Abstract
The global preparedness and response to the rapid escalation to severe acute respiratory syndrome coronavirus (SARS-CoV)-2-related disease (COVID-19) to a pandemic proportion has demanded the formulation of a reliable, useful and evidence-based mechanism for health services prioritisation, to achieve the highest quality standards of care to all patients. The prioritisation of high value cancer interventions must be embedded in the agenda for the pandemic response, ensuring that no inconsistency or discrepancy emerge in the health planning processes.
The aim of this work is to organise health interventions for breast cancer management and research in a tiered framework (high, medium, low value), formulating a scheme of prioritisation per clinical cogency and intrinsic value or magnitude of benefit. The public health tools and schemes for priority setting in oncology have been used as models, aspiring to capture clinical urgency, value in healthcare, community goals and fairness, while respecting the principles of benevolence, non-maleficence, autonomy and justice.
We discuss the priority health interventions across the cancer continuum, giving a perspective on the role and meaning to maintain some services (undeferrable) while temporarily abrogate some others (deferrable). Considerations for implementation and the essential link to pre-existing health services, especially primary healthcare, are addressed, outlining a framework for the development of effective and functional services, such as telemedicine.
The discussion covers the theme of health systems strategising, and why oncology care, in particular breast cancer care, should be maintained in parallel to pandemic control measures, providing a pragmatic clinical model within the broader context of public healthcare schemes.
Keywords: breast cancer, ESMO adapted recommendations, COVID-19
Introduction
The global preparedness and response to the rapid escalation of severe acute respiratory syndrome coronavirus (SARS-CoV)-2-related disease (COVID-19) at a pandemic proportion has demanded the prompt development of a reliable, applicable and evidence-informed mechanism for the priority-setting of health services, to assure the attainment of the highest standards of quality care to all.1 While the response strategy to the global spread of COVID-19 must be immediately integrated into public health policy, these actions require non-COVID-19 health priorities to be urgently identified and defined to reshape health systems, which are struggling to meet the health demands in view of the rapid spread of the pandemic.2
In oncology, the definition of essential services must be developed within a value framework for organisational and clinical decision making—as the clinical response to COVID-19 should never be developed a latere, but integrated in the multisectoral approach, to pursue overarching community goals, while preserving the perspectives of patient-centred care.3 No response from the oncology community should be disconnected from the broader healthcare priority setting mechanisms. A horizontal approach is envisioned, engaging with the continuum of care and embracing the primary healthcare sector to guide patients with cancer at home, including effectively developing telemedicine systems. Globalisation has propelled the rapid spread of the disease, inundating healthcare systems, in particular, intensive healthcare capacities, thus necessitating the abrogation of some health services of deferrable priority as a key strategic approach. Therefore, integrated healthcare planning is the bedrock of success in the preparation and response to COVID-19—including oncology.4
The need for value-based priority setting mechanisms has been envisioned by the global agencies for public health, especially when planning in health environments with impaired availability or accessibility to essential health resources, including epidemic outbreaks and disruptions of procurement chains and intermittent health service delivery—one of the global dividend of equitable quality care between well-resourced and resource-constrained settings.5 In the era of COVID-19-related constraints and the need to meet the goals of population health against threats that challenge the resilience of the health systems as we know them, the development of clinical guidelines must embrace an evidence-based mechanism to make choice, thus refusing the easy ways of perspectives or opinions approached per silos. The key to succeed in the management of cancer, when resources are limited or impaired by extraordinary events, is to define a methodology and create a value-enhanced framework with the most salient cancer interventions—attaining the principles of benevolence, non-maleficence, autonomy and justice.6
Contemporary oncology research and care models are sophisticated and resource intensive in many countries. However, the necessary restrictions posed by the public health interventions in the pandemic control demand a practical framework for resource prioritisation, maintaining high standards of quality cancer care while protecting patients from health-related financial distress. Patients with cancer have been suggested as a more fragile population, namely more vulnerable to SARS-CoV-2 infection, reporting poorer outcomes from COVID-19 for multicomorbid and/or older patients—cancer history positioning as a possible independent prognostic risk factor.7 However, a general under-representation of patients with cancer in the mixed series published to date limits any articulated dissertation.
In such an uncertain area of healthcare, the knowledge of protecting the vulnerable defines the needs for research implementation, addressing the spectrum of non-communicable diseases including cancer, as mandated by WHO in strategising the response to COVID-19 in the operational guidance for maintaining essential health services during an outbreak. The mandate is to ensure the essential services to all, prioritising per value.8
The trajectory of the present work aligns in parallel with the WHO indications to ‘redirect chronic disease management to focus on maintaining supply chains for medications and needed supplies, with a reduction in provider encounters’—approaching across the continuum of care and shaping a multidisciplinary dialogue across societies and diverse stakeholders.8
Methodology for the selection of priority interventions
The present manuscript is the result of an international panel of expert health providers in the management of breast cancer and is proposed to guide healthcare professionals treating patients with breast cancer during the COVID-19 pandemic. The expert consensus-based recommendations are not intended to replace the current guidelines but rather adapt breast cancer care during the COVID-19 pandemic, using a value-based framework to set priorities. All the adaptations and prioritisations have been formulated by the experts via teleconferences and e-mails discussions.
In the aim to provide a framework for the response of the medical community to COVID-19, the European Society for Medical Oncology (ESMO) has established a guidance for clinicians, defining three levels of priorities regarding medical interventions, namely: tier 1 (high priority intervention), tier 2 (intermediate priority) and tier 3 (low priority)—informed by the Ontario Health Cancer Care Ontario framework of resource-prioritisation and by the ESMO Magnitude of Clinical Benefit Scale (MCBS), a public health tool intended to support the uptake of medical interventions in oncology.9., 10.
Overall, the prioritisation has been developed to incorporate both the information on the value-based prioritisation and clinical cogency of the interventions (figure 1 ).
Figure 1.
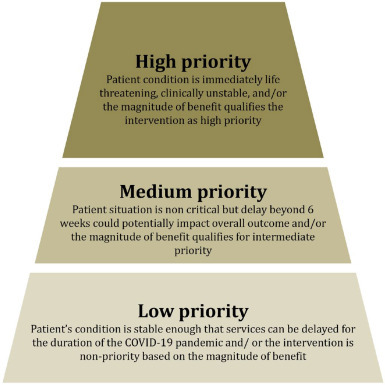
Priority-setting of the health interventions in oncology during COVID-19.
-
•
Tier 1 (high priority): patient’s condition is immediately life-threatening, clinically unstable and/or the magnitude of benefit qualifies the intervention as high priority (eg, significant overall survival (OS) gain and/or substantial improvement of the quality of life (QoL)).
