Abstract
Background
Renin-angiotensin-aldosterone system (RAAS) inhibitors may facilitate host cell entry of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) or attenuate organ injury via RAAS blockade. We aimed to assess the associations between prior use of RAAS inhibitors and clinical outcomes among Korean patients with coronavirus disease 2019 (COVID-19).
Methods
We performed a nationwide population-based cohort study using the Korean Health Insurance Review and Assessment database. Claim records were screened for 69 793 individuals who were tested for COVID-19 until 8 April 2020. Adjusted odds ratios (ORs) were used to compare the clinical outcomes between RAAS inhibitor users and nonusers.
Results
Among 5179 confirmed COVID-19 cases, 762 patients were RAAS inhibitor users and 4417 patients were nonusers. Relative to nonusers, RAAS inhibitor users were more likely to be older, male, and have comorbidities. Among 1954 hospitalized patients with COVID-19, 377 patients were RAAS inhibitor users, and 1577 patients were nonusers. In-hospital mortality was observed for 33 RAAS inhibitor users (9%) and 51 nonusers (3%) (P < .001). However, after adjustment for age, sex, Charlson comorbidity index, immunosuppression, and hospital type, the use of RAAS inhibitors was not associated with a higher risk of mortality (adjusted OR, 0.88; 95% confidence interval, 0.53–1.44; P = .60). No significant differences were observed between RAAS inhibitor users and nonusers in terms of vasopressor use, modes of ventilation, extracorporeal membrane oxygenation, renal replacement therapy, and acute cardiac events.
Conclusions
Our findings suggest that prior use of RAAS inhibitors was not independently associated with mortality among COVID-19 patients in Korea.
Keywords: ACEI/ARB, comorbidity, COVID-19, renin-angiotensin system, SARS-CoV-2
In-hospital mortality rate was higher among Renin-angiotensin-aldosterone System (RAAS) inhibitor users than nonusers. However, RAAS inhibitor users were also older and more likely to have comorbidities. RAAS inhibitor use was not independently associated with a higher risk of mortality among COVID-19 patients.
(See the Editorial Commentary by Patel and Verma on pages 2129–31.)
In late 2019, several pneumonia cases with an undetermined cause were identified in Wuhan (Hubei province, China), and similar cases were rapidly observed in other Chinese provinces and other countries [1, 2]. On 3 January 2020, a novel coronavirus (severe acute respiratory syndrome coronavirus 2 [SARS-CoV-2]) was isolated from these patients [1], and the corresponding disease was subsequently named coronavirus disease 2019 (COVID-19) by the World Health Organization. Severe cases, including cases that resulted in death, have been observed in older patients and patients with comorbidities, including hypertension, coronary artery disease, diabetes, and chronic kidney disease [3–5].
Both SARS-CoV-1 and SARS-CoV-2 require human angiotensin-converting enzyme 2 (ACE2) to enter the host cell [6, 7]. Interestingly, ACE2 expression is increased in patients with diabetes [8] and after treatment using renin-angiotensin-aldosterone system (RAAS) inhibitors, such as angiotensin-converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs) [9]. Given that mechanism, and the fact that several Chinese studies have identified more severe outcomes in COVID-19 patients with comorbidities that meet the indications for ACE inhibitors and ARBs [3–5], there are concerns regarding the use of these drugs in COVID-19 cases [10]. In addition, SARS-CoV-2 appears to downregulate ACE2 expression, which is a key enzyme that counteracts RAAS by degrading angiotensin II to angiotensin-(1–7) and thereby attenuates organ injury [11]. Blockage of RAAS and increased ACE2 expression attenuated lung injury induced by SARS-CoV in preclinical model [11, 12]. Nevertheless, discontinuation of RAAS inhibitor treatment could lead to clinical deterioration in patients with cardiovascular diseases, and clinical data are lacking regarding the theoretical relationships between RAAS inhibitor use and COVID-19 severity. Recent Chinese studies demonstrated that inpatient use of RAAS inhibitors was not associated with mortality in COVID-19 patients with hypertension [13, 14]. However, prehospital drug use was not evaluated in these studies, thereby questioning adequate exposure to RAAS inhibitors. Therefore, this nationwide population-based cohort study evaluated records from the Korean Health Insurance Review and Assessment (HIRA) database to determine whether prior use of RAAS inhibitors was associated with clinical outcomes among patients with COVID-19.
