Abstract
No widely accepted surgical technique for subretinal gene replacement therapy delivery in pediatric patients exists. We present alternative approaches in patients with inherited retinal diseases that aim to improve surgical success and minimize complications.
Inherited retinal diseases (IRD) are a group of rare ocular conditions that result in visual impairment either at birth or progressively over time. Gene replacement therapy has demonstrated the most success since the landmark FDA approval of voretigene neparvovec-rzyl (Luxturna®) for treatment of RPE65-related disorders.1,2
No widely accepted technical approach exists for the subretinal delivery of AAV gene therapy in children. Pediatric retinal surgery is accompanied by several unique challenges that may benefit from adjustments in intraoperative techniques.
The protocol developed by Maguire and Russell et al. utilizes a two-surgeon manual injection approach which lead to the approval of voretigene neparvovec-rzyl.3 Additional instrumentation has since been introduced, such as the single-surgeon foot pedal-controlled automated infusion.2 A demonstration of an alternative surgical approach to the subretinal delivery of AAV gene therapy was performed in pediatric eyes with biallelic RPE65 mutations who received voretigene neparvovec-rzyl.
Methods
This study was compliant with the Health Insurance Portability and Accountability Act of 1996, adhered to the tenets of the Declaration of Helsinki, and was approved by the Institutional Review Board of the University of Miami Miller School of Medicine and Children’s Hospital Los Angeles. Written consent was obtained from all patients.
Surgical Technique
Pre-operative doses of oral prednisone were given for 3 days to 1 week with gastric protection. A standard 23 or 25-gauge (G) vitrectomy was performed with ports inserted 3.0-3.5mm behind the limbus in patients 2-years and older. A posterior vitreous detachment was induced over the optic nerve, and if needed, very cautiously over the macula with the assistance of a Finesse Flex Loop® (Alcon, Fort Worth, TX, USA, see Figure 1 and Video Clip 1, available at https://www.ophthalmologyretina.org). Dilute triamcinolone acetonide was used for visualization of the posterior hyaloid. A peripheral vitrectomy was performed along with a depressed examination of the periphery. At the macula, no pre-elevation of the subretinal space was performed with balanced salt solution (BSS). After loading the AAV vector syringe onto a 38G/25G Subretinal PolyTip® Cannula (MedOne, Sarasota, Florida, USA) and purging all air bubbles, the injector was delivered into the subretinal space with a beveled tip (Video Clip 1, available at https://www.ophthalmologyretina.org). Beveling of the cannula tip was performed with scissors at a 45-degree angle, and the cannula entered into the subretinal space in bevel-up fashion. The EVA Phaco-Vitrectomy System (DORC, Dutch Ophthalmic Research Center, Zuidland, The Netherlands) with the viscous fluid control system was used for a single-surgeon, foot pedal controlled linear infusion of the drug. When bleb tension and risks of macular hole formation is deemed too high, more than one bleb is recommended. With more than one extrafoveal bleb, subretinal delivery began nearer to the arcades but included the macula. In all cases, a total of 300 microliters of vector was released (see Figure 2). Subretinal delivery of the vector was confirmed with intraoperative optical coherence tomography (OCT). If coalescence of multiple blebs occurs, OCT is helpful in ensuring no reflux occurs through the retinotomy sites and that the drug vector remains in the subretinal space. Beveling the subretinal cannula is preferred but can cause the appearance of pseudo-schisis, which has been identified as transient hydration of the outer plexiform layers on OCT (see Figure 3, available at https://www.ophthalmologyretina.org). Therefore, beveling or not beveling the cannula is left to the preference of the surgeon. A subsequent air-fluid exchange was then performed. Standard closure was performed followed by a 40mg injection of sub-Tenons or retrobulbar triamcinolone acetonide.
Figure 1.
