Version Changes
Revised. Amendments from Version 1
Based on the reviews received we corrected some typographical errors, added some useful additional references, revised a few sentences for clarity and emphasised the importance of education to support rabies control programmes and ensure their sustainability.
Abstract
Dog-mediated rabies continues to kill tens of thousands of people every year in low- and middle-income countries despite being an entirely vaccine-preventable disease. WHO and partners have launched a global campaign to reach zero human deaths from dog-mediated rabies by 2030. The primary tools for reaching this target are mass dog vaccination to interrupt transmission in domestic dog populations that maintain infection, appropriate post-exposure prophylaxis (PEP) for rabies-exposed persons to prevent the fatal onset of disease, together with education to support their effective uptake. Models have been developed to assess the feasibility, impact and cost-effectiveness of these measures. From these models, we argue that the 2030 target of zero human rabies deaths is achievable, but will require concerted effort, engagement and investment. A proposed Gavi investment in human rabies vaccines has potential to drive progress towards the 2030 target; however, concomitant investment is needed to scale up mass dog vaccination or this target will be missed. Predicted economic benefits of mass dog vaccination vary according to national PEP provisioning and healthcare access. Integrated Bite Case Management can enhance surveillance and rationalize PEP use, but needs adapting to and integrating within local health systems and international reporting systems to improve PEP accountability, monitor impacts and support verification of disease freedom. Modelling approaches need refining to project realistic and geographically specific timelines for achieving targets. Model iterations informed by data on the implementation of interventions can be used to evaluate progress and guide future strategies. Critically such models are needed to advocate for investment, since the greatest risk to the ‘Zero by 30’ strategy is the limited long-term cross-sectoral or targeted financing to support countries to deliver and sustain mass dog vaccination.
Keywords: canine rabies, WHO guidelines, post-exposure prophylaxis, validation, verification, mass dog vaccination, zoonosis, surveillance, integrated bite case management
Disclaimer
The views expressed in this article are those of the author(s). The opinions expressed herein are those of the authors and do not necessarily reflect the views of the World Health Organisation. Publication in Gates Open Research does not imply endorsement by the Gates Foundation.
Background
In over 120 countries around the world, rabies presents a significant threat to human lives and a considerable public health burden. Around 60,000 people die from rabies annually, whilst tens of millions receive costly post-exposure prophylaxis (PEP) 1. The vast majority of these human rabies cases (>99%) are contracted through the bite of a rabid dog 2. Although fatal following the onset of clinical disease, rabies is entirely preventable. Administration of PEP to rabies exposed persons according to WHO guidelines prevents disease progression and death 3. However, PEP does not reduce transmission in source domestic dog populations. Mass dog vaccination has been the foundation for the successful elimination of rabies from dogs in North America, Western Europe, Japan and much of Central and South America 2. In contrast, mass dog vaccination has barely started in most low- and middle-income countries (LMICs) in Africa and Asia, where the disease remains widespread.
Within the context of neglected tropical diseases (NTDs), zoonoses like rabies have been especially neglected. Rabies was not amongst the diseases prioritized in the 2012 London Declaration, endorsed by international, non-government, government and industry partners that committed over US$785 million to the control or elimination of 10 NTDs by 2020. Investment in the 2012 roadmap overlooked zoonoses requiring control of disease in animal or vector reservoirs 4. However, rabies was listed amongst five Neglected Zoonotic Diseases (NZDs) within the 66th World Health Assembly resolution on NTDs in 2013 5. The lack of progress on rabies control in Africa and Asia reflects the omission from high-level political advocacy 6, and the consequences of minimal investment. A One Health approach is increasingly advocated, recognizing the interactions between human and animal health and the required intersectoral collaboration at local, national and international levels. But concomitant investment in veterinary public health to deal with endemic zoonoses has been limited. Without financing mechanisms, veterinary services in most LMICs will remain focused on livestock diseases considered valuable for trade and will lack the resources needed for mass dog vaccination.
WHO and partners recently launched a global campaign to address the neglect of rabies, with the aim of achieving zero human deaths from dog-mediated rabies by 2030 7. Some regions already had targets to interrupt transmission in dogs or reach zero human deaths. The Americas have progressed remarkably: human rabies deaths in the region have declined by >99% from endemic levels, many countries have not reported human deaths from dog-mediated rabies in over a decade (only three countries reported deaths in 2016 8), and have ostensibly eliminated transmission, with mass dog vaccination discontinued from large areas. Although the Americas had to reset their target dates and are still to verify freedom from dog-mediated rabies, their successes demonstrate what is possible through coordinated mass dog vaccination. The Southeast Asia and Western Pacific region’s targets for zero deaths from 2020 9 have both been reset following limited progress. The Pan-African Rabies Control Network (PARACON) and Association of Southeast Asian Nations (ASEAN) united regional groups across Africa and Southeast Asia, respectively, and are involved in pushing the rabies elimination agenda. However, with a few exceptions 10, countries on both continents have yet to demonstrate significant case reductions. Development of a new NTD roadmap for 2030 that includes rabies provides an opportunity to build on successes and reflect on failures to constructively identify obstacles and overcome them.
