Abstract
Background
Several guidelines to guide clinical practice among esophagogastric surgeons during the COVID-19 pandemic were produced. However, none provide reflection of current service provision. This international survey aimed to clarify the changes observed in esophageal and gastric cancer management and surgery during the COVID-19 pandemic.
Methods
An online survey covering key areas for esophagogastric cancer services, including staging investigations and oncological and surgical therapy before and during (at two separate time-points—24th March 2020 and 18th April 2020) the COVID-19 pandemic were developed.
Results
A total of 234 respondents from 225 centers and 49 countries spanning six continents completed the first round of the online survey, of which 79% (n = 184) completed round 2. There was variation in the availability of staging investigations ranging from 26.5% for endoscopic ultrasound to 62.8% for spiral computed tomography scan. Definitive chemoradiotherapy was offered in 14.8% (adenocarcinoma) and 47.0% (squamous cell carcinoma) of respondents and significantly increased by almost three-fold and two-fold, respectively, in both round 1 and 2. There were uncertainty and heterogeneity surrounding prioritization of patients undergoing cancer resections. Of the surgeons symptomatic with COVID-19, only 40.2% (33/82) had routine access to COVID-19 polymerase chain reaction testing for staff. Of those who had testing available (n = 33), only 12.1% (4/33) had tested positive.
Conclusions
These data highlight management challenges and several practice variations in caring for patients with esophagogastric cancers. Therefore, there is a need for clear consistent guidelines to be in place in the event of a further pandemic to ensure a standardized level of oncological care for patients with esophagogastric cancers.
Keywords: COVID-19, esophageal cancer, esophageal surgery, gastric cancer, pandemic, SARS-CoV-2
INTRODUCTION
The World Health Organization declared a pandemic of coronavirus disease SARS-CoV-2 (COVID-19) on 11 March 2020.1 Rapid spread of COVID-19 heavily affected healthcare systems worldwide, which led to vast and widespread changes in hospitals’ structure to plan for increased intensive care unit (ICU) capacity to cope with the pandemic.2 As a result, this pandemic not only affected COVID-19 patients, but also impacted the entire healthcare system including the provision of care for patients with cancer and surgery.3,4
Recent reports suggest an increased risk for cancer patients to develop severe complications when infected by COVID-19, with a similar pattern observed for those treated with surgery or chemotherapy.5 Pursuing oncological care exposes both healthcare professionals and patients to become infected by COVID-19. However, the inability to receive oncological and/or surgical care seems to be an equally important prognostic threat for cancer patients.6 The aggressive disease biology of esophageal and gastric cancer requires the continuation of oncological therapy during the COVID-19 pandemic.7 However, thoughtful strategies need to be developed to support healthcare professionals in clinical decision-making and ensure resources are allocated appropriately.8
This international survey aimed to clarify the changes observed in esophageal and gastric cancer management during the COVID-19 pandemic. This was performed by surveying international specialist surgeons with a focus on how the pandemic has affected their cancer services and how they have adapted their service with respect to: (1) cancer staging pathways, (2) multidisciplinary team (MDT)/tumor board meetings, (3) oncological therapy, (4) operative intervention, (5) preoperative COVID-19 testing and (6) effect of the pandemic on esophagogastric cancer surgeons and redeployment.
METHODS
Survey
An online survey was developed through a consensus process involving a core group of investigators (Appendix I). The questionnaire was designed to cover a range of key areas for esophageal and gastric cancer, including staging investigations and oncological and surgical therapy before and during (at two separate time-points) the COVID-19 pandemic. This survey was circulated to all members of the international esophageal and gastric associations and study groups: Australian and New Zealand Gastric and Oesophageal Surgery Association, Association of Upper Gastrointestinal Surgeons (AUGIS), Dutch Cancer Audit, European Society of Diseases of Esophagus (ESDE), Gastric Cancer Surgery Trials Reported Outcomes Standardisation (GASTROS), International Society for Disease of Esophagus and Oesophagogastric Anastomosis Audit using Google Forms (Google LLC, Menlo Park, CA, USA). Only consultant or attending surgeons performing esophageal and gastric resections were invited to complete the online survey via email.
The survey consisted of two rounds with 40 questions (Appendix II and III) on baseline characteristics, the local impact of the COVID-19 pandemic on esophageal and gastric surgery (i.e. MDT meeting, availability of staging investigations, choice of oncological therapy, surgical resection and ICU capacity) and statements about prioritization of resection in the current era of the COVID-19 pandemic to gain a consensus. Each statement had to be appraised using the ‘prioritization of patients for cancer surgery’9 from the National Health Service England options: priority level 1 (curative therapy with a high [>50%] chance of success), priority level 2 (curative therapy with an intermediate [15–50%] chance of success), priority level 3 (noncurative therapy with a high [>50%] chance of >1 year of life extension), priority level 4 (curative therapy with a low [0–15%] chance of success or noncurative therapy with an intermediate [15–50%] chance of >1 year life extension), priority level 5 (noncurative therapy with a high [>50%] chance of palliation/temporary tumor control, but <1 year life extension) and priority level 6 (noncurative therapy with an intermediate [15–50%] chance of palliation or temporary tumor control and <1 year life extension).
The first round of the survey was conducted in the last week of March 2020 (Fig. 1A) and the second round was repeated in the last 2 weeks of April 2020 (Fig. 1B). Nonrespondents were sent two reminder emails. Respondents were asked to register their name and institution to prevent overlap of members between the same hospitals.
Fig. 1.
Global map demonstrating countries participating in round 1 and 2 of the survey and total COVID-19 cases per capita globally. (A) Total COVID-19 incidence on 24th March 2020. (B) Total COVID-19 incidence on 18th April 2020. *These international maps demonstrating total COVID-19 confirmed cases per capita were obtained from the Our World in Data from https://ourworldindata.org/coronavirus-data.
