As the coronavirus 2019 (COVID-19) continues to spread globally, hundreds of thousands have been infected, among whom approximately 15% of COVID-19 patients develop severe disease, and 5 to 6% are critically ill [1]. Critical patients of COVID-19 have a dramatically higher case fatality rate than severe cases. Thus, it is increasingly urgent to develop early and effective predictors to distinguish critical patients from severe patients. Storms of inflammatory cytokines and blood clots were reported to associate with severe disease and fatality of COVID-19 patients [2, 3]. We aimed to identify a biomarker for the detection of COVID-19 progression from numerous cytokines and coagulation indicators.
We conducted a retrospective study based on patients with a laboratory-confirmed diagnosis of COVID-19 admitted to the intensive care unit in Beijing Ditan Hospital from January 20, 2020, to March 23, 2020. This study was approved by the Ethics Committee of Beijing Ditan Hospital. The severity of COVID-19 was defined according to the guidelines on the diagnosis and treatment of new coronavirus pneumonia (version 7). All baseline medical record information including demographic, data, complications, and laboratory results were obtained within the first day after hospital admission. Blood samples were collected at baseline and once every 3–7 days during hospitalization. Forty-five cytokines/chemokines/growth factors in serum were measured using Luminex multiplex assay. Random forests machine learning classifier in Python environment was used for variable importance of the feature rankings. A receiver operating characteristic (ROC) curve was generated to evaluate the diagnostic accuracy of a protein.
A total of 24 COVID-19 patients were enrolled in this study, including 14 (58.3%) severe patients and 10 (41.7%) critical patients (Table 1). Compared to the severe group, critical cases were of significantly older ages and showed higher white blood cell counts and neutrophil counts. Levels of VEGF-D, TNF-α, SCF, LIF, IL-2, IL-4, IL-6, IL-8, IL-10, IL-15, IL-17A, IL-18, IL-1β, and IFN-γ were significantly higher in the critical group than in the severe group (Table 1). Additionally, lymphocyte count, CRP, LDH, and coagulation indicators (d-dimer, platelet count, PT, and APTT), which were reported to associate with clinical outcome [4, 5], were also included in the random forests model.
Table 1.
Demographics, baseline characteristics, cytokines, chemokines, and growth factors of COVID-19 patients
| Characteristics | Total (n = 24) | Severe patients (n = 14) | Critical patients (n = 10) | P values |
|---|---|---|---|---|
| Age mean range, years | 68 (36, 88) | 65 (36, 81) | 77 (64, 88) | .003 |
| Gender | .521 | |||
| Male, n (%) | 15 (62.5) | 10 (71.4) | 5 (50) | |
| Female, n (%) | 9 (37.5) | 4 (28.6) | 5 (50) | |
| Admission to ICU, mean (SD), days | 22 (22) | 13 (8) | 35 (28) | .009 |
| SOFA score, mean (SD) | 3.7 (2.4) | 2.8 (1.7) | 4.9 (2.8) | .015 |
| Complications, n (%) | .568 | |||
| Hypertension | 10 (41.7) | 6 (42.9) | 4 (40.0) | 1 |
| Cardiovascular disease | 4 (16.7) | 1 (7.1) | 3 (30.0) | .355 |
| Chronic Pulmonary disease | 6 (25) | 1 (7.1) | 5 (50.0) | .056 |
| Diabetes | 6 (25) | 2 (14.3) | 4 (40.0) | .339 |