For rabies, the new 2030 NTD roadmap, for the first time, identifies time-bound targets for countries to achieve the long-term goal of zero human rabies deaths, intermediate milestones of 50% mortality reduction and operational prerequisites of 70% mass dog vaccination coverage in high-risk areas 11. Modelling contributed to policy development in the build up to and subsequent launch of the ‘Zero by 30’ campaign. In this letter, we describe insights from modelling that can inform progress towards the 2030 NTD roadmap and lay out challenges to meeting these targets.
Insights from modelling supporting policy development
The historic lack of investment in NZDs has previously been attributed to an absence of estimates of their burden on society to motivate policy 12– 14. Human and animal rabies is vastly under-reported in most endemic regions due to a lack of surveillance and reporting capacity, contributing to this cycle of neglect 15, 16. With this in mind, the Global Alliance for Rabies Control commissioned a study to estimate the global burden of dog-mediated rabies. Building on decision tree models 17, 18 and using updated country data, burden estimates were generated that have been widely cited in advocacy 1, 19.
Gavi, the Vaccine Alliance, has considered investing in rabies as part of its mission to increase the equitable use of vaccines for children in the world’s poorest countries. The Gavi Learning Agenda on rabies, the outcome of Gavi’s 2013 Vaccine Investment Strategy decision, complemented key policy processes: a Strategic Advisory Group of Experts (SAGE) Working Group reviewed evidence on rabies vaccines and immunoglobulins 20 (July 2016–April 2018) and a review of models on the impact and cost-effectiveness of rabies prevention strategies was undertaken through the Immunization and Vaccines related Implementation Research Advisory Committee (IVIR-AC) in 2017. Both SAGE and IVIR-AC models highlighted that one strategy under consideration, rabies pre-exposure prophylaxis within a routine Expanded Programme on Immunization (EPI), would be expensive and much less cost-effective than direct prevention through PEP 21. For post-exposure vaccination, intradermal (ID) administration was under all conditions more cost-effective than the widely used intramuscular route 21. An abridged one-week ID regimen was universally preferred over other regimens recognized by WHO as safe and effective, with potential to reduce costs and be more resilient to stockouts during outbreaks 21. A dose sparing approach to administration (wound infiltration only) of rabies immunoglobulins was also demonstrated to be more cost-effective than previous recommendations 21. These outcomes considerably simplified recommendations for rabies PEP, which had previously been regarded as unwieldy and complex, and informed the latest WHO position 3, providing a preferred strategy for improved PEP access under Gavi investment.
Gavi recently announced their support for rabies post-exposure vaccination beginning 2021, subject to funding availability. Models projected that under improved PEP access (Gavi investment), total human rabies vaccine use could remain similar to the status quo across Gavi-eligible countries from 2020–2035 ( ~73 million), but with millions more (17.4 million) vaccinated by switching to the abridged one-week ID regimen 22. This would prevent around half a million rabies deaths, making improved PEP access an extremely cost-effective intervention at around $600 per death averted and costing approximately $403.7 million for the 46 currently Gavi-eligible countries. The counterfactual prediction was that although current PEP provisioning should save around 900,000 lives from 2021–2035, over one million people will otherwise die from rabies. This difference is attributed to addressing the market failure in vaccine supply, with the introduction of free point-of-care access increasing bite victim care seeking and compliance. Models compellingly showed how investing in PEP would be a step to improve equity and commitment to universal health coverage 23. Nonetheless, they also starkly highlighted that without concomitant investment in dog vaccination, the 2030 target of zero dog-mediated rabies deaths cannot be met. Scaling up dog vaccination according to the Zero by 30 strategy is predicted to avert a further 300,000 deaths in addition to those prevented through PEP 22. But without dog vaccination, transmission in dogs will become more entrenched and rabies deaths and PEP use are expected to steadily increase in line with population growth.
Vaccinating 70% of dog populations in high-risk areas is a cost-effective and recognized measure to break the rabies transmission cycle 24, 25. Empirical work shows that achieving 70% coverage is possible 26, 27 and reveals advantages and disadvantages of strategies according to setting 28– 30. Models identified heterogeneity in vaccination coverage, and specifically areas of low coverage, as a major impediment to progress 30, 31. Coverage gaps can result from limited resources, poor planning, neglect of particular communities, non-participation by local government units and a lack of monitoring and evaluation. Effective education, awareness raising and advertising are key to participation in dog vaccination campaigns and achieving high coverage, as well as for ensuring persons at risk of rabies exposure seek PEP. Models further suggest that rabies persists through metapopulation processes 32, 33. Limited geographical extent of dog vaccinations can therefore leave areas vulnerable to incursions from neighbouring endemic populations 33– 35. This highlights the need for sustainability, as mass dog vaccination campaigns need maintaining until elimination is achieved in focal areas and their neighbouring populations.
