Although most of the 4.7 million individuals infected to date with COVID-19 (SARS-CoV-2) worldwide remain asymptomatic or have transient symptoms followed by complete recovery, approximately 316 thousand (6.7%) have died [1], most often from acute respiratory distress syndrome (ARDS) leading to organ failure and death. The components that contribute to morbidity include forceful activation of both innate and adaptive immune pathways that release a cytokine storm which causes extensive damage to lung alveolar endothelium, oxygen insufficiency and downstream bystander damage to the heart, liver and kidneys. Recent publications predicted immunological processes anticipated in COVID-19 patients based on previous findings in severe acute respiratory syndrome (SARS), Middle East respiratory syndrome (MARS) and sepsis patients [2], [3], [4]. To reiterate, upon binding to the ACE2 (angiotensin-converting enzyme-2) receptor via its spike (S) protein, the SARS-CoV-2 virus infects alveolar epithelial cells, triggering the complement cascade and inducing fibrin deposition leading to platelet aggregation and macrophage activation resulting in metabolic changes in arachidonic acid with release of thromboxane A2 and secretion of IL-1β, IL-6, TNF-α, platelet activating factor & nitric oxide. In addition infection induces neutrophil activation, aggregation and degranulation with release of O2 radicals and proteases and CCR4+CCR6+Th17 cell activation with secretion of IL-2, IL-7, IFN-γ, G-CSF and chemokines CCL2, CCL3 and CXCl10. This focused collection of highly inflammatory factors in the lungs causes serious endothelial damage and respiratory failure. As well, ACE2 is expressed on heart, liver and kidney cells that may also be infected and gravely damaged by SARS-CoV-2, leading to fatal multi-organ failure.
In addition to current approaches such as glucocorticoids for treating advanced COVID-19 patients, there may be other drug combinations that could be helpful, some being bifunctional. As relevant examples reviewed in [5], pharmacological drugs for treatment of patients with asthma and/or chronic obstructive pulmonary disease (COPD) were developed to include both β2-agonists to relax airway smooth muscle constriction and muscarinic receptor antagonists to block M3 receptors. Another bifunctional drug was formulated to antagonize the receptors for platelet-activating factor and histamine, and mast cell secretion-blocking effects. A third anti-inflammatory drug combined an antagonist for the thromboxane receptor and cysteinyl-leukotriene antagonists into the same molecule. A bifunctional antibody construct was also developed for IL-4 and IL-13 to reduce the IL-4-dependent rise in serum IgE and IL-13-dependent airway hyperresponsiveness, lung inflammation, mucin gene expression and serum chitinase responses in mice. In this vein, bifunctional immunotherapeutic approaches could be developed for treatment of the cytokine storm in COVID-19 patients with ARDS at high risk of death. Proven and developing therapies that downregulate receptors for IL-1β, TNF-α, IL-6 and IL-2 as well as those which block macrophage migration inhibitory factor (MIF) and its homolog MIF2 through CD74 and potentially CXCR2&4 for MIF are all potential inhibitors of cytokine storm effects and could be combined or designed for bifunctional activity.
