ABSTRACT
Klebsiella pneumoniae carbapenemase (KPC)-producing K. pneumoniae (KPC-KP) have disseminated worldwide and emerged as major threats to public health. Of epidemiological significance, the international pandemic of KPC-KP is primarily associated with CG258 isolates and blaKPC-IncF plasmids. CRISPR-Cas system is an adaptive immune system that can hinder gene expansion driven by horizontal gene transfer. Because of blaKPC-IncF plasmids are favored by CG258 K. pneumoniae, it was of interest to examine the co-distribution of CRISPR and blaKPC-IncF plasmids in such isolates. We collected 459 clinical K. pneumoniae isolates in China and collected 203 global whole-genome sequences in GenBank to determine the prevalence of CRISPR-Cas systems. We observed that CRISPR-Cas system was significantly scarce in the CG258 lineage and blaKPC-positive isolates. Furthermore, the results of conjugation and plasmid stability assay fully demonstrated the CRIPSR-Cas system in K. pneumoniae could effectively hindered blaKPC-IncF plasmids invasion and existence. Notably, most blaKPC-IncF plasmids were also proved to be good targets of CRISPR owing to carry matched and functional protospacers and PAMs. Overall, our work suggests that type I-E CRISPR-Cas systems could impact the spread of blaKPC in K. pneumoniae populations, and the scarcity of CRISPR-Cas system was one of potential factors leading to the propagation of blaKPC-IncF plasmids in CG258 K. pneumoniae.
KEYWORDS: CRISPR-Cas, Klebsiella pneumoniae clonal complex 258, carbapenem resistance, plasmids, horizontal gene transfer
Introduction
Since they were first identified in 2001 [1], Klebsiella pneumoniae carbapenemase (KPC)-producing K. pneumoniae (KPC-KP) have emerged as important nosocomial pathogens and causes of global public health concern because of their prevalence and the associated high rate of mortality [2–5] Therefore, controlling the dissemination of KPC-KP became a critical global issue. Interestingly, although the carbapenemase-encoding blaKPC-harboring plasmid has been detected in numerous K. pneumoniae sequence types (STs), the pandemic of KPC-KP is mainly associated with the clonal group 258 (CG258), which includes ST258, ST11, ST340, ST512, and others [2,6]. In China, the epidemic of KPC-KP is primarily restricted to ST11 K. pneumoniae [7]. Furthermore, blaKPC is mostly located on the incompatibility group F (IncF) plasmids(Figure S2A) [2,7,8], although other plasmid scaffolds harbouring blaKPC-like genes (e.g. IncI2, IncX, IncA/C, IncR, and ColE1) have been identified among CG258 isolates [2,7,8]. The global dissemination of KPC-KP is strongly related to K. pneumoniae CG258 and blaKPC-IncF epidemic plasmids. Although the reasons behind this phenomenon are unclear, the ability of the CG258–blaKPC-IncF linkage to spread swiftly is beyond dispute.
Previous studies have shown that in addition to clonal dissemination, horizontal gene transfer (HGT) also contributes significantly to the pandemic dissemination of the blaKPC gene [2,7,9]. The clustered regularly interspaced short palindromic repeats (CRISPRs) are part of the adaptive immune system in diverse bacteria and archaea, which can cleave foreign DNA in a programmable and sequence-specific manner, and are disadvantageous for HGT-driven gene expansion [10–12]. Thus, we proposed a hypothesis that high-risk CG258 lineage may lack or have lost such endogenous barriers (CRISPR) to HGT. Antibiotic use unintentionally selects for outgrowth of these immunocompromised strains with enhanced abilities to acquire blaKPC genes, thereby assisting their rapid adaptation the hospital environment.
The new CRISPR-Cas classification of CRISPR-Cas includes two classes, five types and 16 subtypes[13]. By analyzing 203 whole genome sequences of K. pneumoniae in GenBank (Supplementary data 1a), only type I-E CRISPR systems were identified [14–16]. The mechanism for type I-E CRISPR-Cas genome defense has been recently reviewed [10,16]and is summarized as follows. CRISPR loci generally consists of short repeat sequences interspersed with unique spacer sequences that are homologous to sequences of invading DNA (“proto-spacers”) and a set of genes encoding nucleases (cas genes) are typically located near the CRISPR. Type I-E CRISPR systems possess eight cas genes (cas1, cas2, cas3, cse1, cse2, cas7e, cas5e, and cas6e) and either one or two CRISPR arrays [14]. On the basis of different layouts, type I-E CRISPR systems were further classified into two distinctive subtypes, type I-E and type I-E*[14]. Type I-E (in the cysH-iap region) is the canonical type I-E CRISPR-Cas system, while the other (Type I-E*, in ABC transport system-glyoxalase region) is variable, occasionally with a transposase-encoding gene integrated into the cas operon (Figure S1). Among the cas genes, cse1, cse2, cas5e, cas6e and cas7e encode proteins required for forming a CRISPR-associated complex for antiviral defense (Cascade) [13,14]. The Cascade–CRISPR RNA(crRNA) complex recognizes and binds to the foreign DNA, and then recruits the Cas3 protein for DNA degradation [13,17]. In addition, for foreign DNA, the matched proto-spacers (for targeted) and functional proto-spacer adjacent motifs (PAMs) (for distinguishing between self and foreign DNA) are necessary for CRISPR interference [18,19].
By analyzing the whole genome sequences of a series of K. pneumoniae, we have found 71 identified CRISPR loci of K. pneumoniae[16] (supplementary data1a) through the CRISPR finder[20]. Furthermore, bioinformatics analysis of 121 blaKPC-bearing plasmids (supplementary data 1b) also showed that the proto-spacer-positive plasmids were more commonly identified in the IncF group (Figure S2B), which possessed 14 proto-spacers (supplementary data 1c) matched for the CRISPR system in K. pneumoniae, than in the non-IncF group. Several matched proto-spacers identified from blaKPC-IncF plasmids suggest that these CRISPR-Cas systems may strongly associated with interfering with the survival of blaKPC-IncF plasmids. However, whether the existence of CRISPR in K. pneumoniae could impede the incursion and survival of such plasmids effectively remained unclear.
In this study, we explored the presence of type I-E CRISPR-Cas system in K. pneumoniae, especially focused on the CG258 lineage. We used the conjugation and plasmid stability assays to explore the function of CRISPR in perturbing the dissemination of the blaKPC-IncF plasmids. Furthermore, we also analyzed whether all blaKPC-IncF plasmids in this study would be good targets for such CRISPR systems. Hence, our goal was to further elucidate the function of CRISPR-Cas in anti- blaKPC-IncF-plasmid immunity and indicate possible associations between the scarcity of CRISPR-Cas system and globally successful dissemination of CG258 harbouring blaKPC plasmids.