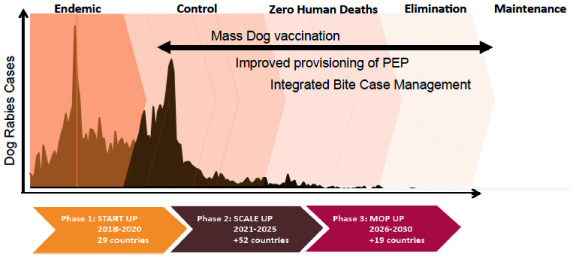
Another challenge is that even with dog vaccination, PEP costs will likely remain high unless strategies to rationalize PEP are implemented. Integrated Bite Case Management (IBCM) is advocated by WHO, to reduce the costs of PEP once rabies has been controlled 36. IBCM is a strategy that formally engages the medical and veterinary sectors to assess the risk of exposure to rabies and requirements for PEP. Implementation of IBCM in Haiti, a resource poor endemic setting, improved patient care by identifying and treating those at risk 37, whilst reducing PEP use by 40–60% 38. Current PEP access differs dramatically by setting. Chronic shortages occur in the poorest countries where the majority of patients seek care for bites by rabid dogs, whereas access is improved in some middle-income countries and sometimes hundreds of patients are treated for healthy dog bites for every rabies exposure 39– 41. In endgame settings, where rabies incidence is very low or absent ( Figure 1), PEP savings could be much higher. Models with conservative implementation of IBCM, i.e. judicious PEP only after countries reach zero human deaths, predict reduced PEP costs by 20–70% 22.
Figure 1. Interventions to reach the Zero by 30 target.
PEP, post-exposure prophylaxis.
Practical implications of the currently proposed goals
To achieve the targets of the 2030 NTD roadmap, considerable progress must be made in the implementation of rabies control and prevention ( Table 1). PEP must be distributed in sufficient quantities and equitably to all rabies-exposed people 22. Gavi support will be crucial and should provide stability and guaranteed funds to maintain production and supply of human vaccines, and catalyse pre-qualification of new suppliers to address ongoing international shortages. In parallel, in most LMICs, mass dog vaccination must be scaled up then maintained at sufficiently high levels through sustained campaigns to interrupt transmission. These efforts should be accompanied by well-planned information campaigns, to ensure that local populations participate and benefit fully from these interventions.
Table 1. Summary of challenges for reaching the neglected tropical diseases roadmap targets for rabies.
|
Current WHO
Goal |
Zero human deaths due to dog-mediated rabies by 2030 and milestones of 50% reductions in human rabies mortality
and 70% mass dog vaccination coverage in high-risk areas. Regional targets set for interruption of transmission in dogs in the Americas and zero deaths in ASEAN and SAARC countries. |
|
Technical
Feasibility |
Technically feasible, but not at current levels of implementation - mass dog vaccination and improved PEP access
needs scaling up in most LMICs. |
| Requirements | 1. Financial support for dog vaccine procurement and delivery at scale, including large-scale information and
awareness raising campaigns 2. Technical assistance for planning, implementation and monitoring of large-scale dog vaccination (e.g. training and supervision, mobile applications, standardized guidance, integrated reporting systems e.g. adaption of DHIS2) 3. Technical assistance for improving PEP access (ID training, DHIS2 adaptation, EPI integration, PEP management and accountability) 4. Targeted approaches for human and animal rabies surveillance (e.g. IBCM, use of RDTs) 5. Novel approaches for dog vaccine delivery in hard-to-reach populations (e.g. ORV, targeted CVR, community-led approaches) |
| Challenges | 1. Funding for dog vaccination
2. Limited veterinary capacity 3. Regulatory issues regarding involvement of (lay) non-veterinarian vaccinators 4. Weak surveillance with underreporting of human rabies deaths and animal rabies cases |
| Risks | 1. Reliance on PEP to prevent deaths without addressing problem at source, or assessing rabies risk
2. Use of low quality/unregulated animal (and human) vaccines 3. Requirements for private dog vaccination (paid for by dog owners) 4. Use of ineffective methods (culling) and ramifications for public trust and expectations of success 5. Limited technical support for effective surveillance and dog vaccination and its monitoring and evaluation |
| Opportunities | 1. Leverage Gavi support for PEP to advocate for dog vaccination
2. Strengthened and harmonized guidance for ‘Zero by 30’ (WHO position paper 3, WHO Technical Report Series, OIE endorsement procedures for dog-mediated rabies national control programmes, OIE Terrestrial Code, the rabies blueprint) 3. Pilot projects where catalytic funding has developed in-country capacity and a legacy of ongoing action (Tanzania, Philippines, South Africa, Haiti, Namibia, Bangladesh) 4. Innovative technical assistance (e.g. customized online and onsite training 43 - Institut Pasteur, Rabies Action Center of Excellence - FAO) 5. United Against Rabies Coalition (WHO/OIE/FAO/GARC) and committed partners including animal welfare organizations, the pharmaceutical industry, academics, civil society and local rabies champions |
WHO, World Health Organization; ASEAN, Association of Southeast Asian Nations; SAARC, South Asian Association for Regional Cooperation; PEP, post-exposure prophylaxis; DHIS2, District Health Information System 2; ID, intradermal; LMIC, low- and middle-income country; EPI, Expanded Programme on Immunization; IBCM, integrated bite case management; RDT, rapid diagnostic test; ORV, oral rabies vaccination; CVR, catch-vaccinate-release; OIE, World Organisation for Animal Health; FAO, Food and Agriculture Organization; GARC, Global Alliance for Rabies Control.
