Abstract
Background
Evidence supports therapeutic drug monitoring (TDM) in improving efficacy and cost-effectiveness of anti-TNF therapy in inflammatory bowel disease (IBD). Data on perceptions and barriers to TDM use are limited and no data are available from India. Our objective was to assess clinicians’ attitudes and barriers to TDM use in IBD.
Methods
A 16-question survey was distributed to members of the Indian Society of Gastroenterology. Information on clinician characteristics, demographics, use and barriers towards TDM with anti-TNFs was collected. Logistic regression was used to predict factors influencing TDM use.
Results
Two hundred and forty-two respondents participated (92.5% male); 83% were consultant gastroenterologists. Of 104 respondents meeting inclusion criteria (treating > 5 IBD patients and at least 1 with an anti-TNF per month), complete responses were available for 101 participants. TDM was utilized by 20% (n = 20) of respondents. Of them, 89.5% (n = 17) used TDM for secondary loss of response; 73.7% (n = 14) for primary non-response and 5.3% (n = 1) proactively. Barriers to TDM use were cost (71.2%), availability (67.8%), time lag in results (58.7%) and the perception that TDM is time-consuming (45.7%). Clinicians treating > 30 IBD patients were more likely to check TDM (OR = 4.9, p = 0.02). Of 81 respondents not using TDM, 97.5% (n = 79) would do so if all the barriers were removed.
Conclusion
Significant barriers to TDM use were availability, cost and time lag for results. If these barriers were removed, almost all the clinicians would use TDM at least reactively and 25% would use proactively. There is an urgent need to address these barriers and optimize anti-TNF therapy for optimal outcomes.
Electronic supplementary material
The online version of this article (10.1007/s12664-020-01047-6) contains supplementary material, which is available to authorized users.
Keywords: Adalimumab, Crohn’s disease, Drug levels, Infliximab, Pharmacokinetics, Ulcerative colitis
Introduction
Anti-TNF therapies have transformed the care of patients with inflammatory bowel disease (IBD). They have re-defined our perceptions around meaningful disease control - moving beyond symptom control to bolder definitions such as mucosal healing, histological and deep remission and an improvement in quality of life [1, 2]. “Treating to target” and achieving mucosal healing when possible is now an important priority [3–5]. Gastroenterologists currently have relatively limited options to achieve this. To contextualize, in India, the mainstay of biological therapy currently is anti-TNF therapy with anti-integrin therapy becoming available soon.
Anti-TNF therapies are immunogenic and are associated with loss of response [6]. Up to 30% of patients have a primary non-response (PNR) and up to 50% will develop a secondary loss of response (SLR) to anti-TNFs [7, 8]. This can be caused by low or undetectable drug concentrations due to immune (anti-drug antibodies) and non-immune clearance [7, 9]. The risk of attrition can make each successive therapy less effective, implying that the first biologic is often most likely to be the most effective. Meanwhile, aiming for higher anti-TNF trough levels may be associated with better outcomes during both maintenance [10–18] and induction [19–22].
Cost of biological therapy has also posed limitations to their use but the approval of biosimilars for infliximab and adalimumab may serve to broaden the reach of these highly effective therapies [23, 24]. Taken together, this emphasizes the need to select wisely and optimize anti-TNF therapy in well-selected patients.
Therapeutic drug monitoring (TDM) involves measuring serum drug trough concentrations and anti-drug antibodies to optimize the use of anti-TNF agents [25–28]. TDM can be either reactive or proactive. Reactive TDM involves checking serum drug trough levels and anti-drug antibodies when there is a suspicion of loss of response to anti-TNF therapy [10–18, 25]. It has been shown to be cost-effective compared to empiric dose escalation [29–31]. Conversely, proactive TDM involves checking serum drug trough levels and anti-drug antibodies at pre-determined time points, irrespective of disease activity, with the aim of preventing “under-dosing” from triggering a disease flare and de-escalating dosing in case of “supra-therapeutic” drug levels [19–22, 25]. The use of TDM, at least reactively, is supported by international IBD guidelines [26–28, 32–34]. Data on attitudes, perceptions and barriers to the use of TDM with anti-TNF therapy are scarce, limited to two studies, from the USA and the UK respectively [35, 36]. India has the second largest IBD population in the world but limited access to biological choices compared with the Western world, making a compelling argument for optimizing the therapies available, through the use of TDM.
We conducted a survey on the use of TDM with anti-TNF therapy in India. Our primary aim was to describe the proportion of gastroenterologists utilizing TDM, the clinical setting in which this was used and to identify barriers to the use of TDM in clinical practice. Our secondary aim was to identify the clinical scenarios in which TDM would be used by gastroenterologists if all perceived barriers to TDM were removed.
