Highlights
-
•
Baricitinib, an anti-JAK1/JAK2, reduces cytokine release and SARS-Co-V2 entry.
-
•
In a retrospective multicenter study baricitinib reduces COVID-19 mortality rate.
-
•
Baricitinib reduces intensive care unit admissions of COVID-19 pneumonia.
-
•
Baricitinb reduces SARS-CoV-2 viral burden detected by nasopharyngeal swab.
-
•
Baricitinib used for 2 weeks was not associated with serious adverse events.
Keywords: COVID-19, Baricitinib, JAK1/2, Pneumonia, Retrospective study, SARS-Co-V2
Graphical abstract
As recently discussed in the Journal,1 baricitinib was safe and improved the clinical conditions in 12 patients with mild-moderate Coronavirus Disease 2019 (COVID-19) pneumonia. It is known that severe symptoms of COVID-19 may develop after a median of 8-days from illness-onset, with a median time to Intensive Care Unit (ICU) admission of 5 days from the dyspnea occurrence.2 Currently, no antiviral therapies or vaccines are available. An uncontrolled immune response3 is observed and is likely involved in tissues injury.4 Baricitinib is an anti-Janus kinase inhibitor-1 and −2, and has a dual action on COVID-19 therapy including the inhibition of cytokine release and SARS-CoV-2 endocytosis.5 Based on this evidence, we conducted an observational, retrospective, longitudinal multicenter-study in consecutive-hospitalized patients with COVID-19 moderate pneumonia to evaluate the 2-week effectiveness and safety of baricitinib combined with antivirals (lopinavir/ritonavir) compared with the standard of care therapy which was hydroxychloroquine and lopinavir/ritonavir. Primary aim was to evaluate the mortality rate; secondary aims were to evaluate the rate of ICU transfer, hospital discharge, improvement of respiratory parameters, adverse events (AEs) occurrence. Moreover, associations between therapy and modification of respiratory parameters and C-Reactive Protein (CRP), interleukin-6 (IL-6), lymphocyte percentage were evaluated.
Clinical charts of patients with moderate COVID-19 pneumonia from 7 Italian hospitals (Hospital of Prato, Hospital of Pistoia, Hospital S.Maria Nuova, Florence, Hospital of Alessandria, Hospital of Fano, Hospital of Pesaro, Hospital of Ariano Irpino (Avellino) were reviewed.
Baricitinib-treated arm included consecutive-hospitalized patients,18 years-older, SARS-CoV-2 naso-pharingeal swab-positive, with a moderate pneumonia characterized by typical symptoms, radiological findings of pneumonia, SpO2 >92% on room air,and PaO2/FiO2 100–300 mmHg, admitted between March 15th-May 5th, 2020. Baricitinib 4 mg/day was provided orally associated with lopinavir/ritonavir tablets 250 mg/bid for 2 weeks.
Control-arm included all consecutive-hospitalized patients from February 20th-March 15th, 2020 with moderate COVID-19 pneumonia, 18 years-older, treated with hydroxychloroquine (HCLR) and lopinavir/ritonavir. Exclusion criteria were: history of thrombophlebitis, latent tuberculosis infection,6 , 7 HBV or HCV infection, current varicella zoster or bacterial infection, pregnancy, lactation, contraceptive pills intake, previous (last 5 years) or current malignancy, neutrophil count<1.0 × 109/L, lymphocyte count<0.2 × 109/L, platelets count<50 × 109/L, transaminases values 4-fold higher than the upper normal limit. Prophylactic anti-thrombotic therapy with low-weight molecular heparin wasadministered. Patients had supportive therapy (O2 supply, rehydration, diuretics, anti-hypertensive, antibiotics) if needed. Corticosteroids were not allowed. If patients were discharged earlier than 2 weeks, they were requested to continue the ongoing therapy until the scheduled 14-days.
Temperature, respiratory and pulse rate, arterial blood gas analysis, blood pressure, blood cell count, liver and kidney tests function were daily assessed. IL-6 serum levels of (RayBioⓇ Human IL-6 ELISA Kit, RayBiotech Co.,USA) were tested at baseline, week 1 and 2. Since low lymphocyte percentage can predict a poor prognosis6, patients were stratified at baseline as: lymphocytes >20%, >5% to <20%, and <5%.
