Abstract
Objectives:
The currently widely used technological devices give rise to electromagnetic fields (EMFs) at various frequencies. Recent studies have reported that EMFs damage the central nervous system. The cerebellum is of considerable importance to human life due to its involvement in motor control, language, and cognitive-sensory functions. Damage occurring in the histological layers of the cerebellar cortex causes various neurological and psychiatric diseases, such as paralysis, tumor, autism, and schizophrenia. Our study involved a histopathological evaluation of the effects of communication systems’ standard 900-MHz EMF on the cerebellum.
Methods:
Sprague–Dawley rats were assigned into two groups containing six animals each: control and EMF. The EMF group was exposed to a 24-h 900-MHz radiofrequency EMF over 20 days with a digital modulation signal generator installed in the middle of their cage. Ten days after EMF application, the rats were sacrificed by cervical dislocation under anesthesia induced with 50 mg/kg ketamine hydrochloride and 10 mg/kg intraperitoneal xylazine HC1.
Results:
Intense caspase-3 expression was seen in the Purkinje cells and granular cells exposed to a 900-MHz frequency EMF (p<0.05). Pyknotic nuclei were notable in the Purkinje and granular cells exposed to a 900-MHz EMF. We also observed a decrease in the cytoplasm of the Purkinje and granular cells. Specimens from the EMF group exhibited decreases in the thickness of the molecular cell layer, Purkinje cell layer, and granular cell layer compared with those from the control group. However, the difference was not statistically significant (p>0.05).
Conclusion:
A 900-MHz EMF causes deleterious effects on the cerebellum by giving rise to apoptosis accompanied by caspase-3 expression in the Purkinje and granular cells in particular.
Keywords: Caspase-3, cerebellum, electromagnetic field, rat
Technological devices that were not used till the 1970s, such as cell phones, base stations, wireless modems, and magnetic resonance imaging systems used in the health sector, have become an indispensable part of our lives now.[1] Recent studies have indicated that these newly introduced technological devices have deleterious effects on human health by creating an electromagnetic field (EMF). Depending on the extent and duration of exposure to EMF, serious problems caused by EMF including mainly apoptosis, oxidative stress, impairment of the structures and functions of enzymes, structural and functional losses in cell organelles, and increase in the number of cases with cancer involving particularly the brain, spinal cord, and heart muscle have been demonstrated.[2–7] Caspase-3 is a key protein, which is involved in the apoptotic mechanism of many cell types including neurons.[8, 9] Besides, caspase-3 has been also associated with neurodegenerative processes.[10, 11] Studies have demonstrated that caspase-3 activation, plays a vital role in apoptosis and may be considered a terminal event before cell death.[9, 12] However, limited information exists on the apoptotic effects of the EMF.[13]
Frei et al.[14] have reported that the EMF increases the risk of cancer particularly in the temporal and occipital regions of the brain in the central nervous system. The cerebellum is of considerable importance to human life due to its involvement in motor control, language, and cognitive-sensory functions.[15–17] Any damage to the cerebellum, which may involve the histological layers of the cerebellar cortex, causes various neurological and psychiatric diseases, such as paralysis, tumor, autism, schizophrenia, and ataxia.[18–20] Iwata et al.[2] have reported that magnetic stimuli activate the Purkinje cells in the cerebellar cortex, leading to the inhibition of dentato-thalamo-cortical pathways. Therefore, noninvasive methods are important as they yield precise anatomical images of the cerebellar cortical architecture, thus providing markers for the diagnosis and follow-up of patients with these pathologies.
Although various frequencies of EMF have been used in relevant studies, we preferred the 900-MHz EMF as it meets the standards of the world communication system and is used in cell phones, which are used in our daily life. We investigated the effects of 900-MHz EMF on the cerebellum using histopathological and immunohistochemical methods.