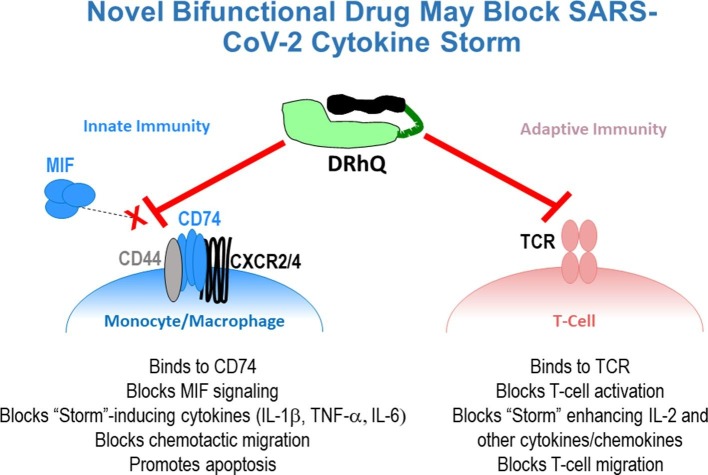
We are developing a second generation bifunctional drug called DRhQ that can simultaneously bind to and inhibit both the TCR and CD74 through distinct regions of the construct [6]. DRhQ is comprised of the HLA-DRα1 domain with an L50Q amino acid substitution (to enhance binding affinity for CD74) linked to an autoantigen peptide (myelin oligodendroglial cell glycoprotein, i.e. MOG-35-55 peptide) (Fig. 1 ). DRhQ was derived from soluble MHC Class II α1-β1-antigenic peptide constructs originally designed to ligate specific T cell receptors as the distal components of the trimolecular complex (called Recombinant TCR ligands – RTLs). As partial TCR agonists these constructs, containing various disease-associated MHC and antigenic peptide components, could indeed inhibit MHC-restricted antigen specific T cells, but translation of RTL1000 for human use in a Phase 1 clinical trial (showing safety and tolerability) required MHC-matched recipients [7]. Thus, the simpler DRhQ construct was designed, retaining just the conserved-in-human DRα1 domain (without the polymorphic HLA-DRβ1 domain) linked to the MOG-35-55 peptide extension, with the added benefit that it can be administered to all recipients without need for tissue type matching. This has enabled use of the DRα1-MOG-35-55 construct to reverse ongoing neuroinflammation and disease signs in animal models of multiple sclerosis, stroke, methamphetamine disorders and traumatic brain injury [8]. These and other studies ([9], [10], [11] & unpublished data) revealed down-regulation of multiple proinflammatory components driven by both innate and adaptive immune responses that also contribute to the SARS-CoV-2 cytokine storm, including complement receptor C5aR1, platelet activation, IL-1β, IL-2, IL-6, TNF-α, CCR2 (receptor for CCL2) and CXCR2. Of further importance, a partial HLA-DP RTL construct could inhibit activated pleural T cell infiltrates from patients with beryllium-induced lung fibroma [12], suggesting more-directly-relevant activity that could be potentially beneficial as a treatment of COVID-19 patients with ARDS.
Fig. 1.
Dual activities of DRhQ that could block the “Cytokine Storm” induced by SARS-CoV-2. DRhQ is a bifunctional drug comprised of the HLA-DRα1 domain covalently linked to human myelin oligodendroglial cell glycoprotein (MOG)-35-55 peptide. Due to its unique design, DRhQ can bind to and inhibit both T cell receptors and the MHC “invariant” chain, CD74, that serves as the receptor for the proinflammatory homologs, MIF and MIF2, resulting in blockade of multiple contributors to the Cytokine Storm. DRhQ image: Green = DRα1L50Q domain; Dark green line = linker; Black = MOG-35-55 peptide extension.
Development of new generation immunologically based cytokine storm inhibitors remains an important endeavor. Even if a successful vaccine is developed for COVID-19 patients, there will be ongoing mortality in vulnerable populations after vaccination as has been observed in flu virus and other current or future coronavirus infections as well as those with sepsis. In these conditions, resolution or chronicity of disease depends upon the strength of regulatory immune processes which appear to be compromised in COVID-19 patients that develop ARDS. To this end, the DRhQ construct could lend a helping hand.
Acknowledgments
Acknowledgement
The authors wish to thank Ashley Headrick for help in preparing and submitting the commentary article.
Funding
This work was funded by the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Biomedical Laboratory Research and Development Merit Review Awards 2I01 BX000226 and IMMA-010-19F (AAV), Senior Research Career Scientist Award 1IK6BX004209 (AAV) and the National Institute of Allergy and Infectious Diseases award R44AI122574 (AAV). The contents do not represent the views of the Department of Veterans Affairs or the US Government.
Availability of data and materials
The datasets used and/or analyzed during previous studies are available from the corresponding author on reasonable request.
Author contributions
The commentary article was written, edited and reviewed by AAV, HO and RMR. All authors read and approved the final article.
