Abstract
Dimethylsulfoniopropionate (DMSP) is a key compound in the marine sulfur cycle, and is produced in large quantities in coral reefs. In addition to Symbiodiniaceae, corals and associated bacteria have recently been shown to play a role in DMSP metabolism. Numerous ecological studies have focused on DMSP concentrations in corals, which led to the hypothesis that increases in DMSP levels might be a general response to stress. Here we used multiple species assemblages of three common Indo-Pacific holobionts, the scleractinian corals Pocillopora damicornis and Acropora cytherea, and the giant clam Tridacna maxima and examined the DMSP concentrations associated with each species within different assemblages and thermal conditions. Results showed that the concentration of DMSP in A. cytherea and T. maxima is modulated according to the complexity of species assemblages. To determine the potential importance of symbiotic dinoflagellates in DMSP production, we then explored the relative abundance of Symbiodiniaceae clades in relation to DMSP levels using metabarcoding, and found no significant correlation between these factors. Finally, this study also revealed the existence of homologs involved in DMSP production in giant clams, suggesting for the first time that, like corals, they may also contribute to DMSP production. Taken together, our results demonstrated that corals and giant clams play important roles in the sulfur cycle. Because DMSP production varies in response to specific species-environment interactions, this study offers new perspectives for future global sulfur cycling research.
Subject terms: Transcriptomics, Marine chemistry, Marine biology
Introduction
Coral reefs have been described as dimethylsulfoniopropionate (DMSP) hotspots1,2. This compound is an important metabolite that plays a central role in the marine sulfur cycle3. It is involved in numerous cellular and ecological processes. Among several known functions, DMSP possesses antioxidant properties4,5 as evidenced by cellular increase of DMSP under CO2 depletion6. Dimethylsulfoniopropionate has other protective physiological functions, notably serving as an osmolyte and cryoprotectant in marine algae7,8. Acting as a signalling molecule, DMSP is also involved in antiviral defense mechanisms and sulfide detoxification9–11. Numerous studies have recently drawn attention to variations of DMSP concentration in organisms subjected to environmental changes. Marine algae and/or coral studies have demonstrated that DMSP concentration changes with light intensity and salinity, as well as in response to oxidative stressors6,12–14. DMSP is therefore believed to be involved in organisms’ stress response, especially in coral species14,15. Dimethylsulfoniopropionate has also been extensively studied for its role in climate regulation16, because it can be converted into dimethylsulfide (DMS), a trace gas that is a source of reduced sulfur and which plays a role in cloud formation17,18. Consequently, precise methodologies such as nuclear magnetic resonance (NMR) spectroscopy have been developed for accurate detection and quantification of DMSP16,19.
Dimethylsulfoniopropionate is found only in certain terrestrial or marine organisms. It has been recorded in graminoids, rhodophytes, mussels and also in benthic flatworms20–23. However, only a few species are able to produce DMSP, and the majority of organisms are believed to accumulate this molecule through their diets or phototrophic symbionts24. To date, pathways of DMSP biosynthesis have been characterized in higher plants22,25, marine algae such as Symbiodiniaceae26,27 and more recently in corals, with methionine as a common precursor15. Higher plants and marine algae use two different DMSP biosynthetic pathways, while corals harbor an algal-like pathway15,26. Similarly to marine algae, coral species such as Acropora sp. likely encode the enzymatic machinery required for biosynthesis of DMSP (specific aminotransferase, reductase, methyltransferase and decarboxylase enzymes)3,15. Even though the specific biosynthesis pathway has not yet been studied in other marine organisms, DMSP has been found in other coral taxa (e.g. Pocilloporidae, Poritidae) and sessile organisms such as giant clams and anemones20,28. The concentrations of DMSP were higher in corals (Acropora sp., Heliopora sp., Pavona sp.) and giant clams (Tridacna maxima and Tridacna squamosa) than in other animals20,28. Dinoflagellates of the family Symbiodiniaceae account for most of the DMSP production in coral reefs24,29. This family is currently classified into nine divergent lineages, hereafter referred to as clades A to I30, of which seven have recently been elevated to genus level31, with clades A-D commonly found in symbiosis with scleractinian corals and giant clams32. Symbiodiniaceae can possess a DMSP-lyase activity leading to the conversion of DMSP into DMS that differs among clades33. Thus, the presence or proportion of different Symbiodiniaceae clades within a host may influence the variation of DMSP concentration often observed between coral species34. Additionally, the differences observed in coral studies could also be the result of other symbionts hosted by corals. Certain bacteria possess crucial genes for DMSP degradation such as homologs of the dmdA gene that are involved in the demethylation pathway or of the dddD gene known to be involved in the degradation of DMSP into DMS35–38. These homologs have been found in Proteobacteria that are common in coral species. Additionally, the occurrence of bacteria involved in DMSP production in coral is possible39, further highlighting the complexity of DMSP-cycling processes in benthic holobionts. Most of these bacteria have been recently described in microbiomes of giant clams40. Further, DMSP production in heterotrophic bacteria has also been recently highlighted41.
It is well known that coral symbionts such as dinoflagellates (Symbiodiniaceae) and bacteria vary according to environmental conditions42–46, and are therefore assumed to play a key role in their host fitness47. In addition to symbionts, interaction between holobionts could also impact the host fitness. While it is well known that marine sessile organisms interact, either directly or indirectly48–50, only a few studies have underlined the role of benthic species assemblages on coral reef dynamics51,52, as well as on giant clams’ fitness40. To better understand how benthic species assemblages could influence their respective fitness, we artificially combined three coral reef builders, including two coral species (Pocillopora damicornis; Linneaus, 1758 and Acropora cytherea; Dana, 1846) and one giant clam species (Tridacna maxima; Röding, 1798), and measured DMSP concentration in each species using NMR spectroscopy. Metabarcoding was used to characterize the Symbiodiniaceae composition within the three studied species and determine the putative role of Symbiodiniaceae in DMSP production. We also examined whether giant clams contribute to DMSP production by screening our previously described transcriptomes of T. maxima53 for enzymes involved in the DMSP biosynthesis. Finally, because DMSP is a stress biomarker, the experiments were performed either at lagoon temperature or under thermal stress.
Material and Methods
Coral and giant clam collection and experimental design
The coral and giant clam samples as well as the experimental design used in this study were described previously52. Briefly, coral species of A. cytherea and P. damicornis were collected in Moorea lagoon, French Polynesia (17°30′S, 149°50′W, Linareva fringing reef)54. For each species, 4 colonies were sampled and nubbinized into 45 small fragments. Giant clams of the species T. maxima were purchased from a French Polynesian nursery on Reao Island (18°28′S, 136°25′W; N°Tahiti: 139 519). Corals nubbins and giant clams were reared in a common garden. A CITES permit was obtained to allow specimen exports (CITES – FR1698700087 – E).