Methods
Experimental animals
Four 6-month-old Sprague–Dawley strain rats weighing 250–300 g were investigated at the Animal Care and Research Unit, Recep Tayyip Erdoğan University. All animals were procured in compliance with the Guide for the Care and Use of Laboratory Animals, which is prepared by National Academy of Sciences and published by National Institute of Health. The study protocol was critically reviewed and approved by the Local Ethics Committee of Animal Research, Faculty of Medicine, Recep Tayyip Erdoğan University (approval number: 2014/2).
Sprague–Dawley strain rats were kept in alternate dark and light cycles of 12 h at 22°C–23°C under 55%–60 % relative humidity. Female rats were kept in plastic cages with the dimensions of 36×23×21 cm. Each cage contained three rats. Commercial rat pellets (BayramoğluYem ve Un Sanayi Ticaret A.Ş., Erzurum, Turkey) and tap water were provided for animals ad libitum. All procedures applied on animals were designed and implemented in compliance with the criteria of Helsinki Declaration concerning National Health Institute Maintenance and Use of Laboratory Animals.
Experimental application
Six healthy control rats and six rats exposed to EMF were assigned into the control and EMF group. The control group was not subjected to any intervention. The EMF group was exposed to a 24-h 900-MHz radiofrequency EMF for 20 days with a digital modulation signal generator (Anritsu MG3670 B type, Japan) installed in the middle of their cage.[21] The peak value of digital modulation signal generator was fixed at 2 Watt (W) during exposure so as to represent the exposure of global mobile communication system. The carrier frequency (900 MHz), modulation frequency (217 Hz), pulse width (577 μs), and maximum peak power (2 W) were set as indicated.[21]
Ten days after EMF application, the rats were sacrificed by cervical dislocation under anesthesia induced with 50 mg/kg ketamine hydrochloride (Ketalar®, Eczacıbaşı Parke Davis, Istanbul, Turkey) and 10 mg/kg intraperitoneal xylazine HC1 (Alfazyne®, Alfasan International BV Woerden®, the Netherlands).
Histopathological follow-up procedure
The cerebellar tissue samples were fixed in 10% neutral formaldehyde solution using routine laboratory methods and then dehydrated with series of increasing concentrations of ethanol (Merck, Darmstadt, Germany). Following the procedure of dehydration, the cerebellar tissues were left in xylol solution (Merck, Darmstadt, Germany) to make them transparent. Finally, the samples were embedded in solid paraffin blocks (Merck, Darmstadt, Germany) at 56°C–58°C. Then, the paraffin blocks were cut using a microtome (Leica RM2125RT, Germany) into 2-µm thick sections. These sections were stained with Harris hematoxylin–eosin (H&E) G dye. Slides stained with H&E were examined under a light microscope (Leica DM6200, Mannheim, Microscope), and the histopathological findings were photographed using Olympus DP20 camera (OlympusCorparation, Tokyo, Japan).
Immunohistochemical analysis
The 2–3-µm thick sections cut from the paraffin blocks with microtomes were placed on slides (Marienfeld-Superior, Germany). Through the action mediated by caspase-activated DNAase enzyme, caspase-3 induces apoptosis via condensation of chromatin and fragmentation of DNA into nucleosomal subunits.[22] Therefore, the sections were stained with immunohistochemical methods (Caspase-3 rabbit polyclonal, ab13847, Abcam UK) to reveal caspase-3 expressions in cerebellar cells. DAB substrate kit (HRP/DAB (ABC) Detection IHC Kit, abcam, ab 64264, UK) was used to visualize the caspase-3 expression.