Methods
Study design
A 16-question survey (Appendix 1) was adapted with permission from a similar study conducted in the UK [36]. The survey underwent a second modifying process in consultation with gastroenterologists (DD, GM and VA) at two large centres in India to ensure its suitability for participants. This was then placed on an online survey tool and an invitation with a link to complete the same was sent out to consultants and higher specialist trainee (Registrar/Fellow) members of the Indian Society of Gastroenterology (ISG) (approximately 1500 members) between June and October 2019. TDM was performed at either All India Institute of Medical Sciences (AIIMS), New Delhi or P D Hinduja Hospital, Mumbai. The study was registered with and approved by the Research and Innovation department of the Pennine Acute Hospitals NHS Trust, UK. No funding was required for this study.
Demographic information sought from the participants included their age, sex, grade (consultant, gastroenterology trainee/registrar, physicians with special interest in gastroenterology), number of years in practice since specialist qualification or accreditation for gastroenterology (as applicable), place of work (government medical college, private medical college, private group practice/corporate hospital or private individual practice) and city of work (tier 1/Metropolitan, tier 2/other state capitals or tier 3/all other cities). Additionally, information was collected from respondents regarding the proportion of patients with IBD seen in their clinical practice, number of patients with IBD treated personally in a 1-month period and numbers treated with anti-TNF therapy per month. We also sought details around the use of TDM using Likert 5-point scales ranging from strongly agree to strongly disagree, to identify levels of agreement or disagreement with potential barriers to using TDM. Participants who treated < 5 IBD patient per month and/or had no patients on anti-TNF therapy every month were excluded from the study.
Statistical analysis
The data were analyzed using R software Version 3.5.2 (R development core team, Vienna, Austria). All variables were categorical and expressed as frequencies and percentages. Univariate logistic regressions were used to examine associations between available variables and the outcomes of interest, use of TDM and proactive TDM. Associations were reported as p-values and odds ratios, along with their 95% confidence intervals. To determine the independent effects of variables associated with the use of reactive and proactive TDM, a multiple binary logistic regression analysis was then performed including variables with a p-value of < 0.1 from univariate analysis.
Results
Responses were received from 242 participants, of whom 104 met inclusion criteria (138 clinicians reported treating less than 5 IBD patients per month and/or having no patient on anti-TNF therapy and were therefore excluded). Baseline characteristics of all the participants are depicted in Table 1.
Table 1.
Participant demographic and clinical characteristics. GI gastrointestinal, IBD inflammatory bowel disease
| Participants | N = 242 |
|---|---|
| Gender | |
| Male | 223 (92.2%) |
| Female | 19 (7.8%) |
| Practice setting (more than one) | |
| Government medical college | 56 (23.1%) |
| Private medical college | 29 (12%) |
| Private group practice/corporate hospital | 92 (38%) |
| Private individual practice | 81 (33.5%) |
| Grade | |
| Consultant gastroenterologist | 201 (83.1%) |
| Physician with special interest in GI practice | 15 (6.2%) |
| Gastroenterology trainee | 18 (7.4%) |
| Other | 8 (3.3%) |
| City of practice | |
| Tier 1 (Metropolitan) | 109 (45%) |
| Tier 2 (other state capitals) | 47 (19.4%) |
| Tier 3 (all other cities and towns) | 86 (35.6%) |
| Age (year) | |
| 25–34 | 45 (18.6%) |
| 35–44 | 82 (33.9%) |
| 45–54 | 46 (19%) |
| 55–64 | 53 (21.9%) |
| > 65 | 16 (6.6%) |
|
Years (post gastroenterology certification) in practice/ Still in training | |
| < 1 | 15 (6.2%) |
| 1–4 | 14 (5.8%) |
| 5–9 | 48 (19.8%) |
| 10–19 | 43 (17.8%) |
| > 20 |
46 (19%) 76 (31.4%) |
| % of patients with IBD in individual practice, (238 responses, 4 skipped) | |
| < 10% | 180 (75.6%) |
| 11–25% | 56 (23.5%) |
| 26–50% | 2 (0.9%) |
| > 50% | 0 (0%) |
Based on the inclusion criteria, only 104 participants were included for further analysis. This is represented as a flow diagram in Fig. 1 and the details are included in Table 2.
Fig. 1.
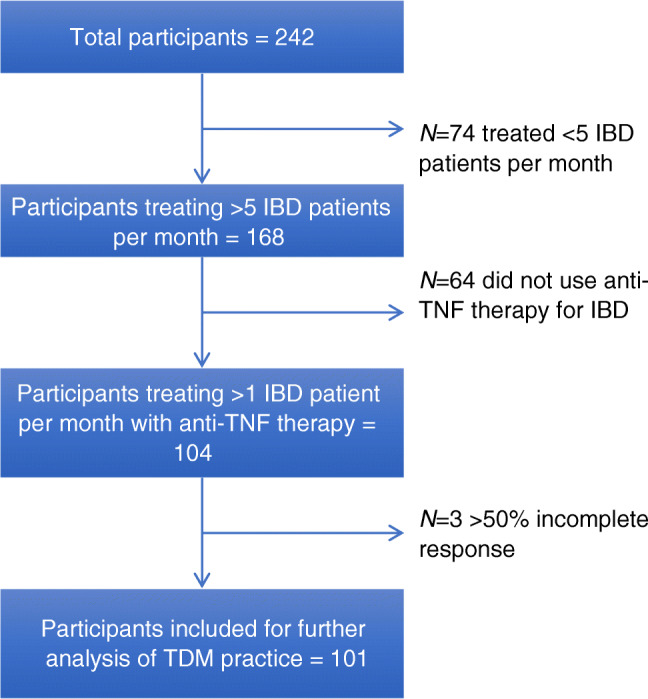
Flow diagram showing inclusion of participants for further analysis. IBD inflammatory bowel disease, TNF tumor necrosis factor, TDM therapeutic drug monitoring
Table 2.