The Zero by 30 strategy ambitiously targets the scale up of dog vaccination in >100 countries over the next decade 36. Given the threat of incursions from neighbouring populations, this coordinated approach should accelerate progress. Coordination of dog vaccination across the Americas provides the model for success, but also illustrates how the poorest states impede progress 42, i.e. circulation persists where implementation of dog vaccination is weakest, seeding outbreaks elsewhere. Dog vaccination campaigns should be effectively targeted and implemented every year, such that high coverage is maintained locally and nationally, but crucially this requires monitoring and evaluation of campaigns. Moreover, delivery methods need tailoring to local contexts, which may require door-to-door vaccinations 44, capture-vaccinate-release 30, and possibly supplementary oral vaccination in well-designated areas 45, 46, in addition to central point methods 28. There is a learning curve to the scaling up of mass dog vaccination. When introduced to communities for the first time, awareness about rabies and trust in dog vaccination may be low, as is the experience and confidence of practitioners, and dog populations estimates may not be very accurate. But over consecutive campaigns dog populations should be better defined and coverage should increase 47. This progressive scaling up is recognized by the operational target for countries to achieve 70% vaccination coverage.
Rabies is highly underreported 16. Validating the targets of zero deaths and 50% mortality reductions will therefore be a challenge. The most recent WHO expert consultation on rabies included, for the first time, chapters with guidance on validation of zero human deaths and verification of dog-mediated rabies freedom 2. The NTD roadmap highlights timely diagnosis and accurate assessment of risks combined with strategic use of rapid diagnostic testing to improve surveillance, i.e. key components of IBCM. Indeed, IBCM is identified as a strategy that can sufficiently enhance surveillance for verification 48. Implementation research is, however, urgently needed to provide guidance on operationalization of IBCM across wide-ranging contexts and best practice given anticipated changes over the time horizon until 2030 ( Figure 1). Regulatory measures will be needed for quality control of rapid diagnostic tests and affordable production for larger-scale use.
Risks
The overwhelming risk to Zero by 30 is the lack of financial support for introduction, implementation and maintenance of mass dog vaccination. Scientific evidence strongly supports the need for sustained high coverage dog vaccination, but most national authorities have not committed sufficient financial resources to support this strategy. Veterinary budgets are typically much smaller than those of health services, and are largely directed towards measures for trade and animal production, not veterinary public health. In most countries that have successfully controlled rabies, the Ministry of Health has led the rabies control programme including financing dog vaccination 49. Unless Ministries of Health recognize dog vaccination as an essential public health intervention for human rabies prevention, the Zero by 2030 target will not be achieved. Instead, continued circulation of rabies in dogs may lead to an over-reliance on PEP. International investment in dog vaccine and technical support including training of public health and veterinary personnel 43, has demonstrably catalyzed action and is needed to overcome barriers that otherwise prevent rabies activities being initiated and adopted as routine by LMIC governments.
To support effective mass dog vaccination, canine vaccines must meet potency and safety standards 50. Use of cheap (substandard) vaccines is a false economy given the much greater costs of vaccine delivery. Furthermore, cheaper, lower quality vaccines can have fatal consequences and dramatically set back elimination programmes 51, 52. Countries in Latin America, with long-standing mass dog vaccination programmes and slow progress towards elimination are notably those using vaccines that do not meet international standards 53. Some countries push for local vaccine production, but quality standards need to be maintained. World Organisation for Animal Health (OIE) vaccine banks are a step towards more affordable procurement of high quality dog vaccines, but budgets need to be committed to dog vaccination for pharmaceuticals to scale up production, and this remains the most enduring challenge 54. Limited regulatory oversight for animal rabies vaccines, which would not be tolerated for human vaccines, is a failure of One Health.
Even in countries with mass dog vaccination, interruptions are common, from financial and logistical challenges to emergencies that divert funds from rabies. Lapses in mass dog vaccination prolong time to elimination 10, 30. Rapid dog population turnover causes coverage to decline and recurrent outbreaks can lead to loss of confidence in vaccination 51. If dog vaccines are not procured nationally, they may be very limited in availability. Local governments may instead promote culling, which is visible but ineffective, or dog owners may be charged for dog vaccination, leading to ineffectual low coverage 55.
A further impediment is that required improvements in surveillance will result in short-term increases in reported cases 56. This can have political implications in rabies-endemic countries and lead to resistance to policies that enhance surveillance. Addressing such perverse incentives is a challenge for elimination programmes, where pressure to reach the end can disincentivize reporting. Effective planning and communication is crucial, recognizing the importance of sustainability. Even under strong surveillance, dog vaccination must be maintained for years after cases have ceased to be reported to prevent re-emergence 57.
