Abstract
Gene duplication facilitates the evolution of biological complexity, as one copy of a gene retains its original function while a duplicate copy can acquire mutations that would otherwise diminish fitness. Duplication has played a particularly important role in the evolution of regulatory networks by permitting novel regulatory interactions and responses to stimuli. The diverse MarR family of transcription factors (MFTFs) illustrates this concept, ranging from highly specific repressors of single operons to pleiotropic global regulators controlling hundreds of genes. MFTFs are often genetically and functionally linked to antimicrobial efflux systems. However, the SlyA MFTF lineage in the Enterobacteriaceae plays little or no role in regulating efflux but rather functions as transcriptional counter-silencers, which alleviate xenogeneic silencing of horizontally-acquired genes and facilitate bacterial evolution by horizontal gene transfer. This review will explore recent advances in our understanding of MFTF traits that have contributed to their functional evolution.
Keywords: Gene regulation, gene duplication, Transcription factor, counter-silencing, Transcription, evolution
Introduction
In his book Evolution by Gene Duplication [1], Susumu Ohno argued that gene duplication is a driving force in evolution, suggesting that “natural selection (has) merely modified, while redundancy created.” He posited that gene duplications and the redundancy they produce provide the evolutionary space necessary for functional diversification and innovation. Duplication allows the exploration of otherwise “forbidden” mutations, generating novel functions unique from those of the ancestral gene, ultimately resulting in greater organismal complexity. In the absence of gene duplication, mutations are limited to those that do not disrupt essential gene function, constraining potential evolutionary trajectories. Although more recent evolutionary studies suggest that horizontal gene transfer has a greater impact on bacterial evolution than classical intragenomic duplication [2–4], Ohno’s ideas are relevant here as well, as the expansion of a gene family by extensive lateral transfer also provides valuable evolutionary space. These processes are exemplified by the evolution of gene regulatory networks. Transcription is typically regulated by transcription factors (TFs), which bind near gene promoters to modulate their transcription by RNA polymerase (RNAP). Although one genome may encode hundreds of unique TFs, these belong to as few as 10 unique TF families [5,6]. The TFs contained within each family are the products of gene duplication, and the maintenance of these duplicates implies a fitness advantage. This can occur through neofunctionalization, wherein a duplicate acquires a novel function not present in the original gene, or subfunctionalization, wherein the function of an original gene is divided between two or more copies via mutational divergence [7]. TF gene duplication allows both in cis variation, resulting from changes in the promoter driving expression of a TF, which affects the binding and activity of upstream TFs and RNAP, or in trans variation, resulting from changes in the coding sequence of the TF, which alters interactions with cognate targets or other interaction partners. The net result of these duplication events and the resulting variation are increasingly complex regulatory networks that are able to respond to a variety of environmental and physiological stimuli.
The MarR (Multiple antibiotic resistance Regulator) family of TFs (MFTFs) exemplifies these processes. MFTFs are ancient, predating the divergence of Archaea and Bacteria [8] and presently comprising one of the most common TF families in bacteria. Although the average bacterial genome encodes 7 unique MFTFs [9], the number can vary widely: Bacillus subtilis and Streptomyces coelicolor encode at least 20 each (depending on the strain), whereas Salmonella enterica serovar Typhimurium encodes 7, and the related enteric species Yersinia pseudotuberculosis encodes only 3. Even endosymbiotic species, which have undergone substantial genome loss, encode multiple MFTFs, including Sodalis glossinidius, which encodes 5 [10,11]. Despite their significant presence in bacteria, MFTFs exhibit limited sequence conservation between lineages, typically with less than 30% identity. This variability may reflect the inherent versatility of the MFTF backbone that allows them to interact with a variety of targets and respond to a variety of physiological and environmental signals. The ubiquity of MFTFs, particularly in the reduced genomes of endosymbiotic species, suggests that they serve an underappreciated role as central regulators of bacterial gene expression.
MFTFs were first recognized when E. coli mutants exhibiting heightened resistance to multiple antibiotics were observed to encode mutations in MarR, the prototypical MFTF [12]. Since that discovery, MFTFs have been found to play a role in a number of important biological processes, including antibiotic resistance, virulence [13], oxidative stress [14], central metabolism [9,15] and the catabolism of a variety of aromatic compounds [9]. Although MFTFs were originally regarded as classical repressors of transcription, often of very small regulons, more recent studies have demonstrated that some members of the family can also function as global regulators, both positively and negatively modulating gene expression [11,16,17]. This scenario is perhaps best exemplified by the SlyA MFTF lineage in Enterobacteriaceae, which has evolved to function as transcriptional counter-silencers [10,16,18,19]. SlyA proteins alleviate xenogeneic silencing of horizontally-acquired genes by proteins such as H-NS [20,21], thereby playing a vital role in the regulatory integration of horizontally-acquired genes. This allows a bacterial cell to realize a potential fitness benefit from horizontally-acquired genes, which might be detrimental if expressed in an unregulated fashion. Notably, SlyA is strongly conserved, as it is present in most species in Enterobacteriaceae, including endosymbionts such as S. glossinidius and Wigglesworthia glossinidia, in which it is under strong selective constraints [22]. The SlyA lineage is not unique in controlling large numbers of genes. ScoC, which belongs to a distinct MFTF lineage, positively and negatively regulates more than 560 genes in B. subtilis, which are involved in sporulation, transport, motility, and metabolism [23], although the mechanistic basis for its pleiotropic function is unknown. The existence of MFTFs functioning as both small regulon repressors and global counter-silencers provides a clear example of functional innovation and diversification as a consequence of gene duplication.
The MFTF DNA binding motif is highly variable.