Materials and Methods
Bacterial strains
Four hundred and fifty-nine non-duplicated K. pneumoniae isolates were randomly isolated from individual patients at seven hospitals in six Chinese provinces, which represented different rates of carbapenem-resistance. (Figure S3), from January 2017 to February 2018. Among these, 247 carbapenem-resistant K. pneumoniae (CR-KP) and 212 carbapenem-sensitive K. pneumoniae (CS-KP) were collected contemporaneously from similar departments (Supplementary data 1d). The presence of blaKPC gene about these strains was determined by the pair of primers listed in Table S1. Susceptibility testing was performed using the VITEK 2 system (bioMérieux, La Balme-les-Grottes, France) or using the broth microdilution method per the Clinical and Laboratory Standards Institute (CLSI) guidelines [21]. Multilocus sequence typing (MLST) was performed according to the protocol described on the Pasteur Institute MLST website for K. pneumoniae. Plasmid incompatibility type was identified by comparing with information in the Plasmid MLST locus/sequence definitions database (https://pubmlst.org/bigsdb?db=pubmlst_plasmid_seqdef). All strains and plasmids used in this study are listed in Table S1.
Bioinformatics analysis
All K. pneumoniae complete genome sequences are publicly available (203 in total) and the sequences of the 121 blaKPC-positive plasmids were downloaded from the NCBI database in April 2018. CRISPR finder [20] was used with default parameters to identify the CRISPR loci in the genomes and determine the number and sequences of the spacers within CRISPR repeat arrays. Nucleotide BLAST was used to identify the Cas genes upstream and downstream of the CRISPR loci. Nucleotide BLAST was also used to search for matched protospacers with a minimum of 90% homology (29/32 nucleotides) on 121 blaKPC-positive plasmids. The proto-spacers and the PAMs located at the 5’end of proto-spacers on the plasmids were searched using Nucleotide BLAST. WebLogo [22] was used to analyze PAMs on blaKPC-IncF plasmids.
Prevalence of CRISPR-Cas systems
The prevalence of K. pneumoniae isolates with CRISPR-Cas was determined using polymerase chain reaction (PCR) with the primers listed in Table S2. The presence of Type I-E CRISPR-Cas were preliminarily tested by amplifying the cysH-iap (Type I-E) and the ABC transport system-glyoxalase region (Type I-E*), as all of the CRISPR-Cas identified in the complete genome sequences of K. pneumoniae available in GenBank were located in cysH-iap or ABC transport system-glyoxalase region(Figure S1), and then further confirmed by amplifying the conserved gene cas1 and cas3, respectively.
Construction of Escherichia coli BW25113 without and with the CRISPR-Cas system mutant strain
The JS681 (BW25113ΔCRISPR) strain, which delete complete original CRISPR-Cas systems in E.coli BW25113, was created also using the lambda RED recombination method as described previously[23]. The plasmid (pCRISPR-KP8) was assembled with three fragments: the two fragments (with 50-bp overlap) of CRISPR-Cas system were amplified from KP8 and the Pi-dependent plasmid backbone (KanR) with one FRT site was amplified from pKD4 using primers with 30-bp homology to CRISPR-Cas fragment of KP8. Then, JS683 (BW25113-KP8CRISPR) was obtained by integrating the CRISPR-Cas system of KP8 (pCRISPR-KP8) into the JS681 strain via FLP-mediated site-specific recombination as described previously[24]. (Figure S4B)
Construction of KP8 cas3 deletion mutants
The cas3 deletion KP8 mutants were constructed using the lambda RED recombination method as described previously [23]. Primers designed to eliminate specific DNA stretches are listed in Table S2. When necessary, the resistance cassette introduced using the gene targeting procedure was eliminated via recombination with plasmid pCP20.
Plasmid construction
The different proto-spacers and PAM sequences (along with same spacer) were synthesized by annealing single-stranded, complementary oligonucleotides and then cloned into a Bsa I site in the pUC-RP4 (Figure S4A). The RP4 Mob site increased the conjugation frequencies and amplified the variation among spacers as described previously[25]. All the primers and synthesized nucleotides used for cloning are listed in Table S2. All the plasmid fragments were combined using the in-fusion cloning method and the NEBuilder HiFi DNA assembly master mix (New England BioLabs).
Conjugation assay
The donors and recipients were cultured to the logarithmic phase, mixed in 1:1 ratio, and then resuspended in 20 µl MgSO4 (10 mM). The resuspension was spotted on the Luria Bertani (LB) plate and incubated at 37°C overnight. Subsequently, the serial dilutions were plated in media with appropriate antibiotics. The conjugation frequency was calculated as the number of transconjugants per recipient.
Analysis of plasmid stability
The strains transformed with the corresponding plasmids were grown overnight in LB broth with appropriate antibiotics. Next, 50 µl of the previous culture were inoculated in 5 ml fresh LB medium (containing 40 mM glucose) after every 12 h (37°C) for several passages[15,26]. For each culture, culture solution was serially diluted, spread on LB plates, and incubated at 37°C overnight and 100 colonies were collected from the LB plates. Single colonies were collected using tips and spotted on LB plates in the presence and absence of imipenem or chloramphenicol. The passages were discontinued only when the percentage of the resistant clones decreased to 50%. Then, the relative plasmid stability was calculated by comparing the number of colonies on the LB agar plate containing antibiotics with that on pure LB agar.
Statistics
Statistical significance was assessed using a two-tailed Student’s t-test, one-way analysis of variance (ANOVA) test, or chi-square test of the GraphPad Prism8 software. p < 0.05 was considered statistically significant.
Results
Type I-E CRISPR systems are extremely rare in KPC-KP and CG258 lineage
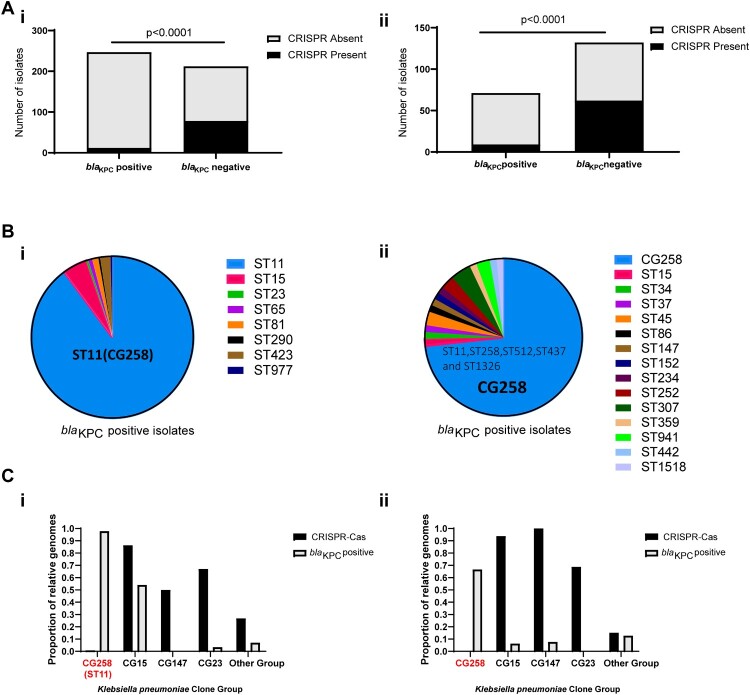
A total of 459 K. pneumoniae isolates was collected from six provinces in China. In parallel, 203 K. pneumoniae whole-genome sequences from worldwide and available over GenBank databases were analyzed to determine the distribution of the CRISPR-Cas loci in K. pneumoniae. We observed that the CRISPR-Cas loci were extremely rare in the KPC-positive isolates, whereas they were abundant among the KPC-negative isolates, either within the Chinese collection of clinical isolates (12/247 vs.78/212, p < 0.0001) or within the Genbank database (9/72 vs.62/131, p < 0.0001) (Figure 1A). These observations indicate a correlation between the scarcity of CRISPR-Cas system and K. pneumoniae with blaKPC.
