Abstract
Background
Angiostrongylus cantonensis is an important food-borne zoonotic parasite. Humans are non-permissive hosts, and this parasite develops into fifth-stage larvae (L5) in the brain and subarachnoid cavity and then induces eosinophilic meningitis and eosinophilic meningoencephalitis. Excretory/secretory products (ESPs) are valuable targets for the investigation of host-parasite interactions. These products contain a wide range of molecules for penetrating defensive barriers and avoiding the immune response of the host. Endoplasmic reticulum (ER) stress has been found to be associated with a wide range of parasitic infections and inflammation. ER stress can increase cell survival via the activation of downstream signalling. However, the mechanisms of ER stress in A. cantonensis infection have not yet been clarified. This study was designed to investigate the molecular mechanisms of ER stress in astrocytes after treatment with the ESPs of A. cantonensis L5.
Results
The results demonstrated that A. cantonensis infection activated astrocytes in the mouse hippocampus and induced the expression of ER stress-related molecules. Next, the data showed that the expression of ER stress-related molecules and the Ca2+ concentration were significantly increased in activated astrocytes after treatment with the ESPs of L5 of A. cantonensis. Ultimately, we found that ESPs induced GRP78 expression via the Sonic hedgehog (Shh) signalling pathway.
Conclusions
These findings suggest that in astrocytes, the ESPs of A. cantonensis L5 induce ER stress and that the Shh signalling pathway plays an important role in this process.
Keywords: Angiostrongylus cantonensis, Fifth-stage larvae, Excretory/secretory products, Astrocytes, Endoplasmic reticulum stress, Sonic hedgehog pathway
Background
Angiostrongylus cantonensis is an important causative agent of human cerebral angiostrongyliasis, such as eosinophilic meningitis and eosinophilic meningoencephalitis. During infection, fifth-stage larvae (L5) can induce a wide range of inflammatory responses, including eosinophil recruitment and cytokine secretion in the brains of humans [1, 2]. The clinical manifestations include headache, fever, nausea, vomiting, neck stiffness and paraesthesia. This disease is considered to be an endemic disease in Southeast Asia and Pacific islands [3, 4]. Recently, human cerebral angiostrongyliasis has become an emerging disease in many parts of the world, including China, Taiwan, Thailand, the USA, including Hawaii, Brazil and the Caribbean islands, including Jamaica [5–13]. Moreover, infections have been recently reported in more than 30 countries [4]. Infection in humans is acquired by ingesting infective third-stage larvae (L3) of A. cantonensis in intermediate hosts or paratenic hosts, such as snails, slugs, frogs, fish, freshwater crustaceans and vegetables [14].
The endoplasmic reticulum (ER) is an organelle that has multiple complex functions, including protein synthesis, cellular calcium (Ca2+) storage, lipid biosynthesis, and membrane biogenesis [15, 16]. The generation of ER stress due to the accumulation of unfolded and misfolded proteins in the ER may activate the unfolded protein response (UPR) and then induce the activation of related signalling pathways. When this stress can be alleviated, the UPR can lead to cell apoptosis [17]. First, ER stress activates an important molecule, glucose-regulated protein 78 (GRP78). GRP78, also called binding immunoglobulin protein (BiP), is an ER stress marker and chaperone in the heat shock protein family [18, 19]. Under ER stress, the activation of GRP78 may increase cell survival through the UPR [20]. In addition, the induction of GRP78 may also protect cells from ER stress-induced apoptosis by activating Bcl-2 and inhibiting Bak, Bax and caspase [21, 22].
In mammalian cells, GRP78 activates three signalling pathways in parallel through transmembrane ER stress sensors (IRE1, PERK and ATF6) [23, 24]. IRE1 can induce the splicing of the cytoplasmic transcription factor XBP1 to spliced XBP1 (XBP1s) and activate the gene expression of chaperones, autophagy, and inflammation. Activated PERK phosphorylates eIF2α to reduce the load of unfolded proteins by attenuating translation. In addition, the transcription factor ATF4 can stimulate autophagy by inducing CHOP expression. On the other hand, ATF6 translocates to the Golgi and reduces protein accumulation by upregulating XBP1 expression.
Hh has three homologs, namely, Sonic hedgehog (Shh), Desert hedgehog (Dhh) and Indian hedgehog (Ihh), but only Shh is broadly expressed in different tissues [25]. Sonic hedgehog (Shh) signalling plays an important role in animal development. Shh signalling can trigger other common signalling pathways. When Shh is activated and secreted, this protein can interact with the transmembrane protein Patched (Ptch). Under these conditions, Smoothened (Smo) and the transcription factor Glioma-associated oncogene-1 (Gli) can be activated [26–28].
In our previous studies, we found that A. cantonensis infection in mice may enhance the expression of GRP78 and that the activation of the Shh signalling pathway can reduce cell death via the GRP78-dependent pathway [29]. On the other hand, oxidative stress and cell apoptosis can be induced in astrocytes after treatment with the excretory/secretory products (ESPs) of A. cantonensis fifth-stage larvae (L5). However, ROS (superoxide and hydrogen superoxide) and the apoptosis of astrocytes are decreased after Shh signalling pathway activation, and the activity of antioxidants is elevated after ESP treatment [30]. Therefore, we demonstrated that the ESPs of A. cantonensis L5 can induce oxidative stress and cell apoptosis and that the Shh signalling plays an important role in the protection of astrocytes. In the present study, we investigated the molecular mechanisms of ER stress in mouse brain astrocytes after treatment with the ESPs of A. cantonensis L5. The results suggested that the ESPs of A. cantonensis L5 induce ER stress in astrocytes and that the activation of the Shh signalling pathway can stimulate GRP78 expression.
Methods
Parasite and animals
Angiostrongylus cantonensis (Taipei strain) has been maintained in our laboratory in Sprague-Dawley (SD) rats and Biomphalaria glabrata snails since 1980 [30, 31]. SD rats and BALB/c mice were purchased from the National Laboratory Animal Center (Taipei, Taiwan) and BioLASCO Taiwan Co., Ltd. (Taipei, Taiwan). Third-stage larvae (L3) of A. cantonensis were collected from infected Biomphalaria glabrata by digestion with 0.6% (w/v) pepsin-HCl (pH 2–3) at 37 °C for 45 min on day 21 post-infection [32]. Each rat or BALB/c mouse was inoculated with L3 via stomach intubation. Rats and mice were kept in plastic cages and provided with food and water ad libitum. The experimental animals were sacrificed by anaesthesia with isoflurane (1 ml/min) (Panion & BF Biotech Inc., Taipei, Taiwan).
Preparation of ESPs of A. cantonensis
Live fifth-stage larvae (L5) of A. cantonensis were isolated from the brain tissues of infected rats by anaesthetisation with isoflurane 21 days post-infection. After the worms were washed with saline, phosphate-buffered saline (PBS), distilled water and RPMI containing a high concentration of antibiotic antimycotic solution (Sigma-Aldrich, St. Louis, USA), they were incubated in RPMI without fetal bovine serum (FBS) for 24, 48 and 72 h. The excretory/secretory products (ESPs) of A. cantonensis L5 were collected from the culture medium and concentrated with Amicon Ultra-15 10K centrifugal filter devices (Merck Millipore, Burlington, USA). The concentration of ESPs was detected by a Bio-Rad Protein Assay Kit (Bio-Rad, Hercules, USA) according to the manufacturer’s instructions. These concentrated ESPs were utilized to treat mouse astrocytes, and mRNA and protein expression levels were detected in astrocytes [33].
