Abstract
Background
Patent ductus arteriosus (PDA) is a common congenital cardiac abnormality in premature infants. In low-birth-weight infants weighing less than 2500 g, if the PDA continues to open, abnormal circulation hemodynamics and pulmonary edema may occur. This study aimed to investigate the role of lung ultrasound score in the assessment of pulmonary edema in low-weight neonates with PDA.
Material/Methods
Two hundred and twenty-one neonates with low birth weight were selected as the subjects, children with PDA as the observation group, and children with closed ductus arteriosus as the control group. On the fourth postnatal day, lung ultrasound examination and 6-segment lung ultrasound scoring were performed.
Results
All 221 infants (94 in the observation group, 127 controls) underwent ultrasound examinations of the lungs. Intergroup differences in gestational age, birth weight, length of hospital stay, and left ventricular ejection fraction were not statistically significant. There was a significant difference in lung ultrasound score (t=0.005, P=0.000) and aortic root ratio to left atrial (t=0.085, P=0.000), which was negatively correlated with gestational age (r=−0.235, P=0.000) and positively correlated with PDA diameter (r=0.261, P=0.011).
Conclusions
Low-birth-weight children often have PDA. Its continued opening changes the circulation hemodynamics in children. Lung ultrasound score can semi-quantitatively evaluate the extravascular lung water content, identifying the need to intervene and follow up the hemodynamic significance of PDA over time.
MeSH Keywords: Low-Birth-Weight Neonates, Lung Ultrasound Score, Patent Ductus Arteriosus
Background
Patent ductus arteriosus (PDA) is a common congenital cardiac abnormality in premature infants. The younger the gestational age and lower the body weight, the higher the incidence of PDA [1]. PDA can cause pulmonary circulation, left cardiac overload, and insufficient systemic perfusion, resulting in circulatory hemodynamic changes [2]. The organs of low-birth-weight infants weighing less than 2500 g are not well developed, and their incidence of PDA is high. If PDA continues to open, abnormal circulation hemodynamics, pulmonary edema, and pulmonary hypertension may occur, resulting in or aggravating hypoxia and causing adverse outcomes. It is essential to establish a reliable hemodynamic evaluation method for low-weight premature infants with continuously opening PDA.
The lung is considered a blind area of ultrasound; however, in recent years, ultrasonography has been used in the diagnosis of lung diseases. Some studies [3,4] found that changes in the ratio of the water content of the alveoli and stroma can confound ultrasound images and create defects, thus reflecting the pathological changes of the lung parenchyma. In recent years, a growing number of studies have shown that lung ultrasound can be used to diagnose and identify anti-diastole in adult lung diseases, as well as to evaluate neonatal lung diseases such as neonatal respiratory distress syndrome, pneumonia of the newborn, wet lung of the newborn, and pulmonary hemorrhage of the newborn [5,6]. The International Consensus on the Diagnosis of Lung Diseases (2012) [7] and the Guidelines for the Diagnosis of Neonatal Lung Disease (2019) [8] indicated that lung ultrasound can be used to diagnose a variety of pulmonary diseases, with higher sensitivity and accuracy than conventional chest X-ray examinations. Using lung ultrasound can quantitatively evaluate pulmonary water content [9]. However, few studies have examined the hemodynamic status of low-birth-weight infants with PDA using lung ultrasound. This study aimed to evaluate the clinical significance of lung ultrasound for evaluating the prognosis of low-birth-weight infants with PDA using lung ultrasound score (LUS).
