Abstract
In the present report, we have broadly outlined the potential advances in the field of skull base surgery, which might occur within the next 20 years based on the many areas of current research in biology and technology. Many of these advances will also be broadly applicable to other areas of neurosurgery. We have grounded our predictions for future developments in an exploration of what patients and surgeons most desire as outcomes for care. We next examined the recent developments in the field and outlined several promising areas of future improvement in skull base surgery, per se, as well as identifying the new hospital support systems needed to accommodate these changes. These include, but are not limited to, advances in imaging, Raman spectroscopy and microscopy, 3-dimensional printing and rapid prototyping, master–slave and semiautonomous robots, artificial intelligence applications in all areas of medicine, telemedicine, and green technologies in hospitals. In addition, we have reviewed the therapeutic approaches using nanotechnology, genetic engineering, antitumor antibodies, and stem cell technologies to repair damage caused by traumatic injuries, tumors, and iatrogenic injuries to the brain and cranial nerves. Additionally, we have discussed the training requirements for future skull base surgeons and stressed the need for adaptability and change. However, the essential requirements for skull base surgeons will remain unchanged, including knowledge, attention to detail, technical skill, innovation, judgment, and compassion. We believe that active involvement in these rapidly evolving technologies will enable us to shape some of the future of our discipline to address the needs of both patients and our profession.
Key words: Artificial intelligence, Genetic engineering and antitumor antibodies, Raman spectroscopy, Skull base surgery, Stem cell technology
Abbreviations and Acronyms: AI, Artificial intelligence; COVID-19, Coronavirus disease 2019; CNS, Central nervous system; CT, Computed tomography; H&E, Hematoxylin and eosin; ICU, Intensive care unit; MRI, Magnetic resonance imaging; OR, Operating room; RS, Raman spectroscopy
Introduction
Surgery for tumors and vascular lesions at the base of the brain has existed since the time of Harvey Cushing; however, such operations were, at times, inadequate and extraordinarily high risk. In the 1980s and 1990s, a number of revolutions occurred as pioneering surgeons and physicians working together in small teams made major advances in the field. These surgeons developed critical innovations through new techniques to expose the skull base, remove tumors safely, repair complex aneurysms and vascular lesions, and safely reconstruct the skull base to promote healing and prevent cerebrospinal fluid leakage and infection. More recent technological introductions have proceeded to revolutionize the treatment of challenging skull base pathology, including the introduction of endoscopic surgery; advances in neuroimaging, radiosurgery, and high-energy focused radiotherapy; the perfection of vascular bypass to replace major arteries and venous sinuses involved by tumors; and the use of skull base approaches to treat complex vascular lesions.1, 2, 3
Through the establishment of organizations such as the North American Skull Base Society, the World Federation of Skull Base Society, and clinical institutions focused on the refinement and teaching of skull base surgery, the knowledge and skillset necessary to properly practice this challenging subspecialty have been effectively disseminated. This long history of innovation has resulted in the safe and effective practice of skull base surgery. However, the discipline remains on the cutting edge of neurosurgery, and many challenges have yet to be addressed. In the present report, we have surveyed the many emerging technologies that appear poised to result in the next revolution in skull base surgery. Many of the advances we have described will also be generally applicable to many areas of neurosurgery.
Although the future will always be difficult to predict, a specialist discussion of the most promising advances could help young surgeons entering the field and, in turn, help to shape the future. A number of techniques that might have an impact on skull base surgery are listed in Table 1 . In the present report, we have focused on some, but not all, of these areas. When thinking about the future of skull base surgery, we need to think about the present needs of patients and surgeons.
Table 1.
Some Areas of Future Advances in Skull Base Surgery
| Advanced imaging techniques, especially using magnetic resonance imaging and ultrasonography |
| Portable imaging technology in operating rooms and intensive care units |
| Simulated Raman microscopy and spectroscopy for rapid diagnosis in operating rooms |
| Three-dimensional printing and rapid prototyping |
| Tissue engineering to fabricate blood vessels, bone, facial tissue, and so forth in conjunction with 3-dimensional printing |
| Nanotechnology to engineer diagnostic and therapeutic particles |
| Rapid molecular and genetic diagnosis of tumors |
| Antitumor antibodies, CAR-T cells, and checkpoint inhibitors to treat malignant tumors |
| CRISPR-CAS-9–based genetic engineering techniques to eliminate inherited syndromes such as neurofibromatosis and von Hippel-Lindau disease |
| Stem cell technologies to repair damage caused by traumatic injuries, tumors, and iatrogenic injuries to the brain and cranial nerves |
| Master–slave and semiautonomous robots for use in operating rooms |
| Humanoid robots as helpers in operating rooms, cleaning services, food services, and nursing services in hospitals |
| Artificial intelligence applications for diagnosis of disease in hospitals and outpatient care facilities |
| Reengineered hospitals that are green, energy self-sufficient, with proper waste disposal and adapted to patients' needs |
| New training methods for residents and surgeons |
CAR-T, chimeric antigen receptor T cells; CRISPR, clustered regularly interspaced short palindromic repeats; Cas9, CRISPR-associated protein-9 nuclease.
What Do Patients and Surgeons Want?
Patients ultimately want their surgical team to cure, control, or, ideally, facilitate the prevention of disease. They favor minimally invasive approaches. When possible, they want illnesses to be treated by medicines only. If further intervention is necessary, they prefer minimally invasive surgery or radiosurgery without tissue damage. When more extensive surgery cannot be avoided, they prefer it to be without undue risk. Patients rightly place a premium on minimizing morbidity, which means no damage to the surrounding brain, cranial nerves, or blood vessels and no cosmetic deformity. Regardless of the approach, they want to minimize time away from work and family and to be treated at a reasonable cost.
Surgeons chose their profession to heal patients and to cure or control disease by performing elegant operations within their limits without major complications. They want to balance this pursuit with their desire to live well and to be healthy and happy with their families. Finally, they want the freedom to operate with the professional autonomy they have earned through their pursuit of highly specialized training without undue interference from the hospital administration or government in their daily practice of medicine.
The best way for surgeons to ensure that the needs of their future patients will be met is to continue to drive the innovation necessary to deliver transformative treatment options that are effective, economic, and minimally disruptive.
Advances in Imaging
During the past 2 decades, we have seen progressive improvements in imaging, and this will continue. At present, a 10.5-Tesla magnetic resonance imaging (MRI) unit is under study at the University of Minnesota's Center for Magnetic Resonance Research. This machine is experimental and has not yet been used for human studies. Also, a 7-Tesla magnet is in use at some centers in the United States and Europe, primarily for research purposes. It will soon be available for clinical use. We can study the brain anatomy, blood vessels, and fiber tracts in detail using this high-field MRI technology. It is likely that antigen or gene imaging of tumors will become commonplace as we find methods to image them.
Advanced imaging technology is also beginning to be used clinically in operating rooms (ORs). At present, hybrid ORs with MRI and intraoperative biplane angiography are in use at some centers for rapid diagnosis and corrections during surgical procedures. Using high-quality intraoperative angiography, a number of new or improved procedures have become possible, such as intraoperative tumor embolization and rapid corrections in vascular bypass procedures. Using intraoperative imaging, more complete tumor resection and the avoidance of some dangerous areas for resection have become possible. Many neurosurgeons and hospitals are still unsure whether to install this technology in their hospitals because the cost-effectiveness has not yet been demonstrated. However, the next 10–20 years will witness the growth of such imaging modalities in ORs and intensive care units (ICUs).
