Abstract
Consistent observations report increased severity of SARS-CoV-2 infection in overweight men with cardiovascular factors. As the visceral fat possesses an intense immune activity, is involved in metabolic syndrome and is at the crossroad between the intestines, the systemic circulation and the lung, we hypothesized that it plays a major role in severe forms of SARS-CoV-2 infection. SARS-CoV2 presents the ability to infect epithelial cells of the respiratory tract as well as the intestinal tract. Several factors may increase intestinal permeability including direct enterocyte damage by SARS-CoV2, systemic inflammatory response syndrome (SIRS) and epithelial ischemia secondary to SARS-CoV2- associated endothelial dysfunction. This increase permeability further leads to translocation of microbial components such as MAMPs (microbial-associated molecular pattern), triggering an inflammatory immune response by TLR-expressing cells of the mesentery fat (mostly macrophages and adipocytes). The pro-inflammatory cytokines produced by the mesentery fat mediates systemic inflammation and aggravate acute respiratory distress syndrome (ARDS) through the mesenteric lymph drainage.
Metabolic syndrome is associated with pejorative outcomes of COVID-19
The recent outbreak of the SARS-CoV-2 has led to a worldwide pandemic responsible for a major health crisis. Coronavirus-induced disease 19 (COVID-19) can evolve to severe forms characterized by hyperinflammation, acute respiratory distress syndrome (ARDS) and death, which are more frequently observed in subsets of patients with comorbidities. Observational studies in Chinese, European and US cohorts consistently highlighted a significant association between intensive care unit (ICU) admission and male gender, as well as hypertension, diabetes and cardiovascular diseases, as compared to non-ICU patients [1], [2]. In a cohort of 138 patients hospitalized in Wuhan, males accounted for 61% of ICU patients (n = 36) as compared to 52% (n = 102) in conventional units; 58%, 25% and 22% of ICU patients had hypertension, diabetes and cardiovascular history, respectively, as compared to 22%, 11% and 6% in non-ICU patients (all p values below 0.05) [2]. Higher risk of death was further reported for males with cardiovascular comorbidities within COVID-19 ICU patients [3], [4]. The average body mass index (BMI) was 33.2 kg.m−2 in a cohort of 24 ICU patients from the Seattle area (WA), thus suggesting obesity as an additional risk factor for severe COVID-19. In a French cohort of 124 ICU patients, 75.8% patients had a BMI >30 kg.m−2. BMI and male sex were independently associated with invasive mechanical ventilation. The odds ratio for orotracheal intubation and mechanical ventilation was 7.36 (1.63–33.14; p = 0.02) for patients with BMI >35 kg.m−2 vs. patients <25 kg.m−2 [5]. Similar results were obtained in a large cohort from New York City (NY), in which obesity was an independent risk-factor for hospitalization in patients under 60 years old who were admitted to the emergency department [6]. In an Italian cohort of more than 1000 ICU patients hospitalized in Lombardia, 83% of patients were males; 50% had high blood pressure and about 20% had type 2 diabetes, hypercholesterolemia and/or a cardiovascular disease [7]. Overall, these studies indicate that overweight males with cardiovascular risk factors are at significantly higher risk of developing severe forms of COVID-19.
As the increased volume of mesentery fat in overweight men play a key role in the occurrence of metabolic syndrome [8], we hypothesized that the visceral adipose tissue plays a central role in severe forms of COVID-19.
COVID-19: a 2-step disease leading to a cytokinic storm
The initial phase of the disease is considered as the viral phase. At that phase, COVID-19 behaves as a classical lymphocytic pneumonia, evidenced by CT-scan and/or lung biopsies [9], [10]. At a later phase of the disease, lung tissue lesions are unusual, presenting notably with major intra-alveolar fibrin deposition and intraluminal loose connective tissue in alveolar ducts and bronchioles. Additionally, vascular damage is significant with cytoplasmic vacuolization of the endothelium and cell detachment in small to medium-sized pulmonary arteries [10].