Astrocyte culture
Astrocytes from the mouse brain (CRL-2535) were obtained from the American Type Culture Collection (ATCC) and employed in this research. The cells were cultured in Dulbecco’s modified Eagle’s medium (Corning, Corning, USA) supplemented with 10% foetal bovine serum and 100 U/ml penicillin/streptomycin in poly-L-lysine-coated culture flasks at 37 °C under 5% CO2. Finally, the cells were pre-treated with recombinant Shh (r-Shh), Shh agonist (SAG), and cyclopamine (Cyclo) for 1 h and then incubated with 250 μg/ml excretory/secretory products (ESPs) of A. cantonensis L5 for 12 h [29].
Brain specimen collection, immunohistochemistry and immunofluorescence staining
After mice were completely anaesthetised by inhalation of 3% (v/v) isoflurane, potassium phosphate-buffered saline (KPBS) was perfused through the heart. The mouse brains were collected from the cranial cavities and then immediately mounted and stored in optimal cutting temperature (OCT) medium (Sakura Finetek, Flemingweg, Netherlands) for further experiments. Before staining, the frozen tissue sections were fixed in 2% (w/v) PFA (paraformaldehyde) and permeabilized in 0.5% (v/v) Triton X-100. The sections were immersed in 5% (v/v) goat serum for 1 h and placed in primary antibody (anti-GFAP) (Abcam, Cambridge, UK) at 4 °C overnight. The sections were placed in secondary antibodies at room temperature for 1 h. avidin-biotin-peroxidase complex reagent (Vector Laboratories, Inc., Burlingame, USA) and DAB (3.3’-diaminobenzidine) reaction solution were added to each section. Finally, the sections were examined by light microscopy.
RNA extraction and cDNA microarray analysis
Total RNA was extracted from astrocytes (in 10 cm culture dishes) treated with the indicated doses of ESPs of A. cantonensis L5 by using the GENEzol TriRNA Pure Kit (Geneaid, New Taipei, Taiwan). The concentration of RNA was determined with a spectrophotometer (OD260 nm). The cDNA targets for hybridization were synthesized by reverse transcription of each RNA sample in the presence of Cy5-dUTP and Cy3-dUTP (Amersham Pharmacia Biotech, Amersham, UK). A customized A. cantonensis cDNA microarray (version 2.0) was utilized, and the data were analysed by QuantArray software (GSI Lumonics, Rugby, UK).
Real-time qPCR
First-strand cDNA was synthesized using the iScript™ Advanced cDNA Synthesis Kit (Bio-Rad) with random hexamers according to the manufacturer’s instructions. Real-time qPCR was performed using iQ™ SYBR® Green Supermix (Bio-Rad) on the CFX Connect™ Real-Time PCR Detection System (Bio-Rad). GAPDH was used as the internal control. Expression levels were detected with specific primers (Table 1).
Table 1.
Primer sequences for real-time qPCR
| Gene | Sequence (5’-3’) | |
|---|---|---|
| Gfap | Forward | CAGATCCGAGAAACCAGCCT |
| Reverse | GAGCCTGGCAAACAGGACTA | |
| Grp78 | Forward | GTGTGTGAGACCAGAACCGT |
| Reverse | AACACACCGACGCAGGAATA | |
| Perk | Forward | TTTCCATCCTCAGCCCCACA |
| Reverse | GGCACTCACGGAGTCGTATT | |
| eif2α | Forward | TTACTGTACGCCTGCGCTTT |
| Reverse | CTTCTCACAGCACCGCACTA | |
| Atf4 | Forward | CGGCTGGTCGTCAACCTATAA |
| Reverse | GGGGTAACTGTGGCGTTAGA | |
| Chop | Forward | GAGCCAGAATAACAGCCGGA |
| Reverse | TCTGCTTTCAGGTGTGGTGG | |
| Ire1 | Forward | CCCGGGAAATACATGAGCCA |
| Reverse | CCAGCGGAGGACAAGGAAAT | |
| Traf2 | Forward | AAGTACCTCTGTTCAGCCTGC |
| Reverse | AGAGAATGGATGCACACCTGA | |
| Atf6 | Forward | GGGAATGGAAGCCTAAAGAGGA |
| Reverse | ACAGAGAAACAAGCTCGGTGT | |
| Gapdh | Forward | GGTCCCAGCTTAGGTTCATCA |
| Reverse | TTTGCCGTGAGTGGAGTCAT |
SDS-PAGE electrophoresis and western blotting analysis
Total protein extracted from astrocytes was separated by 12% SDS-PAGE. The separated proteins were transferred to a nitrocellulose (NC) membrane and incubated with antibodies against GFAP (Proteintech, Rosemont, USA), Shh (Abcam), Ptch (Sigma-Aldrich), Smo (Sigma-Aldrich), Gli-1 (Sigma-Aldrich), GRP78 (Proteintech), PERK, eIF2 (Proteintech), phospho-eIF2 (EnoGene Biotech Co., New York, USA), IRE1 (Signalway Antibody, Baltimore, USA), phospho-IRE1 (Boster, Pleasanton, USA), CHOP (Proteintech), and β-actin (Proteintech). The NC membrane was washed with TBS/T three times and then incubated with a 1:10,000 dilution of anti-rabbit or mouse horseradish peroxidase antibody (Sigma-Aldrich). The bands were detected by ECL reagents (EMD Millipore, Burlington, USA) and captured by a ChemiDoc Imaging System (Bio-Rad). ImageJ software analysis was employed to detect the optical density of the target proteins.
Ca2+ analysis
The concentration of Ca2+ was measured using the Calcium Detection Assay Kit (Abcam). The samples were pre-treated with the ESPs of A. cantonensis L5 and then treated with Chromogenic Reagent and Calcium Assay Buffer at room temperature for 10 min protected from light. After incubation, the samples and standards were analysed with a spectrophotometer (OD575 nm).
Statistical analysis
Student’s t-test was employed to compare the mRNA and protein expression levels using GraphPad Prism 5 software (GraphPad Software, San Diego, CA, USA). The data are expressed as the mean ± standard deviation. P < 0.05 and < 0.01 were considered statistically significant.
Results
The activation of astrocytes in mouse brains after A. cantonensis infection
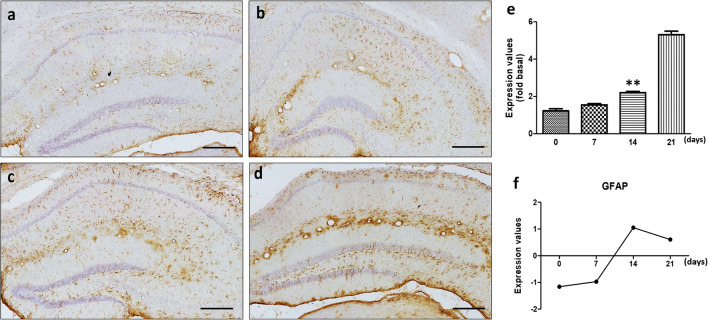
Astrocytes are the most abundant glial cells in the central nervous system (CNS). These cells can regulate the migration and differentiation of neural stem cells or other glial cells by secreting factors [34, 35]. Astrocytes can also reduce neuronal death during oxidative stress [36]. Glial fibrillary acidic protein (GFAP), an intermediate filament protein, is highly expressed in activated astrocytes [37–39]. GFAP is the most commonly used cell-specific marker for astrocytes in neurological studies. This marker can be employed to distinguish activated astrocytes from other brain cells. In this study, we used immunohistochemical (IHC) and cDNA microarray analysis to detect the expression of GFAP in mouse brains after A. cantonensis infection. IHC staining with an anti-GFAP antibody revealed that the expression of GFAP in astrocytes was significantly higher around the hippocampus after day 21 post-infection (t(4) = 7.244, P < 0.01) (Fig. 1a–e). On the other hand, the mRNA expression of GFAP in mouse brains was elevated after A. cantonensis infection (Fig. 1f). These data indicated that astrocytes were activated in the brain after A. cantonensis infection.