-
•
Tier 2 (medium priority): patient’s situation is non-critical but delay beyond 6–8 weeks could potentially impact overall outcome and/or the magnitude of benefit qualifies for intermediate priority.
-
•
Tier 3 (low priority): patient’s condition is stable enough allowing services to delay for the duration of the COVID-19 pandemic and/or the intervention is non-priority based on the magnitude of benefit (eg, no survival gain with no change or reduced QoL). Of note, some interventions included in the tier 3 and considered safely deferrable should be re-discussed after 6–8 weeks, to re-assess the priority and deliberate on the clinical cogency.
The clinical guidance defined by ESMO must be interpreted in the broader context of healthcare response to the pandemic, and always linked to the Global Norms of WHO, the lead public health agencies and health technical governmental boards, for the definition of the strategies for the preparedness and response on populations—including the interventions to ensure the safest conditions for the health workforce, the proper provision of personal protective equipment, the testing strategy for healthcare personnel, patients and communities. Inconsistencies of clinical guidelines developed outside the global strategy and not in coordination with the strategic population policies of pandemic control will inevitably harm communities, with the earliest impact being on the most vulnerable patients—patients with cancer being first among them.11
Priorities for the management of breast cancer: reorganising the outpatient setting and sharing decisions
Meeting the community goals and perspectives of physical distancing and the overall public health measures for the mitigation of SARS-CoV-2 spread may necessitate a reinforcement and reorganisation of the outpatient setting. Triaging patients for fever and COVID-19-related symptoms is mandatory and an entry checkpoint should be considered by all healthcare facilities.
In the outpatient setting, postsurgical haematoma and infectious complications of surgical wounds may prompt ambulatory clinical interventions, like a drainage or an incision, to reduce the burden of serious, but preventable complications from cancer interventions, applying the priority criteria of clinical cogency.
Prioritisation is guided by magnitude of benefit, tumour biology and stage together with clinical scenarios. Quality of care should remain unchanged for the prioritised interventions. For example, for the treatment planning of all patients with cancer, a multidisciplinary board discussion must be assured, as retaining a major prognostic significance: thus while the format may change (eg, videoconferencing), the principle of multidisciplinary care is non-negotiable12 (table 1 ).
Table 1.
Outpatient visit priorities for the management of breast cancer
| High priority | Medium priority | Low priority |
|---|---|---|
| Postoperative unstable clinical scenario (eg, haematoma, infection) | New diagnosis of non-invasive cancer. Convert as many visits as possible to telemedicine visits. | Established patients with no new issues: refer to telemedicine |
| New diagnosis of invasive breast cancer (for multidisciplinary tumour board discussion: biology and stage will drive priority) | Survivorship follow-up: refer to telemedicine | |
| Breast cancer diagnosis during pregnancy | Postoperative visits in patients with no complications | Follow-up for patients at high risk of breast cancer (BRCA carriers, etc) or patients at high risk of relapse |
| On-treatment patients with new symptoms or side effects (depending on severity of symptoms/side effects, burden of progression, etc). Convert as many visits as possible to telemedicine visits. Intensify safety monitoring for those patients on oral chemotherapy or endocrine therapy plus biological agents | Psychological support visits (convert to telemedicine) | |
Visits to hospital should be minimised and if necessary, all protective measures should be taken (eg, physical distancing recommendations and use of masks). Also, there should be a triage for possible symptoms of COVID-19 prior to enter into the hospital premises. The use of home-based services should be considered as much as possible while hospital procedures are still indicated for patients with cancer where delaying treatment may compromise cancer prognosis. For specific conditions resulting from treatment-related toxicities, such as febrile neutropenia13 with clinical and anamnestic adverse prognostic factors, a rapid clinical intervention can make a difference in the prognosis as well as optimise the treatment delivery plan, thus must be included in the priorities.14
Accordingly, the safety monitoring of patients receiving oral treatments (eg, biological agents, endocrine agents and chemotherapeutics) should be organised through a quality system of telemedicine, assuring the requirements for a patient-centred connection with the oncologists and the cancer nurses. All the non-priority outpatient visits may be shifted to the telemedicine platform. The telemedicine delivery is best suited to non-urgent situations for established patients with no new complaints as well as survivorship and follow-up care of patients with cancer and of people referred to high-risk clinics (eg, BRCA carriers) for preventive interventions. In asymptomatic patients, follow-up exams can be delayed unless there are symptomatic clinically emergent conditions.
The patient-centred model of care requires addressing all of a patients care needs and as such, psychosocial supports must be assured and may be converted to telemedicine or other web-based platforms.
Although telemedicine has been recognised as an efficient delivery platform for deferrable healthcare services, limiting factors must be considered. Aspects of legal frameworks and data protection regulations, along with the management of the workforce for tele-health are of prominent interest. The consensus recognises the multiple legal and ethical issues, especially in the area of patient privacy and confidentiality—fostering the engagement with the competent vertices for healthcare professionals, healthcare facilities and academic institutions, communities, health managers and policy makers—to develop a socially accountable infrastructure of cancer service delivery. A public health approach is essentially endorsed, as the implementation of new service delivery models cannot be framed under a pure clinical indication but approached by multiple competent stakeholders. In principle, telemedicine is envisioned as part of the clinical service of the health personnel, thus accounted entirely in the clinical workload. Eventually, although beyond the scope of the present work, sustainability, financing, reimbursability and health impact of telemedicine must be accountable, on the longer term, to inspire a durable set-up of innovative paradigms of healthcare via telehealth applications. From this angle, an assessment of the baseline capacity and interim registration of the efficiency of telemedicine should orient the entire process of capacity building and, if appropriate, prompt the establishment of the programmes. To date, the telemedicine model is interpreted across several applications and web-based platforms, within the national and local perimeters of legal frameworks—variable across the settings, and a definite optimum has not been defined in oncology, as data are still warranted. While the efficiency of the prescription of multiple treatment cycles (eg, several months of endocrine therapy and target agents for patients with metastatic breast cancer) to reduce the healthcare admissions is recognised, it may be critical to intersect the cancer institution activity in a service delivery model based on a primary healthcare approach—engaging with community-based healthcare providers, including general practitioners and nurses.14 Overall, in the context of oncology care, telemedicine is indeed a mechanism to rethink the intersection of cancer care within the existing healthcare system. While providing clinical recommendations for telemedicine, all the key elements of the healthcare contexture are to be included in the discussion, to ensure that the clinical guidance is truly offering a valid alternative option for patients and not an ideal structure unfeasible on the pre-existing community services.
Of note, it is of utmost importance that all medical and strategic decisions regarding a patient’s care programme are made with careful consideration by a multidisciplinary team and shared with the patient. An informed consent process needs to be performed ensuring that the patient fully understand the risks and benefits she/he can expect from any medical intervention in the context of the current public health crisis, with all its limitations.