At this later phase, along with endothelial damage, clinical and biological characteristics are suggestive of a “cytokinic storm”. In critically ill patients with COVID-19 an intense inflammatory response during a secondary respiratory and systemic worsening at day 7 to day 15 from the onset of initial symptoms has been widely described [7], [11], [12].
Myeloid cells-associated cytokines are dramatically increased in severe COVID-19
Hadjadj et al. performed an immunological extensive assessment (immune blood cell phenotyping, whole blood transcriptomic and cytokine quantification) in patients with mild form and severe form of COVID-19 at day 8 to day 12 after onset of symptoms [13]. Interestingly, they found that type 1 interferon response was profoundly impaired. In addition, innate immunity-associated cytokinic pathways were markedly activated. Specifically, they found a strong increase in expression of IL-6-induced genes, such as IL6R, SOCS3 and STAT3 as well as an up-regulation of TNF pathway-related genes, including TNFSF10. Another key finding is the up-regulation of the CCL2-CCR2 pathway suggesting a key role of monocytes and macrophages in the lung and in the intense systemic inflammation state [13].
Other studies described increased plasma concentrations of a series of inflammatory cytokines, such as granulocyte-macrophage colony-stimulating factor (GM-CSF), IL-6 [14], [15], TNF-α, and granulocyte colony-stimulating factor (G-CSF), in severe COVID-19. Further investigations demonstrated that peripheral inflammatory monocytes and T cells through their production of GM-CSF may incite cytokine storm in severe COVID-19 patients [15], [16].
This excess of inflammatory response is a critical cause of mortality from COVID-19. A study of 548 patients showed that, high cytokine levels (IL-2R, IL-6, IL-10, and TNF-α), and high LDH level were significantly associated with severe forms of COVID-19 [17].
Accordingly, anti-IL-6R therapy is being investigated to treat severely ill patients [18], [19] with preliminary results reporting efficacy in a French multicenter randomized clinical trial. Other anti-cytokines therapies are also under investigation.
SARS-CoV-2 binds ACE2 which is vastly expressed in cardiopulmonary tissues, including endothelial cells. Some hematopoietic cells also express it, and particularly monocytes and macrophages [20].
From studies focusing on other coronaviruses, it is known that the infection of cells of the myeloid lineage (monocytes, macrophages and dendritic cells) results in their activation and production of inflammatory cytokines including IL-6 [20].
IL-6 can signal through 2 main pathways, the trans- and the cis- signaling pathways. The cis- pathway involves gp130 and mIL-6R (membrane-bound IL-6R) mainly restricted to immune cells and results in a pleiotropic effect on the cells of the adaptative and innate immunity.
Trans signaling only involves the soluble form of IL-6R (sIL-6R) forming a complex with gp130 which is virtually express on all hematopoietic and non-hematopoietic cell types. Therefore, it activates cells that do not express mIL-6R like endothelial cells, resulting in a cytokinic storm [20].
Overall, the day 7 to day 12 clinical worsening is a typical feature of the course of COVID-19 and involves a major release of cytokines and particularly IL-6 and TNF-α with a key role of myeloid cells. This cytokine release syndrome highly correlates with severity and mortality.
Additionally, A key feature of COVD-19 course, is lymphopenia – a hallmark of severe forms -, suggesting that most of the cytokines released during the cytokinic storm are produced by myeloid cells or non-hematopoietic cells such as adipocytes.
The mesentery fat is a site able to promote the inflammatory response
Visceral adipose tissue is more developed in men, as it represents 10 to 20% of total fat as compared to 5–10% in women [21]. Visceral fat possesses multiple properties that make it a key site for intestinal and systemic immune response. In comparison with subcutaneous adipose tissue, visceral adipose tissue including mesenteric fat has a higher vascularization and contains greater number of immune cells. It also expresses higher level of pro-inflammatory cytokines such as IL-6, TNF-α and IL-8 [21]. Visceral adipose tissue is enriched in macrophages that are skewed toward an M2 phenotype (the pro-inflammatory subtype) [22]. Importantly, in case of visceral obesity, the pro-inflammatory state of the visceral fat is further enhanced [21].