Future modelling priorities
Modelling now has an important role to play within the 2030 NTDs roadmap, requiring both new and better data, as well as technical development. In anticipation of Gavi investment and with country specific data, modelling should inform where populations are underserved in terms of health care to inform improved provisioning of PEP. Considerable uncertainty in how rabies control and prevention measures will be implemented makes their impacts difficult to ascertain. Forecasting stepwise progress towards goals in the context of data on the roll out of dog vaccination is therefore a priority, with the need for expedient and realistic dynamical models that can be scaled to specific geographies. The size and connectivity of dog populations determines progress towards elimination. Nonspatial models are inadequate for capturing the low endemic incidence of rabies (<1% dogs infected/year); thus, more complex, data-intensive modelling approaches are required. There is scope for such models to prioritize areas for scale up, to predict the duration over which dog vaccinations will be required to eliminate disease and to assess when dog vaccination can be relaxed. Improved data available from the roll out of mass dog vaccinations and post-vaccination monitoring should inform model development and calibration, including dog population estimates and measures of coverage. Improved data collection methods will be invaluable to populating these models. Transition to electronic data collection can provide timely spatially resolved data to better understand effective program implementation 58. Programs utilizing mobile applications for large-scale, real-time guidance of vaccination teams has shown effective in both collecting data to improve methodologies and rapidly implementing high-coverage campaigns 29.
Improved dialogue between stakeholders, with a focus on packaging and communicating guidance from models and data, would bring many benefits. Close collaboration between practitioners and modelers to assess the impact and cost-effectiveness of interventions (PEP, dog vaccination, IBCM) could improve implementation, for example, by identifying areas requiring remedial dog vaccination. Models allow the comparison of alternative control strategies, for example, by illustrating the ramifications of low efficacy vaccines or undetected cases from weak surveillance 57. Similarly models could alleviate concerns about increased case detection as surveillance improves and rabies awareness builds. Such models would therefore need to include behavioural responses, including to information and awareness campaigns. Burden estimates rely heavily on models. Improved surveillance, particularly IBCM, will provide better data to inform and validate dynamic models and burden predictions, transparently accounting for PEP use and directly measuring impact.
Increasingly, there are calls for integrated interventions in dog populations to more effectively combat diseases that they vector, such as Leishmaniasis, Echinococcosis and Dracunculiasis. Models could help build cost-effectiveness arguments for programmatic integration and inform how such interventions are targeted. Finally, as the goal of elimination approaches, models can be used to compare alternative endgame strategies and guide contingency planning for rapid response and enhanced surveillance to maintain rabies freedom.
Conclusions
WHO’s 3rd report on NTDs in 2013 highlighted four major obstacles for rabies: improving access to PEP, scaling up of mass dog vaccination, maintaining support for elimination once incidence is no longer a major public health threat, and weak surveillance to monitor progress towards targets 4. These challenges remain, but Gavi investment in human rabies vaccines addresses the first, representing an unprecedented and long-overdue recognition of the entirely vaccine-preventable burden of rabies. IBCM can strengthen surveillance over the elimination timeline and prevent PEP costs from escalating, providing a sustainable exit strategy for Gavi and a direct measure of the impact of investment. However, to achieve Zero by 30, Gavi investment needs leveraging to secure country-level and international financing for scaling up mass dog vaccination. Veterinary capacity is limited in most LMICs, and veterinary services have much smaller budgets than Ministries of health. Regulatory mechanisms for animal vaccines are also much weaker than for human vaccines, even though poor quality dog rabies vaccines and poor implementation of dog vaccination results in the deaths of people. The Zero by 30 strategy relies on countries stepping up their dog vaccination programmes over the next five years. The intermediate target of 70% coverage is the critical directly measurable indicator of whether countries deliver on this commitment and investment will be needed to support countries to do so. If countries do not, modelling will remain a counterfactual exercise to demonstrate what could have been achieved and to starkly measure how many deaths from rabies we, the global community, are willing to tolerate.
Data availability
No data are associated with this article.
Acknowledgements
Members of the WHO rabies modelling consortium:
Katie Hampson 1, Bernadette Abela-Ridder 2, Joel Changalucha 3, Sarah Cleaveland 1, Lea Knopf 2, Kennedy Lushasi 1, 3, Mary Elizabeth Miranda 4, SM Thumbi 5, 6, Kristyna Rysava 7, Tenzin Tenzin 8, Michael Tildesley 7, Ryan Wallace 9, Caroline Trotter 10
Affiliations:
1. Institute of Biodiversity, Animal Health & Comparative Medicine, College of Medical, Veterinary & Life Sciences, University of Glasgow, Glasgow, G12 8QQ, UK
2. Department of the Control of Neglected Tropical Diseases, World Health Organization, Geneva, Switzerland
3. Environmental Health and Ecological Sciences Department, Ifakara Health Institute, Ifakara, Tanzania
4. Field Epidemiology Training Program Alumni Foundation Inc., Quezon City, Philippines
5. Center for Global Health Research, Kenya Medical Research Institute, Kisumu, Kenya
6. Paul G Allen School for Global Animal Health, Washington State University, Pullman, Washington, USA
7. The Zeeman Institute for Systems Biology & Infectious Disease Epidemiology Research, School of Life Sciences and Mathematics Institute, Coventry, UK
8. National Centre for Animal Health, Department of Livestock, Ministry of Agriculture & Forests Serbithang, Babesa, Bhutan
9. Centers for Disease Control and Prevention (CDC), Atlanta, USA
10. Department of Veterinary Medicine, University of Cambridge, Cambridge, UK
Corresponding author: Katie Hampson ( katie.hampson@glasgow.ac.uk)
We are grateful to members of the WHO Rabies Modelling Consortium and of the Partners for Rabies prevention for supportive collaborations, valuable discussions and feedback. We also thank Andreia Vasconcelos for overlooking the development of this article.
