Abstract
Purpose
Accumulating evidence suggests that microbiota dysbiosis induced by antibiotic administration plays a crucial role in regulating the efficacy and toxicity of cancer therapy. We explored the influence of antibiotic administration on the efficacy of chemotherapy in patients with esophageal cancer (EC).
Patients and Methods
EC patients were stratified into two groups: antibiotic-treated group and control group. The antibiotic-treated group included patients who received antibiotics within 60 days before or after chemotherapy initiation, and the control group included patients who did not receive antibiotics within 60 days before or after chemotherapy initiation. Progression-free survival (PFS) and overall survival (OS) curves were constructed using the Kaplan–Meier method. The Cox proportional hazards model was used for univariate and multivariate analyses.
Results
The rate of primary progressive disease in the antibiotic-treated group was significantly higher than that in the control group (36.58% vs 10.45%, p = 0.002) as calculated using the chi-square test. Further, antibiotic administration was associated with shorter PFS (6.7 vs 14.6 months, hazard ratio (HR): 2.545, 95% confidence interval (CI): 1.554–4.168, p < 0.001) and reduced OS (15.0 vs 21.0 months, HR: 2.007, 95% CI: 1.213–3.319, p = 0.007) in univariate analysis. Subsequent multivariate analysis indicated that antibiotic administration was a significant independent prognostic factor for PFS (HR: 2.350, 95% CI: 1.423–3.882, p = 0.001) and OS (HR: 1.900, 95% CI: 1.140–3.167, p = 0.014).
Conclusion
Antibiotic administration was associated with reduced chemotherapy efficacy and poor prognosis in patients with EC.
Keywords: esophageal cancer, antibiotics, PFS, OS
Introduction
Esophageal cancer (EC) is the eighth most common cancer and the sixth leading cause of cancer-related mortality worldwide.1 The prognosis of EC remains poor despite incremental advances in diagnosis and treatment over recent decades.2 The 5-year survival rate of patients with EC is below 20%, with a median overall survival (OS) range of 9–10 months because 50% of the patients present with inoperable advanced or metastatic disease at the initial diagnosis.2,3 The optimal treatment for metastatic EC patients is concurrent chemoradiotherapy or palliative chemotherapy.4 The first-line chemotherapy regimen in East Asia consists of a fluoropyrimidine/platinum combination or the addition of other drugs such as docetaxel, irinotecan, and targeted drugs.4
Microbiota is involved in the host metabolism, immunomodulation, neuronal development, and maintenance of the intestinal mucosal barrier.5 Microbiota, especially gut microbiota, demonstrates great plasticity, and the gut colonization is influenced by the mode of delivery, dietary, and antibiotic exposure of the microbiota.5–7 Antibiotic administration leads to changes in bacterial metabolites, disruption of bacterial signaling, gut immune cell dysregulation, and systemic immune dysfunction by inducing microbiota dysbiosis with loss of distinct species and expansion of pathogens.8 Increasing evidence suggests that gut microbiota could regulate the efficacy and toxicity of cancer therapy including chemotherapy (cyclophosphamide, irinotecan, gemcitabine), radiotherapy, and immunotherapy of cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and anti-programmed cell death 1 ligand 1 (anti-PD-L1).9–14 Antibiotic exposure has been associated with reduced efficacy and poor prognosis of CTLA-4 and anti-PD-1/PD-L1 immunotherapy in several tumors including advanced non-small cell lung cancer (NSCLC), urothelial carcinoma, and advanced renal cell cancer (RCC).15–17 In the present study, we evaluated the influence of antibiotic administration on chemotherapy efficacy in EC patients. We expect that our findings will provide a basis for future therapeutic concepts in EC patients who require antibiotics during chemotherapy.
Patients and Methods
Patients
Patients with metastatic EC who started palliative chemotherapy at the Fourth Hospital of Hebei Medical University between January 2013 and September 2018 were enrolled in this retrospective study. In total, 108 metastatic EC patients were enrolled in this study, including 79 men and 29 women, all of Han nationality and from the Hebei Province (Table 1). All patients received chemotherapy either alone or in combination with radiotherapy. All procedures were supervised and approved by the hospital’s ethics committee. We obtained ethical permission to change or modify their therapy based on the progress of their disease. Written informed consent was obtained from the participants.
Table 1.