Fig. 1.
Astrocyte activation was induced in the brains of Angiostrongylus cantonensis-infected mice. The expression of GFAP was detected in the hippocampus in the absence of infection (a) and on days 7 (b), 14 (c) and 21 (d) post-infection after infection with 25 third-stage larvae. e The expression level of GFAP in the brain was quantified by ImageJ software (**P < 0.01). f The mRNA expression level of GFAP in the brain was detected by microarray analysis
The expression of ER stress-related genes in the mouse brain after A. cantonensis infection
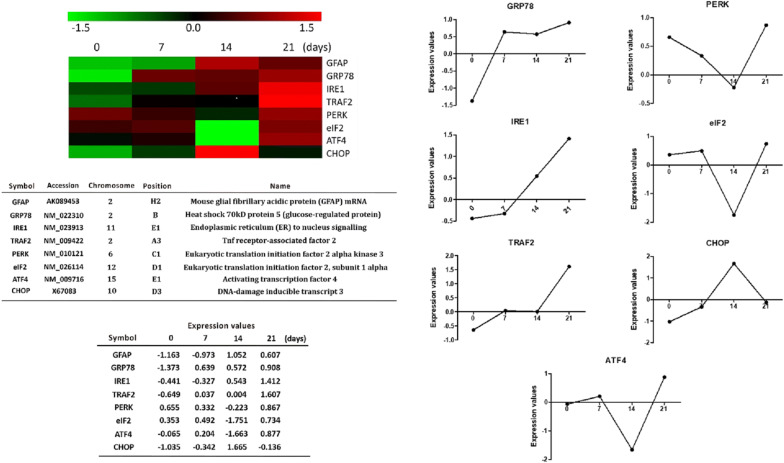
To evaluate the induction of ER stress in the mouse brain after A. cantonensis infection, we collected 25 third-stage larvae of A. cantonensis to infect BALB/c mice. cDNA microarray analysis was utilized to detect the mRNA expression of ER stress-related genes in four mouse brains (day 0, 7, 14 and 21 post-infection), including GRP78, IRE1, TRAF2, PERK, eIF2, ATF4 and CHOP. The data indicated a trend of elevated mRNA expression levels (Fig. 2). These results suggest that ER stress is induced in the mouse brain after A. cantonensis infection.
Fig. 2.
Angiostrongylus cantonensis infection stimulates the expression of ER stress-related molecules in the mouse brain. Mouse brains were collected from A. cantonensis-infected mice on days 0, 7, 14 and 21 post-infection. Then, the mRNA expression levels of ER stress-related molecules were detected by cDNA microarray analysis
ESPs induce astrocyte activation and GRP78 expression
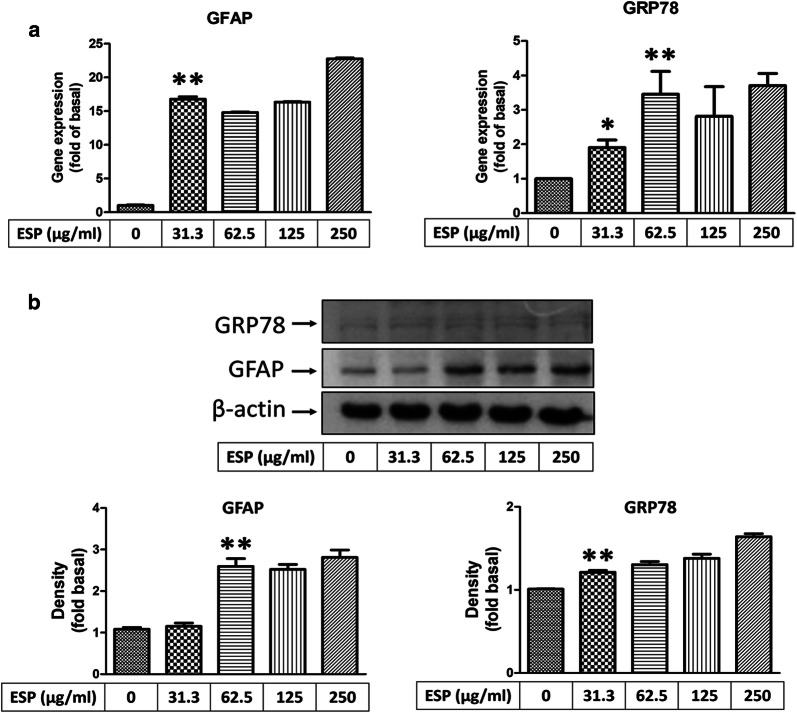
To investigate the activation of astrocytes and GRP78 expression, cells were treated with ESPs for 12 h. Real-time qPCR and western blotting were used to monitor the mRNA and protein expression levels of GFAP (mRNA level: t(4) = 41.44, P < 0.0001; protein level: t(4) = 7.892, P < 0.01) and GRP78 (mRNA level: t(4) = 4.814, P < 0.01; protein level: t(4) = 8.993, P < 0.001). The results showed that the ESPs of A. cantonensis stimulated the expression of GFAP and GRP78 in a dose-dependent manner (Fig. 3a, b).
Fig. 3.
Excretory/secretory products of Angiostrongylus cantonensis L5 stimulate astrocyte activation and GRP78 expression. Cells were treated with 0, 31.3, 62.5, 125 or 250 μg/ml excretory/secretory products (ESPs) of A. cantonensis L5 for 12 h. The mRNA (a) and protein (b) expression levels of GFAP and GRP78 were detected by real-time qPCR and western blotting. The data are expressed as the mean ± SD from three independent experiments (n = 3). *P < 0.05 and **P < 0.01 compared with cells treated with 0 μg/ml ESPs of A. cantonensis L5 for 12 h
ESPs induce the expression of ER stress-related molecules in astrocytes
As shown in Fig. 3, the results demonstrated that the ESPs of A. cantonensis induced astrocyte activation and GRP78 expression. Thus, we further examined whether the ESPs of A. cantonensis activated the ER stress downstream pathway. We detected the expression of ER stress-related genes and proteins (PERK, eIF2, ATF4, CHOP, IRE1, TRAF2 and ATF6) in astrocytes after treatment with ESPs for 12 h. The results of real-time qPCR and western blot analysis showed that the expression levels of ER stress-related molecules in astrocytes were increased in a dose-dependent manner (PERK: mRNA level: t(4) = 20.48, P < 0.0001; protein level: t(4) = 9.671, P < 0.001; eIF2: mRNA level: t(4) = 11.51, P < 0.001; protein level: t(4) = 11.92, P < 0.001; ATF4: t(4) = 13.41, P < 0.001; CHOP: mRNA level: t(4) = 10.70, P < 0.001; protein level: t(4) = 8.752, P < 0.001; IRE1: mRNA level: t(4) = 32.51, P < 0.0001; protein level: t(4) = 8.797, P < 0.001; TRAF2: t(4) = 38.43, P < 0.0001; ATF6: t(4) = 4.841, P < 0.01) (Fig. 4a, b). These data indicated that the ESPs of A. cantonensis induce the elevation of ER stress.