Funding Statement
The authors acknowledge the support of the Bill and Melinda Gates Foundation through the funding of the NTD Modelling Consortium [OPP1184344]. This work was supported by the Wellcome Trust [207569], the Gavi Learning Agenda, the World Health Organization and the Scottish Funding Council.
The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
[version 2; peer review: 3 approved, 1 approved with reservations]
References
- 1. Hampson K, Coudeville L, Lembo T, et al. : Estimating the global burden of endemic canine rabies. PLoS Negl Trop Dis. 2015;9(4):e0003709. 10.1371/journal.pntd.0003709 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. WHO: Expert Consultation on Rabies, Third Report. (WHO, Geneva).2018. Reference Source [Google Scholar]
- 3. WHO: Rabies vaccines and immunoglobulins: WHO position. Weekly Epidemiological Record.2018;93(16):201–220. Reference Source [Google Scholar]
- 4. WHO: Investing to overcome the global impact of neglected tropical diseases: third WHO report on neglected diseases.2015. Reference Source [Google Scholar]
- 5. World Health Assembly: World Health Assembly Resolution WHA 66.12: Neglected tropical diseases. (World Health Assembly, Geneva, Switzerland).2013. Reference Source [Google Scholar]
- 6. WHO: The control of neglected zoonotic diseases: Community based interventions for NZDs prevention and control. Report of the third conference. (WHO Headquarters, Geneva, Switzerland). Accessed 14 March 2014,2011. Reference Source [Google Scholar]
- 7. Abela-Ridder B, Knopf L, Martin S, et al. : 2016: the beginning of the end of rabies? Lancet Glob Health. 2016;4(11):e780–e781. 10.1016/S2214-109X(16)30245-5 [DOI] [PubMed] [Google Scholar]
- 8. Freire de Carvalho M, Vigilato MAN, Pompei JA, et al. : Rabies in the Americas: 1998-2014. PLoS Negl Trop Dis. 2018;12(3):e0006271. 10.1371/journal.pntd.0006271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. WHO: Accelerating Work to Overcome the Global Impact of Neglected Tropical Diseases: A Roadmap for Implementation.2012. Reference Source [Google Scholar]
- 10. Ripani A, Mérot J, Bouguedour R, et al. : Review of rabies situation and control in the North African region with a focus on Tunisia. Rev Sci Tech. 2017;36(3):831–838. 10.20506/rst.36.3.2718 [DOI] [PubMed] [Google Scholar]
- 11. Neglected Tropical Disease Roadmap 2030 Disease Packs.(accessed 18 Feb 2020). Reference Source [Google Scholar]
- 12. WHO: Global Plan to Combat Neglected Tropical Diseases 2008–2015.2007. Reference Source [Google Scholar]
- 13. Okello A, Gibbs EP, Vandersmissen A, et al. : One Health and the neglected zoonoses: turning rhetoric into reality. Vet Rec. 2011;169(11):281–285. 10.1136/vr.d5378 [DOI] [PubMed] [Google Scholar]
- 14. WHO: The control of Neglected Zoonotic Diseases: A route to poverty alleviation. Report of a joint WHO/DFID-AHP Meeting.2006. Reference Source [Google Scholar]
- 15. Banyard AC, Horton DL, Freuling C, et al. : Control and prevention of canine rabies: the need for building laboratory-based surveillance capacity. Antiviral Res. 2013;98(3):357–364. 10.1016/j.antiviral.2013.04.004 [DOI] [PubMed] [Google Scholar]
- 16. Taylor LH, Hampson K, Fahrion A, et al. : Difficulties in estimating the human burden of canine rabies. Acta Trop. 2017;165:133–140. 10.1016/j.actatropica.2015.12.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Cleaveland S, Fèvre EM, Kaare M, et al. : Estimating human rabies mortality in the United Republic of Tanzania from dog bite injuries. Bull World Health Organ. 2002;80(4):304–310. [PMC free article] [PubMed] [Google Scholar]
- 18. Knobel DL, Cleaveland S, Coleman PG, et al. : Re-evaluating the burden of rabies in Africa and Asia. Bull World Health Organ. 2005;83(5):360–368. [PMC free article] [PubMed] [Google Scholar]
- 19. Shwiff S, Hampson K, Anderson A: Potential economic benefits of eliminating canine rabies. Antiviral Res. 2013;98(2):352–356. 10.1016/j.antiviral.2013.03.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. O'Brien KL, Nolan T, SAGE WG on Rabies: The WHO position on rabies immunization - 2018 updates. Vaccine. 2019;37 Suppl 1:A85–A87. 10.1016/j.vaccine.2018.10.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Hampson K, Abela-Ridder B, Bharti O, et al. : Modelling to inform prophylaxis regimens to prevent human rabies. Vaccine. 2019;37 Suppl 1:A166–A173. 10.1016/j.vaccine.2018.11.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. WHO Rabies Modelling Consortium: The potential effect of improved provision of rabies post-exposure prophylaxis in Gavi-eligible countries: a modelling study. Lancet Infect Dis. 2019;19(1):102–111. 