Experiments were conducted in open-circuit aquariums (20 L/h), using distinct assemblages of either one species P. damicornis (P, n = 4 aquariums) or T. maxima (T, n = 4 aquariums), two species P. damicornis + A. cytherea (PA, n = 4 aquariums) or A. cytherea + T. maxima (AT, n = 4 aquariums), or three species P. damicornis + A. cytherea + T. maxima (PAT, n = 4 aquariums; Figure S1a). Three nubbins per coral colony (n = 3 nubbins x 4 colonies per species) and 12 giant clams were used when required in an assemblage. Coral fragments (5–8 cm in height and 2 cm in diameter) and giant clams (4–8 cm in size) were spaced 5 to 10 cm apart to avoid contact. Assemblages were either under thermal stress (32 °C, S, n = 10 aquariums) or at lagoon temperature (27 °C, L, n = 10 aquariums). After 12 days of acclimation at 27 °C (time 0), half of the aquariums (n = 10) were placed under thermal stress by increasing the temperature by 1 °C per day until it reached 32 °C on day 17 (Fig. S1b). At each sampling time (day 12 and 17), half of the sampled giant clam mantles (4 cm2) and 80% of the sampled coral fragments were immediately snap-frozen in liquid nitrogen and stored at −80 °C until further analysis. The remaining portions of the sampled giant clams and corals were stored in 70% ethanol for DNA analysis. Seawater temperature data were recorded every 10 minutes with Temperature/Light Data Loggers (P/N U22–001, Onset, Bourne, Massachusetts; or Ruskin, Ottawa, Canada; Fig. S1b). Seawater temperature was controlled with the Biotherm pro system (Hobby, Stukenbrock, Germany). Health status checks on the coral nubbins were carried out using the standardized coral coloration scale as a proxy of symbiont density and chlorophyll a content variation (Coral Health Chart, www.CoralWatch.org)55,56, and through daily visual observation for giant clams.
Metabolite extraction for dimethylsulfoniopropionate quantification
Coral samples were lyophilized overnight, and 1 to 1.5 g of powder was obtained for extraction. The extraction protocol was adapted from Tapiolas and collaborators19. In total, 51 A. cytherea and 54 P. damicornis samples were extracted with 3 mL of HPLC-grade methanol (CH3OH). After 5 minutes of sonication (35 kHz Transsonic 950/H, Elma, Germany) at room temperature, the extracts were shaken for 3 hours. A second extraction with an additional 2 mL of CH3OH was performed using 5 minutes of sonication and 5 minutes of shaking. The two extracts were then pooled and dried using a vacuum centrifuge drier (Genevac EC-2 plus, Genevac, UK) before being stored at −20 °C until NMR analysis. Samples were lyophilized overnight and weighed to measure the quantity of extracts.
Giant clam samples (n = 42) were lyophilized overnight and 0.1 to 0.5 g of powder was obtained for extraction. Protocol extraction was adapted from Mohamadi57. Solid-liquid extractions were performed using 2 mL of H20 and 0.5 mL of HPLC-grade methanol (CH3OH) prior to vortexing. After adding 1.5 mL of CH3OH and 2 mL of dichloromethane (CH2Cl2), the solutions were mixed using a vortex and sonicated for 10 minutes. The solid-liquid extraction was carried out three times and the three extracts were pooled and centrifuged at 2050 g for 20 min in order to separate the polar and apolar phases. The apolar extracts were pooled as per the polar extracts and lyophilized. In this study, only the hydroalcoholic (polar) extracts of the giant clam samples were analyzed.
All extracts were resolubilized in 500 μL of deuterium oxide (D2O), vortex mixed and transferred into a 5 mm NMR tube, then analyzed immediately by 1H-NMR spectroscopy.
Nuclear magnetic resonance data acquisition and dimethylsulfoniopropionate quantification
All 1H spectra were recorded on a Bruker Avance 1 spectrometer (Bruker, Germany) at 300 MHz in the same conditions with 128 transients; chemical shifts were reported in ppm from tetramethylsilane (TMS). Absolute Area was determined by NMR analysis on the topspin 2.1 Bruker software.
Two external calibration curves of DMSP were performed under the same conditions as the samples with 128 transients (Figure S1c,d). Integration zone A (3.41 ppm to 3.34 ppm) was used for A. cytherea and P. damicornis samples because there were no overlapping signals. Integration zone B was used for T. maxima samples because of overlapping signals on zone A and no overlapping signal on zone B (Fig. S1e–h). The concentrations of DMSP calculated with the calibration curve (in mg per mL) were normalized with the mass ratio corresponding to the dry weight of the extract. Analysis of variance (ANOVA) and ad hoc pairwise comparisons (Tukey test) were performed using R software58.
DNA extraction; PCR amplification and sequencing of Symbiodiniaceae communities
Coral DNA extractions (n = 2 to 9 per assemblage) were performed following a Cethyl Trimethylammonium Bromide (CTAB) based protocol59. Symbiodiniaceae diversity was studied using the internal transcribed spacer 2 (ITS2) marker. The ITS2 region of the nuclear ribosomal array was amplified using the forward primer “its-Dino” (5′-GTGAATTGCAGAACTCCGTG-3′) and the reverse primer “ITS2-Rev2” (5′-GCCTCCGCTTACTTATATGCT-3′)60,61. IlluminaTM overhang adaptors were included in the primers as described in Kozich et al.62. PCR amplification and sequencing were performed as described in Guibert et al.52.
Symbiodiniaceae composition analysis of A. cytherea and P. damicornis samples was adapted from the Arif et al. (2014) pipeline in mothur v1.3963. An ITS2 database64 was used to annotate the sequences via Basic Local Alignment Search Tool (BLASTN). The relative abundance of Symbiodiniaceae subclades was represented using R (Reshape2 and ggplot2 packages).
Symbiodiniaceae composition analysis of T. maxima samples was processed using the same methodology as described in Guibert et al.40.
Identification of candidate genes
A previous predicted peptide data set of T. maxima metatranscriptome53 was blasted against a custom database at a threshold of 10−3. This database contained molluscan and cnidarian sequences extracted from the Protein database of the National Center for Biotechnology Information (NCBI) filtered with the DMSP enzyme names or EC numbers and from the DMSP candidate genes previously identified in Acropora millepora15 (Table S2, sheet 2). The selected sequences were blasted against the whole NCBI Protein database and any sequence with a top hit with a plant, algae or bacterial taxonomy was considered a false positive and removed (Table S2, sheet 1).