Material and Methods
Subjects
From January 2018 to December 2019, lung ultrasonography was performed on low-birth-weight neonates admitted to the Department of Neonatology of the Affiliated Hangzhou First People’s Hospital, Zhejiang University School of Medicine. Inclusion criteria were: premature infants with PDA and a birth weight less than 2500 g; and admission to our hospital within 24 hours of birth. Exclusion criteria were: children with congenital heart disease confirmed by cardiac ultrasound except PDA and patent foramen ovale; children with definite congenital malformations and other chromosome abnormalities diagnosed prenatally; and neonates with pneumothorax and lung bleeding. The premature infants with similar gestational age and a closed arterial line were selected as the control group. The examination was completed on the fourth day of the post-partum period. This study was reviewed by the hospital’s ethics committee (no. 2018-30-1). Informed consent was provided by each child’s immediate family or guardian prior to the examination. All methods were performed in accordance with the Declaration of Helsinki.
Methods
Instrument: Philips CX50 digital Doppler ultrasonic diagnostic apparatus, neonatal cardiac ultrasound probe (12 MHz), high-frequency shallow probe (12 MHz), and micro-convex probe (8 MHz).
Inspection method: The supine and lateral supine positions were taken. Each lung was divided into 3 areas: anterior superior, anterior inferior, and lateral. There were 6 total lung regions. Horizontal and longitudinal scans were taken. The scans were examined by the same ultrasound physician with experience in pediatric lung ultrasound.
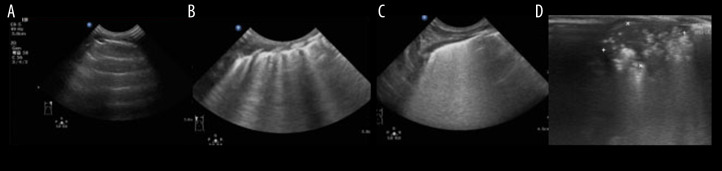
Each area was graded as described by Brat et al. [10]: Pulmonary sliding sign with A line or a small amount of B line, 0 points; more than 3 B lines, spacing between B lines, no fusion, 1 point; diffused B line and fusion B line involving a small amount of subpleural consolidation, 2 points; and wide range of consolidation areas, 3 points. Each region was rated with the most serious performance. The LUS was the sum of the scores of each region, and the highest score of the 6 segments was 18 points (Figure 1).
Figure 1.
(A) Line A, ultrasound score: 0 point. (B) Irregular line B; ultrasound score: 1 point. (C) Diffuse fusion line B; ultrasound score: 2 points. (D) Local solid change, ultrasound score: 3 points.
Statistical methods
The statistical analysis was performed using SPSS version 21.0 software. The t test was used to compare LUS between the ductus arteriosus closure group and the PDA group, while Pearson analysis was used to analyze the correlation between LUS and the data of each group. The ROC curve was established using the sensitivity of the dual lung score to diagnose PDA as the ordinate and 1-specificity as the abscissa.
Results
Comparison between the experimental group and the control group
Two hundred and twenty-one infants were considered eligible and underwent ultrasound examinations of the lungs. Among them, there were 94 cases of low-birth-weight infants with PDA, 51 females, and 43 males. The mean gestational age was 30.90±3.56 weeks, mean birth weight was 1568.94±630.06 g, mean hospital stay was 33.09±23.20 days, mean age at examination was 4.13±1.12, mean lung ultrasound score was 8.88±3.56, lowest score was 2 points, and highest score was 15 points. Two patients died, one with a lung score of 14 and the other with a score of 7. Another patient could not be weaned and underwent PDA ligation. There were 127 cases in the controls group (63 females, 63 males), which had a mean gestational age of 30.33±2.81 weeks, mean birth weight of 1434.69±485.66 g, mean hospitalization stay of 35.71±19.66 days, and mean lung ultrasound score of 6.75±3.66. The mean age at examination was 4.50±1.26 days. There was no significant intergroup difference in gestational age, birth weight, hospitalization length, or left ventricular ejection fraction between the 2 groups, but there was a significant intergroup difference in lung ultrasound score and aortic root ratio to left atrial (Table 1).
Table 1.
