Abstract
In late December 2019 an outbreak of a novel coronavirus (SARS-CoV-2) causing severe pneumonia (COVID-19) was reported in Wuhan, Hubei Province, China. A common finding in most COVID-19 patients is high D-dimer levels which are associated with a worse prognosis. We aimed to evaluate coagulation abnormalities via traditional tests and whole blood thromboelastometry profiles in a group of 22 (mean age 67 ± 8 years, M:F 20:2) consecutive patients admitted to the Intensive Care Unit of Padova University Hospital for acute respiratory failure due to COVID-19. Cases showed significantly higher fibrinogen and D-dimer plasma levels versus healthy controls ( p < 0.0001 in both comparisons). Interestingly enough, markedly hypercoagulable thromboelastometry profiles were observed in COVID-19 patients, as reflected by shorter Clot Formation Time (CFT) in INTEM ( p = 0.0002) and EXTEM ( p = 0.01) and higher Maximum Clot Firmness (MCF) in INTEM, EXTEM and FIBTEM ( p < 0.001 in all comparisons). In conclusion, COVID-19 patients with acute respiratory failure present a severe hypercoagulability rather than consumptive coagulopathy. Fibrin formation and polymerization may predispose to thrombosis and correlate with a worse outcome.
Keywords: thromboelastometry, fibrinogen/fibrin, thrombotic complications
Coronaviruses (CoVs) are enveloped, single-stranded ribonucleic acid viruses that usually infect birds, mammals, and humans. Human CoVs may cause respiratory, enteric, and neurological affections. 1 In late December 2019, a cluster of pneumonia cases of unknown cause was reported in Wuhan, Hubei Provence, China. Thereafter, many countries reported similar cases, leading to the discovery of a novel CoV. In February 2020, the World Health Organization named SARS-CoV-2 as the cause of the Coronavirus Disease 2019 (COVID-19). Early reports showed that very high D-dimer levels are common in COVID-19 pneumonia and correlate with a worse prognosis. 2 To better characterize COVID-19-related coagulation changes, we investigated traditional parameters and whole blood thromboelastometry profiles in all consecutive patients admitted to the intensive care unit (ICU) of Padua University Hospital between March 7 and 19 2020 for acute respiratory distress syndrome due to COVID-19. Exclusion criteria were: known preexisting congenital bleeding or thrombotic disorders and/or preexisting acquired coagulopathies, active cancer and/or chemotherapy, pregnancy, and ongoing anticoagulant therapy. Demographic characteristics, comorbidities, and Sequential Organ Failure Assessment score were recorded. A group of 44 healthy, age-, sex-, and body weight-matched subjects served as controls for laboratory data. Within 30 minutes of ICU admission, venous blood samples were drawn from each enrolled patient into two BD Vacutainer tubes (Becton–Dickinson, Franklin Lakes, New Jersey, United States) containing sodium citrate 109 mmol/L (3.8%) and one BD Vacutainer tube containing ethylenediaminetetraacetic acid 5.4 mg. In all enrolled patients, hemoglobin, platelet count, prothrombin time/international normalized ratio, activated partial thromboplastin time, fibrinogen, antithrombin, and D-dimer were measured. Whole blood thromboelastometry profiles were obtained using a ROTEM delta apparatus (Instrumentation Laboratory – Werfen, Barcelona, Spain), as previously described. 3 INTEM and EXTEM assays (evaluation of intrinsic and extrinsic coagulation pathways) and FIBTEM test (evaluation of fibrinogen contribution to blood clot) were performed in each enrolled patient. The following ROTEM parameters were analyzed: (1) clotting time corresponding to the initiation phase of the clotting process; (2) clot formation time (CFT) reflects the measure of the propagation phase of whole blood clot formation; (3) maximum clot firmness (MCF) is the maximum amplitude in millimeters reached in thromboelastogram; and (4) area under the curve (mm*100), defined as the area under the velocity curve, that is the area under the first derivative curve ending at a time point that corresponds to MCF. 4 The protocol was conducted in compliance with the Helsinki Declaration and notified to the Institutional Ethical Committee of Padua University Hospital. Written informed consent was obtained from each patient when possible or relatives and from each control. Statistical analysis was performed using the PASW Statistics 17.0.2 (SPSS Inc.) for Windows. Continuous variables were expressed as mean ± standard deviation and categorical variables as number and fraction (%). The parametric t -Student's test or the nonparametric Mann–Whitney U was used to test for differences between variables, when appropriate. A p -value of < 0.05 was considered significant.