During surgery, surgeons often require rapid information about the pathology of the tissue they are removing and the tumor margins. The current technique of intraoperative pathological consultation will often be inadequate to answer these questions because of artifacts from tissue cryosectioning and subsequent conventional staining with hematoxylin and eosin (H&E). Various optical technologies have been developed that might find clinical implementation as intraoperative guidance tools in skull base neurosurgery, including optical coherence tomography, fluorescence-based technologies, Raman spectroscopy (RS) and imaging, diffuse reflectance spectroscopy and imaging, and advanced quantitative methods.4, 5, 6
RS is an optical nondestructive and noninvasive technique that provides biochemical information for the tissues studied based on inelastic scattering of light by vibrating molecules. Raman techniques have been identified as promising experimental intraoperative tools in neurosurgery.6 RS can accurately discriminate normal brain tissue from brain neoplasms by the biochemical alterations within tissues, which could help guide decision-making and surgical resection.7 , 8 Stimulated Raman histologic findings can create a complementary new direction for tissue diagnosis, independent of the conventional pathological findings, using an unprocessed label-free optical imaging method to provide the histologic diagnosis in near real-time. Using a fast simultaneous 2-channel/lipid channel at 2850 cm−1 and 3-channel/protein at 2930 cm−1 Raman shift wavenumbers, stimulated Raman scattering microscopy and a pseudo-H&E coloring method might allow pathologists to supplement or replace current frozen section diagnostic tools, which, in turn, has implications for tissue preservation and augmented diagnostic utility.6 , 8 An ex vivo skull base neoplasm examined using a Raman fiberoptic touch probe device to determine the biochemical “fingerprint” of the specimen is shown in Figure 1A and B . The same neoplasm examined using stimulated Raman scattering microscopy compared with a subsequent conventional tissue section stained with H&E is shown in Figure 1C and D. In addition, such imaging modalities can be combined with immediate treatment. Laser thermal ablation is already being used in the MRI suite for epileptic lesions and some brain tumors, with variable results.9 , 10 Lasers or ultrasonic removal can also be combined with rapid intraoperative pathological examination for precise intraoperative tissue removal.
Figure 1.
A patient with skull base chondrosarcoma. (A and B) Raman spectroscopy with an explanation of the various peaks present. (C) Hematoxylin and eosin–stained specimen. (D) Simulated Raman microscopy of fresh tissue from the patient.
Patients in the ICU often require imaging of the head, neck, and other areas of the body to evaluate structural and functional changes. Examples include the use of a portable computed tomography (CT) scanner for imaging studies, CT of the cerebral blood flow, and CT angiography of the brain or lungs.11 More recently, portable MRI technology has been developed and applied in the ICU setting.12 A tremendous amount of nursing effort, time delay, and threat to patient safety is involved in transporting a patient from the ICU to a fixed MRI unit at present. If the imaging quality is good, portable MRI devices could be used in large ICUs in a cost-effective manner.
Genetic Engineering and Antitumor Antibodies
Novel therapeutic approaches have begun to revolutionize cancer treatment.13 Skull base tumors have been considered primarily surgical diseases, and research of nonsurgical approaches to treatment has lagged behind other fields of oncology. With the advent of more sensitive techniques for profiling tumors and the natural transition from generic chemotherapy regimens to targeted approaches, it will be beneficial to apply molecular medicine approaches to skull base disease. A particular focus should be on diseases that are, at present, refractory to even the most advanced surgical techniques.
Skull base neoplasms are divisible into syndromic and sporadic disease processes. Central nervous system (CNS) tumor syndromes (e.g., neurofibromatosis types 1 and 2) have shown a specific predilection for skull base pathology.14 It is challenging to treat such diffuse disease processes in which the molecular basis is a germline mutation present throughout the body. The advent of genome editing techniques, in particular, the CRISPR (clustered regularly interspaced short palindromic repeats)-Cas9 (CRISPR-associated protein-9 nuclease)–based platforms,15 have created the potential for such interventions. CRISPR-Cas9 has already shown promise in editing hematopoietic lineage cells in stem cell transplantation and conferring human immunodeficiency virus resistance.16 One application to skull base tumors would be to correct pathologic point mutations in genes underlying CNS tumor syndromes, such as the NF2 gene, which is associated with multiple tumors in these patients.17 At present, we do not know whether correction of the NF2 gene would be sufficient to halt the growth of NF2-related tumors. As gene editing techniques become more refined, the focus will shift toward safe and specific delivery. For CNS applications, this might require the use of viruses engineered to have cell type-specific tropism to allow them to target the meninges or Schwann cells.18 When these issues have been resolved for syndromic diseases, such techniques could then be used to correct similar mutations in sporadic diseases as well.
Sporadic diseases exhibit multiple mutations, unlike syndromic diseases. They require broader approaches than does the treatment of syndromic diseases. Immunotherapy is an approach often used for other malignancies,19 using antibody-based drugs that target tumor-specific surface antigens. An example is the use of bevacizumab, an antibody that targets the angiogenic growth factor VEGF (vascular endothelial growth factor) in patients with NF2 and bilateral vestibular schwannomas.20 Medical treatment with bevacizumab has already shown some tumor stabilization and hearing preservation in such patients.21 Other antibody-based drugs include those that target immune checkpoint pathways, most commonly PD1 (programmed cell death 1), PDL1 (programmed cell death ligand 1), and CTLA4.22 These drugs serve as immunostimulatory molecules that might generate tumor-specific immune responses. A prerequisite for the use of such immunostimulatory molecules is the presence of unique neoantigens within the tumor, which are proteins generated by mutations that render them different from the normal host proteins.
Overall, lower grade meningiomas and schwannomas will have relatively few mutations, making them poor candidates for such approaches. However, higher grade meningiomas have a significantly greater baseline somatic mutation rate,23 making them potentially amenable to checkpoint inhibitor therapy. Another appealing approach is to perform a first intervention to facilitate antigen release and inflammation (e.g., using radiotherapy) and then a second intervention using checkpoint inhibitor therapy. Clinical trials using the combination of radiotherapy and checkpoint inhibitor therapy are currently being planned. Finally, directed therapy against specific surface antigens with engineered chimeric antigen receptor T cells could be used to target tumors highly specifically.24 This requires the identification of tumor-specific neoantigens and subsequent successful engineering of chimeric antigen receptor T cells able to specifically recognize and attack such an antigen. These approaches are under trial in other malignancies, including glioma, with varying degrees of success.25
The most important step in bringing novel therapeutic approaches to skull base pathology is the detailed molecular characterization of each of the pathologic entities that collectively constitute “skull base tumors.” Such efforts are underway and will expand as investigational techniques become more widely available and cost-effective. For skull base tumors, we will need to tailor our therapeutic interventions according to disease-specific and, even, patient-specific, mutational, transcriptional, and/or epigenetic profiles. This type of “precision medicine” will offer hope to patients for whom our current treatment paradigms are inadequate.
Stem Cell Technology
Stem cell recovery techniques after surgically induced and other neurological deficits will become commonplace within the next 20 years. This might promote more complete resection of some skull base tumors. Thus, in a patient requiring surgery for a vestibular schwannoma, recovery of cranial nerve 7 and 8 function might be better than at present even for those with large or giant tumors. This might also apply to iatrogenic neurological deficits involving the brain or brainstem after tumor or vascular operations. Understanding stem cell–tumor interactions might enable the prevention and cure of some skull base malignant neoplasms.
Different types of stem cells have been used for regenerative research, including embryonic stem cells, induced pluripotent stem cells, and perinatal (cord blood-derived) stem cells. We have already seen increased application of induced mesenchymal pluripotent stem cells and perinatal stem cells.