Macrophages of the mesentery fat from patients with nonalcoholic steatohepatitis (NASH) present elevated levels of cytokines and chemokines compared with healthy subjects suggesting, that mesenteric inflammation may uncouples and mediates inflammation through the portal flow [23].
In addition to the immune response provided by mesentery fat-associated macrophages, adipocytes and stromal cells may also play their own role as immune enhancers [24]. For example, adipokines produced by adipocytes, can regulate metabolic homeostasis and affect immune functions [25].
In Crohn’s disease, it has been shown that mesenteric fat could be a major source of C-reactive protein as well as IL-6. It is also enriched in microbial particles of the microbiota as an evidence of enhanced intestinal microbial translocation [26]. Remarkably, antiTNF-α and antiIL-6 therapies have been shown to reduce intestinal inflammation during Crohn’s disease [27], [28].
Therefore, the visceral adipose tissue is a site with an intense immune activity that is able to produce high level of innate immunity-associated cytokines such as IL-6 and TNF-α. It has –pro-inflammatory properties compared with subcutaneous adipose tissue. A systemic pro-inflammatory potential is further enhanced in case of android obesity. Whether mesenteric fat can be responsible for an intense cytokine production raises the question of the role of the gut in this mesenteric activation.
SARS-CoV-2 intestinal symptomatic infection associates with systemic hyperinflammation
Digestive symptoms are not usually well reported and are probably under-evaluated in patients affected by COVID-19. Despite the fact that they are underreported, several cohorts found a high prevalence of gastrointestinal symptoms including nausea, vomiting, loss of appetite and diarrhea. A meta-analysis performed from 60 studies and 4243 patients, showed a pooled prevalence of diarrhea of 12.5% (95% CI: 9.6–16.0) [29]. Patients with severe forms of COVID-19 had a prevalence of digestive manifestations of 17.6% versus 11% for patients with non-severe forms [29]. In UK, 29% of patients tested positive for SARS-CoV-2 and using RADAR COVID-19, a smartphone application that was launched among the UK general population asking about COVID-19 symptoms, had diarrhea. Digestive symptoms were strong predictors of having a positive PCR test [30].
In Cheung and al. meta-analysis, fecal shedding of SARS-CoV-2 RNA was found in about half of the patients. In the Hong-Kong cohort of 59 patients, viral RNA was more frequently found in the stool of patients with diarrhea as compared with patients without diarrhea (38.5% vs 8.7%; p = 0.019) [29].
It has been demonstrated that SARS-CoV-2 is capable of binding to the angiotensin-converting enzyme 2 (ACE2) which is express in many cell types including the epithelial cells of the respiratory and the gastrointestinal tract [31], [32].
An Austrian team reported that patient with COVD-19-associated diarrhea had an elevated fecal calprotectin with a mean of 123 µg/g in patients with active diarrhea. The fecal calprotectin could reach values above 200 µg/g in some patients without any preexisting digestive condition [33]. Interestingly, the level of fecal calprotectin did not correlate with the level of viral RNA in the stool but positively correlated with the level of IL-6 in plasma [33].
Stanifer et al. provided direct evidence that human intestinal cells could be infected by SARS-CoV-2. Moreover, they showed that the virus can replicate in intestinal epithelial cell lines as well as in human colon organoid models, therefore participating in the spread and enhanced viremia of SARS-CoV-2 [34], [35].
Overall, SARS-CoV-2 has an intestinal tropism and may induce acute intestinal inflammation which associates with systemic inflammation rather than with intestinal viral load.
Mucosal injury leads to alteration in the intestinal barrier and increases intestinal permeability. The ultimate consequence is an increase translocation of the intestinal luminal content (including bacteria of the microbiota) to the mesentery fat and the systemic blood circulation. Therefore, the ability of SARS-CoV2 to directly bind and invade the intestinal epithelium and to induce intestinal mucosa inflammation could lead to enhance mesentery antigenic stimulation.