Participant characteristics based on inclusion criteria. IBD inflammatory bowel disease, TNF tumor necrosis factor
| Number of patients with IBD treated per month, (168 responses) | |
| 5–10 | 81 (34.3%) |
| 11-20 | 39 (16.5%) |
| 20-30 | 27 (11.4%) |
| > 30 | 21 (8.9%) |
| No. of patients with IBD treated with anti-TNF per month, (104 responses) | |
| 1–4 | 94 (56.3%) |
| 5-10 | 10 (6%) |
| 11-20 | 0 (0%) |
Practice of TDM
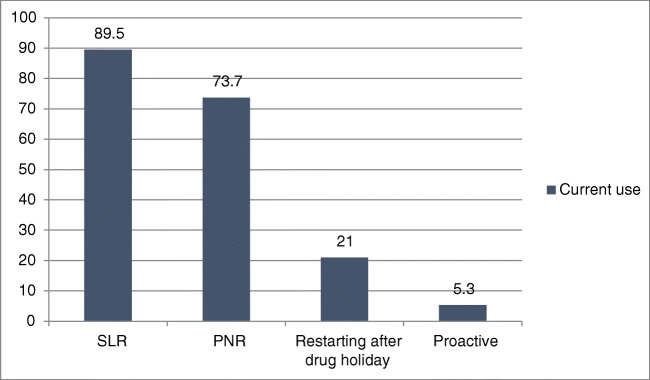
Of the 104 participants included in this analysis, completed responses were available for only 101 participants. TDM was utilized in clinical practice by 20% (n = 20) of respondents. Of them, 89.5% (n = 17) used TDM for SLR; 73.7% (n = 14) used it for PNR; 21% (n = 4) used it before restarting anti-TNF therapy after a drug holiday; and 5.3% (n = 1) used TDM proactively (Fig. 2). Of the 242 initial respondents, 64 clinicians (26.44%) reported not using biological therapies at all to manage their patients with IBD.
Fig. 2.
Current use of therapeutic drug monitoring. PNR primary non-response, SLR secondary loss of response
Multivariate analysis identified practice in tier 2 cities (p = 0.047), having 11% to 25% patients with IBD in their practice (p = 0.018) and seeing/treating a higher number of IBD patients per month (p = 0.036) as factors independently associated with the use of TDM (Table 3).
Table 3.
Univariate and multivariate logistic regression analysis of variables associated with the use of therapeutic drug monitoring. GI gastrointestinal, IBD inflammatory bowel disease, TNF tumor necrosis factor, OR odds ratio, CI confidence interval
| Variables | Univariate analysis | Multivariate analysis | |||||
|---|---|---|---|---|---|---|---|
| p-value | OR | 95% CI | p-value | OR | 95% CI | ||
| Practice setting | 0.890 | ||||||
| Government medical college | 1 | ||||||
| Private medical college | 0.800 | ||||||
| Private group practice/corporate hospital | 0.604 | ||||||
| Private individual practice | 0.601 | ||||||
| City of practice | 0.098 | ||||||
| Tier 1 (Metropolitan) | 1 | 1 | |||||
| Tier 2 (other state capitals) | 0.084 | 0.047 | 0.1 | 0.01–0.97 | |||
| Tier 3 (all other cities and towns) | 0.634 | 0.6 | |||||
| Grade | 0.176 | ||||||
| Consultant gastroenterologist | 1 | ||||||
| Physician with special interest in GI | 0.999 | ||||||
| Gastroenterology trainee | 0.998 | ||||||
| Others/surgeon | 0.999 | ||||||
| Gender | 0.148 | ||||||