Figure 1.
Presence of CRISPR-Cas system in blaKPC-positive /blaKPC-negative groups and CG258/non-CG258 isolates. (A) i. Presence of type I-E CRISPR systems in 459 Chinese clinical isolates collected in this study; ii. Presence of type I-E CRISPR systems in 203 completely sequenced strains available in GenBank. (B) i. MLSTs of blaKPC-positive group in459 Chinese clinical isolates; ii. MLSTs of blaKPC-positive group in 203 completely sequenced strains available in GenBank. (C) i. Presence of type I-E CRISPR systems among different clone groups in 459 Chinese clinical isolates collected in this study; ii. Presence of type I-E systems among different clone groups in 203 completely sequenced strains available in GenBank. p < 0.0001 indicate significant differences between two groups as determined using Chi-square (and Fisher’s exact) test with Bonferroni correction of the GraphPad Prism8 software.
Previous studies demonstrated that the pandemic of KPC-KP is mainly associated with the clonal group 258 (CG258) [2,6]. In this study, we also confirmed these features by analyzing 459 Chinese collection of clinical isolates and 203 global whole genome sequences (Figure 1B). The epidemic of blaKPC is primarily restricted to CG258 K. pneumoniae, but the sequence types of KPC-negative strains were diverse (Supplementary data 1a and Supplementary data 1d). Thus, we speculate the CG258 lineage may be immunocompromised strains lacking type I-E CRISPR systems.
We investigated the co-distribution of CRISPR and acquired blaKPC genes in different K. pneumoniae clone group, including high-risk CG258 group, MDR (Multidrug-Resistant) group (CG15 and CG147), hypervirulent group (CG23) and others. Consistent with our hypothesis, we observed that type I-E CRISPR systems were extremely rare in the CG258 lineages, either within the Chinese collection of clinical isolates or within the Global Genbank database (Figure 1C). Moreover, we found a significant inverse correlation between the presence of CRISPR and blaKPC genes in all K. pneumoniae clone groups. These findings indicated the lack of type I-E CRISPR systems in CG258 group may be related to the blaKPC genes dissemination in such high-risk lineage.
The CRISPR-Cas system in K. pneumoniae impedes IncF blaKPC-harboring plasmid conjugation
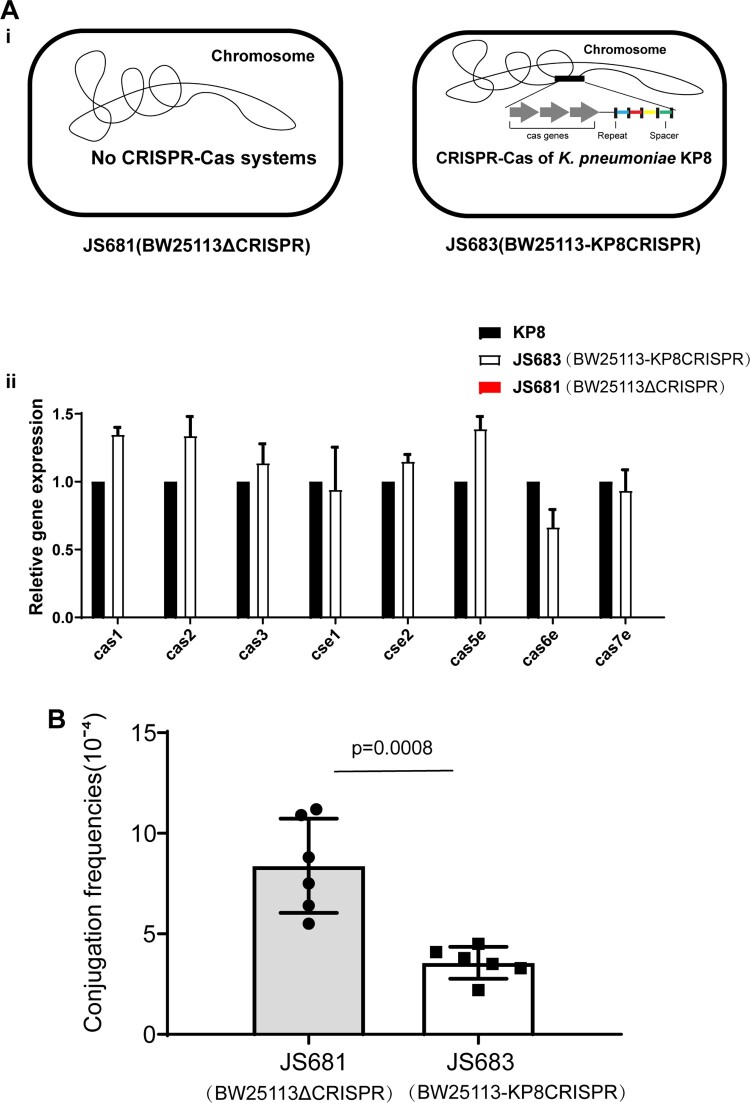
The paucity of the CRISPR-Cas system in both CG258 K. pneumoniae and KPC-positive isolates implied that CRISPR-Cas may be involved in preventing the acquisition of the blaKPC-harboring plasmid. We used conjugation assays to determine whether the CRISPR-Cas system in K. pneumoniae is disadvantageous for the dissemination of blaKPC- positive plasmids. The CRISPR-Cas system in the K. pneumoniae KP8 strain (type I-E, CP025636.1), being blaKPC-negative and belonging to ST458, was used as a model owing to its abundant matched spacers (Table S1). Escherichia coli BW25113 was used as recipient to generate recombinant strains with and without the CRISPR-Cas of strain KP8 (JS681 and JS683, Figure 2A). In addition, E. coli strain JS531 harbouring p187-2 (an IncF conjugative plasmid with blaKPC and matched proto-spacers, CP025468.1) was used as donor (Table. S1).
Figure 2.
Conjugation frequencies of p187-2 in BW25113 strains with or without KP8 CRISPR. (A) (i) Schematic of JS681 and JS683. (ii)Expression of the Cas operon in the KP8, JS683 and JS681 cells. (B) Effect of the KP8 CRISPR on the conjugation frequencies of p187-2(IncF conjugative plasmid with blaKPC and matched proto-spacers). The data represent the mean ± SD for six independent biological replicates. p = 0.0008 indicate significant differences between two groups as determined using two-tailed Student’s t-test.