Definitions
Critical care capacity was defined using the CRITCON level, which was originally developed in 2009 by the North West London Critical Care Network in response to the H1N1 influenza pandemic.10 This grades ICU capacity into the following levels (CRITCON 0–4, corresponding to ‘business as usual’, ‘normal winter’, ‘unprecedented’, ‘last resort’ and ‘triage’). Data for total confirmed cases per capita were derived from the ‘Our World in Data’,11 which is a platform for global data on a broad range of conditions. Data on total confirmed COVID-19 cases per capita were grouped into tertiles (i.e. low [L-CoV], medium [M-CoV] and high [H-CoV]) derived from the database.
Statistical analysis
Data analysis was performed using R Foundation Statistical software (R 3.2.2) (R Foundation for Statistical Computing, Vienna, Austria). Data were reported as number with percentage or as median with interquartile range. Categorical variables were compared using the chi-squared test. Non-normally distributed data were analyzed using the Mann–Whitney U test. Stratified sensitivity analyses were performed based on total confirmed cases per capita for each country on 24th March 2020 (round 1) and 18th April 2020 (round 2). All statistical significance was considered as two-tailed P-value <0.05.
RESULTS
Demographics of survey participants and centers
A total of 234 respondents from 225 centers and 49 countries spanning six continents completed the first round of the online survey (Supplementary data, Fig. S1). Baseline demographics of respondents are presented in Supplementary data, Table S1. Most respondents were upper gastrointestinal or esophagogastric surgeons (n = 158, 67.5%) working in an academic center (n = 217, 92.7%). The majority of centers had 501–1,000 hospital beds (n = 91, 38.9%) and 36.8% (n = 86) of centers had 20 to 50 hospital ICU beds. In round 1, 81.6% had patients in their center with COVID-19 and 29.9% of centers had >51 inpatients with COVID-19. Majority of centers were at CRITCON level 2 ‘unprecedented’ (n = 96, 41.0%), followed by CRITCON level 1 ‘normal winter’ (n = 64, 27.4%) and CRITCON level 0 ‘business as usual’ (n = 38, 16.2%). Baseline characteristics of COVID-19-related demographics are presented in Table 1. Of the 234 respondents from round 1, 184 respondents (79%) completed round 2 of the survey as presented in Supplementary data, Table S1.
Table 1.
Baseline characteristics of COVID-19, cancer protocols, and multidisciplinary team among respondents stratified in round 1 (n = 234) and 2 (n = 184) of the survey
| Round 1 | Round 2 | P-value | ||
|---|---|---|---|---|
| COVID-19 | ||||
| COVID-19 patients | No | 34 (14.5) | 29 (15.8) | 0.2 |
| Yes | 191 (81.6) | 153 (83.2) | ||
| Unknown | 9 (3.8) | 2 (1.1) | ||
| Number of COVID-19 patients | 0–10 | 67 (28.6) | 40 (21.7) | <0.001 |
| 11–20 | 33 (14.1) | 10 (5.4) | ||
| 21–30 | 19 (8.1) | 10 (5.4) | ||
| 31–40 | 18 (7.7) | 10 (5.4) | ||
| 41–50 | 8 (3.4) | 49 (26.6) | ||
| >51 | 70 (29.9) | 59 (32.0) | ||
| Unknown | 19 (8.1) | 6 (3.2) | ||
| CRITCON | Level 0 (normal) | 38 (16.2) | 22 (12.0) | 0.5 |
| Level 1 (bad winter) | 64 (27.4) | 52 (28.3) | ||
| Level 2 (unprecedented) | 96 (41.0) | 87 (47.3) | ||
| Level 3 (full stretch) | 30 (12.8) | 21 (11.4) | ||
| Level 4 (last resort) | 6 (2.6) | 2 (1.1) | ||
| Cancer protocols and MDT | ||||
| Protocol | No | 29 (12.4) | 16 (8.7) | 0.5 |
| Yes—local | 115 (49.1) | 89 (48.4) | ||
| Yes—national | 56 (23.9) | 53 (28.8) | ||
| Yes—local and national | 34 (14.5) | 26 (14.1) | ||
| MDT | No—limited numbers in the room | 87 (37.2) | 51 (27.7) | 0.1 |
| No—they have had to be stopped | 38 (16.2) | 26 (14.1) | ||
| No—virtual MDT (video linkage) | 85 (36.3) | 89 (48.4) | ||
| Yes—face-to-face meetings | 24 (10.3) | 18 (9.8) | ||
| Staging modalities | ||||
| Diagnostic endoscopy | Available | 70 (29.9) | 80 (43.5) | 0.015 |
| Limited or delayed availability | 142 (60.7) | 92 (50.0) | ||
| Unavailable during COVID-19 pandemic | 22 (9.4) | 12 (6.5) | ||
| Therapeutic endoscopy (EMR/ESD/stenting) | Available | 78 (33.3) | 75 (40.8) | 0.2 |
| Limited or delayed availability | 134 (57.3) | 89 (48.4) | ||
| Unavailable during COVID-19 pandemic | 22 (9.4) | 20 (10.9) | ||
| Spiral computed tomography scan | Available | 147 (62.8) | 144 (78.3) | 0.002 |
| Limited or delayed availability | 82 (35.0) | 39 (21.2) | ||
| Unavailable during COVID-19 pandemic | 5 (2.1) | 1 (0.5) | ||
| Endoscopic ultrasound | Available | 62 (26.5) | 52 (28.3) | 0.7 |
| Limited or delayed availability | 118 (50.4) | 96 (52.2) | ||