Comparison of patient characteristics between patent ductus arteriosus and ductus arteriosus closure groups.
| Group | N | Age (days) | Gestational age (weeks) | Weight (kg) | AO/LA | EF (%) | Hospital stay length (days) | LUS |
|---|---|---|---|---|---|---|---|---|
| Patent ductus arteriosus | 127 | 4.13±1.12 | 30.90±3.56 | 1.56±0.63 | 0.83±0.12 | 63.38±5.88 | 33.09±23.20 | 8.88±3.56 |
| Ductus arteriosus closure | 94 | 4.50±1.26 | 30.33±2.81 | 1.43±0.48 | 0.88±0.14 | 64.31±5.95 | 35.71±19.66 | 6.75±3.66 |
| t | 0.776 | 13.094 | 15.819 | 0.085 | 0.012 | 4.784 | 0.005 | |
| P | 0.444 | 0.199 | 0.087 | 0.001 | 0.249 | 0.377 | 0.000 |
Ultrasonic imaging of lungs
In this study, there were different degrees of B line (100%), among which 52 cases showed a diffuse B line in the anterior and lateral chest (24%), with 20 cases of closure and 32 cases of non-closure. Thirty-three cases had different degrees of solid change (15%), with 12 cases of PDA closure and 21 cases of non-closure. No pleural effusions were detected during the examination.
Correlation analysis
The correlation coefficients between lung ultrasound score and gestational age, birth weight, hospitalization length, left ventricular ejection fraction, aortic root ratio to left atrial, and PDA diameter were −0.235, −0.205, 0.268, 0.080, −0.338, and 0.261, respectively. Lung ultrasound score was negatively correlated with gestational age (r=−0.235, P=0.000), negatively correlated with birth weight (r=−0.205, P=0.002), negatively correlated with aortic root ratio to left atrial (r=−0.338, P=0.000), and unrelated to left ventricular ejection fraction (r=0.080, P=0.234); and positively correlated with PDA inner diameter (r=0.261, P=0.011) and length of stay (r=0.268, P=0.000).
ROC curve for assessing whether the pulmonary artery catheter is closed with the lung ultrasound score
When the Youden index was 0.264, the area under the ROC curve was 0.668, and the cut-off value was 9.5, its sensitivity and specificity was 50% and 76.4% (Figure 2).
Figure 2.
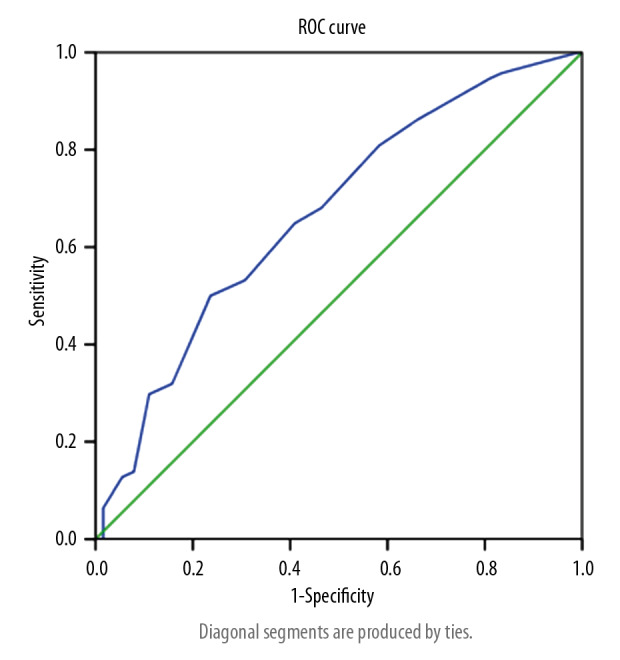
ROC curve for assessing whether the pulmonary artery catheter is closed with the double lung score. The area under the ROC curve is 0.668. When the cut-off value of the lung score is 9.5, the sensitivity of the predicted PDA is 50% and the specificity is 76.4%.