METHODS
Data Sources and Study Subjects
Limited real-world clinical data are available to physicians and policy makers during the COVID-19 pandemic. Therefore, the Republic of Korea’s government decided to share the world’s first nationwide database of de-identified COVID-19 patient data with domestic and international researchers [15]. The database contains claim records (including for a 5-year period before hospitalization) for 69 793 individuals who were tested for COVID-19 and for whom hospitals issued claims to the HIRA until 8 April 2020. The database includes detailed information regarding demographic characteristics, diagnoses, prescriptions, procedures, and patient outcomes. The diagnostic codes were assigned on the basis of the Korean Classification of Diseases, seventh revision (KCD-7), which is a modified version of the tenth revision of the International Classification of Diseases (ICD-10). Use of drugs was identified using Anatomical Therapeutic Chemical codes and HIRA general name codes. The study protocol for analysis of de-identified patient data was exempted from review by the Institutional Review Board of Chung-Ang University (1041078-202004-HR-082-01).
We included patients with COVID-19 who were ≥ 18 years old. Confirmed COVID-19 cases were identified using the following KCD-7/ICD-10 codes: B34.2 (coronavirus infection, unspecified site), B97.2 (coronavirus as the cause of diseases classified to other chapters), U18 (provisional assignment of new diseases of uncertain etiology or emergency use), U18.1 (novel coronavirus infection), and U07.1 (coronavirus disease 2019 [COVID-19]). These diagnostic codes were given only if a definitive diagnosis of COVID-19 was made based on positive nasopharyngeal swab specimens tested with real-time reverse transcription-polymerase chain reaction assays [16]. The index date was defined as the date of the COVID-19 diagnosis. All patients were followed until the first instance of death or 8 April 2020.
RAAS Inhibitors
This study identified all RAAS inhibitors, including ACE inhibitors and ARBs that were prescribed within 1 year before the index date. For the present study, RAAS inhibitor users were defined as patients with RAAS inhibitor use at 1–30 days before the index date, whereas nonusers were defined as patients who had never received RAAS inhibitors or had received them at 31–365 days before the index date. A prescription duration of ≥ 7 days was required to define drug use. The detailed types and codes for RAAS inhibitors are described in Supplementary Table 1.
Data Collection and Definitions
Comorbidities were defined on the basis of claim codes within 1 year before the index date and evaluated on the basis of the Charlson comorbidity index [17]. The presence of hypertension, which is not included in the Charlson comorbidity index, was separately identified using the ICD-10 codes (see Supplementary Table 2). Data were also collected regarding previous use of steroids (oral or intravenous for ≥ 30 days during the previous year) and immunosuppressants (see Supplementary Table 3). Immunosuppression was identified on the basis of an underlying disease or condition that affected the immune system (eg, human immunodeficiency virus infection or malignancy) or if the patient was receiving immunosuppressive therapy. The type of hospital in which a diagnosis of COVID-19 has been established was defined according to the center’s number of beds and medical specialties (see Supplementary Material).
Data were extracted regarding antibiotics, antivirals, hydroxychloroquine, steroids, and vasopressors that were used during the hospitalization (see Supplementary Tables 3 and 4). In-hospital steroid treatment was defined as only intravenous corticosteroid treatment (methylprednisolone or hydrocortisone). Vasopressor use was defined as the administration of norepinephrine, epinephrine, vasopressin, dopamine, or dobutamine during the hospitalization. Procedure codes were also used to identify cases that involved conventional oxygen therapy, high flow nasal cannula, mechanical ventilation, extracorporeal membrane oxygenation (ECMO), and renal replacement therapy (see Supplementary Table 5).
In-hospital mortality was defined as the primary study outcome. The secondary outcomes included vasopressor use, modes of ventilation, ECMO, renal replacement therapy, and acute cardiac events (cardiac arrest, myocardial infarction, and acute heart failure).
Statistical Analysis
Logistic regression models were used to calculate odds ratios (ORs) and 95% confidence interval (CIs) for in-hospital mortality according to RAAS inhibitor use. The association of RAAS inhibitor use with in-hospital mortality was also assessed in the subgroup of patients with hypertension, given the fact that previous studies have identified more severe outcomes in these patients. Multivariate logistic regression analysis was performed to adjust for age, sex, Charlson comorbidity index, immunosuppression, and hospital type. A separate analysis was also performed to ascertain the effects of in-hospital medication exposures, vasopressor use, and mechanical ventilation. The rate at which excess ACE2 would disappear from host cell membranes after withdrawal of RAAS inhibitors is unclear. Thus, as sensitivity analysis, the patients who received RAAS inhibitors at any time during the 365 days before the index date were analyzed to compare the clinical outcomes between RAAS inhibitor users and nonusers. The associations between RAAS inhibitor use and mortality were assessed on the basis of stratification according to age, sex, Charlson comorbidity index, diabetes, hypertension, myocardial infarction, cerebrovascular disease, chronic pulmonary disease, chronic kidney disease, and immunosuppression. Furthermore, we evaluated whether prior RAAS inhibitor use was significantly associated with any of the secondary outcomes. All tests were 2-tailed, and differences were considered statistically significant at P-values of < .05. All statistical analyses were performed using SAS software (version 9.4; SAS Institute, Cary, NC, USA).