Among 30 eligible patients, 8 were excluded: active cancer ( n = 4), anticoagulant therapy ( n = 3), and pregnancy ( n = 1). Table 1 shows the main clinical and laboratory characteristics of the study population. The vast majority of COVID-19 patients were male (91%), obese (86%), with a PaO 2 /FiO 2 < 150 (91%) at admission, and mechanically ventilated (86%) (tidal volume 6 mL × ideal body weight and driving pressure < 12 cm H 2 O). Fibrinogen and D-dimer plasma levels were significantly higher in COVID-19 patients than controls ( p < 0.0001 in both comparisons). Cases showed markedly hypercoagulable ROTEM profiles characterized by significantly shorter CFT in INTEM ( p = 0.0002) and EXTEM ( p = 0.01) and by a higher MCF in INTEM, EXTEM, and FIBTEM ( p < 0.001 in all comparisons). One patient died of multiorgan failure at day +1 after enrolment and presented with the most hypercoagulable ROTEM profiles ( Fig. 1 ).
Table 1. Patient characteristics and laboratory data.
| Cases | Controls | p -Value | |
|---|---|---|---|
| Patients, n | 22 | 44 | – |
| Gender M/F, n | 20/2 | 40/4 | – |
| Age, y | 67 ± 8 | 68 ± 7 | 0.63 |
| BMI, kg/m 2 | 30 ± 6 | 29 ± 4 | 0.49 |
| SOFA score | 4 ± 2 | – | – |
| PT, % | 93 ± 10 | 91 ± 10 | 0.46 |
| INR | 1.08 ± 0.06 | 1.09 ± 0.06 | 0.54 |
| aPTT, s | 26 ± 12 | 26 ± 2 | 0.95 |
| Fibrinogen, mg/dL | 517 ± 148 | 297 ± 78 | < 0.0001 |
| Antithrombin, % | 96 ± 13 | 90 ± 14 | 0.1 |
| D-dimer, ng/L | 5,343 ± 2,099 | 225 ± 158 | < 0.0001 |
| Hb, g/L | 121 ± 16 | 138 ± 15 | 0.0002 |
| Htc, % | 38 ± 4 | 41 ± 4 | 0.008 |
| Plts, ×10 9 /L | 249 ± 119 | 218 ± 67 | 0.27 |
| INTEM | |||
| CT, s | 185 ± 49 | 174 ± 23 | 0.33 |
| CFT, s | 57 ± 13 | 70 ± 18 | 0.0002 |
| MCF, mm | 68 ± 6 | 62 ± 7 | < 0.0001 |
| ML, % (range) | 1 ± 3 | 2 ± 3 | 0.22 |
| AUC, U | 6,808 ± 603 | 6,743 ± 563 | 0.68 |
| EXTEM | |||
| CT, s | 75 ± 16 | 72 ± 8 | 0.11 |
| CFT, s | 66 ± 20 | 78 ± 26 | 0.01 |
| MCF, mm | 69 ± 6 | 64 ± 5 | 0.0003 |
| ML, % (range) | 1 ± 3 | 2 ± 3 | 0.22 |
| AUC, U | 6,924 ± 591 | 6,882 ± 569 | 0.79 |
| FIBTEM | |||
| MCF, mm | 31 ± 9 | 18 ± 6 | < 0.0001 |
| AUC, U | 3,101 ± 852 | 2,249 ± 1072 | 0.001 |
Abbreviations: aPTT, activated partial thromboplastin time; AUC, area under the curve; BMI, body mass index; CFT, clot formation time; CT, clotting time; Hb, hemoglobin; Htc, hematocrit; INR, international normalized ratio; MCF, maximum clot firmness; ML, maximum lysis; Plts, platelet count; PT, prothrombin time; SOFA, Sequential Organ Failure Assessment.
Note: p -Values marked in bold are statistically significant.
Fig. 1.
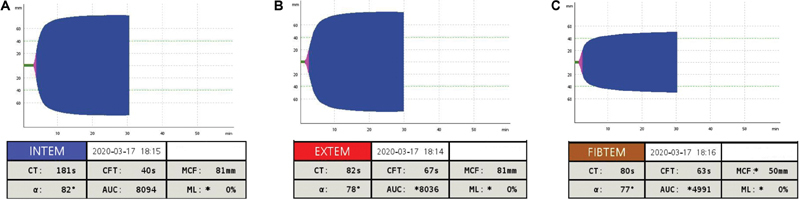
Thromboelastometry profiles of the deceased patient. ( A ) INTEM test; ( B ) EXTEM test; and ( C ) FIBTEM test. ɑ, ɑ-angle; AUC, area under the curve; CFT, clot formation time; CT, clotting time; MCF, maximum clot firmness.