Results of real-time PCR analysis confirmed that the KP8 Cas operon integrated in E. coli BW25113 was expressed successfully. Moreover, these results also proved the negative control JS681(BW25113ΔCRISPR) was useful since there was no Cas operon expression detected in such isolates (Figure 2B). Then, conjugation frequencies of the two E. coli recombinant strains were compared. As show in Figure 2C, the presence of the CRISPR-Cas of KP8 significantly decreased the conjugation frequencies in E. coli JS683 (BW25113-KP8CRISPR) cells by approximately 60%. This suggested that the type I-E CRISPR-Cas system in K. pneumoniae KP8 was involved in perturbing the acquisition of the IncF-blaKPC-harboring plasmids.
The CRISPR-Cas system affects the stability of plasmids harbouring matched spacers
In addition to the plasmid acquisition process, stable plasmid persistence in a given host also plays a critical role in maintenance of antibiotic resistance. Therefore, a plasmid stability test was performed to investigate the survival of target plasmids harbouring matched spacers in both KP8 and JS683 (BW25113-KP8CRISPR) cells. In case a sequence on a plasmid may be targeted by the Cascade–crRNA complex, plasmid replication should be hindered. Therefore, when grown in a medium without antibiotics, plasmid abundance should be reduced, a phenomenon reflected by the lower percentage of resistant clones recovered. Moreover, the interference of the CRISPR-Cas system was conversely estimated by looking at specific antibiotic susceptibility recovery.
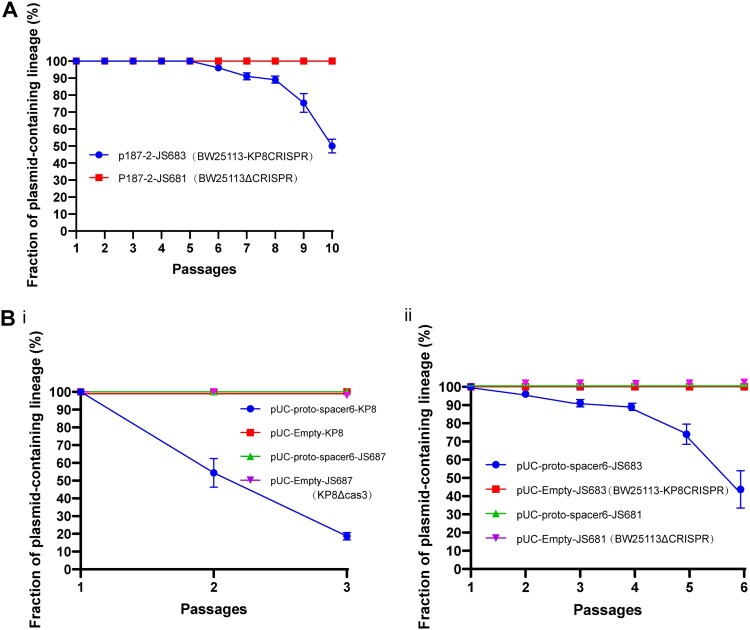
To investigate whether the CRISPR-Cas system of strain KP8 inhibited the presence of the blaKPC-IncF plasmids, the natural plasmid p187-2 was transformed into both E. coli JS683 (BW25113-KP8CRISPR) and E. coli JS681 (BW25113ΔCRISPR) strains and replication interference was assessed. It showed that the percentage of resistant clones in the CRISPR-positive strain ranged from 46% to 54% after 10 passages in LB medium, whereas the CRISPR-negative strain still retained the resistance plasmid after the same number of passages (Figure 3A). In agreement with the results of the plasmid stability assay, minimum inhibitory concentrations (MICs) of imipenem for these clones further confirmed that the CRISPR-Cas system of KP8 significantly and negatively contributed to restrain the retention of IncF-blaKPC plasmids (Table. S3).
Figure 3.
Plasmid stability in KP8 and JS683 (BW25113-KP8CRISPR) cells. (A) The E. coli strains JS683 and JS681 were transformed with a clinical plasmid p187-2 (an IncF conjugative plasmid with blaKPC and matched spacers). Plasmid stability experiment results during10 passages. The number of imipenem-resistant clones was lesser in the CRISPR-positive strain (blue line) at the 10th passage than in the CRISPR-negative strain (red line), in which the number of resistant clones were no altered. (B) Two chloramphenicol-resistant plasmids with or without matched spacer were transformed into KP8, JS683, and their CRISPR-mutant versions(JS687, KP8ΔCas3 and JS681,BW25113ΔCRISPR). Plasmid stability experiment results during 3 passages(i) or 6 passages(ii). The elimination rates of pUC-proto-spacer 6 in the CRISPR-positive strain (blue lines) decreased to variable extents, whereas that of pUC-empty in all strains were identical. The stability of all plasmids was identical in the CRISPR deletion strain(JS687, KP8ΔCas3 and JS681,BW25113ΔCRISPR). All experiments were conducted in triplicate.
As host differences may affect the CRISPR interference, two plasmids (chloramphenicol-resistant) were constructed with or without matched proto-spacers to further evaluate how CRISPR operate in the original K. pneumoniae strain (KP8). Furthermore, the KP8 Cas3 deletion mutant was created as the negative control. Both plasmids were transformed into K. pneumoniae KP8 and KP8 mutants and into E. coli BW25113 with or without CRISPR. This experiment showed that the maintenance ability of matched plasmids in the CRISPR-positive strains, especially in the K. pneumoniae KP8, decreased significantly compared to the negative strains and non-matched plasmid (Figure 3B). Those observations were in full agreement with obtained MIC values (Table. S3). Overall, those findings confirmed that this difference in term of plasmid stability was unequivocally related to the CRISPR-Cas system-mediated interference.
Proto-spacers located on blaKPC-IncF plasmids can be well-interfered by the CRISPR system
The interference between the CRISPR system and the occurrence of the IncF-blaKPC plasmids in K. pneumoniae was investigated. Our aim was to evaluate whether all the IncF blaKPC-harboring plasmids in this study were effectively targeted by CRISPR. Previous studies have shown that the matched proto-spacers on plasmids are the vital clues for CRISPR scanning and cleavage [12]. Therefore, degradation of plasmids depends on its proto-spacers.