RESULTS
Among the 69 793 individuals who were tested, 5179 patients were confirmed to have COVID-19. These patients with COVID-19 were classified as RAAS inhibitor users (762 patients) or nonusers (4417 patients). In total, 732 users (96%) received ARBs, and 110 patients received RAAS inhibitors at 31–365 days before the index date and were classified as nonusers (see Supplementary Table 6). The patients’ baseline characteristics are described in Table 1. The RAAS inhibitor users were more likely to be older, male, and have the comorbidities listed in Table 1. Moreover, RAAS inhibitor users were more likely to be immunosuppressed.
Table 1.
Baseline Characteristics of the 5179 Patients with COVID-19 According to RAAS Inhibitor Use
| RAAS Inhibitor Status | ||||
|---|---|---|---|---|
| All Patients (n = 5179) | Users (n = 762) | Nonusers (n = 4417) | P | |
| Age, years | <.001 | |||
| Mean (SD) | 44.6 (18.0) | 62.5 (14.7) | 41.5 (16.6) | |
| Range | ||||
| 18–39 | 2357 (46) | 49 (6) | 2308 (52) | |
| 40–59 | 1669 (32) | 269 (35) | 1400 (32) | |
| 60–79 | 947 (18) | 343 (45) | 604 (14) | |
| ≥80 | 206 (4) | 101 (13) | 105 (2) | |
| Sex | <.001 | |||
| Female | 2884 (56) | 362 (48) | 2522 (57) | |
| Male | 2295 (44) | 400 (52) | 1895 (43) | |
| Comorbidities | ||||
| Diabetes | 870 (17) | 366 (48) | 504 (11) | <.001 |
| Hypertension | 1157 (22) | 719 (94) | 438 (10) | <.001 |
| Myocardial infarction | 49 (1) | 27 (4) | 22 (1) | <.001 |
| Congestive heart failure | 225 (4) | 109 (14) | 116 (3) | <.001 |
| Cerebrovascular disease | 333 (6) | 141 (19) | 192 (4) | <.001 |
| Chronic pulmonary disease | 1475 (28) | 304 (40) | 1171 (27) | <.001 |
| Chronic liver disease | 1002 (19) | 269 (35) | 733 (17) | <.001 |
| Chronic kidney disease | 263 (5) | 145 (19) | 118 (3) | <.001 |
| Malignancy | 326 (6) | 77 (10) | 249 (6) | <.001 |
| Charlson comorbidity index | <.001 | |||
| Mean (SD) | 1.5 (2.2) | 3.3 (2.8) | 1.2 (1.9) | |
| Range | ||||
| 0–2 | 4142 (80) | 362 (48) | 3780 (86) | |
| 3–5 | 707 (14) | 253 (33) | 454 (10) | |
| ≥6 | 330 (6) | 147 (19) | 183 (4) | |
| Immunosuppression | 999 (19) | 211 (28) | 788 (18) | <.001 |
Data are shown as number (%) or mean (SD), and P-values were calculated using the χ 2 or Fischer exact test, as appropriate.
Abbreviations: COVID-19, coronavirus disease 2019; RAAS, renin-angiotensin-aldosterone system; SD, standard deviation.
Among the 5179 patients with COVID-19, hospitalization was observed for 1954 patients (38%). Based on the absence of claims data, we assume that the remaining patients stayed at home or received out-of-hospital treatment, such as at community treatment centers. The hospitalized patients included 377 patients who used RAAS inhibitors and 1577 patients who were nonusers. And 365 users (97%) received ARBs, and 40 patients received RAAS inhibitors at 31–365 days before the index date and were classified as nonusers (see Supplementary Table 6). The in-hospital treatments included antibiotics (599 patients, 31%), antivirals (782 patients, 40%), hydroxychloroquine (412 patients, 21%), and intravenous steroids (75 patients, 4%) (see Supplementary Table 7). Each treatment type was more common among RAAS inhibitor users.
The overall mortality rate for the entire cohort was 1.6% (84/5179 patients), and in-hospital mortality was observed for 33/377 RAAS inhibitor users (9%) and for 51/1577 nonusers (3%) (P < .001) (Table 2). The survival curves for the entire cohort and the hospitalized patients are shown in Supplementary Figures 1 and 2. The in-hospital procedures included vasopressor treatment (54 patients, 3%), conventional oxygen therapy (276 patients, 14%), high flow nasal cannula (71 patients, 4%), mechanical ventilation (36 patients, 2%), ECMO (3 patients, 0.2%), and renal replacement therapy (13 patients, 1%) (Table 2). Relative to nonusers, RAAS inhibitor users were more likely to develop cardiac arrest and acute heart failure, and these observations were not influenced when we evaluated subjects who received RAAS inhibitors at any time during the 365 days before the index date (see Supplementary Table 8).