A differential gene expression analysis was then performed from previous RNAseq results of T. maxima under thermal stress at 32 °C for 5 days (Bioproject accession number: PRJNA309928). Transcript quantification of the above selected sequences was performed with RNA-Seq by Expectation Maximization (RSEM)65 across 14 samples of the Bioproject (8 biological samples: B0Tx and BT0 as the control group and 6 BTx biological samples as the Heating group53). The differential expression between the 2 groups was performed with the edgeRun package66 with 50,000 iterations.
Results
Analyses of dimethylsulfoniopropionate concentrations according to thermal stress, species and assemblages
Dimethylsulfoniopropionate concentration was evaluated by 1H-NMR spectroscopy and standardized with the mass ratio in the three species: P. damicornis, A. cytherea and T. maxima (Table S3). An ANOVA test (Table 1) revealed significant differences among assemblages (p = 1.01e-11) or species (p < 2e-16) with a significant interaction among them (p = 3.90e-08). No significant differences between assemblages and temperatures were found. Thus, the DMSP data were pooled by assemblages for each species.
Table 1.
Results of ANOVA test on DMSP concentration (mass ratio) by assemblage, species and temperature. Df: degrees of freedom; SumSq: Sum of squares; Mean Sq: mean sum of squares, F value: F statistic.
| Df | Sum Sq | Mean Sq | F value | P value | |
|---|---|---|---|---|---|
| Assemblage | 5 | 184.3 | 36.86 | 15.274 | 1.01e-11*** |
| Temperature | 1 | 2.7 | 2.67 | 1.107 | 0.295 |
| Species | 2 | 339.5 | 169.75 | 70.343 | <2e-16*** |
| Assemblage: Temperature | 4 | 11.3 | 2.81 | 1.166 | 0.329 |
| Assemblage: Species | 2 | 94.6 | 47.28 | 19.594 | 3.90e-08*** |
| Temperature: Species | 2 | 4.1 | 2.05 | 0.848 | 0.431 |
| Assemblage: Temperature: Species | 2 | 0.3 | 0.14 | 0.060 | 0.942 |
| Residuals | 126 | 304.1 | 2.41 |
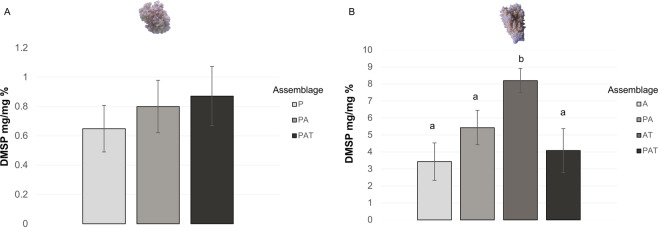
In corals, the concentrations of DMSP were from 5 to 10-fold lower in P. damicornis than in A. cytherea (Tukey test, p < 0.001), with means ranging between 0.65 and 0.87% (% = mg DMSP/100 mg extract) for P. damicornis and between 3.43 and 8.20% for A. cytherea (Fig. 1). In P. damicornis, while mean DMSP concentrations increased with the increasing complexity of assemblages, no significant differences were found (Fig. 1A). However, in A. cytherea, the DMSP concentration increased for the two-species assemblages (PA and AT) but decreased in PAT assemblages. A significant difference was found between AT and the other assemblages (A, p < 0.0001; PA, p < 0.05; PAT, p < 0.001; Fig. 1B).
Figure 1.
Concentration of dimethylsulfoniopropionate (Mass ratio: mg/mg %) of Pocillopora damicornis (A) and Acropora cytherea (B) by assemblage. Assemblages: P: P. damicornis (nA = 25); A: A. cytherea (nB = 12); PA: P. damicornis and A. cytherea (nA = 16, nB = 16); AT: A. cytherea and T. maxima (nB = 8); PAT: P. damicornis, A. cytherea and T. maxima (nA = 13, nB = 15). Letters in lower case indicate significant differences between means (Tukey, p < 0.05). Photographs: Isis Guibert.
Interestingly, in T. maxima, mean DMSP concentrations decreased when exclusively associated with A. cytherea (from 1.97% to 0.98%) but increased in the PAT assemblage (3.22%, Fig. 2). Tridacna maxima exhibited a significantly higher DMSP concentration in the PAT assemblages than in the AT assemblages (p < 0.5).
Figure 2.
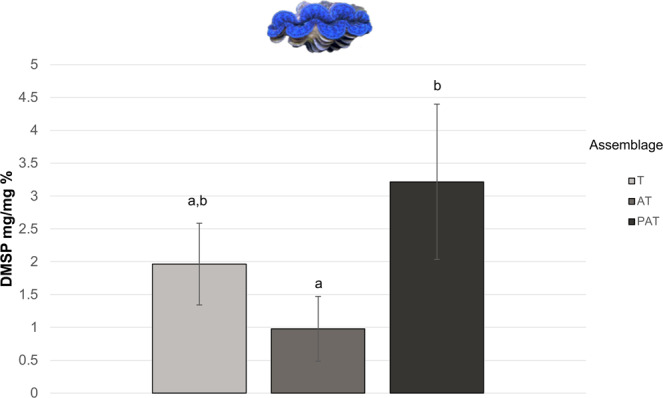
Concentration of dimethylsulfoniopropionate (Mass ratio: mg/mg %) of Tridacna maxima by assemblage. Assemblages: T: T. maxima (n = 19); AT: A. cytherea and T. maxima (n = 9); PAT: P. damicornis, A. cytherea and T. maxima (n = 14). Letters in lower case indicate significant differences between means (Tukey, p < 0,05). Photograph: Isis Guibert.
Symbiodiniaceae composition by assemblage
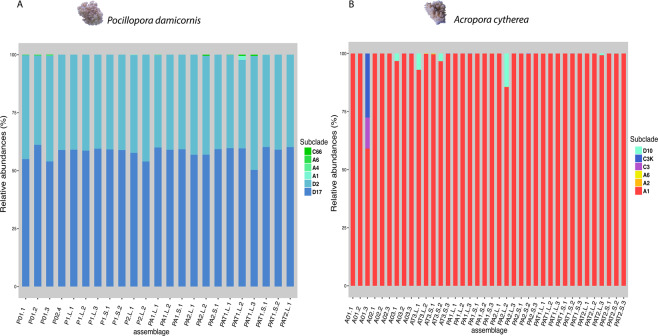
The relative abundances of Symbiodiniaceae genotypes associated with the different assemblages and thermal stress conditions were measured at the clade and subclade levels (Fig. 3 and Table S4). Among the three Symbiodiniaceae clades (A, C and D) detected in the samples, clade D was the dominant lineage in P. damicornis nubbins (> 90%; Fig. 3a). Two D subclades were systematically detected in all nubbins. Subclade D17 represented more than half of the relative abundance (58–60%) and subclade D2 ranged from 39 to 44%. Clades A and C were only detected as background clades with subclades A1, A4, A6 and C66 sporadically present in the nubbins, either alone or associated, regardless of temperature levels and/or assemblages.