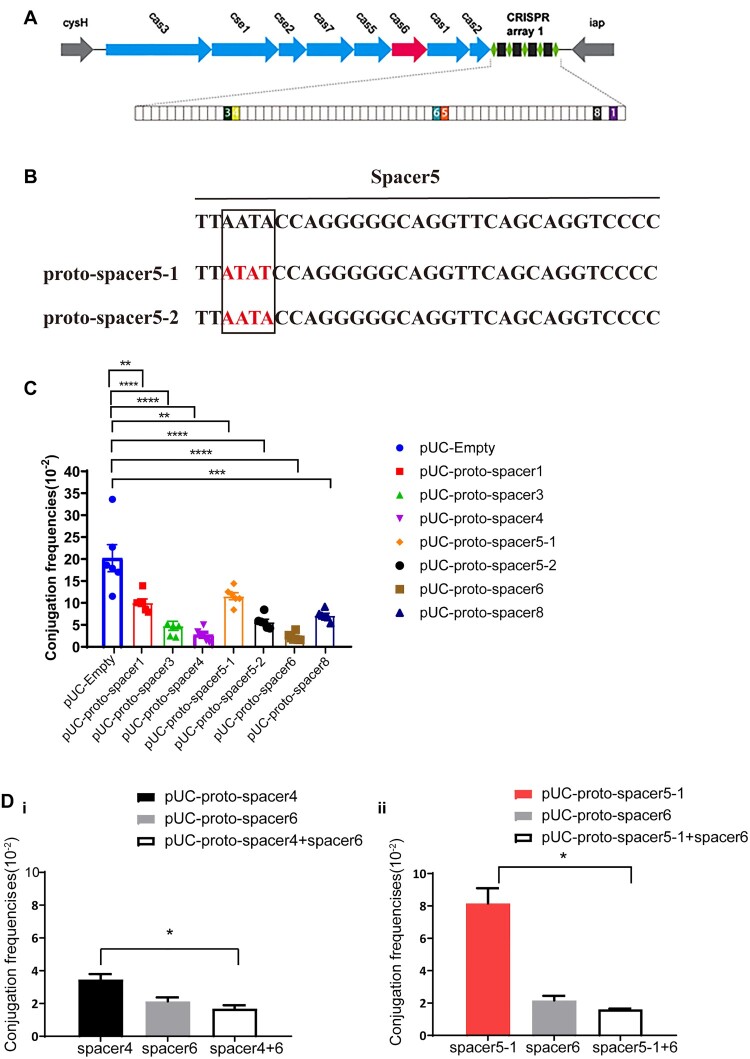
We have previously observed that almost all the blaKPC-IncF plasmids contain more than one type of proto-spacers that matched with the CRISPR array in K. pneumoniae (Supplementary data 1b). To investigate whether all types of proto-spacers present onto IncF plasmids can be targeted, the KP8 CRISPR was used as a model. Six types of proto-spacers (Table S4) that matched the KP8 CRISPR were used, of which proto-spacer 5 had two subtypes (Figure 4).
Figure 4.
Characteristics of proto-spacers located on the blaKPC -IncF plasmids. (A) Schematic showing the CRISPR-Cas system in KP8. Genes are depicted as arrows in different colours and the IncF plasmid-matched spacers are shown as colourful boxes. (B) Two subtypes of proto-spacer 5, in which the nucleotide sequence in red represents the base mutation. (C) Conjugation frequencies of different proto-spacers. (D) Conjugation frequencies of single and combined proto-spacers. The results of the conjugation assay are presented as means ± SD from six independent experiments. * p < 0.05, ** p < 0.01, *** p < 0.001, ****p < 0.0001 indicate significant differences between the strains and the control group (pUC-Empty) as determined using one-way ANOVA with Dunnett correction(C). Statistical significance between the two strains was assessed using a two-tailed Student’s t-test with Bonferroni correction of the GraphPad Prism8 software. * p < 0.05 was considered statistically significant (D).
Conjugation is a stable assay for assessing CRISPR-Cas activity. Hence, we used this assay to monitor how different types of sequences targeted by the crRNA affect plasmid replication. Seven plasmids harbouring variable proto-spacers were constructed and transformed into E. coli S17-1 λpir (RP4+) that was subsequently used as donor (Figure S4A) while E. coli JS683(BW25113-KP8CRISPR) was used as recipient for conjugative assays. Compared to the negative control (pUC-Empty), the frequencies of all seven plasmids decreased significantly, among which the frequency of proto-spacer 6 was reduced the most and that of the proto-spacer 5–1 was reduced the least (90% vs. 42.2%) (Figure 4C). Notably, frequencies of the well-paired proto-spacer 5–2 decreased by 51% compared to that of the proto-spacer 5–1 (containing 3 bp substitutions), although both belonged to the same type. These results indicated that all matched sequences can be targeted, albeit with variations in the CRISPR immune response.
The aforementioned results showed that not all plasmid-borne proto-spacers may be interfered in a similar manner, suggesting that CRISPR tend to leave multiple matched proto-spacers on one plasmid possibly because that may promote effective plasmid elimination. To account for this possibility, two recombinant plasmids were constructed: harbouring either plasmids containing two highly functional proto-spacers (proto-spacer 4 and proto-spacer 6) or containing the supposed strongest and weakest- functional proto-spacers (proto-spacer 5–1 and proto-spacer 6), respectively. Compared to the plasmids harbouring single proto-spacer4 or proto-spacer 5-1, the conjugation frequencies of the combined plasmids all decreased and were similar to the frequencies of proto-spacer 6 (Figure 4D). These findings indicated that the occurrence of high-active proto-spacer 6 made up for the low-active proto-spacers, resulting a more effective plasmid elimination.
Taken together, these results demonstrated that although the CRISPR system may specifically interfere with certain spacers effectively, multiple proto-spacers on a single plasmid may compensate for this bias and accelerate its elimination.
PAMs harboured by the blaKPC-IncF plasmids are conserved and functional
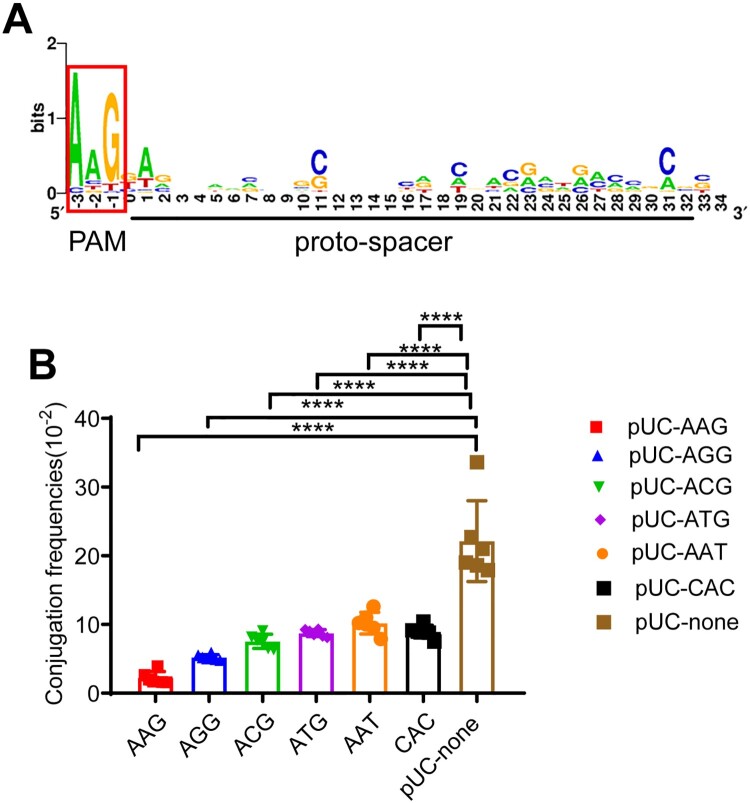
In addition to the matched pro-spacers, functional PAM is also essential for the Cascade–crRNA complex to target the plasmid. To investigate whether PAMs on IncF- blaKPC plasmids were conserved respect to the CRISPR system in K. pneumoniae, all the target proto-spacers of 54 IncF blaKPC-positive plasmids were analyzed (Supplementary data 1b). As shown in Figure 5A, there was a strong bias for the AAG motif, which confirmed the existence of CRISPR-specific PAMs.