Table 2.
Clinical Outcomes Among the 1954 Hospitalized Patients With COVID-19
| RAAS Inhibitor Status | ||||
|---|---|---|---|---|
| All Patients (n = 1954) | Users (n = 377) | Nonusers (n = 1577) | P | |
| Primary outcome | ||||
| In-hospital mortality | 84 (4) | 33 (9) | 51 (3) | <.001 |
| Secondary outcomes | ||||
| Vasopressor use | 54 (3) | 24 (6) | 30 (2) | <.001 |
| Conventional oxygen therapy | 276 (14) | 94 (25) | 182 (12) | <.001 |
| High flow nasal cannula | 71 (4) | 35 (9) | 36 (2) | <.001 |
| Mechanical ventilation | 36 (2) | 14 (4) | 22 (1) | .003 |
| ECMO | 3 (0.2) | 1 (0.3) | 2 (0.1) | .47 |
| Renal replacement therapy | 13 (1) | 4 (1) | 9 (1) | .29 |
| Acute cardiac events | ||||
| Cardiac arrest | 18 (1) | 8 (2) | 10 (1) | .01 |
| Myocardial infarction | 49 (3) | 7 (2) | 42 (3) | .37 |
| Acute heart failure | 135 (7) | 36 (10) | 99 (6) | .02 |
Data are shown as number (%), and P-values were calculated using the χ 2 or Fischer exact test, as appropriate.
Abbreviations: COVID-19, coronavirus disease 2019; ECMO, extracorporeal membrane oxygenation; RAAS, renin-angiotensin-aldosterone system.
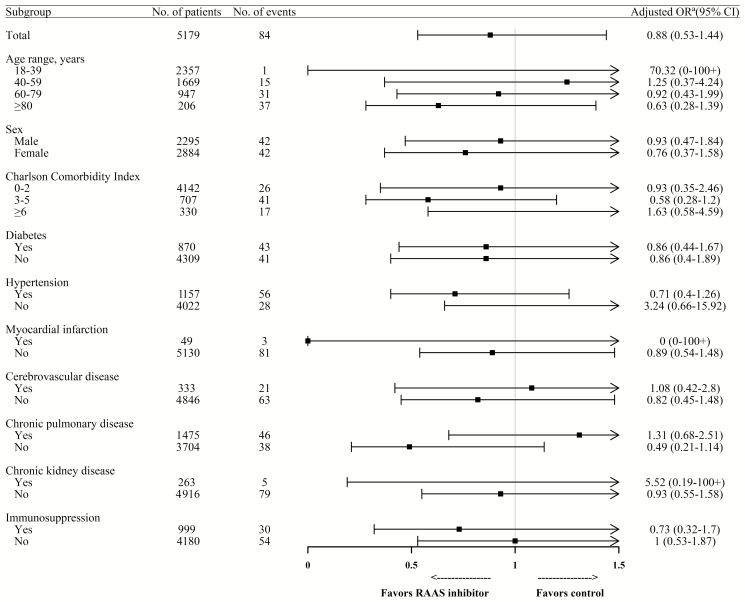
However, multivariate analysis that adjusted for baseline imbalances revealed that RAAS inhibitor use was not independently associated with a higher risk of mortality among COVID-19 patients (adjusted OR, 0.88; 95% CI, .53–1.44; P = .60) (Table 3). This finding was not influenced when we evaluated subjects who received RAAS inhibitors at any time during the 365 days before the index date (see Supplementary Table 9). The analysis that additionally adjusted for in-hospital medications, vasopressor use, and mechanical ventilation was consistent with the overall results (see Supplementary Table 10). The multivariate results were generally consistent across all subgroups (Figure 1), and there were generally no significant differences in the secondary outcomes between the RAAS inhibitor users and nonusers (Table 4).
Table 3.
Crude and Adjusted Odds Ratios for the Risk of In-hospital Mortality According to RAAS Inhibitor Use
| No. of Patients | No. of Events | Unadjusted OR (95% CI) | P | Adjusted ORa (95% CI) | P | |
|---|---|---|---|---|---|---|
| RAAS inhibitor nonusers | 4417 | 51 | 1 (reference) | 1 (reference) | ||
| RAAS inhibitor users | 762 | 33 | 3.88 (2.48–6.05) | <.001 | .88 (.53–1.44) | .60 |
Abbreviations: CI, confidence interval; OR, odds ratio; RAAS, renin-angiotensin-aldosterone system.
aAdjusted for age, sex, Charlson comorbidity index, immunosuppression, and hospital type.