| Unavailable during COVID-19 pandemic | 54 (23.1) | 36 (19.6) | ||
| Position emission topography | Available | 116 (49.6) | 119 (64.7) | 0.006 |
| Limited or delayed availability | 86 (36.8) | 43 (23.4) | ||
| Unavailable during COVID-19 pandemic | 32 (13.7) | 22 (12.0) | ||
| Staging laparoscopy | Available | 91 (38.9) | 85 (46.2) | 0.2 |
| Limited or delayed availability | 96 (41.0) | 61 (33.2) | ||
| Unavailable during COVID-19 pandemic | 47 (20.1) | 38 (20.7) | ||
| Cancer surgery | ||||
| Prioritization | No | 39 (16.7) | 26 (14.1) | 0.5 |
| Yes—local protocol | 149 (63.7) | 114 (62.0) | ||
| Yes—national protocol | 46 (19.7) | 44 (23.9) | ||
| Resection | Consultants are assisted by other consultants (dual operating) | 94 (40.2) | 95 (51.6) | 0.002 |
| Consultants are assisted by trainees | 105 (44.9) | 78 (42.4) | ||
| Consultants are training trainees | 9 (3.8) | 7 (3.8) | ||
| Resectional surgery has stopped | 26 (11.1) | 4 (2.2) | ||
| Postoperative management | ||||
| Postoperative ICU | Never | 15 (6.4) | 13 (7.1) | 0.3 |
| Routinely | 106 (45.3) | 96 (52.2) | ||
| Selectively | 113 (48.3) | 75 (40.8) | ||
| Postoperative destination changes during COVID-19 | No | 61 (26.1) | 80 (43.5) | <0.001 |
| Yes—consider operating or send patients to a different hospital | 6 (2.6) | 11 (6.0) | ||
| Yes—consider or send patients back to the ward or alternatives to ICU | 124 (53.0) | 77 (41.8) | ||
| Yes—operative surgery will be extremely rare | 43 (18.4) | 16 (8.7) | ||
| Perceived morbidity with COVID-19 in esophagogastric cancer patients | Higher morbidity, but similar mortality | 31 (13.2) | 23 (12.5) | 0.7 |
| Higher mortality | 178 (76.1) | 142 (77.2) | ||
| Similar risks to usual | 9 (3.8) | 10 (5.4) | ||
| Unknown | 16 (6.8) | 9 (4.9) | ||
| Estimated postoperative COVID-19 infections in esophagogastric cancer patients* | 0% | — | 56 (30.4) | — |
| 1–10% | — | 32 (17.4) | ||
| >10% | — | 14 (7.6) | ||
| Unknown | — | 82 (44.6) | ||
| Estimated major complications associated with COVID-19 infections in esophagogastric cancer patients* | 0% | — | 51 (27.7) | — |
| 1–10% | — | 20 (10.9) | ||
| >10% | — | 35 (19.0) | ||
| Unknown | — | 78 (42.4) | ||
| Estimated 30-day mortality associated with COVID-19 infections in esophagogastric cancer patients* | 0% | — | 53 (28.8) | — |
| 1–10% | — | 20 (10.9) | ||
| >10% | — | 33 (17.9) | ||
| Unknown | — | 78 (42.4) | ||
*These questions were only asked in round 2 of the survey.
Cancer protocols and MDT
In round 1, 88.6% of centers had standardized protocols available for the treatment of esophagogastric cancer patients during COVID-19 and these increased to 92.3% in round 2 (Table 1). Protocols were based on national guidelines in 23.9% and 28.8% of centers in round 1 and 2 respectively. There was a substantial increase in virtual MDT meetings from 36.3% to 48.4% from round 1 to 2, respectively (Table 1). In stratified analyses, rates of virtual MDT among centers were significantly higher in countries with M-CoV and H-CoV incidence cases compared to L-CoV incidence cases in both round 1 (47.4% vs. 37.7% vs. 24.1%, P < 0.001) and 2 (49.4% vs. 51.2% vs. 34.8%, P < 0.001) (Supplementary data, Table S2).
Staging investigations
There was variation in the availability of staging investigations in round 1 ranging from 26.5% for endoscopic ultrasound to 62.8% for spiral computed tomography (CT) scan (Fig. 2). Round 2 demonstrated significant increases in rates of diagnostic endoscopy (29.9% vs. 43.5%, P = 0.015), spiral CT scan (62.8% vs. 78.3%, P = 0.002) and position emission topography scan (49.6% vs. 64.7%, P = 0.006) (Fig. 2; Table 1) in comparison to round 1. Limited variations were observed in stratified analyses by total COVID-19 cases between rounds (Supplementary data, Table S3).
Fig. 2.
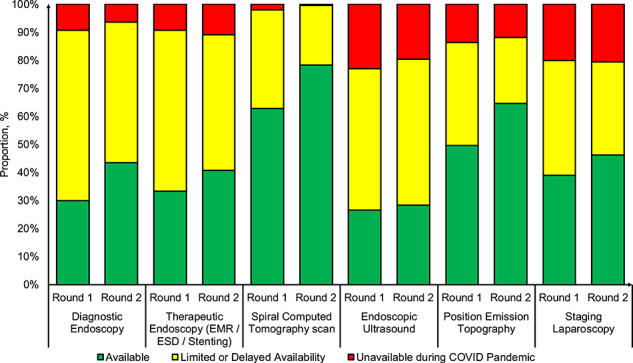
Distribution of staging investigations availability across centers from round 1 and 2 of the survey during the COVID-19 pandemic.
Oncological therapy
Esophageal adenocarcinoma
Standard pre-COVID-19 oncological therapies available for potentially curative esophageal adenocarcinoma included neoadjuvant chemotherapy (NCS) (n = 163, 69.1%) or chemoradiotherapy (NCRS) (n = 152, 64.4%) (Fig. 3A). Definitive chemoradiotherapy (DCRT) was offered in 14.8% of centers and significantly increased during the COVID-19 pandemic by almost three-fold in both round 1 and 2 (39.7% and 38.0%, P < 0.001). Stratified analyses demonstrated higher rates of adoption of DCRT in H-CoV centers in both round 1 and 2 compared to standard for esophageal adenocarcinoma (Supplementary data, Fig. S2, Table S4).
Fig. 3.
