; two studies, 1552 persons). Further, the risk ratio of the incidence of CM among persons with CD4+ T cell counts between 100 and 200 cells/μL receiving placebo or no intervention was 1.15 times compared to those receiving antifungal treatment (95%CI: 0.16–8.13, p = 0.97, I2 = 0%; three studies; 140 persons). The risk ratio of the all-cause mortality among persons with CD4+ T cell counts between 100 and 200 cells/μL receiving antifungal treatment was 0.27 compared to those receiving placebo or no intervention (95%CI: 0.01–4.93, p and I2 not applicable; one study; seven persons). Details are shown in Table 3.
Table 3.
Comparisons of CrAg positivity, incidence of CM and all-cause mortality among HIV-infected persons stratified by CD4+ T cell count
| Author, year | Prevalence of CrAg positivity | Incidence of CM | All-cause mortality | |||||||
| CD4<100 (cells/μL) | CD4 100 ~ 200 (cells/μL) | CD4<100 (cells/μL) | CD4 100 ~ 200 (cells/μL) | CD4<100 (cells/μL) | CD4 100 ~ 200 (cells/μL) | |||||
| with antifungal treatment | without antifungal treatment | with antifungal treatment | without antifungal treatment | with antifungal treatment | without antifungal treatment | with antifungal treatment | without antifungal treatment | |||
| Chariyalertsak, 2002 [25] | Not reported | 0 of 40 | 6 of 45 | 0 of 23 | 0 of 21 | Not reported | Not reported | |||
| Parkes-Ratanshi, 2011 [18] | Not reported | 17 of 698 | 2 of 821 | 3a of 698 | 0a of 821 | |||||
| McKinsey, 1999 [19] | Not reported | 0 of 101 | 7 of 103 | 1 of 48 | 1 of 43 | Not reported | Not reported | |||
| Meya, 2010 [21] | 26 of 295 | 7 of 298 | 3 of 21 | 2 of 5 | Not reported | 6 of 21 | 5 of 5 | 0 of 4 | 1 of 3 | |
| Govender, 2015 [1] | 20 of 708 | 6 of 371 | Not reported | Not reported | Not reported | Not reported | ||||
| Borges, 2019 [24] | 12 of 159 | 5 of 5 | 0 of 9 | 0 of 3 | 0 of 1 | 1 of 4 | Not reported | Not reported | ||
|
Pooled CrAg + prevalence: 0.06 [−0.02, 0.11], p = 0.001, I2 = 86.5% |
Pooled CrAg + prevalence: 0.02 [0.01, 0.03], p = 0.146, I2 = 48% |
Risk ratio with 95%CI: 7.67 [2.03, 29.05], p = 0.36, I2 = 1% |
Risk ratio with 95%CI: 1.15 [0.16, 8.13], p = 0.97, I2 = 0% |
Risk ratio with 95%CI: 0.32 [0.16, 0.64], p and I2 not applicable |
Risk ratio with 95%CI: 0.27 [0.01, 4.93], p and I2 not applicable |
|||||
| Risk ratio with 95%CI | 1.82 [0.77, 4.30], p = 0.007, I2 = 63% | 2.53 [0.50, 12.71], p = 0.26, I2 = 59% | 4.15 [0.89, 19.42], p = 0.07, I2 = 0% | |||||||
a Died within 4 weeks
Discussion
Several meta-analyses have been conducted in the past designed to evaluate the necessity of CrAg screening and administration of pre-emptive antifungal treatment among HIV-infected CrAg+ persons with varying low CD4 levels. For example, Temfack et al investigated the effectiveness of CrAg detection and the initiation of pre-emptive fluconazole treatment in HIV-infected persons with cryptococcal antigenemia and CD4+ T cell levels< 100 cells/μL [16]. Their results suggested that administration of fluconazole pre-emptive therapy to CrAg+ persons greatly reduced the risk of incident CM, and may have specific survival benefits [16]. Another meta-analysis conducted by Ssekitoleko et al also suggested that in resource-limited settings, CrAg+ persons should routinely receive primary antifungal prophylaxis [25], but they failed to clarify at which specific CD4+ T cell count antifungal prophylaxis should be initiated. Ford et al’s [26] meta-analysis only reported the combined prevalence of cryptococcal antigenemia among HIV-infected persons with CD4+ T cell counts≤100 cells/μL, and with CD4+ T cell counts between 101 ~ 200 cells/μL. Importantly, their study did not mention whether pre-emptive antifungal treatment was necessary or effective among HIV-infected persons with cryptococcal antigenemia at these two CD4+ T cell count strata. From the above studies, it may be gathered that the prudence and benefits of CrAg screening and pre-emptive antifungal therapy remain unclear at higher CD4+ T cell counts. The objective of our meta-analysis was to investigate the prevalence of cryptococcal antigenemia in HIV-infected patients with CD4+ T cell counts< 200 cells/μL, and the potential benefit of pre-emptive antifungal therapy among HIV-infected persons with cryptococcal antigenemia and CD4+ T cell counts< 200 cells/μL.