Figure 3.
Relative abundance of Symbiodiniaceae subclades in Pocillopora damicornis (A) and Acropora cytherea (B). All samples were collected at day 17 except those with a 0 in their name that were collected at day 12. PAT: P. damicornis, A. cytherea and T. maxima; AT: A. cytherea and T. maxima; A: A. cytherea; P: P. damicornis; 0: time 0 - day 12; (1–3): number of the experiment; L: lagoon temperature; S: thermal stress; (1–4): number of sample. Photograph: Isis Guibert.
Three clades (A, C and D) were also detected in A. cytherea (Fig. 3b), although clade A largely dominated in all nubbins (n = 38), 24 of which harbored only this clade. Subclade A1 was dominant in the Symbiodiniaceae composition of all A. cytherea nubbins, with the presence of A2 subclade only once, and A6, 5 times, with both subclades occurring at a background level (<1%). Clade C was detected in 9 nubbins at the background level (<0.2%) except for one nubbin (A01.3 composed of 59% A and 41% C). Two C subclades, C3 and C3K were found concomitantly (in 4 nubbins) or separately (in 2 and 3 nubbins, respectively). Finally, clade D (subclade D10) was found in 5 nubbins, two of which contained D10 above the background level (7% and 14%). No significant correlation between Symbiodiniaceae clade/subclade and host assemblages or temperature was found. Moreover, no variation of nubbin color, was recorded according to temperature levels and/or assemblages meaning that there was no drastic variation in Symbiodiniaceae densities.
Identification of candidate genes for dimethylsulfoniopropionate biosynthesis in Tridacna maxima
The blasting of the predicted peptide T. maxima dataset against a DMSP enzyme custom database allowed the selection of 475 predicted peptides potentially involved in pathways of DMSP biosynthesis (Table S2 and S5, sheet 2). After functional clustering, all identified peptides with potential roles in DMSP synthesis were found to exhibit at least 1 homolog per step. Among them, 69 were significantly (False Discovery Rate <0.05) up-regulated during a thermal stress event (Table S5, sheet 1). More precisely, 29 peptides were identified as homologs to those of the coral pathway: 6 homologs of AT1 and AT5 for transamination step, 16 for the reduction step, 6 homologs of REDOX2,3,4,6 and 10, for the methylation step of METHYL 1 and 2, and one homolog of DECARB3 for the decarboxylation step (Fig. 4).
Figure 4.
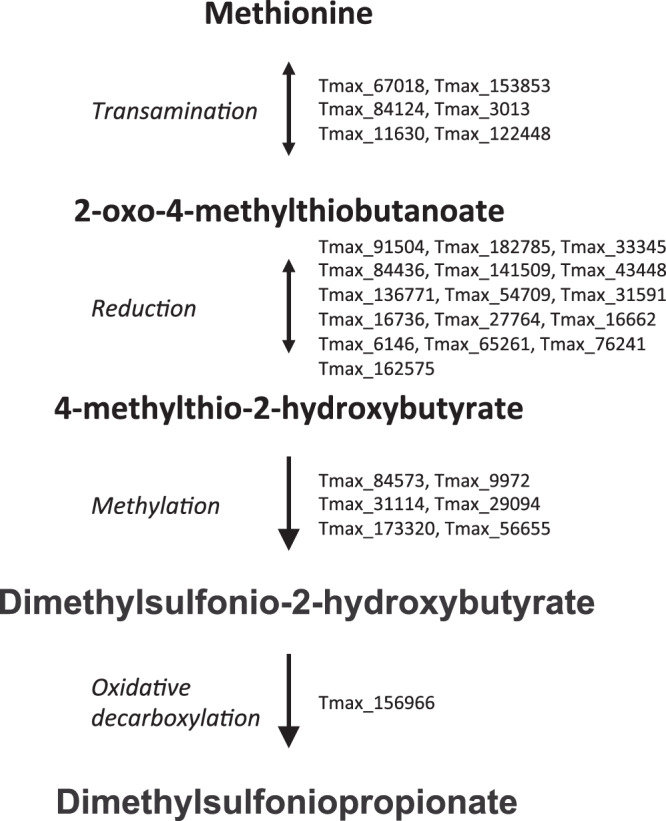
Putative pathway of dimethylsulfoniopropionate biosynthesis in Tridacna maxima. ID numbers of T. maxima peptides are indicated for each step.
Discussion
Dimethylsulfoniopropionate concentrations differ according to species and assemblage
Dimethylsulfoniopropionate concentrations varied among the three invertebrate species analyzed. In A. cytherea, DMSP concentrations were 5 to 10-fold higher than in P. damicornis. A previous study performed on Pocilloporidae and Acroporidae, measured DMSP concentrations of, respectively, 0.333 nmol/mm2 and 2.473 nmol/mm2 in P. damicornis and A. millepora19, which is consistent with our results. Also, similar DMSP concentrations were found in three different species from the Pocilloporidae family, while they were highly variable in Acroporidae19. As we used different NMR quantification approaches for corals and giant clams, we were not able to directly compare concentrations between them. Nevertheless, beyond differences between species, our results revealed that the DMSP concentrations might vary in a species according to its close environment, namely interspecies assemblages.