The problems with stem cell therapy are twofold. The goal is for the stem cells to transform into the target cells and to maintain potency after multiple divisions. However, their uncontrolled growth can result in neoplasms, which occurred after the unregulated use of fetal cell allografts for spinal cord injury in China (personal communication, Rostomily R, 2017). An immune-mediated rejection (host vs. graft or graft vs. host disease) can also occur. However, induced or genetically engineered mesenchymal stem cells are more likely to find early applications for neural repair.
Gary Steinberg and his team at Stanford University have used induced pluripotent stem cells for recovery after stroke, using the human neural stem cell product, NR1 stem cells, in phase I human trials. The Steinberg laboratory experiments have demonstrated that these stem cells will survive and migrate over time. In addition, patients with stroke have derived benefits even many years after the stroke.26 , 27 The effectiveness of the stem cells might result from local trophic factors secreted by the cells rather than by direct multiplication. A phase I/IIa open-label trial of the genetically modified adult bone marrow-derived cell SB623 was conducted at 2 U.S. university medical centers (personal communication, Steinberg GK, 2020, of press release from SanBio Co., Ltd., 2019). These cells were implanted stereotactically into the peri-infarction areas of the brain in patients with motor deficits due to ischemic stroke that had occurred 6–60 months previously. That study showed that such implants were safe, and clinically significant improvements were found using the European Stroke Scale, National Institutes of Health Stroke Scale, Fugl-Meyer motor function scale, and Fugl-Meyer assessment total score. However, a phase IIb trial of SB623 failed to achieve the primary endpoints for patients with ischemic stroke (personal communication, Steinberg GK, 2020, of press release from SanBio Co., Ltd., 2019).
These cells were more effective in patients with traumatic brain injury in a 12-month phase II, randomized controlled trial conducted at U.S. and Japanese centers. The SB623 cells had been implanted around the site of brain injury. The primary endpoint was reached in the study; the patients had achieved an average 8.3-point improvement compared with their baseline Fugl-Meyer motor scale compared with an improvement of 2.3 in the control group at 24 weeks (P = 0.040). The SanBio company is planning to study this further in phase III clinical trials (personal communication, Steinberg GK, 2020, of press release from SanBio Co., Ltd., 2019). Clinical trials with stem cells are similar to the development of new drugs or vaccines, will require investments of money and time, and many setbacks can be expected.
Three-Dimensional Printing
The use of 3-dimensional (3D) printing has been rapidly increasing in many industries. In the neurosurgical field, 3D printing is already used to make implants for cranioplasty, brain models, and aneurysm and tumor models for teaching and surgical planning.28 , 29 3D printing of patient-specific bioresorbable intravascular stents has been developed by the Northwestern University School of Engineering.30 A proof-of-concept 3D-printed bionic ear was developed using a cell-seeded hydrogel matrix in the anatomic shape of a human ear with an intertwined polymer of infused nanoparticles, which allowed for the culturing of cartilage tissue around an inductive coil antenna in the ear.31 The printed ear exhibited enhanced auditory sensing of radiofrequency.31
We envision increased applications for 3D printing and rapid prototyping of implants and stem cell-based bioimplants in neurosurgery in the near future.
Nanotechnology
Nanotechnology is the design and fabrication of submicroscopic particles 1–100 nano-microns in diameter. In nanomedicine, fabricated nanoparticles can be useful for diagnosis and targeted therapy of drugs, gene delivery, neuromodulation, and neural regeneration. However, many issues remain in the practical applications of this advance, similar to other new drugs.32
Artificial Intelligence
Artificial intelligence (AI) is a multidisciplinary science focused on developing computer systems able to perform tasks that would ordinarily require human intelligence. Human intelligence has several components. Sensory organs collect data about the environment, leading to perception and the formation of models that can be learned, remembered, and forgotten. Conscious thought links perception and modeling with a capacity for problem solving and action. Humans have highly evolved language, speech, and symbolism. Imagination is a unique ability, which can aid in problem solving by facilitating the creation of new cognitive experiences and the consideration of alternative or future realities in the mind. The harnessing of emotion and the interpretation of social interactions comprise a part of “emotional intelligence.” The subconscious includes the execution of cognitive processes required for normal functioning that do not rise to the level of awareness, including intuition, automation, and premonition. Using meditation, an individual is able to reach an elevated state of consciousness and practice the purposeful focus of attention. Finally, “consciousness,” or the awareness of self and one's ongoing experience, is poorly understood but has been shown to be computationally efficient and to confer a biological advantage by functioning as a “flexible response mechanism” by which data can be manipulated to produce decisions superior to those likely to be produced by automatic “subconscious” programs.33 , 34
Alan Turing, the mathematician and cryptanalyst, shortly after breaking the German encryption machine Enigma, established a vision for AI in his 1950 article “Computing Machinery and Intelligence.”35 From its inception in the 1950s, AI has branched into a dizzying array of fields. Most recently, progress has occurred in the development of 2 methods, known more specifically as machine learning and deep learning (Figure 2 ). In broad terms, machine learning is a method by which an algorithm is fed data and uses statistical techniques to “learn” how to become progressively better at a task for which it was not specifically programmed. Although machine learning has demonstrated promise for many purposes, it generally requires the user to identify and select certain “features” by which the algorithm will “learn” to complete a task—such as identifying a pattern within a high-dimensional dataset. The process of feature extraction and selection is time consuming and has necessarily limited the power of machine learning for certain applications. Deep learning is a subset of machine learning by which these “features” are not prespecified, and the algorithm identifies the optimal features for a classification task from the data without human intervention.27 These new methods have facilitated improved machine vision and natural language processing, which have yielded breakthroughs in medical imaging analysis, augmented clinical decision-making, and robotic performance. Better-than-human diagnostic accuracy has already been demonstrated for a variety of tasks, including classification of Alzheimer disease, dermatological lesions, and interpretation of basic neurophysiological reflexes.36, 37, 38
Figure 2.
The modern design of an artificial intelligence network compared with the human brain, which processes external signals and responds appropriately through the effector organs in the body.
AI could have tremendous implications in skull base surgery and health care. In the outpatient area, “chatbots” could be used for preoperative education and daily (or at a frequency determined by the patient) discourse with postoperative patients to discuss symptoms, evaluate wounds, provide psychological support, and report to health care providers when needed. Such chatbots have been successfully deployed to talk with patients with post-traumatic stress syndrome.39 AI can also be used to communicate with various types of providers and healthy populations of patients. In epidemic situations, AI could be used to rapidly detect the trends in the occurrence of disease, populations, movements of people, and so forth. Similar techniques can also be used to study referral patterns and for clinical research of outpatients.
In the hospital, AI will become increasingly essential. Robotics and AI will combine to influence every aspect of health care (see Medical Robotics). AI could be used in the ICU and general ward to monitor patient care, in addition to providing clinical decision-making support to doctors and nurses. For example, if a postoperative patient has developed a fever, severe headache, or rapid heart rate, AI could guide a nurse or resident doctor to act quickly in determining which investigations should be performed and what early actions are necessary, while waiting for the attending physician to respond. AI systems can progressively learn about the accuracy of such actions by examining the outcomes and then guide all clinicians about the utility of their actions.40 , 41
AI has made great strides in diagnostic radiology, and radiologists will soon work in tandem with AI tools to reduce the number of “human” reading required and make imaging diagnoses not previously possible.42 , 43 Additionally, AI tools can analyze images uploaded online from anywhere in the world, resulting in the democratization of radiological expertise and the provision of high-level diagnostic services in low-resource settings. Such diagnostics could also be combined with treatment algorithms, which could help primary care and emergency department physicians in choosing the appropriate referrals to specialists.44
In the past decade, great advances have occurred owing to the computing power delivered by handheld devices and the efficacy of internet-based search engines. This information is very useful to medical professionals. However, these data are usually unfiltered and have not been sorted by levels of evidence. An AI system could perform such filtering and sorting and deliver such information rapidly to physicians through portable devices.