Regardless of their specific function, MFTFs are generally defined by four common features: (1) a single globular domain, containing both (2) a winged-helix-turn-helix (wHTH) DNA-binding motif [24,25] and (3) a ligand-binding site that allows allosteric inhibition by environmental or physiological signals [26], and (4) genetic linkage to a multi-drug efflux pump. The chimeric wHTH (Fig. 1) domain consists of a classical prokaryotic helix-turn-helix motif, in which the recognition helix (α4) engages extensively with the major groove of the DNA duplex, acting as the primary determinant of specificity. The wing domain, more commonly observed in eukaryotic proteins, interacts closely with the minor groove and appears to increase the affinity of the interaction via indirect readout, wherein the shape of the DNA rather than the DNA sequence itself is the primary determinant of the DNA-TF interaction [25,27–30]. This is notable, as xenogeneic silencers such as H-NS and Lsr2 [31] also rely upon indirect readout, detecting a narrowing of the minor groove that is associated with TpA steps in AT-rich DNA. The emergence of MFTFs as counter-silencers may be due in part to the ability of the wing region to recognize sequences similar in structure to those recognized by xenogeneic silencers. Upon MFTF binding, the DNA major groove widens by 2–4Å to accommodate insertion of the recognition helix, while the minor grove can similarly widen and overtwist as a result of interactions with the wing [25,32]. These structural changes distort the DNA duplex, bending the DNA by ~15° and underwinding it by 1.2 to 1.4° [25,32]. This distortion may be important for counter-silencing function, which is accompanied by the formation of a bend within the silenced H-NS DNA complex [18]. A helix-helix (HH) motif, comprised of helices 03B11 and α2 and their connecting residues, also contributes to the MarR-DNA interaction via contacts with the phosphate backbone of the DNA duplex [25,28]. Genome-wide data characterizing MFTF-DNA interactions are limited. However, a number of studies have examined MFTF binding at specific loci and reveal that MFTFs typically recognize palindromic binding sites with an elevated AT-content [33–39]. This is partially due to a conserved arginine residue in the wing of many MFTFs (Fig. 1), which is essential for SlyA function [40], This residue makes multiple contacts with adenine and thymine bases in the minor groove [27]. However, the α4 recognition helix can vary significantly among MFTFs in both the number and type of interactions made with the major groove. Some MFTFs, such as OhrR of B. subtilis and SCO3205 of S. coelicolor, encode multiple residues (4 and 3, respectively) in α4, which form relatively sequence-specific hydrogen bonds with DNA. However, others are more reliant on the less specific or lower energy van der Waals interactions to form a MFTF-DNA complex. These include SlyA, which has only one hydrogen bond-forming residue but two van der Waals-interacting residues, and MepR of S. aureus, which does not form any hydrogen bonds but is instead reliant on 4 van der Waals contacts [27]. MarR, which exhibits high sequence specificity with only a single binding site in the E. coli chromosome [41] exhibits both types of interaction, forming 3 hydrogen bonds and 5 van der Waals contacts [28]. More unusual interactions are also possible, as the recognition helix of ST1170, an MFTF from Sulfolobus tokodaii, does not insert into the major groove at all, but rather makes non-specific contacts with the sugar-phosphate backbone of DNA [42]. The DNA passes over the wHTH domain of ST1170 to make contact only with the wing. Given these observations, it is conceivable that MFTF gene duplication has allowed α4 to become reliant upon hydrogen bonds when more specific interactions of a classical repressor are required, or upon van der Waals contacts when performing a more pleiotropic global role, as in SlyA and other counter-silencers (Fig. 2).
Figure 1. MFTF DNA-protein interaction.
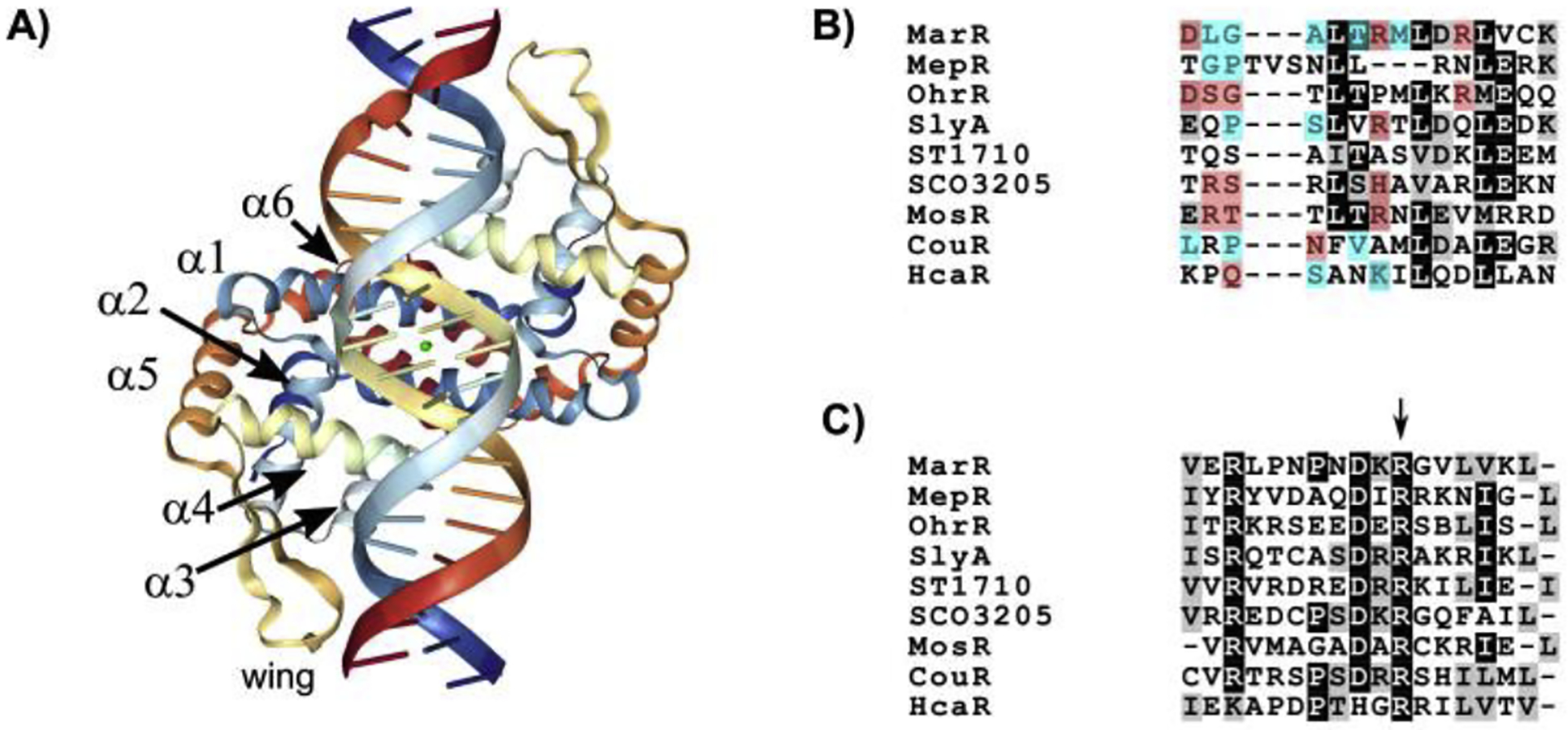
The crystal structure of MarR C80S is shown in complex with its operator DNA (PDB ID: 5H3R) [28] (A). The image was generated using the NGL viewer [67]. The axis of symmetry is indicated by a green circle. Helices and the wing of one MarR monomer are indicated. The aligned recognition helices (α4) of representative MFTFs are shown in (B). Residues forming hydrogen bonds with the DNA major groove are highlighted in red. Residues forming weaker and less specific van der Waals contacts are highlighted in blue, based on data originally presented by [27] and the following structural data: E. coli MarR [28]; S. aureus MepR [27]; B. subtilis OhrR [25]; S. Typhimurium SlyA [32]; S. tokodaii ST1710 [42]; S. coelicolor SCO3205 [68]; Mycobacterium tuberculosis MosR [69]; Rhodopseudomonas palustris CouR [70]; Acinetobacter sp. ADP1 HcaR [71]. The aligned wings of the same MFTFs are shown in (C). A conserved arginine residue responsible for interacting with the DNA minor groove is indicated with an arrow.