In our study, the slides were left in xylol solution (Merck, Darmstadt, Germany) and then subjected to paraffinization procedure. Then the slides were left in 80°, 90°, and absolute alcohol solutions in that order (Etanol, Merck, Darmstadt, Germany). Following the stage of deparaffinization, the sections were placed in a citrate buffer solution (×100 citrate buffer, pH=6, ab 93678, Abcam, UK) and subjected to antigen retrieval procedure in a microwave oven. The slides were washed with phosphate buffer saline solution (10× Phosphate Buffered Saline (PBS), pH=7.4, ab128983, Abcam, UK). Subsequently, the slides were incubated in 3% H202 solution (H202 blocking, ab64264, Abcam, UK) to block endogenous peroxidases. The slides were washed with a PBS buffer solution, and then left in a protein blocking solution (Abcam, ab64264, Abcam, UK). Afterward, they were incubated in a primary antibody solution for 60 min. The slides were again washed with the PBS solution and incubated in a secondary antibody solution (Biotinylated Goat Anti-Polyvalent, ab64264, Abcam, UK) for 30 min. The sections were washed with the PBS solution and left first in a streptavidine peroxidase solution (Abcam, ab64264, Abcam, UK) and then in DAB-chromogen kit solution. Then, the slides washed with PBS and counterstained with H&E. The sections were washed with water, closed with water-based closure and coverslip, examined under light microscope (Leica DM6200, Mannheim, Germany), and photographed using Olympus DP20 brand (Olympus Corporation, Tokyo, Japan) digital camera build in the microscope.
Quantitative analysis
In our study, the measurements of molecular, Purkinje cell, and granular layers and white matter area were made using arbitrary line probe of the Olympus DP2-BSW (v.2.1 to v.2.2, Build 6212, Tokyo, Japan) program linked to the light microscope. This measurement system consists of a camera (Olympus DP20, Olympus Corporation, Tokyo, Japan) attached to a light microscope (Leicia DM6200, DM6200, Mannheim, Germany) and an Olympus DP2-BSW software program. The limits of quantification of H&E-stained slides were determined by two independent histopathologists (Fig. 1).
Figure 1.
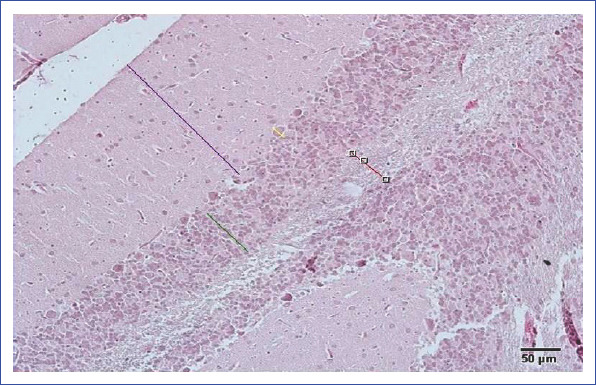
Method for measuring the cerebellar thickness.
Semiquantitative analysis
Caspase-3 positive cells were evaluated by two independent histologists as slightly positive (+), moderately positive (++), strongly positive (+++), very strongly positive (++++) (Table 1).
Table 1.
Caspase-3 positivity scoring
| % | Explanation | Caspase-3 positivity score |
|---|---|---|
| <5* | Mild | (+) |
| <25 | Moderate | (++) |
| <50 | Severe | (+++) |
| <75 | Very severe | (++++) |
Statistical Analysis
Measurement data of the scoring of histological layers of cerebellum, gray matter, and caspase-3 positive cells were estimated using SPSS 20.00 (IBM, New York, USA) program and expressed as median±standard deviation. Data with and without normal distribution patterns were evaluated using Student t test and Mann–Whitney U test, respectively. In all measurements, p<0.05 was considered statistically significant.
Results
Histopathological findings
The samples of the cerebellar tissue belonging to the control group were examined under the light microscope, and the cerebellar cortex that consists of molecular cells, Purkinje cells, and granular layers cells and white matter was found to have a normal structural composition. The Purkinje cells had euchromatic nuclei with a markedly visible cytoplasm. Other cerebellar cells had retained their normal anatomy (Fig. 2a-b). However, especially, as a remarkable finding, the Purkinje cells and granular layer cells in the cerebellar samples of the EMF group had pycnotic nuclei. Besides, we observed marked decrease in the cytoplasmic contents of Purkinje and granular cells of the EMF group (Fig. 2c-d).