Discussion
In premature infants, the arterial duct tissue is immature and PDA often occurs. When PDA occurs, blood is shunted from the aorta to the pulmonary artery through the arterial catheter during systole and diastole. Persistent severe left-to-right shunts cause left ventricular enlargement and diastolic blood pressure to increase, resulting in congestion of the left atrium and pulmonary vascular bed, causing pulmonary edema. In recent years, lung ultrasound has been widely used in the diagnosis of neonatal lung diseases. B line can better reflect the degree of pulmonary edema, but the utility of lung ultrasound score in diagnosis is controversial [11]. This study attempted to use LUS to assess extravascular lung water content in neonates with PDA and to analyze the prognosis. We found that lung ultrasound score is correlated with gestational age, birth weight, and length of hospitalization, so it can help clinicians analyze the prognosis of children. In the evaluation of PDA-related conditions, the lung ultrasound score is correlated with the inner diameter of the PDA and the ratio of the aorta to the left atrium, indicating that it can semi-quantitatively evaluate the degree of pulmonary edema and can indirectly reflect the increase in left atrial pressure. Therefore, it can help clinicians understand the severity of the disease and help observe the evolution of the disease. However, the sensitivity of the ROC curve is not high and it cannot accurately reflect the severity of the child’s condition.
The fourth postnatal day was selected as the time of examination. Because of the rapid decrease in pulmonary artery pressure after birth, the pulmonary artery pressure decreased rapidly, but it remained beyond the normal adult level (<30 mmHg) [12] at 72 h. Therefore, it is possible to reduce the effect of hemodynamic changes on the results after the third postnatal day. In addition, the newborn’s condition changes quickly, and an examination at a different time may result in a different score; therefore, the examination was completed on the fourth postnatal day.
There are different methods of zoning for lung ultrasound examination that use 12-zone and 6-zone scores. The 6-zone score was selected for this study. First, the body surface area of low-weight premature children is small, and the selection of a 12-zone score may lead to overlap of the examination areas. Second, some children who required respiratory aids are at an increased risk of extubation when turned over during an ultrasound examination. In addition, the hemodynamic instability caused by PDA patency was mainly pulmonary edema [13,14]. The dense B line was mainly located in the anterior chest, and the main lesion area could be observed with the use of 6 areas.
No significant differences in birth age, birth weight, length of hospital stay, or left ventricular ejection fraction were seen between low-birth-weight neonates with open versus closed PDA. However, significant intergroup differences were seen in lung ultrasound scores. There was no correlation between LUS and birth age, birth weight, length of hospitalization, or left ventricular ejection fraction, but the inner diameter of the LUS and PDA were positively correlated. Therefore, lung ultrasound can be used to assess the early lung condition of low-birth-weight neonates with PDA. Lung ultrasound score was negatively correlated with aortic root ratio to left atrial. The ratio of left atrium to aorta can indirectly reflect left atrial pressure. Increased left atrial pressure can cause anterior blood flow in the pulmonary veins to be blocked, resulting in increased pulmonary circulation resistance and increased lung fluid. Cardiac ultrasound with lung ultrasound indirectly evaluates the cycle stability of the PDA and provides a basis for early intervention, with hemodynamic significance.
In this study, the mean lung ultrasound score (8.88±3.56) was significantly higher in the PDA group than in the ductus arteriosus closure group (6.75±3.66). When the newborn has a continuous unclosed arterial catheter, blood is diverted from the aorta to the pulmonary artery. The increase in pulmonary artery pressure, pulmonary congestion, and pulmonary venous pressure lead to pulmonary congestion, which leads to increased pulmonary interstitial fluid, dyspnea, and heart failure [15]. Because the number of B lines can reflect extravascular lung water, LUS can be used to evaluate the aggravation of pulmonary edema in PDA.
Because lung ultrasound is simple and can effectively minimize radiation exposure, it has been widely used in recent years [16,17]. Some experts believe that ultrasound is more sensitive, accurate, and reliable than traditional X-ray in the diagnosis of neonatal lung diseases [18–21].