In corals, increases in DMSP have been associated with direct sunlight, thermal stress, air exposure or oxidative stressors3,5,14. Measurements of DMSP concentration in Acropora species (A. aspera, A. tenuis and A. millepora) after 467 and 53 days at 31 °C revealed an increase of DMSP concentration in bleached corals. Here we show that after 2 days at >31 °C the DMSP production, in still unbleached A. Cytherea, P. damicornis and T. maxima, is not sufficiently enhanced to detect a significant difference in DMSP concentrations between stressed and unstressed corals and giant clams. This indicates that at least three days of temperature stress and/or the concurrent loss/gain of a certain quantity of Symbiodiniaceae/bacteria might be required for enhancing DMSP production. However, significant variations of DMSP concentrations were observed among the three species. These variations occurred in different environmental contexts. For A. cytherea, DMSP concentration increased significantly in AT assemblages but not in PA and PAT assemblages, whereas for T. maxima, it increased in PAT assemblages but decreased in association with A. cytherea. These previous results suggest that DMSP concentration in coral reef holobionts is dependent on neighboring species, possibly acting as a stressor14 or an activator of the sulfur metabolism. For P. damicornis, the association with A. cytherea alone or with T. maxima does not lead to a highly significant increase in DMSP. For A. cytherea, an increase of DMSP is observed in the AT assemblage. However, when P. damicornis is also present in the assemblage (PA or PAT), this effect disappeared. Therefore, the presence of P. damicornis prevents the clams effect’, highlighting possible yet uncharacterized interspecies communication responses. It is also known that, depending on the species, the variation in DMSP is not always observed under the same environmental factor. For example, a decrease in salinity leads to a decrease of DMSP in A. millepora, but such an effect had not previously been observed in Stylophora pistillata and P. damicornis68. In the present study, contrary to A. cytherea, the DMSP concentration in giant clams was increased in PAT assemblages rather than in AT assemblages. For giant clams, the presence of the two coral species seems to have a stronger effect than the sole presence of A. cytherea. In the case of DMSP as an indicator of stress, we have previously shown that giant clam mortality occurred in presence of A. cytherea, in both AT and PAT assemblages40. However, giant clam DMSP increases occurred only in PAT assemblages and we did not find any correlation between health status and DMSP levels in giant clams. Thus, the “health status” (i.e. declining clams) does not appear to systematically lead to a significant change in DMSP level. Taken together, our results suggest that DMSP concentration in the holobionts is influenced by their neighboring species, modifying the metabolism of the sulfur pathway.
Dimethylsulfoniopropionate production
The variations in DMSP concentrations among invertebrates harboring Symbiodiniaceae have been essentially attributed to the types and densities of their symbiotic dinoflagellates20. Significant DMSP variation also exists between Symbiodiniaceae clades5,20,33,67. Consequently, the types and densities of Symbiodiniaceae possibly impact the DMSP concentrations in holobionts, by translocating DMSP to their hosts69,70. Nevertheless, DMSP concentrations and the types of clades in our samples were not correlated. While the DMSP concentration of A. cytherea was high in AT assemblages and low in PAT assemblages, the relative abundance of clade A and D were similar in both assemblages. If Symbiodiniaceae are indeed responsible for the DMSP concentrations, we have to assume that their density should be higher in A. cytherea nubbins in AT than in other assemblages. Moreover, as DMSP concentrations in P. damicornis nubbins are between 5 and 10-fold lower than in A. cytherea nubbins, and since clade D has been described as a lower DMSP producer than clade C67, we can also hypothesize that clade D is a lower DMSP producer than clade A, a permanent clade in A. cytherea. Another non-exclusive alternative hypothesis for explaining the difference in DMSP production between A. cytherea and P. damicornis relies on a difference in Symbiodiniaceae densities between these two coral species, as observed in our previous work, A. cytherea harboring more Symbiodiniaceae than P. damicornis in Moorea59. Regarding giant clams, as for corals, Symbiodiniaceae types are not sufficient to account for DMSP concentrations variations between assemblages. At the individual level, giant clams in the same assemblage, with either 99% of subclade C66 or 99% of subclade A6 did not exhibit any difference in DMSP concentration40. Thus, even if the Symbiodiniaceae are well known DMSP producers, our results suggested that Symbiodiniaceae genotypes did not account for the DMSP variations observed in our study.
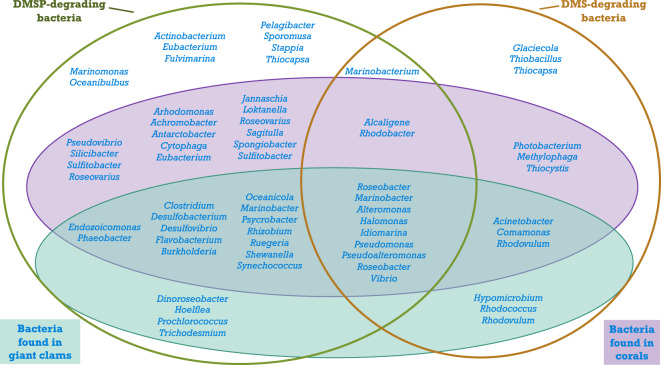
In addition to Symbiodiniaceae, corals and giant clams harbor numerous other associated organisms, including various families of bacteria. Raina and collaborators37 reported that more than 65% of the bacteria genera playing a role in DMSP/DMS metabolism can be found in association with coral species. The bacterial communities associated within the giant clams of this experiment have already been characterized40. By overlapping the data from both studies, we demonstrated that several bacterial genera involved in the degradation of DMSP or DMS in corals were also associated with T. maxima (Fig. 5). This suggests that the microbial communities of giant clams are also structured by the presence of DMSP and could indicate that bacteria influence the composition and variation in DMSP concentration between assemblages. However, as for Symbiodiniaceae, no link was found between bacterial composition and assemblages in T. maxima. Altogether our results suggested that the variation in DMSP concentration observed here did not result from Symbiodiniaceae or bacterial genera present in the sample, but might result from differences in their relative densities.
Figure 5.
Marine bacterial genera degrading DMSP or DMS in the water column, associated or not with corals and/or giant clams. DMSP-degrading (green circle) and DMS-degrading (brown circle) found in the water column, bacterial genera found in corals (blue oval) and in giant clams (green oval) (adapted from37,40; and further developed from71–77).
Recent studies have highlighted the existence of DMSP biosynthesis pathways in animals, and in corals notably3,15. The increase in DMSP concentration measured in some assemblages might also be due to an enhancement in DMSP biosynthesis by the hosts, including giant clams. Of relevance, we showed for the first time that, similarly to corals, the giant clam T. maxima possesses algal gene homologs putatively involved in DMSP biosynthesis pathways. Candidate genes were not only homologous to DMSP alga-like pathway genes (top blast hit), but were also overexpressed during a thermal stress event. As at least one candidate gene for each step of the alga-like pathway has been found, we assume that the whole pathway is present in T. maxima. Future studies on complete holobiont communities and gene expression by assemblage will be required to decipher the involvement of each holobiont partner in DMSP biosynthesis.
This study explored the influence of interspecies assemblages on DMSP concentrations in corals and giant clams. We showed that DMSP concentration is higher in A. cytherea when associated with T. maxima and higher in T. maxima when they are in a three species assemblage. Because the balance between Symbiodiniaceae genera was maintained regardless of host assemblages, our results also show that microbial communities cannot solely account for the DMSP differences observed within and among assemblages. Further studies are required to decipher if symbiont densities and/or the host alga-like DMSP biosynthesis pathway contribute to the differences in DMSP production between assemblages.