Competing Interests
Drs. Vandenbark, Offner, Meza-Romero, and OHSU have a significant financial interest in Artielle ImmunoTherapeutics, Inc., a company that may have a commercial interest in the results of this research and technology. This potential conflict of interest has been reviewed and managed by the OHSU and VA Portland Health Care System Conflict of Interest in Research Committees.
References
- 1.World Health Organization, Coronavirus Disease 2019 (COVID-19) A Situation Report—121. 2020, May 20. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports/.
- 2.Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet J. 2020;395(10229):1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Xie P., Ma W., Tang H., Liu D. Severe COVID-19: a review of recent progress with a look toward the future. Front. Public Health. 2020;8(189):1–7. doi: 10.3389/fpubh.2020.00189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.InvivoGen, Spotlight on COVID-19: Predicted immune responses. 2020, May 20. http://www.invivogen.com/spotlight-covid-19-predicted-immune-responses?utm_source=emailing-covid03&utm_medium=mail&utm_campaign=covid19-3-us&utm_content=img.
- 5.Page C., Cazzola M. Bifunctional drugs for the treatment of asthma and chronic obstructive pulmonary disease. Eur. Respir. J. 2014;44:475–482. doi: 10.1183/09031936.00003814. [DOI] [PubMed] [Google Scholar]
- 6.Meza-Romero R., Benedek G., Gerstner G., Kent G., Nguyen H., Offner H., Vandenbark A.A. Increased CD74 binding and EAE treatment efficacy of a modified DRα1 molecular construct. Metab. Brain Dis. 2019 Feb;34(1):153–164. doi: 10.1007/s11011-018-0331-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yadav V., Bourdette D.N., Bowen J.D., Lynch S.G., Mattson D., Preiningerova J., Bever C.T., Jr., Simon J., Goldstein A., Burrows G.G., Offner H., Ferro A.J., Vandenbark A.A. Recombinant T-cell receptor ligand (RTL) for treatment of multiple sclerosis: a phase I, dose-escalation study. Autoimmune Dis. 2012;954739:1–11. doi: 10.1155/2012/954739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Vandenbark A.A., Meza-Romero R., Benedek G., Offner H. A novel neurotherapeutic for multiple sclerosis, ischemic injury, methamphetamine addiction, and traumatic brain injury. J. Neuroinflammation. 2019;16:14. doi: 10.1186/s12974-018-1393-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Benedek G., Meza-Romero R., Jordan K., Zhang Y., Nguyen H., Kent G., Li J., Siu E., Frazer J., Piecychna M., Du X., Sreih A., Leng L., Wiedrick J., Caillier S.J., Offner H., Oksenberg J.R., Yadav V., Bourdette D., Bucala R., Vandenbark A.A. MIF and D-DT are potential disease severity modifiers in male MS subjects. Proc. Natl. Acad. Sci. U. S. A. 2017;114(40):E8421–E8429. doi: 10.1073/pnas.1712288114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sinha S., Subramanian S., Proctor T.M., Kaler L.J., Grafe M., Dahan R., Huan J., Vandenbark A.A., Burrows G.G., Offner H. A promising therapeutic approach for multiple sclerosis: recombinant TCR ligands modulate experimental autoimmune encephalomyelitis by reducing IL-17 production and inhibiting migration of encephalitogenic cells into the CNS. J. Neurosci. 2007;27(46):12531–12539. doi: 10.1523/JNEUROSCI.3599-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Itakura A., Aslan J.E., Sinha S., White-Adams T.C., Berny M.A., Vandenbark A.A., Burrows G.G., Offner H., McCarty O.J.T. Characterization of human platelet binding of recombinant T cell receptor ligand. J. Neuroinflammation. 2010;7:75. doi: 10.1186/1742-2094-7-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fontenot A.P., Keizer T.S., McCleskey M., Mack D.G., Meza-Romero R., Huan J., Edwards D.M., Chou Y.K., Vandenbark A.A., Scott B., Burrows G.G. Recombinant HLA-DP2 binds beryllium and tolerizes beryllium-specific pathogenic CD4+ T cells. J. Immunol. 2006;177(6):3874–3883. doi: 10.4049/jimmunol.177.6.3874. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during previous studies are available from the corresponding author on reasonable request.