Priorities for the radiological and pathological diagnosis of breast cancer
Understanding the implications of the delay in diagnosis and access to treatment of breast cancer cannot be entirely captured unless contextualised to the biology of the cancer and patterns of clinical presentation, for example, the stage and the setting of care.15 All patients presenting with a new breast lump with high suspicion of malignancy or who have already undergone a screening procedure with an imaging finding highly suspicious for malignancy (eg, BIRADS 5 at mammography) should be promptly referred for tissue diagnosis and imaging and pathology are to be designated as high priority (table 2 ).
Table 2.
Priorities for breast diseases: diagnostics and imaging
| High priority | Medium priority | Low priority |
|---|---|---|
| Self-diagnosis of breast lump or other symptoms suggestive of malignancy | Further diagnostic imaging for BIRADS 4 screening mammogram in asymptomatic subjects. | Mammography-based population screening and risk-adapted breast screening programmes for asymptomatic subjects (eg, MRI or ultrasound). |
| Clinical evidence of locoregional relapse with surgical radical approach feasible (according to stage, histology and biological features of the disease) | Image-guided or clinically guided biopsy to ascertain a suspect of metastatic relapse. | Patients with abnormal findings at screening mammograms who can go to 6-month interval imaging (BIRADS 3). |
| Pathology assessment (histopathology or cytopathology) for abnormal mammograms or breast symptoms or a symptomatic metastatic relapse | Initial metastatic workup (according to stage and biological features) in patients with early stage invasive breast cancer. | In patients with early stage breast cancer, follow-up imaging, restaging studies, echocardiograms, ECGs and bone density scans can be delayed if clinically asymptomatic. |
| In patients with metastatic breast cancer, we recommend symptoms-oriented follow-up. Imaging, restaging studies, echocardiograms and ECGs can be delayed or done at lengthened intervals. Implement telemedicine follow-up. | ||
| Further diagnostic imaging for BIRADS 5 screening mammogram in asymptomatic subjects | Echocardiograms in patients with early stage invasive breast cancer requiring with indication to anthracycline-based or anti-HER2 treatment. |
BIRADS, Breast Imaging-Reporting and Data System.
Similarly, patients experiencing locoregional disease recurrence of breast cancer can still pursue a radical intention for the treatment—with a combination of locoregional treatments, either surgery and/or radiation therapy, and complemented with risk-reducing medical therapies: targeted, endocrine or chemotherapy.16., 17., 18. In this setting, it becomes essential to share the clinical decision, informed by the primary intention of the approach. Although often complex across a spectrum of clinical presentations, some disease recurrence patterns are more prone to radical approaches and, long-lasting disease-free intervals can be expected. Thus, based on clinical and pathological criteria, priority must be based on the intention of the care to identify the undeferrable cases, always in a multidisciplinary assessment.
Overall, histopathology diagnosis can have an immediate impact and sometimes cannot be deferred to complete the differential diagnosis. For patients with symptomatic metastatic relapse, whenever the provision of a treatment can be life-saving and/or significantly modify the quality of life, a histopathology diagnosis will be included in the set of undeferrable health services.
Besides the primary treatment intentions, the safe delivery and safety monitoring of treatments may require specific imaging techniques. Commonly, patients under treatment with anthracyclines or anti-HER2 agents require a regular assessment of cardiac function, prompting treatment interruptions, dose-reductions or cardio-oncology interventions, where indicated.19 Similarly, the ECG assessment for potentially arrhythmogenic changes, including the QT tract prolongation, should be considered in selected patients receiving some biological agents or carrying specific comorbidities.20 Overall, the cardio-oncology consultation could be reshaped in a risk-adapted scheme, adapting the monitoring intensity to the baseline risk of cardiac adverse events and the treatments received.
During the pandemic outbreak, mammography-based screening for breast cancer in asymptomatic women should be temporarily abrogated, as the risk-benefit ratio (risk of infection related to healthcare admissions for second-level procedures vs lives saved by screening) is not predicted to be high during the pandemic and appointments may be postponed by a few weeks or months without disproportionate predicted harms. Similarly, imaging for follow-up of patients with early breast cancer and metastatic patients may be delayed if necessary, with tailoring of radiological assessments per symptoms or other suspicion of progression, as clinically assessed—while still preferring telemedicine for non-priority complains and interventions.21
Priorities for breast surgical oncology
Cancer surgery is one of the cancer interventions bringing the greatest benefits for patients with cancer, principally in the curative setting, resulting in gains in cancer survival.22 The priority-setting framework of breast cancer surgery aims to understand how reasonably and for how long cancer surgery can be delayed, assuring the attainment of the highest standards for quality care delivery. Once again, the question must take into account cancer biology, clinical presentation pattern, patients’ conditions and preference (table 3 ).23
Table 3.
Priorities for breast disease: surgical oncology
| High priority | Medium priority | Low priority |
|---|---|---|
| Breast cancer surgery complication with bleeding or indication to incision and drainage of a breast abscess and/or haematoma | Clinically low-risk primary breast cancer (eg, stage I/II ER-positive/PR-positive/HER2-negative, low-grade/low proliferative index tumours). After multidisciplinary tumour board discussion consider starting neoadjuvant/preoperative endocrine therapy according to menopausal status and delay surgery. | Excision of benign lesions and duct excision (fibroadenomas, atypia, papillomas). |
| Complications of reconstructing surgery (eg, ischaemia) | Discordant biopsies likely to be malignant. | Surgery of non-invasive breast cancer (in situ) except for extended high-grade DCIS. |
| Surgery in patients who have completed neoadjuvant chemotherapy-based treatment (or, in exceptional cases, with progression of disease during neoadjuvant treatment) | - | Discordant biopsies likely to be benign. |
| - | - | Immediate or delayed breast reconstruction with autologous tissue and/or implants. |
| Surgery in patients with invasive cancer for whom multidisciplinary tumour board may decide, case-by-case, to proceed with upfront surgery | - | Prophylactic surgery for asymptomatic high-risk patients. |
| Excision of malignant recurrence (depending on phenotype and extent) | ||
| Breast cancer surgery during pregnancy (multidisciplinary treatment should be individualised according to stage and biology) | - | - |
DCIS, ductal carcinoma in situ.
The clinical cogency for intervention is dictated by the risk for serious complications or irreversible health conditions, like for postsurgical breast cancer complications. Where relatively simple surgical interventions can determine a change in the natural process of a potentially adverse pathological condition at rapid evolution, it represents a surgical priority. This also includes surgery complications demanding haemostatic procedures, incision, drainage or packing material insertion and certain wound dressing. However, relatively more complex procedures can sometimes still retain a priority, when inaction is associated with potentially fatal outcomes (eg, for the delay of adjuvant treatments).24 Several respected professional societies have proposed classifications of urgency/benefit/postponability.25 26
Based on the magnitude of benefit criteria, patients completing neoadjuvant chemotherapy-based treatments or, less commonly, progressing during such treatments, should receive curative surgery with no postponement. In situations in which this is absolutely impossible in terms of crisis healthcare resources, adding another cycle of neoadjuvant therapy in patients responding well to therapy may ‘protect’ the patient from the postponement of surgery.