This inflammation of the intestinal tract and subsequently of the mesentery fat could be aggravated by other COVID-19-associated features such as blood coagulopathy and microthrombotic disorders [36].
Another key digestive characteristic of COVD-19 is the high prevalence of hepatic cytolysis that is associated with severe forms of the disease [37]. While the underlying mechanism is not well understood, multiple factors are likely involved. Increased intestinal translocation to the portal circulation may be one of the participating factors of liver damage.
Overall, the gastrointestinal tract is a key organ with probable direct viral invasion evidenced by fecal shedding of SARS-CoV-2 RNA and prevalent digestive symptoms during COVID-19 course, which are associated with a pejorative outcome
Inflammatory mediators from the gut are transmitted to the lung
Patients with systemic inflammatory response syndrome (SIRS) have increase intestinal permeability even if they don’t have any gastrointestinal condition [38]. Therefore, there is an increase bacterial translocation of either live bacteria, or MAMPs (microbial-associated molecular patterns) that will trigger the innate immunity through toll-like receptors. Increased intestinal permeability was found in ICU patients by measuring urinary excretion of orally administered lactulose or mannitol [39]. Interestingly, increased intestinal permeability was associated with clinical worsening including multiorgan failure in critically ill patients.
Portal venous and the lymph flows are the two main routes that could explain how this modified intestinal permeability as well as the bacterial translocation impact other organs such as the lungs. In a study published in 1991, Moore and al. performed a venous catheterism in the portal vein of critically ill patients to do sequential blood cultures. While 60% of patients were in shock and 30% developed multiorgan failure, only 2% of portal venous blood cultures were positive suggesting that the portal circulation is not the main way for intestinal bacterial translocation [40]. An alternative route of translocation could be the intestinal lymphatics. Intestinal lymph is drained from the mesenteric lymphatics to the thoracic duct, subsequently merging with the blood circulation in the left subclavian vein. Therefore, products drained by the intestinal lymph are first in contact with the pulmonary arterial vasculature.
In that regard, it is relevant to note that critically ill patients often develop ARDS even without any primary respiratory condition or infections such as patients with severe pancreatitis or trauma developing ARDS. Interestingly, experiments performed in a hemorrhagic shock model in rats showed that lymph collected in the mesenteric lymphatics but not blood from the portal vein could have negative effect in vitro on the endothelium permeability as well as on neutrophils activation and several other relevant cell types. Reinjection of lymph but not portal blood from shocked rat to other rats could worsen lung failure in vivo. Strikingly, the ligation of the major intestinal lymph duct could prevent early lung damage [41], [42], [43].
Therefore, multiple experimental in vivo and ex vivo data both in human and animal models support a gut lymph-lung axis. SARS-CoV-2 that has a direct effect on the intestine and that triggers a SIRS may then critically increase intestinal permeability leading to the recirculation of pro-inflammatory mediators to the circulation and primarily to the lungs.
Additionally, extensively reported SARS-CoV-2-associated vascular injury and coagulopathy [10], [36] may lead to ischemia of the intestinal epithelium, further favoring microbial translocation.
Conclusion and working hypothesis
Male sex, advanced age and metabolic syndrome are factors strongly associated with the severity of COVID-19. They are associated with a high mortality rate. The daily expanding literature shows overwhelming data on the secondary worsening associated with high systemic inflammation including TNF-α and IL-6 as two of the key cytokines. Moreover, it is more and more evident that the intestine is targeted by the SARS-CoV-2 and that the intestinal phase may be critical in severe forms of COVID-19.