Fig. 4.
Excretory/secretory products of Angiostrongylus cantonensis L5 stimulate the activation of ER stress-related pathways in astrocytes. Cells were treated with 0, 31.3, 62.5, 125 or 250 μg/ml excretory/secretory products (ESPs) of A. cantonensis L5 for 12 h. The mRNA (a) and protein (b) expression levels of ER stress-related molecules were detected by real-time qPCR and western blotting. The data are expressed as the mean ± SD from three independent experiments (n = 3). *P < 0.05 and **P < 0.01 compared cells treated with 0 μg/ml ESPs of A. cantonensis L5 for 12 h
ESPs induce Ca2+ release in astrocytes
Some previous studies demonstrated that a loss of Ca2+ cellular homeostasis can induce ER stress and ER stress-related apoptosis [40–42]. To examine whether the ESPs of A. cantonensis induce the elevation of the Ca2+ concentration in astrocytes, we used the calcium detection assay kit to detect the concentration of Ca2+ in different dose of ESPs treatment (0, 31.3, 62.5, 125 or 250 μg/ml). The results showed that the concentration of Ca2+ was increased in astrocytes in a dose-dependent manner (2.92 to 3.58 mM) (t(4) = 11.39, P < 0.001) (Fig. 5).
Fig. 5.

Excretory/secretory products of Angiostrongylus cantonensis L5 stimulate calcium secretion in astrocytes. Cells were treated with 0, 31.3, 62.5, 125 or 250 μg/ml excretory/secretory products (ESPs) of A. cantonensis L5 for 12 h. The concentration of calcium was measured by the Calcium Assay Kit. The data are expressed as the mean ± SD from three independent experiments (n = 3). **P < 0.01 compared with cells treated with 0 μg/ml ESPs of A. cantonensis L5 for 12 h
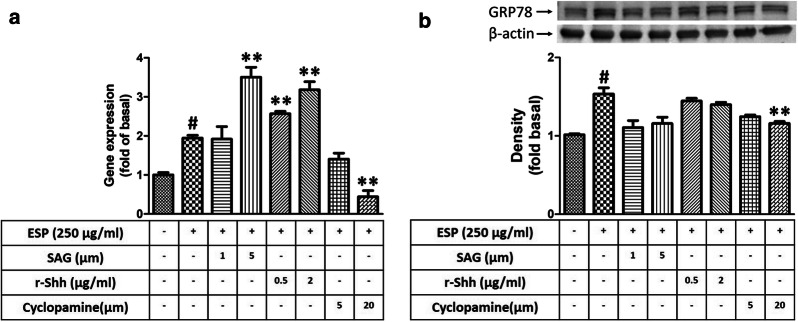
ESPs induce the expression of GRP78 via the Shh signalling pathway
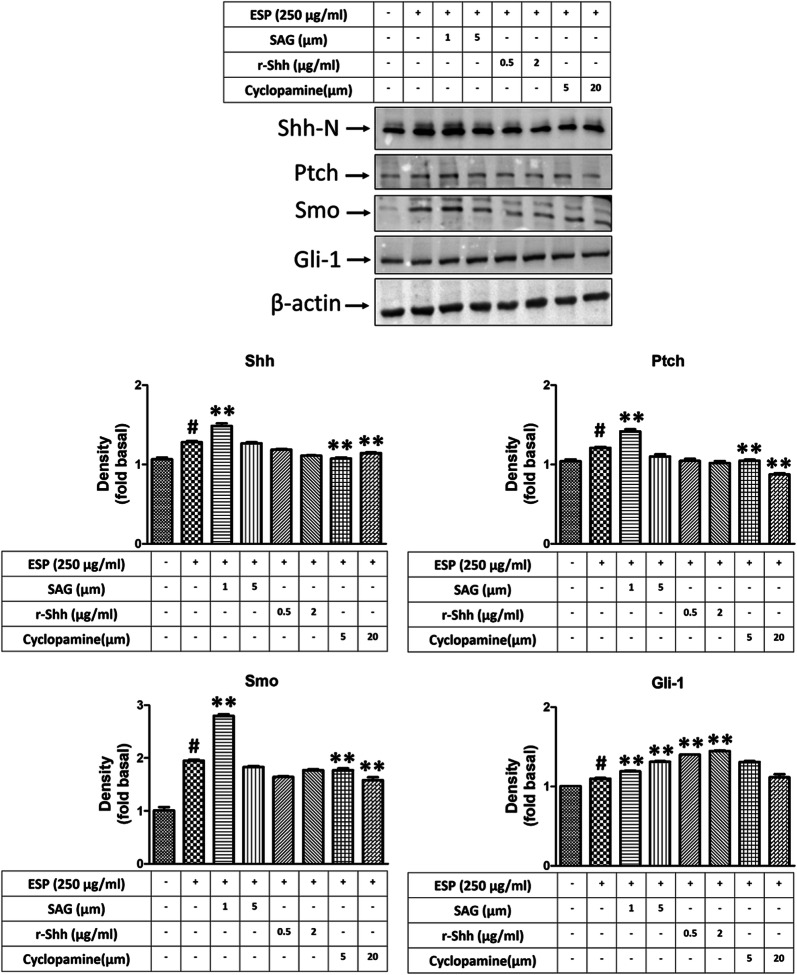
To determine whether Shh signalling can induce ER stress generation in astrocytes after treatment with the ESPs of A. cantonensis, cells were pre-treated with a Smo agonist (SAG), recombinant Shh (r-Shh), and an Shh pathway inhibitor (cyclopamine) and then treated with ESPs. First, we wanted to evaluate the effectiveness of an activator and inhibitor on the Shh signalling pathway. Western blotting analysis was used to confirm the protein expression of Shh pathway-related molecules, including Shh-N, Ptch, Smo, and Gli-1. The data showed that the expression of Shh pathway-related proteins was elevated in astrocytes after SAG (Shh-N: t(4) = 5.908, P < 0.01; Ptch: t(4) = 7.054, P < 0.01; Smo: t(4) = 23.42, P < 0.0001; Gli-1: t(4) =16.95, P < 0.0001) and r-Shh treatment (Gli-1: t(4) = 26.93, P < 0.0001). Conversely, the expression of Shh pathway-related proteins was decreased by cyclopamine treatment (Shh-N: t(4) = 9.347, P < 0.001; Ptch: t(4) = 18.12, P < 0.0001; Smo: t(4) = 6.351, P < 0.01) (Fig. 6). Next, real-time qPCR and western blotting analysis were employed to detect the expression of GRP78 in astrocytes after treatment with the ESPs of A. cantonensis. The results showed that the expression level of GRP78 was significantly changed following Shh pathway activation (SAG: t(4) = 5.989, P < 0.01; r-Shh: t(4) = 6.600, P < 0.01) or inactivation (mRNA level: t(4) = 8.710, P < 0.001; protein level: t(4) = 4.743, P < 0.01) after ESPs treatment (Fig. 7). These data indicate that the ESPs of A. cantonensis L5 induced ER stress in astrocytes through the Shh signalling pathway.
Fig. 6.