Patients provided a written-informed consent. The off-label use of baricitinib was approved by the Hospital-Committee and Ethical-Committee of Toscana-Region (Code: BARIC-off; 17,261; approval date: May 5th 2020).
Mann-Whitney U test was used to compare quantitative variables; Wilcoxon test for paired data; Chi-squared or Fisher's exact test for categorical variables. A two-tailed p-value <0.05 was considered significant.
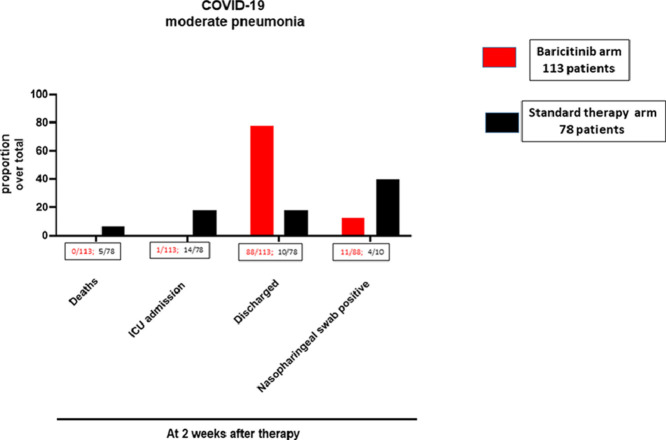
At baseline, 113 patients were in the baricitinib-arm, and 78 in the control-arm (Table 1 ). The results indicate that the 2-week case fatality rate was significantly lower in the baricitinib-arm compared with controls [0% (0/113) vs 6.4% (5/78) (p-value: 0.010; 95%CI 0.0000–0.4569)] (Table 2 ). ICU admission was requested in 0.88% (1/113) vs 17.9% (14/78) patients in the baricitinib-arm compared to the control-arm (week 1, p-value: 0.019; 95%CI 0.0092–0.6818), (week 2, p-value: <0.0001; 95%CI 0.0038–0.2624). Discharge rate was significantly higher in the baricitinib-arm at week 1 [9.7% (11/113) vs 1.3% (1/78) (p-value: 0.039; 95%CI: 1.41–90.71)], and at week 2 [77.8% (88/113) vs 12.8% (10/78) (p:<0.0001; 95%CI 10.79–51.74)].
Table 1.
Baseline demographic, clinical, and laboratory characteristics of COVID-19 patients treated with lopinavir/ritonavir and either baricitinib or with standard of care therapy.
| Feature | Baricitinib Combined with lopinavir/ritonavir treated arm a |
Hydroxychloroquine Combined with lopinavir/ritonavir treated arm (standard of care-treated arm) b |
P value |
|---|---|---|---|
| Patient number, No (%) | 113 (100) | 78 (100) | |
| Male/female, No (%) | 73/40 (64.6/35.4) | 46/32 (59/41) | 0.524 |
| Age years, median (IQR) | 68 (57–76) | 63 (55.5–69.5) | 0.602 |
| Interval from symptoms onset and therapy starting, days, No | 7 (5–10) | 6.5 (4–9) | 0.915 |
| Cough, No (%) | 75/113 (66.4) | 53/78 (67.9) | 0.943 |
| Dyspnea, No (%) | 84/113 (74.3) | 58/78 (74.4) | 0.869 |
| Sputum production, No (%) | 41/113 (36.2) | 29/78 (37.2) | 0.979 |
| Headache, No (%) | 56/113 (49.5) | 31/78 (39.7) | 0.234 |
| Diarrhea, No (%) | 26/113 (23) | 11/78 (14.1) | 0.179 |
| Ageusia/anosmia, No (%) | 44/113 (38.9) | 29/78 (37.1) | 0.925 |
| Hypertension, No (%) | 32/113 (28.3) | 21/78 (26.9) | 0.962 |
| Diabetes, No (%) | 18/113 (15.9) | 13/78 (16.6) | 0.949 |
| COPD, No (%) | 16/113 (14.1) | 14/78 (17.9) | 0.613 |
| CVD, No (%) | 10/113 (8.5) | 5/78 (6.4) | 0.732 |
| Fever °C, median (IQR) | 37.9 (36.7–38.3) | 37.8 (37.7–38.7) | 0.915 |