Figure 1.
Forest plot of the association between RAAS inhibitor use and in-hospital mortality according to subgroup. Abbreviations: CI, confidence interval; OR, odds ratio; RAAS, renin-angiotensin-aldosterone system. aAdjusted for age, sex, Charlson comorbidity index, immunosuppression, and hospital type.
Table 4.
Crude and Adjusted Odds Ratios for the Risk of Secondary Outcomes According to RAAS Inhibitor Use
| No. of Patients | No. of Events | Unadjusted OR (95% CI) | P | Adjusted ORa (95% CI) | P | |
|---|---|---|---|---|---|---|
| Vasopressor use | 5179 | 54 | 4.76 (2.77–8.18) | <.001 | 1.33 (.74–2.38) | .34 |
| Conventional oxygen therapy | 5179 | 276 | 3.31 (2.55–4.30) | <.001 | 1.08 (.80–1.45) | .62 |
| High flow nasal cannula | 5179 | 71 | 5.86 (3.66–9.39) | <.001 | 1.78 (1.06–2.99) | .03 |
| Mechanical ventilation | 5179 | 36 | 3.74 (1.91–7.34) | <.001 | 1.03 (.50–2.13) | .93 |
| ECMO | 5179 | 3 | 2.90 (.26–32.03) | .38 | 1.44 (.10–20.06) | .79 |
| Renal replacement therapy | 5179 | 13 | 2.59 (.79–8.41) | .11 | .74 (.21–2.57) | .63 |
| Acute cardiac event | 5179 | 194 | 1.69 (1.19–2.39) | .003 | .88 (.59–1.31) | .53 |
Abbreviations: CI, confidence interval; ECMO, extracorporeal membrane oxygenation; OR, odds ratio; RAAS, renin-angiotensin-aldosterone system.
aAdjusted for age, sex, Charlson comorbidity index, immunosuppression, and hospital type.
Among the 1157 patients with hypertension and COVID-19, there were 719 patients who used RAAS inhibitors and 438 patients who were nonusers. The RAAS inhibitor users were more likely to have diabetes, and nonusers were more likely to have malignancy and be immunosuppressed (see Supplementary Table 11). In-hospital mortality was observed for 31/348 RAAS inhibitor users (9%) and for 25/194 nonusers (13%) (P = .14) (see Supplementary Table 12). Multivariate analysis that adjusted for baseline imbalances revealed that RAAS inhibitor use was not independently associated with a higher risk of mortality among hypertensive COVID-19 patients (adjusted OR, 0.71; 95% CI, .40–1.26; P = .25) (see Supplementary Table 13).
DISCUSSION
The present study revealed a significantly higher mortality rate among patients with COVID-19 who were using RAAS inhibitors, relative to patients who were not receiving RAAS inhibitors. However, RAAS inhibitor users were older, had more comorbidities, and were more likely to receive in-hospital treatments. The elevated risk of mortality among RAAS inhibitor users disappeared after adjusting for these confounding factors.
The use of RAAS inhibitors is thought to upregulate ACE2 expression and facilitate host cell entry of SARS-CoV [6, 7]. Furthermore, ACE2 is predominantly expressed in lung alveolar epithelial cells and in many extrapulmonary tissues, including the heart and kidneys [18]. However, preclinical models have revealed inconsistent findings regarding the effects of RAAS inhibitors on ACE2 expression [9, 19]. Moreover, ACE2 may protect against organ injury by reducing angiotensin II activity, and experimental models have indicated that decreased ACE2 activity may contribute to respiratory virus-induced lung injury [20]. A recent study also suggested that patients with COVID-19 appeared to exhibit RAAS activation, increased viral load levels, and subsequent lung injury [21]. The present study revealed that, relative to nonusers, RAAS inhibitor users had a higher mortality rate and more advanced ventilatory support. Nevertheless, the potential for confounding precludes causal inference regarding the relationship between RAAS inhibitor use and lung injury. In addition, the effects of ARBs on ACE2 expression are inconsistent in animal models [9, 19], whereas ACE inhibitors do not affect directly the activity of ACE2 [22]. Thus, it would be interesting to perform subgroup analysis according to specific RAAS inhibitors. However, our study had a relatively small sample of ACE inhibitor users, relative to ARB users (see Supplementary Table 6), limiting conclusions for the association of ACE inhibitors with mortality. Additional studies are required to define the mechanisms through which RAAS inhibitors modulate ACE2 and exert beneficial or harmful effects among patients with COVID-19.