Evaluation of the effectiveness of an activator or inhibitor on the Shh signalling pathway. Cells were pre-treated with recombinant Shh (r-Shh), Shh agonist (SAG) and cyclopamine (Cyclo) for 1 h, then incubated with 250 μg/ml excretory/secretory products (ESPs) of A. cantonensis L5 for 12 h. The protein expression levels were detected by western blotting. The data are expressed as the mean ± SD from three independent experiments (n = 3). #P < 0.01 compared with the control; **P < 0.01 compared with cells exposed to ESPs
Fig. 7.
Excretory/secretory products induce GRP78 expression through the Shh signalling pathway. Cells were pre-treated with recombinant Shh (r-Shh), Shh agonist (SAG) and cyclopamine (Cyclo) for 1 h, then incubated with 250 μg/ml excretory/secretory products (ESPs) of A. cantonensis L5 for 12 h. The mRNA (a) and protein (b) expression levels were detected by real-time qPCR and western blotting. The data are expressed as the means ± SD from three independent experiments (n = 3). #P < 0.01 compared with the control; **P < 0.01 compared with cells exposed to ESPs
Discussion
In the life-cycle of A. cantonensis, the first-stage larvae (L1) are released into the faeces of the definitive host (rat). L1 in the faeces can infect the intermediate host and become third-stage larvae (L3). When humans are infected with A. cantonensis by eating L3 in intermediate hosts or paratenic hosts, infective L3 can penetrate and migrate into the central nervous system (CNS) through the circulatory system. Afterwards, L3 develop into fifth-stage larvae (L5) and remain in the CNS of the host indefinitely. In this condition, eosinophilic meningitis and eosinophilic meningoencephalitis can be induced in the brain of the host [33]. In our histopathological study, we found that L5 were surrounded by eosinophils in the anterior cerebral fissure, hippocampus, posterior cerebral fissure, and cerebellar fissure on day 14 post-infection [43]. On the other hand, A. cantonensis infection can induce a wide range of pathological changes in the CNS, including infiltration of eosinophils and congestion in the meninges, infiltration of lymphocytes and eosinophils in the meninges and choroid plexus, and necrosis and perivascular cuffing of lymphocytes in the brain parenchyma [44]. In this study, we also demonstrated that astrocytes were significantly activated in the hippocampus after A. cantonensis infection.
In the present study, the experiments were performed to determine the influence of the ESPs of A. cantonensis L5 in mouse astrocytes. The excretory/secretory products (ESPs) of nematodes, trematodes and cestodes contain a wide range of molecules, including proteins, lipids, glycans, and nucleic acids, and they can aid in the penetration of host defensive barriers, the avoidance of the host immune response and establishment and survival in host tissues [45–49]. ESPs are also useful targets for investigating the interaction between parasitic helminths and hosts [50–52]. In studies on the ESPs of A. cantonensis, proteomic analysis has been employed to determine the composition of ESPs in adults. Aspartyl protease inhibitor, cathepsin B-like cysteine proteinase, haemoglobinase- type cysteine proteinase, and heat shock protein 70 have been detected in ESPs [53]. On the other hand, our study identified approximately 51 protein spots and found immunoreactivity for protein disulfide-isomerase, a putative aspartic protease, and annexin in A. cantonensis L5 [33]. Here, we found that the ESPs of A. cantonensis L5 can stimulate the ER stress generation and the Ca2+ concentration in activated astrocytes.
ER stress has been found to be associated with a wide range of parasitic infections, including Trichinella spiralis, Toxoplasma gondii, Trypanosoma brucei and Plasmodium falciparum, and inflammation [54–58]. Some studies on parasitic infections have demonstrated that parasites can induce ER stress and subsequent cell apoptosis through the upregulation of GRP78 and caspase 3 expression in infected hosts [59–61]. However, some studies have shown that under damaging pathological conditions, GRP78 has protective effects on tissues or organs via Bcl-2 activation [62, 63]. In the central nervous system (CNS), the blood-brain barrier (BBB), which is composed of endothelial cells and astrocytes, is present at blood vessels. This barrier separates the circulating blood from brain tissue and regulates CNS homeostasis. The BBB allows the diffusion of only small hydrophobic molecules (O2, CO2 and hormones) [64]. Several studies on parasites have demonstrated that pathogens such as Toxoplasma gondii, Toxocara canis, Trypanosoma brucei spp. and malaria can penetrate into the CNS by causing BBB dysfunction [65–67]. Some studies on A. cantonensis have found that matrix metalloproteinase induces BBB breakdown and inflammation in cerebral angiostrongyliasis [68, 69]. In this study, we also found that the ESPs of A. cantonensis L5 induced the expression of GRP78 and downstream ER stress pathways, including the IRE1, PERK and ATF6 pathways in mouse astrocytes.
Finally, some studies have shown that the Shh pathway has a protective effect on the BBB. Astrocyte-derived Shh proteins can upregulate BBB formation through the stimulation of tight junction protein expression and inhibit proinflammatory cell entry [70–72]. Moreover, Shh signalling protects neurons by inhibiting cell apoptosis in oxidative stress and brain injury. Shh signalling can elevate the expression of antioxidants and anti-apoptotic proteins, including superoxide dismutase, glutathione peroxidase and Bcl-2 [73–77]. In our study, these data indicated that the Shh pathway influenced ER stress by regulating GRP78 expression after treatment with the ESPs of A. cantonensis L5 in astrocytes.
Conclusions
In conclusion, this study found that the ESPs of A. cantonensis L5 induce ER stress, upregulate the expression of GRP78 and then activate three ER stress-related pathways, including the IRE1, PERK, and ATF6 pathways. On the other hand, the sonic hedgehog signalling pathway plays an important role in protecting astrocytes by increasing GRP78 expression.
Acknowledgements
This study was supported in part by grants from the National Science Council, Executive Yuan, ROC (NSC105-2320-B-182-028-MY3 and 107-2320-B-039-070-MY2), the Chang Gung Memorial Hospital Research Grant (CMRPD1H0342 and CMRPD1H0442), and the China Medical University Research Grant (CMU108-S-45).
Abbreviations
- L5
fifth-stage larvae
- ESPs
excretory/secretory products
- ER
endoplasmic reticulum
- Shh
Sonic hedgehog
- L3
third-stage larvae
- UPR
unfolded protein response
- GRP78
glucose-regulated protein 78
- BiP
binding immunoglobulin protein
- Dhh
Desert hedgehog
- Ihh
Indian hedgehog
- Ptch
Patched
- Smo
Smoothened
- Gli
Glioma-associated oncogene
- FBS
fetal bovine serum
- KPBS
potassium phosphate-buffered saline
- PFA
paraformaldehyde
- CNS
central nervous system
- GFAP
Glial fibrillary acidic protein
- IHC
immunohistochemical
- SAG
Smoothened agonist
- r-Shh
recombinant Shh
- L1
first-stage larvae
Authors’ contributions
KYC and LCW conceived the study and designed the research. KYC, YJC, CJC and KYJ performed the experiments and analysis. KYC wrote the manuscript. All authors read and approved the final manuscript.
Funding
This work was supported in part by grants from the National Science Council, Executive Yuan, ROC (NSC105-2320-B-182-028-MY3 and 107-2320-B-039-070-MY2), the Chang Gung Memorial Hospital Research Grant (CMRPD1H0342 and CMRPD1H0442), and the China Medical University Research Grant (CMU108-S-45).
Availability of data and materials
The data supporting the conclusions of this article are included within the article.
Ethics approval and consent to participate
All animal procedures and animal care protocols in this study were approved by the Chang Gung University Institutional Animal Care and Use Committee (IACUC) in Taiwan (CGU107-086) and followed the Guide for Laboratory Animal Facilities and Care (The Council of Agriculture. Executive Yuan, ROC).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Kuang-Yao Chen, Email: kychen@mail.cmu.edu.tw.