| Hyperlipemia | 0 | 0 | 0 | |
| Chronic kidney disease | 3 (12.5) | 2 (14.3) | 1 (10.0) | 1 |
| Immune disorders | 3 (12.5) | 3 (21.4) | 0 | .348 |
| Others | 1 (4.2) | 1 (7.1) | 0 | 1 |
| Laboratory data, mean (SD) | ||||
| WBC, 109/L | 7.72 (5.12) | 5.82 (2.13) | 10.20 (6.8) | .039 |
| Lymphocyte, 109/L | 1.08 (0.47) | 0.93 (0.45) | 1.27 (0.44) | .089 |
| Neutrophil, 109/L | 6.34 (4.91) | 4.59 (1.74) | 8.63 (6.7) | .048 |
| Platelets, 109/L | 211 (98) | 206 (85) | 218 (117) | .785 |
| PT, s | 13.1 (1.7) | 13.5 (1.9) | 12.7 (1.4) | .291 |
| APTT, s | 33.8 (8.6) | 33.9 (7.2) | 33.7 (10.4) | .973 |
| d-dimer, mg/L | 4.9 (7.6) | 2.9 (5.2) | 7.1 (9.3) | .213 |
| CRP, mg/L | 69.4 (66.1) | 55.3 (32.7) | 85.1 (89.6) | .315 |
| LDH, U/L | 403.6 (129.6) | 398.3 (66.1) | 406.3 (155.5) | .916 |
| Serum creatinine, μmol/L | 108.1 (172.4) | 67.9 (12.9) | 148.1 (242.9) | .331 |
| ALT, U/L | 40.5 (13.9) | 44.8 (23.2) | 41.9 (31.5) | .809 |
| Blood potassium, mmol/L | 4.0 (0.5) | 3.9 (0.4) | 4.2 (0.5) | .299 |
| Blood sodium, mmol/L | 137.4 (5.9) | 137.1 (8.2) | 137.8 (2.7) | .772 |
| Cytokines, chemokines, and growth factors, median (IQR), pg/mL | ||||
| VEGF-D | 40.1 (17.7, 64.8) | 25.9 (12.3, 40.6) | 62.9 (45.8, 79.6) | .0048 |
| TNF-α | 25.3 (3.2, 67.9) | 8.6 (0, 48.4) | 54.8 (15.3, 131.0) | .027 |
| SCF | 17.1 (9.2, 20.7) | 13.9 (7.7, 18.4) | 20.1 (16.2, 68.3) | .019 |
| LIF | 18.4 (4.2, 64.9) | 7.4 (1.9, 21.9) | 56.5 (10.8, 96.9) | .0089 |
| IL-2 | 35.2 (8.7, 59.1) | 17.5 (4.7, 43.5) | 55.2 (23.5, 90.2) | .018 |
| IL-4 | 2.1 (0, 20.2) | 0 (0, 9.8) | 143.7 (47.2, 203.9) | .033 |
| IL-6 | 54.2 (26.7, 157.8) | 35.4 (19.5, 76.9) | 143.7 (47.2, 203.9) | .019 |
| IL-8 | 20.1 (0.1, 44.2) | 2.6 (0, 13.0) | 13.0 (5.4, 17.8) | .039 |
| IL-10 | 6.0 (1.4, 15.3) | 3.9 (0.9, 6.2) | 8.4 (3.3, 24.1) | .038 |
| IL-15 | 20.1 (4.8, 44.2) | 10.7 (1.8, 26.3) | 38.7 (18.1, 83.3) | .018 |
| IL-17A | 19.8 (0.7, 55.3) | 9.5 (0, 26.2) | 50.0 (16.4, 109.4) | .021 |
| IL-18 | 86.0 (19.8, 185.6) | 29.2 (18.5, 109.1) | 158.9 (91.8, 209.2) | .046 |
| IL-1β | 8.8 (2.1, 25.2) | 4.4 (1.6, 15.8) | 22.8 (7.8, 52.7) | .022 |
| IFN-γ | 17.6 (6.2, 29.9) | 9.1 (3.6, 24.2) | 26.4 (12.9, 53.2) | .013 |
WBC white blood cells, CRP C-reactive protein, LDH lactate dehydrogenase, PT prothrombin time, APTT activated partial thromboplastin time, ALT alanine aminotransferase, VEGF vascular endothelial growth factor; TNF-α tumor necrosis factor-alpha, SCF stem cell factor; LIF leukemia inhibitory factor, IL interleukin, IFN interferon
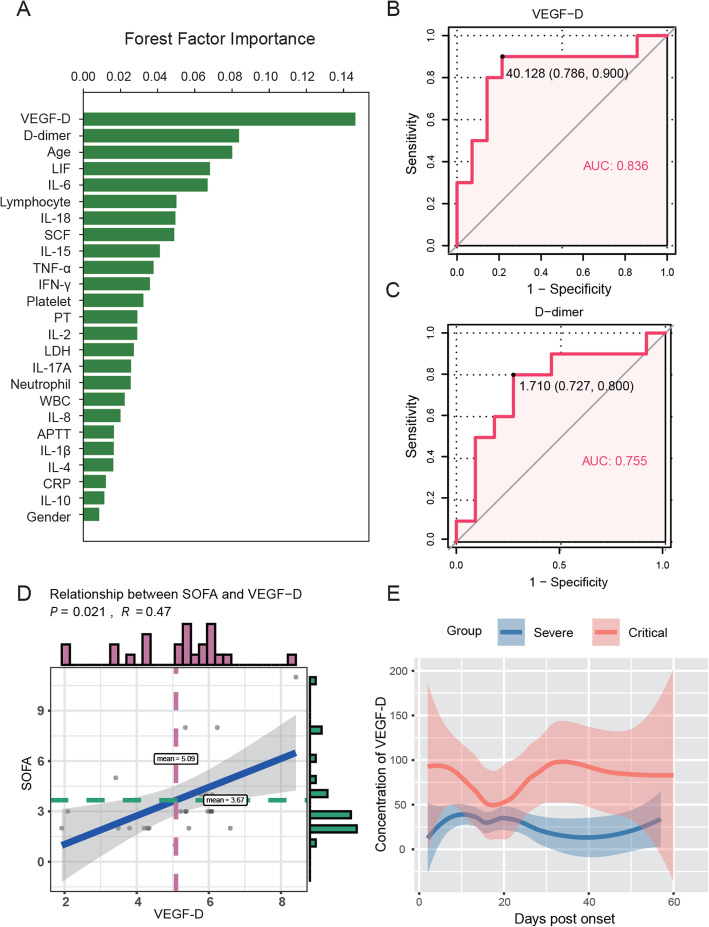
Strikingly, VEGF-D was identified as the most important indicator related to the severity of COVID-19 (ranked as 1, Fig. 1a). As expected, d-dimer, age, IL-6, and lymphocyte count associated with clinical outcomes of COVID-19 patients reported previously were also highly ranked. VEGF-D had a higher area under the curve (AUC) (0.836 (95% CI 0.648–1); Fig. 1b) than d-dimer (0.755 (95% CI 0.527–0.982); Fig. 1c). Consistently, VEGF-D levels were positively correlated with sequential organ failure assessment (SOFA) scores (Fig. 1d). As shown in Fig. 1e, critical patients had higher levels of VEGF-D than the severe cases during the whole course of hospitalization.