| Female | 1 | ||||||
| Male | 0.132 | ||||||
| Age Group | 0.171 | ||||||
| 25–34 years | 1 | ||||||
| 35–44 years | 0.058 | ||||||
| 45–54 years | 0.234 | ||||||
| 55–64 years | 0.199 | ||||||
| > 65 | 0.998 | ||||||
| Years of practice | 0.249 | ||||||
| Still in training | 1 | ||||||
| < 1 years | 0.273 | ||||||
| 1–4 years | 0.919 | ||||||
| 5–9 years | 0.288 | ||||||
| 10–19 years | 0.728 | ||||||
| > 20 years | 0.552 | ||||||
| Percentage of patients with IBD | 0.009 | ||||||
| < 10 | 1 | ||||||
| 11–25% | 0.004 | 4.8 | 1.7–13.9 | 0.018 | 3.9 | 1.3–12.4 | |
| 26–50% | 0.998 | ||||||
| No. of patients with IBD reviewed/treated per month | 0.035 | ||||||
| 5–10 | 1 | 1 | |||||
| 11–20 | 0.686 | 0.745 | |||||
| 21–30 | 0.154 | 0.261 | |||||
| > 30 | 0.021 | 4.9 | 1.3–20 | 0.036 | 3.5 | 0.8–16.1 | |
| No. of patients with IBD on anti-TNF therapy per month | 0.282 | ||||||
| 0 | 1 | ||||||
| 1–4 | 0.890 | ||||||
| 5–10 | 0.214 | ||||||
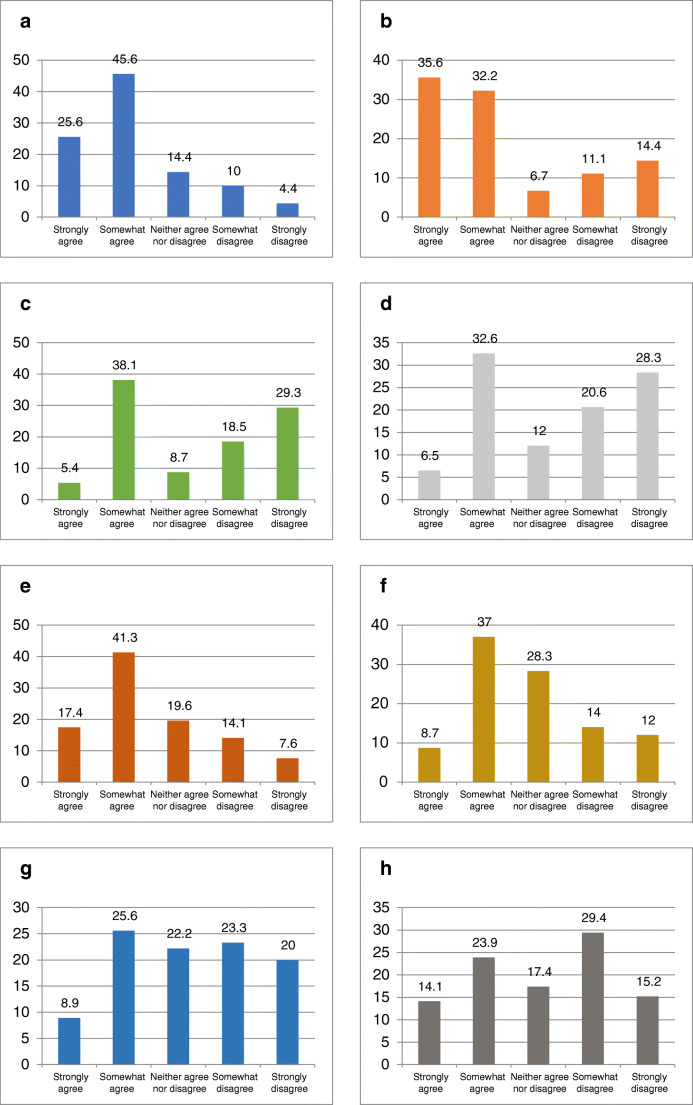
The main barriers for TDM use reported by the respondents were cost (71.2%), uncertainty about availability (67.8%), time lag in receiving results (58.7%) and the perception that TDM is cumbersome and time consuming (45.7%). Respondents mostly disagreed or strongly disagreed that lack of overall knowledge of TDM (47.8%), lack of knowledge regarding how to interpret TDM and what to do with results of TDM (48.9%), lack of awareness of clinical guidelines (44.6%) and perceived lack of an evidence base for TDM use (43.3%) were barriers to its use (Fig. 3).
Fig. 3.
a Barriers to therapeutic drug monitoring (TDM): test is expensive; b uncertainty about availability in my practice; c lack of overall knowledge of TDM; d lack of knowledge on how to interpret and what to do with the results of TDM; e time lag from serum sampling to results of TDM; f TDM is cumbersome and/or time-consuming; g lack of good evidence-based medicine of the usefulness of TDM in inflammatory bowel disease; h lack of clinical guidelines recommending the use of TDM
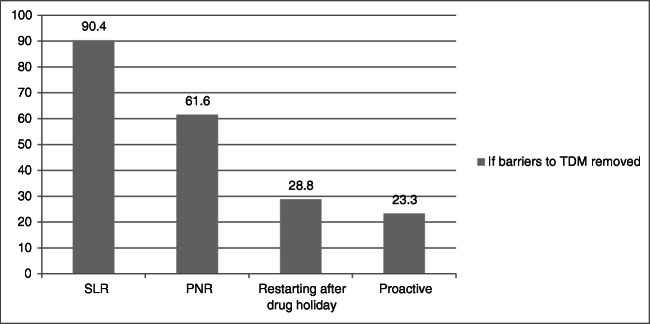
If all the barriers to TDM use were removed, 79 out of the 81 respondents currently not practising TDM would perform it more frequently. Amongst them, 90.4% would check TDM for SLR, 61.6% for PNR, 28.8% when restarting after a drug holiday and 23.3% would check it proactively (Fig. 4). 79.3% of these would check TDM proactively at least once a year if all barriers were removed.