Yi-Ju Chen, Email: yiru830318@gmail.com.
Chien-Ju Cheng, Email: d000014521@cgu.edu.tw.
Kai-Yuan Jhan, Email: ed790101@hotmail.com.
Lian-Chen Wang, Email: wanglc@mail.cgu.edu.tw.
References
- 1.Wang LC, Jung SM, Chen KY, Wang TY, Li CH. Temporal-spatial pathological changes in the brains of permissive and non-permissive hosts experimentally infected with Angiostrongylus cantonensis. Exp Parasitol. 2015;157:177–184. doi: 10.1016/j.exppara.2015.08.006. [DOI] [PubMed] [Google Scholar]
- 2.Wang TY, Chen KY, Jhan KY, Li CH, Jung SM, Wang LC. Temporal-spatial expressions of interleukin-4, interleukin-10, and interleukin-13 in the brains of C57BL/6 and BALB/c mice infected with Angiostrongylus cantonensis: an immunohistochemical study. J Microbiol Immunol Infect. 2018;S1684–1182:30173–30177. doi: 10.1016/j.jmii.2018.10.010. [DOI] [PubMed] [Google Scholar]
- 3.Rosen L, Loison G, Laigret J, Wallace GD. Studies on eosinophilic meningitis. 3. Epidemiologic and clinical observations on Pacific islands and the possible etiologic role of Angiostrongylus cantonensis. Am J Epidemiol. 1967;85:17–44. doi: 10.1093/oxfordjournals.aje.a120673. [DOI] [PubMed] [Google Scholar]
- 4.Wang QP, Lai DH, Zhu XQ, Chen XG, Lun ZR. Human angiostrongyliasis. Lancet Infect Dis. 2008;8:621–630. doi: 10.1016/S1473-3099(08)70229-9. [DOI] [PubMed] [Google Scholar]
- 5.Chen XG, Li H, Lun ZR. Angiostrongyliasis, Mainland China. Emerg Infect Dis. 2005;11:1645–1647. doi: 10.3201/eid1110.041338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Federspiel F, Skovmand S, Skarphedinsson S. Eosinophilic meningitis due to Angiostrongylus cantonensis in Europe. Int J Infect Dis. 2020;S1201–9712:30014. doi: 10.1016/j.ijid.2020.01.012. [DOI] [PubMed] [Google Scholar]
- 7.Johnston DI, Dixon MC, Elm JL, Calimlim PS, Sciulli RH, Park SY. Review of cases of angiostrongyliasis in Hawaii, 2007–2017. Am J Trop Med Hyg. 2019;101:608–616. doi: 10.4269/ajtmh.19-0280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lindo JF, Waugh C, Hall J, Cunningham-Myrie C, Ashley D, Eberhard ML, et al. Enzootic Angiostrongylus cantonensis in rats and snails after an outbreak of human eosinophilic meningitis, Jamaica. Emerg Infect Dis. 2002;8:324–326. doi: 10.3201/eid0803.010316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Morassutti AL, Thiengo SC, Fernandez M, Sawanyawisuth K, Graeff-Teixeira C. Eosinophilic meningitis caused by Angiostrongylus cantonensis: an emergent disease in Brazil. Mem Inst Oswaldo Cruz. 2014;109:399–407. doi: 10.1590/0074-0276140023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Qu ZY, Yang X, Cheng M, Lin YF, Liu XM, He A, et al. Enzootic angiostrongyliasis, Guangdong, China, 2008–2009. Emerg Infect Dis. 2011;17:1335–1336. doi: 10.3201/eid1707.100714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sinawat S, Trisakul T, Choi S, Morley M, Sinawat S, Yospaiboon Y. Ocular angiostrongyliasis in Thailand: a retrospective analysis over two decades. Clin Ophthalmol. 2019;13:1027–1031. doi: 10.2147/OPTH.S204380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tsai HC, Chen YS, Yen CM. Human parasitic meningitis caused by Angiostrongylus cantonensis infection in Taiwan. Hawaii J Med Public Health. 2013;72:26–27. [PMC free article] [PubMed] [Google Scholar]
- 13.Underwood EB, Walker MJ, Darden TL, Kingsley-Smith PR. Frequency of occurrence of the rat lungworm parasite in the invasive island apple snail in South Carolina, USA. J Aquat Anim Health. 2019;31:168–172. doi: 10.1002/aah.10063. [DOI] [PubMed] [Google Scholar]
- 14.Wang QP, Wu ZD, Wei J, Owen RL, Lun ZR. Human Angiostrongylus cantonensis: an update. Eur J Clin Microbiol Infect Dis. 2012;31:389–395. doi: 10.1007/s10096-011-1328-5. [DOI] [PubMed] [Google Scholar]
- 15.Chen TH, Chiang YH, Hou JN, Cheng CC, Sofiyatun E, Chiu CH, et al. XBP1-mediated BiP/GRP78 upregulation copes with oxidative stress in mosquito cells during dengue 2 virus infection. Biomed Res Int. 2017;2017:3519158. doi: 10.1155/2017/3519158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rodriguez D, Rojas-Rivera D, Hetz C. Integrating stress signals at the endoplasmic reticulum: The BCL-2 protein family rheostat. Biochim Biophys Acta. 2011;813:564–574. doi: 10.1016/j.bbamcr.2010.11.012. [DOI] [PubMed] [Google Scholar]
- 17.Fribley A, Zhang K, Kaufman RJ. Regulation of apoptosis by the unfolded protein response. Methods Mol Biol. 2009;559:191–204. doi: 10.1007/978-1-60327-017-5_14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.de Ridder G, Ray R, Misra UK, Pizzo SV. Modulation of the unfolded protein response by GRP78 in prostate cancer. Methods Enzymol. 2011;489:245–257. doi: 10.1016/B978-0-12-385116-1.00014-5. [DOI] [PubMed] [Google Scholar]
- 19.Kim R, Emi M, Tanabe K, Murakami S. Role of the unfolded protein response in cell death. Apoptosis. 2011;11:5–13. doi: 10.1007/s10495-005-3088-0. [DOI] [PubMed] [Google Scholar]
- 20.Li J, Lee AS. Stress induction of GRP78/BiP and its role in cancer. Curr Mol Med. 2006;6:45–54. doi: 10.2174/156652406775574523. [DOI] [PubMed] [Google Scholar]
- 21.Bruchmann A, Roller C, Walther TV, Schäfer G, Lehmusvaara S, Visakorpi T, et al. Bcl-2 associated athanogene 5 (Bag5) is overexpressed in prostate cancer and inhibits ER-stress induced apoptosis. BMC Cancer. 2013;13:96. doi: 10.1186/1471-2407-13-96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wu LF, Guo YT, Zhang QH, Xiang MQ, Deng W, Ye YQ, et al. Enhanced antitumor effects of adenoviral-mediated siRNA against GRP78 gene on adenosine-induced apoptosis in human hepatoma HepG2 cells. Int J Mol Sci. 2014;15:525–544. doi: 10.3390/ijms15010525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Galluzzi L, Diotallevi A, Magnani M. Endoplasmic reticulum stress and unfolded protein response in infection by intracellular parasites. Future Sci OA. 2017;3:FSO198. doi: 10.4155/fsoa-2017-0020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lebeaupin C, Vallée D, Hazari Y, Hetz C, Chevet E, Bailly-Maitre B. Endoplasmic reticulum stress signaling and the pathogenesis of non-alcoholic fatty liver disease. J Hepatol. 2018;69:927–947. doi: 10.1016/j.jhep.2018.06.008. [DOI] [PubMed] [Google Scholar]
- 25.Abramyan J. Hedgehog signaling and embryonic craniofacial disorders. J Dev Biol. 2019;7:9. doi: 10.3390/jdb7020009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kelleher FC. Hedgehog signaling and therapeutics in pancreatic cancer. Carcinogenesis. 2011;32:445–451. doi: 10.1093/carcin/bgq280. [DOI] [PubMed] [Google Scholar]