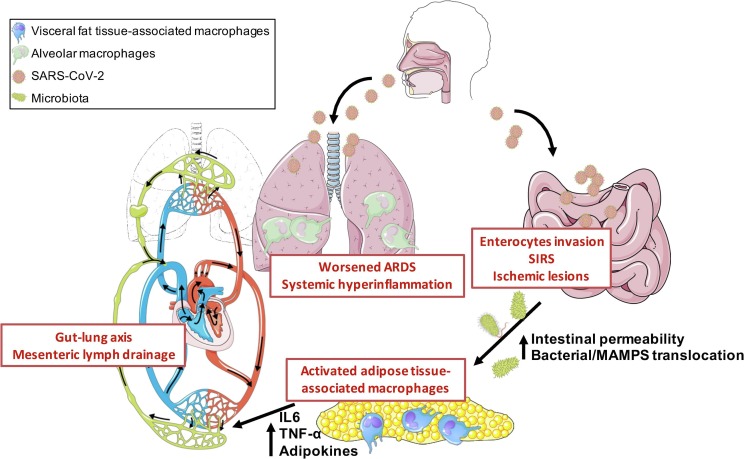
Therefore, we propose a pathophysiogeny model of severe forms (Fig. 1 ) where the visceral fat, which is already in a pro-inflammatory state in patients with dysmetabolic syndrome, acts as an enhancer of inflammation, being an uncoupling link between the intestine and systemic and pulmonary inflammation through the mesenteric lymphatics. A better understanding of the way that the mesentery fat aggravates the course of COVID-19 may open new and innovative therapeutic avenues.
Fig. 1.
The gut lymph-lung axis promotes hyperinflammation and aggravates pulmonary lesions during SARS-CoV2 infection. SARS-CoV2 presents the ability to infect epithelial cells of the respiratory tract as well as the intestinal tract. Several factors may increase intestinal permeability including, direct enterocyte damage by SARS-CoV2, systemic inflammatory response syndrome (SIRS) and epithelial ischemia secondary to SARS-CoV2-associated endothelial dysfunction. This increase permeability further leads to translocation of microbial components such as MAMPs (microbial-associated molecular pattern), triggering an inflammatory immune response by TLR-expressing cells of the mesentery fat (mostly macrophages and adipocytes). The pro-inflammatory cytokines produced by the mesentery fat mediates systemic inflammation and aggravate acute respiratory distress syndrome (ARDS) through the mesenteric lymph drainage.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Guan W., Ni Z., Hu Y. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wang D., Hu B., Hu C. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323:1061. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chen T., Wu D., Chen H. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. BMJ. 2020:m1091. doi: 10.1136/bmj.m1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wu Z., McGoogan J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China. JAMA. 2020;323:1239. doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 5.Simonnet A., Chetboun M., Poissy J. High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity (Silver Spring) 2020 doi: 10.1002/oby.22831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lighter J., Phillips M., Hochman S. Obesity in patients younger than 60 years is a risk factor for Covid-19 hospital admission. Clin Infect Dis. 2020 doi: 10.1093/cid/ciaa415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Grasselli G., Zangrillo A., Zanella A. Baseline characteristics and outcomes of 1591 patients infected with SARS-CoV-2 admitted to ICUs of the Lombardy Region, Italy. JAMA. 2020;323:1574. doi: 10.1001/jama.2020.5394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Liu K.H., Chan Y.L., Chan W.B., Chan J.C.N., Chu C.W.W. Mesenteric fat thickness is an independent determinant of metabolic syndrome and identifies subjects with increased carotid intima-media thickness. Diabetes Care. 2006;29:379–384. doi: 10.2337/diacare.29.02.06.dc05-1578. [DOI] [PubMed] [Google Scholar]
- 9.Gattinoni L., Chiumello D., Caironi P. COVID-19 pneumonia: different respiratory treatments for different phenotypes? Intensive Care Med. 2020 doi: 10.1007/s00134-020-06033-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Copin M.-C., Parmentier E., Duburcq T., Poissy J., Mathieu D. Time to consider histologic pattern of lung injury to treat critically ill patients with COVID-19 infection. Intensive Care Med. 2020 doi: 10.1007/s00134-020-06057-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Li Q., Guan X., Wu P. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N Engl J Med. 2020;382:1199–1207. doi: 10.1056/NEJMoa2001316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yang X., Yu Y., Xu J. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020;8:475–481. doi: 10.1016/S2213-2600(20)30079-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hadjadj J, Yatim N, Barnabei L, et al. Impaired type I interferon activity and exacerbated inflammatory responses in severe Covid-19 patients. medRxiv 2020; 2020.04.19.20068015. [DOI] [PMC free article] [PubMed]