Figure 5.
Characteristics of PAMs adjunct to the proto-spacers harboured by blaKPC-IncF plasmids. (A) WebLogo was used to analyze PAMs from blaKPC-IncF plasmids. The first nucleotide of the proto-spacer is at position 0. WebLog of the proto-spacer, as well as those 3 nucleotides upstream and downstream of the proto-spacer are shown. The relative letter size indicates the base frequency at that position. (B) Conjugation frequencies of proto-spacers with different PAMs. The data represent the mean ± SD for six independent biological replicates. * p < 0.05, ** p < 0.01, *** p < 0.001, ****p < 0.0001 indicate significant differences between the strains and the control group (pUC-none) as determined using one-way ANOVA with Dunnett correction.
As the AAG motif was the most abundant PAM (56.7%, 389/687) (Figure 5A), we speculated that it was also subject to the strongest CRISPR-interference. To verify this hypothesis, six plasmids were constructed containing different PAMs (AAG, AGG, ACG, ATG, AAT, and CAC) respectively (Figure S4A) originating from the IncF plasmids and conjugation assay were performed to assess their function. In agreement with our assumption, the frequency of degradation of the AAG-PAM (90.3%) was higher among the 6 PAMs (Figure.5B) compared to those of plasmids with no PAM (pUC-none). These observations further demonstrate that the functions of different PAMs are consistent with the PAM preferences of the CRISPR cascade.
Discussion
The widespread K. pneumoniae CG258 isolates are important human pathogens known to cause urinary tract infections, respiratory tract infections, and bloodstream infections, which have spread extensively throughout the world [2,6]. Several studies have demonstrated that the CG258 strains and related blaKPC-IncF plasmids are responsible for the sudden increase in the population of multidrug resistance among K. pneumoniae isolates [8,27]. However, little is known about the exact factors of CG258-blaKPC successful dissemination. Our results indicated the type I-E CRISPR-Cas systems impacted the spread of blaKPC-plasmids in K. pneumonia. The scarcity of type I-E CRISPR-Cas systems in CG258 lineage, allowing them to readily acquire and adapt to blaKPC-plasmids.
Type I-E CRISPR-Cas systems identified in K. pneumoniae are categorized into subtypes type I-E and type I-E*[14], and the anti-plasmid immunity in the type I-E*-CRISPR-positive K. pneumoniae NTUH-K2044 strain (ST23) have been well-described [15]. However, their relationship with blaKPC plasmids was not understood. Hence, in this study, we investigated the type I-E CRISPR prevalence in K. pneumoniae at present and observed dearth of drug-resistant K. pneumoniae CG258 strains. Furthermore, the proto-spacers harboured by the IncF-blaKPC plasmids, matched for CRISPR, were collected and analyzed. Results indicated that more than one matched sequence were present in all these plasmids (Supplementary data 1c). As the role of type I-E*-CRISPR activity in anti-plasmid immunity has already been confirmed [15], the type I-E CRISPR in the KP8 strain was selected as a model for further study.
Conjugation and plasmid stability assays were used to comprehensively assess the ability of CRISPR to impede the transmission of IncF-blaKPC plasmids. The results demonstrated that the CRISPR harboured by K. pneumoniae resulted in effective immunity to drug-resistance plasmids containing matched proto-spacers. The results of the plasmid stability assay also revealed that complete plasmid elimination required cumulative CRISPR interference, consistent with the results of former studies [15]. Interestingly, although the Cas operon transcript of an alternative isolate (JS683,E. coli BW25513-KP8 CRISPR) and original strain (K. pneumoniae KP8) were similar, according to the results of the plasmid stability assay the CRISPR system in KP8 cells was more competent than that in JS683(E.coli BW25513-KP8 CRISPR), which may be ascribed to differences in host regulators in these two organisms. In addition, this can explain the absence of a sharp decrease in the conjugation frequency of the substitute strains (E. coli) and the more significant decrease in the frequency of the KP8(K. pneumoniae) isolate could be speculated.
Previous studies have demonstrated that the IncF plasmids are the most predominant blaKPC-containing plasmid types [2,7]. In this study, we observed that the IncF-blaKPC plasmids were also favoured by the proto-spacers in the 121 blaKPC plasmids we studied (Supplementary data 1b and Figure S2B). To clarify whether the CRISPR system can effectively prevent blaKPC dissemination, we should further validate whether all the proto-spacers and PAMs harboured by blaKPC-IncF-plasmids are functionally recognized. Our results elucidated that all matched sequences can be targeted, although not all can be flawlessly interfered with. Many studies suggest that the base pairing at the −1 position and the seed region (nucleotides +1 to +5, +7, and +8) are critical for target recognition by Cascade [28], which is in agreement with the observation that proto-spacer 5–2 generated more effective immune response than proto-spacer 5–1 (Figure 4). In addition to the well base-paired, the positions at which spacers are inserted in the CRISPR array and spacer GC content (62.5% was optimal), are also essential for CRISPR interference activity [29]. Among these selected proto-spacers, the CRISPR-interference increased as the GC content in the spacer until reached 62.5% [29], and the more proximal a spacer is to the leader sequence, the more efficiently can it be transcribed [10,30]. Therefore, although each matched sequence was targeted, not all proto-spacers could trigger interference with equal efficiency. These observations explained the presence of multiple spacers in the plasmids, which were required to compensate for the presence of low-active mismatch-containing spacers [10]. Our observations also support the view that multiple proto-spacers on plasmids promote the efficiency of CRISPR-mediated degradation. In summary, almost all the blaKPC -IncF plasmids can be well-targeted as they harbour manifold proto-spacers.
Interestingly, both type I-E CRISPR-Cas systems identified in E. coli [31] and K. pneumoniae are prone to targeting the AAG PAM, and PAMs located on IncF plasmids predominantly contain the AAG motif (Figure 5A). In such CRISPR systems, the PAM selectivity of the CRISPR adaptation machinery has co-evolved with the CRISPR interference machinery; thus, both the structural basis of cas1-cas2 (adaption) and cse1 (interference) are responsible for PAM determination, which specifically recognize the AAG as the consensus PAM sequence [10,32]. In this study, we demonstrated that the AAG PAM was conserved in blaKPC-IncF plasmids and that the AAG motif showed the strongest CRISPR interference. As previous studies have demonstrated that only the proto-spacers were flanked by a functional PAM, they can be effectively recognized by the cascade [33]. Our observations regarding PAMs further verified that the blaKPC-IncF plasmids are good targets for CRISPR.