Clinical Characteristics of EC Patients
| Characteristics | Total | Antibiotic-Treated Group | Control Group | p-value | |
|---|---|---|---|---|---|
| n=108 (%) | n=41 (%) | n=67 (%) | |||
| Gender | Male | 79 (73.15) | 26 (63.41) | 53 (79.10) | 0.074 |
| Female | 29 (26.85) | 15 (36.59) | 14 (20.90) | ||
| Age, year | <60 | 36 (33.33) | 14 (34.15) | 22 (32.84) | 0.888 |
| ≥60 | 72 (66.67) | 27 (65.85) | 45 (67.16) | ||
| Primary Tumor Location | Upper | 12 (11.11) | 5 (12.20) | 7 (10.45) | 0.763 |
| Middle-lower | 96 (88.89) | 36 (87.80) | 60 (89.55) | ||
| Primary Tumor Length (cm) | <5 | 62 (57.41) | 26 (63.41) | 36 (53.73) | 0.493 |
| 5 ≤ < 10 | 37 (34.26) | 13 (31.71) | 24 (35.82) | ||
| 10≤ | 9 (8.33) | 2 (4.88) | 7 (10.45) | ||
| Histology | Squamous | 100 (92.59) | 38 (92.68) | 62 (92.54) | 0.493 |
| Small-cell | 6 (5.56) | 3 (7.32) | 3 (4.48) | ||
| Sarcoma + Mucoepidermoid | 2 (1.85) | 0 (0.00) | 2 (2.98) | ||
| ECOG Performance Status | 0 | 32 (29.63) | 16 (39.02) | 16 (23.88) | 0.271 |
| 1 | 72 (66.67) | 24 (58.54) | 48 (71.64) | ||
| 2 | 4 (3.70) | 1 (2.44) | 3 (4.48) | ||
| Treatment | Chemotherapy + Radiotherapy | 31 (28.70) | 15 (36.59) | 16 (23.88) | 0.157 |
| Chemotherapy | 77 (71.30) | 26 (63.41) | 51 (76.12) | ||
| Chemotherapy | Fluoropyrimidine + Platinum | 35 (32.41) | 15 (36.58) | 20 (29.85) | 0.763 |
| Paclitaxel + Platinum | 65 (60.18) | 23 (56.10) | 42 (62.69) | ||
| Etoposide + Platinum | 8 (7.41) | 3 (7.32) | 5 (7.46) | ||
Abbreviations: EC, esophageal cancer; ECOG, Eastern Cooperative Oncology Group.
Medical records of all patients were reviewed to determine whether any antibiotic administration occurred within 60 days before or after chemotherapy initiation. Data of the specific time of antibiotic exposure, antibiotic class, indication, route of administration, and duration were collected (Table S1). Patients who received antibiotics within 60 days before or after chemotherapy initiation were included in the antibiotic-treated group while patients who did not receive antibiotics within 60 days before or after chemotherapy initiation were included in the control group. Data of additional parameters including age, sex, location and extent of the tumor, histological type, performance status (PS) of Eastern Cooperative Oncology Group (ECOG), detailed treatment regimen, and metastatic status were also collected.
All patients were followed up every three months until death or until the database was closed (May 1, 2020). Computed tomography (CT) scans and gastroscopy findings (if necessary) were reviewed regularly to evaluate the tumor response according to the Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1.18
Statistical Analysis
Clinicopathologic features and tumor response were compared using Fisher’s exact test or chi-square test. Progression-free survival (PFS) was defined as the time interval from chemotherapy initiation to the date of disease progression. Overall survival (OS) was defined as the time interval from chemotherapy initiation to the date of death. At the deadline, the number of patients lost to follow-up was 3 (2.8%) in the PFS analysis and 9 (8.3%) in the OS analysis. PFS and OS curves were estimated by the Kaplan–Meier method and compared using the Log rank test. The Cox proportional hazards model was used to calculate the hazard ratio (HR) and 95% confidence interval (CI) in univariate and multivariate analyses. Variables with p < 0.1 in univariate analysis were chosen to be included in the multivariate analysis. All statistical analyses were performed using SPSS software, version 19.0 (IBM Corporation, Armonk, NY); p < 0.05 was considered to indicate statistical significance.