| Breath rate n/min, median (IQR), | 20 (17–22) | 21 (19–24) | 0.825 |
| SpO2 (%),median (IQR) | 95 (92–98) | 93 (92–97) | 0.234 |
| PaO2/FiO2, median (IQR) | 265.7 (202–330) | 267.5 (263.1–301) | 0.522 |
| Pulse rate, median (IQR) | 80 (70–90) | 85 (79–92) | 0.129 |
| SBP mm/hg, median (IQR) | 125 (112–135) | 115 (105–130) | 0.121 |
| DBP mm/hg, median (IQR) | 70 (65–80) | 65 (60–72) | 0.232 |
| WBC (x109/l), median (IQR) | 6.5 (4.9–8.3) | 7.2 (5.8–8.6) | 0.708 |
| Neutrophils (x109/l), median (IQR) | 4.7 (3.5–6.5) | 4.9 (4.4–6.9) | 0.911 |
| Lymphocytes (x109/l), median (IQR) | 0.93 (0.7–1.2) | 0.88 (0.7–0.9) | 0.728 |
| Lymphocyte percentage | |||
| >20 N (%) | 39 (34.5) | 24 (30.8) | 0.701 |
| >5-<20 | 70 (61.9) | 51 (65.4) | 0.740 |
| <5 | 4 (3.6) | 3 (3.8) | 0.779 |
| Hemoglobin (g/l), median (IQR) | 118 (101–134) | 125 (108–133) | 0.426 |
| Platelets (x109/l), median (IQR) | 213 (167–308) | 268 (156–392) | 0.234 |
| Alt (u/l), median (IQR) | 29 (19–49) | 34 (25–52) | 0.168 |
| Ast (u/l), median (IQR) | 34 (25–51) | 40 (34–47) | 0.628 |
| Creatinine (mg/dl), median (IQR) | 0.9 (0.7–1.1) | 1.00 (0.9–1.2) | 0.925 |
| Crp (mg/dl), median (IQR) | 8.2 (4.1–14.5) | 6.3 (2.8–13.6) | 0.129 |
| IL-6 (PG/ML), MEDIAN (IQR) c | 29.4 (16–45) | 32.6 (25–61) | 0.225 |
| Procalcitonin (ng/ml), median (IQR) | 0.6 (0.3–1.2) | 0.9 (0.8–2.1) | 0.802 |
| Mews, median (IQR) | 1 (0–2) | 2 (1–3) | 0.225 |
Abbreviations and symbols: No= number;%= percentage; °C: grade Celsius; min= minute; SpO2= peripheral capillary oxygen saturation; PaO2/FiO2= ratio of arterial oxygen partial pressure to fractional inspired oxygen; SBP= systolic blood pressure; DBP= diastolic blood pressure; WBC= white blood cells; AST= serum glutamic oxaloacetic transaminase; ALT= serum alanine aminotransferase; MEWS= Modified Early Warning Score; IQR: Interquartile range.
Baricitinib-treated arm: Baricitinib-therapy was given 4 mg/day orally combined with lopinavir/ritonavir tablets 250 mg/bid.
Consecutive patients treated with standard of care therapy (hydroxychloroquine 200 mg/bid with lopinavir/ritonavir tablets 250 mg/bi) during the previous weeks before the first baricitnib-treated patient served as controls.
IL-6 values were available in 58 patients of the baricitinib-treated group and in 36 controls.
Table 2.
Clinical, laboratory and respiratory parameters of COVID-19 patients after 1- or 2-week treatment in the baricitinib-treated group and in the standard-treated group: comparison within the same treatment group and between the 2 different treatment groups.