Figure 2. Model of functional evolution by gene duplication.
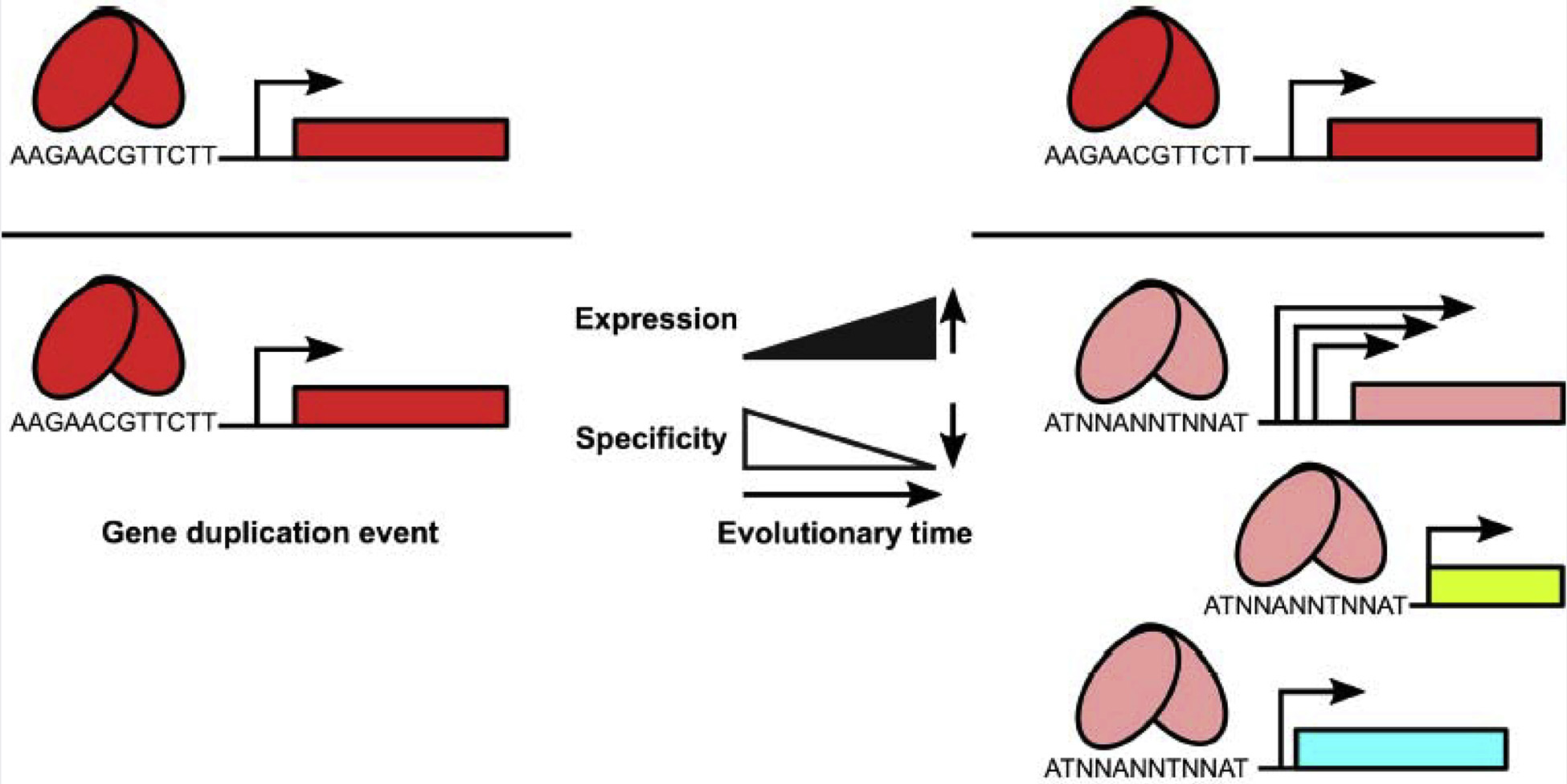
Pleiotropic regulators (pink ovals; pink box represents the coding sequence) such as SlyA may have arisen following duplication of a gene encoding a more specific classical repressor (red ovals; red box represents the coding sequence). This duplication allows one gene copy to accumulate mutations that reduce its binding specificity and thereby increase its potential target repertoire, as well as increasing its expression level, while the other copy retains its ancestral function. Eventually, the duplicate copy evolves to perform a unique role as a regulator of multiple genes.
MFTFs are allosterically inhibited by multiple stimuli.
The mechanism of allosteric inhibition, which sensitizes MFTFs to physiological and environmental stimuli, is a point of conjecture for MarR. Early studies observed that multi-drug resistance could be induced by the addition of salicylate to E. coli cultures [43], which was later shown to result from the inhibition of MarR-mediated DNA binding and repression [44]. Because MFTFs are comprised of a singular globular domain, ligand binding can easily impact interaction with DNA. The binding of small molecules like salicylate causes the α4 recognition helix to rotate out of register with the major groove of DNA, thereby inhibiting DNA binding [10,24,45,46]. Subsequent studies have identified endogenous aromatic acid metabolites that are structurally similar to salicylate and can also inhibit MarR [47,48]. More recently, it has been postulated that MarR is inhibited by cytoplasmic copper(II) liberated from membrane-bound proteins during envelope stress [38]. These copper(II) ions oxidize the conserved C80 cysteine residue of MarR, promoting the formation of disulfide bonds between MarR dimers to form a tetramer that is unable to interact with the DNA major groove. Inhibition by aromatic carboxylates was also suggested by these authors to result from cysteine oxidation, perhaps as a result of salicylate-induced membrane stress and copper(II) release. However, although the cysteine oxidation model of inhibition is attractive, as similar models have been observed for other MFTFs such as OhrR of B. subtilis, which utilizes cysteine oxidation to sense organic hydroperoxides [14,49], it is not universal. Mutation of the lone conserved cysteine residue in SlyA has no effect on its counter-silencing activity nor on its inhibition by aromatic carboxylates [10]. Mutagenesis of an analogous cysteine residue in the Staphylococcus aureus MFTF MhqR similarly had no impact on its function as a quinone-responsive regulator of antimicrobial resistance [50], and non-cysteine-dependent aromatic carboxylate inhibitory mechanisms have been described for an array of MFTFs [9,51]. Although we cannot specify ancestral endogenous ligands with certainty, we note that both archaeal MFTFs and counter-silencing MFTFs such as SlyA and RovA have retained the ability to bind and be inhibited by aromatic carboxylates [10,42,45], suggesting that the ancestral ligand(s) is a similar molecule. It is possible that some MFTFs are subject to inhibition by both cysteine and aromatic ligand binding, due to an inherent promiscuity of the ligand-binding site [46]. This may be the case with MarR [24,38], and if true for other MFTFs, could contribute to their functional versatility.