Primary surgery of low-risk early breast cancer can safely be postponed up to 12 weeks, based on experts’ opinion, although no firm data exist, unless very aggressive tumour biology is present. In latter situations, neoadjuvant treatment approaches may be considered anyway. For luminal-like breast cancers, using appropriate preoperative endocrine treatment might be an option for avoiding harm due to the delay of surgery in selected cases. The psychological strain for patients who experience delays of their surgical procedures must not be underestimated, and appropriate psychological support offered.
Surgery for in situ ductal carcinomas could be transiently deferred in some cases, and the use of endocrine therapy if hormone receptor positive may be considered. Reconstructive procedures can/should be postponed, also because their perioperative morbidity risk is higher than that of standard breast surgery.27
As discussed above, patients experiencing locoregional relapse should be discussed in a multidisciplinary setting to weigh the potential benefits of offering an immediate radical surgical excision with subsequent durable survival gain versus postponing surgery after a primary systemic treatment.
As for all the other clinical presentations, the discussion about surgical indications must be individualised. Of note, all decisions must be shared with the patients, assessing the preference and expectations while informing on the threats and advantages of the adapted treatment plans in the context of the COVID-19 crisis.
Priorities for breast radiation oncology
The indications for radiation therapy in breast oncology are wide, and must be interpreted in the multidisciplinary care settings. According to clinical prioritisation, immediate radiation therapy should be initiated, in accordance with current clinical practice, in patients with acute spinal cord compression, symptomatic brain metastases not improving with steroidal medication and any urgent irradiation with an expected impact in survival or a modifying effect on the risk of disabling sequelae and/or quality of life (table 4 ). While the control of breast mass-related symptoms for fungating or bleeding neoplasia can usually be obtained with supportive care interventions, including advanced wound dressings, there may exist a need to provide surgery or radiation therapy for haemostasis or to alleviate pain due to inoperable breast masses for improving the quality of life for patients with advanced disease.
Table 4.
Priorities for breast cancer: radiation oncology
| High priority | Medium priority | Low priority |
|---|---|---|
| Palliative treatment of acute spinal cord compression, symptomatic brain metastases, bleeding/painful inoperable breast masses or any urgent palliative radiation therapy, when control of symptoms cannot be achieved pharmacologically | Adjuvant postoperative radiation therapy for low-risk/intermediate-risk patients with breast cancer (aged <65 years and stage I/II luminal cancer, ER positive regardless of nodal status or positive margins). Use of hypofractionated regimens should be considered to reduce hospital visits. Endocrine therapy can be started during the waiting interval. | Elderly patients with low-risk breast cancer (aged >70 years, with low-risk stage I ER-positive/HER2-negative breast cancer): starting adjuvant endocrine therapy is recommended while postponing radiation therapy. |
| Carcinoma in situ. | ||
| Patients already on radiation treatment | - | - |
| Adjuvant postoperative radiation therapy for high-risk patients with breast cancer (inflammatory disease at diagnosis, node-positive disease, triple-negative or HER2-positive breast cancer, residual disease at surgery postneoadjuvant therapy, young age (<40 years) | - | - |
The optimisation of locoregional control and the improvement of survival define the priority of the interventions in radiation therapy. Thus, post-operative radiation therapy for high-risk patients (eg, inflammatory breast cancer, node-positive or high-risk biology) should be scheduled as high priority, respecting the highest standards of quality for radiation therapy when proposing alternative (shorter) radiation regimens.28 Most patients are eligible for short-course treatments using hypo-fractionated schedules, being the treatment of choice. Accelerated partial breast irradiation should be proposed for low-risk patients, when indicated and technically feasible, given available technology and the capacity of the centres. Also for this, shorter treatment courses should be favoured, including single dose intra-operative electron radiation therapy or up to 5 fractions of preferably external beam radiation therapy and not brachytherapy as this implicates a second intervention and more intense hospital visits. Where the expected clinical benefit of irradiation is very low, as in the older population with low-risk breast cancer under adjuvant endocrine therapy, deferral is possible or omission could even be considered in some circumstances.29 The aim is to maintain a balance between respecting COVID-19 restrictive measures such as limiting number of contacts and physical distance, while at the same time preventing jeopardising the outcomes of cancer interventions, considering that radiation therapy usually requires a sometimes long series of treatments with repeated admissions to the institutions. This should be an additional argument for applying the abundance of evidence in favour of the use of hypofractionation in clinical practice.
Priorities for breast medical oncology in the early setting of care
The role of presurgical, perisurgical and postsurgical systemic therapies has evolved to become the backbone of care in breast oncology, and over the last decade a paradigm shift in systemic care has occurred, with the tailoring of type and timing of care to tumour subtypes. The delivery of systemic treatments usually demands a resource-intense effort of the health institutions, built around the patient, with optimal safety conditions—stating that the quality of medical oncology must attain the highest standards, when priority interventions are established (table 5 ).
Table 5.
Priorities for breast cancer: medical oncology—early breast cancer
| High priority | Medium priority | Low priority |
|---|---|---|
| Neoadjuvant and adjuvant chemotherapy for patients with triple-negative breast cancer | For postmenopausal women with stage I cancers, with low-intermediate grade tumours, lobular breast cancers endocrine therapy may be started first while surgery can be delayed. | Follow-up imaging, restaging studies, echocardiograms, ECGs and bone density scans can be delayed if patients clinically asymptomatic or clinical signs of response in the neoadjuvant setting. |
| Neoadjuvant and adjuvant chemotherapy in combination with targeted therapy for HER2-positive patients with breast cancer | ||
| Neoadjuvant and adjuvant endocrine therapy±chemotherapy for high-risk ER-positive/HER2-negative breast cancer as defined by current guidelines | ||
| Completion of neoadjuvant chemotherapy (with or without anti-HER2 therapy) that has already been initiated | For patients with low-risk genomic signatures/score prefer endocrine therapy alone. | - |
| Continuation of adjuvant capecitabine treatment in patients with high-risk triple-negative breast cancer, and T-DM1 in HER2-positive patients with high-risk breast cancer | Ongoing adjuvant trastuzumab alone may be postponed by 6–8 weeks in patients at high risk of complicated COVID-19 infection. | - |
| Continuation of standard adjuvant endocrine therapy in premenopausal and postmenopausal setting. Use telemedicine to manage potential toxicity reported by patients | ||
| Continuation of treatment in the context of a clinical trial, provided patient benefits overweight risks, with possible adaptation of procedures without affecting patient safety and study conduct. Regulatory agencies and sponsors may provide guidance on rules on study conduct during the pandemics | - | - |
During pandemics, the disruption of the procurement chain for medicines and the changed contexts of drug selection can have an impact on the availability and accessibility of medicines, including irregular provision or shortage of medicines for the supportive care.30 Optimising the medical oncology service during COVID-19 means assuring the assignment of the treatments at the highest benefit to the eligible population, in the safest conditions and guaranteeing punctual monitoring mechanisms for the adverse effects, implemented by telemedicine systems and engaging with primary healthcare sectors—an effort of one health system working in synchrony.