A number of epidemiological studies have shown that PDA is a risk factor [22,23] that occurs in bronchopulmonary dysplasia (BPD). PDA with hemodynamic significance can also lead to an increased risk of pulmonary bleeding in premature infants [24]. Therefore, some experts used pulmonary ultrasound to evaluate BPD [25] and respiratory distress syndrome [26] in low-birth-weight neonates. However, the pulmonary ultrasound findings were not specific. All of the studied children had different degrees of B line, considering that their lung water contents were relatively high. In addition to pulmonary edema caused by PDA patency, the wet lung of neonates could also show an increase in B line. Common respiratory distress syndrome occurs in low-weight preterm infants as well as in those with meconium inhalation or neonatal pneumonia, and lung ultrasound can show pulmonary consolidations. Therefore, ultrasound findings must be combined with clinical specific analysis results.
Simple ultrasound showed no quantitative assessment of the lung condition. In 2015, Brat first proposed an application [10] for direct exogenous pulmonary surfactant and continuous positive airway pressure ventilation with a lung ultrasound score [10]. In 2018, Rouby further introduced this scoring system in detail [27], but the semi-quantitative method had many influencing factors [28]. This study showed that in the case of PDA-related disease assessment, the lung ultrasound score could be used for the semi-quantitative assessment of the degree of pulmonary edema, which could help the clinician understand disease severity and benefit the observation of the condition. Some scholars do not recommend the use of lung ultrasound scores in newborns. They believe that for severe diseases such as pneumothorax and pulmonary hemorrhage, lung ultrasound scores cannot reflect the severity of the disease, but they do not deny that lung scores can quantitatively evaluate pulmonary edema [11]. This study excluded diseases such as pneumothorax and pulmonary hemorrhage. The main research object was low-birth-weight infants with PDA. The purpose was not to diagnose, but to evaluate extravascular lung water content in specific diseases.
This study showed that when the cut-off value of the double lung score of the ROC curve is 9.5, the sensitivity of the predicted PDA is 50% and the specificity is 76.4%. The use of lung ultrasound scores to determine whether a PDA is open is unreliable. The neonatal lung is complicated, and various diseases can lead to increased lung water content. Therefore, the specificity of lung ultrasound scores is not particularly high.
There are some defects in this study. First, the lung ultrasonic score was semi-quantitative, and there are no accepted quantitative standards for the number of B lines and the scope of the change of the lung. Second, the scores were affected by the subjective factors of the operator, and the scores for the same child may differ among operators. The scans in this study were assessed by the same sonographer. However, it was difficult to collect large quantities of data at multiple centers. Third, low-birth-weight neonates often have other lung problems and a variety of complex factors that can affect score accuracy.
Conclusions
Low-birth-weight neonates often have PDA. A continuously opening PDA affects hemodynamic changes of the circulation in children. Cardiac ultrasound can observe whether the PDA is closed and the thickness of the catheter. The left atrial pressure is estimated indirectly based on the ratio of the left atrium and the aorta. Lung ultrasound scores can semi-quantitatively evaluate extravascular lung water content, but have limited value for hemodynamic evaluation. Relying solely on lung ultrasound scores cannot accurately reflect the severity of the child’s condition, and it is necessary to further combine the patient’s lung ultrasound performance and medical history. The 6-segment lung scoring combined with echocardiography is simple and noninvasive. It can be used as an effective method for early diagnosis of hemodynamically significant PDA in neonates, assessing the severity of the disease and observing the prognosis, and provides a more timely and intuitive imaging basis for clinical use. It can dynamically monitor the effect of clinical treatment, reduce the number of X-ray follow-up visits in the short term, and reduce the exposure of newborns and NICU medical staff to X-rays.
Acknowledgments
The authors thank Dr. Ding Zhongxiang for his help in editing the article.