Results
Clinical Features of EC Patients
In total, 108 metastatic EC patients were enrolled in this study. The predominant histological EC subtype was esophageal squamous cell carcinoma, present in 100 patients (92.59%) (Table 1). Of these patients, 35 (32.41%) received the fluoropyrimidine/platinum combination, 65 (60.18%) received paclitaxel plus platinum, and 8 (7.41%) received etoposide plus platinum (Table 1). The distribution of clinicopathologic characteristics was well balanced between the antibiotic-treated group and the control group (Table 1). The most frequently administered antibiotics were β-lactams ± inhibitors (Table S1).
Impact of Antibiotic Administration on the Clinical Outcome of EC Patients
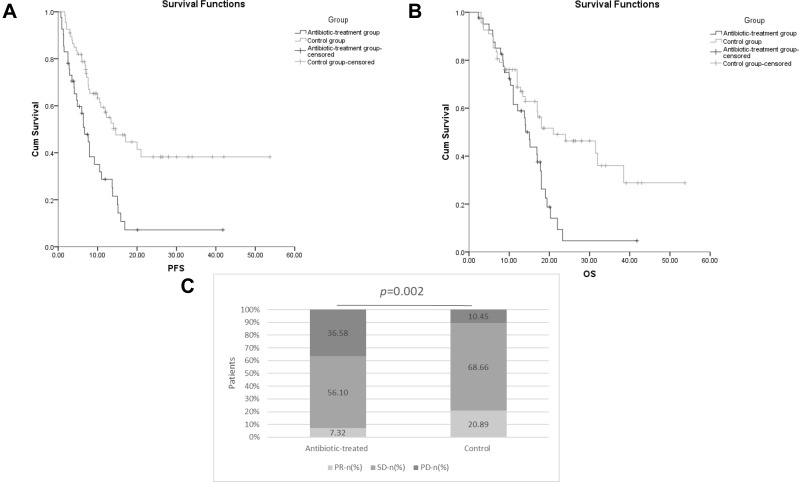
The effect of antibiotic administration on the efficacy of chemotherapy was evaluated in EC patients. The rate of primary progressive disease (PD) in the antibiotic-treated group was significantly higher than that in the control group (36.58% vs 10.45%, p = 0.002) (Figure 1C). Further, univariate analysis with Kaplan–Meier calculation demonstrated shorter PFS (median PFS: 6.7 vs 14.6 months, HR: 2.545, 95% CI: 1.554–4.168, p < 0.001) and lower OS (median OS: 15.0 vs 21.0 months, HR: 2.007, 95% CI: 1.213–3.319, p = 0.007) in the antibiotic-treated group than in the control group (Tables 2 and 3). The histology of the tumor was also identified for its association with PFS and OS using univariate analysis. OS decreased dramatically in the antibiotic-treated group compared with that in the control group (Figure 1A and B). Our data implied that antibiotic administration might change the outcome of EC patients by reducing the efficacy of chemotherapy.
Figure 1.
The impact of antibiotic administration on the clinical outcome of EC patients. (A) The Kaplan–Meier curve of progression free survival (PFS). (B) The Kaplan–Meier curve of overall survival (OS). (C) Analysis of chemotherapy response in EC patients. PR: partial response, SD: stable disease, PD: progressive disease.
Table 2.
Univariate and Multivariate Analyses for PFS in EC Patients
| Prognostic Factor | Univariate Analysis | p-value | Multivariate Analysis | p-value |
|---|---|---|---|---|
| PFS | PFS | |||
| HR (95% CI) | HR (95% CI) | |||
| Antibiotic /Control | 2.545 (1.554–4.168) | <0.001 | 2.350 (1.423–3.882) | 0.001 |
| Gender | 1.066 (0.619–1.838) | 0.817 | - | - |
| Male/Female | ||||
| Age | 0.935 (0.555–1.575) | 0.801 | - | - |
| <60 year/≥60 year | ||||
| Primary Tumor Location | 1.151 (0.524–2.528) | 0.726 | - | - |
| Upper/Middle-lower | ||||
| Primary Tumor Length (cm) | 1.170 (0.712–1.923) | 0.535 | - | - |
| <5/≥5 | ||||
| Histology | 0.258 (0.113–0.588) | 0.001 | 0.357 (0.154–0.829) | 0.017 |
| Squamous/Non-squamous | ||||
| ECOG Performance Status | 1.630 (0.979–2.713) | 0.060 | 1.432 (0.848–2.417) | 0.179 |
| 0 | ||||
| 1 or 2 | ||||
| Treatment | 1.105 (0.618–1.973) | 0.737 | - | - |
| Chemotherapy-Radiotherapy/Chemotherapy |
Abbreviations: EC, esophageal cancer; PFS, progression free survival; HR, hazard ratio; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group.