Decisions at national and institutional level can be readily informed by public health tools of selection and prioritisation of the medical interventions, including the ESMO MCBS.10 According to the MCBS, the priority interventions in the curative setting are scored A, on a scale from A to C, from high to low priority, respectively. While the score has been applied to a several novel compounds for breast cancer treatment, mostly in the advanced setting, less scoring has been performed in the early breast cancer setting, particularly with chemotherapy regimens, and this should be considered when balancing risk and benefits.10
Patients with the highest risk breast cancers are to be prioritised for systemic treatments (ie, either triple-negative or HER2-positive early breast cancer). Overall, adjuvant endocrine treatments are not expected to be substantially changed, not impacting significantly on immune functions, while the monitoring for side effects and treatment compliance could be readily performed via telemedicine. Of note, in case of SARS-CoV-2 infection in a patient with breast cancer taking tamoxifen, the hormone therapy should be suspended and preventive measures against thrombosis proposed, given the high risk of thromboembolic events in patients with COVID-19 with severe disease, and the well-known increased thromboembolic risk associated with tamoxifen.31
For some patients, a neoadjuvant endocrine approach could be considered, according to the current clinical recommendations, and possibly to delay the time to surgery in selected clinical presentations. This group encompasses patients with postmenopausal stage I cancers, low-grade to intermediate-grade tumours and lobular breast histology variants, deferring the surgery up to 6–12 months, as indicated in existing guidelines.32 Aromatase inhibitors (AI) should be preferred. While for postmenopausal women neoadjuvant endocrine therapy is an established approach, this is not so for premenopausal women but may be considered on a case-by-case basis under these extenuating circumstances. All premenopausal women receiving an AI should receive LHRH (Luteinizing hormone-releasing hormone) analogue. Monthly injections should be preferred but in some cases (particularly women in their mid-late 40s), one may discuss 3-monthly formulation of LHRH analogue, in order to reduce patient visits. Overall, home-based monthly administrations would be the preferred choice. The possibility to deliver treatments at home, including some antineoplastic agents (eg, subcutaneous trastuzumab, LHRH agonists) is framed in the national and regional competencies in the matter of healthcare organisation and regulations on medicines—conditioning the grades of operational flexibilities for the treatment delivery at the time of COVID-19.33 However, any initiative for home-based care should be built under the COVID-19 agenda, to ensure consistency with the physical distancing measures and protection of both the patients and the workforce.
Moreover, for selected HER2-positive breast cancer, low-risk or elderly patients with cardiovascular or other comorbidities adjuvant trastuzumab may reasonably be discontinued after 6 months instead of 12 months of treatment, according to clinical guidelines.34
Ongoing treatments should be fully completed, as any compromise in dose density and intensity may adversely impact prognosis. For triple-negative and HER2-positive patients with breast cancer treated with neoadjuvant therapies and not reaching a pathological complete response, postneoadjuvant systemic treatments are highly recommended as they provide durable disease control and improved survival.
The selection of the most appropriate schedules of chemotherapy will make considerations of at least three factors: the number of monthly admissions to the institutions, preferring 2-weekly or 3-weekly schedules; the need to escalate immunosuppressive supportive medications (eg, steroids for premedication); the informed patient’s preference. In the midst of uncertainties of the impact of immune-suppressive agents or steroids used for premedication at non-immunosuppressive doses on the risk of SARS-CoV-2 infection and COVID-19 outcome, the use of these agents should be carefully evaluated. The use of granulocyte growth factors may be considered to minimise neutropenia.
Priorities for breast medical oncology in the advanced and metastatic setting of care
The decision making for priority positioning in the metastatic setting is more complicated. In the clinical landscape of effective treatments that impact on the quality of life and the survival, the best treatment approaches should be part of the health priorities (table 6 ).
Table 6.
Priorities for breast cancer: medical oncology—metastatic breast cancer
| High priority | Medium priority | Low priority |
|---|---|---|
| Early line chemotherapy, endocrine therapy, targeted agents or immune-checkpoint inhibitors likely to improve outcomes in metastatic disease (high priority to pertuzumab/trastuzumab plus chemotherapy in HER2-positive breast cancer). Consider also, discussing case-by-case in multidisciplinary tumour board, endocrine therapy with CDK 4–6 inhibitors in ER-positive/HER2-negative breast cancer, chemotherapy plus atezolizumab in PD-L1-positive TNBC |
Second, third, beyond third-line treatment when therapy may provide clinical benefit and impact on outcome. | Bone agents therapy (zoledronic acid, denosumab) that are not needed urgently for hypercalcaemia, or not needed for pain control and in patients who are otherwise not in need for coming to the hospital (ie, receiving oral chemotherapy or endocrine therapy). Bone agents can be administered every 3 months. |
| Visceral crisis | Consider avoiding or delaying the addition of mTOR inhibitors or PIK3CA inhibitors (still not approved in European Union) to endocrine therapy, particularly in elderly patients with comorbidities. | If clinically asymptomatic follow-up imaging, restaging studies, echocardiograms and ECGs can be delayed or done at lengthened intervals. |
| Continuation of treatment in the context of a clinical trial, provided patient benefits overweight risks, with possible adaptation of procedures without affecting patient safety and study conduct. Regulatory agencies and sponsors may provide guidance on rules on study conduct during the pandemics. |
Consider, discussing case-by-case, inclusion in a clinical trial, provided patient benefits overweight risks, with possible adaptation of procedures without affecting patient safety and study conduct. | - |
mTOR, mammalian target of rapamycin; PD-L1, programmed death-ligand 1; PIK3CA, phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha; TNBC, triple-negative breast cancer.
The health cogency criterion demands prompt cancer treatments in patients where a delay can result in fatal outcomes. For example, patients experiencing cancer-related treatment organ dysfunctions or at increased risk of it, namely patients in visceral crisis, should be prioritised for the start of antineoplastic treatments.