Collaborative researchers from Harvard and the National Center for Science and Technology Beijing developed a water-based electrical circuit.45 This was injected into the brain of rats and connected to an external electrical circuit. The brain cells grew around it.45 A company called Neuralink (Neuralink Co., San Francisco, California, USA) has been established to develop implantable brain–computer interfaces. Extensive integration between advanced neuroelectrodes and ultraminiaturized electronics will make bidirectional brain–computer interfaces possible.46 Although still largely experimental, these types of advances will revolutionize medicine and surgery as practiced today.
The use of AI systems will be able to create medical education on demand to patients, physicians, trainees, and nursing professionals according to individual patient situations. For example, when a patient presents to the primary care physician's office with hearing loss and tinnitus, the diagnostic options could be presented to the primary care physician (Figures 3 and 4 ). Such a diagnostic workup could include an audiogram and MRI. If a vestibular schwannoma is diagnosed, an AI-based algorithm could educate the primary care physician (and the patient) about the treatment options available, such as observation, radiosurgery, or microsurgical removal. The AI system could also find specialists nearby who provide the needed services, their experience, and their outcomes. If desired, the AI system could schedule the next available appointment for the patient, with the possibility to override by the patient or physician. A trainee about to perform an operation could learn all the appropriate steps and potential pitfalls and complications for a tumor of its size and location by recalling information from a large video library. The trainee might even be able to practice some of the steps using virtual reality. Holographic images could further the understanding of anatomy and simulations of surgery. UpsurgeOn Academy (available at: Upsurgeon.com) is a hybrid simulation platform, integrating digital and physical tools for neurosurgical training. It includes a series of applications for teaching neurosurgical trainees many types of procedures. It also provides a physical neurosurgical simulation device, the Brainbox, which replicates human intracranial anatomy in morphology and consistency. The use of augmented reality further enhances the experience.47 Recent work by members of this group used AI techniques of optimization to develop surgical approaches to designated skull base pathologies, which have minimized morbidity and simultaneously maximized the distance from critical structures such as optic nerves and the interior carotid artery.48
Figure 3.
(A) Artificial intelligence (AI) evaluation and treatment to guide a patient and primary care physician (PCP) through the diagnosis, required investigations, and appropriate referrals. (B) The AI system can also help trainees before surgery, educate and prepare the patient and employer, and, finally, provide feedback to the primary care physician and specialist. CISS, constructive interference in steady state; FIESTA, fast imaging employing steady-state acquisition; CNS, central nervous system; MRI, magnetic resonance imaging.
Figure 4.
The Raven II table-mounted robot for abdominal surgery in use experimentally (not approved for use in humans). It is table mountable and can accommodate 4 arms. A rapid tool changer has also been designed to change the surgical equipment.
The potential of AI to transform the practice of skull base neurosurgery and medicine more generally cannot be overstated, and the focus of neurosurgeons and trainees should be in driving the optimal development and implementations of these applications.
Medical Robotics
Robotics will find increasing applications in all areas of surgery, including skull base and neurosurgery. To be adopted widely, a medical robot should be able to perform a task as well as or better than a human, should not cause any harm to the patient or human health care workers, should be able to adjust to the human environment, and should able to be rapidly cleaned and sterilized. Tasks requiring great accuracy, repetitive but monotonous actions, working in dangerous (e.g., infectious) environments, and requiring rapid scaling up or down are ideally suited for medical robotics.
Medical robots are divisible into 3 categories: passive robots or master–slave robots, active or autonomous robots, and semiautonomous robots. Master–slave robots are completely controlled by the surgeon. An example is the da Vinci robotic system (Intuitive Surgical, Sunnyvale, California, USA), now commonly used for urological, colorectal, gynecological, cardiac, and transoral surgery.49 Active robots are programmed to sense and evaluate data from their environment and complete their tasks appropriately. Two examples are the THINK Surgical Robot (Think Surgical, Inc., Fremont, California, USA) used in hip replacement surgery (previously called the ROBODOC) and the iROBOT (iRobot Corp., Bedford, Massachusetts, USA) used as an automatic vacuum cleaner. Semiactive robots are robots in which the surgeon retains partial control, and the robot provides assistive guidance in performing the procedure. The NeuroMate robotic system (Renishaw, Inc., Gloucestershire, United Kingdom) has been used for stereotactic procedures, including electrode implantation for deep brain stimulation, stereo-encephalography, and stereotactic biopsy.
The Rosa robot (Medtech-Zimmer Biomet Co., Warsaw, Indiana, USA) has been used for a variety of procedures, including stereoelectroencephalographic electrode placement and laser interstitial thermal therapy for epilepsy, without the need for a stereotactic frame. The advantage is that no human error in frame placement is possible. When performing multiple electrode placements, the time per electrode placement has decreased steadily when using a ROSA compared with a stereotactic frame. Also, a marked benefit was found with the use of the robot with the placement of ≥4 electrodes (personal communication, Andrew Ko, 2020). The Mazor X Stealth spine robot (Medtronic Co., Minneapolis, Minnesota, USA) has proved to be very useful for the precise placement of pedicle screws in transforaminal lumbar interbody fusion surgery and minimally invasive spine surgery.50
For cranial surgery, the NeuroArm developed by Garnette Sutherland, University of Calgary (Calgary, Alberta, Canada), is an elegant tool consisting of a robot controlled by the surgeon, from a distance if necessary, to perform surgical procedures inside the MRI suite.51 The Shinshu University NeuRobot (jointly developed by the Mechanical Engineering Research Laboratory, Hitachi Ltd., Tokyo Women's Medical University, University of Tokyo, and Shinshu University School of Medicine) is a master–slave micromanipulator system with a rigid endoscope and 3 robot arms that can be used through a small burr hole. It was successfully used for some surgical procedures and has demonstrated its feasibility for telesurgery between hospitals, albeit with a 1-ms delay.52
Surgical robotics has been a major topic of research at the University of Washington with collaborations between the School of Medicine and the School of Engineering. The RAVEN I and the RAVEN II are compact OR table-mounted surgical robots that were developed for abdominal minimally invasive surgery. They have been used primarily for research purposes (Figure 5 ).53 , 54 The collaboration among the Departments of Neurological Surgery, Electrical Engineering, Mechanical Engineering, and Otolaryngology led to the development of a bendable endoscopic sheath with tools such as the Roboscope (Figures 6 and 7 ). The same team developed an expandable snake-like introducer device for the Roboscope (Figure 8 ) and a remote console device to control these or other instruments patented by several of the investigators.55
Figure 5.
(A) The Roboscope is shown with the actuator mechanism. (B) The bendable sheath has 6 channels at present. The top 2 (SFE 1.4 mm) are for the 2 laser endoscopes, the middle 2 (2.0 mm) are for the instruments (Inst), and the bottom 2 (1.1 mm) are for suction (S) devices. The channels can be modified to suit the surgical requirements.
Figure 6.
(A) The Roboscope with 2 different dimensions (14 and 8 mm). (B) The Roboscope shown bent, with the 2 tools in close up.
Figure 7.
The Karns introducer device for the Roboscope is shown with (A) the tulip closed and (B) the tulip open.
Figure 8.