Supplementary information
Acknowledgements
This work was supported by LabexCorail “SynTox”. I. Guibert was a fellow of Sorbonne University – Doctoral School 129. We acknowledge the InterContinental Resort & Spa Moorea and the Moorea Dolphin Center for providing a coral garden protected area. We also thank Franck Lerouvreur, Pascal Ung, Ewen Morin and all the students from CRIOBE who helped us in the field. We are much indebted to Roy Benyon for English improvement of the manuscript.
Author contributions
Conceived and designed the experiments: I.G., V.B., P.R., G.L., Performed the experiments: I.G., V.D., V.B., Collected the samples: I.G., Processed the samples: I.G., F.B., G.L., I.B., Generated the data: F.B., V.D., X.P., G.L., Analysed the data: I.G., G.L., Wrote the manuscript: I.G., V.B., G.L., I.B., X.P.
Data availability
Supplementary data for this article can be found at Aquatic Sciences Website. All data are available upon request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Summary statement Various assemblages of corals and giant clams modulate dimethylsulfoniopropionate concentration, a key compound in the marine sulfur cycle.
Supplementary information
is available for this paper at 10.1038/s41598-020-66290-5.
References
- 1.Broadbent AD, Jones GB, Jones RJ. DMSP in corals and benthic algae from the Great Barrier Reef. Estuar. Coast. Shelf Sci. 2002;55:547–555. [Google Scholar]
- 2.Hill RW, Dacey JWH, Krupp DA. Dimethylsulfoniopropionate in reef corals. Bull. Mar. Sci. 1995;57:489–494. [Google Scholar]
- 3.Raina JB, et al. DMSP biosynthesis by an animal and its role in coral thermal stress response. Nature. 2013;502:677–80. doi: 10.1038/nature12677. [DOI] [PubMed] [Google Scholar]
- 4.Downs CA, et al. Oxidative stress and seasonal coral bleaching. Free Radic. Biol. Med. 2002;33:533–543. doi: 10.1016/s0891-5849(02)00907-3. [DOI] [PubMed] [Google Scholar]
- 5.Deschaseaux ESM, et al. Effects of environmental factors on dimethylated sulfur compounds and their potential role in the antioxidant system of the coral holobiont. Limnol. Oceanogr. 2014;59:758–768. [Google Scholar]
- 6.Sunda W, Kieber DJ, Kiene RP, Huntsman S. An antioxidant function for DMSP and DMS in marine algae. Nature. 2002;418:317–320. doi: 10.1038/nature00851. [DOI] [PubMed] [Google Scholar]
- 7.Kirst GO, et al. Dimethylsulfoniopropionate (DMSP) in ice-algae and its possible biological role. Mar. Chem. 1991;35:381–388. [Google Scholar]
- 8.Trevena AJ, Jones GB, Wright SW, Van Den Enden RL. Profiles of DMSP, algal pigments, nutrients and salinity in pack ice from eastern Antarctica. J. Sea Res. 2000;43:265–273. [Google Scholar]
- 9.Evans C, Malin G, Wilson WH, Liss PS. Infectious titers of Emiliania huxleyi virus 86 are reduced by exposure to millimolar dimethyl sulfide and acrylic acid. Limnol. Oceanogr. 2006;51:2468–2471. [Google Scholar]
- 10.Havill DC, Ingold A, Pearson J. Sulphide tolerance in coastal halophytes. Vegetatio. 1985;62:279–285. [Google Scholar]
- 11.Seymour JR, Simó R, Ahmed T, Stocker R. Chemoattraction todimethylsulfoniopropionate throughout the marine microbial food web. Science. 2010;329:342–346. doi: 10.1126/science.1188418. [DOI] [PubMed] [Google Scholar]
- 12.Van Rijssel M, Gieskes WWC. Temperature, light, and the dimethylsulfoniopropionate (DMSP) content of Emiliania huxleyi (Prymnesiophyceae) J. Sea Res. 2002;48:17–27. [Google Scholar]
- 13.Van Alstyne KL, Gifford SA, Dohman JM, Savedo MM. Effects of environmental changes, tissue types and reproduction on the emissions of dimethyl sulfide from seaweeds that form green tides. Environ. Chem. 2016;13:220–230. [Google Scholar]
- 14.Yost DM, Jones RJ, Mitchelmore CL. Alterations in dimethylsulfoniopropionate (DMSP) levels in the coral Montastraea franksi in response to copper exposure. Aquat. Toxicol. 2010;98:367–373. doi: 10.1016/j.aquatox.2010.03.005. [DOI] [PubMed] [Google Scholar]
- 15.Aguilar C, et al. Transcriptomic analysis of the response of Acropora millepora to hypo-osmotic stress provides insights into DMSP biosynthesis by corals. BMC Genomics. 2017;18:612–626. doi: 10.1186/s12864-017-3959-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rap A, et al. Natural aerosol direct and indirect radiative effects. Geophys. Res. Lett. 2013;40:3297–3301. [Google Scholar]
- 17.Nunes J, et al. Two intertidal, non-calcifying macroalgae (Palmaria palmata and Saccharina latissima) show complex and variable responses to short-term CO2 acidification. ICES J. Mar. Sci. 2016;73:887–896. [Google Scholar]
- 18.Simó R. Production of atmospheric sulfur by oceanic plankton: Biogeochemical, ecological and evolutionary links. Trends Ecol. Evol. 2001;16:287–294. doi: 10.1016/s0169-5347(01)02152-8. [DOI] [PubMed] [Google Scholar]
- 19.Tapiolas DM, Raina JB, Lutz A, Willis BL, Motti CA. Direct measurement of dimethylsulfoniopropionate (DMSP) in reef-building corals using quantitative nuclear magnetic resonance (qNMR) spectroscopy. J. Exp. Mar. Bio. Ecol. 2013;443:85–89. [Google Scholar]
- 20.Van Alstyne KL, Schupp P, Slattery M. The distribution of dimethylsulfoniopropionate in tropical Pacific coral reef invertebrates. Coral Reefs. 2006;25:321–327. [Google Scholar]
- 21.Becker S, et al. The biology of an Antarctic rhodophyte, Palmaria decipiens: recent advances. Antarct. Sci. 2011;23:419–430. [Google Scholar]