According to the MCBS, the priority interventions in the advanced setting are scored 3, 4 or 5, in a descending scale for value, from 5 to 1.10 For such, the magnitude of benefit and the expected treatment benefits should guide the clinical indications. Early line chemotherapy, endocrine therapy and targeted agents with higher MCBS score are of choice, and should be delivered in the eligible population—for example, in the prescription of the double anti-HER2 blockade plus chemotherapy as first line for patients with HER2-positive disease, where affordably accessible (MCBS score 4).
The use of immunotherapy (eg, atezolizumab) and CDK4/6 inhibitors and endocrine therapy (MCBS scores of 3–4), should be considered after a case-by-case discussion taking into account baseline risk factors of the patient and their functional status, along with considerations on the disease pattern of spread and biology—to ensure a critical decision making by tailoring for the single patient, in accordance with local practice and resource availability. In the midst of uncertainty related to some immune-modulating agents with limited value in cancer treatment, avoiding or delaying the addition of mTOR inhibitors to endocrine therapy could be prudent, especially in multicomorbid older patients (MCBS score for everolimus: 2).
This means that the contingency plan during the pandemic could result in the temporary withdrawal of some interventions, or predefined safe delays. The use of oral bisphosphonate or subcutaneous denosumab can be considered in some cases, as to the choice of longer intervals for intravenous bisphosphonates, for example, every 3 months. Similarly, in late treatment lines, patients with poorer prognosis and impaired functional status could be considered for a best supportive care (BSC) approach and/or drug holiday periods—with consideration given for the benefits in terms of quality of life, while attaining to the highest standards of care: an early start of home-based supportive care plan and the judicious referral of some patients to BSC is a priority, overall, when the available therapies are known to provide only a narrow benefit and they may incur on the patient excessive of toxicity.35 Treatment de-escalation and/or maintenance with oral agents may be considered, although largely based on experts’ opinions.
COVID-19 and its impact on cancer research
The continuation of treatment in the context of a clinical trial is valuable for cancer clinical research, provided patient benefits outweigh risks, with possible adaptation of trial procedures without affecting patient safety and study conduct.
To date, the social distancing measures have partially frozen the preclinical laboratory-based research in many countries, in response to the urgent need to prioritise the services with an immediate role in COVID-19 response and to serve community needs. Overall, the national and regional regulatory agencies have formulated guidance for the management of patients enrolled in clinical trials, providing recommendations on the exploitation of the study procedures and the safe delivery of the study medications, including concrete information on changes and protocol deviations which may be needed in the conduct of clinical trials to deal with extraordinary situations. In such a context, protocol deviations are expected and some are justified, when not exposing patients to safety issues and facilitating procedural barriers or bureaucratic aspects of study procedures: a real moment to re-think the clinical trial conductions tout court.36 For instance, in Europe the recommendations of the European Medicines Agency can be found at: https://www.ema.europa.eu/en/news/guidance-sponsors-how-manage-clinical-trials-during-covid-19-pandemic In the USA, the Food and Drug Administration also released guidance on clinical research and COVID-19 (https://www.fda.gov/media/136238/download).
While avoiding a total distraction of the cancer research community from the goals and research questions and slowing the progress in cancer care—research questions of immediate clinical interest for cancer and COVID-19 have been implemented, bridging the need to provide responses to patients with cancer and the advancement of knowledge.
Several ongoing trials are addressing the safety and trying to dissect the interplay of anticancer and antiviral immune responses. Generating quality evidence is now the real priority, to enhance the clinical decision-making: the observational series of the first waves are now expected to leave room for controlled studies, mechanistic explanations and translational experiences, ensuring the best research at the service of the cancer and pandemic response. There is an ongoing list of clinical trials testing different strategies in patients with COVID-19 and they can be found at http://www.redo-project.org/COVID-19_db-summaries/
Conclusions
For cancer care, COVID-19 is presenting a challenging period in medicine, demanding a re-focus on the value and priorities of health interventions, including the reshaping of cancer care—catalysing a review of value-based and patient-centred decision making. Setting priorities based on the intrinsic value of the interventions about patients’ outcomes and ensuring simultaneously population and societal benefits is the perspective in which the response to COVID-19 has been designed.
The priority-setting mechanism that is envisioned aligns with the global, regional and national standards proposed in several work streams, including the development and implementation of the MCBS scale, which is based on patient-related end points and aimed to provide a statement and guidance on priority setting, to support and enable decision making when resources need to be rationed and cautiously allocated.
WHO has defined differentiated preparedness and response plans, according to the disease spread in single countries, providing global guidance and guidance for local action. This means that the degree of SARS-CoV-2 spread in a country dictates the type and intensity of the public health interventions and should also orient the selection and prioritisation of the health interventions in oncology—with a tiered approach.
The perimeters and limits of this work should be interpreted and adapted within the national and regional dispositions in terms of the ability towards health system reorganisation and reshaping of the existing model. While some countries have interpreted a segregation model, and suggestions from WHO seems to propend more for the identification of a COVID-19 health pathway for prompt referral, policies can vary across countries, related to the stage of the pandemic as well as the health sector specificities.
In such a complex setting, this clinical guidance for breast cancer management is intended to orient and not decree local guidelines, and guide the development of action plans to maintain a quality cancer service, setting a minimal array of required interventions. Useful information can be found at https://www.esmo.org/guidelines/cancer-patient-management-during-the-covid-19-pandemic/breast-cancer-in-the-covid-19-era
Acknowledgements
The authors would like to thank ESMO for allowing the use of the tables in this manuscript.
Footnotes
Twitter: @E_de_Azambuja, @darioT_, @MichaelGnant
EdA and DT contributed equally.
Correction notice: This paper has been updated since first published to correct name of author 'Philip Poortmans'.