- 14.Merad M., Martin J.C. Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nat Rev Immunol. 2020 doi: 10.1038/s41577-020-0331-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhou Y., Fu B., Zheng X. Pathogenic T cells and inflammatory monocytes incite inflammatory storm in severe COVID-19 patients. Natl Sci Rev. 2020 doi: 10.1093/nsr/nwaa041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Li G., Fan Y., Lai Y. Coronavirus infections and immune responses. J Med Virol. 2020;92:424–432. doi: 10.1002/jmv.25685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Li X., Xu S., Yu M. Risk factors for severity and mortality in adult COVID-19 inpatients in Wuhan. J Allergy Clin Immunol. 2020 doi: 10.1016/j.jaci.2020.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Toniati P., Piva S., Cattalini M. Tocilizumab for the treatment of severe COVID-19 pneumonia with hyperinflammatory syndrome and acute respiratory failure: A single center study of 100 patients in Brescia, Italy. Autoimmun Rev. 2020:102568. doi: 10.1016/j.autrev.2020.102568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Klopfenstein T., Zayet S., Lohse A. Tocilizumab therapy reduced intensive care unit admissions and/or mortality in COVID-19 patients. Médecine Mal Infect. 2020 doi: 10.1016/j.medmal.2020.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Moore JB, June CH. Cytokine release syndrome in severe COVID-19. Science (80-) 2020; 368:473–474. [DOI] [PubMed]
- 21.Kredel L.I., Siegmund B. Adipose-tissue and intestinal inflammation – visceral obesity and creeping fat. Front Immunol. 2014:5. doi: 10.3389/fimmu.2014.00462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zeyda M., Stulnig T.M. Adipose tissue macrophages. Immunol Lett. 2007;112:61–67. doi: 10.1016/j.imlet.2007.07.003. [DOI] [PubMed] [Google Scholar]
- 23.du Plessis J., van Pelt J., Korf H. Association of adipose tissue inflammation with histologic severity of nonalcoholic fatty liver disease. Gastroenterology. 2015;149 doi: 10.1053/j.gastro.2015.05.044. 635-648.e14. [DOI] [PubMed] [Google Scholar]
- 24.Permana P.A., Menge C., Reaven P.D. Macrophage-secreted factors induce adipocyte inflammation and insulin resistance. Biochem Biophys Res Commun. 2006;341:507–514. doi: 10.1016/j.bbrc.2006.01.012. [DOI] [PubMed] [Google Scholar]
- 25.Weidinger C., Ziegler J.F., Letizia M., Schmidt F., Siegmund B. Adipokines and their role in intestinal inflammation. Front Immunol. 2018:9. doi: 10.3389/fimmu.2018.01974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Peyrin-Biroulet L., Gonzalez F., Dubuquoy L. Mesenteric fat as a source of C reactive protein and as a target for bacterial translocation in Crohn’s disease. Gut. 2012;61:78–85. doi: 10.1136/gutjnl-2011-300370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Present D.H., Rutgeerts P., Targan S. Infliximab for the Treatment of Fistulas in Patients with Crohn’s Disease. N Engl J Med. 1999;340:1398–1405. doi: 10.1056/NEJM199905063401804. [DOI] [PubMed] [Google Scholar]