Fig. 4.
If barriers to therapeutic drug monitoring removed. PNR primary non-response, SLR secondary loss of response, TDM therapeutic drug monitoring
Discussion
This is the first National Survey of TDM use amongst clinicians treating IBD in India and only the third such survey in the world; so far, only two studies, one from the USA and the other from UK, has been published, underscoring the general lack of information on clinicians’ attitudes, perceptions and barriers to the use of anti-TNF TDM [35, 36].
Despite an increasing therapeutic armamentarium to treat IBD including biologicals and small molecule, treatment options in IBD remain largely limited as compared to other immune-mediated diseases. Moreover, anti-TNF therapies are currently the only available biological treatment option in India, with anti-integrin therapy (VDZ) likely to become available imminently. Progressive nature of IBD and the risk of attrition of response when changing from one treatment to another emphasize the need to optimize therapy before changing drugs within or outside class. Substantial variation in anti-TNF drug exposure and response to treatment underscores the importance of treatment optimization based on TDM. Consequently, TDM has emerged as the new standard of care for optimizing anti-TNF therapy in IBD, with reactive TDM being endorsed for assessment of PNR and SLR by recent international guidelines [26–28, 32–34]. Despite this, its use in clinical practice, since it first became available for use in 2016 in India, has not been assessed.
We found that only 20% of respondents reported using TDM in their practice. This is in contrast to 90.1% and 96.6% in the USA and UK studies, respectively [35, 36]. Of those respondents using TDM, 89.5% used it to assess for secondary loss of response and 73.7% for primary non-response. These figures are comparable with the recent outcomes from a Western population of IBD clinicians (96% and 72% in UK, 87% and 66% in USA).
Working in smaller (tier 2) cities, having between 11% and 25% of one’s practice made up of IBD patients and seeing/treating a higher number of IBD patients per month were factors independently associated with using TDM. This suggests that clinicians working in more manageable environments (lower overall population of patients and lower burden of IBD) who are able to follow up their IBD patients more frequently are more likely to use TDM. This contrasts to the UK study, which identified an association between clinicians having a larger IBD patient population (> 50% of their practice) and TDM use [36].
Only 5.3% (n = 1) (compared with 54% and 36.6% in UK and US surveys, respectively) used TDM proactively [35, 36]. Asia Pacific guidance on the use of biologics supports reactive TDM in patients with active IBD to help guide management [37]. However, there is a growing body of evidence supporting proactive TDM during maintenance treatment [19, 21, 22, 38].
We assessed the predominant barriers to the use of TDM amongst our respondents and found these to be cost (71.2%), availability (67.8%), time lag from serum sampling to results (58.7%) and “cumbersome” nature of performing TDM (45.7%). Time lag to results was the only barrier common to clinicians working in UK, USA and India [35, 36]. In addition to the cost of the biologic agent, which is approximately INR 18,000 (biosimilar) to INR 24,000 (originator) per month, for a 60-kg person during maintenance, the cost of TDM is INR 14,000 for measurement of both drug levels and drug antibodies. Respondents may also be deliberating the need for repeated monitoring when considering cost as a barrier to TDM use. From our respondents not using TDM, if all barriers were removed, 97.5% would start using it as an IBD management strategy. Of them, most would use it reactively but up to one quarter would use TDM proactively. Therefore, a more affordable ‘point of care’ assay would enable wider adoption of TDM-based treatment optimization.
Lack of knowledge, awareness of guidelines and evidence were not barriers to the use of TDM in the majority of respondents from India. This probably highlights the fact that IBD care is a niche area practiced by clinicians with a special interest. Presumably, these clinicians are well aware of current guidelines on the management of IBD patients. Notably, a study exploring understanding and interpretation of TDM using TDM-based clinical scenarios demonstrated marked heterogeneity in its practical use, understanding and interpretation [39]. This makes sense when one acknowledges that TDM is a relatively newer concept, albeit integrated through evidence into standard of care, and that its use may still be limited by experience and awareness of various assays and the heterogeneity therein. It also makes a compelling case for a more robust approach through multidisciplinary care provided by experienced IBD clinicians and an unmet training need.
Additionally, the potential for population pharmacokinetics to identify parameters and sources of variability with dosing may enable clinicians to apply individual dosing schedules using a dashboard system to calculate the exact dose a patient should receive and at what time to maintain optimal drug concentrations [40, 41]. Meanwhile, ‘point of care’ assays may be able to rapidly measure trough concentrations enabling efficacy through speedy and accurate dose optimization [25, 42]. Reassuringly, TDM has been shown to be cost-effective compared to empiric dose escalation [29–31].
A significant strength of our survey is the involvement of respondents across different practice settings and experience levels in India. Despite our wide reach through the ISG membership, we acknowledge the possibility of a selection bias, which may apply to most survey-based studies [35, 36]. We were limited by the overall number of IBD patients treated by individual clinicians. This resulted in a significant number of respondents being excluded from the study, a small number of respondents looking after > 25% IBD patients within their practice and few treating more than 5 patients per month with an anti-TNF. Consequently, our sample size was small.