Table 3.
Univariate and Multivariate Analyses for OS in EC Patients
| Prognostic Factor | Univariate Analysis | p-value | Multivariate Analysis | p-value |
|---|---|---|---|---|
| OS | OS | |||
| HR (95% CI) | HR (95% CI) | |||
| Antibiotic/Control | 2.007 (1.213–3.319) | 0.007 | 1.900 (1.140–3.167) | 0.014 |
| Gender | 1.187 (0.685–2.056) | 0.541 | - | - |
| Male/Female | ||||
| Age | 0.818 (0.478–1.399) | 0.462 | - | - |
| <60 year/≥60 year | ||||
| Primary Tumor Location | 1.147 (0.566–2.324) | 0.703 | - | - |
| Upper/Middle-lower | ||||
| Primary Tumor Length (cm) | 1.036 (0.629–1.705) | 0.890 | - | - |
| <5/≥5 | ||||
| Histology | 0.375 (0.159–0.889) | 0.026 | 0.453 (0.190–1.080) | 0.074 |
| Squamous/Non-squamous | ||||
| ECOG Performance Status | 1.157 (0.681–1.965) | 0.589 | - | - |
| 0 | ||||
| 1 or 2 | ||||
| Treatment | 0.783 (0.469–1.306) | 0.348 | - | - |
| Chemotherapy-Radiotherapy/Chemotherapy |
Abbreviations: EC, esophageal cancer; OS, overall survival; HR, hazard ratio; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group.
The impact of antibiotic administration on PFS and OS was further investigated within individual subgroups of patients. As noted, the antibiotic-treated group displayed a trend of reduced PFS (Figure 2A) and OS (Figure 2B) within most subgroups.
Figure 2.
Subgroup analyses of independent prognostic factors for PFS (A) and OS (B).
The potential outcome predictors including antibiotic administration and histology were analyzed using the Cox proportional hazards model. As shown in Tables 2 and 3, antibiotic administration was identified as a significant independent prognostic factor for PFS (HR: 2.350, 95% CI 1.423–3.882, p = 0.001) and OS (HR: 1.900, 95% CI: 1.140–3.167, p = 0.014). Histology was an independent prognostic factor only for PFS (HR: 0.357, 95% CI: 0.154–0.829, p = 0.017) but not for OS (HR: 0.453, 95% CI: 0.190–1.080, p = 0.074) (Tables 2 and 3). These data indicated that antibiotic administration exhibited a negative independent effect on the outcome of chemotherapy-receiving EC patients.
Discussion
Approximately 10% of cancer patients undergoing chemotherapy may require antibiotic treatment due to the immunosuppressive nature of their malignancies and the lymphodepletion induced by anticancer therapies.17,19 We evaluated the influence of antibiotic administration on chemotherapy in EC patients and found that it was associated with reduced PFS and OS as well as higher rates of PD. Our data implied that antibiotics might modulate the chemotherapy efficiency by changing the equilibrium of the gut microbiota to modify the EC outcome. Cancer patients, especially those with gastrointestinal cancers, may sometimes need antibiotic treatment because malnutrition and cachexia make them susceptible to infections. In addition, chemotherapy-related agranulocytosis also requires antibiotic treatment. Previous studies have shown that antibiotic-related changes in Akkermansia muciniphila abundance are associated with poor prognosis of advanced NSCLC and RCC patients who receive PD-1/PD-L1 monoclonal antibodies.15 The deleterious effects of antibiotics on PFS and OS were also found in patients with advanced NSCLC or RCC treated with immune checkpoint inhibitors (ICI).16 Few studies have explored the effect of antibiotic administration on the efficacy of chemotherapy. We found that antibiotic administration has a negative influence on the chemotherapy efficacy in EC patients.