- 28.Danese S., Vermeire S., Hellstern P. Randomised trial and open-label extension study of an anti-interleukin-6 antibody in Crohn’s disease (ANDANTE I and II) Gut. 2019;68:40–48. doi: 10.1136/gutjnl-2017-314562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cheung K.S., Hung I.F., Chan P.P. Gastrointestinal Manifestations of SARS-CoV-2 Infection and Virus Load in Fecal Samples from the Hong Kong Cohort and Systematic Review and Meta-analysis. Gastroenterology. 2020 doi: 10.1053/j.gastro.2020.03.065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Menni C, Valdes AM, Freidin MB, et al. Real-time tracking of self-reported symptoms to predict potential COVID-19. Nat Med 2020; 1–4. [DOI] [PMC free article] [PubMed]
- 31.Liang W., Feng Z., Rao S. Diarrhoea may be underestimated: a missing link in 2019 novel coronavirus. Gut. 2020;69:1141–1143. doi: 10.1136/gutjnl-2020-320832. [DOI] [PubMed] [Google Scholar]
- 32.Wan Y., Shang J., Graham R., Baric R.S., Li F. Receptor Recognition by the Novel Coronavirus from Wuhan: an Analysis Based on Decade-Long Structural Studies of SARS Coronavirus. J Virol. 2020;94 doi: 10.1128/JVI.00127-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Effenberger M, Grabherr F, Mayr L, et al. Faecal calprotectin indicates intestinal inflammation in COVID-19. Gut 2020; gutjnl-2020-321388. [DOI] [PMC free article] [PubMed]
- 34.Stanifer ML, Kee C, Cortese M, et al. Critical role of type III interferon in controlling SARS-CoV-2 infection, replication and spread in primary human intestinal epithelial cells. bioRxiv 2020; 2020.04.24.059667. [DOI] [PMC free article] [PubMed]
- 35.Lamers MM, Beumer J, van der Vaart J, et al. SARS-CoV-2 productively infects human gut enterocytes. Science (80-) 2020; eabc1669. [DOI] [PMC free article] [PubMed]
- 36.Zhang Y., Xiao M., Zhang S. Coagulopathy and antiphospholipid antibodies in patients with Covid-19. N Engl J Med. 2020;382 doi: 10.1056/NEJMc2007575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zhang C., Shi L., Wang F.-S. Liver injury in COVID-19: management and challenges. Lancet Gastroenterol Hepatol. 2020;5:428–430. doi: 10.1016/S2468-1253(20)30057-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Assimakopoulos S.F., Triantos C., Thomopoulos K. Gut-origin sepsis in the critically ill patient: pathophysiology and treatment. Infection. 2018;46:751–760. doi: 10.1007/s15010-018-1178-5. [DOI] [PubMed] [Google Scholar]
- 39.Doig C.J., Sutherland L.R., Sandham J.D., Fick G.H., Verhoef M., Meddings J.B. Increased intestinal permeability is associated with the development of multiple organ dysfunction syndrome in critically ill ICU patients. Am J Respir Crit Care Med. 1998;158:444–451. doi: 10.1164/ajrccm.158.2.9710092. [DOI] [PubMed] [Google Scholar]
- 40.Moore F.A., Moore E.E., Poggetti R. Gut bacterial translocation via the portal vein: a clinical perspective with major torso trauma. J Trauma - Inj Infect Crit Care. 1991;31:629–638. doi: 10.1097/00005373-199105000-00006. [DOI] [PubMed] [Google Scholar]
- 41.Magnotti L.J., Upperman J.S., Xu D.Z., Lu Q., Deitch E.A. Gut-derived mesenteric lymph but not portal blood increases endothelial cell permeability and promotes lung injury after hemorrhagic shock. Ann Surg. 1998:518–527. doi: 10.1097/00000658-199810000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Upperman J.S., Deitch E.A., Guo W., Lu Q., Xu D. Post-hemorrhagic shock mesenteric lymph is cytotoxic to endothelial cells and activates neutrophils. Shock. 1998;10:407–414. doi: 10.1097/00024382-199812000-00005. [DOI] [PubMed] [Google Scholar]
- 43.Deitch E.A., Xu D.Z., Kaiser V.L. Role of the gut in the development of injury- and shock induced SIRS and MODS: The gut-lymph hypothesis, a review. Front Biosci. 2006;11:520–528. doi: 10.2741/1816. [DOI] [PubMed] [Google Scholar]