Inconsistencies with the use of TDM for anti-TNF therapy, despite international guidelines endorsing their applicability for optimizing therapy, are an important area of unmet need, which should be addressed through educational initiatives, seminars and publication with wide access to practising gastroenterologists [26–28, 32–34, 39–41]. Collaborative working, discussion within IBD multidisciplinary teams and access to more specialized units will promote best practice and achieve more optimal patient outcomes.
In conclusion, we found that only 1 in 5 surveyed gastroenterologists in India are using TDM within their IBD practice. Significant barriers were availability, cost and time lag between test and results. Removal of these barriers would result in almost all clinicians using TDM at least reactively and a shift from 5% to around 25% using proactive TDM. The development of low-cost assays would inevitably result in a surge in TDM use paralleling the effect that biosimilars have had in increasing biologic use in the West. Meanwhile, dashboard systems and novel approaches using population pharmacokinetics may serve to optimize drug exposure through predictive modelling.
The rising incidence and prevalence of IBD in India, coupled with increasing complexity of disease phenotypes and availability of biosimilar versions of anti-TNF, will improve access to therapy making a compelling argument to optimize available therapies to enable best possible patient outcomes. The real-world impact of these rapid strides and the altruistic pursuit of meaningful targets, however, hinges on the wider adoption of treatment optimization in practice and, in doing so, exemplifying the virtues of personalized medicine.
Electronic supplementary material
(PDF 57 kb)
Acknowledgements
We acknowledge the support of the Indian Society of Gastroenterology for circulating the study questionnaire to its members.
Author contribution
All authors approved the final version.
Compliance with ethical standards
Conflict of interest
RNP, GBN, RGJ, DD, GM, VA, and JKM declare that they have no conflict of interest.
Disclosures
The authors have no relevant disclosures.
Ethics statement
The study was performed conforming to the Helsinki declaration of 1975, as revised in 2000 and 2008 concerning human and animal rights, and the authors followed the policy concerning informed consent as shown on Springer.com.
Disclaimer
The authors are solely responsible for the data and the contents of the paper. In no way, the Honorary Editor-in-Chief, Editorial Board Members, or the printer/publishers are responsible for the results/findings and content of this article.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Cholapranee A, Hazlewood GS, Kaplan GG, Peyrin-Biroulet L, Ananthakrishnan AN. Systematic review with meta-analysis: comparative efficacy of biologics for induction and maintenance of mucosal healing in Crohn’s disease and ulcerative colitis controlled trials. Aliment Pharmacol Ther. 2017;45:1291–302. [DOI] [PMC free article] [PubMed]
- 2.Feagan BG, Reinisch W, Rutgeerts P, et al. The effects of infliximab therapy on health-related quality of life in ulcerative colitis patients. Am J Gastroenterol. 2007;102:794–802. [DOI] [PubMed]
- 3.Peyrin-Biroulet L, Sandborn W, Sands BE, et al. Selecting therapeutic targets in inflammatory bowel disease (STRIDE): determining therapeutic goals for treat-to-target. Am J Gastroenterol. 2015;110:1324–38. [DOI] [PubMed]
- 4.Shah SC, Colombel J-F, Sands BE, Narula N. Mucosal healing is associated with improved long-term outcomes of patients with ulcerative colitis: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2016;14:1245–55. [DOI] [PubMed]
- 5.Shah SC, Colombel J, Sands BE, Narula N. Systematic review with meta-analysis: mucosal healing is associated with improved long-term outcomes in Crohn’s disease. Aliment Pharmacol Ther. 2016;43:317–33. [DOI] [PubMed]
- 6.Baert F, Noman M, Vermeire S, et al. Influence of immunogenicity on the long-term efficacy of infliximab in Crohn’s disease. N Engl J Med. 2003;348:601–8. [DOI] [PubMed]
- 7.Ben-Horin S, Chowers Y. Review article: loss of response to anti-TNF treatments in Crohn’s disease. Aliment Pharmacol Ther. 2011;33:987–95. [DOI] [PubMed]
- 8.Kennedy NA, Heap GA, Green HD, et al. Predictors of anti-TNF treatment failure in anti-TNF-naive patients with active luminal Crohn’s disease: a prospective, multicentre, cohort study. Lancet Gastroenterol Hepatol. 2019;4:341-53. [DOI] [PubMed]