Genetic linkage to transporters suggests a physiological function for MFTFs.
The third common feature of MFTFs, genetic linkage to efflux pumps or transporters, may reflect the primordial function of this protein family. In S. Typhimurium, 4 out of 7 MFTF genes are genetically linked to multi-drug efflux pump or transporter-encoding genes (Fig. 3), including the pleiotropic counter-silencer SlyA, although this linkage has been lost in most other enteric species, including endosymbiotic species. This suggests that, although SlyA may be under selective constraints [22], the YdhIJK efflux pump is no longer important for SlyA function [10]. Similar linkages to pumps and transporters are observed in other species, including Archaea. In Sulfolobus sulfataricus P2, 4 out of 6 MFTFs are linked to MFS or ABC family transporters. Even when MFTFs are not directly linked to efflux pump coding genes, they are often functionally linked to efflux, as is MarR. Although the marRAB operon is not genetically linked to efflux pump genes, MarR represses the expression of the activator MarA, which up-regulates synthesis of the AcrAB-TolC efflux pump, a primary determinant of intrinsic antibiotic resistance in E. coli [52,53]. This suggests an ancestral role for MFTFs in maintaining physiological homeostasis by sensing and regulating the efflux of both toxic metabolites as well as xenobiotics, a hypothesis supported by studies identifying aromatic metabolites as endogenous ligands of MarR [47,48]. However, it is notable that these transporters are not all genetically related. AcrAB, regulated by MarR, is a member of the RND (Resistance-Nodulation-Division) superfamily, whereas many other regulated transporters are members of the Major Facilitator Superfamily (MFS), suggesting that some of these linkages are a product of convergent evolution.
Figure 3. MFTFs linked to transporters.
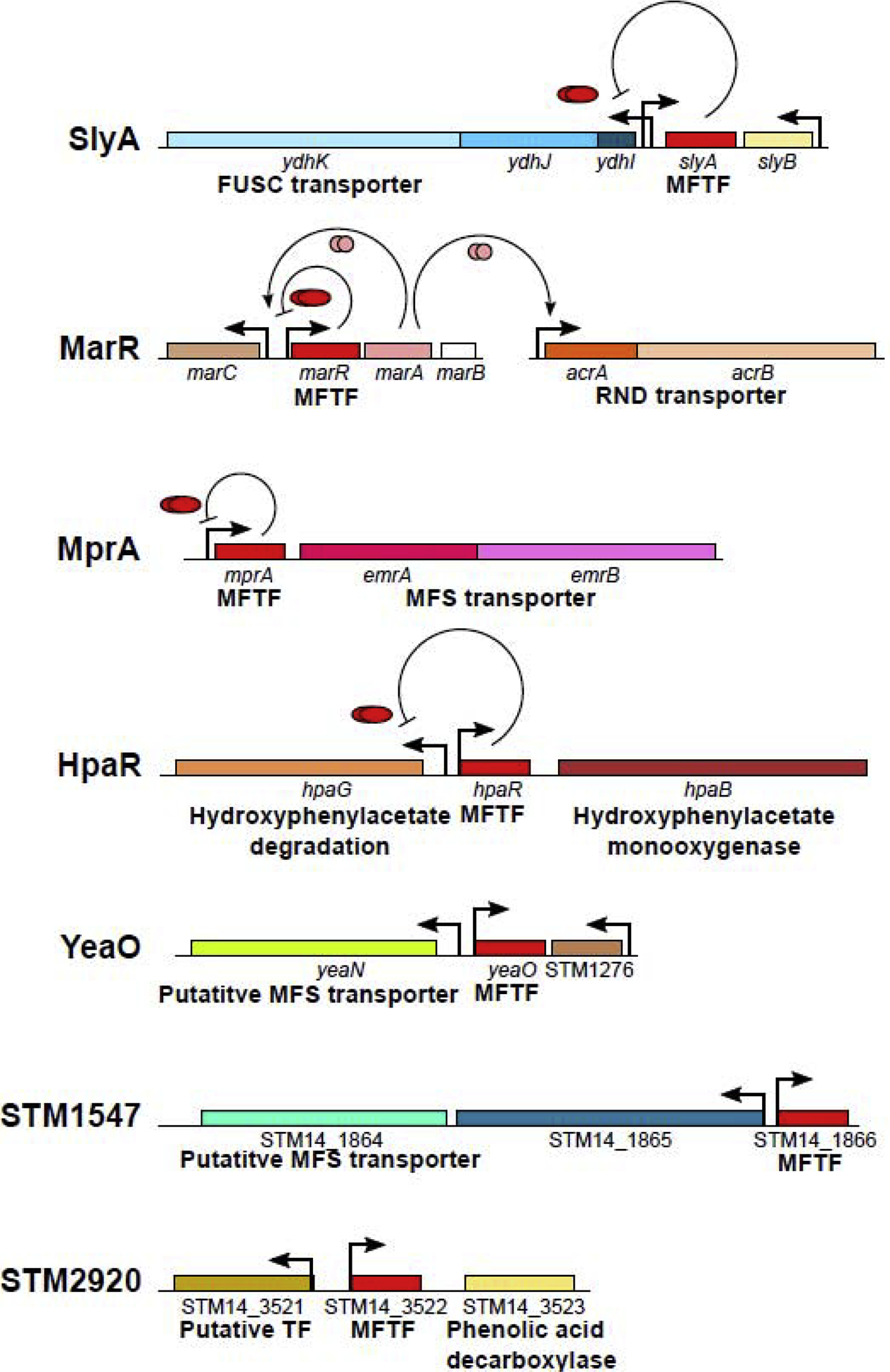
S. Typhimurium encodes 7 MFTFs, each of which is depicted with its relevant linked gene, with known or hypothetical functions indicated below. Promoters are indicated by arrows. Local regulatory functions are shown if characterized and indicated by curved lines ending in bars (repression) or arrows (activation; e.g., MarA). The slyA gene is divergently transcribed from the ydhIJK operon, which encodes an FUSC superfamily transporter [72]. The marR gene is not linked to a pump gene but indirectly regulates acrAB, encoding an RND superfamily transporter [73], via MarA [74]. MprA functions as an autorepressor and is co-transcribed with emrAB, which encodes an MFS family transporter [75,76]. yeaO and STM1547 are also linked to putative MFS family transporters. Diagram is not to scale.