Fig. 1.
A high level of VEGF-D predicted progression of COVID-19. a Eleven clinical indicators and 14 cytokines were considered for inclusion and ranked by importance using random forest. b, c Receiver operating characteristic (ROC) analyses for VEGF-D (b) and d-dimer (c) in COVID-19 patients. d Relationship between VEGF-D and SOFA scores in severe and critical COVID-19 patients was analyzed by the Spearman rank correlation test. e Temporal changes of VEGF-D levels in each group during hospitalization. The median values of each time point (the day from onset) were shown. The 95% interval was plotted as a colored shadow
To our knowledge, this is the first report of VEGF-D as a potential biomarker for detecting the progression of COVID-19. Despite limited evidence in COVID-19, previous studies demonstrated an important role of VEGF in the pathogenesis of acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) by its properties to increase vascular permeability. Furthermore, VEGF is regarded as an indirect procoagulant for altering the hemostatic features of endothelial cells [6]. We hypothesized that elevated VEGF-D level might potentially relate to the storm of blood clots occurring in COVID-19 patients. Notably, it is of great interest to investigate the therapeutic effects of VEGF inhibitor in COVID-19 patients.
This study has limitations, including the small sample size, a single-center experience, and a variable time interval of each patient from admission to symptoms onset. Studies based on a larger cohort in additional sites are needed to verify our findings.
Acknowledgements
We thank all patients included in this study. We appreciate the works from Dr. Yao Sun from Intensive Care Medicine, Beijing Ditan Hospital; Professor Fujie Zhang, Dr. Di Wang, Dr. Rui Song, and Dr. Yangzi Song from Clinical and Research Center of Infectious Diseases, Beijing Ditan Hospital; and Dr. Mingxi Hua, Dr. Guoli Li, Dr. Juan Du, Dr. Ju Zhang, Kai Han, and Yonghong Yan from Institute of Infectious Diseases, Beijing Ditan Hospital in recruiting the patients, extracting the clinical information, and providing technical supports.
Authors’ contributions
YK and H. Zhang designed the study and wrote the manuscript. JL conducted the study and recruited the patients. YK and JH collected the samples, performed the experiments, and analyzed the data. XW analyzed the data and performed the statistical analysis. H. Zeng participated in the critical review of the manuscript. All authors read and approved the final manuscript.
Funding
This work was supported by the Beijing Municipal Science and Technology Commission (Z201100005420012), Capital Clinical Characteristic Application (Z171100001017052), National Key Sci-Tech Special Project of China (2018ZX10302207), and Beijing Municipal Administration of Hospital Clinical Medicine Development of Special Funding Support (ZYLX201802).
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Ethics approval and consent to participate
This study was approved by the Committee of Ethics at Beijing Ditan Hospital, Capital Medical University, Beijing, China, and each patient gave written informed consent.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yaxian Kong, Junyan Han and Xueying Wu contributed equally to this work.
Contributor Information
Yaxian Kong, Email: kongyaxian@ccmu.edu.cn.
Jingyuan Liu, Email: dtyyicu@ccmu.edu.cn.
Henghui Zhang, Email: zhhbao@ccmu.edu.cn.
References
- 1.Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y et al: Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323(11):1061–9. [DOI] [PMC free article] [PubMed]
- 2.Moore JB, June CH. Cytokine release syndrome in severe COVID-19. Science. 2020;368(6490):473–474. doi: 10.1126/science.abb8925. [DOI] [PubMed] [Google Scholar]
- 3.Willyard C. Coronavirus blood-clot mystery intensifies. Nature. 2020;581(7808):250. doi: 10.1038/d41586-020-01403-8. [DOI] [PubMed] [Google Scholar]
- 4.Wynants L, Van Calster B, Bonten MMJ, Collins GS, Debray TPA, De Vos M, Haller MC, Heinze G, Moons KGM, Riley RD, et al. Prediction models for diagnosis and prognosis of covid-19 infection: systematic review and critical appraisal. BMJ. 2020;369:m1328. doi: 10.1136/bmj.m1328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhang L, Yan X, Fan Q, Liu H, Liu X, Liu Z, Zhang Z: D-dimer levels on admission to predict in-hospital mortality in patients with Covid-19. J Thromb Haemost. 2020;18(6):1324–9. [DOI] [PMC free article] [PubMed]
- 6.Verheul HM, Hoekman K, Lupu F, Broxterman HJ, van der Valk P, Kakkar AK, Pinedo HM. Platelet and coagulation activation with vascular endothelial growth factor generation in soft tissue sarcomas. Clin Cancer Res. 2000;6(1):166–171. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.