- 9.Ordás I, Mould DR, Feagan BG, Sandborn WJ. Anti-TNF monoclonal antibodies in inflammatory bowel disease: pharmacokinetics-based dosing paradigms. Clin Pharmacol Ther. 2012;91:635–46. [DOI] [PubMed]
- 10.Bortlik M, Duricova D, Malickova K, et al. Infliximab trough levels may predict sustained response to infliximab in patients with Crohn’s disease. J Crohn's Colitis. 2013;7:736–43. [DOI] [PubMed]
- 11.Cornillie F, Hanauer SB, Diamond RH, et al. Postinduction serum infliximab trough level and decrease of C-reactive protein level are associated with durable sustained response to infliximab: a retrospective analysis of the ACCENT I trial. Gut. 2014;63:1721–7. [DOI] [PMC free article] [PubMed]
- 12.Levesque BG, Greenberg GR, Zou G, et al. A prospective cohort study to determine the relationship between serum infliximab concentration and efficacy in patients with luminal Crohn’s disease. Aliment Pharmacol Ther. 2014;39:1126–35. [DOI] [PubMed]
- 13.Papamichael K, Rakowsky S, Rivera C, Cheifetz AS, Osterman MT. Association between serum infliximab trough concentrations during maintenance therapy and biochemical, endoscopic, and histologic remission in Crohn’s disease. Inflamm Bowel Dis. 2018;24:2266–71. [DOI] [PMC free article] [PubMed]
- 14.Papamichael K, Rakowsky S, Rivera C, Cheifetz AS, Osterman MT. Infliximab trough concentrations during maintenance therapy are associated with endoscopic and histologic healing in ulcerative colitis. Aliment Pharmacol Ther. 2018;47:478–84. [DOI] [PMC free article] [PubMed]
- 15.Yarur AJ, Kanagala V, Stein DJ, et al. Higher infliximab trough levels are associated with perianal fistula healing in patients with Crohn’s disease. Aliment Pharmacol Ther. 2017;45:933–40. [DOI] [PubMed]
- 16.Strik AS, Löwenberg M, Buskens CJ, et al. Higher anti-TNF serum levels are associated with perianal fistula closure in Crohn’s disease patients. Scand J Gastroenterol. 2019;54:453–8. [DOI] [PubMed]
- 17.Paul S, Moreau AC, Del Tedesco E, et al. Pharmacokinetics of adalimumab in inflammatory bowel diseases: a systematic review and meta-analysis. Inflamm Bowel Dis. 2014;20:1288–95. [DOI] [PubMed]
- 18.Juncadella A, Papamichael K, Vaughn BP, Cheifetz AS. Maintenance adalimumab concentrations are associated with biochemical, endoscopic, and histologic remission in inflammatory bowel disease. Dig Dis Sci. 2018;63:3067–73. [DOI] [PMC free article] [PubMed]
- 19.Vande Casteele N, Ferrante M, Van Assche G, et al. Trough concentrations of infliximab guide dosing for patients with inflammatory bowel disease. Gastroenterology. 2015;148:1320–9. [DOI] [PubMed]
- 20.D’Haens G, Vermeire S, Lambrecht G, et al. Increasing infliximab dose based on symptoms, biomarkers, and serum drug concentrations does not increase clinical, endoscopic, and corticosteroid-free remission in patients with active luminal Crohn’s disease. Gastroenterology. 2018;154:1343–51. [DOI] [PubMed]
- 21.Papamichael K, Vajravelu RK, Vaughn BP, Osterman MT, Cheifetz AS. Proactive infliximab monitoring following reactive testing is associated with better clinical outcomes than reactive testing alone in patients with inflammatory bowel disease. J Crohn's Colitis. 2018;12:804–10. [DOI] [PMC free article] [PubMed]
- 22.Vaughn BP, Martinez-Vazquez M, Patwardhan VR, Moss AC, Sandborn WJ, Cheifetz AS. Proactive therapeutic concentration monitoring of infliximab may improve outcomes for patients with inflammatory bowel disease: results from a pilot observational study. Inflamm Bowel Dis. 2014;20:1996–2003. [DOI] [PMC free article] [PubMed]
- 23.Jorgensen KK, Olsen IC, Goll GL, et al. Switching from originator infliximab to biosimilar CT-P13 compared with maintained treatment with originator infliximab (NOR-SWITCH): a 52-week, randomised, double-blind, non-inferiority trial. Lancet. 2017;389:2304–16. [DOI] [PubMed]
- 24.Fiorino G, Ruiz-Argüello MB, Maguregui A, et al. Full interchangeability in regard to immunogenicity between the infliximab reference biologic and biosimilars CT-P13 and SB2 in inflammatory bowel disease. Inflamm Bowel Dis. 2018;24:601–6. [DOI] [PubMed]
- 25.Strik AS, Berends SE, Löwenberg M. Therapeutic drug monitoring-based dosing of TNF inhibitors in inflammatory bowel disease: the way forward? Expert Rev Clin Pharmacol. 2019;12:885–91. [DOI] [PubMed]
- 26.Feuerstein JD, Nguyen GC, Kupfer SS, Falck-Ytter Y, Singh S, American Gastroenterological Association Institute Clinical Guidelines Committee. American Gastroenterological Association Institute Guideline on Therapeutic Drug Monitoring in inflammatory bowel disease. Gastroenterology. 2017;153:827–34. [DOI] [PubMed]