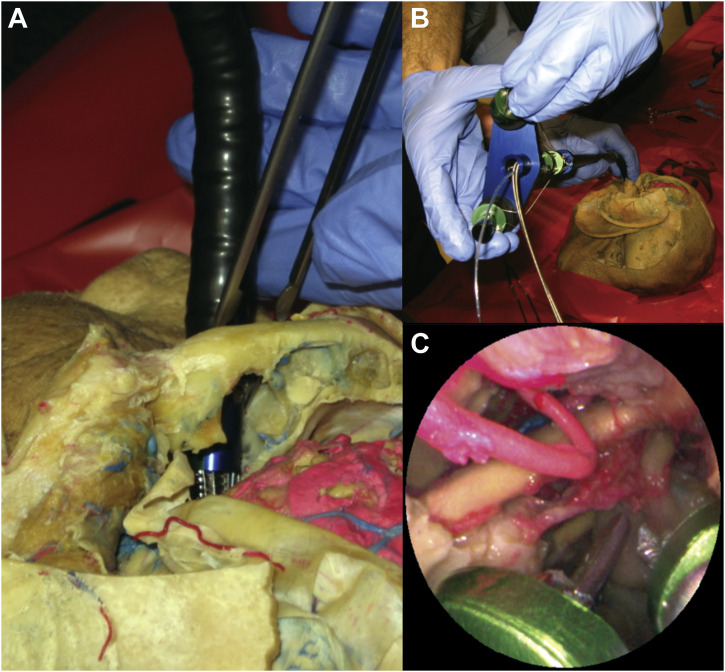
Cadaveric use of the Roboscope. (A) Introduction of the Roboscope through an opening in the skull base of a cadaver. (B) Remote manipulation of the controls. (C) The view of the structures through the laser fiberoptic endoscope (courtesy of Eric Seibel, PhD).
At present, a University of Washington team is working on an autonomous robot conceptually called the Artificially Intelligent Neurosurgical Robotic Assistant (Figure 9 ). The goal for this robot is for it to be able to perform functions typically performed by a microneurosurgical assistant, such as pulling on tissue gently and providing suction in a particular surgical space. The robotic assistant should act intuitively according to the surgeon's needs or from voice commands, similar to a human surgical assistant. The challenge in this project has been to decipher how the surgeon interacts with the assistant during an operation. The team has been recording the surgeon's voice and tool movement under the microscope and deciphering them using convolutional neural networks56 , 57 and Python speech application program interfaces, respectively. The instruments in a surgical field are visually identified based on their type and tracked across the frames at a pixel level58 (Figure 10 ). This information is used to predict the intended direction of movement of the surgeon while working with a surgical assistant. To provide an intuitive interface between the surgeon and the robotic assistant, parsing the medical terms is of prime importance. The speech processing community has principally focused on converting everyday speech to text. These models were fine-tuned to convert important medical speech terms for natural language parsing. In a recent study, the team demonstrated fusion of semiautonomous robotic therapy with a tumor-specific biomarker.59 , 60 “Tumor paint” is a biomarker specific to brain tumors derived from a component of scorpion toxin.61 In bench-top studies reported by Hu et al.,59 , 60 a robotic system was used to survey a simulated tumor margin for spots labeled for positive tumor cells and perform an automated ablation motion pathway after approval of the plan by the surgeon.
Figure 9.
Concept of the artificially intelligent robotic assistant showing (A) the surgeon and robotic assistant and (B) the surgeon, a human assistant, and a robotic assistant.
Figure 10.
Ground truth (GT) annotation for identifying instruments in a surgical field through the NeuroID dataset generated by the University of Washington team.45 (A) Input frame. (B) Annotations were created using the LabelMe annotation tool.58 (C) GT for distinguishing tool versus background (e.g., tissue, gauze). (D) GT for locating each class of instrument in pixel space.53
It is widely expected that the use of AI combined with robotics in the OR will increase during the next 20 years. The great benefits will be for procedures that require great precision, procedures performed through a small space, and procedures that require repetitive actions with precision (e.g., removal of a tumor over a large area). The use of AI could also assist in surgical decision-making regarding the apparent anatomy. During an operation, surgeons might be able to access a vast library of similar operations performed by them or other experts to help in difficult situations.
In Japan, humanoid robots, specifically nurse robots and robotic assistants, are being developed owing to a lack of medical personnel and caregiver resources. In the United States and other countries, such humanoid robots have also been developed for other uses. Hansen Robotics (Hansen Robotics Co., Hong Kong, China) has created human-looking and speaking humanoid robots, endowed with AI, notably those named Jules and Sophia. These robots have been given speech recognition skills, responses, and some other skills, although not all the elements of intelligence displayed by humans. Humanoid robotic nursing assistants will be developed and widely used in future because of health care worker shortages, patients' desire to have 24/7 nursing assistance, and the need to minimize human–human contact because of infectious diseases. Humanoid robots will find extensive applications in hospitals in the care of patients in ICUs and isolation wards. They can provide 24/7 nursing and transmit vital information to health care providers. Doctors and nurses can experience haptic feedback provided by robot-assisted examination of the patients. In turn, the patient will be able to feel the touch or soothing movements of nurses or loved ones not actually inside the room (Figure 11 ).
Figure 11.
Conceptualization of the android robotic nurse helper (NH) for a patient in isolation because of an infection. The physician (P) and nurse (N) are able to remotely view the patient and all vital signs, sense palpation using haptic sensors, and instruct the robotic nurse helper. The android robotic nurse helper is present with the patient continuously around the clock and is able to sterilize itself using ultraviolet (UV) light or other methods.
In China, an AI-powered robotic doctor was developed called Xiaoyi (“little doctor”), designed by iFlytek Co. (Hefei, China) and Tsinghua University (Beijing, China). This robot passed the medical licensing examination in China with high marks. The Fourth Medical Military University and Beihang University in Beijing have collaborated to devise a robot that can perform automated dental surgery. This robot demonstrated the ability to place 3D-printed dental implants in humans with 0.2–0.3-mm accuracy under the watchful eyes and supervision of human dentists (available at: https://wonderfulengineering.com/chinese-robot-just-performed-first-ever-fully-automated-dental-surgery/).
We expect that hospitals will also use robots to replace or supplement employees such as internal delivery workers, cleaners, and other positions inside the hospital, which require regular or rapid maintenance, such as with the hospital's sewage, water, and/or electrical supply. Cleaning of ORs or other hospital rooms that do not have patients in them can be performed efficiently and rapidly with ultraviolet light or other techniques using robots.62 , 63 Such robots will need to be supervised by their human counterparts. However, they will learn steadily with use, and such knowledge can be transmitted readily to other robots like them. It is to be expected that, initially, there will be resistance to the deployment of these robots. However, over time, people will gradually accept them, primarily because of the increased safety and lowered costs of health care, although both of these putative benefits will need to be demonstrated scientifically.
The human–robot interface and communication will create new problems and require special education and training to achieve efficacy in practice. Hospitals will require a new set of workers tasked with repairing and maintaining these robots, although eventually AI-powered robots themselves will be able to perform the repairs and maintenance.
Telemedicine-based consultations with patients and other doctors have already experienced a boost owing to the coronavirus disease 2019 (COVID-19) epidemic. An evolution of this could also be the performance of patient rounds using telemedicine by doctors who will be able to see all the vital signs and laboratory values of a patient and examine the patient with the assistance of a nurse, or a robot, transmitted live to the physicians.
Like all new technologies, the safety issues of AI and robotics will need to be studied carefully to protect other health care workers and our patients. The legal ramifications of errors by such beings will be intriguing.