Contributors: GC developed the first outline and coordinated the authors’ contributions, engaged for competencies and expertise in the management of breast cancer, development of adapted guidelines in special circumstances and its methodology. GC and DT formulated the first draft to organise the authors’ work, and with EdA developed a draft zero. All the authors were involved in writing and, when the first draft had been developed, all the authors refined the contents with feedbacks and comments, incorporated by GC, EdA and DT. All the authors approved the final draft before the submission. While the authors are affiliated to different institutions for cancer research and care, the perspectives expressed in the present paper should not be intended to mirror the societal and institutional positions.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: GC has received honoraria from Pfizer, Novartis, Eli Lilly, Roche; fees for expert testimony and medical education from Pfizer and has participated in advisory boards for Pfizer, Roche, Eli Lilly, Novartis, Seattle Genetics, Celltrion. SD reports grants, institutional fees from Roche/Genentech, Pfizer, Puma, AstraZeneca, Novartis, Amgen, Sanofi, Eli Lilly, MSD, BMS, Daichi and non-financial support from Roche/Genentech, Pfizer, AstraZeneca. SDC has received fees for medical education from Novartis and Pierre-Fabre and is the recipient of the IG 20774 of Fondazione AIRC. MG reports personal fees/travel support from Amgen, AstraZeneca, Celgene, Eli Lilly, Invectys, Pfizer, Novartis, Puma, Nanostring, Roche, Medison, LifeBrain, all outside the submitted work; an immediate family member is employed by Sandoz. PP is medical advisor of Sordina IORT Technologies. ES has received honoraria from Amgen, AstraZeneca, Clinigen, Egis, Eli Lilly, Genomic Health, Novartis, Pfizer, Pierre-Fabre, Roche, Sandoz, TLC Biopharmaceuticals and travel support from Amgen, AstraZeneca, Egis, Novartis, Pfizer, Roche. FC has consultancy role for Amgen, Astellas/Medivation, AstraZeneca, Celgene, Daiichi-Sankyo, Eisai, GE Oncology, Genentech, GlaxoSmithKline, Macrogenics, Medscape, Merck-Sharp, Merus BV, Mylan, Mundipharma, Novartis, Pfizer, Pierre-Fabre, prIME Oncology, Roche, Sanofi, Samsung Bioepis, Seattle Genetics, Teva. EdA honoraria and/or advisory board from Roche/GNE, Novartis, SeaGen and Zodiac; travel grants from Roche/GNE and GSK/Novartis; research grant to the institution from Roche/GNE, AstraZeneca, GSK/Novartis and Servier. CC declares consultancy/advisory role/speaker's bureau: Pfizer, Eli Lilly, Roche, Novartis. SP-S discloses honoraria, consultancy and speaker’s bureau: Roche, Novartis, Pfizer, Eli Lilly, AstraZeneca, Nanostring.
Patient consent for publication: Not required.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Burki T.K. Cancer guidelines during the COVID-19 pandemic. Lancet Oncol. 2020;21:629–630. doi: 10.1016/S1470-2045(20)30217-5. doi:10.1016/S1470-2045(20)30217-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Emanuel E.J., Persad G., Upshur R. Fair allocation of scarce medical resources in the time of Covid-19. N Engl J Med. 2020 doi: 10.1056/NEJMsb2005114. http://www.ncbi.nlm.nih.gov/pubmed/32202722 doi:10.1056/NEJMsb2005114 doi [Epub ahead of print 23 Mar 2020] [DOI] [PubMed] [Google Scholar]
- 3.Ferguson N.M., Laydon D., Nedjati-Gilani G. Imperial College London; London: 2020. Impact of non-pharmaceutical interventions (NPIs) to reduce COVID-19 mortality and healthcare demand. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baker T., Schell C.O., Petersen D.B. Essential care of critical illness must not be forgotten in the COVID-19 pandemic. Lancet. 2020;395:1253–1254. doi: 10.1016/S0140-6736(20)30793-5. http://www.ncbi.nlm.nih.gov/pubmed/32246914 doi:10.1016/S0140-6736(20)30793-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.GBD 2016 Healthcare Access and Quality Collaborators Measuring performance on the healthcare access and quality index for 195 countries and territories and selected subnational locations: a systematic analysis from the global burden of disease study 2016. Lancet. 2018;391:2236–2271. doi: 10.1016/S0140-6736(18)30994-2. http://www.ncbi.nlm.nih.gov/pubmed/29893224 doi:10.1016/S0140-6736(18)30994-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hanna T.P., Evans G.A., Booth C.M. Cancer, COVID-19 and the precautionary principle: prioritizing treatment during a global pandemic. Nat Rev Clin Oncol. 2020;17:268–270. doi: 10.1038/s41571-020-0362-6. http://www.ncbi.nlm.nih.gov/pubmed/32242095 doi:10.1038/s41571-020-0362-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liang W., Guan W., Chen R. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol. 2020;21:335–337. doi: 10.1016/S1470-2045(20)30096-6. http://www.ncbi.nlm.nih.gov/pubmed/32066541 doi:10.1016/S1470-2045(20)30096-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.WHO COVID-19: operational guidance for maintaining essential health services during an outbreak interim guidance. 2020 https://apps.who.int/iris/handle/10665/331561 Available[Accessed ], 10 Apr 2020. [Google Scholar]
- 9.Cancer Care Ontario. Identify priority problems and goals to improve the model of care. Available: file:///C:/Users/pc/Downloads/PEPPAToolkit_Step4.pdf [Accessed 10 Apr 2020].
- 10.Cherny N.I., Dafni U., Bogaerts J. ESMO-Magnitude of clinical benefit scale version 1.1. Ann Oncol. 2017;28:2340–2366. doi: 10.1093/annonc/mdx310. http://www.ncbi.nlm.nih.gov/pubmed/28945867 doi:10.1093/annonc/mdx310 [DOI] [PubMed] [Google Scholar]
- 11.OECD. Tackling coronavirus (COVID–19). Contributing to a global effort. Available: https://www.oecd.org/coronavirus/en/ [Accessed 10 Apr 2020].
- 12.Kesson E.M., Allardice G.M., George W.D. Effects of multidisciplinary team working on breast cancer survival: retrospective, comparative, interventional cohort study of 13 722 women. BMJ. 2012;344:e2718. doi: 10.1136/bmj.e2718. http://www.ncbi.nlm.nih.gov/pubmed/22539013 doi:10.1136/bmj.e2718 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Georges Q., Azoulay E., Mokart D. Influence of neutropenia on mortality of critically ill cancer patients: results of a meta-analysis on individual data. Crit Care. 2018;22:326. doi: 10.1186/s13054-018-2076-z. http://www.ncbi.nlm.nih.gov/pubmed/30514339 doi:10.1186/s13054-018-2076-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Carrera P.M., Kantarjian H.M., Blinder V.S. The financial burden and distress of patients with cancer: understanding and stepping-up action on the financial toxicity of cancer treatment. CA Cancer J Clin. 2018;68:153–165. doi: 10.3322/caac.21443. http://www.ncbi.nlm.nih.gov/pubmed/29338071 doi:10.3322/caac.21443 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Caplan L. Delay in breast cancer: implications for stage at diagnosis and survival. Front Public Health. 2014;2 doi: 10.3389/fpubh.2014.00087. http://www.ncbi.nlm.nih.gov/pubmed/25121080 doi:10.3389/fpubh.2014.00087 87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Anderson S.J., Wapnir I., Dignam J.J. Prognosis after ipsilateral breast tumor recurrence and locoregional recurrences in patients treated by breast-conserving therapy in five national surgical adjuvant breast and bowel project protocols of node-negative breast cancer. J Clin Oncol. 2009;27:2466–2473. doi: 10.1200/JCO.2008.19.8424. http://www.ncbi.nlm.nih.gov/pubmed/19349544 doi:10.1200/JCO.2008.19.8424 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Waeber M., Castiglione-Gertsch M., Dietrich D. Adjuvant therapy after excision and radiation of isolated postmastectomy locoregional breast cancer recurrence: definitive results of a phase III randomized trial (SAKK 23/82) comparing tamoxifen with observation. Ann Oncol. 2003;14:1215–1221. doi: 10.1093/annonc/mdg347. http://www.ncbi.nlm.nih.gov/pubmed/12881382 doi:10.1093/annonc/mdg347 [DOI] [PubMed] [Google Scholar]
- 18.Wapnir I.L., Price K.N., Anderson S.J. Efficacy of chemotherapy for ER-negative and ER-positive isolated locoregional recurrence of breast cancer: final analysis of the CALOR trial. J Clin Oncol. 2018;36:1073–1079. doi: 10.1200/JCO.2017.76.5719. http://www.ncbi.nlm.nih.gov/pubmed/29443653 doi:10.1200/JCO.2017.76.5719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Curigliano G., Lenihan D., Fradley M. Management of cardiac disease in cancer patients throughout oncological treatment: ESMO consensus recommendations. Ann Oncol. 2020;31:171–190. doi: 10.1016/j.annonc.2019.10.023. http://www.ncbi.nlm.nih.gov/pubmed/31959335 doi:10.1016/j.annonc.2019.10.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Americal College of Cardiology (ACC). Risk score for drug-associated QTc prolongation. ventricular arrhythmia risk due to Hydroxychloroquine-Azithromycin treatment for COVID-19. Available: https://www.acc.org/latest-in-cardiology/articles/2020/03/27/14/00/ventricular-arrhythmia-risk-due-to-hydroxychloroquine-azithromycin-treatment-for-covid-19 [Accessed 10 Apr 2020].