Figure 2 (a, d).
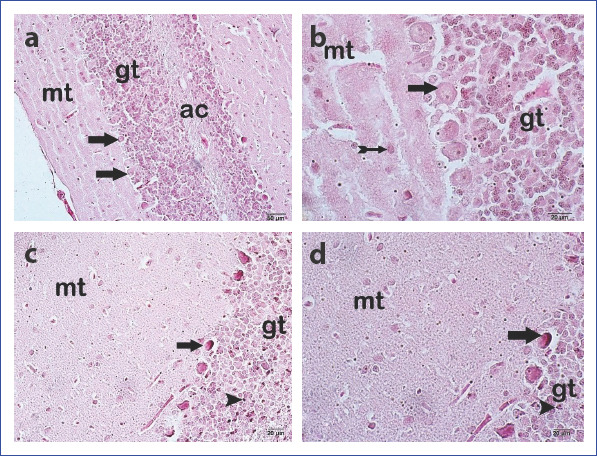
Appearance of the cerebellum under light microscope. Control Group: (a) Purkinje cell (arrow) 200× (b) Purkinje cell (arrow) basket cell (arrow with tail) 400×. Electromagnetic field group (d) Degenerative Purkinje cells with pycnotic nuclei (arrow) and granular cells with pycnotic nuclei (arrowhead) 200×. (e) Degenerative Purkinje cells with pycnotic nuclei (arrow) and granular cells with pycnotic nuclei (arrowhead). Mt, molecular layer; Gt, granular layer; Ac, white matter (H&E).
Immunohistochemical findings
We did not observe caspase-3 expression in the Purkinje cells, basket cells in the molecular layer, and granular cells in the granular layer of the control group samples of cerebellar tissue (Fig. 3a-b). However, very intense caspase-3 expression was detected mainly in the Purkinje cells and granular layer cells in the EMF group (Fig. 3a-b).
Figure 3 (a, d).
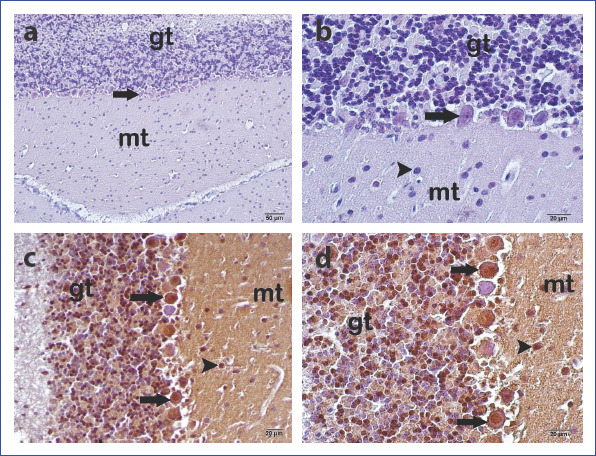
Appearance of the cerebellum under light microscope. Control Group: (a) Immune-negative Purkinje cell (arrow) 200×. (b) Immune-negative Purkinje cell (arrow) and immune-negative granular cell (arrowhead) 400×. Electromagnetic field group: (c) Immune-positive Purkinje cells (arrow) and immune-positive granular cells (arrowhead) 200×. (d) Immune-positive Purkinje cells (arrow) and immune-positive granular cells (arrowhead). Mt, molecular layer; Gt, granular layer. (Caspase-3 immunohistochemical staining).
Results of statistical analysis
Measurements of the samples of the EMF group revealed decreases in the thickness of molecular cell layer, Purkinje cell layer, and granular cell layer compared with those of the control group without any statistically significant intergroup difference (p>0.05 ) (Table 2) (Fig. 3). The number of caspase-3 positive cells in the cerebellar tissue samples obtained from the EMF group were significantly higher than those from the control group (p=0.00) (Table 3).