Education of Students and Residents
Technology will transform the education of students, residents, and surgeons in the future. Students will be able to study anatomy and physiology in 3, 4, and 5 dimensions. The fourth dimension reflects the time-related changes in 3D images (e.g., carotid artery anatomy as the heart beats), along with physiology. The fifth dimension indicates the 3D images changing in time with simulated pathology. Advanced virtual reality and surgical simulations and one-on-one coaching by AI-enabled robots will supplement traditional teacher–student learning. Flexibility in adapting to the quickly evolving and increasingly sophisticated systems of cognitive offloading will be critical to student success. In the future, imagination, problem solving, and the ability to work in teams with diverse members (including AI-enabled robots) will be more important than the accumulation of knowledge. This is because massive knowledge storage will be available for quick access. Master AI brains will assist in all our medical work to varying degrees. This will reduce medical errors, increase work efficiency, and improve on the job learning.
The educational qualifications and training for trainee neurosurgeons will be different in the next decade. Mathematics, biology, physics, chemistry, and logical thinking will still form the building blocks of education in science. However, residents will also need to learn computer science, software development, robotics, and AI. Skills in video gaming and surgical simulations will be very beneficial. Studying ethics and philosophy and developing communication skills will also be important, because these qualities will distinguish humans and the various types of AI-enabled robots. Trainees and developing surgeons can reduce both physical and mental stress by the practice of meditation, yoga, Tai Chi, and so forth.
Hospital Care
When surgical procedures become less invasive and outpatient care has been enhanced to provide rapid patient access and monitoring, the need for hospitalizations will decrease. Methods to reduce the spread of infection to other patients and to health care workers will need to be developed. Techniques to reduce postoperative pain and immobility and technologies to provide home-based rehabilitation for patients will allow for shorter lengths of stay.
Hospitals of the next 20 years will be very different from the hospitals today. We envision that they will be smaller and closer to patients, with only the most difficult cases transferred to central hospitals. All hospitals will be environmentally friendly and carbon neutral, deriving their entire energy requirements from renewable energy. This will also apply to the products used in hospitals. Hospitals will be providing an enhanced and optimal healing environment for patients. For example, the patient rooms could be optimized to each patient with use of colors, plants, music, sunlight, and other features. Better methods of medical waste disposal will need to be developed to avoid environmental contamination and the spread of infection into communities.
The Future of Health Care Workers in the Age of Robotics and AI
In parallel with many other industries, fewer health care workers will be required to perform manual and highly repetitive jobs, and AI-enabled robots will replace some of these workers. Health care workers in hospitals will require greater skills and education. Hospital employees will be happier, work less, and supervise robotic workers. Fewer radiologists, pathologists, family doctors, and others will be required with the use of robotic assistants. Home visits could be performed by a humanoid AI-enhanced robot exhibiting great knowledge, empathy, and no prejudices. Surgeons, trainees, and other health care workers will also work collaboratively with such robots and AI systems because they will have become commonplace.
Surgeons will be performing less invasive, but more technically complex, procedures, with great emphasis on master–slave robots and supervision of autonomous robots performing procedures. A great role will exist for innovators and a constant retraining will be required for the newer procedures.
With the increasing use of AI and robotics, will humans still be involved in patient care? Because patients are humans, a need will always exist for humans to care for them. However, we believe we will see an evolutionary change in health care professionals during the next 20 years. Adaptability, cooperation in the work environment, compassion, and a special set of skills will be required of surgeons. Some of these will not be obvious until the new reality has emerged.
The current COVID-19 epidemic has suddenly enhanced the use of many technologies that had been developed but not deployed on a large scale. These include teleconsultations, teleworking, different requirements for sterilization of hospitals, and home based learning. The lead author's team has also developed a low-cost “home microsurgery laboratory” for resident trainees and proposed this as the seventh competency in resident training in the United States.64 Many of these changes will influence patient comfort and safety, the costs of medical care, and the need for particular types of health care workers.
Conclusions
Thirty years ago, skull base surgery did not exist as a specialty. Dramatic changes have occurred since then, not only in this field of surgery, but also in all areas of medicine, technology, and our lives. Change is constant, and our adaptation to the demands of our patients and society will define our survival as surgeons. We expect that health care requirements will be different in developed economies than in less developed economies. However, the evolutionary steps are the same, based on the development of the economy. Major changes in health care occur in spurts, with some setbacks. Economic-, political-, and population-based trends are also important. Crises such as the current COVID-19 pandemic could usher in major changes.
Skull base surgeons and neurosurgeons of the future will need to be nimble, adopting newer technologies as they become available. However, the essential characteristics will remain unchanged. These are knowledge, innovation, technical skill, judgment, and compassion. Our active involvement in these technologies will enable us to shape some of the future in our field.
Innovation will be an important requirement of future and current doctors. The innovations might not be major but found instead in the small items that affect our day-to-day work, or they might be related to clinical surgery, the basic neurosciences, workflow and efficiency, outpatient and hospital infrastructure, patient satisfaction, and quality improvements. Young surgeons must constantly strive to leave their field better than they found them. Surgeons should be actively involved in hospital and health care administration to guide these changes.
The peace mantra, which is from the Brihadāranyaka Upanishad (verse 1.4.14), summarizes the central ethos of our work.
May all be happy; May all be without disease; May all have well-being; May none have misery of any sort. Om Peace, Peace, Peace.
Acknowledgments
We wish to thank Raja Sekhar for his review of our report and comments, and Jennifer Pryll for the drawings. The copyright for the drawings belongs to Laligam N. Sekhar, who provided permission for their reproduction.
Footnotes
Conflict of interest statement: The present study was supported by the National Institutes of Health (NIH), National Institute of Biomedical Imaging and Bioengineering (grant RO1 EBO 16457), NIH, National Cancer Institute, Small Business Innovation Research Phase I (grant R43 CA211086); the Department of Defense (Phase I Army Small Business Technology Transfer [STTR] grant W81XWH-09-C-0159; Phase I Army STTR grant A09A-028-0022; Phase I Army STTR grant A09A-028-0022; Phase II Army STTR grant W81XWH-09-0159); and a University of Washington CoMotion Amazon Catalyst Grant – The Artificially Intelligent Robotic Surgical Assistant (no grant number assigned).