- 21.Lafranconi A., Pylkkänen L., Deandrea S. Intensive follow-up for women with breast cancer: review of clinical, economic and patient's preference domains through evidence to decision framework. Health Qual Life Outcomes. 2017;15 doi: 10.1186/s12955-017-0779-5. http://www.ncbi.nlm.nih.gov/pubmed/29052503 doi:10.1186/s12955-017-0779-5 206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.World Health Organization Updated appendix 3 of the who global Ncd action plan 2013-2020. Technical Annex. 2017 [Google Scholar]
- 23.Harbeck N., Gnant M. Breast cancer. Lancet. 2017;389:1134–1150. doi: 10.1016/S0140-6736(16)31891-8. http://www.ncbi.nlm.nih.gov/pubmed/27865536 doi:10.1016/S0140-6736(16)31891-8 [DOI] [PubMed] [Google Scholar]
- 24.Robertson S.A., Jeevaratnam J.A., Agrawal A. Mastectomy skin flap necrosis: challenges and solutions. Breast Cancer. 2017;9:141–152. doi: 10.2147/BCTT.S81712. http://www.ncbi.nlm.nih.gov/pubmed/28331365 doi:10.2147/BCTT.S81712 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.American College of Surgeons COVID 19: elective case triage guidelines for surgical care. released March 2020. 2020 https://www.facs.org/covid-19/clinical-guidance/triage 14 Apr 2020. Available[Accessed ] [Google Scholar]
- 26.Jiang Zefei L.J. Ten hot issues of breast cancer during the COVID-19 pandemic. Recommendations by the Chinese Society of clinical oncology breast cancer (CSCO bc) 2020 [Google Scholar]
- 27.Gnant M. Challenges and controversies in breast surgery. Breast Care. 2019;14:185–187. doi: 10.1159/000502134. http://www.ncbi.nlm.nih.gov/pubmed/31558891 doi:10.1159/000502134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Coles C.E., Aristei C., Bliss J. International guidelines on radiation therapy for breast cancer during the COVID-19 pandemic. Clin Oncol. 2020;32:279–281. doi: 10.1016/j.clon.2020.03.006. doi:10.1016/j.clon.2020.03.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kunkler I.H., Williams L.J., Jack W.J.L. Breast-Conserving surgery with or without irradiation in women aged 65 years or older with early breast cancer (prime II): a randomised controlled trial. Lancet Oncol. 2015;16:266–273. doi: 10.1016/S1470-2045(14)71221-5. http://www.ncbi.nlm.nih.gov/pubmed/25637340 doi:10.1016/S1470-2045(14)71221-5 [DOI] [PubMed] [Google Scholar]
- 30.Linton T., Vakil B. Coronavirus is proving we need more resilient supply chains. Harvard business review. 2020 https://hbr.org/2020/03/coronavirus-is-proving-that-we-need-more-resilient-supply-chains 5 Apr 2020. Available[Accessed ] [Google Scholar]
- 31.Tang N., Bai H., Chen X. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy [published online. J Thromb Haemost. 2020 doi: 10.1111/jth.14817. 10.1111/jth.14817, ahead of print, 2020 Mar 27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cardoso F., Kyriakides S., Ohno S. Early breast cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2019;30:1674. doi: 10.1093/annonc/mdz189. http://www.ncbi.nlm.nih.gov/pubmed/31236598 doi:10.1093/annonc/mdz189 [DOI] [PubMed] [Google Scholar]
- 33.Cocquyt V.F., Martinez-Mena C.L., Martens M.T. Belis: safety and tolerability of at home administration of trastuzumab (Herceptin) subcutaneous for the treatment of patients with her2-positive early breast cancer [abstract P4-21-17] Cancer Res. 2017;77 [Google Scholar]
- 34.Earl H.M., Hiller L., Vallier A.-L. 6 versus 12 months of adjuvant trastuzumab for HER2-positive early breast cancer (Persephone): 4-year disease-free survival results of a randomised phase 3 non-inferiority trial. Lancet. 2019;393:2599–2612. doi: 10.1016/S0140-6736(19)30650-6. http://www.ncbi.nlm.nih.gov/pubmed/31178152 doi:10.1016/S0140-6736(19)30650-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cinausero M., Gerratana L., De Carlo E. Determinants of Last-line treatment in metastatic breast cancer. Clin Breast Cancer. 2018;18:205–213. doi: 10.1016/j.clbc.2017.07.008. http://www.ncbi.nlm.nih.gov/pubmed/28781022 doi:10.1016/j.clbc.2017.07.008 [DOI] [PubMed] [Google Scholar]
- 36.Tarantino P., Trapani D., Curigliano G. Conducting phase 1 cancer clinical trials during the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)-related disease pandemic. Eur J Cancer. 2020;132:8–10. doi: 10.1016/j.ejca.2020.03.023. http://www.ncbi.nlm.nih.gov/pubmed/32305011 doi:10.1016/j.ejca.2020.03.023 [DOI] [PMC free article] [PubMed] [Google Scholar]