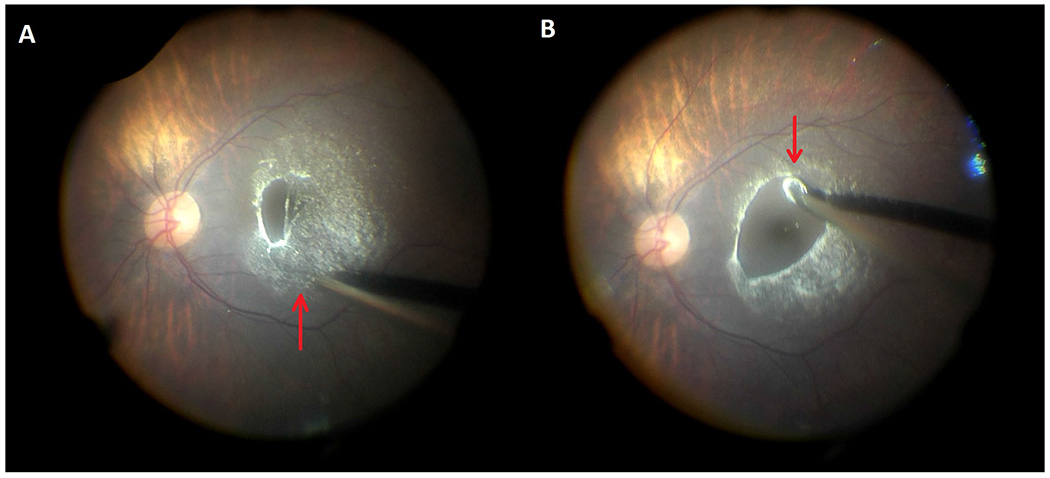
Careful induction of a posterior vitreous detachment with Flex Loop instrumentation over the macula is performed. In (A), the loop size is made larger (red up arrow) to allow for less tension on the instrument and better visualization whereas in (B), the loop size is smaller (red down arrow) to permit for better tractional force and to separate the hyaloid off the macula.
Figure 2.
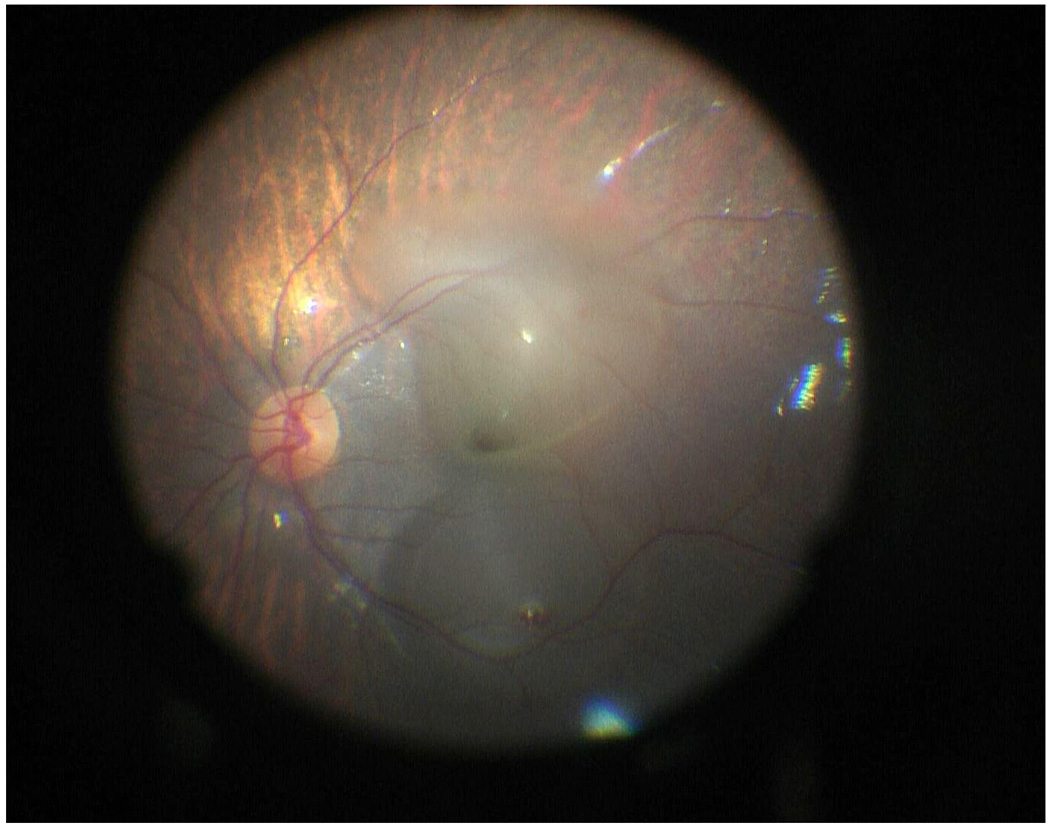
Multiple elevated subretinal blebs with a foveal detachment after injection of gene vector therapy is shown. Initial subretinal injection sites were perimacular.