Footnotes
Source of support: This study was supported by Zhejiang Medical and Health Science and Technology Project (2020ky690) and Zhejiang Natural Science Public Welfare Fund (LGF20H020004)
References
- 1.Benitz WE Committee on Fetus and Newborn, American Academy of Pediatrics. Patent ductus arteriosus in preterm infants. Pediatrics. 2016;137(1) doi: 10.1542/peds.2015-3730. [DOI] [PubMed] [Google Scholar]
- 2.Almeida-Jones M, Tang NY, Reddy A, Zahn E. Overview of transcatheter patent ductus arteriosus closure in preterm infants. Congenit Heart Dis. 2019;14(1):60–64. doi: 10.1111/chd.12712. [DOI] [PubMed] [Google Scholar]
- 3.Kurepa D, Zaghloul N, Watkins L, et al. Neonatal lung ultrasound exam guidelines. J Perinatol. 2017;38(1):11–22. doi: 10.1038/jp.2017.140. [DOI] [PubMed] [Google Scholar]
- 4.Lichtenstein DA. Current misconceptions in lung ultrasound: A short guide for experts. Chest. 2019;156(1):21–25. doi: 10.1016/j.chest.2019.02.332. [DOI] [PubMed] [Google Scholar]
- 5.De Martino L, Yousef N, Ben-Ammar R, et al. Lung ultrasound score predicts surfactant need in extremely preterm neonates. Pediatrics. 2018;142(3):e20180463. doi: 10.1542/peds.2018-0463. [DOI] [PubMed] [Google Scholar]
- 6.Liu J, Cao HY, Fu W. Lung ultrasonography to diagnose meconium aspiration syndrome of the newborn. J Int Med Res. 2016;44:1534–42. doi: 10.1177/0300060516663954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Felippe P, Cassiano F. Lung ultrasound in critically ill patients: A new diagnostic tool. J Bras Pneumol. 2012;38:246–56. doi: 10.1590/s1806-37132012000200015. [DOI] [PubMed] [Google Scholar]
- 8.Liu J, Copetti R, Sorantin E, et al. Protocol and guidelines for point-of-care lung ultrasound in diagnosing neonatal pulmonary diseases based on international expert consensus. J Vis Exp. 2019;(145) doi: 10.3791/58990. [DOI] [PubMed] [Google Scholar]
- 9.Zong HF, Guo G, Liu J, et al. Using lung ultrasound to quantitatively evaluate pulmonary water content. Pediatr Pulmonol. 2020;55(3):729–39. doi: 10.1002/ppul.24635. [DOI] [PubMed] [Google Scholar]
- 10.Brat R, Yousef N, Klifa R, et al. Lung ultrasonography score to evaluate oxygenation and surfactant need in neonates treated with continuous positive airway pressure. JAMA Pediatr. 2015;169:e151797. doi: 10.1001/jamapediatrics.2015.1797. [DOI] [PubMed] [Google Scholar]
- 11.Liu J. The lung ultrasound score cannot accurately evaluate the severity of neonatal lung disease. J Ultrasound Med. 2019 doi: 10.1002/jum.15176. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 12.Sharma D, Farahbakhsh N. Role of chest ultrasound in neonatal lung disease: A review of current evidences. J Matern Fetal Neonatal Med. 2019;32:310–16. doi: 10.1080/14767058.2017.1376317. [DOI] [PubMed] [Google Scholar]
- 13.Clyman RI. Patent ductus arteriosus, its treatments, and the risks of pulmonary morbidity. Semin Perinatol. 2018;42(4):235–42. doi: 10.1053/j.semperi.2018.05.006. [DOI] [PubMed] [Google Scholar]
- 14.Letshwiti JB, Semberova J, Pichova K, et al. A conservative treatment of patent ductus arteriosus in very low birth weight infants. Early Hum Dev. 2017;104:45–49. doi: 10.1016/j.earlhumdev.2016.12.008. [DOI] [PubMed] [Google Scholar]