Three Chinese studies of patients with severe COVID-19 revealed that hypertension, diabetes, and coronary artery disease were the most common comorbidities, and that older age was a risk factor for mortality [3–5]. An Italian cohort of critically ill patients were predominantly older men, and hypertension was the most common comorbidity, which was followed by cardiovascular disorders, hypercholesterolemia, and diabetes [23]. In Korea, the case fatality rate was higher among older men with comorbidities that included cardiovascular disease, diabetes, and neurologic disease [24]. These poor outcomes are likely related to age-dependent defects in immune function and an excessive host response to viral infection [25]. The protective effects of both sex hormones and a second X chromosome, which carries several genes encoding for innate immune molecules, may explain the low susceptibility of female patients to viral infections [26]. Moreover, ACE2 is located on the X chromosome, and there is a possibility that the dosage effect of sex chromosome may impact ACE2 activity due to escape from X-inactivation [27]. The present study revealed that 22% of patients were ≥60 years old, and 44% of patients were men, which are similar to the official statistics from the Korea Centers for Disease Control and Prevention as of 5 April 2020 (≥60 years old: 24%, men: 40%). We did not separately estimate the individual risk factors for mortality, although it would be reasonable to assume that baseline factors (eg, age, sex, and comorbidities) did affect the study outcomes, given that the associations with RAAS inhibitor use disappeared after adjustment for these factors.
In clinical practice, RAAS inhibitors are typically used to treat cardiovascular diseases, diabetes, and chronic kidney disease [28, 29]. Unfortunately, the withdrawal of RAAS inhibitors may result in severe clinical decompensation in patients with heart failure [30]. In addition, myocardial dysfunction has been observed during the course of COVID-19 [31]. Dysregulation of ACE2 may play a role in myocardial injury, as ACE2 knockout in animal models led to angiotensin II-induced injury and adverse left ventricular remodeling [32]. Furthermore, ACE2 expression was reduced in viral RNA-positive heart samples from patients who died of SARS [33]. The present study revealed RAAS inhibitor users to have higher proportions of vasopressor use, cardiac arrest, and acute heart failure, although these relationships were not significant after adjustment for confounding factors. Therefore, our findings suggest that RAAS inhibitors should be continued in COVID-19 patients, especially patients with high risks of heart failure and myocardial infarction. Future studies are needed to evaluate the association between RAAS inhibitor discontinuation and outcomes among COVID-19 patients.
To the best of our knowledge, ours is the first comprehensive study to evaluate whether prior RAAS inhibitor use was associated with outcomes in a nationwide cohort of Asian COVID-19 patients. However, the findings suggest that RAAS inhibitor use was not independently associated with COVID-19 outcomes after adjusting for baseline demographic characteristics, comorbidities, and in-hospital treatments. These data support the recommendations of major medical societies, which suggest continuation of RAAS inhibitors in patients with COVID-19 [34, 35]. Thus, our “real-world” evidence regarding this relationship is crucial, given the concerns regarding ill-advised discontinuation among patients who rely on these drugs [36]. At the time of publication, there have been 3 large retrospective studies investigating the associations between the use of RAAS inhibitors and the risk of COVID-19 or in-hospital mortality in international, Italian, and New York City cohorts [37–39]. Although these studies provide information similar to ours, the evaluation of the associations of RAAS inhibitors with COVID-19-related outcomes in an Asian population is clinically relevant, given that the East Asian populations have higher ACE2 expression in tissues than other populations under similar conditions [40].
The present study has several limitations. First, the accuracy of diagnostic codes may be limited, and there is always a possibility of over- or undercoding. However, the Korean HIRA Service vigorously audits insurance claims, and numerous peer-reviewed publications have been based on HIRA data. It is also unlikely that there were discrepancies between the database disease codes and the actual diseases, as epidemic investigations were performed for all COVID-19 cases. Moreover, RAAS inhibitors are not available as over-the-counter drugs or via self-pay prescriptions in Korea. Second, the retrospective observational design cannot exclude the possibility of residual confounding and precludes causal inference regarding the relationship between RAAS inhibitors and COVID-19-related outcomes. Thus, randomized controlled studies would be needed to establish causality, although it would not likely be feasible to enroll a sufficient number of patients in a short period. Thus, observational data are currently the best available evidence regarding RAAS inhibitor use among patients with COVID-19. Third, database limitations made it impossible to obtain information regarding a potential control group (eg, patients who were not sick enough to be tested for COVID-19). In addition, the database did not include information regarding smoking status and laboratory results. Nevertheless, our primary objective was to evaluate whether RAAS inhibitor use was associated with outcomes among patients with COVID-19. Finally, additional information regarding the cause of death was not available in the database. In this study, the probability of survival dropped abruptly in the first few days for the entire cohort and the hospitalized patients. It is possible that early mortality may be related to acute cardiac event or acute pulmonary embolism rather than COVID-19-related respiratory disease.