10.1016/S1473-3099(18)30512-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Wentworth D, Hampson K, Thumbi SM, et al. : A social justice perspective on access to human rabies vaccines. Vaccine. 2019;37 Suppl 1:A3–A5. 10.1016/j.vaccine.2019.01.065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Fitzpatrick MC, Hampson K, Cleaveland S, et al. : Cost-effectiveness of canine vaccination to prevent human rabies in rural Tanzania. Ann Intern Med. 2014;160(2):91–100. 10.7326/M13-0542 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Mindekem R, Lechenne MS, Naissengar KS, et al. : Cost Description and Comparative Cost Efficiency of Post-Exposure Prophylaxis and Canine Mass Vaccination against Rabies in N'Djamena, Chad. Front Vet Sci. 2017;4:38. 10.3389/fvets.2017.00038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Davlin SL, Vonville HM: Canine rabies vaccination and domestic dog population characteristics in the developing world: a systematic review. Vaccine. 2012;30(24):3492–3502. 10.1016/j.vaccine.2012.03.069 [DOI] [PubMed] [Google Scholar]
- 27. Arief RA, Hampson K, Jatikusumah A, et al. : Determinants of Vaccination Coverage and Consequences for Rabies Control in Bali, Indonesia. Front Vet Sci. 2017;3:123. 10.3389/fvets.2016.00123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Kaare M, Lembo T, Hampson K, et al. : Rabies control in rural Africa: evaluating strategies for effective domestic dog vaccination. Vaccine. 2009;27(1):152–160. 10.1016/j.vaccine.2008.09.054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Gibson AD, Ohal P, Shervell K, et al. : Vaccinate-assess-move method of mass canine rabies vaccination utilising mobile technology data collection in Ranchi, India. BMC Infect Dis. 2015;15:589. 10.1186/s12879-015-1320-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Townsend SE, Sumantra IP, Pudjiatmoko, et al. : Designing programs for eliminating canine rabies from islands: Bali, Indonesia as a case study. PLoS Negl Trop Dis. 2013;7(8):e2372. 10.1371/journal.pntd.0002372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Ferguson EA, Hampson K, Cleaveland S, et al. : Heterogeneity in the spread and control of infectious disease: consequences for the elimination of canine rabies. Sci Rep. 2015;5:18232. 10.1038/srep18232 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Beyer HL, Hampson K, Lembo T, et al. : Metapopulation dynamics of rabies and the efficacy of vaccination. Proc Biol Sci. 2011;278(1715):2182–2190. 10.1098/rspb.2010.2312 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Bourhy H, Nakouné E, Hall M, et al. : Revealing the Micro-scale Signature of Endemic Zoonotic Disease Transmission in an African Urban Setting. Plos Pathog. 2016;12(4):e1005525. 10.1371/journal.ppat.1005525 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Zinsstag J, Lechenne M, Laager M, et al. : Vaccination of dogs in an African city interrupts rabies transmission and reduces human exposure. Sci Transl Med. 2017;9(421): pii: eaaf6984. 10.1126/scitranslmed.aaf6984 [DOI] [PubMed] [Google Scholar]
- 35. Tohma K, Saito M, Demetria CS, et al. : Molecular and mathematical modeling analyses of inter-island transmission of rabies into a previously rabies-free island in the Philippines. Infect Genet Evol. 2016;38:22–28. 10.1016/j.meegid.2015.12.001 [DOI] [PubMed] [Google Scholar]
- 36. WHO, FAO, OIE, et al.: Zero by 30: The global strategic plan to end human deaths from dog-mediated rabies by 2030.2018. Reference Source [Google Scholar]
- 37. Etheart MD, Kligerman M, Augustin PD, et al. : Effect of counselling on health-care-seeking behaviours and rabies vaccination adherence after dog bites in Haiti, 2014-15: a retrospective follow-up survey. Lancet Glob Health. 2017;5(10):e1017–e1025. 10.1016/S2214-109X(17)30321-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Undurraga EA, Meltzer MI, Tran CH, et al. : Cost-Effectiveness Evaluation of a Novel Integrated Bite Case Management Program for the Control of Human Rabies, Haiti 2014-2015. Am J Trop Med Hyg. 2017;96(6):1307–1317. 10.4269/ajtmh.16-0785 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Rysava K, Miranda ME, Zapatos R, et al. : On the path to rabies elimination: The need for risk assessments to improve administration of post-exposure prophylaxis. Vaccine. 2019;37 Suppl 1:A64–A72. 10.1016/j.vaccine.2018.11.066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Amparo ACB, Jayme S, Roces MCR, et al. : The evaluation of operating Animal Bite Treatment Centers in the Philippines from a health provider perspective. PLoS One. 2018;13(7):e0199186. 