- 22.Kocsis MG, et al. Dimethylsulfoniopropionate biosynthesis in Spartina alterniflora. Evidence that S-methylmethionine and dimethylsulfoniopropylamine are intermediates. Plant Physiol. 1998;117:273–281. doi: 10.1104/pp.117.1.273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.White BA, Lansing E, Hill RW, Dacey JWH. Accumulation of Dimethylsulfonioprionate in Geukensia demissa Depends on trophic Interactions. Nature. 1995;189:235–236. doi: 10.1086/BBLv189n2p235. [DOI] [PubMed] [Google Scholar]
- 24.Van Alstyne KL, Dominique VJ, Muller-Parker G. Is dimethylsulfoniopropionate (DMSP) produced by the symbionts or the host in an anemone-zooxanthella symbiosis? Coral Reefs. 2009;28:167–176. [Google Scholar]
- 25.Hanson AD, Rivoal J, Paquet L, Cage DA. Biosynthesis of 3-Dimethylsulfoniopropionate in Wollastonia biflora (L.) DC. Plant Physiol. 1994;105:103–110. doi: 10.1104/pp.105.1.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Stefels J. Physiological aspects of the production and conversion of DMSP in marine algae and higher plants. J. Sea Res. 2000;43:183–197. [Google Scholar]
- 27.Keller MD, Bellows WK, Guillard RRL. Dimethyl sulfide production in marine phytoplankton. Biog. sulfur Environ. 1989;393:167–182. [Google Scholar]
- 28.Hill RW, Dacey JWH, Edward A. Dimethylsulfoniopropionate in giant clams (Tridacnidae) Biol. Bull. 2000;199:108–115. doi: 10.2307/1542870. [DOI] [PubMed] [Google Scholar]
- 29.Steinke M, Brading P, Kerrison P, Warner ME, Suggett DJ. Concentrations of dimethylsulfoniopropionate and dimethyl sulfide are strain-specific in symbiotic dinoflagellates (Symbiodinium sp., dinophyceae) J. Phycol. 2011;47:775–783. doi: 10.1111/j.1529-8817.2011.01011.x. [DOI] [PubMed] [Google Scholar]
- 30.Pochon X, Gates RD. A new Symbiodinium clade (Dinophyceae) from soritid foraminifera in Hawai’i. Mol. Phylogenet. Evol. 2010;56:492–497. doi: 10.1016/j.ympev.2010.03.040. [DOI] [PubMed] [Google Scholar]
- 31.LaJeunesse TC, et al. Systematic revision of Symbiodiniaceae highlights the antiquity and diversity of coral endosymbionts. Curr. Biol. 2018;28:1–11. doi: 10.1016/j.cub.2018.07.008. [DOI] [PubMed] [Google Scholar]
- 32.Neo ML, Eckman W, Vicentuan K, Teo SLM, Todd PA. The ecological significance of giant clams in coral reef ecosystems. Biol. Conserv. 2015;181:111–123. [Google Scholar]
- 33.Yost DM, Mitchelmore CL. Dimethylsulfoniopropionate (DMSP) lyase activity in different strains of the symbiotic alga Symbiodinium microadriaticum. Mar. Ecol. Prog. Ser. 2009;386:61–70. [Google Scholar]
- 34.Van Alstyne KL, Puglisi MP. DMSP in marine macroalgae and macroinvertebrates: Distribution, function, and ecological impacts. Aquat. Sci. 2007;69:394–402. [Google Scholar]
- 35.Howard EC, et al. Bacterial Taxa That Limit Sulfur Flux from the Ocean. Science. 2006;314:649–652. doi: 10.1126/science.1130657. [DOI] [PubMed] [Google Scholar]
- 36.Littman RA, Willis BL, Pfeffer C, Bourne DG. Diversities of coral-associated bacteria differ with location, but not species, for three acroporid corals on the Great Barrier Reef. FEMS Microbiol. Ecol. 2009;68:152–163. doi: 10.1111/j.1574-6941.2009.00666.x. [DOI] [PubMed] [Google Scholar]
- 37.Raina JB, Dinsdale EA, Willis BL, Bourne DG. Do the organic sulfur compounds DMSP and DMS drive coral microbial associations? Trends Microbiol. 2010;18:101–108. doi: 10.1016/j.tim.2009.12.002. [DOI] [PubMed] [Google Scholar]
- 38.Todd JD, et al. Structural and regulatory genes required to make the gas dimethyl sulfide in bacteria. Science. 2007;315:666–669. doi: 10.1126/science.1135370. [DOI] [PubMed] [Google Scholar]
- 39.Raina JB, Tapiolas D, Willis BL, Bourne DG. Coral-associated bacteria and their role in the biogeochemical cycling of sulfur. Appl. Environ. Microbiol. 2009;75:3492–3501. doi: 10.1128/AEM.02567-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Guibert, I., Pochon, X., Lecellier, G. & Berteaux-lecellier, V. Metabarcoding reveals distinct microbiotypes in the giant clam Tridacna maxima. Microbiome 8, 57 (2020). [DOI] [PMC free article] [PubMed]
- 41.Curson ARJ, et al. Dimethylsulphoniopropionate biosynthesis in marine bacteria and identification of the key gene in this process. Nat. Microbiol. 2017;2:1–35. doi: 10.1038/nmicrobiol.2017.9. [DOI] [PubMed] [Google Scholar]
- 42.Silverstein RN, Cunning R, Baker AC. Change in algal symbiont communities after bleaching, not prior heat exposure, increases heat tolerance of reef corals. Glob. Chang. Biol. 2015;21:236–249. doi: 10.1111/gcb.12706. [DOI] [PubMed] [Google Scholar]
- 43.Cunning R, Yost DM, Guarinello ML, Putnam HM, Gates RD. Variability of Symbiodinium communities in waters, sediments, and corals of thermally distinct reef pools in American Samoa. PLoS One. 2015;10:1–17. doi: 10.1371/journal.pone.0145099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ziegler M, Seneca FO, Yum LK, Palumbi SR, Voolstra CR. Bacterial community dynamics are linked to patterns of coral heat tolerance. Nat. Commun. 2017;8:14213. doi: 10.1038/ncomms14213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sharp KH, Pratte ZA, Kerwin AH, Rotjan RD, Stewart FJ. Season, but not symbiont state, drives microbiome structure in the temperate coral Astrangia poculata. Microbiome. 2017;5:120. doi: 10.1186/s40168-017-0329-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Camp, E. F. et al. Corals exhibit distinct patterns of microbial reorganisation to thrive in an extreme inshore environment. Coral Reefs 3, 1–16 (2020).