| Baricitinib combined with lopinavir/ritonavir treated arm 113 patients |
Hydroxychloroquine combined with lopinavir/ritonavir treated arm (Standard of care-treated arm) 78 patients |
Baricitinib-based therapy arm vs standard of care therapy arm |
|||||||
|---|---|---|---|---|---|---|---|---|---|
| Clinical, laboratory, respiratory parameters | Baseline | Week 1 | Week 2 | P value Baseline values vs Week 1 a or vs Week 2 b values |
Baseline | Week 1 | Week 2 | P value Baseline values vs Week 1 a or vs Week 2 b values |
P value Week 1 a Week 2 b |
| Cough, No (%) | 75 (66.4) | 25 (22.1) | 4 (3.5) | 0.000 a 0.000 b |
53 (67.9) | 39 (50) | 15 (19.2) | 0.034 a 0.000 b |
0.000 a 0.014 b |
| Dyspnea, No (%) | 84 (74.3) | 20 (1.7) | 4 (3.5) | 0.000 a 0.000 b |
58 (74.4) | 51 (65.3) | 39 (50) | 0.295 a 0.003 b |
0.000 a 0.000 b |
| Sputum production, No (%) | 41 (36.2) | 15 (13.2) | 10 (8.8) | 0.000 a 0.000 b |
29 (37.2) | 18 (23) | 12 (15.3) | 0.081a 0.004 b |
0.117 a 0.246 b |
| Headache, No (%) | 56 (49.5) | 12 (10.6) | 2 (1.7) | 0.000 a 0.000 b |
31 (39.7) | 24 (30.7) | 10 (12.8) | 0.315 a 0.002 b |
0.000 a 0.005 b |
| Diarrhea, No (%) | 26 (23) | 2 (1.7) | 0 | 0.000 a 0.000 b |
11 (14.1) | 1 (1.3) | 0 | 0.007 a 0.002 b |
0.745 a NA b |
| Ageusia/Anosmia, No (%) | 44 (38.9) | 32 (28.3) | 24 (21.2) | 0.121 a 0.006 b |
29 (37.1) | 25 (32) | 19 (24.3) | 0.614 a 0.118 b |
0.694 a 0.740 b |
| Fever °C, median (IQR) | 38 (37.4–38.2) |
36.1 (36–36.4) |
36 (36–36.1) |
0.001 a 0.001 b |
38 (37.7–38.7) |
37.5 (36.5–38.1) |
37 (36.3–37.4) |
0.516 a 0.129 b |
0.000 a 0.000 b |
| Breath, N/min, median (IQR) | 20 (17–22) |
18 (15–20) |
16 (14–18) |
0.003 a 0.002 b |
21 (19–24) | 19 (18–22) |
17 (16–21.7) |
0.083 a 0.058 b |
0.724 a 0.225 b |
| SpO2%, median (IQR) | 95 (92–98) | 96 (95–98) |
97 (96–98) |
0.191 a 0.0018 b |
93 (92–97) | 93.8 (91.8–94.7) |
93.6 (86.7–94.3) |
0.678 a 0.715 b |
0.000 a 0.000 b |
| PaO2/FiO2 value, median (IQR) | 265.7 (202–330) | 336.5 (245.5–452) |
395 (320–452.3) |
0.0016 a 0.0000 b |
267.5 (263.1–301) | 278.2 (258.3–302.4) |
293.5 (244.2–315.1) |
0.514 a 0.376 b |
0.001 a 0.000 b |
| Pulse rate, No/min. median (IQR) | 80 (70–90) | 76 (68.5–81) |
74.5 (68–80) |
0.433 a 0.177 b |
85 (79–92) | 82.5 (76.5–91) |
83 (77–96.4) |
0.433 a 0.903 b |
0.129 a 0.004 b |
| WBC, x109/L, median (IQR) | 6.5 (4.9–8.3) | 7.0 (5.7–9.2) | 7.0 (5.8–8.6) | 0.481 a 0.225 b |
7.2 (5.8–8.6) | 6.9 (6.5–7.4) |
7.2 (6.4–8.6) |
0.789 a 0.922 b |
0.389 a 0.533 b |
| Neutrophils, x109/L, median (IQR) | 4.7 (3.5–6.5) | 5 (3.3–7) | 4.4 (3.2–6.9) |
0.720 a 0.876 b |
4.9 (4.4–6.9) | 5.4 (5.1–6.2) |
6.7 (6.3–7.3) | 0.054 a 0.002 b |
0.136 a 0.068 b |
| Lymphocytes, x109/L, median (IQR) | 0.93 (0.7–1.2) | 1.11 (0.8–1.9) | 1.3 (1–2.1) | 0.0023 a 0.0017 b |
0.88 (0.7–0.9) | 0.86 (0.5–1) | 0.9 (0.69–1.0) | 0.524 a 0.836 b |
0.023 * 0.004 † |
| Lymphocytes, No (%) | |||||||||
| >20 | 39 (34.5) |
57 (50.4) |
78 (69.0) |
0.022 a 0.000 b |
24 (30.8) | 32 (41.0) |