References
- 1.Sekhar L.N., Tzortzidis F., Bejjani G.K., Schessel G.A. Saphenous vein graft bypass of the sigmoid sinus and the jugular bulb during the removal of glomus jugulare tumors. J Neurosurg. 1997;86:1036–1041. doi: 10.3171/jns.1997.86.6.1036. [DOI] [PubMed] [Google Scholar]
- 2.Sindou M.P., Alverina J.E. Results of attempted radical tumor resection and venous repair in 100 consecutive meningiomas involving the major dural sinuses. J Neurosurg. 2006;105:514–525. doi: 10.3171/jns.2006.105.4.514. [DOI] [PubMed] [Google Scholar]
- 3.Yang T., Tariq F., Chabot J., Madhok R., Sekhar L.N. Cerebral revascularization for difficult skull base tumors: a contemporary series of 18 patients. World Neurosurg. 2013;82:660–671. doi: 10.1016/j.wneu.2013.02.028. [DOI] [PubMed] [Google Scholar]
- 4.Broadbent B., Tseng J., Kast R. Shining light on neurosurgery diagnostics using Raman spectroscopy. J Neurooncol. 2016;130:1–9. doi: 10.1007/s11060-016-2223-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shin K.S., Francis A.T., Hill A.H. Intraoperative assessment of skull base tumors using stimulated Raman scattering microscopy. Sci Rep. 2019;9:20392. doi: 10.1038/s41598-019-56932-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hollon T.C., Pandian B., Adapa A.R. Near real-time intraoperative brain tumor diagnosis using stimulated Raman histology and deep neural networks. Nat Med. 2020;26:52–58. doi: 10.1038/s41591-019-0715-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Polis B., Imiela A., Polis L., Abramczyk H. Raman spectroscopy for medulloblastoma. Childs Nerv Syst. 2018;34:2425–2430. doi: 10.1007/s00381-018-3906-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jermyn M., Mok K., Mercier J. Intraoperative brain cancer detection with Raman spectroscopy in humans. Sci Transl Med. 2015;7:274. doi: 10.1126/scitranslmed.aaa2384. [DOI] [PubMed] [Google Scholar]
- 9.Ivan M.E., Mohammadi A.M., De Deugd N. Laser ablation of newly diagnosed malignant gliomas: a meta-analysis. Neurosurgery. 2016;79:S17–S23. doi: 10.1227/NEU.0000000000001446. [DOI] [PubMed] [Google Scholar]
- 10.McCracken D.J., Willie J.T., Fernald B. Gross magnetic resonance thermometry-guided stereotactic laser ablation of cavernous malformations in drug-resistant epilepsy: imaging and clinical results. Op Neurosurg. 2016;12:39–48. doi: 10.1227/NEU.0000000000001033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Peace K., Maloney E., Frangos S. The use of a portable head CT scanner in the intensive care unit. J Neurosci Nurs. 2010;42:109–116. doi: 10.1097/jnn.0b013e3181ce5c5b. [DOI] [PubMed] [Google Scholar]
- 12.Hyperfine. Hyperfine and Yale School of Medicine collaborate on world’s first portable MRI technology. Eurekalert! News Release, October 14, 2019. https://www.eurekalert.org/pub_releases/2019-10/cl-sn_1101119.php Available at:
- 13.Schilsky R.L. Implementing personalized cancer care. Nat Rev Clin Oncol. 2014;11:432–438. doi: 10.1038/nrclinonc.2014.54. [DOI] [PubMed] [Google Scholar]
- 14.Kresak J.L., Walsh M. Neurofibromatosis: a review of NF1, NF2, and schwannomatosis. J Pediatr Genet. 2016;5:98–104. doi: 10.1055/s-0036-1579766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cox D.B.T., Platt R.J., Zhang F. Therapeutic genome editing: prospects and challenges. Nat Med. 2015;21:121–131. doi: 10.1038/nm.3793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Xu L., Wang J., Liu Y. CRISPR-edited stem cells in a patient with HIV and acute lymphocytic leukemia. N Engl J Med. 2019;381:1240–1247. doi: 10.1056/NEJMoa1817426. [DOI] [PubMed] [Google Scholar]
- 17.Coy S., Rashid R., Stemmer-Rachamimov A., Santagata S. An update on the CNS manifestations of neurofibromatosis type 2. Acta Neuropathol. 2020;139:643–665. doi: 10.1007/s00401-019-02029-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hrvatin S., Tzeng C.P., Nagy M.A. A scalable platform for the development of cell-type-specific viral drivers. Elife. 2019;8:e48089. doi: 10.7554/eLife.48089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rodríguez Pérez Á., Campillo-Davo D., Van Tendeloo V.F.I., Benítez-Ribas D. Cellular immunotherapy: a clinical state-of-the-art of a new paradigm for cancer treatment. https://link.springer.com/10.1007/s12094-020-02344-4 [e-pub ahead of print]. Clin Trans Oncol. accessed April 7, 2020. [DOI] [PubMed]
- 20.Ferrara N., Hillan K.J., Gerber H.-P., Novotny W. Discovery and development of bevacizumab, an anti-VEGF antibody for treating cancer. Nat Rev Drug Discov. 2004;3:391–400. doi: 10.1038/nrd1381. [DOI] [PubMed] [Google Scholar]
- 21.Plotkin S.R., Stemmer-Rachamimov A.O., Barker F.G., II Hearing improvement after bevacizumab in patients with neurofibromatosis type 2. N Engl J Med. 2009;361:358–367. doi: 10.1056/NEJMoa0902579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Havel J.J., Chowell D., Chan T.A. The evolving landscape of biomarkers for checkpoint inhibitor immunotherapy. Nat Rev Cancer. 2019;19:133–150. doi: 10.1038/s41568-019-0116-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bi W.L., Greenwald N.F., Abedalthagafi M. Genomic landscape of high-grade meningiomas. NPJ Genom Med. 2017;2:15. doi: 10.1038/s41525-017-0014-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Newick K., O’Brien S., Moon E., Albelda S.M. CAR T cell therapy for solid tumors. Ann Rev Med. 2017;68:139–152. doi: 10.1146/annurev-med-062315-120245. [DOI] [PubMed] [Google Scholar]
- 25.Bagley S.J., Desai A.S., Linette G.P., June C.H., O’Rourke D.M. CAR T-cell therapy for glioblastoma: recent clinical advances and future challenges. Neuro Oncol. 2018;20:1429–1438. doi: 10.1093/neuonc/noy032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Horie N., Niizuma K., Pereira M.P. Transplanted stem cell-secreted VEGF effects post-stroke recovery, inflammation, and vascular repair. Stem Cells. 2011;29:274–285. doi: 10.1002/stem.584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Steinberg G.K., Kondziolka D., Wechsler L.R. Clinical outcomes of a transplanted modified bone marrow-derived mesenchymal stem cells in stroke: a phase 1/2a study. Stroke. 2016;47:1817–1824. doi: 10.1161/STROKEAHA.116.012995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ploch C.C., Mansi C.S.S.A., Jayamohan J., Kuhl E. Using 3D printing to create personalized brain models for neurosurgical training and preoperative planning. World Neurosurg. 2016;90:668–674. doi: 10.1016/j.wneu.2016.02.081. [DOI] [PubMed] [Google Scholar]
- 29.Lan Q., Chen A., Zhang T. Development of three-dimensional printed models for simulated neurosurgery. World Neurosurg. 2016;91:434–442. doi: 10.1016/j.wneu.2016.04.069. [DOI] [PubMed] [Google Scholar]
- 30.Lith R.V., Baker E., Ware H. 3-D printing strong high-resolution anti-oxidant bioresorbable vascular stents. Adv Mater Technol. 2016;1:1600138. [Google Scholar]
- 31.Mannoor M.S., Jiang Z., James T. 3 D printed bionic ears. Nano Lett. 2013;13:2634–2639. doi: 10.1021/nl4007744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tan A., Jeyaraj R., Ashkan K. Nanotechnology in neurosurgery: thinking small, dreaming big. Br J Neurosurg. 2017;31:538–550. doi: 10.1080/02688697.2017.1327017. [DOI] [PubMed] [Google Scholar]
- 33.Mathis D.W., Mozer M.C. On the computational utility of consciousness. In: Tesauro G., Touretzky D.S., Leen T.K., editors. Advances in Neural Information Processing Systems 7. MIT Press; Denver: 1997. pp. 11–18. [Google Scholar]