Although the CRISPR could influence the obtention of blaKPC-IncF plasmids in K. pneumoniae, the type I-E CRISPR systems were not flawless barriers to plasmid transfer. We could found other K. pneumoniae lineages such as CG 65, CG86 and CG40, also seem to lack type I-E CRISPR-Cas[33], but they were not favoured by blaKPC-IncF plasmids, that may be caused by polysaccharide capsule (CG 65 and CG86, hypervirulent clones) or harbouring R-M (Restriction-Modification) systems (the other defense system)[34]. Notably, in addition to the high-risk CG258 lineage, other clone groups including CG15 and CG147 also be considered to be important causes of carbapenem-resistant infections[35]. However, these MDR groups could acquire several resistance genes such as blaVIM, blaIMP and blaOXA-48[35], successfully evaded restriction by CRISPR systems. Moreover, previous studies also found CRISPR systems could not influence the invasion of all antibiotic resistance genes into K. pneumoniae [36]. These defense failures may be attributed to the no-matched spacers of invading resistance genes or involved with CRISPR tolerance[11].
Remarkably, unlike the inverse relationship we found between blaKPC genes and CRISPR, the coexistence of a CRISPR array and multiple copies of the same β-lactamase genes in the chromosome were found in several K. pneumoniae isolates [37]. These novel findings proposed a potential mechanism that CRISPR could promote the antimicrobial resistance gene mobilization from plasmids into the chromosome, through degradation the targeting plasmids[37]. As well as the CRISPR systems, the other immune systems (R-M systems) carried by the bacteria also play an important role in regulating the plasmid dissemination. Previous study in E. faecalis indicated that CRISPR-Cas defense and R-M defense individually contribute significantly to anti-plasmid genome defense[38]. Over all, these observations all verified that the CRISPR-Cas system was not the only limiting factor for controlling plasmid attack, as other differences between the resistance-plasmids and several host-specific variations may also affect plasmid intrusion, which will be the focus of future work.
In addition to exploring the relationship between blaKPC and CRISPR in K. pneumoniae, we also found the hypervirulent group, especially the CG23 lineage, was preferred by the type I-E CRISPR, in keeping with the results of former studies [36]. These interesting discoveries were also worth further study.
In conclusion, our work demonstrates the scarcity of type I-E CRISPR-Cas systems is a probable factor leading to blaKPC-IncF plasmids can be propagated in K. pneumoniae CG258 lineage. Meanwhile, antibiotic use inadvertently selects for these strains with blaKPC-plasmids, which provide a potential explanation for the CG258 epidemic success.
Supplementary Material
Acknowledgements
The authors thank Dr. Jian-feng Zhang and Dr. Ying-zhou Xie (State Key Laboratory for Microbial Metabolism and School of Life Sciences & Biotechnology, Shanghai Jiao Tong University, Shanghai, China) for kindly experiment assisting in this study.
Glossary
ABBREVIATIONS: CG258: Clonal group 258; CRISPR: Clustered regularly interspaced short palindromic repeats; IncF: Incompatibility group F; KPC: Klebsiella pneumoniae carbapenemase; KPC-KP: KPC-producing K. pneumoniae; LB: Luria Bertani; MICs: Minimum inhibitory concentrations; PCR: Polymerase chain reaction; PAMs: Proto-spacer adjacent motifs; ST: Sequence type
Funding Statement
This work was supported by research grants National Natural Science Foundation of China [grant number 81871692 and 81572031] and by the University of Fribourg and by the Science and Technology Commission of Shanghai Municipality [grant number 19JC1413002].
Disclosure statement
No potential conflict of interest was reported by the author(s).
References
- 1.McKenna M. Antibiotic resistance: the last resort. Nature. 2013 Jul 25;499(7459):394–396. doi: 10.1038/499394a [DOI] [PubMed] [Google Scholar]
- 2.Chen L, Mathema B, Chavda KD, et al. Carbapenemase-producing Klebsiella pneumoniae: molecular and genetic decoding. Trends Microbiol. 2014 Dec 1;22(12):686–696. doi: 10.1016/j.tim.2014.09.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kuehn BM. “Nightmare” bacteria on the rise in US hospitals, long-term care facilities. JAMA. 2013 Apr 7;309(15):1573–1574. doi: 10.1001/jama.2013.2922 [DOI] [PubMed] [Google Scholar]
- 4.Logan LK, Weinstein RA.. The epidemiology of carbapenem-resistant enterobacteriaceae: the impact and evolution of a global menace. J Infect Dis. 2017 Feb 15;215(suppl_1):S28–S36. doi: 10.1093/infdis/jiw282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Munoz-Price LS, Poirel L, Bonomo RA, et al. Clinical epidemiology of the global expansion of Klebsiella pneumoniae carbapenemases. Lancet Infect Dis. 2013 Sept 1;13(9):785–796. doi: 10.1016/S1473-3099(13)70190-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chen L, Mathema B, Pitout JD, et al. Epidemic Klebsiella pneumoniae ST258 is a hybrid strain. MBio. 2014 Jun 24;5(3):e1314–e1355. doi: 10.1128/mBio.01355-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fu P, Tang Y, Li G, et al. Pandemic spread of blaKPC-2 among Klebsiella pneumoniae ST11 in China is associated with horizontal transfer mediated by IncFII-like plasmids. Int J Antimicrob Agents. 2019 Aug;54(2):117–124. doi: 10.1016/j.ijantimicag.2019.03.014 [DOI] [PubMed] [Google Scholar]
- 8.Peirano G, Bradford PA, Kazmierczak KM, et al. Importance of clonal complex 258 and IncFK2-like plasmids among a global collection of Klebsiella pneumoniae with blaKPC. Antimicrob Agents Chemother. 2017 Apr 1;61(4):e02610-16. 10.1128/AAC.02610-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Adler A, Khabra E, Paikin S, et al. Dissemination of the blaKPC gene by clonal spread and horizontal gene transfer: comparative study of incidence and molecular mechanisms. J Antimicrob Chemother. 2016 Aug 1;71(8):2143–2146. doi: 10.1093/jac/dkw106 [DOI] [PubMed] [Google Scholar]
- 10.Amitai G, Sorek R.. CRISPR-Cas adaptation: insights into the mechanism of action. Nat Rev Microbiol. 2016 Feb 1;14(2):67–76. doi: 10.1038/nrmicro.2015.14 [DOI] [PubMed] [Google Scholar]
- 11.Hullahalli K, Rodrigues M, Nguyen UT, et al. An Attenuated CRISPR-Cas system in enterococcus faecalis permits DNA acquisition. MBio. 2018 May 1;9(3):e00414-18. doi: 10.1128/mBio.00414-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sampson TR, Saroj SD, Llewellyn AC, et al. A CRISPR/Cas system mediates bacterial innate immune evasion and virulence. Nature. 2013 May 9;497(7448):254–257. doi: 10.1038/nature12048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Makarova KS, Wolf YI, Alkhnbashi OS, et al. An updated evolutionary classification of CRISPR-Cas systems. Nat Rev Microbiol. 2015 Nov 1;13(11):722–736. doi: 10.1038/nrmicro3569 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Shen J, Lv L, Wang X, et al. Comparative analysis of CRISPR-Cas systems in Klebsiella genomes. J Basic Microbiol. 2017 Apr 1;57(4):325–336. doi: 10.1002/jobm.201600589 [DOI] [PubMed] [Google Scholar]