Variation in cis has contributed to pleiotropic function.
As mentioned above, cis-level variation, which alters the expression of individual MFTFs, can also contribute to regulatory evolution and functional adaptation. This was recently demonstrated in an analysis of SlyA alleles from S. Typhimurium and E. coli. Although allelic exchange demonstrated that both alleles are capable of functioning as counter-silencers in S. Typhimurium [10], SlyA does not play a significant role in the E. coli regulatory network [46–48], apparently because of low levels of expression. The S. Typhimurium slyA promoter has evolved to provide high expression levels under conditions found in the intra-phagosomal environment [10], which corresponds to the essential role of SlyA in resistance to macrophage killing [54]. This is supported by studies of hlyE, which encodes a cytolytic toxin in S. enterica and E. coli. Although non-pathogenic E. coli strains encode a functional hlyE allele, it is silent except in the absence of hns [55] or during the over-expression of slyA [56,57]. This was shown to be due to SlyA-mediated counter-silencing of the hlyE promoter [58]. However, non-pathogenic E. coli strains are unable to express slyA at levels sufficient for counter-silencing. In contrast, even under non-inducing conditions, expression levels of SlyA in S. Typhimurium are typically higher than that of any other MFTF [59] (Fig. 4). Similarly, the Y. pseudotuberculosis SlyA ortholog, called RovA, is the most strongly expressed MFTF in that species [60]. Even in B. subtilis, where the functions of most MFTFs are presently uncharacterized, the pleiotropic regulators ScoC [23] and MhqR [61,62] are highly expressed [63].
Figure 4. SlyA is the most highly expressed S. Typhimurium MFTF in late exponential and early stationary phases of growth.

Raw expression levels (in transcripts per million) of each S Typhimurium MFTF were measured during early exponential phase (EEP), mid-exponential phase (MEP), late exponential phase (LEP), early stationary phase (ESP) and late stationary phase (LSP). Data are from previously published transcriptomic data [59], which are available online (http://bioinf.gen.tcd.ie/cgi-bin/salcom.pl?_HL).
Conclusions
Gene duplication appears to have facilitated the asymmetrical adaptation of MFTF lineages (Fig. 2). Although recent studies have highlighted the prominence of horizontal gene transfer in bacterial evolution [2,4], these studies are typically limited to the last 100 million years. We do not dispute that horizontal gene transfer has played a role in MFTF expansion, as some lineages are suggested to have been acquired via horizontal gene transfer, such as HucR of Deinococcus radiodurans [64]. However, MFTFs, along with the AsnC family, represent the original wHTH TFs and are thought to have been present in the last universal common ancestor before the divergence of Archaea and Bacteria, more than 3 billion years ago [8,65,66]. Gene duplication must have occurred to allow the emergence of other wHTH TF families and their subsequent expansion. Indeed, evolutionary studies acknowledge that intragenomic gene duplication may have played a more prominent role in initial network establishment [4]. Horizontal gene transfer may have then allowed the subsequent refinement of MFTF function. Ohno’s model [1] remains applicable, with extensive horizontal transfer providing new evolutionary space for functional adaptation. Although some MFTFs such as MarR exhibit tightly controlled expression circuits and highly specific DNA interactions with the α4 helix, others such as SlyA possess a promiscuous recognition helix and more robust expression levels to accommodate their global regulatory roles. The conserved feature of allosteric MFTF regulation by small aromatic molecules provides a potential mechanistic linkage between the regulation of drug resistance and virulence. The further analysis of this fascinating ancient family of regulators promises to provide important new insights into transcriptional regulation as a driving force in bacterial evolution.
Highlights.
MarR family transcription factors (MFTFs) are ancient and ubiquitous regulatory proteins, predating the divergence of Archaea and Bacteria.
Gene duplication has accommodated the adaptation of MFTFs to multiple regulatory functions.
Allosteric inhibition, typically by small aromatic molecules, confers MFTF responsiveness to environmental and physiological stimuli.
Variation in DNA-binding domains and promoters contributes to MFTF specificity.
Acknowledgements
The National Institutes of Health provided support to F.C.F. (Grant numbers: AI39557, AI44486, AI118962, and AI112640).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflict of interest statement
Nothing declared.
References and recommended reading
Papers of particular interest, published within the period of review, have been highlighted as:
• of special interest
•• of outstanding interest
- 1.Ohno S: Evolution by Gene Duplication New York: Springer; 1970. [Google Scholar]
- 2.Treangen TJ, Rocha EP: Horizontal transfer, not duplication, drives the expansion of protein families in prokaryotes. PLoS Genet 2011, 7:e1001284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dagan T, Artzy-Randrup Y, Martin W: Modular networks and cumulative impact of lateral transfer in prokaryote genome evolution. Proc Natl Acad Sci U S A 2008, 105:10039–10044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pál C, Papp B, Lercher MJ: Adaptive evolution of bacterial metabolic networks by horizontal gene transfer. Nat Genet 2005, 37:1372–1375. [DOI] [PubMed] [Google Scholar]
- 5.Charoensawan V, Wilson D, Teichmann SA: Genomic repertoires of DNA-binding transcription factors across the tree of life. Nucleic Acids Res 2010, 38:7364–7377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Charoensawan V, Wilson D, Teichmann SA: Lineage-specific expansion of DNA-binding transcription factor families. Trends Genet 2010, 26:388–393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Konrad A, Teufel AI, Grahnen JA, Liberles DA: Toward a general model for the evolutionary dynamics of gene duplicates. Genome Biol Evol 2011, 3:1197–1209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pérez-Rueda E, Collado-Vides J: Common history at the origin of the position-function correlation in transcriptional regulators in archaea and bacteria. J Mol Evol 2001, 53:172–179. [DOI] [PubMed] [Google Scholar]
- 9.Grove A: Regulation of Metabolic Pathways by MarR Family Transcription Factors. Comput Struct Biotechnol J 2017, 15:366–371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. ••.Will WR, Brzovic P, Le Trong I, Stenkamp RE, Lawrenz MB, Karlinsey JE, Navarre WW, Main-Hester K, Miller VL, Libby SJ, et al. : The evolution of SlyA/RovA transcription factors from repressors to countersilencers in Enterobacteriaceae. MBio 2019, 10:e00009–19. [DOI] [PMC free article] [PubMed] [Google Scholar]; This study examined the SlyA lineage of MFTFs, demonstrating that they are conserved throughout the Enterobacteriaceae, where they likely function as counter-silencers. Counter-silencing function is at least partly attributable to in cis adaptation, as the SlyA lineage is strongly expressed in species where it plays a significant regulatory role.