- 27.Sasai N, Toriyama M, Kondo T. Hedgehog signal and genetic disorders. Front Genet. 2019;10:1103. doi: 10.3389/fgene.2019.01103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wang C, Cassandras M, Peng T. The role of hedgehog signaling in adult lung regeneration and maintenance. J Dev Biol. 2019;7:14. doi: 10.3390/jdb7030014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chen KY, Cheng CJ, Wang LC. activation of sonic hedgehog leads to survival enhancement of astrocytes via the GRP78-dependent pathway in mice infected with Angiostrongylus cantonensis. Biomed Res Int. 2015;2015:674371. doi: 10.1155/2015/674371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chen KY, Chiu CH, Wang LC. Anti-apoptotic effects of Sonic hedgehog signalling through oxidative stress reduction in astrocytes co-cultured with excretory-secretory products of larval Angiostrongylus cantonensis. Sci Rep. 2017;7:41574. doi: 10.1038/srep41574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang LC, Chao D, Chen ER. Acquired immunity in rats against Angiostrongylus cantonensis infection. Int J Parasitol. 1989;19:617–620. doi: 10.1016/0020-7519(89)90039-8. [DOI] [PubMed] [Google Scholar]
- 32.Wang LC, Chao D, Chen ER. Experimental infection routes of Angiostrongylus cantonensis in mice. J Helminthol. 1991;65:296–300. doi: 10.1017/s0022149x00010890. [DOI] [PubMed] [Google Scholar]
- 33.Chen KY, Lu PJ, Cheng CJ, Jhan KY, Yeh SC, Wang LC. Proteomic analysis of excretory-secretory products from young adults of Angiostrongylus cantonensis. Mem Inst Oswaldo Cruz. 2019;114:e180556. doi: 10.1590/0074-02760180556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Abbott NJ. Astrocyte-endothelial interactions and blood-brain barrier permeability. J Anat. 2002;200:629–638. doi: 10.1046/j.1469-7580.2002.00064.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kang W, Hébert JM. Signaling pathways in reactive astrocytes, a genetic perspective. Mol Neurobiol. 2011;43:147–154. doi: 10.1007/s12035-011-8163-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Desagher S, Glowinski J, Premont J. Astrocytes protect neurons from hydrogen peroxide toxicity. J Neurosci. 1996;16:2553–2562. doi: 10.1523/JNEUROSCI.16-08-02553.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ben Haim L, Carrillo-de Sauvage MA, Ceyzériat K, Escartin C. Elusive roles for reactive astrocytes in neurodegenerative diseases. Front Cell Neurosci. 2015;9:278. doi: 10.3389/fncel.2015.00278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Filous AR, Silver J. Targeting astrocytes in CNS injury and disease: a translational research approach. Prog Neurobiol. 2016;144:173–187. doi: 10.1016/j.pneurobio.2016.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wilson EH, Hunter CA. The role of astrocytes in the immunopathogenesis of toxoplasmic encephalitis. Int J Parasitol. 2004;34:543–548. doi: 10.1016/j.ijpara.2003.12.010. [DOI] [PubMed] [Google Scholar]
- 40.Bahar E, Kim H, Yoon H. ER Stress-mediated signaling: action potential and Ca(2+) as key players. Int J Mol Sci. 2016;17:1558. doi: 10.3390/ijms17091558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Carreras-Sureda A, Pihán P, Hetz C. Calcium signaling at the endoplasmic reticulum: fine-tuning stress responses. Cell Calcium. 2018;70:24–31. doi: 10.1016/j.ceca.2017.08.004. [DOI] [PubMed] [Google Scholar]
- 42.Krebs J, Agellon LB, Michalak M. Ca(2+) homeostasis and endoplasmic reticulum (ER) stress: an integrated view of calcium signaling. Biochem Biophys Res Commun. 2015;460:114–121. doi: 10.1016/j.bbrc.2015.02.004. [DOI] [PubMed] [Google Scholar]
- 43.Wang LC, Wan DP, Jung SM, Chen CC, Wong HF, Wan YL. Magnetic resonance imaging findings in the brains of rabbits infected with Angiostrongylus cantonensis: a long-term investigation. J Parasitol. 2005;91:1237–1239. doi: 10.1645/GE-450R1.1. [DOI] [PubMed] [Google Scholar]
- 44.Wang LC, Jung SM, Chen CC, Wong HF, Wan DP, Wan YL. Pathological changes in the brains of rabbits experimentally infected with Angiostrongylus cantonensis after albendazole treatment: histopathological and magnetic resonance imaging studies. J Antimicrob Chemother. 2006;57:294–300. doi: 10.1093/jac/dki430. [DOI] [PubMed] [Google Scholar]
- 45.Crowe J, Lumb FE, Harnett MM, Harnett W. Parasite excretory-secretory products and their effects on metabolic syndrome. Parasite Immunol. 2017;39:e12410. doi: 10.1111/pim.12410. [DOI] [PubMed] [Google Scholar]
- 46.Dzik JM. Molecules released by helminth parasites involved in host colonization. Acta Biochim Pol. 2006;53:33–64. [PubMed] [Google Scholar]
- 47.Fang W, Xu S, Wang Y, Ni F, Zhang S, Liu J, et al. ES proteins analysis of Angiostrongylus cantonensis: products of the potential parasitism genes? Parasitol Res. 2010;106:1027–1032. doi: 10.1007/s00436-010-1751-z. [DOI] [PubMed] [Google Scholar]
- 48.Hewitson JP, Grainger JR, Maizels RM. Helminth immunoregulation: the role of parasite secreted proteins in modulating host immunity. Mol Biochem Parasitol. 2009;167:1–11. doi: 10.1016/j.molbiopara.2009.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Morassutti AL, Graeff-Teixeira C. Interface molecules of Angiostrongylus cantonensis: their role in parasite survival and modulation of host defenses. Int J Inflam. 2010;2012:512097. doi: 10.1155/2012/512097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bai X, Wu X, Wang X, Guan Z, Gao F, Yu J, et al. Regulation of cytokine expression in murine macrophages stimulated by excretory/secretory products from Trichinella spiralis in vitro. Mol Cell Biochem. 2012;360:79–88. doi: 10.1007/s11010-011-1046-4. [DOI] [PubMed] [Google Scholar]
- 51.Lightowlers MW, Rickard MD. Excretory-secretory products of helminth parasites: effects on host immune responses. Parasitology. 1988;96(Supp. l):S123–S166. doi: 10.1017/s0031182000086017. [DOI] [PubMed] [Google Scholar]
- 52.Sun XM, Guo K, Hao CY, Zhan B, Huang JJ, Zhu X. Trichinella spiralis excretory-secretory products stimulate host regulatory T cell differentiation through activating dendritic cells. Cells. 2019;8:1404. doi: 10.3390/cells8111404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Morassutti AL, Levert K, Pinto PM, da Silva AJ, Wilkins P, Graeff-Teixeira C. Characterization of Angiostrongylus cantonensis excretory-secretory proteins as potential diagnostic targets. Exp Parasitol. 2012;130:26–31. doi: 10.1016/j.exppara.2011.10.003. [DOI] [PubMed] [Google Scholar]