- 15.Lin TL, Pan YJ, Hsieh PF, et al. Imipenem represses CRISPR-Cas interference of DNA acquisition through H-NS stimulation in Klebsiella pneumoniae. Sci Rep. 2016 Aug 17;6:31644. doi: 10.1038/srep31644 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tang Y, Fu P, Zhou Y, et al. Absence of the type I-E CRISPR-Cas system in Klebsiella pneumoniae clonal complex 258 is associated with dissemination of IncF epidemic resistance plasmids in this clonal complex. J Antimicrob Chemother. 2020 Apr 1;75(4):890–895. doi: 10.1093/jac/dkz538 [DOI] [PubMed] [Google Scholar]
- 17.Hochstrasser ML, Taylor DW, Bhat P, et al. Casa mediates Cas3-catalyzed target degradation during CRISPR RNA-guided interference. Proc Natl Acad Sci U S A. 2014 May 6;111(18):6618–6623. doi: 10.1073/pnas.1405079111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Westra ER, Semenova E, Datsenko KA, et al. Type I-E CRISPR-cas systems discriminate target from non-target DNA through base pairing-independent PAM recognition. PLoS Genet. 2013 Jan 20;9(9):e1003742. doi: 10.1371/journal.pgen.1003742 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yosef I, Goren MG, Qimron U.. Proteins and DNA elements essential for the CRISPR adaptation process in Escherichia coli. Nucleic Acids Res. 2012 Jul 1;40(12):5569–5576. doi: 10.1093/nar/gks216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Grissa I, Vergnaud G, Pourcel C.. The CRISPRdb database and tools to display CRISPRs and to generate dictionaries of spacers and repeats. BMC Bioinformatics. 2007 May 23;8:172. doi: 10.1186/1471-2105-8-172 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Performance Standards for Antimicrobial Susceptibility Testing, 28th Edition. CLSI:M100-M128.
- 22.Crooks GE, Hon G, Chandonia JM, et al. Weblogo: a sequence logo generator. Genome Res. 2004 Jun 1;14(6):1188–1190. doi: 10.1101/gr.849004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Datsenko KA, Wanner BL.. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc Natl Acad Sci U S A. 2000 Jun 6;97(12):6640–6645. doi: 10.1073/pnas.120163297 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ellermeier CD, Janakiraman A, Slauch JM.. Construction of targeted single copy lac fusions using lambda Red and FLP-mediated site-specific recombination in bacteria. Gene. 2002 May 15;290(1-2):153–161. doi: 10.1016/S0378-1119(02)00551-6 [DOI] [PubMed] [Google Scholar]
- 25.van Aartsen JJ, Rajakumar K.. An optimized method for suicide vector-based allelic exchange in Klebsiella pneumoniae. J Microbiol Methods. 2011 Sept 1;86(3):313–319. doi: 10.1016/j.mimet.2011.06.006 [DOI] [PubMed] [Google Scholar]
- 26.Yang CD, Chen YH, Huang HY, et al. CRP represses the CRISPR/Cas system in Escherichia coli: evidence that endogenous CRISPR spacers impede phage P1 replication. Mol Microbiol. 2014 Jun 1;92(5):1072–1091. doi: 10.1111/mmi.12614 [DOI] [PubMed] [Google Scholar]
- 27.Tzouvelekis LS, Markogiannakis A, Psichogiou M, et al. Carbapenemases in Klebsiella pneumoniae and other Enterobacteriaceae: an evolving crisis of global dimensions. Clin Microbiol Rev. 2012 Oct 1;25(4):682–707. doi: 10.1128/CMR.05035-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Semenova E, Jore MM, Datsenko KA, et al. Interference by clustered regularly interspaced short palindromic repeat (CRISPR) RNA is governed by a seed sequence. Proc Natl Acad Sci U S A. 2011 Jun 21;108(25):10098–10103. doi: 10.1073/pnas.1104144108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fu BX, Wainberg M, Kundaje A, et al. High-throughput characterization of cascade type I-E CRISPR Guide Efficacy Reveals Unexpected PAM Diversity and target sequence preferences. Genetics. 2017 Aug 1;206(4):1727–1738. doi: 10.1534/genetics.117.202580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pougach K, Semenova E, Bogdanova E, et al. Transcription, processing and function of CRISPR cassettes in Escherichia coli. Mol Microbiol. 2010 Sept 1;77(6):1367–1379. doi: 10.1111/j.1365-2958.2010.07265.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Musharova O, Sitnik V, Vlot M, et al. Systematic analysis of type I-E Escherichia coli CRISPR-Cas PAM sequences ability to promote interference and primed adaptation. Mol Microbiol. 2019 Jun 1;111(6):1558–1570. doi: 10.1111/mmi.14237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hayes RP, Xiao Y, Ding F, et al. Structural basis for promiscuous PAM recognition in type I-E Cascade from E. coli. Nature. 2016 Feb 25;530(7591):499–503. doi: 10.1038/nature16995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Westra ER, van Erp PB, Kunne T, et al. CRISPR immunity relies on the consecutive binding and degradation of negatively supercoiled invader DNA by Cascade and Cas3. Mol Cell. 2012 Jun 8;46(5):595–605. doi: 10.1016/j.molcel.2012.03.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wyres KL, Wick RR, Judd LM, et al. Distinct evolutionary dynamics of horizontal gene transfer in drug resistant and virulent clones of Klebsiella pneumoniae. PLoS Genet. 2019 Apr 1;15(4):e1008114. doi: 10.1371/journal.pgen.1008114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.David S, Reuter S, Harris SR, et al. Epidemic of carbapenem-resistant Klebsiella pneumoniae in Europe is driven by nosocomial spread. Nat Microbiol. 2019 Jan 1;4(11):1919–1929. doi: 10.1038/s41564-019-0492-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Liao W, Liu Y, Chen C, et al. Distribution of CRISPR-Cas systems in clinical carbapenem-resistant Klebsiella pneumonia strains in a Chinese Tertiary Hospital and Its potential relationship with virulence. Microb Drug Resist. 2019 Dec 13. doi: 10.1089/mdr.2019.0276 [DOI] [PubMed] [Google Scholar]
- 37.Huang W, Wang G, Sebra R, et al. Emergence and Evolution of multidrug-resistant Klebsiella pneumoniae with both blaKPC and blaCTX-M integrated in the chromosome. Antimicrob Agents Chemother. 2017 Jul 1;61(7):e00076-17. doi: 10.1128/AAC.00076-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Price VJ, Huo W, Sharifi A, et al. CRISPR-Cas and restriction-modification act additively against conjugative antibiotic resistance plasmid transfer in enterococcus faecalis. mSphere. 2016 May 1;1(3):e00064-16. doi: 10.1128/mSphere.00064-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.