35 (44.9) | 0.243 a 0.099 b |
0.256 a 0.001 b |
| >5-<20 |
70 (61.9) |
49 (43.4) |
28 (24.8) |
0.008 a 0.000 b |
51 (65.4) | 39 (50.0) | 35 (44.9) | 0.075 a 0.016 b |
0.449 a 0.006 b |
| <5 | 4 (3.6) | 7 (6.2) | 7 (6.2) | 0.536 a 0.536 b |
3 (3.8) | 7 (9.0) | 8 (10.2) | 0.327 a 0.211 b |
0.658 a 0.452 b |
| Hb, g/L, median (IQR) | 118 (101–134) | 120 (106–112) |
125 (118–131) |
0.234 a 0.129 b |
125 (108–133) | 122 (112–125.2) |
124 (114–128) |
0.812 a 0.534 b |
0.925 a 0.746 b |
| Platelets, Nox109/L, median (IQR) | 213 (167–308) | 347 (265–426) |
284 (205–419) |
0.121 a 0.189 b |
268 (156–392) | 328 (321–461) | 359 (316–423) |
0.268 a 0.189 b |
0.786 a 0.144 b |
| ALT, U/L, median (IQR) | 29 (19–49) | 45 (29–68) (43.2–57.2) |
45 (26.7–71 (43–83.7) |
0.065 a 0.076 b |
34 (25–52) | 57.5 (38–69.4) |
54.8 (39.5–54.5) |
0.049 a 0.057 b |
0.533 a 0.144 b |
| AST, U/L, median (IQR) | 34 (25–51) | 36 (25–49.2) |
30 (23–43.5) |
0.965 a 0.764 b |
40 (34–47) | 46.5 (41.7–52.5) |
48.5 (42–55.8) |
0.624 a 0.019 b |
0.076 a 0.129 b |
| Creatinine, mg/dl, median (IQR) | 0.9 (0.7–1.1) | 0.86 (0.69–1.0) |
0.88 (0.7–1.0) |
0.969 a 0.934 b |
1.00 (0.9–1.2) | 1.0 (0.9–1.1) |
1.1 (1–1.2) |
0.956 a 0.783 b |
0.433 a 0.246 b |
| CRP, mg/dl median (IQR) |
8.2 (4.1–14.5) | 0.96 (0.51–2.28) |
0.3 (0.13–0.88) |
0.000 a 0.000 b |
6.3 (2.8–13.6) | 5.4 (2.8–8.9) |
4.6 (2.3–6.4) |
0.871 a 0.433 b |
0.003 a 0.000 b |
| IL-6 (pg/ml), median (IQR) c | 29.4 (16–45) | 5 (2–9) | 2.3 (0–4.2 | 0.0001 a 0.0001 b |
32.6 (25–61) | 29.3 (23–35.2) | 16.3 (12–20.5) | 0.189 a 0.087 b |
0.001 a 0.000 b |
| Procalcitonin, ng/ml, median (IQR) | 0.6 (0.3–1.2) | 0.9 (0.6–1.4) |
0.8 (0.6–1.8) | 0.625 a 0.567 b |
0.9 (0.8–2.1) | 1.3 (0.7–1.9) |
1.2 (0.8–1.4) |
0.278 a 0.124 b |
0.276 a 0.146 b |
| MEWS, median (IQR) | 1 (0–2) | 01 (0–0) |
0 (0–0) |
0.000 a 0.000 b |
2 (1–3) | 1 (1–2) |
1 (0–2) |
0.643 a 0.184 b |
0.004 a 0.012 b |
| ICU transfer, No (%) | 0 | 1 (0.88) d | 0 (0) | 0.000 a NA b |
0 | 6 (7.7) | 14 (17.9) | 0.037 a 0.000 b |
0.019 a 0.000 b |
| Discharged, No (%) |
0 | 11 (9.7) | 88 (77.8) | 0.002 a 0.000 b |
0 | 1 (1.3) | 10 (12.8) | 1.000 a 0.003 b |
0.039 a 0.000 b |
| Positive RT-PCR swabs at discharge, No (%) e | 11 (12.5) | NA | 4 (40) | NA | 0.043 | ||||
| Deaths, No (%) |
0 | 0 | 0 | NA | 0 | 2 (2.6) | 5 (6.4) | 0.477 * 0.069 † |
0.323 a 0.010 b |
Abbreviations and symbols: No: number;%: percentage; °C: grade Celsius; min: minute; SpO2: peripheral capillary oxygen saturation; PaO2/FiO2: ratio of arterial oxygen partial pressure to fractional inspired oxygen; SBP: systolic blood pressure; DBP: diastolic blood pressure; WBC: white blood cells; AST: serum glutamic oxaloacetic transaminase; ALT: serum alanine aminotransferase; IU: international unit; MEWS: Modified Early Warning Score; IQR: Interquartile range. Statistical analysis was performed using the Wilcoxon test (for paired comparisons) or the Mann-Whitney test. P value was considered significant if <0.05.