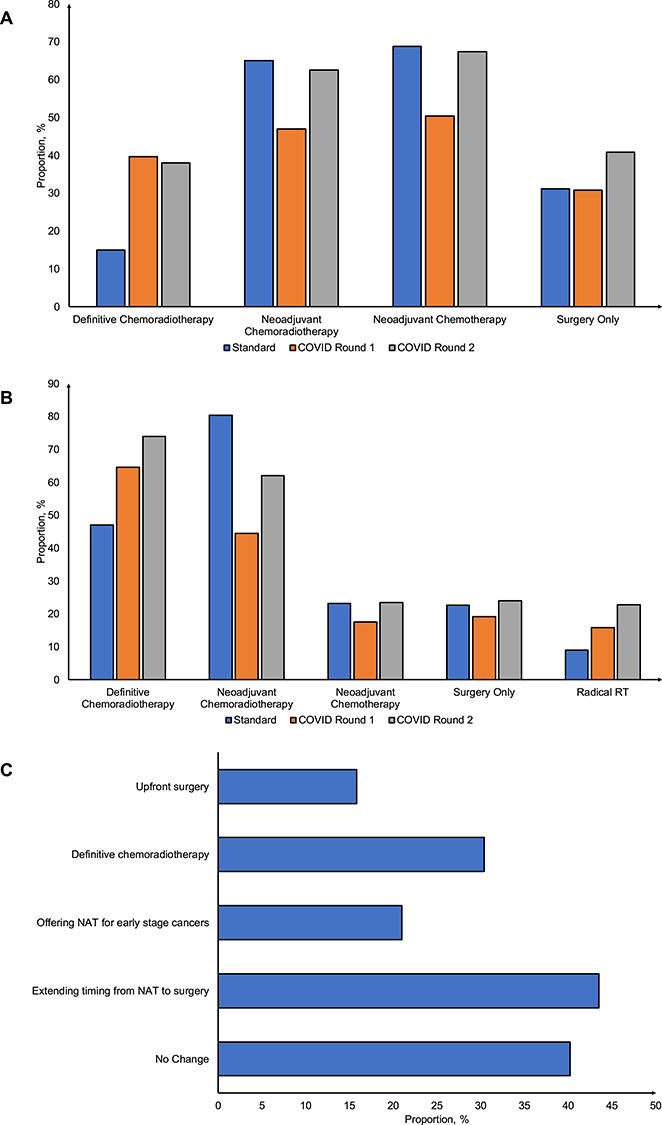
Distribution of oncological therapy available for esophageal cancers across centers from round 1 and 2 of the survey during the COVID-19 pandemic. (A) Esophageal adenocarcinoma. (B) Esophageal squamous cell carcinoma. (C) Overall changes to treatment strategies in esophagogastric cancer management to cope with the COVID-19 pandemic.
Esophageal squamous cell carcinoma
Standard pre-COVID-19 oncological therapies available for potentially curative esophageal squamous cell carcinoma included NCRS (n = 188, 80.3%) and DCRT (n = 110, 47.0%) (Fig. 3B). DCRT was offered in 47.0% of centers and significantly increased during the COVID-19 pandemic in both round 1 and 2 (64.5% and 73.9%, P < 0.001). Stratified analyses demonstrated significantly higher rates of adoption of DCRT and H-CoV centers in both round 1 and 2 compared to standard for esophageal squamous cell carcinoma (Supplementary data, Fig. S2, Table S4).
Timing of surgery after neoadjuvant therapy
For timing of surgery after neoadjuvant therapy as standard pre-COVID practice, the majority of centers offered surgery 6 to 8 weeks following neoadjuvant therapy (n = 132, 56.4%) and only 4.3% and 0.9% of centers offered surgery after 10 to 12 weeks and >12 weeks, respectively. However, there was a five-fold significant increase in centers offering surgery 10 to 12 weeks after neoadjuvant therapy in round 1 and 2 (26.5% vs. 21.7%, P < 0.001) (Table 2).
Table 2.
Changes in oncological therapy for esophageal cancer among respondents stratified in round 1 (n = 234) and 2 (n = 184) of the survey
| Standard | Round 1 | Round 2 | P-value | ||
|---|---|---|---|---|---|
| Esophageal adenocarcinoma | |||||
| Definitive chemoradiotherapy | No | 199 (85.0) | 141 (60.3) | 114 (62.0) | <0.001 |
| Yes | 35 (15.0) | 93 (39.7) | 70 (38.0) | ||
| Neoadjuvant chemoradiotherapy | No | 82 (35.0) | 124 (53.0) | 69 (37.5) | <0.001 |
| Yes | 152 (65.0) | 110 (47.0) | 115 (62.5) | ||
| Neoadjuvant chemotherapy | No | 73 (31.2) | 116 (49.6) | 60 (32.6) | <0.001 |
| Yes | 161 (68.8) | 118 (50.4) | 124 (67.4) | ||
| Surgery only | No | 161 (68.8) | 162 (69.2) | 109 (59.2) | 0.059 |
| Yes | 73 (31.2) | 72 (30.8) | 75 (40.8) | ||
| Esophageal squamous cell carcinoma | |||||
| Definitive chemoradiotherapy | No | 124 (53.0) | 83 (35.5) | 48 (26.1) | <0.001 |
| Yes | 110 (47.0) | 151 (64.5) | 136 (73.9) | ||
| Neoadjuvant chemoradiotherapy | No | 46 (19.7) | 130 (55.6) | 70 (38.0) | <0.001 |
| Yes | 188 (80.3) | 104 (44.4) | 114 (62.0) | ||
| Neoadjuvant chemotherapy | No | 180 (76.9) | 193 (82.5) | 141 (76.6) | 0.2 |
| Yes | 54 (23.1) | 41 (17.5) | 43 (23.4) | ||
| Surgery only | No | 181 (77.4) | 189 (80.8) | 140 (76.1) | 0.5 |
| Yes | 53 (22.6) | 45 (19.2) | 44 (23.9) | ||
| Radical radiotherapy | No | 213 (91.0) | 197 (84.2) | 142 (77.2) | <0.001 |
| Yes | 21 (9.0) | 37 (15.8) | 42 (22.8) | ||
| Timing of surgery after neoadjuvant therapy | |||||
| <6 weeks | 59 (25.2) | 24 (10.3) | 24 (13.0) | <0.001 | |
| 6–8 weeks | 132 (56.4) | 46 (19.7) | 69 (37.5) | ||
| 8–10 weeks | 30 (12.8) | 51 (21.8) | 43 (23.4) | ||
| 10–12 weeks | 10 (4.3) | 62 (26.5) | 40 (21.7) | ||
| >12 weeks | 2 (0.9) | 44 (18.8) | 8 (4.3) | ||
| Neoadjuvant therapy is unavailable | 1 (0.4) | 7 (3.0) | 0 (0) | ||
Standard treatment was defined as oncological therapy offered to patients by their center prior to the SARS CoV-2 pandemic whereas in round 1 and round 2 focusses on oncological therapy used during the SARS CoV-2 pandemic. P-values in tables represent statistical testing across each variable.