- 11. •.Clayton AL, Enomoto S, Su Y, Dale C: The regulation of antimicrobial peptide resistance in the transition to insect symbiosis. Mol Microbiol 2017, 103:958–972. [DOI] [PMC free article] [PubMed] [Google Scholar]; This study describes a MFTF encoded by both the insect endosymbiont S. glossinidius and its free-living relative, S. praecaptivus, where it is a pleiotropic regulator required for antimicrobial peptide resistance and intracellular growth in the insect host. The presence of a pleiotropic MFTF in an insect endosymbiont suggests that it serves an essential role in bacterial regulatory networks.
- 12.Cohen SP, Yan W, Levy SB: A multidrug resistance regulatory chromosomal locus is widespread among enteric bacteria. J Infect Dis 1993, 168:484–488. [DOI] [PubMed] [Google Scholar]
- 13.Ellison DW, Miller VL: H-NS represses inv transcription in Yersinia enterocolitica through competition with RovA and interaction with YmoA. J Bacteriol 2006, 188:5101–5112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fuangthong M, Helmann JD: The OhrR repressor senses organic hydroperoxides by reversible formation of a cysteine-sulfenic acid derivative. Proceedings of the National Academy of Sciences 2002, 99:6690–6695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Huang H, Sivapragasam S, Grove A: The regulatory role of Streptomyces coelicolor TamR in central metabolism. Biochemical Journal 2015, 466:347–358. [DOI] [PubMed] [Google Scholar]
- 16.Navarre WW, Halsey TA, Walthers D, Frye J, McClelland M, Potter JL, Kenney LJ, Gunn JS, Fang FC, Libby SJ: Co-regulation of Salmonella enterica genes required for virulence and resistance to antimicrobial peptides by SlyA and PhoP/PhoQ. Mol Microbiol 2005, 56:492–508. [DOI] [PubMed] [Google Scholar]
- 17.Cathelyn JS, Crosby SD, Lathem WW, Goldman WE, Miller VL: RovA, a global regulator of Yersinia pestis, specifically required for bubonic plague. Proc Natl Acad Sci U S A 2006, 103:13514–13519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. •.Will WR, Bale DH, Reid PJ, Libby SJ, Fang FC: Evolutionary expansion of a regulatory network by counter-silencing. Nat Commun 2014, 5:5270. [DOI] [PMC free article] [PubMed] [Google Scholar]; This study demonstrated the mechanistic basis for the novel regulatory role of SlyA in alleviating xenogeneic silencing.
- 19.Will WR, Navarre WW, Fang FC: Integrated circuits: how transcriptional silencing and counter-silencing facilitate bacterial evolution. Curr Opin Microbiol 2015, 23:8–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Navarre WW, Porwollik S, Wang Y, McClelland M, Rosen H, Libby SJ, Fang FC: Selective silencing of foreign DNA with low GC content by the H-NS protein in Salmonella. Science 2006, 313:236–238. [DOI] [PubMed] [Google Scholar]
- 21.Lucchini S, Rowley G, Goldberg MD, Hurd D, Harrison M, Hinton JC: H-NS mediates the silencing of laterally acquired genes in bacteria. PLoS Pathog 2006, 2:e81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. •.Sabater-Muñoz B, Toft C, Alvarez-Ponce D, Fares MA: Chance and necessity in the genome evolution of endosymbiotic bacteria of insects. ISME J 2017, 11:1291–1304. [DOI] [PMC free article] [PubMed] [Google Scholar]; This study sought to define the impact of genetic drift on enodosymbiotic species, which exhibit elevated rates of mutation. The primary conclusion of this study is that genetic drift has played a primary role in determining endosymbiotic associations, while the insect host has had a minor effect. However, SlyA is under strong selective constraints in W. glossinidia despite increased drift, whereas the linked YdhIJK, efflux pump has been lost, suggesting that SlyA now performs a critical function unrelated to YdhIJK.
- 23.Caldwell R, Sapolsky R, Weyler W, Maile RR, Causey SC, Ferrari E: Correlation between Bacillus subtilis scoC phenotype and gene expression determined using microarrays for transcriptome analysis. J Bacteriol 2001, 183:7329–7340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Alekshun MN, Levy SB, Mealy TR, Seaton BA, Head JF: The crystal structure of MarR, a regulator of multiple antibiotic resistance, at 2.3 A resolution. Nat Struct Biol 2001, 8:710–714. [DOI] [PubMed] [Google Scholar]
- 25.Hong M, Fuangthong M, Helmann JD, Brennan RG: Structure of an OhrR-ohrA operator complex reveals the DNA binding mechanism of the MarR family. Mol Cell 2005, 20:131–141. [DOI] [PubMed] [Google Scholar]
- 26.Cohen SP, Levy SB, Foulds J, Rosner JL: Salicylate induction of antibiotic resistance in Escherichia coli: activation of the mar operon and a mar-independent pathway. J Bacteriol 1993, 175:7856–7862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. ••.Birukou I, Seo SM, Schindler BD, Kaatz GW, Brennan RG: Structural mechanism of transcription regulation of the Staphylococcus aureus multidrug efflux operon mepRA by the MarR family repressor MepR. Nucleic Acids Res 2014, 42:2774–2788. [DOI] [PMC free article] [PubMed] [Google Scholar]; This structural characterization of MepR and its comparison to other proteins explains how MFTFs might evolve their DNA-binding surfaces to become less specific and more pleiotropic.