The antibiotic-induced microbiota dysbiosis might be responsible for a reduced chemotherapy efficiency in EC patients, but the true mechanism remains uncertain. The platinum compounds cisplatin and oxaliplatin initiate cancer cell toxicity by forming platinum DNA adducts and overproduction of reactive oxygen species (ROS).20 Tumor-infiltrating myeloid-derived cells respond poorly to platinum compounds with lower cytokine production, deficient production of ROS, and cytotoxicity in antibiotic-treated or germ-free mice.20 Additionally, concomitant antibiotic treatment was associated with the attenuated efficacy of cisplatin in a lung cancer mouse model by downregulating the expression of BAX and cyclin-dependent kinase inhibitor 1B (CDKN1B) and upregulating that of vascular endothelial growth factor A (VEGF-A); the antibiotic-induced chemotherapy inefficiency could be rescued by concomitant administration of Lactobacillus bacteria.21 Furthermore, antibiotic administration could induce microbiota dysbiosis with increased pathogenic bacteria (including bacteria from the Escherichia, Shigella, and Enterobacter genera), thus impairing the efficacy of 5-fluorouracil by downregulating the expression of genes involved in the amino acid metabolism.22 The aforementioned findings indicate that antibiotics might initiate microbiota dysbiosis by downregulating the expression of the ROS, BAX, CDKN1B, and genes participating in the amino acid metabolism and upregulating that of VEGF-A to reduce the efficacy of chemotherapy in EC patients.
Conclusion
Antibiotic administration was associated with reduced chemotherapy efficacy and poor prognosis in EC patients. These findings suggest that antibiotic administration should be limited to strict indications in EC patients receiving chemotherapy. More studies are warranted to confirm the effect of antibiotics in a larger prospective trial and identify the key members of the microbiota that modulate the efficacy of chemotherapy. The discovery of targeted bacteria capable of rescuing unfavorable antibiotic-associated dysbiosis made it possible to promote the antitumor efficacy of chemotherapy by modulating microbiome diversity.
Funding Statement
This work was supported by the Natural Science Foundation of China of Hebei Province (Grant No. H2019206428).
Abbreviations
EC, esophageal cancer; PFS, progression-free survival; OS, overall survival; HR, hazard ratio; CI, confidence interval; PD, primary progressive disease; CTLA-4, cytotoxic T-lymphocyte-associated protein 4; anti-PD-L1, anti-programmed cell death 1 ligand 1; NSCLC, non-small cell lung cancer; RCC, advanced renal cell cancer; PS, performance status; ECOG, Eastern Cooperative Oncology Group; CT, computed tomographic scans; RECIST, response evaluation criteria in solid tumors; ESCC, esophageal squamous cell carcinoma; mAb, monoclonal antibody; ICI, Immune checkpoint inhibitor; ROS, reactive oxygen species; CDKN1B, cyclin dependent kinase inhibitor 1B; VEGFA, vascular endothelial growth factor.
Data Sharing Statement
All data generated or analyzed during this study are included in this published article (more information please contact the corresponding author).
Ethics Approval and Informed Consent
All procedures were supervised and approved by the hospital’s Ethics Committee (No. 2020KS001). Written informed consents were provided from the participants.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Ferlay J, Soerjomataram I, Fau - Dikshit R, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN. Int j Cancer. 2012;136(5):E359–E386. doi: 10.1002/ijc.29210 [DOI] [PubMed] [Google Scholar]
- 2.Lin EW, Karakasheva TA, Hicks PD, Bass AJ, Rustgi AK. The tumor microenvironment in esophageal cancer. Oncogene. 2016;35(41):5337–5349. doi: 10.1038/onc.2016.34 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.van Rossum PSN, Mohammad NH, Vleggaar FP, van Hillegersberg R. Treatment for unresectable or metastatic oesophageal cancer: current evidence and trends. Nat Rev Gastroenterol Hepatol. 2018;15(4):235–249. doi: 10.1038/nrgastro.2017.162 [DOI] [PubMed] [Google Scholar]
- 4.Ku GY. Systemic therapy for esophageal cancer: chemotherapy. Chin Clin Oncol. 2017;6(5):49. doi: 10.21037/cco.2017.07.06 [DOI] [PubMed] [Google Scholar]