In conclusion, our results indicate that prior use of RAAS inhibitors was not independently associated with mortality among Korean patients with COVID-19. However, controversy regarding the role of RAAS blockade in COVID-19 cases calls for urgent multicenter trials.
Supplementary Data
Supplementary materials are available at Clinical Infectious Diseases online. Consisting of data provided by the authors to benefit the reader, the posted materials are not copyedited and are the sole responsibility of the authors, so questions or comments should be addressed to the corresponding author.
Notes
Author contributions. S. Y. J. participated in the data acquisition, data analysis and interpretation, and drafting of the manuscript. J. C. C. participated in the data interpretation and helped to revise the manuscript for important intellectual content. S. H. Y. participated in the data acquisition and data analysis and interpretation. W. Y. K. participated in the study’s conception and design, data interpretation, and drafting of the manuscript. All authors read and approved the final manuscript.
Acknowledgments. The authors thank the healthcare professionals dedicated to treating COVID-19 patients in Korea, and the Ministry of Health and Welfare and the Health Insurance Review and Assessment Service of Korea for sharing invaluable national health insurance claims data in a prompt manner.
Potential conflicts of interest. The authors: No reported conflicts of interest. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest.
References
- 1. Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med 2020; 382:727–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Holshue ML, DeBolt C, Lindquist S, et al. First case of 2019 novel coronavirus in the United States. N Engl J Med 2020; 382:929–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020; 382:1708–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020; 395:1054–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Du Y, Tu L, Zhu P, et al. Clinical features of 85 fatal cases of COVID-19 from Wuhan: a retrospective observational study [manuscript published online ahead of print 3 Apr 2020]. Am J Respir Crit Care Med 2020. doi: 10.1164/rccm.202003-0543OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Li W, Moore MJ, Vasilieva N, et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 2003; 426:450–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Hoffmann M, Kleine-Weber H, Schroeder S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020; 181:271–280.e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Wan Y, Shang J, Graham R, Baric RS, Li F. Receptor recognition by the novel coronavirus from Wuhan: an analysis based on decade-long structural studies of SARS coronavirus. J Virol 2020; 94:e00127-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Ferrario CM, Jessup J, Chappell MC, et al. Effect of angiotensin-converting enzyme inhibition and angiotensin II receptor blockers on cardiac angiotensin-converting enzyme 2. Circulation 2005; 111:2605–10. [DOI] [PubMed] [Google Scholar]
- 10. Fang L, Karakiulakis G, Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir Med 2020; 8:e21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Kuba K, Imai Y, Rao S, et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat Med 2005; 11:875–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Imai Y, Kuba K, Rao S, et al. Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature 2005; 436:112–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Zhang P, Zhu L, Cai J, et al. Association of inpatient use of angiotensin converting enzyme inhibitors and angiotensin II receptor blockers with mortality among patients with hypertension hospitalized with COVID-19 [manuscript published online ahead of print 17 Apr 2020]. Circ Res 2020. doi: 10.1161/CIRCRESAHA.120.317134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Li J, Wang X, Chen J, Zhang H, Deng A. Association of renin-angiotensin system inhibitors with severity or risk of death in patients with hypertension hospitalized for coronavirus disease 2019 (COVID-19) infection in Wuhan, China [manuscript published online ahead of print 23 Apr 2020]. JAMA Cardiol 2020. doi: 10.1001/jamacardio.2020.1624 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Ministry of Health and Welfare, Health Insurance Review and Assessment Service. #opendata4covid19 Available at: https://covid19data.hira.or.kr. Accessed 20 April 2020.