Figure 3.
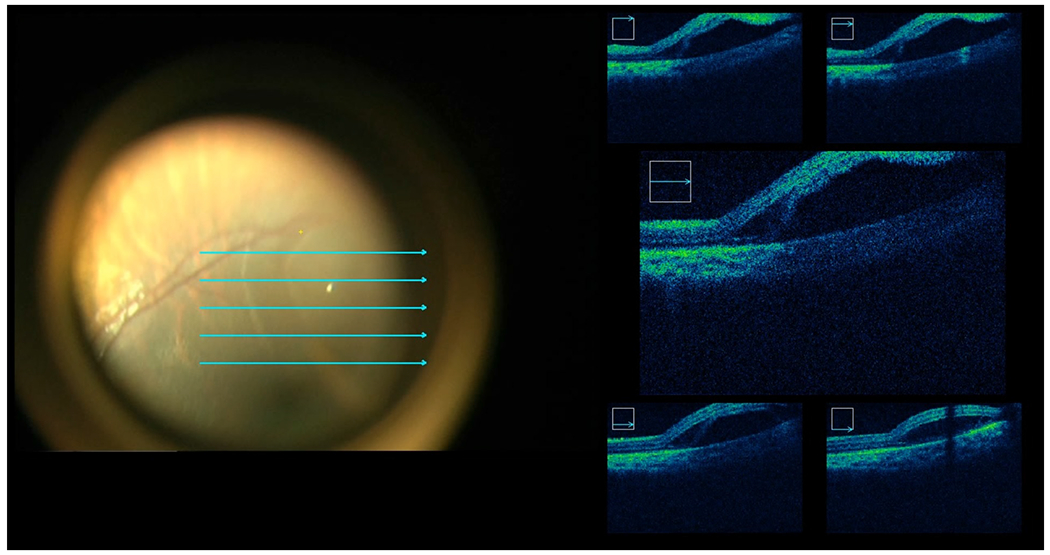
Intraoperative optical coherence tomography (OCT) is shown with confirmation of the delivery of gene vector in the subretinal bleb. Intraoperative OCT images also help confirm findings such as schisis, as well as avoidance of complications like macular holes and multiple or enlarged retinotomy sites.
Discussion
Posterior Vitreous Detachment Induction
In the pediatric patient, induction of a PVD is very challenging. The authors recommend using triamcinolone and initiating a PVD from the optic nerve. When the vitreous is taut, dissection of the hyaloid over the macula can be performed with extreme caution in a radial fashion spreading outward from the central fovea. If the hyaloid behaves more adherently at the macula, the authors suggest use of the Finesse Flex Loop to tease off the vitreous face. Additionally, unlike other instruments, the pressure applied by surgeon and the depth of the instrument loop tip can be easily adjusted and visualized.
Method of Subretinal Delivery
Subretinal injection has become the prevailing method of gene therapy delivery due to poor internal limiting membrane (ILM) penetration of the AAV vector during intravitreal injection.4,5 Differences exist in the technique of the subretinal bleb formation in pediatric patients.
In contrast to the authors’ current practice, some clinical trial protocols recommend using BSS to separate the retina from the RPE before re-entering with drug delivery through the same retinotomy.2,4–6 The 2-step BSS subretinal bleb method creates the scenario for potential trauma to the retinotomy site, enlargement of the first retinotomy with risk of vector reflux, or accidental creation of a second retinotomy. The intended retinotomy created by the 38G injector is self-sealing,4 and thus can be difficult to find and re-enter for a second injection. Additionally, overstretching the retina and inducing a macular hole is one of the surgeon’s greatest concerns in this already tenuous procedure.1,2,4–7 With a single injection, the potential bleb space is filled with only gene vector and therefore stay within critical bleb height. Theoretically, this can reduce the total number of blebs required as well as the rate of complications.