Coagulation profiles observed in our study population reflect a severe hypercoagulability rather than a consumptive coagulopathy (e.g., disseminated intravascular coagulation). Such a laboratory pattern and association can be linked to both markedly increased levels of fibrinogen and an excessive fibrin polymerization due to the infection. SARS-CoV-2 is likely to promote massive fibrin formation and deposition which can also account for the very high D-dimer levels found in these patients. 2 Fibrin deposition in alveolar and interstitial lung spaces, in addition to microcirculation thrombosis, 5 may contribute to worsen respiratory failure resulting in prolonged mechanical ventilation, poor prognosis, and death. Furthermore, other major venous thromboembolic events and arterial complications (e.g., acute myocardial infarction) have been reported 6 and are likely to be largely underestimated. Notably, 5 (23%) of our patients developed an in-hospital deep vein thrombosis despite anticoagulant prophylaxis. In this regard, anticoagulant therapy may improve the prognosis in COVID-19 patients as reported by Tang et al. 7 In light of the severe hypercoagulable state observed in these patients, effective anticoagulant prophylaxis should be considered to reduce the risk of thrombotic complications. Unfortunately, our data did not allow to assess the impact of adequate dosages of anticoagulants on clotting parameters. Measuring antifactor Xa (anti-Xa) activity in plasma was deemed as the most accurate way to monitor therapeutic dosing of low molecular weight heparin (LMWH, the anticoagulant used in our study). However, we were not able to evaluate anti-Xa activity in our study as it should have been measured 4 to 6 hours after the last injection of LMWH (peak level) whereas we collected blood samples immediately after admission to the ICU and thus at varying times from the last administration of heparin. It bears noting that if on the one hand thromboelastometry has the advantage to provide a global assessment of whole blood's ability to clot, on the other hand it is not able to evaluate the contribution to clot formation of each element (e.g., endothelium, platelets, and clotting factors). In conclusion, COVID-19 patients with acute respiratory failure present with severe hypercoagulability due to hyperfibrinogenemia resulting in increased fibrin formation and polymerization that may predispose to thrombosis. Larger studies are needed to define new therapeutic strategies to limit hypercoagulability and improve outcomes.
Acknowledgment
The authors want to dedicate their study to all the Italian doctors, nurses, paramedics, and other volunteers who are fighting COVID-19.
Conflict of Interest None declared.
Authors' Contributions
L.S. and A.B. study design, data analysis, and drafting; A.B., F.P., L.C., and I.T. patient enrolment; L.S. and E.C. performed laboratory tests; P.N. and P.S. study supervision and final approval.
References
- 1.Singh S K. Middle East respiratory syndrome virus pathogenesis. Semin Respir Crit Care Med. 2016;37(04):572–577. doi: 10.1055/s-0036-1584796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18(04):844–847. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Spiezia L, Radu C, Marchioro P et al. Peculiar whole blood rotation thromboelastometry (Rotem) profile in 40 sideropenic anaemia patients. Thromb Haemost. 2008;100(06):1106–1110. [PubMed] [Google Scholar]
- 4.Sørensen B, Johansen P, Christiansen K, Woelke M, Ingerslev J. Whole blood coagulation thrombelastographic profiles employing minimal tissue factor activation. J Thromb Haemost. 2003;1(03):551–558. doi: 10.1046/j.1538-7836.2003.00075.x. [DOI] [PubMed] [Google Scholar]
- 5.Chen S, Huang B, Luo D J et al. Pregnant women with new coronavirus infection: a clinical characteristics and placental pathological analysis of three cases [in Chinese] Zhonghua Bing Li Xue Za Zhi. 2020;49(00):E005. doi: 10.3760/cma.j.cn112151-20200225-00138. [DOI] [PubMed] [Google Scholar]
- 6.Zhou F, Yu T, Du Ret al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study Lancet 2020pii: S0140–6736(20)30566–3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tang N, Bai H, Chen X, Gong J, Li D, Sun Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost. 2020;18(05):1094–1099. doi: 10.1111/jth.14817. [DOI] [PMC free article] [PubMed] [Google Scholar]