The pooled prevalence of CrAg positivity in HIV-infected persons with CD4+ T cell counts< 200cells/μL was 5% (5 studies) in our meta-analysis, which was similar to 6% (31 studies) among HIV-infected persons with CD4+ T cell counts< 100cells/μL in Temfack’s meta-analysis [16] and 6.5% (60 studies) among HIV-infected persons with CD4+ T cell counts < 100 cells/μL in Ford’s meta-analysis [26]. Therefore, antifungal prophylaxis seems imperative for HIV-infected persons with cryptococcal antigenemia who have CD4+ T cell counts< 200 cells/μL.
Our results have demonstrated that, in persons with CD4+ T cell count< 200 cells/μL, the risk ratio of CM events among those who received placebo or no intervention was significant higher than those who received antifungal therapy, suggesting that antifungal prophylaxis significantly reduce the risk of CM events in CrAg+ persons with a higher CD4+ T-cell counts. However, the very limited data among persons with CD4+ T cell counts between 101 ~ 200 cells/μL restricted our capacity to investigate it further. Thus, more specific data are needed to demonstrate the benefit of antifungal treatment in HIV-infected persons with CD4+ T cell counts between 100 and 200 cells/μL, and warrants further investigation.
No significant difference in all-cause mortality was found in our meta-analysis among CrAg+ persons who received pre-emptive antifungal therapy versus placebo or no intervention. This is a somewhat surprising outcome, and the reason of this may be associated with the discrepant sample sizes in these two groups (396 vs. 1092).
We considered the following possible reasons for clinical and methodological heterogeneity: discrepancies in follow-up time for reporting CM events and death events, variations in drug dosing, regimens, or drug class of prescribed antifungal therapy, ART status of subjects, and risk of bias. For example, the study durations ranged from104 weeks to 6 years, and the dosing of azole antifungal treatments ranged from 100 mg/d to 900 mg/d. With regards to reporting bias, it is possible that the unformed funnel plot for all-cause mortality could be a consequence of the varied ART status of study participants, different dosage regimens and duration of treatment and the different follow-up periods in each of the individual studies.
There are some limitations in our study. Firstly, the data supporting the association between prevalence of CrAg positivity and occurrence of adverse outcomes in HIV-infected persons with CD4+ T-cell counts between 100 and 200 cells cells/μL is sparse. Secondly, there exists a paucity of new data regarding CrAg positivity prevalence, CM incidence, and all-cause mortality in HIV-infected persons with CD4+ T-cell counts< 200 cells/μL since 2015 [27], and our pooled outcome analyses relied heavily on older studies, which may be less applicable to the modern test-and-treat era. And thirdly, the dosage and durations of azole therapy was not assessed in our meta-analysis. The preceding limitations may contribute to the clinical and methodological heterogeneity in our study.
Conclusions
In our meta-analysis, the incidence of CM was significantly reduced by pre-emptive antifungal therapy in CrAg+ persons with CD4+ T cell counts< 200 cells/μL. Nevertheless, more specific intervention data are needed in persons with CD4+ T cell counts between 101 ~ 200 cells/μL to better clarify the benefit of CrAg screening and pre-emptive antifungal treating in CrAg- persons with CD4+ T cell counts< 200 cells/μL more clear.