Location of Subretinal Delivery
The precise location for subretinal delivery in the reported literature is subject to some variability. Few guidelines exist; typically, subretinal delivery is performed within the confines of the superior arcade and spares vascular structures.2,7 Though some authors have recommend direct subfoveal detachment with the injection and bleb,2 others warn against foveal detachment due the unpredictable behavior of retina in IRDs (thin overlying retina, and risk of macular hole).2,6 We recommend at least two extrafoveal injections of gene vector within the arcades with or without foveal detachment. The intention is not to explicitly detach the fovea, however it is acceptable if detachment occurs. During this step, the authors use intraoperative OCT over the area of injection and bleb site to gauge the height of the bleb, a crucial step to avoid overstretching the retina.
Conclusion
Surgical approaches for novel treatments in retinal gene therapy for IRDs are constantly evolving. Our proposed surgical techniques eliminate the need for the two-step BSS bleb. In summary, when performing subretinal delivery of AAV vector in pediatric patients, the authors believe the aforementioned techniques may be helpful in maximizing success and minimizing adverse outcomes.
Supplementary Material
Video demonstrating the alternative surgical techniques described in the manuscript regarding the subretinal delivery of gene therapy in pediatric patients. mpeg2 file.
Acknowledgments
Financial Support: The research was supported in part by the National Eye Institute Center Core Grant (P30EY014801) and an unrestricted grant from the Research to Prevent Blindness, Inc., New York, NY to the Department of Ophthalmology, University of Miami Miller School of Medicine. This work was also supported in part by an unrestricted grant to the Department of Ophthalmology at the USC Keck School of Medicine from Research to Prevent Blindness, New York NY (AN), the Las Madrinas Endowment in Experimental Therapeutics for Ophthalmology (AN), and the Knights Templar Eye Foundation (AN).The funding organizations had no role in the design or conduct of this research.
Footnotes
Meeting Presentations: None
Conflicts of Interest: No conflicting relationship exists for any authors.
This article contains a video as additional online-only material. The following should appear online-only: Figure 1, Figure 3, Video Clip 1.
References
- 1.Gupta PR, Huckfeldt RM. Gene therapy for inherited retinal degenerations: initial successes and future challenges. J Neural Eng. 2017;14(5):051002. [DOI] [PubMed] [Google Scholar]
- 2.Xue K, Groppe M, Salvetti AP, MacLaren RE. Technique of retinal gene therapy: delivery of viral vector into the subretinal space. Eye (Lond). 2017;31(9):1308–1316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Russell S, Bennett J, Wellman JA, et al. Efficacy and safety of voretigene neparvovec (AAV2-hRPE65v2) in patients with RPE65-mediated inherited retinal dystrophy: a randomised, controlled, open-label, phase 3 trial. Lancet. 2017;390(10097):849–860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gregori NZ, Lam BL, Davis JL. Intraoperative Use of Microscope-Integrated Optical Coherence Tomography for Subretinal Gene Therapy Delivery. Retina. 2019. [DOI] [PubMed] [Google Scholar]
- 5.Ochakovski GA, Bartz-Schmidt KU, Fischer MD. Retinal Gene Therapy: Surgical Vector Delivery in the Translation to Clinical Trials. Front Neurosci. 2017;11:174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lam BL, Davis JL, Gregori NZ, et al. Choroideremia Gene Therapy Phase 2 Clinical Trial: 24-Month Results. Am J Ophthalmol. 2019;197:65–73. [DOI] [PubMed] [Google Scholar]
- 7.Ong T, Pennesi ME, Birch DG, Lam BL, Tsang SH. Adeno-Associated Viral Gene Therapy for Inherited Retinal Disease. Pharm Res. 2019;36(2):34. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Video demonstrating the alternative surgical techniques described in the manuscript regarding the subretinal delivery of gene therapy in pediatric patients. mpeg2 file.