- 5.Jandhyala SM, Talukdar R, Subramanyam C, Vuyyuru H, Sasikala M, Nageshwar Reddy D. Role of the normal gut microbiota. World J Gastroenterol. 2015;21(29):8787–8803. doi: 10.3748/wjg.v21.i29.8787 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zmora N, Suez J, Elinav E. You are what you eat: diet, health and the gut microbiota. Nat Rev Gastroenterol Hepatol. 2019;16(1):35–56. [DOI] [PubMed] [Google Scholar]
- 7.Nogacka AM, Salazar N, Arboleya S, et al. Early microbiota, antibiotics and health. Cell Mol Life Sci. 2018;75(1):83–91. doi: 10.1007/s00018-017-2670-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhang S, Chen D-C. Facing a new challenge: the adverse effects of antibiotics on gut microbiota and host immunity. Chin Med J (Engl). 2019;132(10):1135–1138. doi: 10.1097/CM9.0000000000000245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Roy S, Trinchieri G. Microbiota: a key orchestrator of cancer therapy. Nat Rev Cancer. 2017;17(5):271–285. doi: 10.1038/nrc.2017.13 [DOI] [PubMed] [Google Scholar]
- 10.Alexander JL, Wilson ID, Teare J, Marchesi JR, Nicholson JK, Kinross JM. Gut microbiota modulation of chemotherapy efficacy and toxicity. Nat Rev Gastroenterol Hepatol. 2017;14(6):356–365. doi: 10.1038/nrgastro.2017.20 [DOI] [PubMed] [Google Scholar]
- 11.Viaud S, Saccheri F, Mignot G, et al. The intestinal microbiota modulates the anticancer immune effects of cyclophosphamide. Science. 2013;342(6161):971–976. doi: 10.1126/science.1240537 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sivan A, Corrales L, Hubert N, et al. Commensal Bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science. 2015;350(6264):1084–1089. doi: 10.1126/science.aac4255 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gupta E, Lestingi TM, Mick R, Ramirez J, Vokes EE, Ratain MJ. Metabolic fate of irinotecan in humans: correlation of glucuronidation with diarrhea. Cancer Res. 1994;54(14):3723–3725. [PubMed] [Google Scholar]
- 14.Vétizou M, Pitt JM, Daillère R, et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science. 2015;350(6264):1079–1084. doi: 10.1126/science.aad1329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Routy B, Le Chatelier E, Derosa L, et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science. 2018;359(6371):91–97. doi: 10.1126/science.aan3706 [DOI] [PubMed] [Google Scholar]
- 16.Derosa L, Hellmann MD, Spaziano M, et al. Negative association of antibiotics on clinical activity of immune checkpoint inhibitors in patients with advanced renal cell and non-small-cell lung cancer. Ann Oncol. 2018;29(6):1437–1444. doi: 10.1093/annonc/mdy103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhao S, Gao G, Li W, et al. Antibiotics are associated with attenuated efficacy of anti-PD-1/PD-L1 therapies in Chinese patients with advanced non-small cell lung cancer. Lung Cancer. 2019;130:10–17. doi: 10.1016/j.lungcan.2019.01.017 [DOI] [PubMed] [Google Scholar]
- 18.Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228–247. doi: 10.1016/j.ejca.2008.10.026 [DOI] [PubMed] [Google Scholar]
- 19.Teillant A, Gandra S, Barter D, Morgan DJ, Laxminarayan R. Potential burden of antibiotic resistance on surgery and cancer chemotherapy antibiotic prophylaxis in the USA: a literature review and modelling study. Lancet Infect Dis. 2015;15(12):1429–1437. doi: 10.1016/S1473-3099(15)00270-4 [DOI] [PubMed] [Google Scholar]
- 20.Iida N, Dzutsev A, Stewart CA, et al. Commensal bacteria control cancer response to therapy by modulating the tumor microenvironment. Science. 2013;342(6161):967–970. doi: 10.1126/science.1240527 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gui QF, Lu HF, Zhang CX, Xu ZR, Yang YH. Well-balanced commensal microbiota contributes to anti-cancer response in a lung cancer mouse model. Genet Mol Res. 2015;14(2):5642–5651. doi: 10.4238/2015.May.25.16 [DOI] [PubMed] [Google Scholar]
- 22.Yuan L, Zhang S, Li H, et al. The influence of gut microbiota dysbiosis to the efficacy of 5-Fluorouracil treatment on colorectal cancer. Biomed Pharmacother. 2018;108:184–193. doi: 10.1016/j.biopha.2018.08.165 [DOI] [PubMed] [Google Scholar]