Changes to overall treatment
Overall changes to curative treatment for esophagogastric cancers are displayed in Figure 3C. While 40.2% of respondents did not consider changes to overall treatment, 43.5% respondents considered extending timing from neoadjuvant therapy, 30.4% of respondents considered changing to DCRT and 21.0% considered offering neoadjuvant therapy for early stage cancers (i.e. T2N0 cancers). Despite these changes, when presented with case vignettes, majority of respondents favored proceeding with resection as planned or proceeding to surgery (Supplementary data, Fig. S3).
Perioperative cancer surgery
Surgical prioritization
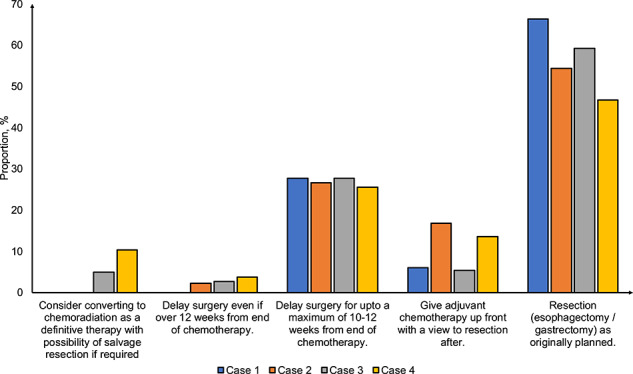
Prioritization of cancer resections was based on a national protocol in 19.7% of centers in round 1 and 23.9% in round 2 (Table 1). There were no significant differences in stratified analyses by total cases of COVID-19 per capita (Supplementary data, Table S5). We also assessed factors used in prioritization of cancer resections. In round 1, patient fitness was ranked first by 35.0% of respondents and tumor stage was ranked first by 34.4% of respondents. There were no significant differences in proportion of respondents ranking both patient fitness and tumor stage first in round 2 (Fig. 4A). However, there was a significant increase in proportion of respondents ranking neoadjuvant timing as first from round 1 to round 2 (16.7% vs. 27.7%, P = 0.011). To assess variation in prioritization of esophagogastric cancer resections, respondents were asked to prioritize case vignettes (Fig. 4B). Overall, there were no significant differences in ranking between cases in round 1 and 2. Prioritization of resections was based on early stage cancers or good physical fitness.
Fig. 4.
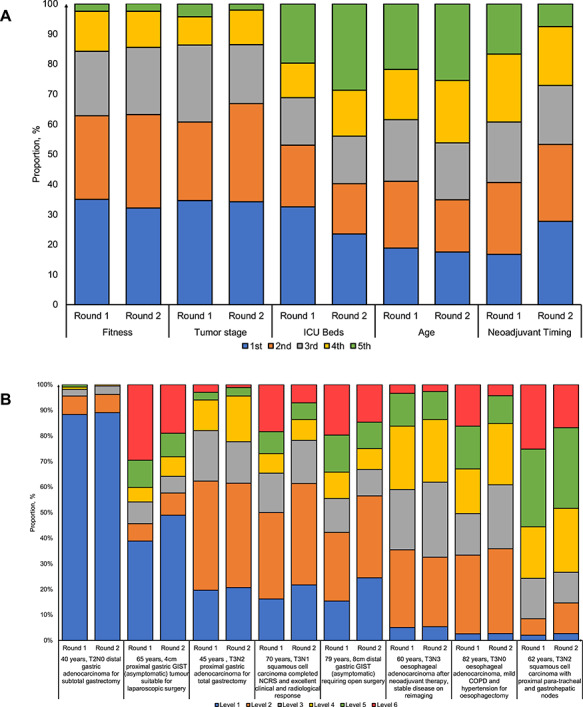
Distribution of ranking priority for esophagogastric cancer resections across centers from round 1 and 2 of the survey during the COVID-19 pandemic. (A) Prioritization factors. (B) Case vignettes. *For Figure 4B, the definition of each levels are as follows: level 1 (curative therapy with a high [>50%] chance of success); level 2 (curative therapy with an intermediate (15–50%) chance of success); level 3 (noncurative therapy with a high [>50%] chance of >1 year of life extension); level 4 (curative therapy with a low [0–15%] chance of success or noncurative therapy with an intermediate [15–50%] chance of >1 year life extension); level 5 (noncurative therapy with a high [>50%] chance of palliation/temporary tumor control but <1 year life extension); and level 6 (noncurative therapy with an intermediate [15–50%] chance of palliation or temporary tumor control and <1 year life extension).
Routine COVID-19 testing of patients
Only 14.7% (n = 27) were not performing routine testing of patients, 6.0% (n = 11) used routine CT scanning only and 33.2% (n = 61) used polymerase chain reaction (PCR) swab testing only. H-CoV centers had significantly higher rates of PCR swab testing than L-CoV or M-CoV centers (48.8% vs. 13.0% vs. 35.5%, P = 0.001). In 32.1% of centers, there was a combination of either symptom assessment, use of CT scanning or PCR swab testing. COVID-19 PCR swab testing was available for centers in 92.2% of respondents. The COVID-19 PCR swab testing was available within 6 hours for centers in 36.4% of respondents, significantly quicker availability rates of in H-CoV centers compared to M-CoV and L-CoV (53.8% vs. 25.9% vs. 13.0%, P < 0.001) (Supplementary data, Table S5).
Location of resection
Most respondents (46.7%, n = 86) were performing cancer resections in the same hospital (usual theatres) while 27.7% (n = 51) were performing cancer resections in the same hospital but separate theatres and 12.5% (n = 23) were performing resections at separate COVID-19 cold/private sites. However, there were no significant differences in location of performing resection between total COVID-19 cases per capita (Supplementary data, Table S5).