Differences between the values at baseline and after 1 week.
Differences between the values at baseline and after 2 weeks. Standard of care therapy-treated group: COVID-19 patients under standard respiratory therapy and lopinavir/ritonavir and hydroxychloroquine treatment that were admitted in the hospital the week before starting the therapy with baricitinib and lopinavir/ritonavir.
IL-6 values were available in 58 patients of the baricitinib-treated group and in 36 controls.
This patient required intubation 2 days after baricitinib starting; she remained 2 day in ICU and then she prosecuted the drug. She was discharged 2 days after the treatment completion with baricitinib.
RT-PCR= real time Reverse Transcription-Polymerase Chain Reaction.
Except ageusia/anosmia, all clinical, laboratory and respiratory functions significantly improved at week 1. SpO2 significantly improved at week 2 (p-value: 0.0018); PaO2/FiO2 significantly improved at weeks 1 and 2 compared with baseline-values (p-value: 0.0016 and <0.0001, respectively). Significant differences resulted from the comparison between the baricitinib-arm and the control-group (SpO2, week 1 p-value: <0.0001; week 2 p-value: <0.0001; PaO2/FiO2, week 1 p-value: 0.001; week 2 p-value: <0.0001). CRP and IL-6 levels significantly decreased in the baricitinib-arm (CRP at week 1, p-value: 0.003; at week 2, p-value: <0.0001; IL-6 at week 1, p-value: 0.001; at week 2, p-value: <0.0001). Lymphocytes significantly increased in the baricitinib-arm, with the exception of patients with a baseline proportions-value <5% (Table 2).
At discharge, the proportion of patients [positive to viral naso-pharingeal swabs was significantly lower [12.5% (11/88)] in the baricitinib-arm compared to the control-arm [40% (4/10)] (p-value: 0.043; 95%CI 0.06044–0.7737).
Seven AEs, not requiring the therapy discontinuation, were recorded in the baricitinib-arm, including transaminase increase in 4 (3.5%) patients, epistaxis due to heparin overdose in 1 patient, urinary infection in 1 patient, and oral candidiasis in 1 patient.
The results of the present observational, retrospective, longitudinal, multicenter-study in 113 consecutive-hospitalized patients with moderate pneumonia confirm the effectiveness and safety of baricitinib in patients with moderate COVID-19 pneumonia previously reported.1 Baricitinib-therapy was started in the early phase of COVID-19 disease (median: 7-days from symptoms onset), and the early treatment and the rapid action of the drug may explain the low number of ICU admissions and deaths. The importance of early starting COVID-19 therapies is highlighted in trials of tociluzimab showing a higher efficacy to reduce ICU admission if administered during the initial phase of pneumonia.8 , 9
Interestingly, a significant reduction of positive naso-pharingeal swabs was observed in the baricitinib-arm at discharge, with only 12.5% positive-swabs compared to 40% in the control-group, confirming the anti-inflammatory and anti-viral effects of the baricitinib recently described in 4 patients.10
The short-term administration of the drug compared to the long-term treatment in rheumatoid arthritis, may probably explain the absence of serious AEs.
In conclusion, baricitinib is a promising and safe therapy in patients with moderate COVID-19 pneumonia. A randomized clinical trial is needed to confirm our findings.
Declaration of Competing Interest
All Authors have nothing to disclose.
Acknowledgments
Funding
This work was not supported by any funding.
Acknowledgements
We are grateful to engineers Alessandra Cantini and Marco Bicchi for their assistance in data imputing in the electronic database, and in the statistical analysis.
The authors are grateful to all the patients, nurses and physicians who helped to perform this study.