Table 2.
Histopathological measurements (median±standard deviation)
| Group | Thickness of molecular cell layer (µm) | Thickness of Purkinje cell layer (µm) | Thickness of granular cell layer (µm) | Thickness of white matter (µm) |
|---|---|---|---|---|
| Control | 210.89±19.25a | 23.17±3.77a | 151.50±37.32a | 133.60±16.50a |
| EMF | 197.26±31.05a | 18.36±3.96a | 113.77±20.92a | 105.08±36.56a |
P<0.05 for the electromagnetic field group in comparison with the control group; EMF, electromagnetic field.
Table 3.
Caspase-3 measurements (median±standard deviation)
P<0.05 for the electromagnetic field group in comparison with the control group; EMF, electromagnetic field.
Discussion
As reported in recent studies, EMF decreases the number of Purkinje cells in the cerebellum.[7, 23] As reported in histopathological and biochemical studies, EMF induces apoptosis in the Purkinje cells.[1] All studies performed concerning the effects of EMF on the cerebellum have generally focused on the Purkinje cells.[1, 5–7, 22] However, as an overlooked issue, we think that since the Purkinje cells are supported by the basket cells and because of their position in the cerebellum, the Purkinje cells are exposed to the adverse effects of EMF less severely than other cerebellar cells. Particularly in the global communication systems, a GSM-900 band width protocol and a minimum 900-MHz frequency EMF is used in mobile phones.[4, 6, 24] Thus, to reveal the total effects of low-frequency EMF on the cerebellum, all histological layers of the cerebellum were analyzed.
In EMF studies, a decrease in the number of Purkinje cells and apoptosis has been reported.[1, 6, 7] Caspase-3 is an important protease, which exerts biological and molecular effects on the mechanism of apoptosis. In our study, we detected caspase-3 expression in the Purkinje cells, in compliance with the findings of other studies. In addition, we observed apoptotic cells accompanying the caspase-3 expression in the granular cells. However, we did not detect caspase-3 activation in the basket cells in the molecular layer. In consideration of its position in the cerebellum, although the basket cells are most severely exposed to the adverse effects of EMF, inability to observe any destructive change in the basket cells may be attributed to their higher resistance against stimuli emitted from EMF than Purkinje and other cerebellar cells. Albeit, the granular layer of cerebellar cortex also contains caspase-3 expression and pycnotic nuclei any destructive change was not observed in other granular layer cells.
The small number of stereological studies performed on this subject has rather focused on the number of Purkinje cells and reported that EMF decreases the number of Purkinje cells.[1, 5–7, 22] In our study, a decrease in the thickness of molecular cell layer, granular cell layer, and Purkinje cell layer was observed without any statistically significant intergroup difference. Despite the appearance of apoptotic Purkinje cells, we think that since supportive cells were not injured, any significant difference in the thickness of molecular cell layer, Purkinje cell layer, and granular cell layer was not observed.
Our study was a Phase 1 trial that analyzed cerebellum as a whole, and as an outcome, the adverse effects of 900-Mhz frequency EMF on cerebellar cells and mainly on Purkinje and granular cells have been demonstrated. Further investigations on the molecular mechanism of cerebellar injury should be performed to emphasize the outcomes of our study.
Disclosures
Ethics Committee Approval: The study protocol was critically reviewed and approved by the Local Ethics Committee of Animal Research, Faculty of Medicine, Recep Tayyip Erdoğan University (approval number: 2014/2).
Peer-review: Externally peer-reviewed.
Conflict of Interest: None declared.
Authorship contributions: Concept – H.F., L.T., T.M.; Design – H.F., L.T., T.M.; Supervision – H.F., L.T., T.M., M.G.A., A.Y.; Materials – H.F., L.T., T.M., M.G.A., A.Y.; Data collection &/or processing – T.M., A.Y., K.A.; Analysis and/or interpretation – H.F., L.T., T.M., M.G.A., A.Y.; Literature search – H.F., L.T., T.M., M.G.A., A.Y.; Writing – H.F., T.M., M.G.A., M.O.; Critical review – H.F., L.T., T.M., M.G.A., A.Y.