- 47.Rosenberg E, Zilber-Rosenberg I. Symbiosis and development: The hologenome concept. Birth Defects Res. Part C - Embryo Today Rev. 2011;93:56–66. doi: 10.1002/bdrc.20196. [DOI] [PubMed] [Google Scholar]
- 48.Chornesky E. Induced development of sweeper tentacles on the reef coral Agaricia agaricites: A response to direct competition. Biol. Bull. 1983;165:569–581. doi: 10.2307/1541466. [DOI] [PubMed] [Google Scholar]
- 49.Connell JH, et al. A long-term study od competition and diversity of corals. Ecol. Monogr. 2004;74:179–210. [Google Scholar]
- 50.Lang J. Interspecific aggression by scleractinian corals. 2. why the race is not only to the swift. Bull. Mar. Sci. 1973;23:260–279. [Google Scholar]
- 51.Adjeroud M, Briand MJ, Kayal M, Dumas P. Coral assemblages in Tonga: Spatial patterns, replenishment capacities, and implications for conservation strategies. Environ. Monit. Assess. 2013;185:5763–5773. doi: 10.1007/s10661-012-2982-5. [DOI] [PubMed] [Google Scholar]
- 52.Guibert I, et al. Differential effects of coral-giant clam assemblages on biofouling formation. Sci. Rep. 2019;9:1–12. doi: 10.1038/s41598-019-39268-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Dubousquet V, et al. Changes in fatty acid composition in the giant clam Tridacna maxima in response to thermal stress. Biol. Open. 2016;5:1400–1407. doi: 10.1242/bio.017921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Rouzé H, Lecellier G, Langlade M, Planes S, Berteaux-Lecellier V. Fringing reefs exposed to different levels of eutrophication and sedimentation can support the same benthic communities. Mar. Pollut. Bull. 2015;92:212–221. doi: 10.1016/j.marpolbul.2014.12.016. [DOI] [PubMed] [Google Scholar]
- 55.Siebeck UE, Marshall NJ, Klüter A, Hoegh-Guldberg O. Monitoring coral bleaching using a colour reference card. Coral Reefs. 2006;25:453–460. [Google Scholar]
- 56.Siebeck, U. E., Logan, D. & Marshall, N. J. CoralWatch - a flexible coral bleaching monitoring tool for you and your group. Proc. 11th Int. Coral Reef Symp. 549–553 (2008).
- 57.Mohamadi, F. La métabolomique appliquée à l’étude de l’impact de stress environnementaux sur les coraux scléractiniaires (2014).
- 58.R core team. A language and environment for statistical computing (2014).
- 59.Rouzé H, et al. An updated assessment of Symbiodinium spp. that associate with common scleractinian corals from Moorea (French Polynesia) reveals high diversity among background symbionts and a novel finding of clade B. PeerJ. 2017;5:1–22. doi: 10.7717/peerj.2856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Pochon X, Pawlowski J, Zaninetti L, Rowan R. High genetic diversity and relative specificity among Symbiodinium-like endosymbiotic dinoflagellates in soritid foraminiferans. Mar. Biol. 2001;139:1069–1078. [Google Scholar]
- 61.Stat M, Pochon X, Cowie ROM, Gates RD. Specificity in communities of Symbiodinium in corals from Johnston Atoll. Mar. Ecol. Prog. Ser. 2009;386:83–96. [Google Scholar]
- 62.Kozich JJ, Westcott SL, Baxter NT, Highlander SK, Schloss PD. Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the miseq illumina sequencing platform. Appl. Environ. Microbiol. 2013;79:5112–5120. doi: 10.1128/AEM.01043-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Schloss PD, et al. Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 2009;75:7537–7541. doi: 10.1128/AEM.01541-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Arif C, et al. Assessing Symbiodinium diversity in scleractinian corals via next-generation sequencing-based genotyping of the ITS2 rDNA region. Mol. Ecol. 2014;23:4418–4433. doi: 10.1111/mec.12869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Li B, Dewey CN. RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinformatics. 2011;12:323. doi: 10.1186/1471-2105-12-323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Dimont E, Shi J, Kirchner R, Hide W. edgeRun: An R package for sensitive, functionally relevant differential expression discovery using an unconditional exact test. Bioinformatics. 2015;31:2589–90. doi: 10.1093/bioinformatics/btv209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Deschaseaux ESM, et al. Comparative response of DMS and DMSP concentrations in Symbiodinium clades C1 and D1 under thermal stress. J. Exp. Mar. Bio. Ecol. 2014;459:181–189. [Google Scholar]
- 68.Gardner SG, et al. Dimethylsulfoniopropionate, superoxide dismutase and glutathione as stress response indicators in three corals under short-term hyposalinity stress. Proc. R. Soc. B Biol. Sci. 2016;283:1–9. doi: 10.1098/rspb.2015.2418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hill RW, Dacey JW, Hill SD, Edward A, Hicks WA. Dimethylsulfoniopropionate in six species of giant clams and the evolution of dimethylsulfide after death. Can. J. Fish. Aquat. Sci. 2004;61:758–764. [Google Scholar]
- 70.Yost DM, Mitchelmore CL. Determination of total and particulate dimethylsulfoniopropionate (DMSP) concentrations in four scleractinian coral species: A comparison of methods. J. Exp. Mar. Bio. Ecol. 2010;395:72–79. [Google Scholar]
- 71.Alex A, Antunes A. Genus-wide comparison of Pseudovibrio bacterial genomes reveal diverse adaptations to different marine invertebrate hosts. PLoS One. 2018;13:1–25. doi: 10.1371/journal.pone.0194368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Zeng YX, Qiao ZY, Yu Y, Li HR, Luo W. Diversity of bacterial dimethylsulfoniopropionate degradation genes in surface seawater of Arctic Kongsfjorden. Sci. Rep. 2016;6:1–10. doi: 10.1038/srep33031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.González JM, et al. Silicibacter pomeroyi sp. nov. and Roseovarius nubinhibens sp. nov., dimethylsulfoniopropionate-demethylating bacteria from marine environments. Int. J. Syst. Evol. Microbiol. 2003;53:1261–1269. doi: 10.1099/ijs.0.02491-0. [DOI] [PubMed] [Google Scholar]
- 74.Rossbach, S., Cardenas, A., Perna, G., Duarte, C. M. & Voolstra, C. R. Tissue-specific microbiomes of the Red Sea giant clam Tridacna maxima highlight differential abundance of Endozoicomonadaceae. Front. Microbiol. 10 (2019). [DOI] [PMC free article] [PubMed]
- 75.Shiu JH, et al. A newly designed primer revealed high phylogenetic diversity of endozoicomonas in coral reefs. Microbes Environ. 2018;33:172–185. doi: 10.1264/jsme2.ME18054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Schäfer H. Isolation of Methylophaga spp. from marine dimethylsulfide-degrading enrichment cultures and identification of polypeptides induced during growth on dimethylsulfide. Appl. Environ. Microbiol. 2007;73:2580–2591. doi: 10.1128/AEM.02074-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Cai L, et al. Season structures prokaryotic partners but not algal symbionts in subtropical hard corals. Appl. Microbiol. Biotechnol. 2018;102:4963–4973. doi: 10.1007/s00253-018-8909-5. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Supplementary data for this article can be found at Aquatic Sciences Website. All data are available upon request.