- 15.Hu Q, Ren WD, Mao J, et al. Changes in pulmonary artery pressure during early transitional circulation in healthy full-term newborns. Ultrasonics. 2015;56:524–29. doi: 10.1016/j.ultras.2014.10.005. [DOI] [PubMed] [Google Scholar]
- 16.Liu J, Chen SW, Liu F, et al. The diagnosis of neonatal pulmonary atelectasis using lung ultrasonography. Chest. 2015;147:1013–19. doi: 10.1378/chest.14-1306. [DOI] [PubMed] [Google Scholar]
- 17.Tavares J, Ivo R, Gonzalez F, et al. Global Ultrasound Check for the Critically lll (GUCCI) – a new systematized protocol unifying point-of-care ultrasound in critically ill patients based on clinical presentation. Open Access Emerg Med. 2019;11:133–45. doi: 10.2147/OAEM.S199137. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 18.Iorio G, Capasso M, De Luca G, et al. Lung ultrasound in the diagnosis of pneumonia in children: Proposal for a new diagnostic algorithm. Peer J. 2015;3:e1374. doi: 10.7717/peerj.1374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jones BP, Tay ET, Elikashvili I, et al. Feasibility and safety of substituting lung ultrasound for chest X-ray when diagnosing pneumonia in children: A randomized controlled trial. Chest. 2016;150:131–38. doi: 10.1016/j.chest.2016.02.643. [DOI] [PubMed] [Google Scholar]
- 20.Noonan M, Turek JW, Dagle JM, et al. Intraoperative high-frequency jet ventilation is equivalent to conventional ventilation during patent ductus arteriosus ligation. World J Pediatr Congenit Heart Surg. 2017;8:570–74. doi: 10.1177/2150135117717974. [DOI] [PubMed] [Google Scholar]
- 21.Chen SW, Fu W, Liu J, et al. Routine application of lung ultrasonography in the neonatal intensive care unit. Medicine. 2017;96:e5826. doi: 10.1097/MD.0000000000005826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Vakrilova L, Hitrova S, Dimitrova V, et al. Bronchopulmonary dysplasia in very low birth weight infants – the old new problem. Akush Ginekol (Sofiia) 2015;54:29–36. [PubMed] [Google Scholar]
- 23.Schena F, Francescato G, Cappelleri A, et al. Association between hemodynamically significant patent ductus arteriosus and bronchopulmonary dysplasia. J Pediatr. 2015;166:1488–92. doi: 10.1016/j.jpeds.2015.03.012. [DOI] [PubMed] [Google Scholar]
- 24.Sivanandan S, Agarwal R. Pharmacological closure of patent ductus arteriosus: Selecting the agent and route of administration. Paediatr Drugs. 2016;18:123–38. doi: 10.1007/s40272-016-0165-5. [DOI] [PubMed] [Google Scholar]
- 25.Alonso-Ojembarrena A, Lubián-López SP. Lung ultrasound score as early predictor of bronchopulmonary dysplasia in very low birth weight infants. Pediatr Pulmonol. 2019;54:1404–9. doi: 10.1002/ppul.24410. [DOI] [PubMed] [Google Scholar]
- 26.Pang H, Zhang B, Shi J, et al. Diagnostic value of lung ultrasound in evaluating the severity of neonatal respiratory distress syndrome. Eur J Radiol. 2019;116:186–91. doi: 10.1016/j.ejrad.2019.05.004. [DOI] [PubMed] [Google Scholar]
- 27.Rouby JJ, Arbelot C, Gao Y, et al. Training for lung ultrasound score measurement in critically ill patients. Am J Respir Crit Care Med. 2018 doi: 10.1164/rccm.201802-0227LE. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Taveira M, Yousef N, Miatello J, et al. [Can a simple lung ultrasound score predict length of ventilation for infants with severe acute viral bronchiolitis]. Arch Pediatr. 2018;25:112–17. doi: 10.1016/j.arcped.2017.11.005. [DOI] [PubMed] [Google Scholar]