- 16. Corman VM, Landt O, Kaiser M, et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill 2020; 25:2000045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987; 40:373–83. [DOI] [PubMed] [Google Scholar]
- 18. Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus: a first step in understanding SARS pathogenesis. J Pathol 2004; 203:631–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Burchill LJ, Velkoska E, Dean RG, Griggs K, Patel SK, Burrell LM. Combination renin-angiotensin system blockade and angiotensin-converting enzyme 2 in experimental myocardial infarction: implications for future therapeutic directions. Clin Sci (Lond) 2012; 123:649–58. [DOI] [PubMed] [Google Scholar]
- 20. Zou Z, Yan Y, Shu Y, et al. Angiotensin-converting enzyme 2 protects from lethal avian influenza A H5N1 infections. Nat Commun 2014; 5:3594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Liu Y, Yang Y, Zhang C, et al. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci 2020; 63:364–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Rice GI, Thomas DA, Grant PJ, Turner AJ, Hooper NM. Evaluation of angiotensin-converting enzyme (ACE), its homologue ACE2 and neprilysin in angiotensin peptide metabolism. Biochem J 2004; 383:45–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Grasselli G, Zangrillo A, Zanella A, et al. Baseline characteristics and outcomes of 1591 patients infected with SARS-CoV-2 admitted to ICUs of the Lombardy Region, Italy [manuscript published online ahead of print 6 Apr 2020]. JAMA 2020. doi: 10.1001/jama.2020.5394 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Korean Society of Infectious Diseases and Korea Centers for Disease Control and Prevention. Analysis on 54 mortality cases of coronavirus disease 2019 in the Republic of Korea from January 19 to March 10, 2020. J Korean Med Sci 2020; 35:e132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Opal SM, Girard TD, Ely EW. The immunopathogenesis of sepsis in elderly patients. Clin Infect Dis 2005; 41 Suppl 7:S504–12. [DOI] [PubMed] [Google Scholar]
- 26. Jaillon S, Berthenet K, Garlanda C. Sexual dimorphism in innate immunity. Clin Rev Allergy Immunol 2019; 56:308–21. [DOI] [PubMed] [Google Scholar]
- 27. Liu J, Ji H, Zheng W, et al. Sex differences in renal angiotensin converting enzyme 2 (ACE2) activity are 17β-oestradiol-dependent and sex chromosome-independent. Biol Sex Differ 2010; 1:6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American college of cardiology/American heart association task force on clinical practice guidelines. Hypertension 2018; 71:e13–e115. [DOI] [PubMed] [Google Scholar]
- 29. Ruggenenti P, Cravedi P, Remuzzi G. The RAAS in the pathogenesis and treatment of diabetic nephropathy. Nat Rev Nephrol 2010; 6:319–30. [DOI] [PubMed] [Google Scholar]
- 30. Halliday BP, Wassall R, Lota AS, et al. Withdrawal of pharmacological treatment for heart failure in patients with recovered dilated cardiomyopathy (TRED-HF): an open-label, pilot, randomised trial. Lancet 2019; 393:61–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Shi S, Qin M, Shen B, et al. Association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan, China [manuscript published online ahead of print 25 Mar 2020]. JAMA Cardiol 2020. doi: 10.1001/jamacardio.2020.0950 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Kassiri Z, Zhong J, Guo D, et al. Loss of angiotensin-converting enzyme 2 accelerates maladaptive left ventricular remodeling in response to myocardial infarction. Circ Heart Fail 2009; 2:446–55. [DOI] [PubMed] [Google Scholar]
- 33. Oudit GY, Kassiri Z, Jiang C, et al. SARS-coronavirus modulation of myocardial ACE2 expression and inflammation in patients with SARS. Eur J Clin Invest 2009; 39:618–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. American College of Cardiology. HFSA/ACC/AHA statement addresses concerns re: using RAAS antagonists in COVID-19 March 17, 2020. Available at: https://www.acc.org/latest-in-cardiology/articles/2020/03/17/08/59/hfsa-acc-aha-statement-addresses-concerns-re-using-raas-antagonists-in-covid-19. Accessed 6 May 2020.
- 35. European Society of Cardiology. Position statement of the ESC Council on Hypertension on ACE-inhibitors and angiotensin receptor blockers March 13, 2020. Available at: https://www.escardio.org/Councils/Council-on-Hypertension-(CHT)/News/position-statement-of-the-esc-council-on-hypertension-on-ace-inhibitors-and-ang. Accessed 6 May 2020. [DOI] [PubMed]
- 36. Tignanelli CJ, Ingraham NE, Sparks MA, et al. Antihypertensive drugs and risk of COVID-19? [manuscript published online ahead of print 26 Mar 2020]. Lancet Respir Med 2020. doi: 10.1016/S2213-2600(20)30153-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Mehra MR, Desai SS, Kuy S, Henry TD, Patel AN. Cardiovascular disease, drug therapy, and mortality in Covid-19 [manuscript published online ahead of print 1 May 2020]. N Engl J Med 2020. doi: 10.1056/NEJMoa2007621 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 38. Mancia G, Rea F, Ludergnani M, Apolone G, Corrao G. Renin-angiotensin-aldosterone system blockers and the risk of Covid-19 [manuscript published online ahead of print 1 May 2020]. N Engl J Med 2020. doi: 10.1056/NEJMoa2006923 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Reynolds HR, Adhikari S, Pulgarin C, et al. Renin-angiotensin-aldosterone system inhibitors and risk of Covid-19 [manuscript published online ahead of print 1 May 2020]. N Engl J Med 2020. doi: 10.1056/NEJMoa2008975 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Cao Y, Li L, Feng Z, et al. Comparative genetic analysis of the novel coronavirus (2019-nCoV/SARS-CoV-2) receptor ACE2 in different populations. Cell Discov 2020; 6:11. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.