References
- 1.Köktürk S, Yardimoglu M, Celikozlu SD, Dolanbay EG, Cimbiz A. Effect of Lycopersicon esculentum extract on apoptosis in the rat cerebellum, following prenatal and postnatal exposure to an electromagnetic field. Exp Ther Med. 2013;6:52–6. doi: 10.3892/etm.2013.1123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Iwata NK, Hanajima R, Furubayashi T, Terao Y, Uesugi H, Shiio Y, et al. Facilitatory effect on the motor cortex by electrical stimulation over the cerebellum in humans. Exp Brain Res. 2004;159:418–24. doi: 10.1007/s00221-004-1979-x. [DOI] [PubMed] [Google Scholar]
- 3.Ikinci A, Mercantepe T, Unal D, Erol HS, Sahin A, Aslan A, et al. Morphological and antioxidant impairments in the spinal cord of male offspring rats following exposure to a continuous 900MHz electromagnetic field during early and mid-adolescence. J Chem Neuroanat. 2016;75:99–104. doi: 10.1016/j.jchemneu.2015.11.006. [DOI] [PubMed] [Google Scholar]
- 4.Kerimoğlu G, Mercantepe T, Erol HS, Turgut A, Kaya H, Çolakoğlu S, et al. Effects of long-term exposure to 900 megahertz electromagnetic field on heart morphology and biochemistry of male adolescent rats. Biotech Histochem. 2016;91:445–54. doi: 10.1080/10520295.2016.1216165. [DOI] [PubMed] [Google Scholar]
- 5.Oda T, Koike T. Magnetic field exposure saves rat cerebellar granule neurons from apoptosis in vitro. Neurosci Lett. 2004;365:83–6. doi: 10.1016/j.neulet.2004.04.068. [DOI] [PubMed] [Google Scholar]
- 6.Odaci E, Hanci H, Ikinci A, Sönmez OF, Aslan A, Sahin A, et al. Maternal exposure to a continuous 900-MHz electromagnetic field provokes neuronal loss and pathological changes in cerebellum of 32-day-old female rat offspring. J Chem Neuroanat. 2016;75:105–10. doi: 10.1016/j.jchemneu.2015.09.002. [DOI] [PubMed] [Google Scholar]
- 7.Rağbetli MC, Aydinlioğlu A, Koyun N, Rağbetli C, Bektas S, Ozdemir S. The effect of mobile phone on the number of Purkinje cells: a stereological study. Int J Radiat Biol. 2010;86:548–54. doi: 10.3109/09553001003734527. [DOI] [PubMed] [Google Scholar]
- 8.Mackenzie SH, Clark AC. Death by Caspase Dimerization. In: Matthews JM, editor. Protein Dimerization and Oligomerization in Biology. New York: Springer; 2012. pp. 55–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dinçel GÇ, Kul O. Pathologic apoptosis and diagnostic methods [Article in Turkish] Gümüshane Üniversitesi Sağlik Bilimleri Dergisi. 2016;5:86–108. [Google Scholar]
- 10.Folch J, Alvira D, López-querol M, Tajes M, Sureda FX, Forsby A, et al. Evaluation of transcriptional activity of caspase-3 gene as a marker of acute neurotoxicity in rat cerebellar granular cells. Toxicol Vitro. 2010;24:465–71. doi: 10.1016/j.tiv.2009.09.023. [DOI] [PubMed] [Google Scholar]