Personal protective equipment
Of the respondents surveyed in round 2, 40.8% of respondents used personal protective equipment (PPE) for all resections and 44.6% only for those with suspected or confirmed COVID-19 patients. Only 14.7% of respondents did not use PPE. There were no significant differences in rates of PPE by total COVID-19 cases per capita across groups (Supplementary data, Table S5).
Resection approach
There was a significant increase toward dual consultant operating from round 1 to round 2 (40.2% vs. 51.6%, P = 0.002), which was significantly higher in H-CoV centers for round 1. However, this was not the case for round 2 as rates of dual consultant operating consistently increased across centers in countries with L-CoV, M-CoV and H-CoV case incidence (45.0% vs. 54.1% vs. 58.2%, P = 0.5) (Supplementary data, Table S5). In 52.2% (n = 96) of centers, there was still ongoing practice of minimally invasive surgery while only 12.0% (n = 22) and 12.5% (n = 23) were avoiding minimally invasive surgery in all or selected cases, respectively. In H-CoV centers, there was a significant reduction in the use of minimally invasive surgery in all cases compared to L-CoV or M-CoV (7.5% vs. 13.0% vs. 16.0%, P = 0.002).
Postoperative management
Postoperative destination
Among respondents in round 1, only 45.3% of respondents send patients to ICU routinely and 48.3% of respondents send patients to ICU selectively following esophagogastric cancer resections (Table 1). In regards to postoperative destination, 2.6% of respondents were considering sending patients to a different hospital postoperatively during the COVID-19 pandemic during round 1, which significantly increased to 6.0% during round 2 (P < 0.001) (Table 1). There were no significant differences in responses when stratified by total COVID-19 cases per capita (Supplementary data, Table S6).
Impact of COVID-19 and surgeons
In round 2, we surveyed the impact of COVID-19 on esophagogastric surgeons. Among surgeons, 44.6% (82/184) have been symptomatic with COVID-19. Of the surgeons symptomatic with COVID-19, only 40.2% (33/82) had routine access to COVID-19 PCR testing for staff. Of those who had testing available (n = 33), only 12.1% (4/33) had tested positive. Figure 3 illustrates redeployment of surgeons during the COVID-19 pandemic. While 67.9% remained in the same department, some 20.0% and 13.8% were redeployed to emergency department and HDU/ICU, respectively (Fig. 5).
Fig. 5.
Distribution of redeployment of surgeons across centers from round 2 (n = 184) of the survey during the COVID-19 pandemic stratified by total COVID-19 cases by low (L-CoV), middle (M-CoV) and high (H-CoV) groups.
DISCUSSION
This international cross-sectional survey across 234 respondents from 225 centers and 49 countries spanning six continents demonstrates a high level of variation in availability of staging investigations, oncological therapy, decision-making regarding prioritization of cancer resections during the COVID-19 pandemic and perioperative management in cancer resections. The main findings of this survey include: (1) wide adoption of DCRT for esophageal adenocarcinoma and squamous cell carcinoma; (2) uncertainty and heterogeneity surrounding prioritization of patients undergoing cancer resections; and (3) high rates (44.6%) of workforce symptomatic from COVID-19 likely to impact delivery of cancer services during a pandemic. These data highlight the management challenges and several practice variations in caring for patients with esophagogastric cancers. Dissemination of data from this survey will improve understanding of current international clinical practice during the COVID-19 pandemic. Further, this study has identified the need for clear consistent national or international guidelines to be in place in the event of a further pandemic to ensure a standardized level of oncological care for patients with esophagogastric cancers.
Oncological therapy
This survey has demonstrated broad shifts in oncological management of patients with both esophageal adenocarcinoma and squamous cell carcinoma toward use of DCRT. This likely reflects the uncertainty related to surgical capacity owing to the lack of HDU or ICU capacity. In the absence of robust randomized trial data, DCRT and neoadjuvant treatment followed by surgery have equivalent survival outcomes for esophageal squamous cell carcinoma.12,13 The evidence base for DCRT is less strong for esophageal adenocarcinoma,13 but reasonable outcomes were seen for this group in the SCOPE1 trial.14,15 In this survey, there are also broad shifts to delaying time to surgery following neoadjuvant therapy, which could potentially affect prognosis. A meta-analysis of 13 studies involving 15,086 patients showed an interval longer than 7 to 8 weeks between the end of neoadjuvant CRT and surgery was significantly associated with an improved pathological complete response rate, but lower 2-year and 5-year overall survival.16 The effect of delay after NCS is less certain, however, a recent US National Cancer Database study demonstrated that an additional delay to surgery by at least 4 weeks may not have a significant impact on patient survival or cancer progression.17 In addition, data presented for esophageal and gastric cancers suggest that surgery could be potentially safe to postpone up to 28 and 30 weeks after diagnosis in patients receiving neoadjuvant treatment.17 The ongoing NeoRes-II randomized controlled trial evaluating timing of surgery after NCRS will provide level 1 evidence.18
Cancer resection prioritization
There is a concern that limited HDU/ICU bed availability and the risk of postoperative SARS-CoV-2 infection will severely limit or preclude surgical intervention for esophagogastric patients. Therefore, prioritization of cancer resection patients most likely to benefit and be at low risk of complications is important. Several groups have developed guidelines and prioritization models, including AUGIS,19 ESDE,20 Thoracic Surgery Outcomes Research Network21 and other groups22–24 specifically for this patient group. The medically necessary, time-sensitive (MeNTS) score has been developed and systematically scores several factors (procedure timing and resource use, disease urgency and patient risk factors) for prioritizing surgical procedures across all surgical specialties.24 This scoring system aims to triage MeNTS procedures, and appropriately weighs individual patient risks with the ethical necessity of optimizing resource allocation during the pandemic. This approach is applicable across a broad range of hospital settings (academic and community, urban and rural) and may be able to inform case triage as operating room capacity resumes once the acute phase of the pandemic subsides.