Supplementary information
Additional file 1: Figure S1. Incidence of CM and all-cause mortality among CrAg+ persons with and without antifungal therapy, (a) Incidence of CM among CrAg+ persons; (b) all-cause mortality among CrAg+ persons; (c) Incidence of CM among persons with antifungal therapy and without antifungal therapy (d); (e) all-cause mortality among persons with antifungal therapy and without antifungal therapy (f). Figure S2. Funnel plots. Funnel plots of the incidence of CM and all-cause mortality among patients with CD4 < 200 cells/μL. Table S1. Quality assessment of 8 included studies by using the JBI Critical Appraisal Checklist for Cohort Studies. Table S2. Risk of bias of the 1 included RCT.
Acknowledgements
Thanks to all authors for their contributions to this manuscript. We wish to thank Vijay Harypursat of Chongqing Public Health Medical Center for his help in language improvement to this manuscript.
Abbreviations
- CrAg
Cryptococcal antigen
- HIV
Human immunodeficiency virus
- CrAg+
CrAg positive
- RCTs
Randomized controlled studies
- RR
Risk ratio
- 95%CI
Confidence interval
- CrAg-
CrAg negative
- CM
Cryptococcal meningitis
- ART
Antiretroviral therapy
- WHO
World Health Organization
- PROSPERO
Prospective Register of Systematic Reviews (PROSPERO)
Authors’ contributions
YL, XJH, HW, XFY and YKC conceived and designed the study. YL, YYQ, JHH and AXL identified studies to be screened. XJH and HC identified studies for eligibility, extracted data, and assessed the methodological quality of included studies. YL performed the data analysis with assistance from XJH, and YKC. All authors read and approved the final manuscript.
Funding
This work was supported by the National Science and Technology Major Project of China during the 13th Five-year Plan Period (2018ZX10302104, 2017ZX10201101), key project of joint medical research project of science and health in Chongqing in 2019 (2019ZDXM012), Major Project of Beijing Municipal Science and Technology Committee (D161100000416003, D171100000517003).
Availability of data and materials
All the data and materials are available from Pubmed, Cochrane Library, MEDLINE/EMBASE and Web of Science.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
All authors declare that this research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yao Li and Xiaojie Huang contributed equally to this work.
Supplementary information
Supplementary information accompanies this paper at 10.1186/s12879-020-05126-z.
References
- 1.Govender NP, Roy M, Mendes JF, Zulu TG, Chiller TM, Karstaedt AS. Evaluation of screening and treatment of cryptococcal antigenaemia among HIV-infected persons in Soweto, South Africa. HIV Med. 2015;16(8):468–476. doi: 10.1111/hiv.12245. [DOI] [PubMed] [Google Scholar]
- 2.Longley N, Jarvis JN, Meintjes G, Boulle A, Cross A, Kelly N, et al. Cryptococcal antigen screening in persons initiating ART in South Africa: a prospective cohort study. Clin Infect Dis. 2016;62(5):581–587. doi: 10.1093/cid/civ936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rajasingham R, Smith RM, Park BJ, Jarvis JN, Govender NP, Chiller TM, et al. Global burden of disease of HIV-associated cryptococcal meningitis: an updated analysis. Lancet Infect Dis. 2017;17(8):873–881. doi: 10.1016/S1473-3099(17)30243-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.French N, Gray K, Watera C, Nakiyingi J, Lugada E, Moore M, et al. Cryptococcal infection in a cohort of HIV-1-infected Ugandan adults. AIDS. 2002;16(7):1031–1038. doi: 10.1097/00002030-200205030-00009. [DOI] [PubMed] [Google Scholar]