- 11.Snigdha S, Smith ED, Prieto GA, Cotman CW. Caspase-3 activation as a bifurcation point between plasticity and cell death. Neurosci Bull. 2012;28:14–24. doi: 10.1007/s12264-012-1057-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pace V, Bellizzi D, Giordano F, Panno ML, De Benedictis G. Experimental testing of a mathematical model relevant to the extrinsic pathway of apoptosis. Cell Stress Chaperones. 2010;15:13–23. doi: 10.1007/s12192-009-0118-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Akdag MZ, Dasdag S, Ulukaya E, Uzunlar AK, Kurt MA, Taskin A. Effects of extremely low-frequency magnetic field on caspase activities and oxidative stress values in rat brain. Biol Trace Elem Res. 2010;138:238–49. doi: 10.1007/s12011-010-8615-3. [DOI] [PubMed] [Google Scholar]
- 14.Frei MR, Berger RE, Dusch SJ, Guel V, Jauchem JR, Merritt JH, et al. Chronic exposure of cancer-prone mice to low-level 2450 MHz radiofrequency radiation. Bioelectromagnetics. 1998;19:20–31. [PubMed] [Google Scholar]
- 15.Marques JP, Gruetter R, van der Zwaag W. In vivo structural imaging of the cerebellum, the contribution of ultra-high fields. Cerebellum. 2012;11:384–91. doi: 10.1007/s12311-010-0189-2. [DOI] [PubMed] [Google Scholar]
- 16.Schmahmann JD, Weilburg JB, Sherman JC. The neuropsychiatry of the cerebellum - insights from the clinic. Cerebellum. 2007;6:254–67. doi: 10.1080/14734220701490995. [DOI] [PubMed] [Google Scholar]
- 17.Steinlin M. Cerebellar disorders in childhood: Cognitive problems. Cerebellum. 2008;7:607–10. doi: 10.1007/s12311-008-0083-3. [DOI] [PubMed] [Google Scholar]
- 18.Schmahmann JD. Disorders of the cerebellum: ataxia, dysmetria of thought, and the cerebellar cognitive affective syndrome. J Neuropsychiatry Clin Neurosci. 2004;16:367–78. doi: 10.1176/jnp.16.3.367. [DOI] [PubMed] [Google Scholar]
- 19.Patel BN, Dunn RJ, Jeong SY, Zhu Q, Julien JP, David S. Ceruloplasmin regulates iron levels in the CNS and prevents free radical injury. J Neurosci. 2002;22:6578–86. doi: 10.1523/JNEUROSCI.22-15-06578.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Grisoli M, Piperno A, Chiapparini L, Mariani R, Savoiardo M. MR imaging of cerebral cortical involvement in aceruloplasminemia. AJNR Am J Neuroradiol. 2005;26:657–61. [PMC free article] [PubMed] [Google Scholar]
- 21.Bedir R, Tumkaya L, Sehitoğlu I, Kalkan Y, Yilmaz A, Sahin OZ, et al. The effect of exposure of rats during prenatal period to radiation spreading from mobile phones on renal development. Ren Fail. 2015;37:305–9. doi: 10.3109/0886022X.2014.985995. [DOI] [PubMed] [Google Scholar]
- 22.Coskun G, Özgür H. Molecular Mechanism of Apoptosis and Necrosis [Article in Turkish] Arsiv Kaynak Tarama Derg. 2014;20:145–58. [Google Scholar]
- 23.Sonmez OF, Odaci E, Bas O, Kaplan S. Purkinje cell number decreases in the adult female rat cerebellum following exposure to 900 MHz electromagnetic field. Brain Res. 2010;1356:95–101. doi: 10.1016/j.brainres.2010.07.103. [DOI] [PubMed] [Google Scholar]
- 24.Topal Z, Hanci H, Mercantepe T, Erol HS, Keles ON, Kaya H, et al. The effects of prenatal long-duration exposure to 900-MHz electromagnetic field on the 21-day-old newborn male rat liver. Turkish J Med Sci. 2015;45:291–7. doi: 10.3906/sag-1404-168. [DOI] [PubMed] [Google Scholar]