This survey highlights the prioritization of cancer resections by centers is based on patient fitness and tumor stage. In our survey, surgeons appear to prioritize advanced tumors (i.e. high-risk nodal disease) due to high risk of disease progression.17 Treatment decisions are further complicated by the fact most of the patients with esophagogastric cancer are in a ‘high-risk’ category for poor outcomes if they developed COVID-19 (elderly, cardiorespiratory comorbidities and obesity). In addition, the surgery (especially thoracotomy) can both impair lung function (i.e. one lung isolation, postoperative pain and pneumonia) and potentially expose clinical teams to aerosolized viral load (bronchoscopy, double-lumen endotracheal tube placement and endoscopy).21
Screening and protection in COVID-19
Due to the high prevalence of pulmonary complications following esophagogastric surgery,25 and the implications of postoperative COVID-19 infection, rates of asymptomatic infection, there is a convincing case for accurate preoperative testing of these patients prior to surgery. The Corona Virus Global Surgical Collaborative recommend performing routine COVID-19 testing for all patients who will undergo a surgical or interventional endoscopic procedure in institutions seeing high volumes of COVID-19 patients.26 In addition, American College of Surgeons advised to wait for the results of COVID-19 testing in patients who may be infected, but no further recommendation surrounding cancellation or postponement of surgery in patients testing positive.27 Routine screening may include symptomatic assessment via telephone triage several days before elective cancer surgery for risk stratification and preoperative CT scanning owing to high sensitivity up to 97%.28
Minimally invasive surgery
Although previous research has shown that laparoscopy can lead to aerosolization of blood-borne viruses,29–31 no specific robust research has been published in COVID-19 patients. Current available guidance from the Intercollegiate Royal College of Surgeons32 and Society of American Gastrointestinal and Endoscopic Surgeons26 offer inconsistent guidance that can be open to interpretation on performing minimally invasive surgery. This is reflected in this survey where 24.5% of respondents stopping minimally invasive surgery in all or selected cases.
Surgical workforce
In our survey, 44.6% have been symptomatic with COVID-19. Of the surgeons symptomatic with COVID-19, only 40.2% (33/82) had routine access to testing for staff. Of those who had testing available (n = 33), only 12.1% (4/33) had tested positive. Esophageal surgeons have a duty to self-isolate if they develop COVID symptoms and get appropriate testing to avoid spreading the virus to other staff and patients in the hospital.
Strengths and limitations
This survey has several strengths including its large sample size and that it performed two rounds capturing changes during the pandemic. In addition, a wide range of countries and continents, with differing COVID-19 disease incidence were captured. This allowed stratified analyses to be performed for total cases per capita to gain extra insight into the uncertainty in current practice among esophageal surgeons. By its nature, this survey will have limitations. First, despite widely advertising this survey via multiple specialist organizations and social media, it is possible that selection bias has occurred. Our survey was specifically designed for surgeons and as such the thoughts of patients, oncologists or other healthcare professionals involved in the treatment of esophagogastric surgery have not been elicited. Second, this study did not collect specific patient outcomes for esophageal and gastric cancers surgery and COVID-19. However, research from collaborative groups such as CovidSurg33 and PanSurg34 collaborative will provide real-time data to further supplement data from this survey. Finally, this study attempted to stratify current practices by total confirmed cases per capita, but this is likely limited by testing rates available in each country.
CONCLUSION
In summary, this survey highlights the management challenges and several practice variations in caring for patients with esophagogastric cancers. Dissemination of data from this survey will improve understanding of current international clinical practice during the COVID-19 pandemic. Further, this study has identified the need for clear, consistent national or international guidelines to be in place in the event of a further pandemic to ensure a standardized level of oncological care for patients with esophagogastric cancers.
Supplementary Material
ACKNOWLEDGMENTS
We would like to thank the Australian and New Zealand Gastric and Oesophageal Surgery Association (ANZGOSA), Association of Upper Gastrointestinal Surgeons (AUGIS), Dutch Cancer Audit (DUCA), European Society of Diseases of Esophagus (ESDE), Gastric Cancer Surgery Trials Reported Outcomes Standardisation (GASTROS), International Society for Disease of Esophagus (ISDE) and Oesophagogastric Anastomosis Audit (OGAA) for emailing this survey out.
Conflicts of interest: The authors declare that they have no conflict of interest.
References
- 1. World Health Organization Coronavirus Disease 2019 (COVID-19): Situation Report, 2020, 103 [Cited 2 May 2020]Available fromhttps://www.who.int/docs/default-source/coronaviruse/situation-reports/20200502-covid-19-sitrep-103.pdf?sfvrsn=d95e76d8_4
- 2. Grasselli G, Pesenti A, Cecconi M. Critical care utilization for the COVID-19 outbreak in Lombardy, Italy: early experience and forecast during an emergency response. JAMA 2020. [DOI] [PubMed] [Google Scholar]
- 3. Oba A, Stoop T F, Lohr M et al. . Global survey on pancreatic surgery during the COVID-19 pandemic. Ann Surg 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Pellino G, Spinelli A. How COVID-19 outbreak is impacting colorectal cancer patients in Italy: a long shadow beyond infection. Dis Colon Rectum 2020. [DOI] [PubMed] [Google Scholar]