- 5.Larson BA, Rockers PC, Bonawitz R, Sriruttan C, Glencross DK, Cassim N, et al. Screening HIV-infected persons with low CD4 counts for cryptococcal antigenemia prior to initiation of antiretroviral therapy: cost effectiveness of alternative screening strategies in South Africa. PLoS One. 2016;11(7):e0158986. doi: 10.1371/journal.pone.0158986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Awotiwon AA, Johnson S, Rutherford GW, Meintjes G, Eshun-Wilson I. Primary antifungal prophylaxis for cryptococcal disease in HIV-positive people. Cochrane Database Syst Rev. 2018;2018(8):CD004773. doi: 10.1002/14651858.CD004773.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jarvis JN, Meintjes G, Williams A, Brown Y, Crede T, Harrison TS. Adult meningitis in a setting of high HIV and TB prevalence: findings from 4961 suspected cases. BMC Infect Dis. 2010;10:67. doi: 10.1186/1471-2334-10-67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sathyanarayanan V, Razak A, Chakraborty J. Clinical profile of disseminated cryptococcal infection – a case series. Asian Pacific Trop Med. 2010;3:818–820. doi: 10.1016/S1995-7645(10)60197-6. [DOI] [Google Scholar]
- 9.Kaplan JE, Vallabhaneni S, Smith RM, Chideya-chihota S, Chehab J, Park B. Cryptococcal antigen screening and early antifungal treatment to prevent cryptococcal meningitis: a review of the literature. J Acquir Immune Defic Syndr. 2015;68(Suppl 3):331–339. doi: 10.1097/QAI.0000000000000484. [DOI] [PubMed] [Google Scholar]
- 10.Beyene T, Woldeamanuel Y, Asrat D, Ayana G, Boulware DR. Comparison of cryptococcal antigenemia between antiretroviral naïve and antiretroviral experienced HIV positive persons at two hospitals in Ethiopia. PLoS One. 2013;8(10):e75585. doi: 10.1371/journal.pone.0075585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jarvis JN, Lawn SD, Vogt M, Bangani N, Wood R, Harrison TS. Screening for cryptococcal antigenemia in persons accessing an antiretroviral treatment program in South Africa. Clin Infect Dis. 2009;48(7):856–862. doi: 10.1086/597262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Meya D, Rajasingham R, Nalintya E, Tenforde M, Jarvis JN. Preventing cryptococcosis shifting the paradigm in the era of highly active antiretroviral therapy. Curr Trop Med Rep. 2015;2(2):81–89. doi: 10.1007/s40475-015-0045-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.World Health Organization. Guidelines for the diagnosis, prevention and management of Cryptococcal disease in HIV-infected adults, adolescents and children. 2018. http://www.who.int/hiv/pub/guidelines/cryptococcal-disease/en/. Accessed 30 Mar 2018. [PubMed]
- 14.The Joanna Briggs Institute . Joanna Briggs institute reviewers’ manual: 2016 edition. Australia: The Joanna Briggs Institute; 2016. [Google Scholar]
- 15.Higgins JP, Altman DG, Gotzsche PC, Jüni P, Moher D, Oxman AD, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. doi: 10.1136/bmj.d5928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Temfack E, Bigna JJ, Luma HN, Spijker R, Meintjes G, Jarvis JN, et al. Impact of routine cryptococcal antigen screening and targeted pre-emptive fluconazole therapy in antiretroviral naive HIV-infected adults with less than 100 CD4 cells/μL: a systematic review and meta-analysis. Clin Infect Dis. 2018. 10.1093/cid/ciy567. [DOI] [PubMed]
- 17.Huang Y, Huang X, Luo Y, Zhou Y, Tao X, Chen H, et al. Assessing the efficacy of lopinavir/ritonavir-based preferred and alternative second-line regimens in HIV-infected persons: a meta-analysis of key evidence to support WHO recommendations. Front Pharmacol. 2018;9:890. doi: 10.3389/fphar.2018.00890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chariyalertsak S, Supparatpinyo K, Sirisanthana T, Nelson KE. A controlled trial of itraconazole as primary prophylaxis for systemic fungal infections in persons with advanced human immunodeficiency virus infection in Thailand. Clin Infect Dis. 2002;34(2):277–284. doi: 10.1086/338154. [DOI] [PubMed] [Google Scholar]