10.1371/journal.pone.0199186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Benavides JA, Megid J, Campos A, et al. : An evaluation of Brazil's surveillance and prophylaxis of canine rabies between 2008 and 2017. PLoS Negl Trop Dis. 2019;13(8):e0007564. 10.1371/journal.pntd.0007564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Del Rio Vilas VJ, Freire de Carvalho MJ, Vigilato MA, et al. : Tribulations of the Last Mile: Sides from a Regional Program. Front Vet Sci. 2017;4:4. 10.3389/fvets.2017.00004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Bourhy H, Troupin C, Faye O, et al. : Customized online and onsite training for rabies-control officers. Bull World Health Organ. 2015;93(7):503–506. 10.2471/BLT.14.149849 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. LeRoux K, Stewart D, Perrett KD, et al. : Rabies control in KwaZulu-Natal: South Africa. Bull World Health Organ. 2018;96(5):360–365. 10.2471/BLT.17.194886 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Gibson AD, Yale G, Vos A, et al. : Oral bait handout as a method to access roaming dogs for rabies vaccination in Goa, India: A proof of principle study. Vaccine X. 2019;1:100015. 10.1016/j.jvacx.2019.100015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Cliquet F, Guiot AL, Aubert M, et al. : Correction to: Oral vaccination of dogs: a well-studied and undervalued tool for achieving human and dog rabies elimination. Vet Res. 2018;49(1):84. 10.1186/s13567-018-0579-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Mpolya EA, Lembo T, Lushasi K, et al. : Toward Elimination of Dog-Mediated Human Rabies: Experiences from Implementing a Large-scale Demonstration Project in Southern Tanzania. Front Vet Sci. 2017;4:21. 10.3389/fvets.2017.00021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Hampson K, Abela-Ridder B, Brunker K, et al. : Surveillance to Establish Elimination of Transmission and Freedom from Dog-mediated Rabies. bioRxiv. 2016. 10.1101/096883 [DOI] [Google Scholar]
- 49. Vigilato MA, Clavijo A, Knobl T, et al. : Progress towards eliminating canine rabies: policies and perspectives from Latin America and the Caribbean. Philos Trans R Soc Lond B Biol Sci. 2013;368(1623):20120143. 10.1098/rstb.2012.0143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Hu RL, Fooks AR, Zhang SF, et al. : Inferior rabies vaccine quality and low immunization coverage in dogs ( Canis familiaris) in China. Epidemiol Infect. 2008;136(11):1556–1563. 10.1017/S0950268807000131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Suseno PP, Rysava K, Brum E, et al. : Lessons for rabies control and elimination programmes – a decade of experience from Bali, Indonesia. OIE Scientific and Technical Review.(in press). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Taylor E, Banyard AC, Bourhy H, et al. : Avoiding preventable deaths: The scourge of counterfeit rabies vaccines. Vaccine. 2019;37(17):2285–2287. 10.1016/j.vaccine.2019.03.037 [DOI] [PubMed] [Google Scholar]
- 53. WHO Expert Consultation on Rabies: Third report (WHO Technical Report Series, No. 1012). (World Health Organization, Geneva).2018. Reference Source [Google Scholar]
- 54. Wallace RM, Undurraga EA, Blanton JD, et al. : Elimination of Dog-Mediated Human Rabies Deaths by 2030: Needs Assessment and Alternatives for Progress Based on Dog Vaccination. Front Vet Sci. 2017;4:9. 10.3389/fvets.2017.00009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Durr S, Mindekem R, Kaninga Y, et al. : Effectiveness of dog rabies vaccination programmes: comparison of owner-charged and free vaccination campaigns. Epidemiol Infect. 2009;137(11):1558–1567. 10.1017/S0950268809002386 [DOI] [PubMed] [Google Scholar]
- 56. Wallace R, Etheart M, Ludder F, et al. : The Health Impact of Rabies in Haiti and Recent Developments on the Path Toward Elimination, 2010-2015. Am J Trop Med Hyg. 2017;97(4_Suppl):76–83. 10.4269/ajtmh.16-0647 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Townsend SE, Lembo T, Cleaveland S, et al. : Surveillance guidelines for disease elimination: a case study of canine rabies. Comp Immunol Microbiol Infect Dis. 2013;36(3):249–261. 10.1016/j.cimid.2012.10.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Gibson AD, Mazeri S, Lohr F, et al. : One million dog vaccinations recorded on mHealth innovation used to direct teams in numerous rabies control campaigns. PLoS One. 2018;13(7):e0200942. 10.1371/journal.pone.0200942 [DOI] [PMC free article] [PubMed] [Google Scholar]