- 27.Mitrev N, Vande Casteele N, Seow CH. IBD Sydney Organisation and the Australian Inflammatory Bowel Diseases Consensus Working Group. Review article: consensus statements on therapeutic drug monitoring of anti-tumour necrosis factor therapy in inflammatory bowel diseases. Aliment Pharmacol Ther. 2017;46:1037–1053. doi: 10.1111/apt.14368. [DOI] [PubMed] [Google Scholar]
- 28.Papamichael K, Cheifetz AS, Melmed GY, et al. Appropriate therapeutic drug monitoring of biologic agents for patients with inflammatory bowel diseases. Clin Gastroenterol Hepatol 2019;17:1655–68. [DOI] [PMC free article] [PubMed]
- 29.Steenholdt C, Brynskov J, Thomsen OØ, et al. Individualised therapy is more cost-effective than dose intensification in patients with Crohn’s disease who lose response to anti-TNF treatment: a randomised, controlled trial. Gut. 2014;63:919–27. [DOI] [PubMed]
- 30.Velayos FS, Kahn JG, Sandborn WJ, Feagan BG. A test-based strategy is more cost effective than empiric dose escalation for patients with Crohn’s disease who lose responsiveness to infliximab. Clin Gastroenterol Hepatol. 2013;11:654–66. [DOI] [PubMed]
- 31.Martelli L, Olivera P, Roblin X, Attar A, Peyrin-Biroulet L. Cost-effectiveness of drug monitoring of anti-TNF therapy in inflammatory bowel disease and rheumatoid arthritis: a systematic review. J Gastroenterol. 2017;52:19–25. [DOI] [PubMed]
- 32.Lichtenstein GR, Loftus EV, Isaacs KL, Regueiro MD, Gerson LB, Sands BE. ACG clinical guideline: management of Crohn’s disease in adults. Am J Gastroenterol. 2018;113:481–517. [DOI] [PubMed]
- 33.Magro F, Gionchetti P, Eliakim R, et al. Third European evidence-based consensus on diagnosis and management of ulcerative colitis. Part 1: definitions, diagnosis, extra-intestinal manifestations, pregnancy, cancer surveillance, surgery, and ileo-anal pouch disorders. J Crohn's Colitis. 2017;11:649–70. [DOI] [PubMed]
- 34.Lamb CA, Kennedy NA, Raine T, Hendy PA, Smith PJ, Limdi JK, Hayee B’H, Lomer MCE, Parkes GC, Selinger C, Barrett KJ, Davies RJ, Bennett C, Gittens S, Dunlop MG, Faiz O, Fraser A, Garrick V, Johnston PD, Parkes M, Sanderson J, Terry H, IBD guidelines eDelphi consensus group. Gaya DR, Iqbal TH, Taylor SA, Smith M, Brookes M, Hansen R, Hawthorne AB. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults. Gut. 2019;68:s1–106. doi: 10.1136/gutjnl-2019-318484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Grossberg LB, Papamichael K, Feuerstein JD, Siegel CA, Ullman TA, Cheifetz AS. A survey study of gastroenterologists’ attitudes and barriers toward therapeutic drug monitoring of anti-TNF therapy in inflammatory bowel disease. Inflamm Bowel Dis. 2017;24:191–7. [DOI] [PubMed]
- 36.Nigam GB, Nayeemuddin S, Kontopantelis E, Hayee B, Limdi JK. UK National Survey of Gastroenterologists’ attitudes and barriers toward therapeutic drug monitoring of anti-TNF therapy in inflammatory bowel disease. Frontline Gastroenterol. 2020;flgastro-2019-101372. [DOI] [PMC free article] [PubMed]
- 37.Ooi CJ, Hilmi I, Banerjee R, et al. Best practices on immunomodulators and biologic agents for ulcerative colitis and Crohn’s disease in Asia. Int Res. 2019;34:1296–315. [DOI] [PubMed]
- 38.Papamichael K, Chachu KA, Vajravelu RK, et al. Improved long-term outcomes of patients with inflammatory bowel disease receiving proactive compared with reactive monitoring of serum concentrations of infliximab. Clin Gastroenterol Hepatol. 2017;15:1580–88.e3. [DOI] [PMC free article] [PubMed]
- 39.Samaan MA, Arkir Z, Ahmad T, Irving PM. Wide variation in the use and understanding of therapeutic drug monitoring for anti-TNF agents in inflammatory bowel disease: an inexact science? Expert Opin Biol Ther. 2018;18:1271–9. [DOI] [PubMed]
- 40.Dubinsky MC, Phan BL, Singh N, Rabizadeh S, Mould DR. Pharmacokinetic dashboard-recommended dosing is different than standard of care dosing in infliximab-treated pediatric IBD patients. AAPS J. 2017;19:215–22. [DOI] [PubMed]
- 41.Wojciechowski J, Upton RN, Mould DR, Wiese MD, Foster DJR. Infliximab maintenance dosing in inflammatory bowel disease: an example for in Silico assessment of adaptive dosing strategies. AAPS J. 2017;19:1136–47. [DOI] [PubMed]
- 42.Van Stappen T, Bollen L, Vande Casteele N, et al. Rapid test for infliximab drug concentration allows immediate dose adaptation. Clin Transl Gastroenterol. 2016;7:e206. [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(PDF 57 kb)