- 54.Chaubey S, Grover M, Tatu U. Endoplasmic reticulum stress triggers gametocytogenesis in the malaria parasite. J Biol Chem. 2014;289:16662–16674. doi: 10.1074/jbc.M114.551549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Michaeli S. The response of trypanosomes and other eukaryotes to ER stress and the spliced leader RNA silencing (SLS) pathway in Trypanosoma brucei. Crit Rev Biochem Mol Biol. 2015;50:256–267. doi: 10.3109/10409238.2015.1042541. [DOI] [PubMed] [Google Scholar]
- 56.Wei W, Zhang F, Chen H, Tang Y, Xing T, Luo Q, et al. Toxoplasma gondii dense granule protein 15 induces apoptosis in choriocarcinoma JEG-3 cells through endoplasmic reticulum stress. Parasit Vectors. 2018;11:251. doi: 10.1186/s13071-018-2835-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Yu YR, Deng MJ, Lu WW, Zhang JS, Jia MZ, Huang J, et al. Endoplasmic reticulum stress-mediated apoptosis is activated in intestines of mice with Trichinella spiralis infection. Exp Parasitol. 2014;145:1–6. doi: 10.1016/j.exppara.2014.06.017. [DOI] [PubMed] [Google Scholar]
- 58.Zhang K, Kaufman RJ. From endoplasmic-reticulum stress to the inflammatory response. Nature. 2008;454:455–462. doi: 10.1038/nature07203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Morada M, Pendyala L, Wu G, Merali S, Yarlett N. Cryptosporidium parvum induces an endoplasmic stress response in the intestinal adenocarcinoma HCT-8 cell line. J Biol Chem. 2013;288:30356–30364. doi: 10.1074/jbc.M113.459735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Nicolao MC, Loos JA, Rodriguez Rodrigues C, Beas V, Cumino AC. Bortezomib initiates endoplasmic reticulum stress, elicits autophagy and death in Echinococcus granulosus larval stage. PLoS ONE. 2017;12:e0181528. doi: 10.1371/journal.pone.0181528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wan L, Gong L, Wang W, An R, Zheng M, Jiang Z, et al. T. gondii rhoptry protein ROP18 induces apoptosis of neural cells via endoplasmic reticulum stress pathway. Parasit Vectors. 2015;8:554. doi: 10.1186/s13071-015-1103-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Jeon M, Choi H, Lee SI, Kim JS, Park M, Kim K, et al. GRP78 is required for cell proliferation and protection from apoptosis in chicken embryo fibroblast cells. Poult Sci. 2016;95:1129–1136. doi: 10.3382/ps/pew016. [DOI] [PubMed] [Google Scholar]
- 63.Park CH, Choi MS, Ha JY, Kim BH, Park CH, Kim CI. Effect of overexpression of glucose-regulated protein 78 and bcl-2 on recurrence and survival in patients with ureter tumors. Korean J Urol. 2013;54:671–676. doi: 10.4111/kju.2013.54.10.671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Rénia L, Howland SW, Claser C, Charlotte Gruner A, Suwanarusk R, Hui Teo T, et al. Cerebral malaria: mysteries at the blood-brain barrier. Virulence. 2012;3:193–201. doi: 10.4161/viru.19013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Feustel SM, Meissner M, Liesenfeld O. Toxoplasma gondii and the blood-brain barrier. Virulence. 2012;3:182–192. doi: 10.4161/viru.19004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Hamilton CM, Brandes S, Holland CV, Pinelli E. Cytokine expression in the brains of Toxocara canis-infected mice. Parasite Immunol. 2008;30:181–185. doi: 10.1111/j.1365-3024.2007.01002.x. [DOI] [PubMed] [Google Scholar]
- 67.Masocha W, Kristensson K. Passage of parasites across the blood-brain barrier. Virulence. 2012;3:202–212. doi: 10.4161/viru.19178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Lai SC, Twu JJ, Jiang ST, Hsu JD, Chen KM, Chiaing HC, et al. Induction of matrix metalloproteinase-9 in murine eosinophilic meningitis caused by Angiostrongylus cantonensis. Ann Trop Med Parasitol. 2004;98:715–724. doi: 10.1179/000349804225021479. [DOI] [PubMed] [Google Scholar]
- 69.Lee HH, Chou HL, Chen KM, Lai SC. Association of matrix metalloproteinase-9 in eosinophilic meningitis of BALB/c mice caused by Angiostrongylus cantonensis. Parasitol Res. 2004;94:321–328. doi: 10.1007/s00436-004-1196-3. [DOI] [PubMed] [Google Scholar]
- 70.Alvarez JI, Dodelet-Devillers A, Kebir H, Ifergan I, Fabre PJ, Terouz S, et al. The hedgehog pathway promotes blood-brain barrier integrity and CNS immune quiescence. Science. 2011;334:1727–1731. doi: 10.1126/science.1206936. [DOI] [PubMed] [Google Scholar]
- 71.Brilha S, Ong CWM, Weksler B, Romero N, Couraud PO, Friedland JS. Matrix metalloproteinase-9 activity and a downregulated hedgehog pathway impair blood-brain barrier function in an in vitro model of CNS tuberculosis. Sci Rep. 2017;7:16031. doi: 10.1038/s41598-017-16250-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Liu S, Chang L, Wei C. The sonic hedgehog pathway mediates Tongxinluo capsule-induced protection against blood-brain barrier disruption after ischaemic stroke in mice. Basic Clin Pharmacol Toxicol. 2019;124:660–669. doi: 10.1111/bcpt.13186. [DOI] [PubMed] [Google Scholar]
- 73.Amankulor NM, Hambardzumyan D, Pyonteck SM, Becher OJ, Joyce JA, Holland EC. Sonic hedgehog pathway activation is induced by acute brain injury and regulated by injury-related inflammation. J Neurosci. 2009;29:10299–10308. doi: 10.1523/JNEUROSCI.2500-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Dai RL, Zhu SY, Xia YP, Mao L, Mei YW, Yao YF, et al. Sonic hedgehog protects cortical neurons against oxidative stress. Neurochem Res. 2011;36:67–75. doi: 10.1007/s11064-010-0264-6. [DOI] [PubMed] [Google Scholar]
- 75.Ghanizadeh A. Malondialdehyde, Bcl-2, superoxide dismutase and glutathione peroxidase may mediate the association of sonic hedgehog protein and oxidative stress in autism. Neurochem Res. 2012;37:899–901. doi: 10.1007/s11064-011-0667-z. [DOI] [PubMed] [Google Scholar]
- 76.Heine VM, Rowitch DH. Hedgehog signaling has a protective effect in glucocorticoid-induced mouse neonatal brain injury through an 11betaHSD2-dependent mechanism. J Clin Invest. 2009;2119:267–277. doi: 10.1172/JCI36376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Xia YP, Dai RL, Li YN, Mao L, Xue YM, He QW, et al. The protective effect of sonic hedgehog is mediated by the phosphoinositide 3-kinase/AKT/Bcl-2 pathway in cultured rat astrocytes under oxidative stress. Neuroscience. 2012;209:1–11. doi: 10.1016/j.neuroscience.2012.02.019. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data supporting the conclusions of this article are included within the article.