Footnotes
Collaborators. Vieri Vannucchi MD, Division of Internal Medicine, Azienda USL Toscana Centro, S.Maria Nuova Hospital, Florence; Monica Todoerti, MD, Rheumatology Unit, AO SS. Antonio e Biagio e C. Arrigo, Alessandria; Massimiliano Parodi, MD, Rheumatology Unit, AO SS. Antonio e Biagio e C. Arrigo, Alessandria, Paola Rossi, MD, Rheumatology Unit, AO SS. Antonio e Biagio e C. Arrigo, Alessandria; Piero Davio, MD, Internal Medicine Department, AO SS. Antonio e Biagio e C. Arrigo, Alessandria; Pinuccia Omodeo, MD, Pulmonary Diseases Department, AO SS. Antonio e Biagio e C. Arrigo, Alessandria; Guido Chichino, MD, Infectious Diseases Department, AO SS. Antonio e Biagio e C. Arrigo, Alessandria, Ernesto Cristiano Lauritano, MD, Emergency Department, AO SS. Antonio e Biagio e C. Arrigo, Alessandria; Antonio Maconi, IRFI Infrastructure Research, Education, Innovation, AO SS. Antonio e Biagio e C. Arrigo – Alessandria; Anna Maria Di Carlo, Consultant, Division of Internal Medicine, Azienda Ospedaliera Ospedali RiunitiMarche Nord, Fano; Rita Girolimetti, Consultant, Division of Internal Medicine, Azienda Ospedaliera Ospedali Riuniti Marche Nord, Pesaro; Chiara Pelosi, Consultant, Division of Internal Medicine, Hospital of Ariano Irpino, Avellino; Annamaria Bellizzi, Consultant, Division of Internal Medicine, Hospital of Ariano Irpino, Avellino; Luca Franco, Division of Internal Medicine, Hospital of Ariano Irpino, Avellino; Italy
ClinicalTrials.gov Identifier: NCT04358614.
References
- 1.Cantini F., Niccoli L., Matarrese D., Nicastri E., Stobbione P., Goletti D. Baricitinib therapy in COVID-19: a pilot study on safety and clinical impact. J Infect. 2020 doi: 10.1016/j.jinf.2020.04.017. Apr 23Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sun X., Wang T., Cai D. Cytokine storm intervention in the early stages of COVID-19 pneumonia. Cytokine Growth Factor Rev. 2020 doi: 10.1016/j.cytogfr.2020.04.002. Apr. 25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Adachi T., Chong J.M., Nakajima N. Clinicopathological and immunohistochemical findings from autopsy of patient with COVID-19, Japan. Emerg Infect Dis. 2020;26(9) doi: 10.3201/eid2609.201353. May 15[Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stebbing J., Phelan A., Griffin I., Tucker C., Oechsle O., Richardson P. COVID-19: combining antiviral and anti-inflammatory treatments. Lancet Infect Dis 2020. 2020 doi: 10.1016/51473-3099(20)30132-8. Published online February 27https//doi.org/ [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.QuantiFERONⓇ-TB Gold Plus, ELISA package insert, quiagen. http://www.quantiferon.com/irm/content/PI/QFT/PLUS/2PK-Elisa/UK.pdf.
- 7.Goletti D., Lee M.R., Wang J.Y., Walter N., Ottenhoff T.H.M. Update on tuberculosis biomarkers: from correlates of risk, to correlates of active disease and of cure from disease. Respirology. 2018;23:455–466. doi: 10.1111/resp.13272. [DOI] [PubMed] [Google Scholar]
- 8.Luo P., Liu Y., Qiu L., Liu X., Liu D., Li J. Tocilizumab treatment in COVID-19: a single center experience. J Med Virol. 2020 doi: 10.1002/jmv.25801. Apr 6Online ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Xu X., Han M., Li T., Sun W., Wang D., Fu B. Effective treatment of severe COVID-19 patients with tocilizumab. Proc Natl Acad Sci U S A. 2020 doi: 10.1073/pnas.2005615117. Apr 29pii: 202005615[Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Stebbing J., Krishnan V., DE Bono S. Mechanism of baricitinib supports artificial intelligence‐predicted testing in COVID‐19 patients. EMBO Mol Med. 2020 doi: 10.15252/emmm.202012697. 0. [DOI] [PMC free article] [PubMed] [Google Scholar]