- 19.Manfredi R, Mastroianni A, Coronado OV, Chiodo F. Fluconazole as prophylaxis against fungal infection in persons with advanced HIV infection. Arch Intern Med. 1997;157(1):64–69. doi: 10.1001/archinte.1997.00440220068009. [DOI] [PubMed] [Google Scholar]
- 20.Parkes-Ratanshi R, Wakeham K, Levin J, Namusoke D, Whitworth J, Coutinho A, et al. Primary prophylaxis of cryptococcal disease with fluconazole in HIV-positive Ugandan adults: a double-blind, randomised, placebo-controlled trial. Lancet Infect Dis. 2011;11:933–941. doi: 10.1016/S1473-3099(11)70245-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.McKinsey DS, Wheat LJ, Cloud GA, Pierce M, Black JR, Bamberger DM, et al. Itraconazole prophylaxis for fungal infections in persons with advanced human immunodeficiency virus infection: randomized, placebo-controlled, double-blind study. National Institute of Allergy and Infectious Diseases Mycoses Study Group. Clin Infect Dis. 1999;28(5):1049–1056. doi: 10.1086/514744. [DOI] [PubMed] [Google Scholar]
- 22.Meya DB, Manabe YC, Castelnuovo B, Cook BA, Elbireer AM, Kambugu A, et al. Cost-effectiveness of serum cryptococcal antigen screening to prevent deaths among HIV-infected persons with a CD4+ cell count < or = 100 cells/microL who start HIV therapy in resource-limited settings. Clin Infect Dis. 2010;51:448–455. doi: 10.1086/655143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kapoor SW, Magambo KA, Kalluvya SE, Fitzgerald DW, Peck RN, Downs JA. Six-month outcomes of HIV-infected persons given short-course fluconazole therapy for asymptomatic cryptococcal antigenemia. AIDS. 2015;29(18):2473–2478. doi: 10.1097/QAD.0000000000000872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Borges MASB, de Araújo Filho JA, Oliveira BJS, Moreira IS, de Paula VV, de Bastos AL, et al. Prospective cohort of AIDS patients screened for cryptococcal antigenaemia, pre-emptively treated and followed in Brazil. PLoS One. 2019;14(7):e0219928. doi: 10.1371/journal.pone.0219928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ssekitoleko R, Kamya MR, Reingold AL. Primary prophylaxis for cryptococcal meningitis and impact on mortality in HIV: a systematic review and meta-analysis. Future Virol. 2013;8(9). 10.2217/fvl.13.71. [DOI] [PMC free article] [PubMed]
- 26.Ford N, Shubber Z, Jarvis JN, Chiller T, Greene G, Migone C, et al. CD4 cell count threshold for cryptococcal antigen screening of HIV-infected individuals: a systematic review and meta-analysis. Clin Infect Dis. 2018;66(suppl_2):S152–S159. doi: 10.1093/cid/cix1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Guideline on when to start antiretroviral therapy and on pre-exposure prophylaxis for HIV. Geneva: World Health Organization. 2015. http://www.who.int/hiv/pub/guidelines/earlyrelease-arv/en. Accessed 29 Sept 2015. [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Figure S1. Incidence of CM and all-cause mortality among CrAg+ persons with and without antifungal therapy, (a) Incidence of CM among CrAg+ persons; (b) all-cause mortality among CrAg+ persons; (c) Incidence of CM among persons with antifungal therapy and without antifungal therapy (d); (e) all-cause mortality among persons with antifungal therapy and without antifungal therapy (f). Figure S2. Funnel plots. Funnel plots of the incidence of CM and all-cause mortality among patients with CD4 < 200 cells/μL. Table S1. Quality assessment of 8 included studies by using the JBI Critical Appraisal Checklist for Cohort Studies. Table S2. Risk of bias of the 1 included RCT.
Data Availability Statement
All the data and materials are available from Pubmed, Cochrane Library, MEDLINE/EMBASE and Web of Science.