- 28.Zhu R, Hao Z, Lou H, Song Y, Zhao J, Chen Y, Zhu J, Chen PR: Structural characterization of the DNA-binding mechanism underlying the copper(II)-sensing MarR transcriptional regulator. J Biol Inorg Chem 2017, 22:685–693. [DOI] [PubMed] [Google Scholar]
- 29.Dorman CJ, Dorman MJ: Control of virulence gene transcription by indirect readout in Vibrio cholerae and Salmonella enterica serovar Typhimurium. Environ Microbiol 2017, 19:3834–3845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rohs R, West SM, Sosinsky A, Liu P, Mann RS, Honig B: The role of DNA shape in protein-DNA recognition. Nature 2009, 461:1248–1253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gordon BR, Li Y, Cote A, Weirauch MT, Ding P, Hughes TR, Navarre WW, Xia B, Liu J: Structural basis for recognition of AT-rich DNA by unrelated xenogeneic silencing proteins. Proc Natl Acad Sci U S A 2011, 108:10690–10695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. •.Dolan KT, Duguid EM, He C: Crystal structures of SlyA protein, a master virulence regulator of Salmonella, in free and DNA-bound states. J Biol Chem 2011, 286:22178–22185. [DOI] [PMC free article] [PubMed] [Google Scholar]; A structural characterization of the SlyA-DNA complex suggests that some MFTFs may be more reliant on indirect readout to bind DNA.
- 33.Perera IC, Grove A: MarR homologs with urate-binding signature. Protein Sci 2011, 20:621–629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bussmann M, Baumgart M, Bott M: RosR (Cg1324), a hydrogen peroxide-sensitive MarR-type transcriptional regulator of Corynebacterium glutamicum. J Biol Chem 2010, 285:29305–29318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rouanet C, Reverchon S, Rodionov DA, Nasser W: Definition of a consensus DNA-binding site for PecS, a global regulator of virulence gene expression in Erwinia chrysanthemi and identification of new members of the PecS regulon. J Biol Chem 2004, 279:30158–30167. [DOI] [PubMed] [Google Scholar]
- 36.Roy A, Ranjan A: HosA, a MarR family transcriptional regulator, represses nonoxidative hydroxyarylic acid decarboxylase operon and is modulated by 4-hydroxybenzoic acid. Biochemistry 2016, 55:1120–1134. [DOI] [PubMed] [Google Scholar]
- 37.Guo J, Zhang X, Lu X, Liu W, Chen Z, Li J, Deng L, Wen Y: SAV4189, a MarR-Family Regulator in Streptomyces avermitilis, activates avermectin biosynthesis. Front Microbiol 2018, 9:1358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. •.Hao Z, Lou H, Zhu R, Zhu J, Zhang D, Zhao BS, Zeng S, Chen X, Chan J, He C, et al. : The multiple antibiotic resistance regulator MarR is a copper sensor in Escherichia coli. Nat Chem Biol 2014, 10:21–28. [DOI] [PubMed] [Google Scholar]; This biochemical study of MarR suggests a novel inhibition mechanism for MFTFs that are inhibited by aromatic carboxylates: free intracellular copper(II), liberated as a result of envelope stress, oxidizes a cysteine residue to promote disulfide bond formation, tetramerization and subsequent inhibtion.
- 39.Stapleton MR, Norte VA, Read RC, Green J: Interaction of the Salmonella typhimurium transcription and virulence factor SlyA with target DNA and identification of members of the SlyA regulon. J Biol Chem 2002, 277:17630–17637. [DOI] [PubMed] [Google Scholar]
- 40.Haider F, Lithgow JK, Stapleton MR, Norte VA, Roberts RE, Green J: DNA recognition by the Salmonella enterica serovar Typhimurium transcription factor SlyA. Int Microbiol 2008, 11:245–250. [DOI] [PubMed] [Google Scholar]
- 41.Sharma P, Haycocks JRJ, Middlemiss AD, Kettles RA, Sellars LE, Ricci V, Piddock LJV, Grainger DC: The multiple antibiotic resistance operon of enteric bacteria controls DNA repair and outer membrane integrity. Nat Commun 2017, 8:1444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kumarevel T, Tanaka T, Umehara T, Yokoyama S: ST1710-DNA complex crystal structure reveals the DNA binding mechanism of the MarR family of regulators. Nucleic Acids Res 2009, 37:4723–4735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Rosner JL: Nonheritable resistance to chloramphenicol and other antibiotics induced by salicylates and other chemotactic repellents in Escherichia coli K-12. Proc Natl Acad Sci U S A 1985, 82:8771–8774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Martin RG, Rosner JL: Binding of purified multiple antibiotic-resistance repressor protein (MarR) to mar operator sequences. Proc Natl Acad Sci U S A 1995, 92:5456–5460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Saridakis V, Shahinas D, Xu X, Christendat D: Structural insight on the mechanism of regulation of the MarR family of proteins: high-resolution crystal structure of a transcriptional repressor from Methanobacterium thermoautotrophicum. J Mol Biol 2008, 377:655–667. [DOI] [PubMed] [Google Scholar]
- 46. •.Chang YM, Chen CK, Ko TP, Chang-Chien MW, Wang AH: Structural analysis of the antibiotic-recognition mechanism of MarR proteins. Acta Crystallogr D Biol Crystallogr 2013, 69:1138–1149. [DOI] [PubMed] [Google Scholar]; A structural analysis of TcaR of S. epidermidis proposes a mechanism for binding of a variety of ligands to MFTFs.