- 34.Earl B. The biological function of consciousness. Front Psychol. 2014;5:697. doi: 10.3389/fpsyg.2014.00697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ramscar M. Computing machinery and understanding. Cogn Sci. 2010;34:966–971. doi: 10.1111/j.1551-6709.2010.01120.x. [DOI] [PubMed] [Google Scholar]
- 36.Jo T., Nho K., Saykin A.J. Deep learning in Alzheimer's disease: diagnostic classification and prognostic prediction using neuroimaging data. Front Aging Neurosci. 2019;11:220. doi: 10.3389/fnagi.2019.00220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Haenssle H.A., Fink C., Schneiderbauer R. Man against machine: diagnostic performance of a deep learning convolutional neural network for dermoscopic melanoma recognition in comparison to 58 dermatologists. Ann Oncol. 2018;29:1836–1842. doi: 10.1093/annonc/mdy166. [DOI] [PubMed] [Google Scholar]
- 38.Mariakakis A., Baudin J., Whitmire E. Vol. 1. 2017. PupilScreen: using smartphones to assess traumatic brain injury. Proceedings of the ACM on Interactive, Mobile, Wearable and Ubiquitous Technologies; pp. 1–27. [Google Scholar]
- 39.Lucas G.M., Rizzo A., Gratch J. Reporting health symptoms: breaking down barriers to care with virtual human interviewers. Front Robot AI. 2017;4:1–9. [Google Scholar]
- 40.Lasko T.A., Denny J.C., Levy M.A. Computational phenotype discovery using unsupervised feature learning over noisy, sparse, and irregular clinical data. PLoS One. 2013;8:e66341. doi: 10.1371/journal.pone.0066341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rajkomar A., Oren E., Chen K. Scalable and accurate deep learning with electronic health records. NPJ Digit Med. 2018;1:18. doi: 10.1038/s41746-018-0029-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ortiz A., Munilla J., Gorriz J.M., Ramirez J. Ensembles of deep learning architectures for the early diagnosis of the Alzheimer's disease. Int J Neural Syst. 2016;26:1650025. doi: 10.1142/S0129065716500258. [DOI] [PubMed] [Google Scholar]
- 43.Suk H.-I., Lee S.-W., Shen D. Hierarchical feature representation and multimodal fusion with deep learning for AD/MCI diagnosis. NeuroImage. 2014;101:569–582. doi: 10.1016/j.neuroimage.2014.06.077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lee L.I.T., Kanthasamy S., Ayyalaraju R.S., Ganatra R. The current state of artificial intelligence in medical imaging and nuclear medicine. BJR Open. 2019;1:20190037. doi: 10.1259/bjro.20190037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Joseph J.R., Smith B.W., Liu X., Park P. Current applications of robotics in spine surgery: a systematic review of the literature. Neurosurg Focus. 2017;42:E2. doi: 10.3171/2017.2.FOCUS16544. [DOI] [PubMed] [Google Scholar]
- 46.Moritz C.T., Ruther P., Goering S. New perspectives on neuroengineering and neurotechnologies: NSF-DFG workshop report. IEEE Trans Biomed Eng. 2016;63:1354–1367. doi: 10.1109/TBME.2016.2543662. [DOI] [PubMed] [Google Scholar]
- 47.Bernardo A. Virtual reality and simulation in neurosurgical training. World Neurosurg. 2017;106:1015–1029. doi: 10.1016/j.wneu.2017.06.140. [DOI] [PubMed] [Google Scholar]
- 48.Aghdasi N., Whipple M., Humphreys I.M., Moe K.S., Hannaford B., Bly R.A. Automated surgical approach planning for complex skull base targets: development and validation of a cost function and semantic atlas. Surg Innov. 2018;25:476–484. doi: 10.1177/1553350618782287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.O’Malley B.W., Jr., Weinstein G.S., Snyder W., Hockstein N.G. Transoral robotic surgery (TORS) for base of tongue neoplasms. Laryngoscope. 2006;116:1465–1472. doi: 10.1097/01.mlg.0000227184.90514.1a. [DOI] [PubMed] [Google Scholar]
- 50.Staub B.N., Sadrameli S.S. The use of robotics in minimally invasive spine surgery. J Spine Surg. 2019;5:S31–S40. doi: 10.21037/jss.2019.04.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sutherland G.R., Lama S., Gan L.S., Wolfsberger S., Zereinia K. Merging machines with microsurgery: clinical experience with neuroArm. J Neurosurg. 2013;118:521–529. doi: 10.3171/2012.11.JNS12877. [DOI] [PubMed] [Google Scholar]
- 52.Goto T., Miyahara T., Toyoda K. Telesurgery of microscopic micromanipulator system “NeuRobot” in neurosurgery: interhospital preliminary study. J Brain Dis. 2009;1:45–53. doi: 10.4137/jcnsd.s2552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lum M.J.H., Friedman D.C.W., Sankaranarayanan G. The RAVEN: design and validation of a telesurgery system. Int J Robot Res. 2009;28:1183–1197. [Google Scholar]
- 54.Hannaford B., Rosen J., Friedman D.C. Raven-II: an open platform for surgical robotics research. IEEE Trans Biomed Eng. 2013;60:954–959. doi: 10.1109/TBME.2012.2228858. [DOI] [PubMed] [Google Scholar]
- 55.Hannaford B., Kim L., Lendvay T.S. Inventors. Surgical Cockpit Comprising Multisensory and Multimodal Interfaces for Robotic Surgery and Methods Related Thereto. US Patent 9,474,580, October 26, 2016. https://neurosurgery.uw.edu/node/394 Information available at:
- 56.Kalavakonda N., Hannaford B., Qazi Z., Sekhar L. Autonomous neurosurgical instrument segmentation using end-to-end learning. Presented at the IEEE Conference on Computer Vision and Pattern Recognition (CVPR) Workshops, 2019. http://openaccess.thecvf.com/content_CVPRW_2019/html/WiCV/Kalavakonda_Autonomous_Neurosurgical_Instrument_Segmentation_Using_End-To-End_Learning_CVPRW_2019_paper.html Available at:
- 57.Allan M., Shvets A., Kurmann T. 2017 Robotic Instrument Segmentation Challenge. https://arxiv.org/abs/1902.06426 Available at:
- 58.Russell B.C., Torralba A., Murphy K.P., Freeman W.T. LabelMe: a database and web-based tool for image annotation. Int J Comput Vision. 2008;77:157–173. [Google Scholar]
- 59.Hu D., Gong Y., Hannaford B., Seibel E.J. Semi-autonomous simulated brain tumor ablation with RAVENII surgical robot using behavior tree. Presented at the 2015 IEEE International Conference on Robotics and Automation (ICRA), Seattle, WA. IEEE Int Conf Robot Autom. 2015;2015:3868–3875. doi: 10.1109/ICRA.2015.7139738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hu D., Gong Y., Hannaford B., Seibel E.J. Vol. 2015. Rep US; 2015. Path planning for semi-automated simulated robotic neurosurgery. Presented at the 2015 IEEE/RSJ International Conference on Intelligent Robots and Systems (IROS), Hamburg; pp. 2639–2645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Veiseh M., Gabikian P., Bahrami S.-B. Tumor paint: a chlorotoxin: Cy5.5 bioconjugate for intraoperative visualization of cancer foci. Cancer Res. 2007;67:6882–6888. doi: 10.1158/0008-5472.CAN-06-3948. [DOI] [PubMed] [Google Scholar]
- 62.Trapani S.R. Inventor. Room sterilization method and system. US Patent 9,345,798, May 24, 2016. https://patents.google.com/patent/US9345798 Available at:
- 63.Begić A. Application of service robots for disinfection in medical institutions. In: Hadžikadić M., Avdaković S., editors. Advanced Technologies, Systems, and Applications II. IAT 2017. Lecture Notes in Networks and Systems. Vol 28. Springer; Cham: 2018. pp. 1056–1065. [Google Scholar]
- 64.Abecassis J., Sen R.D., Ellenbogen R., Sekhar L.N. Developing microsurgical milestones for psychomotor skills in neurological surgery residents as an adjunct to operative training: the home microsurgery lab. J Neurosurg. 2020:1–11. doi: 10.3171/2020.5.JNS201590. [DOI] [PubMed] [Google Scholar]