- 5. Liang W, Guan W, Chen R et al. . Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol 2020; 21(3): 335–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Wang H, Zhang L. Risk of COVID-19 for patients with cancer. Lancet Oncol 2020; 21: e181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Ueda M, Martins R, Hendrie P C et al. . Managing cancer care during the COVID-19 pandemic: agility and collaboration toward a common goal. J Natl Compr Canc Netw 2020; 1–4. [DOI] [PubMed] [Google Scholar]
- 8. Brindle M, Gawande A. Managing COVID-19 in surgical systems. Ann Surg 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Coronavirus NEaNI Specialty Guides For Patient Management, 2020. [Cited 4 May 2020]Available fromhttps://www.england.nhs.uk/coronavirus/publication/specialty-guides/
- 10. Network NLCC CRITCON, 2020. [Cited 2 May 2020]Available fromhttps://www.londonccn.nhs.uk/managing-the-unit/capacity-escalation/critcon/
- 11. Roser M, Ritchie H. Coronavirus Pandemic (COVID-19)—The Data, 2020. [Cited 2 May 2020]Available fromhttps://ourworldindata.org/coronavirus-data?country=ARG+AUS+AUT+BEL+BRA+CMR+CAN+COL+CZE+DNK+EGY+FIN+DEU+GBR+GRC+HKG+HUN+IND+IRN+IRL+ITA+CIV+JPN+KEN+LBN+LBY+LTU+LUX+MYS+MEX+NLD+NZL+NGA+PAK+PER+POL+PRT+KOR+ROU+RUS+SGP+ESP+SWE+SDN+CHE+TUR+USA#confirmed-cases
- 12. Rackley T, Leong T, Foo M, Crosby T. Definitive chemoradiotherapy for oesophageal cancer—a promising start on an exciting journey. Clin Oncol (R Coll Radiol) 2014; 26(9): 533–40. [DOI] [PubMed] [Google Scholar]
- 13. Kamarajah S K, Phillips A W, Hanna G B, Low D E, Markar S R. Definitive Chemoradiotherapy compared to neoadjuvant Chemoradiotherapy with Esophagectomy for Locoregional esophageal cancer: National Population-Based Cohort Study. Ann Surg 2020. [DOI] [PubMed] [Google Scholar]
- 14. Crosby T, Hurt C N, Falk S et al. . Chemoradiotherapy with or without cetuximab in patients with oesophageal cancer (SCOPE1): a multicentre, phase 2/3 randomised trial. Lancet Oncol 2013; 14: 627–37. [DOI] [PubMed] [Google Scholar]
- 15. Crosby T, Hurt C N, Falk S et al. . Long-term results and recurrence patterns from SCOPE-1: a phase II/III randomised trial of definitive chemoradiotherapy +/− cetuximab in oesophageal cancer. Br J Cancer 2017; 116(6): 709–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Qin Q, Xu H, Liu J et al. . Does timing of esophagectomy following neoadjuvant chemoradiation affect outcomes? A meta-analysis. Int J Surg 2018; 59: 11–8. [DOI] [PubMed] [Google Scholar]
- 17. Turaga K K, Girotra S. Are we harming cancer patients by delaying their cancer surgery during the COVID-19 pandemic? Ann Surg 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Nilsson M. Timing of Resective Surgery After Neoadjuvant Chemoradiotherapy in Esophageal Cancer (NeoResII), 2020. [Cited 6 May 2020]Available fromhttps://clinicaltrials.gov/ct2/show/NCT02415101
- 19. Ireland AoUGSoGBa Surgical Priority in Oesophageal and Gastric Cancer, 2020. [Cited 2 May 2020]Available fromhttps://www.augis.org/wp-content/uploads/2020/03/Surgical-Priority-in-Oesophageal-and-Gastric-Cancer.pdf
- 20. Barbieri L, Talavera Urquijo E, Parise P, Nilsson M, Reynolds J V, Rosati R. Esophageal oncologic surgery in SARS-CoV-2 (COVID-19) emergency. Dis Esophagus 2020; 3692(20): 30758–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Thoracic Surgery Outcomes Research Network, Inc COVID-19 guidance for triage of operations for thoracic malignancies: a consensus statement from thoracic surgery outcomes research network. Ann Thorac Surg 2020; 22(5). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Qadan M, Hong T S, Tanabe K K, Ryan D P, Lillemoe K D. A multidisciplinary team approach for triage of elective cancer surgery at the Massachusetts General Hospital during the novel coronavirus COVID-19 outbreak. Ann Surg 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Brindle M E, Doherty G, Lillemoe K, Gawande A. Approaching surgical triage during the COVID-19 pandemic. Ann Surg 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Prachand V N, Milner R, Angelos P et al. . Medically necessary, time-sensitive procedures: scoring system to ethically and efficiently manage resource scarcity and provider risk during the COVID-19 pandemic. J Am Coll Surg 2020; 7515(20): 30317–23.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Mariette C, Markar S R, Dabakuyo-Yonli T S et al. . Hybrid minimally invasive Esophagectomy for esophageal cancer. N Engl J Med 2019; 380(2): 152–62. [DOI] [PubMed] [Google Scholar]
- 26. SoAGaES (SAGES) SAGES and EAES Recommendations Regarding Surgical Response to COVID-19 Crisis, 2020. [Cited 3 May 2020]Available fromhttps://www.sages.org/recommendations-surgical-response-covid-19/
- 27. ACoS (ACS) COVID-19: Elective Case Triage Guidelines for Surgical Care, 2020. [Cited 3 May 2020]Available fromhttps://www.facs.org/covid-19/clinical-guidance/elective-case
- 28. Ai T, Yang Z, Hou H et al. . Correlation of chest CT and RT-PCR testing in coronavirus disease 2019 (COVID-19) in China: a report of 1014 cases. Radiology 2020; 200642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Alp E, Bijl D, Bleichrodt R P, Hansson B, Voss A. Surgical smoke and infection control. J Hosp Infect 2006; 62: 1–5. [DOI] [PubMed] [Google Scholar]
- 30. Kwak H D, Kim S H, Seo Y S, Song K J. Detecting hepatitis B virus in surgical smoke emitted during laparoscopic surgery. Occup Environ Med 2016; 73: 857–63. [DOI] [PubMed] [Google Scholar]
- 31. Choi S H, Kwon T G, Chung S K, Kim T H. Surgical smoke may be a biohazard to surgeons performing laparoscopic surgery. Surg Endosc 2014; 28: 2374–80. [DOI] [PubMed] [Google Scholar]
- 32. Edinburgh TRCoSo Intercollegiate General Surgery Guidance on COVID-19 UPDATE, 2020. [Cited 6 May 2020]Available fromhttps://www.rcsed.ac.uk/news-public-affairs/news/2020/march/intercollegiate-general-surgery-guidance-on-covid-19-update
- 33. CovidSurg Collaborative Global guidance for surgical care during the COVID-19 pandemic. Br J Surg 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Markar S, Martin G, Penna M et al. . Changing the paradigm of surgical research during a pandemic. Ann Surg 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