- 47.Chubiz LM, Rao CV: Aromatic acid metabolites of Escherichia coli K-12 can induce the marRAB operon. J Bacteriol 2010, 192:4786–4789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ruiz C, Levy SB: Regulation of acrAB expression by cellular metabolites in Escherichia coli. J Antimicrob Chemother 2014, 69:390–399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Fuangthong M, Atichartpongkul S, Mongkolsuk S, Helmann JD: OhrR is a repressor of ohrA, a key organic hydroperoxide resistance determinant in Bacillus subtilis. J Bacteriol 2001, 183:4134–4141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Fritsch VN, Loi VV, Busche T, Sommer A, Tedin K, Nurnberg DJ, Kalinowski J, Bernhardt J, Fulde M, Antelmann H: The MarR-Type repressor MhqR confers quinone and antimicrobial resistance in Staphylococcus aureus. Antioxid Redox Signal 2019, 31:1235–1252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Tropel D, van der Meer JR: Bacterial transcriptional regulators for degradation pathways of aromatic compounds. Microbiol Mol Biol Rev 2004, 68:474–500, table of contents. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sulavik MC, Houseweart C, Cramer C, Jiwani N, Murgolo N, Greene J, DiDomenico B, Shaw KJ, Miller GH, Hare R, et al. : Antibiotic susceptibility profiles of Escherichia coli strains lacking multidrug efflux pump genes. Antimicrob Agents Chemother 2001, 45:1126–1136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Swick MC, Morgan-Linnell SK, Carlson KM, Zechiedrich L: Expression of multidrug efflux pump genes acrAB-tolC, mdfA, and norE in Escherichia coli clinical isolates as a function of fluoroquinolone and multidrug resistance. Antimicrob Agents Chemother 2011, 55:921–924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Libby SJ, Goebel W, Ludwig A, Buchmeier N, Bowe F, Fang FC, Guiney DG, Songer JG, Heffron F: A cytolysin encoded by Salmonella is required for survival within macrophages. Proc Natl Acad Sci U S A 1994, 91:489–493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Westermark M, Oscarsson J, Mizunoe Y, Urbonaviciene J, Uhlin BE: Silencing and activation of ClyA cytotoxin expression in Escherichia coli. J Bacteriol 2000, 182:6347–6357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ludwig A, Tengel C, Bauer S, Bubert A, Benz R, Mollenkopf HJ, Goebel W: SlyA, a regulatory protein from Salmonella typhimurium, induces a haemolytic and pore-forming protein in Escherichia coli. Mol Gen Genet 1995, 249:474–486. [DOI] [PubMed] [Google Scholar]
- 57.del Castillo FJ, Leal SC, Moreno F, del Castillo I: The Escherichia coli K-12 sheA gene encodes a 34-kDa secreted haemolysin. Mol Microbiol 1997, 25:107–115. [DOI] [PubMed] [Google Scholar]
- 58.Lithgow JK, Haider F, Roberts IS, Green J: Alternate SlyA and H-NS nucleoprotein complexes control hlyE expression in Escherichia coli K-12. Mol Microbiol 2007, 66:685–698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kröger C, Colgan A, Srikumar S, Händler K, Sivasankaran SK, Hammarlöf DL, Canals R, Grissom JE, Conway T, Hokamp K, et al. : An infection-relevant transcriptomic compendium for Salmonella enterica Serovar Typhimurium. Cell Host Microbe 2013, 14:683–695. [DOI] [PubMed] [Google Scholar]
- 60.Avican K, Fahlgren A, Huss M, Heroven AK, Beckstette M, Dersch P, Fällman M: Reprogramming of Yersinia from virulent to persistent mode revealed by complex in vivo RNA-seq analysis. PLoS Pathog 2015, 11:e1004600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Towe S, Leelakriangsak M, Kobayashi K, Van Duy N, Hecker M, Zuber P, Antelmann H: The MarR-type repressor MhqR (YkvE) regulates multiple dioxygenases/glyoxalases and an azoreductase which confer resistance to 2-methylhydroquinone and catechol in Bacillus subtilis. Mol Microbiol 2007, 66:40–54. [DOI] [PubMed] [Google Scholar]
- 62.Leelakriangsak M, Huyen NT, Towe S, van Duy N, Becher D, Hecker M, Antelmann H, Zuber P: Regulation of quinone detoxification by the thiol stress sensing DUF24/MarR-like repressor, YodB in Bacillus subtilis. Mol Microbiol 2008, 67:1108–1124. [DOI] [PubMed] [Google Scholar]
- 63.Nicolas P, Mader U, Dervyn E, Rochat T, Leduc A, Pigeonneau N, Bidnenko E, Marchadier E, Hoebeke M, Aymerich S, et al. : Condition-dependent transcriptome reveals high-level regulatory architecture in Bacillus subtilis. Science 2012, 335:1103–1106. [DOI] [PubMed] [Google Scholar]
- 64.Wilkinson SP, Grove A: HucR, a novel uric acid-responsive member of the MarR family of transcriptional regulators from Deinococcus radiodurans. J Biol Chem 2004, 279:51442–51450. [DOI] [PubMed] [Google Scholar]
- 65.Lemmens L, Maklad HR, Bervoets I, Peeters E: Transcription regulators in archaea: homologies and differences with bacterial regulators. J Mol Biol 2019, 431:4132–4146. [DOI] [PubMed] [Google Scholar]
- 66.Feng DF, Cho G, Doolittle RF: Determining divergence times with a protein clock: update and reevaluation. Proc Natl Acad Sci U S A 1997, 94:13028–13033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Rose AS, Bradley AR, Valasatava Y, Duarte JM, Prlic A, Rose PW: NGL viewer: web-based molecular graphics for large complexes. Bioinformatics 2018, 34:3755–3758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Stevenson CE, Assaad A, Chandra G, Le TB, Greive SJ, Bibb MJ, Lawson DM: Investigation of DNA sequence recognition by a streptomycete MarR family transcriptional regulator through surface plasmon resonance and X-ray crystallography. Nucleic Acids Res 2013, 41:7009–7022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Brugarolas P, Movahedzadeh F, Wang Y, Zhang N, Bartek IL, Gao YN, Voskuil MI, Franzblau SG, He C: The oxidation-sensing regulator (MosR) is a new redox-dependent transcription factor in Mycobacterium tuberculosis. J Biol Chem 2012, 287:37703–37712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Cogan DP, Baraquet C, Harwood CS, Nair SK: Structural basis of transcriptional regulation by CouR, a repressor of coumarate catabolism, in Rhodopseudomonas palustris. J Biol Chem 2018, 293:11727–11735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kim Y, Joachimiak G, Bigelow L, Babnigg G, Joachimiak A: How aromatic compounds block dna binding of hcar catabolite regulator. J Biol Chem 2016, 291:13243–13256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Utsumi R, Yagi T, Katayama S, Katsuragi K, Tachibana K, Toyoda H, Ouchi S, Obata K, Shibano Y, Noda M: Molecular cloning and characterization of the fusaric acid-resistance gene from Pseudomonas cepacia. Agric Biol Chem 1991, 55:1913–1918. [PubMed] [Google Scholar]
- 73.Nikaido H: RND transporters in the living world. Res Microbiol 2018, 169:363–371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Alekshun MN, Levy SB: The mar regulon: multiple resistance to antibiotics and other toxic chemicals. Trends Microbiol 1999, 7:410–413. [DOI] [PubMed] [Google Scholar]
- 75.Lomovskaya O, Lewis K, Matin A: EmrR is a negative regulator of the Escherichia coli multidrug resistance pump EmrAB. J Bacteriol 1995, 177:2328–2334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Pao SS, Paulsen IT, Saier MH Jr.: Major facilitator superfamily. Microbiol Mol Biol Rev 1998, 62:1–34. [DOI] [PMC free article] [PubMed] [Google Scholar]


